Introduction
Gastric cancer (GC) is the fifth most common type of
cancer worldwide and the third leading cause of cancer-related
mortality, with approximately 951,600 cases and 723,100 GC-related
deaths in 2012 (1). Common
therapeutic strategies for GC include surgical resection,
chemotherapy, radiotherapy and anti-angiogenic therapy (2). However, the efficacy of current
treatment regimens for GC are hindered by multiple factors such as
chemotherapy and radiotherapy resistance, and tumor relapse
(3). At present, cancer stem cells
(CSCs) are regarded as a crucial population contributing to the
irresponsiveness to GC treatment and the poor prognosis (4). CSCs can initiate tumor formation, and
promote self-renewal, therapy resistance, metastasis and tumor
recurrence (5,6). As a result, it is of great importance to
determine the key molecules controlling the malignant properties of
gastric CSCs (GCSCs).
GCSCs were first identified in 2007 by Yang et
al (7). CD44, CD24/CD44 and
aldehyde dehydrogenase (ALDH)1 have been identified as GCSCs
markers (8,9). Numerous previous studies have revealed
the therapeutic value of targeting CSC markers for GC intervention;
for example, Gong et al (10)
reported that leucine rich repeat containing G protein-coupled
receptor 5 (LGR5) antibody conjugates induced cytotoxicity in GC
cells overexpressing LGR5. In addition, all-trans retinoic
acid treatment inhibited GC progression in mouse xenograft models
by downregulating the expression levels of CD44 and ALDH1 (11). Recently, an increasing number of
therapeutics targeting stemness-associated functions have been
designed to specifically eradicate GCSCs, including those that
inhibit stemness-associated genes, block self-renewal signaling
pathways and microenvironment-based anti-GCSC therapies (12). One of the most characterized pathways
contributing to the function of CSCs is the Wnt signaling pathway,
and the aberrant activation of the Wnt signaling pathway has been
revealed to promote stem cell characteristics of CSCs and initiate
the epithelial-mesenchymal transition (EMT) process (13). Due to the pivotal role of CSCs in
tumor progression and metastasis, an improved understanding of the
regulatory elements that control the malignant behaviors of GCSCs
may lead to the development of effective therapies for patients
with GC.
Retinoic acid-related orphan receptor β (RORβ) is a
member of the orphan nuclear receptor family (14). RORβ was originally considered to be
expressed solely in the central nervous system (CNS), mainly in the
regions modulating the circadian rhythm (15). However, RORβ has since been
demonstrated to be expressed in other regions of the body,
including bone tissue, pancreatic cancer tissue and colorectal
cancer tissue (16,17). Risinger et al reported that the
expression levels of RORβ were upregulated in women with
endometrial cancer compared with healthy women (18). However, despite the reported
expressional changes of RORβ, the pathological significance of RORβ
remains largely unknown. Therefore, the present study aimed to
investigate the expression levels and function of RORβ in GC, and
identified RORβ as a novel suppressor of GCSCs. These findings may
help to propose a valuable target for the stem cell-based therapy
of GC in the future.
Materials and methods
Patient studies
A total of 32 patients with GC at the General
Surgery Department of Sir Run Run Shaw Hospital (Hangzhou, China)
were selected from January 2019 to December 2019. There were 18
males and 14 females with an age range of 34–76 years (average age
61.5 years old). The fresh operative GC tissues and corresponding
para-cancerous tissues were collected. The pathological stage was
determined according to the American Joint Committee on Cancer
(AJCC) and the International Union against Cancer (UICC) seventh
edition TNM staging system (19). The
inclusion criteria were as follows: i) radical gastrectomy or
palliative surgery for GC and pathological diagnosis was GC; ii)
preoperative radiotherapy, and chemotherapy were not performed;
iii) complete clinicopathological data were available. The
exclusion criteria were as follows: i) gastric cancer recurrence or
residual gastric cancer; ii) combined with organ dysfunction or
other tumors. All patients provided written informed consent prior
to participation and the present study was approved by the
Institutional Review Board from Sir Run Run Shaw Hospital (approval
no. 20210429-30).
Cell culture
GC cell lines, AGS and MKN45, were purchased from
The Cell Bank of Type Culture Collection of the Chinese Academy of
Sciences. AGS cells were cultured in DMEM (Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% FBS (Thermo Fisher
Scientific, Inc.), while MKN45 cells were cultured in RPMI-1640
medium (Gibco; Thermo Fisher Scientific, Inc.) supplemented with
10% FBS. All GC cells were incubated at 37°C and 5% CO2.
GCSCs were cultured from GC cells at a density of 1×105
cells/ml in serum-free medium. EGF (20 g/l; Invitrogen; Thermo
Fisher Scientific, Inc.), bFGF (20 g/l; Invitrogen; Thermo Fisher
Scientific, Inc.), B27 (2%; Invitrogen; Thermo Fisher Scientific,
Inc.), BSA (0.4%; Roche Diagnostics), insulin (4 mg/l; Invitrogen;
Thermo Fisher Scientific, Inc.) and gentamicin (200 IU/ml;, Sangon
Biotech, Co., Ltd.) were added to DMEM/F-12 (Gibco; Thermo Fisher
Scientific, Inc.) in 4-well low adhesion culture plates in an
incubator at 37°C with 5% CO2 for 7–10 days.
Screening and identification of stable
RORβ-overexpression GC cells
The RORβ gene coding sequence from RORβ/pReceiver
plasmid (GeneCopoeia, Inc.) was inserted into pEGFP-C1 vector at
the BamHI site and identified by DNA sequencing (Sangon
Biotech Co., Ltd.). GC cells were seeded into 6-well plates at a
density of 1×106 cells/well at 37°C with 5%
CO2 for 24 h. RORβ/pEGFP-C1 (4 µg/µl) and pEGFP-C1
vector (4 µg/µl) were transfected into GC cells using
Lipofectamine® 3000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.). After 2 days, the cells were cultured in the
presence of 500 µg/ml G418 (Invitrogen; Thermo Fisher Scientific,
Inc.) for 2 weeks to obtain stably transfected GC cells.
RORβ-overexpression GC cells were further verified by western
blotting.
Screening stable RORβ-knockdown GC
cells
GC cells were transfected with 4 µg/µl RORβ-short
hairpin (shRNA) and 4 µg/µl control shRNA (both from Santa Cruz
Biotechnology, Inc.) using Lipofectamine® 3000 reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) for 2 days and then
the cells were screened with puromycin (5 µg/ml) for 14 days to
obtain the stable RORβ-knockdown cells further verified by western
blotting.
Nuclear/cytosolic fractionation
Ice-precooled CER I (200 µl) was added to GC cells
and the cells were incubated on ice for 10 min after vortex
oscillation for 15 sec using nuclear and cytoplasmic extraction
reagents (NE-PER) (Pierce; Thermo Fisher Scientific, Inc.).
Ice-precooled CER II (11 µl) was added and incubated, then swirled
and shaken for 5 sec, and incubated on ice for 1 min. The
supernatant (cytoplasm) was transferred to a clean ice-precooled
centrifuge tube for preservation on ice. The sediment was suspended
with ice-precooled NER 100 µl, incubated on ice for 10 min, and
vortex oscillated 4 times for 15 sec. After centrifugation at
16,000 × g for 10 min at 4°C, the supernatant was transferred to a
clean ice-precooled centrifuge tube (cell nucleus), and then
western blotting was performed.
