Introduction
Hepatocellular carcinoma (HCC) is the second leading
cause of cancer-associated mortality worldwide, and its morbidity
and mortality rates are steadily increasing (1,2).
Although the therapeutic approaches for HCC have improved during
the past few years, the prognosis of HCC patients remains
unsatisfactory since diagnosis is often made at an advanced stage
and, therefore, these patients cannot be treated with surgery
(3).
Sorafenib (Nexavar), a multikinase inhibitor, is the
first and only systemic agent approved for the treatment of
patients with advanced HCC (4,5). It
inhibits multiple cell surface tyrosine kinases and downstream
intracellular serine/threonine kinases in the mitogen-activated
protein kinase cascade, including vascular endothelial growth
factor receptor (VEGFR)-1, VEGFR-2 and VEGFR-3, as well as
platelet-derived growth factor receptor-β (4,6,7).
Furthermore, previous studies have demonstrated that certain
microRNA (miR) molecules, such as miR-1274a and miR-125a-5p, are
involved in HCC target-chemotherapy with sorafenib (8,9).
Although it has been reported that sorafenib improves the overall
survival of patients with advanced HCC, the majority of patients
ultimately develop resistance to sorafenib (4,5,10).
Recently, sorafenib has been administered in combination with other
agents. Previous research has revealed that the combination of
sorafenib and triptolide is superior to single drug treatment in
increasing cell death and apoptosis in vitro, and inhibiting
tumor growth in vivo (11).
The combination of sorafenib and aspirin resulted in a synergistic
antitumor effect against liver tumors both in vitro and
in vivo (12). However,
clinical investigations associated with these studies have not been
conducted to date. Therefore, it is necessary to develop novel
combination therapies to potentiate the anticancer effect of
sorafenib and facilitate its clinical application.
Berberine is an isoquinoline alkaloid isolated from
species belonging to various plant families, including the
Berberidaceae, Ranunculaceae and Papaveraceae families (13,14).
It is traditionally used as an antibiotic to treat dysentery,
gastrointestinal diseases and diarrhea in China, and is a
relatively cheap compound (14,15).
Previous studies have reported that berberine exerts an anticancer
activity in multiple types of cancer and inhibits the proliferation
of cancer cells by inducing apoptosis (16–19).
The possible molecular mechanism of the effect of berberine
involves its interaction with numerous molecular targets, including
transcription factors, cytokines, enzymes, receptors, oncomiRs and
tumor-suppressive miRs (20).
Furthermore, it has been reported that berberine suppresses tumor
development when used in combination with other chemotherapeutic
agents. For instance, the combination of berberine and evodiamine
enhanced the apoptosis of SMMC-7721 cells (21), while berberine in combination with
cisplatin exhibited high synergistic inhibitory effects on the
growth of HeLa cells (22). These
findings suggest that berberine is a potential antitumor drug with
fewer side effects and a low price. However, the combined efficacy
of berberine and sorafenib in the treatment of HCC has not been
reported.
In the present study, the aim was to explore the
effect of the combination of berberine and sorafenib on the growth
of HCC cells. The data of this study indicated for the first time
that berberine in combination with sorafenib exerts an inhibitory
effect on the proliferation of liver cancer cells and induces
cellular apoptosis. Thus, combined treatment with sorafenib and
berberine may be a novel therapeutic strategy for HCC patients with
sorafenib resistance.
Materials and methods
Cells and reagents
Two HCC cell lines (SMMC-7721 and HepG2) were
obtained from the American Type Culture Collection (ATCC; Manassas,
VA, USA). The cells were cultured in Dulbecco's modified Eagle's
medium (DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA,
USA; cat. no. 8117220;) supplemented with 10% (v/v) fetal bovine
serum (FBS) and 1% (v/v) penicillin/streptomycin (Gibco; Thermo
Fisher Scientific, Inc.; cat. no. 10-378-016) and maintained at
37°C in a 5% CO2 incubator. Berberine was purchased from
Selleck Chemicals (Houston, TX, USA; cat. no. S2271). Sorafenib was
purchased from MedChemExpress (Monmouth Junction, NJ, USA; cat. no.
Y-10201). Dimethyl sulfoxide (DMSO) was obtained from Sigma-Aldrich
(Merck KGaA, Darmstadt, Germany; cat. no. 67-68-5).
Cell viability assays
The cells were seeded at a density of 3,000
cells/well in a 96-well plate. After 24 h of incubation, the
indicated concentrations of sorafenib (0, 1, 2, 4 and 8 µM) or
berberine (0, 5, 25, 50 and 100 µM) alone, or their combination (1
µM sorafenib + 5 µM berberine; 1 µM sorafenib + 25 µM berberine; 1
µM sorafenib + 50 µM berberine; 1 µM sorafenib + 100 µM berberine;
2 µM sorafenib + 5 µM berberine; 2 µM sorafenib + 25 µM berberine;
2 µM sorafenib + 50 µM berberine; 2 µM sorafenib + 100 µM
berberine; 4 µM sorafenib + 5 µM berberine; 4 µM sorafenib + 25 µM
berberine; 4 µM sorafenib + 50 µM berberine; 4 µM sorafenib + 100
µM berberine; 8 µM sorafenib + 5 µM berberine; 8 µM sorafenib + 25
µM berberine; 8 µM sorafenib + 50 µM berberine; 8 µM sorafenib +
100 µM berberine) were added to each well and incubated for 48 h.
Mock treatment with an identical volume of DMSO was used as a
control. In addition, treatment with a combination of 4 µM
sorafenib and 100 µM berberine was tested after incubation for
three different time-points (24, 48 and 72 h). Next, the cell
viability was measured with the MTS method. Briefly, 20 µl MTS dye
solution was mixed with 100 µl DMEM in each well and cultured for
2.5 h at 37°C. The optical density (OD) values were read at 490 nm
using a Synergy H1/Epoch microplate reader (BioTek Instruments,
Inc., Winooski, VT, USA). Cell viability rate was calculated
according to the following formula: Cell viability (%) = (the mean
OD value of drug - treated sample/the mean OD value of control
sample) × 100%.
EdU incorporation assay
For evaluation of cell proliferation, the
Cell-Light™ EdU Apollo®567 in vitro Imaging kit
(Guangzhou RiboBio Co., Ltd., China) was used, and the EVOS FL High
Content Imaging System was used to obtain and analyze images of the
cell cultures. Briefly, following the drug treatment, 50 µM EdU was
added to each well and incubated for 2 h to facilitate its
integration into the S phase of DNA. The cells in the 96-well plate
were then washed twice with phosphate-buffered saline (PBS) and
fixed with 4% paraformaldehyde for 30 min at 25°C. Next, the cells
were washed once with 2 mg/ml glycine and treated with 0.5% Triton
X-100 at 25°C for 10 min. Following permeabilization, the cells
were incubated with 100 µl 1× Apollo reaction mixture at 25°C for
30 min in the dark, washed with 0.5% Triton X-100 twice for 10 min,
and then washed twice with methanol for 5 min. The cell nucleus was
stained with Hoechst 33342 reaction mixture and washed with PBS for
10 min. Finally, the EVOS FL High Content Imaging System was used
to obtain and analyze images of the cells.