Sphere formation assay
RORβ-overexpression and RORβ-knockdown GC cells and
control cells were digested and cultured in 24-well plates at a
density of 200 cells/well in serum-free medium incubated at 37°C
with 5% CO2 for 7 days. The medium was replaced every
3–4 days. Following the incubation, the spheres were counted
manually using a light microscope with a magnification of ×20.
CCK-8 assay
RORβ-overexpression and RORβ-knockdown cells and
control cells were seeded into 96-well plates at a density of
1×104 at 37°C with 5% CO2 for 24 h. Following
incubation, 10 µl CCK-8 reagent (Abcam) was added/well and
incubated for 4 h. Subsequently, the absorbance value was measured
with a microplate reader at a wavelength of 460 nm.
Flow cytometric analysis of
apoptosis
RORβ-overexpression GC cells and control cells were
trypsinized, and 1×106/ml cells were collected by
centrifugation (300 × g, 5 min at 4°C). Cells were washed with PBS
twice and incubated in 500 µl binding buffer, 5 µl Annexin V-EGFP
and 10 µl propidium iodide at room temperature in the dark for 15
min using Annexin V-FITC/PI Apoptosis Detection Kit (Beyotime
Institute of Biotechnology) according to the manufacturer's
instructions. Apoptotic cells were visualized and quantified using
FACSAria (BD Biosciences) with FlowJo software (version 7.6.1; Tree
Star, Inc.).
Nude mouse xenograft model
All animal experiments were approved by the
Institutional Animal Care and Use Committee of Zhejiang University
(Hangzhou, China) and complied with the Animal Welfare Act
(20). BALB/c nude mice (30 females;
4 weeks old; body mass 17–20 g) were purchased from Shanghai
Laboratory Animal Center. The mice were bred at the Laboratory
Animal Research Center at Sir Run Run Shaw Hospital in a barrier
environment, specific pathogen-free (SPF) grade, with humidity
approximately 30 to 50% at 22°C. In addition, water and food was
provided ad libitum. A total of 0.2 ml RORβ-overexpression
GC cells and control cells (at the density of 1×105,
5×105 and 1×106) were subcutaneously
inoculated into the backs of nude mice. There were five mice in
each group. Tumor growth was observed weekly, and rats were
sacrificed by cervical spinal cord transection after 4 weeks, and
the tumor body was measured and analyzed. The tumor volumes were
assessed according to the diameters of the tumors.
Immunohistochemistry (IHC)
All GC tissue specimens were fixed with 10% neutral
formaldehyde solution for 4–6 h at room temperature, and were
routinely dehydrated, waxed and wrapped. Then, the tissues were cut
into 3-µm sections. The tissue sections were baked at 60°C
overnight. Then, the tissue sections were subsequently
deparaffinized by the following method: After baking, the tissue
slices were immersed in xylene at room temperature twice for 10 min
each. Then they were immersed in 100% ethanol for 5 min. Next, the
tissue sections were rehydrated in 100, 85 and 70% ethanol at room
temperature for 2 min respectively. Finally, the slices were
incubated at 90°C in deionized water for 30 min. Subsequently, the
slices were boiled in citric acid buffer solution and maintained at
a low heat for 30 min. The sections were then sealed with 5% BSA
(Sangon Biotech Co., Ltd.) in a wet box at 37°C for 30 min and
air-dried. The sections were then incubated in a wet box at 4°C
overnight with an anti-RORβ antibody (cat. no. NBP1-82532; 1:300;
Novus Biologicals, LLC). Following the primary antibody incubation,
the sections were rinsed thrice with PBS for 5 min and incubated
with 50 µl of an HRP-conjugated secondary goat anti-rabbit antibody
(product code ab6721; 1:1,000; Epitomics; Abcam) at room
temperature for 20 min. Then the sections were rinsed thrice with
PBS for 3 min. The slides were observed under a light microscope
with a magnification of ×100.
Wound healing assay
MKN45 and AGS cells were plated into 96-well plates
and incubated overnight. Approximately 1×104 cells in
each well were serum-starved overnight, and then the head of the
pipette tip was used to produce a scratch in the cell monolayer.
The cells were then washed with PBS 3 times to remove the
unattached cells and subsequently incubated in fresh culture medium
without FBS for indicated time-points at 37°C. Images of the wound
were captured at 0, 12 and 24 h using a light microscope
(magnification, ×20). The percentage of migration area was
calculated using ImageJ software (1.52v; National Institutes of
Health).
F-actin polymerization experiment
AGC cells grown on cover-slips inside a petri dish
were transfected with RORβ/pReceiver plasmid and control and were
incubated at 37°C for 2 days using Lipofectamine® 3000
reagent (Invitrogen; Thermo Fisher Scientific, Inc.). The cells
were washed once with PBS. Then, the cells were incubated with 3–4%
formaldehyde in PBS at room temperature for 30 min. The staining
solution was aspirated carefully and the fixed cells were washed
2–3 times in PBS. Conjugate working solution 1X Phalloidin-iFluor™
488 (AAT Bioquest, Inc.) was added to the fixed cells.
Subsequently, the cells were incubated at room temperature for 60
min. DAPI staining solution (BIOSS) was then added and the cells
were incubated at room temperature for 5 min. The cells were rinsed
gently 2–3 times with PBS to remove excess phalloidin conjugate.
Mounting medium was added and sealed. The cells were observed at a
magnification of ×400 under a fluorescence microscope (Olympus
Corporation) with an FITC filter set.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA from GC cells was extracted using RNeasy
Mini Kit (cat. no. 74104; Qiagen GmbH) and then reverse transcribed
into cDNA using PrimeScript RT reagent kit (cat. no. RR037A; Takara
Bio, Inc.) according to the manufacturer's protocol. RT-qPCR was
performed using Premix Ex Taq (cat. no. RR420A; Takara Bio, Inc.).
The thermocycling conditions were as follows: Initial denaturation
at 95°C for 5 min; annealing and elongation at 95°C for 30 sec,
95°C for 5 sec, 60°C for 34 sec for 40 cycles; and final extension
at 60°C for 10 min. The sequences of the primer pairs used were:
Oct4 forward, 5′-GTGGAGGAAGCTGACAACAA-3′, reverse,
5′-AACAAATTCTCCAGGTTGCC-3′ and probe,
5′-Fam-TCTCTTTCGGGCCTGCACGA-3′Tamra; Sox2 forward,
5′-AATGCCTTCATGGTGTGG−3′, reverse, 5′-CTTCTCCGTCTCCGACAAA-3′, and
probe, 5′Fam-AGTTTCCACTCGGCGCCCAG-3′Tamra; KLF4 forward,
5′-GGCACTACCGTAAACACACG-3′, reverse, 5′-CTGGCAGTGTGGGTCATATC-3′,
and probe, 5′Fam-CAGGTCGGACCACCTCGCCT-3′Tamra; CD44 forward,
5′-TGGCAACAGATGGCATGAGG-3′, reverse, 5′-CCTTGCATTGGATGGCTGGT-3′ and
probe, 5′Fam-AACAGGGACAGCTGCAGCCTCAGCT-3′Tamra; ALDH1 forward,
5′-GCTGGCGACAATGGAGTCAA-3′, reverse, 5′-TGTACGGCCCTGGATCTTGT-3′ and
probe, 5′Fam-ACATTGCGCTACTGTGCAGGTTGGGCT−3′ Tamra; Slug forward,
5′-TCACTGTGTGGACTACCGCT-3′, reverse, 5′-TCGCCCCAAAGATGAGGAGT-3′ and
probe, 5′Fam-ATTCCACGCCCAGCTACCCAATGGCCT−3′Tamra; Twist forward,
5′-CCCTCGGACAAGCTGAGCAA-3′, reverse, 5′-TCGCTCTGGAGGACCTGGTA-3′ and
probe, 5′Fam-ATTCAGACCCTCAAGCTGGCGGCCA-3′Tamra; vimentin forward,
5′-CAATGAGTCCCTGGAACGCC-3′, reverse, 5′-GGTGACGAGCCATTTCCTCC-3′ and
probe, 5′Fam-ACCAAGACACTATTGGCCGCCTGCAGG−3′Tamra; GAPDH forward,
5′-ATCATCCCTGCCTCTACTGG-3′, reverse, 5′-GTCAGGTCCACCACTGACAC-3′ and
probe, 5′Fam-ACCTTGCCCACAGCCTTGGC-3′Tamra; The relative mRNA
expression levels were calculated using 2−ΔΔCq method
(21).
Western blotting
Cells were lysed using RIPA Lysis Buffer (Beyotime
Institute of Biotechnology). The protein concentrations were
determined using the bicinchoninic acid (BCA) assay. Proteins were
separated via 12% SDS-PAGE and blocked with 5% bovine serum albumin
at 4°C overnight. A total of 20 ml of 12% separation gel was
prepared, and the 5% stacking gel was added on top of the
separation gel. In total, 20–40 µl protein samples with 5 µl
protein marker were added to the corresponding lanes. Then the
protein samples were transferred to the cellulose nitrate membranes
by electrophoresis. The cellulose nitrate membranes were incubated
with the following primary antibodies at 4°C overnight: Anti-c-Myc
(product code ab32072; 1:2,000), anti-cyclin D1 (product code
ab16663; 1:2,000), anti-Slug (product code ab51772; 1:2,000),
anti-Twist (product code ab50887; 1:2,000), anti-E-cadherin
(product code ab40772; 1:2,000), anti-N-cadherin (product code
ab76011; 1:2,000), anti-vimentin (product code ab92547; 1:2,000),
anti-Sox2 (product code ab92494; 1:2,000), anti-Oct4 (product code
ab200834; 1:2,000), anti-KLF4 (product code ab215036; 1:2,000),
anti-ALDH1 (product code ab177463; 1:2,000), anti-CD44 (product
code ab189524; 1:2,000), anti-phosphorylated (p)-β-catenin (product
code ab81305; 1:2,000) and anti-RORβ antibody (product code
ab228650; 1:1,000; all from Epitomics; Abcam), anti-Bcl-2 like
protein 11 (BCL2L11; cat. no. AP8553c; 1:1,000; Abgent, Inc.) and
anti-GAPDH (cat. no. KC-5G5; 1:5,000; KangChen Biotech Co., Ltd.).
Then the cellulose nitrate membranes were incubated with the
secondary antibodies at room temperature for 1 h: Goat anti-rabbit
IgG H&L (HRP) (product code ab6721; 1:2,000) or goat anti-mouse
IgG H&L (HRP) (product code ab6789; 1:2,000; both from
Epitomics; Abcam). Protein bands were visualized with ECL reagent
(PerkinElmer, Inc.), according to the manufacturer's protocol.
BandScan software (V5.0; Glyko Biomedical, Ltd.) was used for
densitometric analysis.
Gene chip detection and Gene Ontology
(GO) analysis
GC cells overexpressing RORβ were lysed by 1 ml
TRIzol reagent (Sangon Biotech, Co., Ltd.) and total RNA was
extracted using PrimeScript RT reagent Kit gDNAEraser (cat. no.
RR047A; Takara Bio, Inc.) according to the manufacturer's
instructions. A GeneChip (HTA 2.0; Affymetrix; Thermo Fisher
Scientific, Inc.) was used to identify differentially expressed
mRNAs. Then 460 differentially expressed genes in the Genechip were
imputed into DAVID (http://david.abcc.ncifcrf,gov/home.jsp) to perform the
GO analysis (22).
Gene Set Enrichment Analysis
(GSEA)
All 200 mRNA GeneChip databases containing patients
with GC were obtained from the City of Hope National Medical
Center, Los Angeles, USA. GSEA (23)
was used to analyze the association between RORβ expression levels
and GCSC markers.
Top/Fop luciferase reporter assay
RORβ-overexpression GC cells were seeded into 6-well
plates at a density of 1×106 at 37°C with 5%
CO2 for 24 h. A total of 8 µl Fugene 6 (Roche
Diagnostics) was incubated with 100 µl OPTI-MEM (Invitrogen; Thermo
Fisher Scientific, Inc.) at room temperature for 5 min. Then, a
total of 2 µg plasmid mixture including TOP (MilliporeSigma), FOP
(MilliporeSigma) and PRL (Promega Corporation) (4:4:1) were added
to the aforementioned mixture and incubated at room temperature for
15 min. Then the mixture of Fugene 6 and plasmids were added to the
6-well plate and mixed, and incubated at 37°C in a 5%
CO2 incubator for 24 h. The cell lysates were
transferred to the centrifuge tube after cell lysis. GloMax 20/20
Luminometer was used to detect the fluorescence activity. A total
of 20 µl of the aforementioned cell lysates were added with 100 µl
of LAR II. After mixing, firefly luciferase activity was detected.
Then 100 µl Stop&Glo Reagent was added to detect Renilla
luciferase activity. The Dual-Luciferase® Reporter
(DLR™) Assay System (Promega Corporation) was used.
Statistical analysis
Statistical analysis was performed using SPSS 19.0
software (IBM Corp.). Statistical differences between groups were
performed using one way ANOVA (with least significant difference
test or Tamhane's T2 post hoc test) or unpaired Student's t-test.
P<0.05 was considered to indicate a statistically significant
difference. All data are presented as the mean ± SD of triplicate
experiments.