Colony formation assay
In total, 1,000 cells were cultured per well in a
6-well plate. After 10 days of growth, the cells were treated with
4 µM sorafenib and 100 µM berberine alone or in combination for 72
h. Mock treatment with an identical volume of DMSO was used as a
control. The cells were washed with PBS twice, fixed with methanol
for 15 min at room temperature, and stained with crystal violet for
15 min. Colonies with >50 cells were counted under an ordinary
optical microscope.
Western blotting
The cells were seeded in a 60-mm plate at the
density of 1.0×105 cells/well and cultured in DMEM with
10% FBS and 1% (v/v) penicillin/streptomycin. Following treatment
with 4 µM sorafenib and 100 µM berberine alone or in combination
for 72 h, the cells were washed with cold PBS and harvested. The
harvested cells were lysed with ice-cold radioimmunoprecipitation
assay buffer containing 2% protease/phosphatase inhibitor, 1%
phenylmethylsulfonyl fluoride and 1% protease inhibitor cocktail
(Thermo Fisher Scientific, Inc.) for 20 min. Subsequent to lysis by
ultrasonication (Sonics Vibra-Cell; Sonics & Materials, Inc.,
Newtown, CT, USA), the lysates were centrifuged at 13,400 × g for
15 min at 4°C. Total protein concentrations of the lysates were
assessed using the BCA protein quantification kit (Thermo Fisher
Scientific, Inc.). Protein extracts (50 µg) were separated by 8%
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
(SDS-PAGE), and then transferred onto methanol-activated
polyvinylidene fluoride (PVDF) membranes at 90 V for 1.5 h. After
blocking the PVDF membranes with 5% non-fat milk for 1 h at 25°C,
the membranes were probed with primary antibodies at 4°C for 12 h,
including anti-poly(ADP-ribose) polymerase (PARP; 1:1,000; cat. no.
9532; Cell Signaling Technology, Inc., Inc.), cleaved-caspase-3
(1:1,000; cat. no. 9664; Cell Signaling Technology, Inc.),
anti-B-cell lymphoma 2 (Bcl-2; 1:1,000; cat. no. 2827; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), anti-VEGF antibodies (1:500;
cat. no. 46154; Abcam, Cambridge UK) and anti-GAPDH (1:1,000; cat.
no. 2118; Cell Signaling Technology, Inc.). Subsequently, the
membranes were incubated with horseradish peroxidase-conjugated
anti-rabbit IgG (1:5,000; cat. no. 7074; Cell Signaling Technology,
Inc.) secondary antibody for 1 h at 25°C. Finally, the protein
bands were detected using enhanced chemiluminescence (SuperSignal
West Pico Chemiluminescent Substrate; Pierce; Thermo Fisher
Scientific, Inc.). All the protein bands were densitometrically
scanned and analyzed with ImageJ software (National Institues of
Health, Bethesda, MD, USA). The results of western blotting are
representative of at least three independent experiments.
Statistical analysis
All data are expressed as the mean ± standard
deviation of three independent sets of experiments. Student's
t-test was used to analyze all experimental data in GraphPad Prism
software, version 6 (GraphPad Software, Inc., La Jolla, CA, USA).
P<0.05 was considered to be an indicator of a statistically
significant difference.
Results
Sorafenib and berberine inhibits the
proliferation of HCC cells in a dose-dependent manner
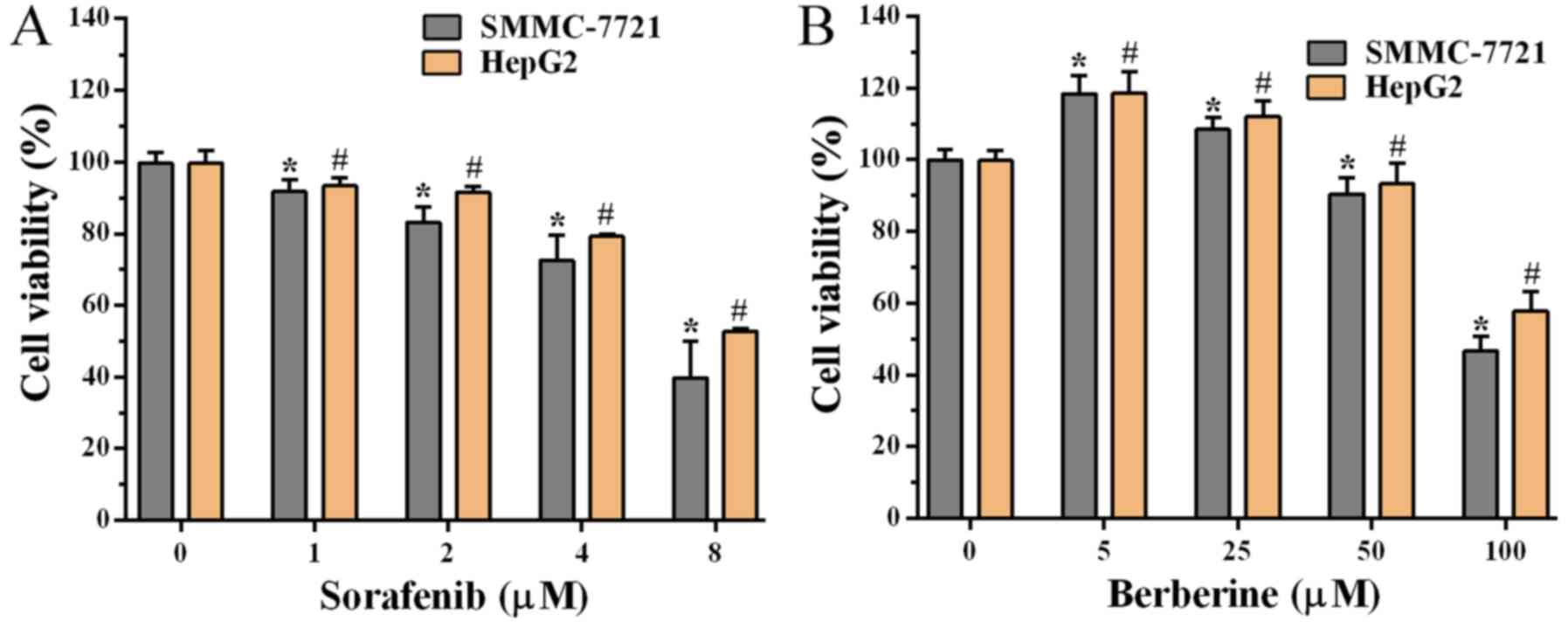
To explore the growth inhibitory effect of sorafenib
and berberine, an MTS assay was utilized to assess the viability of
HCC cells treated with sorafenib or berberine. The results
demonstrated that sorafenib inhibited the growth of SMMC-7721 and
HepG2 cells in a dose-dependent manner (Fig. 1A), and that the viability of cells
treated with 4 and 8 µM sorafenib was reduced to almost 80 and 50%,
respectively. It was also observed that treatment with 5 and 25 µM
berberine enhanced the viability of SMMC-7721 and HepG2 cells by
10–20% (Fig. 1B). However,
cytotoxicity was gradually observed with increasing doses of
berberine. Cell viability was inhibited following treatment with 50
and 100 µM berberine, while the viability of cells was reduced to
~50% when treated with 100 µM berberine (Fig. 1B). Thus, these findings indicated
that sorafenib inhibited the growth of HCC cells in a
dose-dependent manner, whereas berberine had a dose-dependent
inhibitory effect at sufficiently high concentrations.