Results
Expression levels of RORβ are
downregulated in GC
RORβ was previously reported to be expressed in the
CNS (24); however, the expression
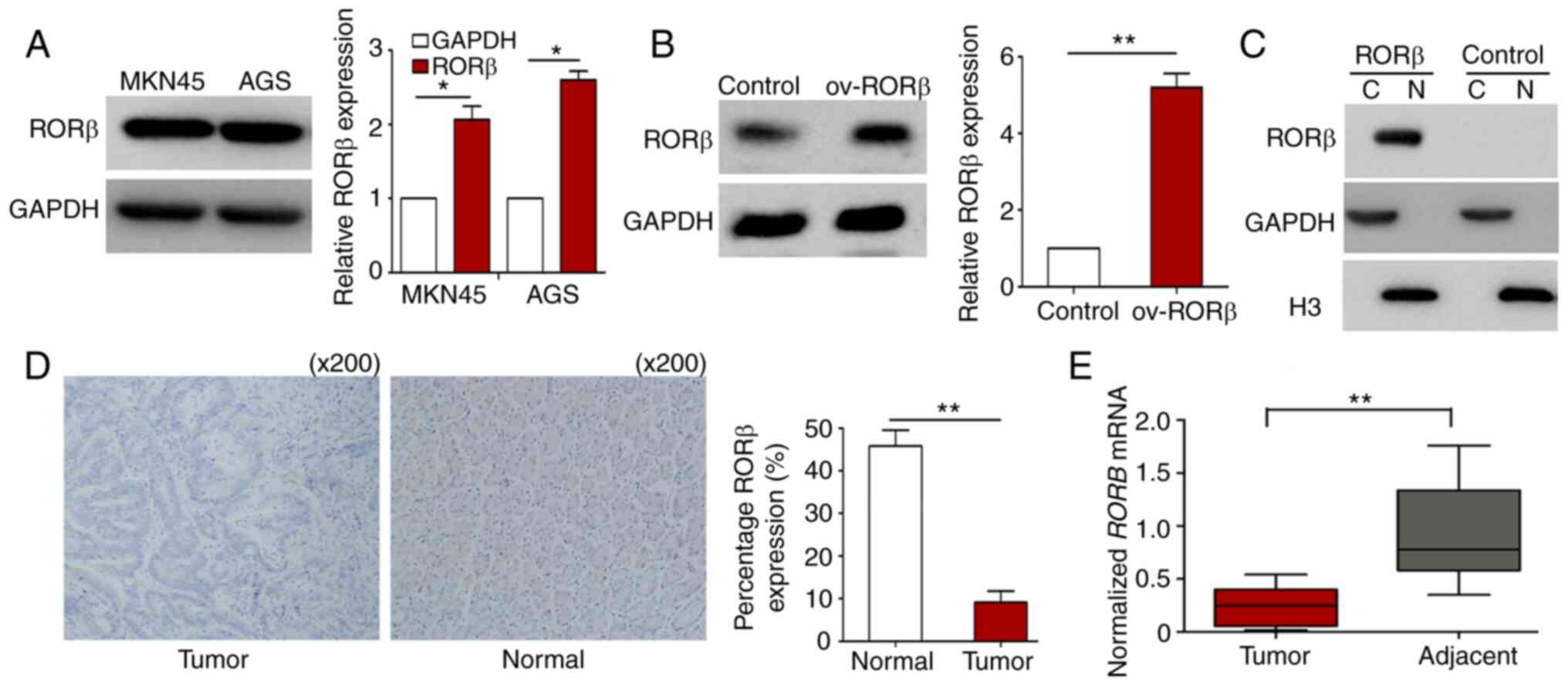
levels of RORβ in GC cells remain unknown. As demonstrated in
Fig. 1A, RORβ was expressed in the
two GC cell lines, MKN45 and AGS. AGS cells were subsequently
transfected with the RORβ-pEGFP-C1 plasmid and western blotting was
used for confirmation (Fig. 1B). The
nuclear localization of RORβ was further confirmed using western
blotting following nuclear/cytosolic fractionation (Fig. 1C). IHC staining of clinical GC
specimens revealed that the expression levels of RORβ were
significantly downregulated in GC tissues compared with
para-carcinoma tissues (Fig. 1D).
RT-qPCR analysis further determined that RORβ expression levels
were downregulated in GC tissues (Fig.
1E). These results indicated that RORβ expression levels may be
downregulated during GC development.
RORβ expression levels are inversely
associated with GCSC markers
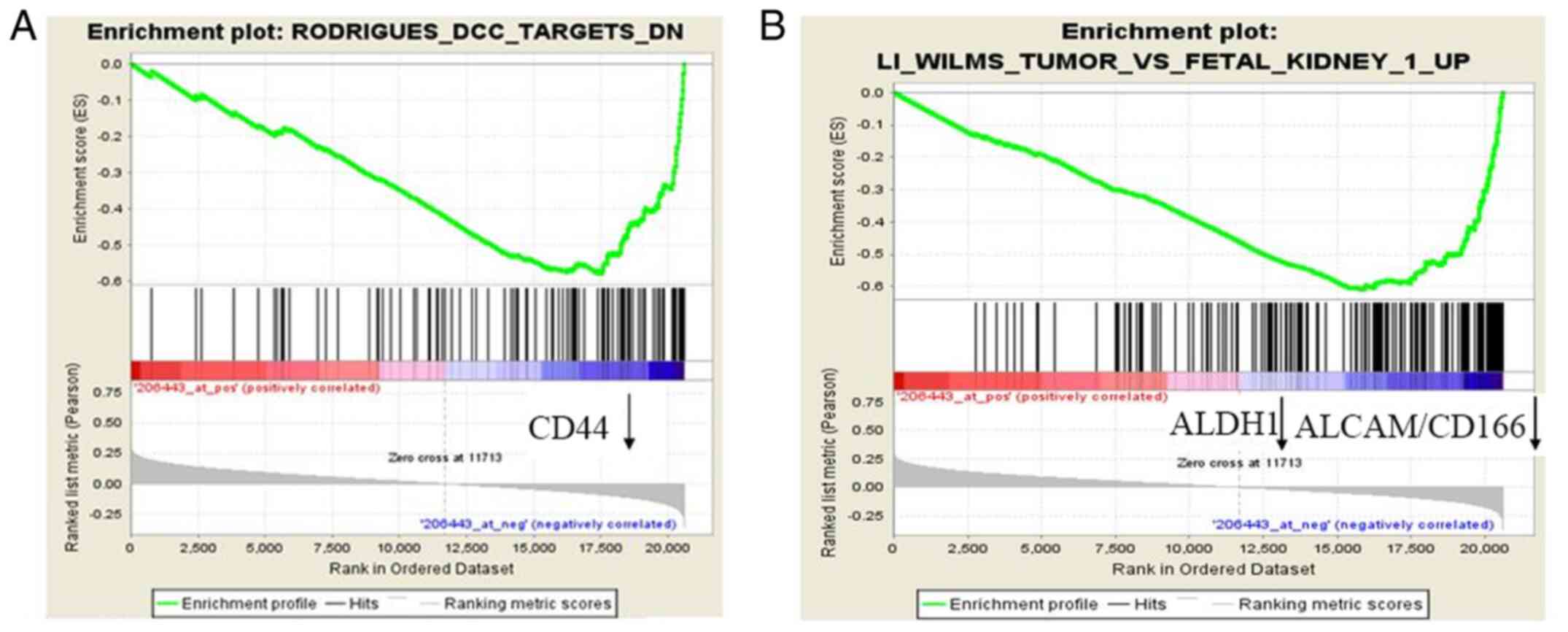
The mRNA expression levels of RORβ in 200 GC tissues
were divided into two groups: RORβ high (RORβhigh) and
low (RORβlow) expression groups. Differentially
expressed genes were identified using GSEA. The results revealed
that the expression levels of CSC markers, including ALDH1, CD44
and activated leukocyte cell adhesion molecule (ALCAM)/CD166, were
all downregulated in the RORβhigh cohort compared with
the RORβlow cohort (Fig. 2A
and B). These results indicated that RORβ may be a cell
differentiation inducer.