Berberine synergistically sensitize
HCC cells to sorafenib
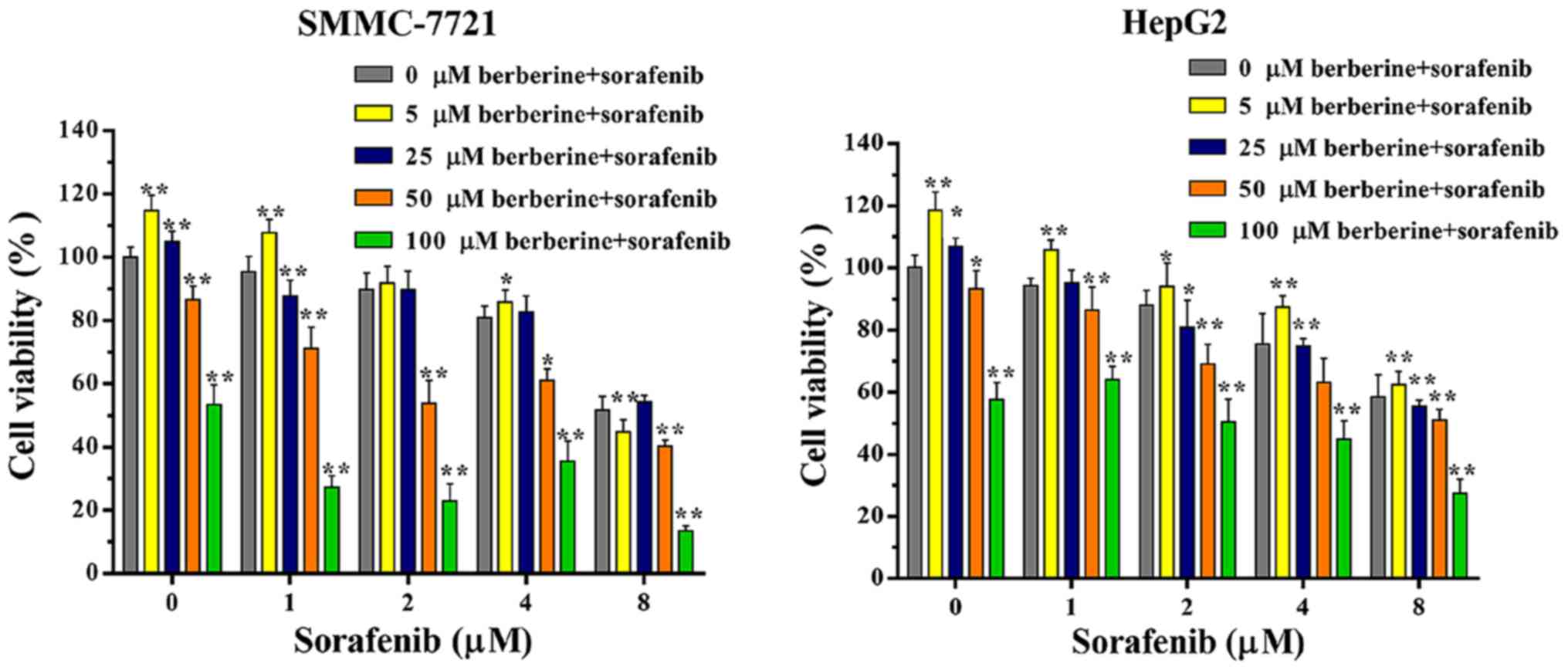
To investigate the synergistic effect of sorafenib
and berberine, SMMC-7721 and HepG2 cells were exposed to different
concentrations of sorafenib (0, 1, 2, 4 and 8 µM) and berberine (0,
5, 25, 50 and 100 µM) alone or in combination. The results of cell
viability assay revealed that, with the exception of the 2 or 8 µM
sorafenib and 5 µM berberine combined groups in SMMC-7721 cells,
the combination of sorafenib (1, 2, 4 and 8 µM) and berberine (5
µM) promoted cell growth when compared with vehicle or sorafenib
treatment alone (Fig. 2A and B).
With the increase of berberine concentration, the
anti-proliferative effect of the combined treatment was markedly
higher in comparison with that of sorafenib treatment alone. The
combination of 8 µM sorafenib and 100 µM berberine had the most
marked inhibitory effect (Fig. 2A and
B). These results demonstrated that berberine synergistically
sensitized HCC cells to sorafenib.