RORβ induces growth inhibition and
apoptosis in GC cells
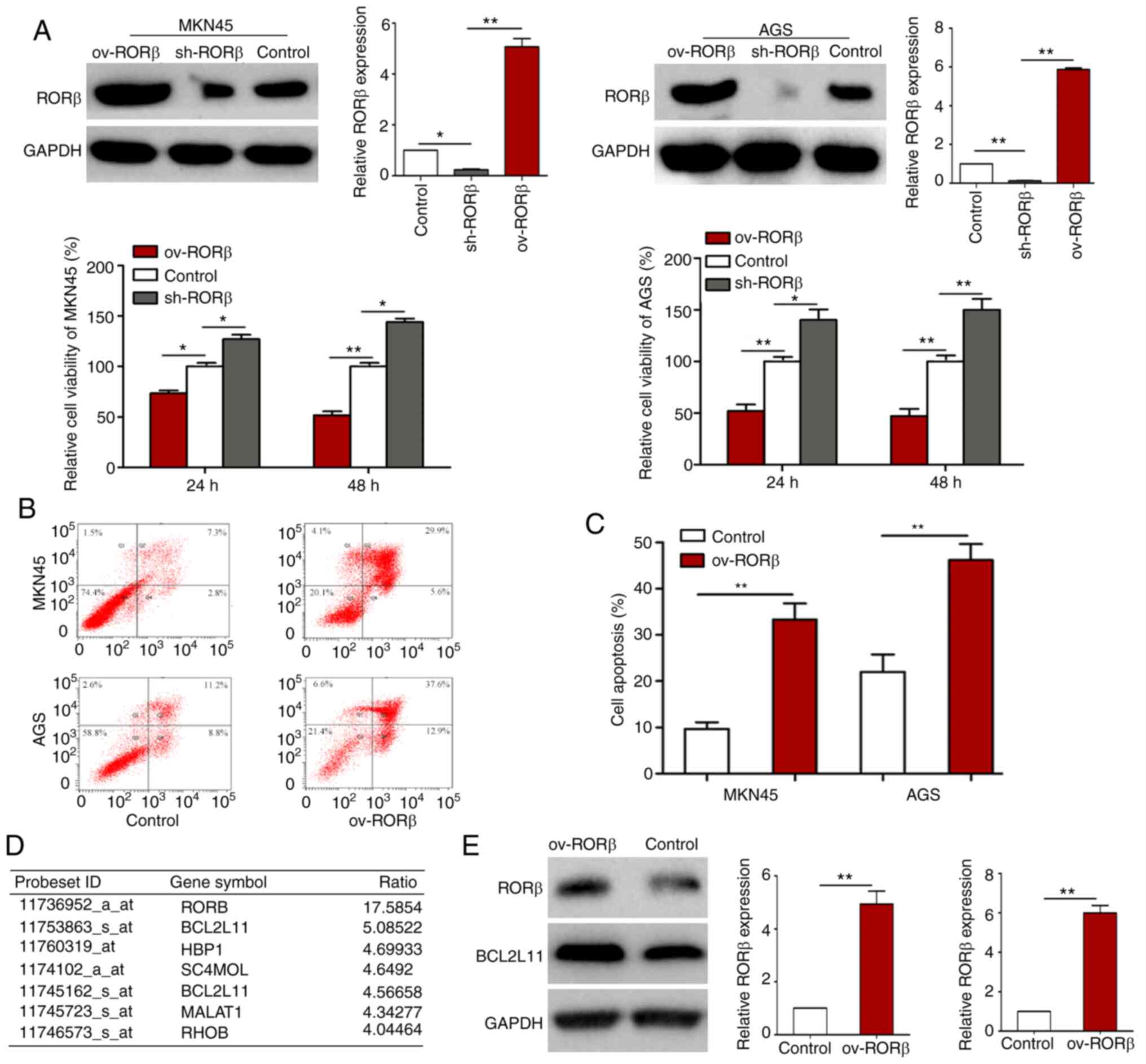
To determine the functional impact of RORβ on GC
cells, a CCK-8 assay was performed. The viability of GC cells was
significantly inhibited following the overexpression of RORβ
(Fig. 3A). Conversely, the
percentages of Annexin V-positive and PI-positive apoptotic GC
cells were significantly increased in GC cells overexpressing RORβ
(Fig. 3B and C). Subsequently, the
changes in the expression levels of genes between control and
RORβ-overexpressing AGS cells were analyzed using GeneChip. The
results revealed that BCL2L11, a pro-apoptotic gene, was the most
significantly upregulated gene in cells overexpressing RORβ, which
was further verified using western blotting (Fig. 3D and E). In addition, GO analysis
discovered that RORβ was mainly involved in ‘nucleoside’,
‘nucleotide’, ‘nucleic acid metabolism and mRNA transcription’,
‘cell cycle’, ‘cell proliferation and differentiation’ and ‘tumor
formation’ (data not shown). These results indicated that RORβ may
inhibit viability, but trigger BCL2L11-dependent apoptosis in GC
cells.
RORβ confers resistance to EMT in GC
cells
Previous studies have reported that EMT may be
related to the stemness of CSCs (25–27).
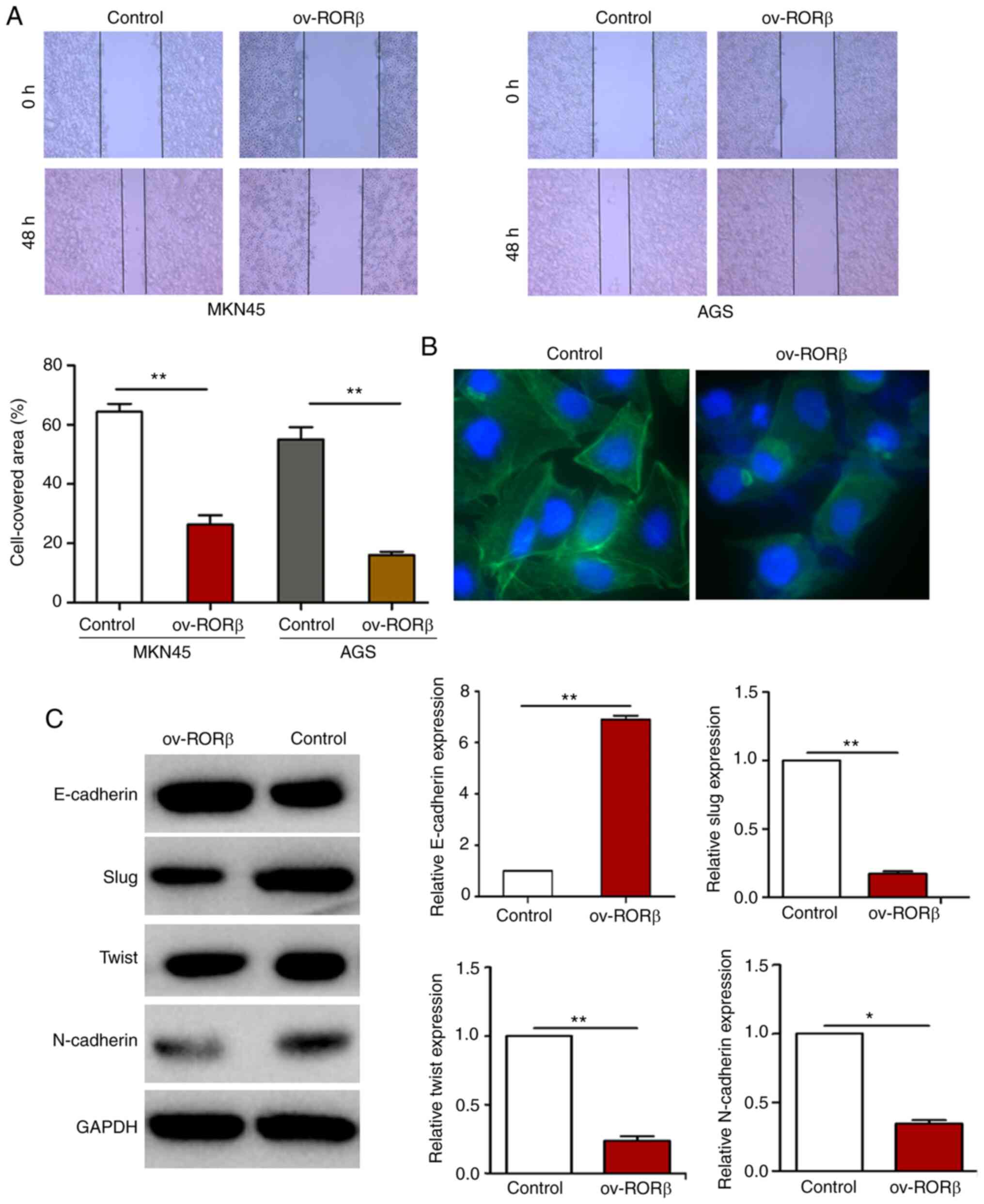
Therefore, whether RORβ played a role in modulating the metastatic
ability of GC cells was investigated. The results of the wound
healing assay demonstrated that the migratory ability was decreased
in GC cells overexpressing RORβ compared with control cells
(Fig. 4A). Immunofluorescence
staining of F-actin revealed a downregulated fluorescence signal in
RORβ overexpression GC cells, which indicated RORβ overexpression
inhibited F-actin polymerization and arrangement (Fig. 4B). Consistent with the increased cell
migratory ability, RORβ overexpression downregulated the expression
levels of EMT-related factors (Slug, Twist and N-cadherin) and
upregulated the expression levels of E-cadherin (Fig. 4C). These findings indicated that RORβ
may suppress EMT in GC cells.