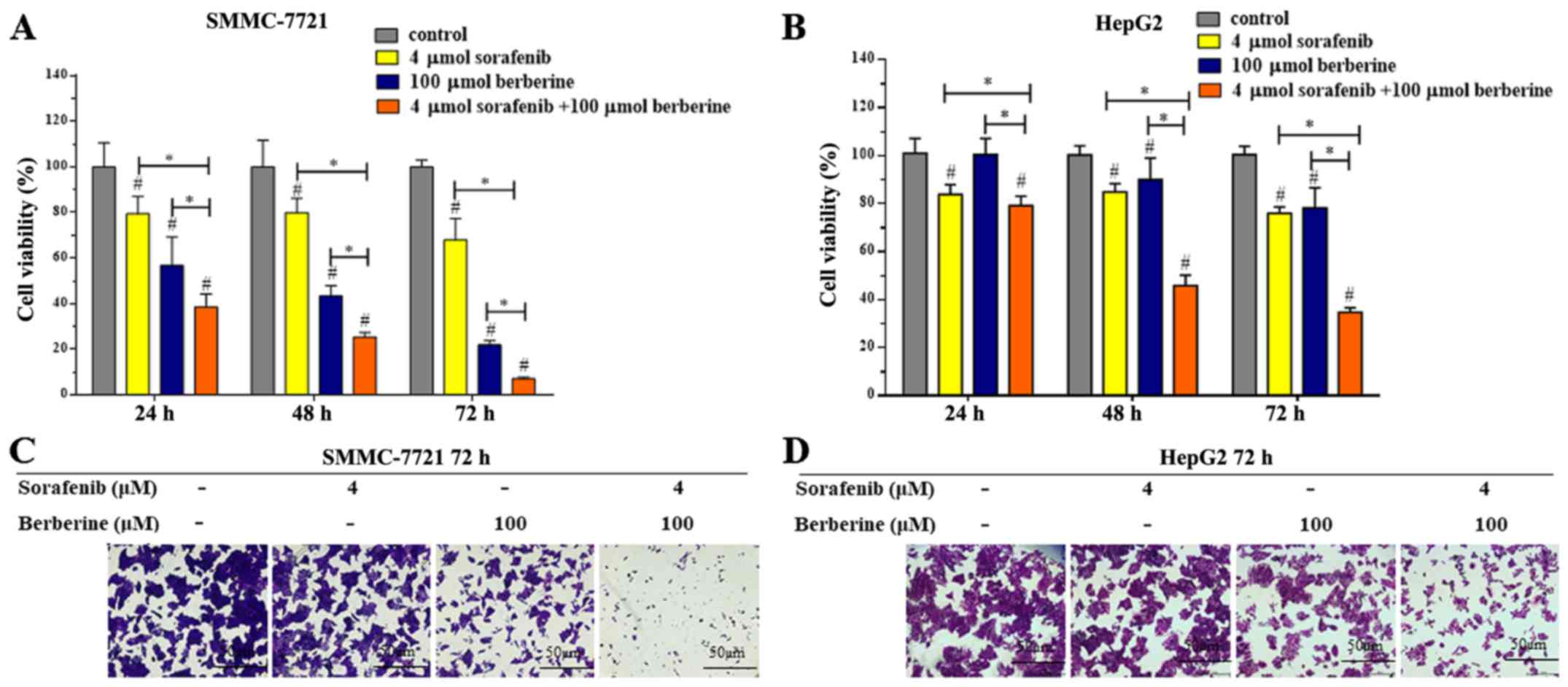
Considering that the combination of 4 µM sorafenib
and 100 µM berberine not only inhibited HCC cell proliferation, but
also increases the sensitivity of HCC cells to sorafenib, the
combination of 4 µM sorafenib and 100 µM berberine was selected for
subsequent experiments. To further confirm the synergistic effect
of sorafenib and berberine, SMMC-7721 and HepG2 cells were treated
with 4 µM sorafenib and 100 µM berberine alone or in combination
for 24, 48 and 72 h, respectively. The results revealed that the
anti-proliferative effect of the combination treatment was
significantly greater compared with that of single agent treatment,
and that this effect was time-dependent (Fig. 3A and B). The viability of SMMC-7721
and HepG2 cells decreased evidently in the group treated with a
combination of 4 µM sorafenib and 100 µM berberine for 72 h. In
addition, crystal violet staining was applied for screening cell
viability. Consistent with the findings of the MTS assay, cell
proliferation was markedly suppressed in the combined treatment
group (Fig. 3C). Similar results
were observed for HepG2 cells in the MTS and crystal violet assays
(Fig. 3B and 3D). Collectively, berberine plus sorafenib
may be a potential therapeutic combination for the inhibition of
HCC cell growth.
Combination of berberine and sorafenib
suppresses the proliferation and colony formation of HCC cells
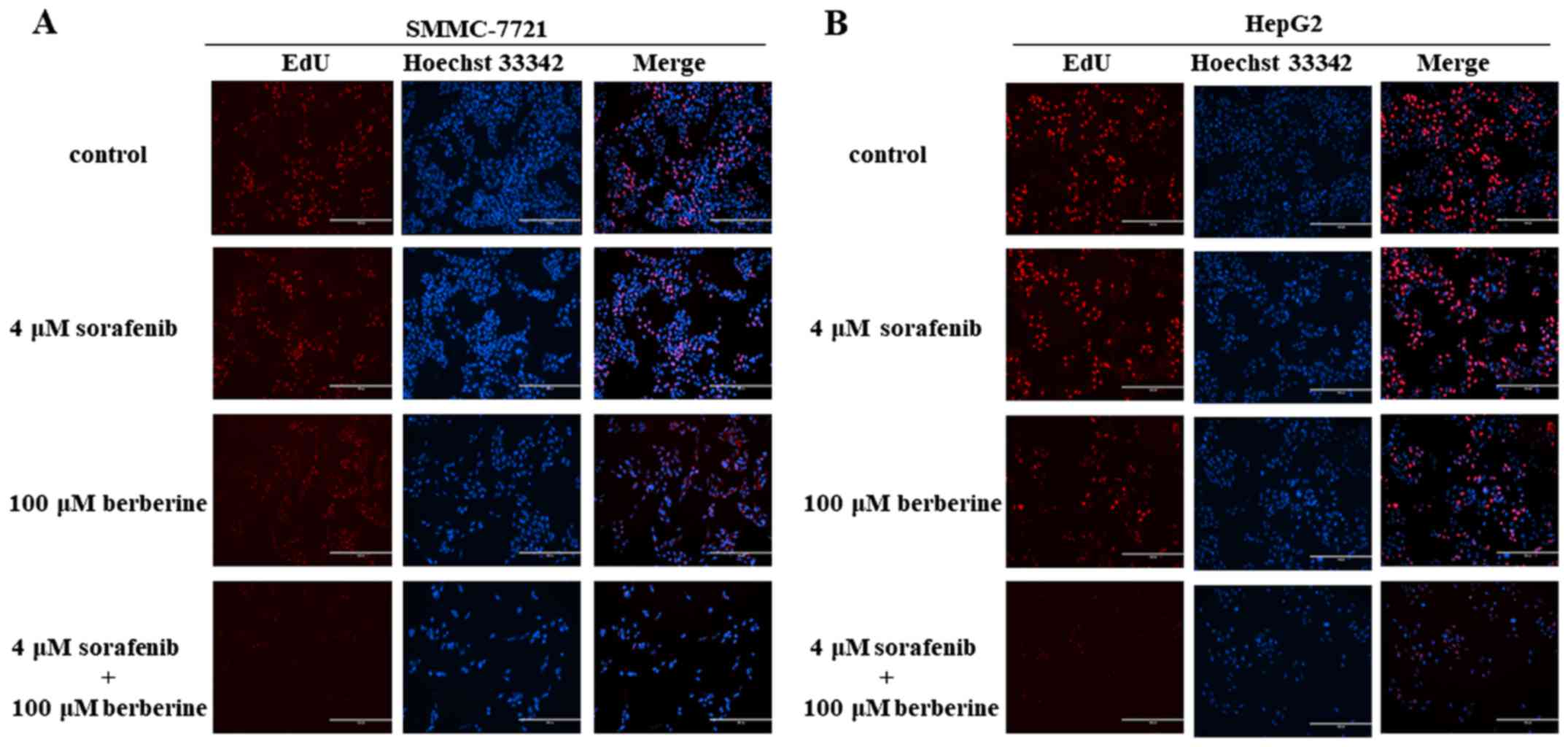
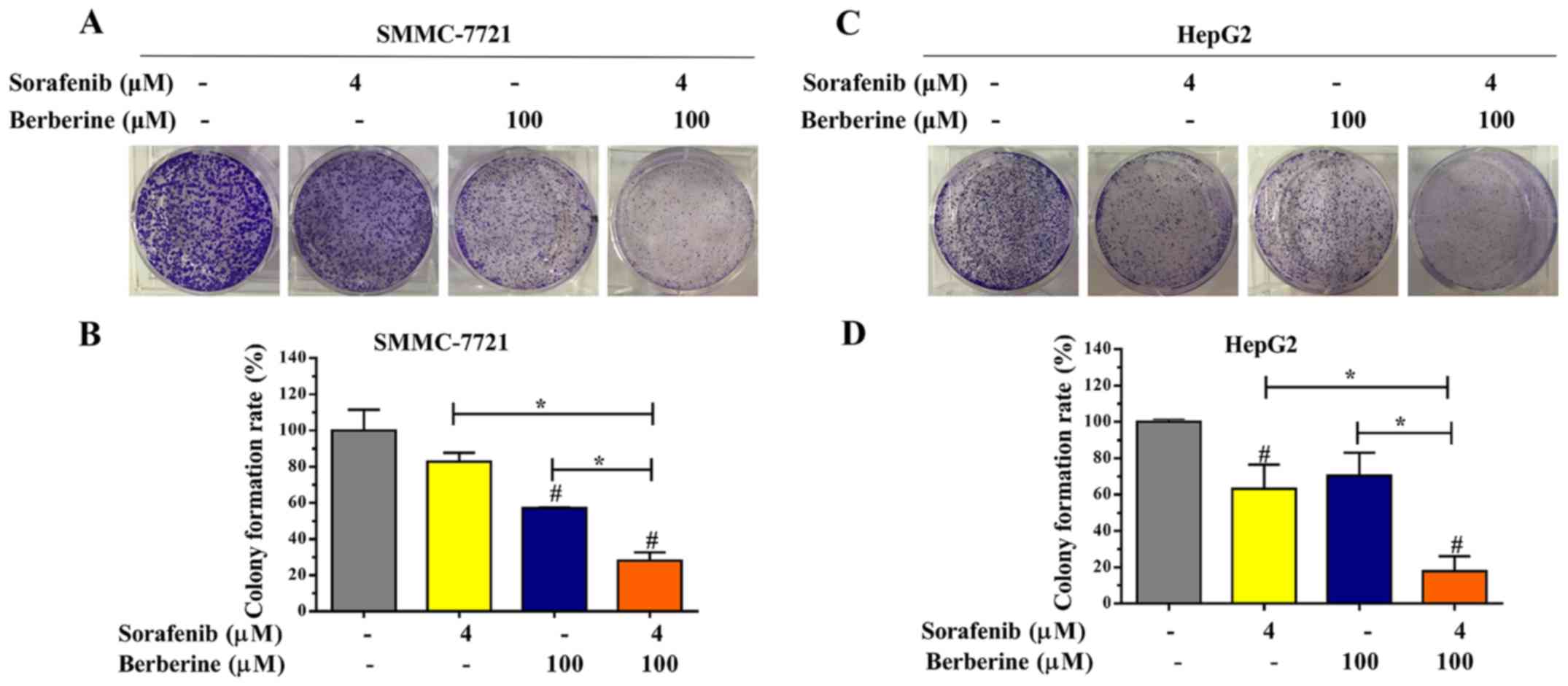
Edu and colony formation assays were conducted to
assess the short-term and long-term effects of combined treatment
with berberine and sorafenib on HCC cell proliferation. As shown in
Fig. 4A, the combination of 100 µM
berberine and 4 µM sorafenib significantly decreased the
proliferation of SMMC-7721 cells when compared with that of the
control and single agent groups. Furthermore, it was observed that
HepG2 cells treated with the combination of 100 µM berberine and 4
µM sorafenib exhibited a lower proliferative ability in comparison
with the other groups (Fig.
4B).
Next, the synergistic potential of berberine and
sorafenib was further evaluated based on the colony formation
efficiency. The results revealed that the largest number of
SMMC-7721 cell colonies was formed in the control group, while the
lowest number of SMMC-7721 cell colonies was observed in the group
treated with the combination of 100 µM berberine and 4 µM sorafenib
(Fig. 5A). Significant differences
were observed in the number of colonies between the four groups
(Fig. 5B; P<0.05). Meanwhile,
similar results were observed in HepG2 cells using the colony
formation assay (Fig. 5C and D;
P<0.05). Taken together, the aforementioned results revealed
that the combination of berberine and sorafenib inhibited the
proliferation and colony formation of HCC cells.