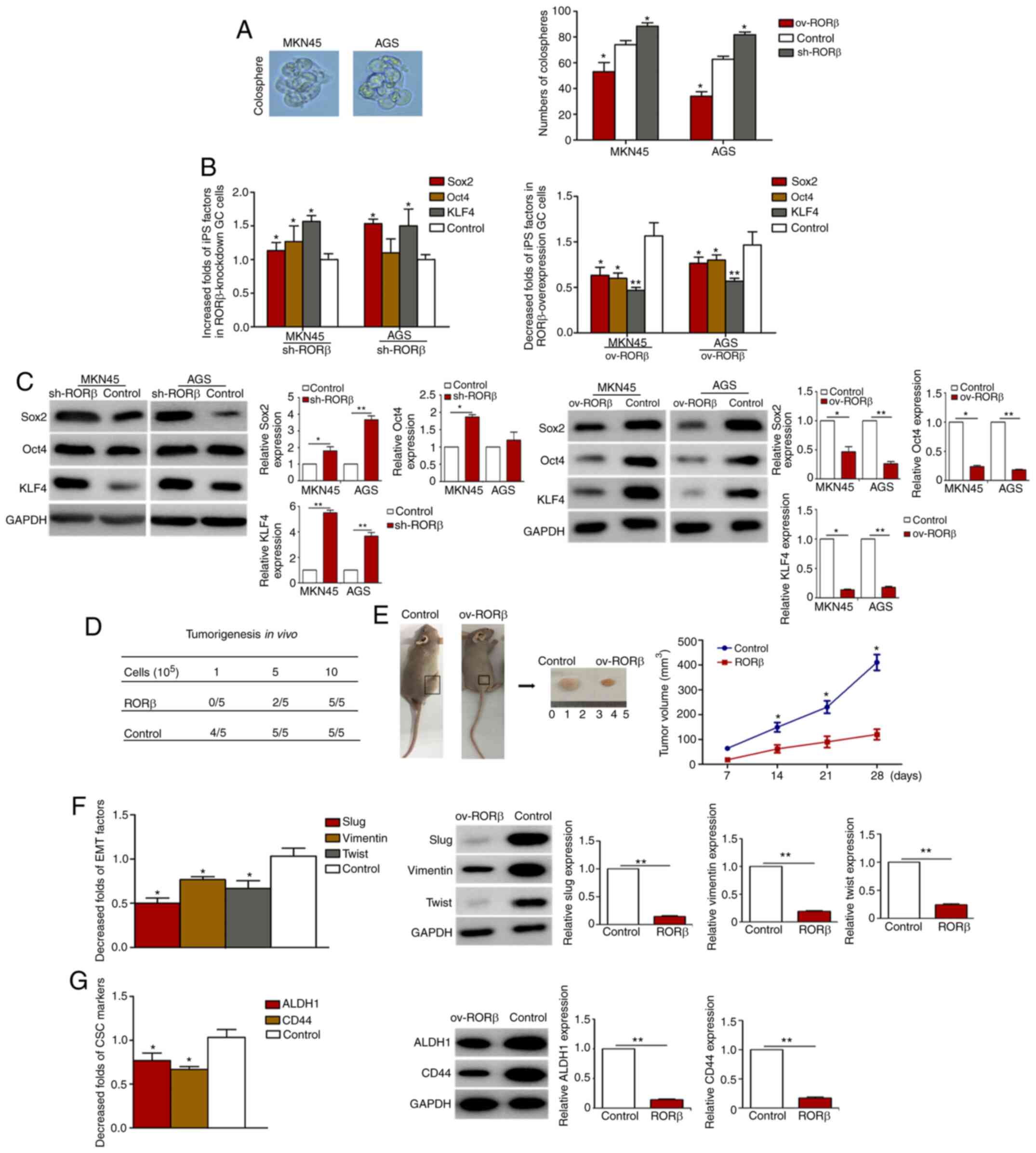
RORβ decreases the stemness of
GCSCs
As an inverse association was identified between
RORβ expression levels and several GCSC markers, it was
subsequently investigated whether RORβ was able to affect the
stemness of GCSCs. The results demonstrated that the sphere forming
ability was significantly decreased in GC cells following RORβ
overexpression, while the genetic silencing of RORβ using shRNA
caused the opposite effect, indicating the inhibitory role of RORβ
on cell stemness (Fig. 5A).
Similarly, the expression levels of iPS cell-related factors (Sox2,
Oct4 and KLF4) were downregulated in GC cells overexpressing RORβ
and upregulated in GC cells following the knockdown of RORβ, which
was further verified using western blotting (Fig. 5B and C). RORβ-overexpressing AGS cells
were subsequently inoculated into nude mice to evaluate the effect
on the tumorigenic capacity and stemness of GC cells in
vivo. The nude mice were inoculated with RORβ overexpression GC
cells and control cells (at the density of 1×105,
5×105 and 1×106) cells. At the fourth week,
there was no significant difference in the tumor-forming rate of
the third group (at the density of 1×106), while there
were significant differences in the tumor-forming rate of the first
(at the density of 1×105) and second groups (at the
density of 5×105) (Fig.
5D). As shown in Fig. 5E-G, RORβ
overexpression markedly inhibited the tumor growth of GC cells and
downregulated the expression of EMT factors and GCSC markers of
tumors in mice. These results indicated that RORβ may disrupt the
stemness of GCSCs.