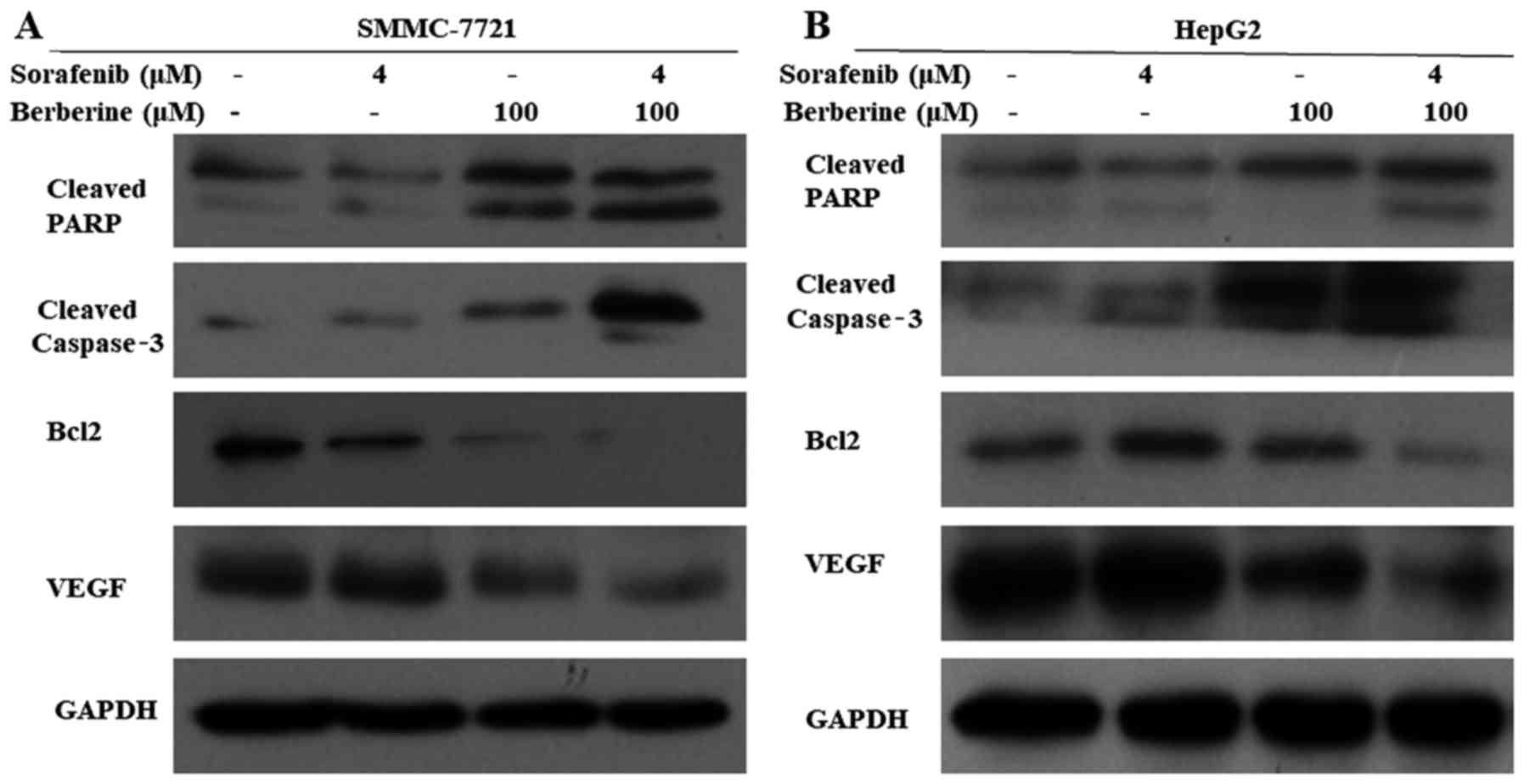
Berberine enhances sorafenib-induced
apoptosis in HCC cells
To investigate cellular apoptosis induced by
combination treatment, SMMC-7721 and HepG2 cells were treated with
100 µM berberine and 4 µM sorafenib alone or in combination for 72
h. Subsequently, the expression levels of apoptosis-associated
proteins were detected by western blot analysis. As shown in
Fig. 6A and B, the expression
levels of the apoptosis-associated proteins cleaved PARP and
cleaved caspase-3 increased, and the expression of the
anti-apoptotic protein Bcl-2 decreased in the combined treatment
group when compared with the other groups. In addition, the
expression level of VEGF, which is involved in the development of
tumor vasculature in the liver, was evidently reduced in the
combined treatment group compared with the other groups. These
results suggested that berberine increased the sensitivity of HCC
cells to sorafenib by inducing cellular apoptosis and inhibiting
tumor angiogenesis.
Discussion
In the present study, the role of berberine in
sensitizing HCC cells to sorafenib-induced apoptosis was
investigated in vitro. The results indicated that the
combination of berberine and sorafenib exhibited synergistic
inhibition of the SMMC-7721 and HepG2 cell growth. In addition,
berberine enhanced the sorafenib-induced apoptosis in HCC cells.
Thus, these findings suggest the therapeutic value of the
combination of berberine and sorafenib for patients with HCC.
Sorafenib is an orally available multikinase
inhibitor that has been clinically approved for the treatment of
patients with advanced HCC. Nevertheless, the survival benefit
correlated with sorafenib treatment is limited to approximately
three months (23). Almost all
patients become refractory within a few months, and adverse effects
are common, which is associated with the poor prognosis. Therefore,
research has focused on the combination of sorafenib and other
chemotherapeutic agents or inhibitors. It has been reported that
sorafenib in combination with cisplatin or fluorouracil (5-FU) was
beneficial for patients with advanced HCC (24). In the present study, it was found
that the combination of 4 µM sorafenib and 100 µM berberine had a
significant inhibitory effect on HCC cell growth, and that the cell
viability was reduced by approximately 50–70%. This result
indicates that the combination of sorafenib and berberine can
increase the sensitivity of HCC cells to sorafenib. In addition, it
should be pointed out that we did not find that the typical dose of
sorafenib was currently used in cell experiments. To the best of
our knowledge, the IC50 concentration of sorafenib
varies from different cell lines. It has been reported that the
IC50 of sorafenib in HepG2 is 4.3 µM, in Hep3B is 3.1
µM, in HuH7 is 7.3 µM, in HuH6 is 2.8 µM, and in PLC/PRF5 is 5.5 µM
(25). Another report showed that
the IC50 of sorafenib is 20.85±2.81 µM, 10.38±1.52 µM,
10.70±2.35 µM and 9.11±2.44 µM in SMMC-7721, MHCC97-L, MHCC97-H and
HCCLM6 cells, respectively (26).
Therefore, more research is needed to further confirm this
study.
Recently, a growing number of studies have reported
the anticancer activity of traditional agents extracted from herbal
plants (27–29). Berberine is a phytochemical compound
isolated from numerous types of medicinal plants and exhibits
antitumor properties against various malignancies (14,30).
Berberine inhibited the growth of a human colon carcinoma xenograft
in nude mice via exhibiting an inhibitory effect on the
proliferation of colon cancer cells by binding retinoid ×
receptor-α to suppress β-catenin signaling (17). In breast cancer, berberine inhibited
the growth and migration of breast cancer cells via binding to
vasodilator-stimulated phosphoprotein (31). In addition, berberine modulated the
sensitivity of cisplatin through the miR-93/PTEN/AKT signaling
pathway in ovarian cancer cells (32) and attenuated the antitumor activity
of 5-FU, camptothecin and paclitaxel (33). Berberine was also found to sensitize
cancer cells to PARP and epidermal growth factor receptor
inhibitors (18,34). In the present study, berberine and
sorafenib were found to exert anti-proliferative effects on HCC
cells in a dose- and time-dependent manner. Compared with groups
treated with berberine or sorafenib alone, the combined treatment
group inhibited cell proliferation and induced apoptosis, which
demonstrated that berberine and sorafenib exert a synergistic
inhibitory effect on the growth of HCC cells. However, it was
observed that 5 µM berberine increased cell proliferation instead
of suppressing the growth of HCC cells (Fig. 1B). Similar results were detected
with combined treatment of 5 µM berberine and different
concentrations of sorafenib, when compared with the group treated
with sorafenib alone (Fig. 2A and
B). The above results may be caused by the hormetic effect of
berberine. A previous study revealed that low doses (1.25-5 µM) of
berberine promoted cell proliferation to 112–170% of the untreated
control value in cancer cells, but inhibited cell proliferation at
high doses (10–80 µM) (33).
Furthermore, the degree of growth stimulation and the dosage range
of berberine notably varied among different types of cancer cells
(33). These may be the reasons for
the increase in proliferation of HCC cells by 5 µM berberine that
was observed in the present study.
Chemotherapy-induced apoptosis is a common
phenomenon regulated by various apoptosis-associated proteins
(35). It has been reported that
berberine can induce cellular apoptosis in various types of cancer
cells. For instance, Yu et al (36)reported that berberine induced early
and late apoptosis of HepG2 cells, leading to caspase-3 cleavage
and Bcl-2 degradation. Furthermore, it has been demonstrated that
berberine can increase the expression levels of cleaved caspase-3
and cleaved PARP, and decrease Bcl-2 protein levels in lung cancer
cells (37). Consistent with the
findings of previous studies, the results of the present study
revealed that 100 µM berberine induced apoptosis in SMMC-7721 and
HepG2 cells. In addition, combined treatment with 100 µM berberine
and 4 µM sorafenib had a more pronounced effect on apoptosis of HCC
cells. It was also observed that the protein expression levels of
cleaved PARP and cleaved caspase-3 increased, while that of
anti-apoptotic protein Bcl-2 decreased in the combined treatment
group compared with the groups treated with berberine or sorafenib
alone. VEGF has been identified as one of the major initiators in
the development and progression of the vascular system (38). Previous research indicated that
berberine was able to inhibit the protein expression of VEGF
(37). Consistent with this
finding, the current study results demonstrated that berberine
suppressed the expression of VEGF, which was significantly
downregulated in the combined treatment group. VEGF is involved in
the development of tumor vasculature in the liver and the
infiltration of cancer cells into the tumor capsule in HCC
(39). These results suggest that
combined treatment with berberine and sorafenib exerts synergistic
effects on apoptosis and tumor angiogenesis inhibition.
In conclusion, the present study demonstrated for
the first time that berberine synergistically sensitized HCC cells
to sorafenib. Combined treatment with berberine and sorafenib
inhibited the proliferation of HCC cells and markedly induced
cellular apoptosis. These results provide a theoretical basis for
the use of berberine and sorafenib in combination as a new
chemotherapy regimen for HCC. Further investigations are required
to elucidate the molecular and biochemical mechanisms underlying
the anticancer effects of combined treatment with berberine and
sorafenib, and to provide evidence on the efficacy of the combined
treatment in HCC patients.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the
National Natural Science Foundation of China (nos. 81372634 and
81600350), the Guangdong Natural Science Funds for Distinguished
Young Scholar (no. S2013050014121), a research project from the
Guangdong Province Office of Education (no. 2015KTSCX117) and the
Science and Technology Program of Guangzhou (no. 201707010470).
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
YH and KW generated, analysed and interpretated the
data and prepared the manuscript. YH, CG, GY, DZ and WM performed
the experiments. YZ, SL and YN analysed the data and prepared the
manuscript. KW and YH wrote the manuscript. HY generated the idea,
designed the study, analysed and interpretated the data, reviewed
and edited the manuscript. All authors read and approved the
manuscript and agreed to be responsible for all aspects of the
study, ensuring that the accuracy or integrity of any part of the
work is properly investigated and resolved.
Ethics approval and consent to
participate
This study does not contain any experiments with
human subjects or animals.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Siegel RL, Ward EM and Jemal A:
Global cancer incidence and mortality rates and trends-an update.
Cancer Epidemiol Biomarkers Prev. 25:16–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sia D, Villanueva A, Friedman SL and
Llovet JM: Liver cancer cell of origin, molecular class, and
effects on patient prognosis. Gastroenterology. 152:745–761. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dutta R and Mahato RI: Recent advances in
hepatocellular carcinoma therapy. Pharmacol Ther. 173:106–117.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Keating GM: Sorafenib: A review in
hepatocellular carcinoma. Target Oncol. 12:243–253. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Finn RS, Zhu AX, Farah W, Almasri J, Zaiem
F, Prokop LJ, Murad MH and Mohammed K: Therapies for advanced stage
hepatocellular carcinoma with macrovascular invasion or metastatic
disease: A systematic review and meta-analysis. Hepatology.
67:422–435. 2017. View Article : Google Scholar
|
|
6
|
Adnane L, Trail PA, Taylor I and Wilhelm
SM: Sorafenib (BAY 43–9006, Nexavar), a dual-action inhibitor that
targets RAF/MEK/ERK pathway in tumor cells and tyrosine kinases
VEGFR/PDGFR in tumor vasculature. Methods Enzymol. 407:597–612.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wilhelm S, Carter C, Lynch M, Lowinger T,
Dumas J, Smith RA, Schwartz B, Simantov R and Kelley S: Discovery
and development of sorafenib: A multikinase inhibitor for treating
cancer. Nat Rev Drug Discov. 5:835–844. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhou C, Liu J, Li Y, Liu L, Zhang X, Ma
CY, Hua SC, Yang M and Yuan Q: microRNA-1274a, a modulator of
sorafenib induced a disintegrin and metalloproteinase 9 (ADAM9)
down-regulation in hepatocellular carcinoma. Febs Lett.
585:1828–1834. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Potenza N, Mosca N, Zappavigna S,
Castiello F, Panella M, Ferri C, Vanacore D, Giordano A, Stiuso P,
Caraglia M and Russo A: MicroRNA-125a-5p is a downstream effector
of sorafenib in its antiproliferative activity toward human
hepatocellular carcinoma cells. J Cell Physiol. 232:1907–1913.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen J, Jin R, Zhao J, Liu J, Ying H, Yan
H, Zhou S, Liang Y, Huang D, Liang X, et al: Potential molecular,
cellular and microenvironmental mechanism of sorafenib resistance
in hepatocellular carcinoma. Cancer Lett. 367:1–11. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Alsaied OA, Sangwan V, Banerjee S, Krosch
TC, Chugh R, Saluja A, Vickers SM and Jensen EH: Sorafenib and
triptolide as combination therapy for hepatocellular carcinoma.