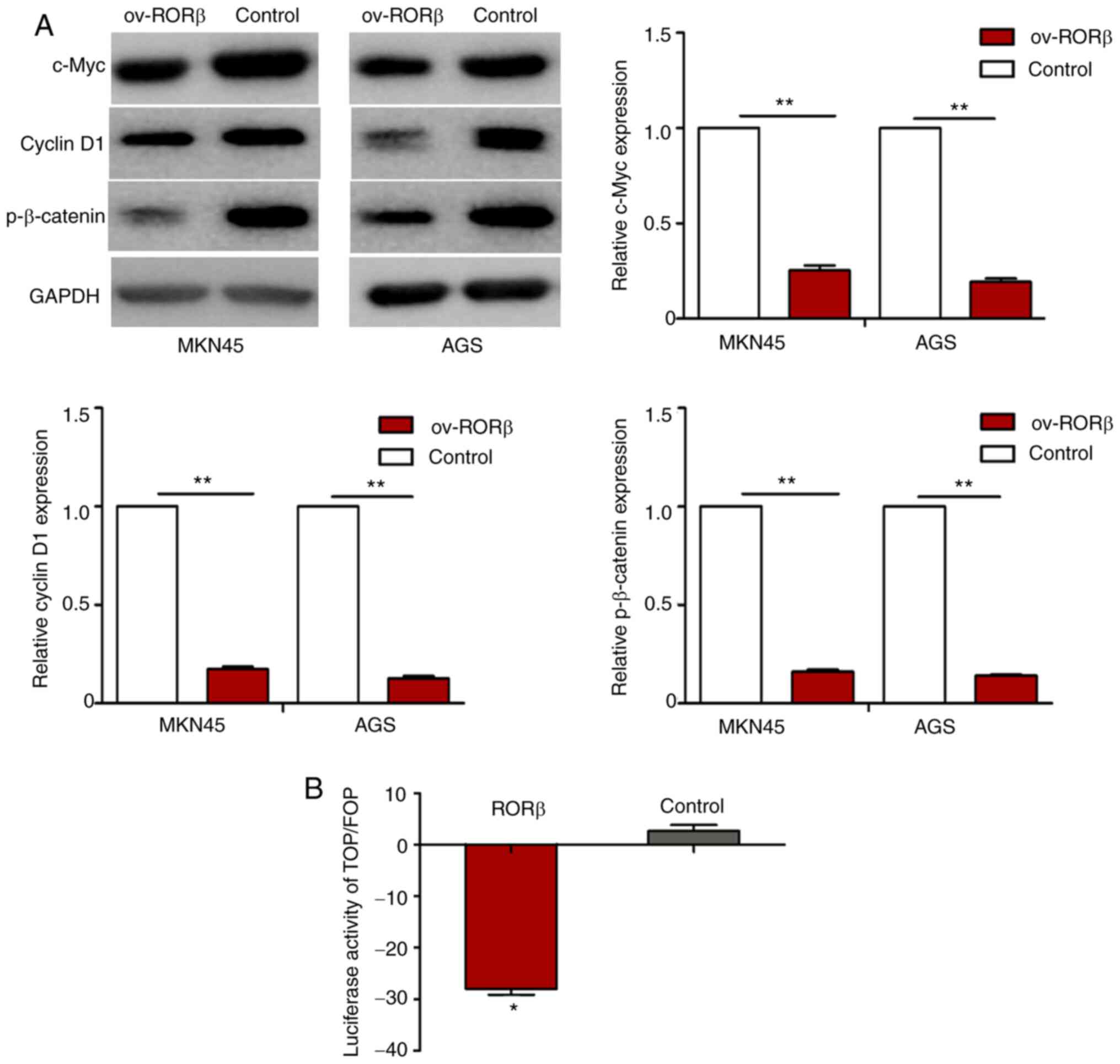
RORβ negatively regulates the activity
of the Wnt signaling pathway
Finally, the molecular mechanisms underlying the
suppressive effect of RORβ on GCSCs were investigated. As a
well-characterized oncogenic pathway, the abnormal activation of
the Wnt signaling pathway has been implicated in the stemness
maintenance of CSCs (13). The
expression levels of c-Myc, cyclin D1 and p-β-catenin, two target
genes downstream of the Wnt signaling pathway (28), were significantly downregulated in GC
cells overexpressing RORβ (Fig. 6A).
The results of the luciferase reporter assay revealed that the
overexpression of RORβ significantly reduced the Top/Fop luciferase
activity in GC cells (Fig. 6B). These
findings suggested that RORβ may impair the stemness of GCSCs by
hindering Wnt/β-catenin signaling.
Discussion
It is widely acknowledged that CSCs, a small group
of cells with self-renewal and strong tumorigenic properties, serve
important roles in tumorigenesis, metastasis, recurrence and
chemoradiotherapy resistance (29,30). GCSCs
were first identified by Yang et al (7). Since this discovery, further studies
have identified the presence of GCSCs (8–11).
However, the mechanisms of GCSCs remain unknown. RORβ was firstly
identified to be expressed in GC in the present study, where it
served as a novel inhibitor of EMT and CSC properties by
downregulating the Wnt signaling pathway.
RORβ is a member of the orphan nuclear receptor
family. RORβ was previously considered to be only expressed in the
CNS (15); however, accumulating
studies have reported that RORβ is also localized in various other
systems such as bone tissue, pancreatic cancer tissue and
colorectal cancer tissue (16,17), where
it participated in physiological functions, such as regulating bone
formation and the circadian rhythm. In addition, RORβ was
demonstrated to be involved in the development of numerous types of
cancer (15). To date, to the best of
our knowledge, there are no current previous studies reporting the
biological function of RORβ in GC. The present study was the first
to reveal that the expression levels of RORβ were significantly
downregulated in GC, suggesting that RORβ may serve as a tumor
suppressor. Through GSEA, the expression levels of stem cell
surface markers were discovered to be downregulated in tissues with
upregulated expression levels of RORβ compared with control
tissues, suggesting that RORβ may be a cell differentiation
inducer.
In the present study, RORβ inhibited GC cell
viability, as determined by a CCK-8 assay. GeneChip-scanning
experiments have previously reported that BCL2L11, a pro-apoptotic
protein, mainly functioned by inhibiting Bcl-2-induced apoptosis
and inactivating the expression levels of Bax and Bak (31). Therefore, it was hypothesized that
RORβ may promote the apoptosis of GC cells. The results of GO
analysis indicated that RORβ, as a transcription factor, may also
be involved in tumorigenesis, regulating the cell cycle, cell
proliferation and differentiation. In addition, the overexpression
of RORβ in GC cells decreased the GC cells stemness by
downregulating the expression levels of CSC surface markers,
inhibiting the sphere forming ability, decreasing the tumor growth
rate and downregulating the expression levels of EMT-related
factors. These findings strongly indicated that RORβ may regulate
the occurrence and development of GC by inhibiting the stemness of
CSCs.
Previous studies have reported that the Wnt
signaling pathway is an important regulatory pathway in GCSCs
(31,32). Tan et al (33) revealed that CSCs in the
AQP5+ tissues promoted GC in vivo by activating
the Wnt signaling pathway in newly generated AQP5-creERT2 model
mice. In the present study, RORβ downregulated the expression
levels of downstream molecules of the Wnt signaling pathway in
GCSCs, which suggested that RORβ regulated the stemness of GCSCs by
downregulating the activity of the Wnt signaling pathway.
Therefore, RORβ, as a Wnt inhibitor, may represent a novel therapy
to reduce CSC activity in GC cases with upregulated expression
levels of RORβ.
In conclusion, the findings of the present study
revealed that RORβ significantly inhibited the stemness properties
of GC cells by downregulating the Wnt signaling pathway. Therefore,
RORβ may represent a potential novel antitumor agent for the
treatment of GC. However, the downstream target molecule of RORβ as
an antitumor agent for the treatment of GC needs to be further
clarified in the future.
Acknowledgements
Not applicable.
Funding
The present study was financially supported by the
Zhejiang Provincial Medical and Health science Foundation (grant
nos. 2021441200, 2015KYB020 and 2020380312), the National Science
of Foundation Committee of China (grant no. 81602586) and the
Zhejiang Provincial Traditional Chinese Medicine Science Research
Foundation (grant no. 2015ZA010).
Availability of data and materials
All datasets generated and analyzed during the
present study are available from the corresponding author on
reasonable request.
Authors' contributions
ZW, MC and WG conceived and designed the study. PD,
KG and YF performed the statistical analysis. MG and QW performed
the experiments. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
All animal experiments were approved by the
Institutional Animal Care and Use Committee of Zhejiang University
(Hangzhou, China) and complied with the Animal Welfare Act to
euthanize all animals in the experiment. GC tissues were collected
at the Sir Run Run Shaw Hospital of the Zhejiang University
following ethical approval from the Institutional Review Board from
Sir Run Run Shaw Hospital (Hangzhou, China). All patients provided
written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics. CA Cancer
J Clin. 65:87–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mariette C, Renaud F, Piessen G, Gele P,
Copin MC, Leteurtre E, Delaeter C, Dib M, Clisant S, Harter V, et
al: The FREGAT biobank: A Clinico-biological database dedicated to
esophageal and gastric cancers. BMC Cancer. 18:1392018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Pan Y, Ma S, Cao K, Zhou S, Zhao A, Li M,
Qian F and Zhu C: Therapeutic approaches targeting cancer stem
cells. J Cancer Res Ther. 14:1469–1475. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Taniguchi H, Moriya C, Igarashi H, Saitoh
A, Yamamoto H, Adachi Y and Imai K: Cancer stem cells in human
gastrointestinal cancer. Cancer Sci. 107:1556–1562. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guo Y, Feng K, Wang Y and Han W: Targeting
cancer stem cells by using chimeric antigen receptor-modified T
cells: A potential and curable approach for cancer treatment.
Protein Cell. 9:516–526. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang Y, Wu KE, Zhao E, Li W, Shi L, Xie G,
Jiang B, Wang Y, Li R, Zhang P, et al: B7-H1 enhances proliferation
ability of gastric cancer stem-like cells as a receptor. Oncol
Lett. 9:1833–1838. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang YC, Wang SW, Hung HY, Chang CC, Wu
IC, Huang YL, Lin TM, Tsai JL, Chen A, Kuo FC, et al: Isolation and
characterization of human gastric cell lines with stem cell
phenotypes. J Gastroenterol Hepatol. 22:1460–1468. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bekaii-Saab T and El-Rayes B: Identifying
and targeting cancer stem cells in the treatment of gastric cancer.
Cancer. 123:1303–1312. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nguyen PH, Giraud J, Chambonnier L, Dubus
P, Wittkop L, Belleannée G, Collet D, Soubeyran I, Evrard S,
Rousseau B, et al: Characterization of biomarkers of tumorigenic
and chemoresistant cancer stem cells in human gastric carcinoma.
Clin Cancer Res. 23:1586–1597. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gong X, Azhdarinia A, Ghosh SC, Xiong W,
An ZQ, Liu QY and Carmon KS: LGR5-targeted antibody-drug conjugate
eradicates gastrointestinal tumors and prevents recurrence. Mol
Cancer Ther. 15:1580–1590. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nguyen PH, Giraud J, Staedel C,
Chambonnier L, Dubus P, Chevret E, Bœuf H, Gauthereau X, Rousseau
B, Fevre M, et al: All-trans retinoic acid targets gastric cancer
stem cells and inhibits patient-derived gastric carcinoma tumor
growth. Oncogene. 35:5619–5628. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fu Y, Du PZ, Zhao J, Hu CE, Qin YY and
Huang GJ: Gastric Cancer Stem cells: Mechanisms and therapeutic
approaches. Yonsei Med J. 59:1150–1158. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhan T, Rindtorff N and Boutros M: Wnt
signaling in cancer. Oncogene. 36:1461–1473. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Carlberg C, Hooft van Huijsduijnen R,
Staple JK, DeLamarter JF and Becker-André M: RZRs, a new family of
retinoid-related orphan receptors that function as both monomers
and homodimers. Mol Endocrinol. 8:757–770. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Feng SJ, Xu S, Wen ZZ and Zhu YL: Retinoic
acid-related orphan receptor RORβ, circadian rhythm abnormalities
and tumorigenesis (Review). Int J Mol Med. 35:1493–1500. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Mühlbauer E, Bazwinsky-Wutschke I, Wolgast
S, Labucay K and Peschke E: Differential and day-time dependent
expression of nuclear receptors RORα, RORβ, RORγ and RXRα in the
rodent pancreas and islet. Mol Cell Endocrinol. 365:129–138. 2013.
View Article : Google Scholar
|
|
17
|
Wen ZZ, Pan TH, Yang SS, Liu JW, Tao HY,
Zhao YM, Xu DT, Shao W, Wu J, Liu XY, et al: Up-regulated NRIP2 in
colorectal cancer initiating cells modulates the Wnt pathway by
targeting RORβ. Mol Cancer. 16:202017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Risinger JI, Allard J, Chandran U, Day R,
Chandramouli GVR, Miller C, Zahn C, Oliver J, Litzi T, Marcus C, et
al: Gene expression analysis of early stage endometrial cancers
reveals unique transcripts associated with grade and histology but
not depth of invasion. Front Oncol. 3:1392013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Edge SB and Compton CC: The American Joint
Committee on Cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mulcahy DM: The animal welfare act and the
conduct and publishing of Wildlife research in the United States.
ILAR J. 58:371–378. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang da W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Subramanian A, Tamayo P, Mootha VK,
Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub
TR, Lander ES and Mesirov JP: Gene set enrichment analysis: A
knowledge-based approach for interpreting genome-wide expression
profiles. Proc Natl Acad Sci USA. 102:15545–15550. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
André E, Gawlas K and Becker-André M: A
novel isoform of the orphan nuclear receptor RORbeta is
specifically expressed in pineal gland and retina. Gene.
216:277–283. 1998. View Article : Google Scholar
|
|
25
|
Tiffon C, Giraud J, Molina-Castro SE, Peru
S, Seeneevassen L, Sifré E, Staedel C, Bessède E, Dubus P, Mégraud
F, et al: TAZ Controls Helicobacter pylori-induced
epithelial-mesenchymal transition and cancer stem cell-like
invasive and tumorigenic properties. Cells. 9:14622020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang M, Jin M, Li K, Liu H, Yang X, Zhang
X, Zhang B, Gong A and Bie Q: TRAF6 promotes gastric cancer cell
self-renewal, proliferation, and migration. Stem Cells Int.
2020:32961922020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Azimi M, Totonchi M, Rahimi M, Firouzi J,
Sahranavard P, Emami Razavi A, Memari F, Kamali F and Ebrahimi M:
An integrated analysis to predict micro-RNAs targeting both
stemness and metastasis in human gastric cancer. J Gastroenterol
Hepatol. 36:436–445. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Slusarski DC, Corces VG and Moon RT:
Interaction of Wnt and a Frizzled homologue triggers
G-protein-linked phosphatidylinositol signalling. Nature.
390:410–413. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nguyen LV, Vanner R, Dirks P and Eaves CJ:
Cancer Stem cells: An evolving concept. Nat Rev Cancer. 12:133–143.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Schulenburg A, Blatt K, Cerny-Reiterer S,
Sadovnik I, Herrmann H, Marian B, Grunt TW, Zielinski CC and Valent
P: Cancer stem cells in basic science and in translational
oncology: Can we translate into clinical application? J Hematol
Oncol. 8:162015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Luo SQ and Rubinsztein DC: BCL2L11/BIM: A
novel molecular link between autophagy and apoptosis. Autophagy.
9:104–105. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mao J, Fan S, Ma W, Fan P, Wang B, Zhang
J, Wang H, Tang B, Zhang Q, Yu X, et al: Roles of Wnt/β-catenin
signaling in the gastric cancer stem cells proliferation and
salinomycin treatment. Cell Death Dis. 5:e10392014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tan SH, Swathi Y, Shawna Tan S, Goh J,
Seishima R, Murakami K, Oshima M, Tsuji T, Phuah P, Tan LT, et al:
AQP5 enriches for stem cells and cancer origins in the distal
stomach. Nature. 578:437–443. 2020. View Article : Google Scholar : PubMed/NCBI
|