Surgery. 156:270–279. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li S, Dai W, Mo W, Li J, Feng J, Wu L, Liu
T, Yu Q, Xu S, Wang W, et al: By inhibiting PFKFB3, aspirin
overcomes sorafenib resistance in hepatocellular carcinoma. Int J
Cancer. 141:2571–2584. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ayati SH, Fazeli B, Momtazi-Borojeni AA,
Cicero A, Pirro M and Sahebkar A: Regulatory effects of berberine
on microRNome in Cancer and other conditions. Crit Rev Oncol
Hematol. 116:147–158. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tillhon M, Guaman OL, Lombardi P and
Scovassi AI: Berberine: New perspectives for old remedies. Biochem
Pharmacol. 84:1260–1267. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li-Weber M: Targeting apoptosis pathways
in cancer by Chinese medicine. Cancer Lett. 332:304–312. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tan W, Li Y, Chen M and Wang Y: Berberine
hydrochloride: Anticancer activity and nanoparticulate delivery
system. Int J Nanomedicine. 6:1773–1777. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ruan H, Zhan YY, Hou J, Xu B, Chen B, Tian
Y, Wu D, Zhao Y, Zhang Y, Chen X, et al: Berberine binds RXRα to
suppress β-catenin signaling in colon cancer cells. Oncogene.
36:6906–6918. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang J, Yang S, Cai X, Dong J, Chen Z,
Wang R, Zhang S, Cao H, Lu D, Jin T, et al: Berberine inhibits EGFR
signaling and enhances the antitumor effects of EGFR inhibitors in
gastric cancer. Oncotarget. 7:76076–76086. 2016.PubMed/NCBI
|
|
19
|
Li M, Zhang M, Zhang ZL, Liu N, Han XY,
Liu QC, Deng WJ and Liao CX: Induction of apoptosis by berberine in
hepatocellular carcinoma HepG2 cells via downregulation of NF-κB.
Oncol Res. 25:233–239. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ayati SH, Fazeli B, Momtazi-Borojeni AA,
Cicero A, Pirro M and Sahebkar A: Regulatory effects of berberine
on microRNome in Cancer and other conditions. Crit Rev Oncol
Hematol. 116:147–158. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang XN, Han X, Xu LN, Yin LH, Xu YW, Qi Y
and Peng JY: Enhancement of apoptosis of human hepatocellular
carcinoma SMMC-7721 cells through synergy of berberine and
evodiamine. Phytomedicine. 15:1062–1668. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Youn MJ, So HS, Cho HJ, Kim HJ, Kim Y, Lee
JH, Sohn JS, Kim YK, Chung SY and Park R: Berberine, a natural
product, combined with cisplatin enhanced apoptosis through a
mitochondria/caspase-mediated pathway in HeLa cells. Biol Pharm
Bull. 31:789–795. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Llovet JM, Ricci S, Mazzaferro V, Hilgard
P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A,
et al: Sorafenib in advanced hepatocellular carcinoma. N Engl J
Med. 359:378–390. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ueshima K, Kudo M, Tanaka M, Kumada T,
Chung H, Hagiwara S, Inoue T, Yada N and Kitai S: Phase I/II study
of sorafenib in combination with hepatic arterial infusion
chemotherapy using low-dose cisplatin and 5-fluorouracil. Liver
Cancer. 4:263–273. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Blivet-Van EM, Chettouh H, Fartoux L,
Aoudjehane L, Barbu V, Rey C, Priam S, Housset C, Rosmorduc O and
Desbois-Mouthon C: Epidermal growth factor receptor and HER-3
restrict cell response to sorafenib in hepatocellular carcinoma
cells. J Hepatol. 57:108–115. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang Z, Zhou X, Shen H, Wang D and Wang
Y: Phosphorylated ERK is a potential predictor of sensitivity to
sorafenib when treating hepatocellular carcinoma: Evidence from an
in vitro study. BMC Med. 7:412009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang X and Wu XZ: Main antitumor
angiogenesis agents isolated from chinese herbal medicines. Mini
Rev Med Chem. 15:1011–1023. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lin M, Bi H, Yan Y, Huang W, Zhang G,
Zhang G, Tang S, Liu Y, Zhang L, Ma J, et al: Parthenolide
suppresses non-small cell lung cancer GLC-82 cells growth via
B-Raf/MAPK/Erk pathway. Oncotarget. 8:23436–23447. 2017.PubMed/NCBI
|
|
29
|
He QL, Minn I, Wang Q, Xu P, Head SA,
Datan E, Yu B, Pomper MG and Liu JO: Targeted delivery and
sustained antitumor activity of triptolide through glucose
conjugation. Angew Chem Int Ed Engl. 55:12035–12039. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sun Y, Xun K, Wang Y and Chen X: A
systematic review of the anticancer properties of berberine, a
natural product from Chinese herbs. Anticancer Drugs. 20:757–769.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Su K, Hu P, Wang X, Kuang C, Xiang Q, Yang
F, Xiang J, Zhu S, Wei L and Zhang J: Tumor suppressor berberine
binds VASP to inhibit cell migration in basal-like breast cancer.
Oncotarget. 7:45849–45862. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen Q, Qin R, Fang Y and Li H: Berberine
sensitizes human ovarian cancer cells to cisplatin through
miR-93/PTEN/Akt signaling pathway. Cell Physiol Biochem.
36:956–965. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bao J, Huang B, Zou L, Chen S, Zhang C,
Zhang Y, Chen M, Wan JB, Su H, Wang Y and He C: Hormetic effect of
berberine attenuates the anticancer activity of chemotherapeutic
agents. PLoS One. 10:e1392982015. View Article : Google Scholar
|
|
34
|
Hou D, Xu G, Zhang C, Li B, Qin J, Hao X,
Liu Q, Zhang X, Liu J, Wei J, et al: Berberine induces oxidative
DNA damage and impairs homologous recombination repair in ovarian
cancer cells to confer increased sensitivity to PARP inhibition.
Cell Death Dis. 8:e30702017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Das T, Sa G, Saha B and Das K: Multifocal
signal modulation therapy of cancer: Ancient weapon targets. Mol
Cell Biochem. 336:85–95. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yu R, Zhang ZQ, Wang B, Jiang HX, Cheng L
and Shen LM: Berberine-induced apoptotic and autophagic death of
HepG2 cells requires AMPK activation. Cancer Cell Int. 14:492014.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fu L, Chen W, Guo W, Wang J, Tian Y, Shi
D, Zhang X, Qiu H, Xiao X, Kang T, et al: Berberine Targets
AP-2/hTERT, NF-κB/COX-2, HIF-1α/VEGF and cytochrome-c/caspase
signaling to suppress human cancer cell growth. PLoS One.
8:e692402013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ferrara N, Gerber HP and LeCouter J: The
biology of VEGF and its receptors. Nat Med. 9:669–676. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wang H, Zhang C, Ning Z, Xu L, Zhu X and
Meng Z: Bufalin enhances anti-angiogenic effect of sorafenib via
AKT/VEGF signaling. Int J Oncol. 48:1229–12241. 2016. View Article : Google Scholar : PubMed/NCBI
|