Introduction
Renal clear cell carcinoma (RCCC) commonly occurs in
patients with VHL disease (1).
Tumorigenesis occurs secondary to germline VHL gene mutation
and inactivation of the VHL wild-type allele in the cancer cell of
origin. Biology and topography of the cell of origin have remained
controversial (2). Following
original histochemical (3), and
immunofluorescent (4,5) studies, the main approach for
determination of the RCCC cell of origin has been the
immunohistochemical characterization of the neoplastic clear cells
with a series of markers specific for proximal or distal tubules,
or collecting ducts. Several such studies were conducted with
remarkably different results. Certain studies firmly concluded the
clear cancer cells to be derived from proximal tubules (3–8),
while other studies identified the origin of neoplastic clear cells
in the distal tubular system (9–11).
Others found evidence for both proximal and distal tubular
differentiation and hypothesized that the RCCC cell of origin may
exhibit different degrees of differentiation (12–14);
this hypothesis implied that immunohistochemical study may be more
appropriate for study of cancer cell differentiation potential
rather than for study of cancer cell origin.
A new approach was applied by Mandriota et al
(15) who studied microscopic
nests of clear cell accumulations in kidney tissue of VHL patients.
By immunohistochemical analysis for carbonic anhydrase IX (CAIX),
the authors were able to verify VHL inactivation in the clear
cells, while immunohistochemistry for Tamm-Horsfall distal tubule
protein (THP) identified associated distal tubular cells.
Importantly, the authors concluded that groups of clear cells were
exclusively observed in the distal tubular system, and not present
in the proximal tubular system suggesting proliferative activity to
be confined to the distal tubular system. Although these seminal
findings were published in 2002, authoritative literature has been
holding on to the concept of proximal tubular origin (16–20).
Main arguments for proximal tubular origin continue to be shared
positive immunoreactivity of cancer cells with markers for proximal
tubules (17,20), or shared gene expression of cancer
cells with proximal tubules (19).
Certain degree of confusion has, however, been appreciated
(17), as RCCCs may express or
co-express a variety of markers of different tubular segments
including the distal tubular system (17).
Histologic features were recently captured in entire
tissue blocks of normal kidney tissue of VHL patients; quantitative
numerical analysis of independent events of VHL-inactivation
provided strong evidence that groups of VHL-inactivated cells in
the human nephron are the result of cellular amplification
(21). Our collection of
H&E-stained sections at 50 µm intervals has allowed us to
re-examine the question of origin of clear cell proliferation by a
different and independent approach. First, authors are fully
informed about the three-dimensional extension of every single
histopathologic structure under study, and it could be claimed with
absolute certainty that clear cell proliferations do not represent
extensions of larger pathologic processes invisible on the original
section. Secondly, H&E-stained sections for structures of
interest were primarily analyzed, followed by investigation of
CAIX+ structures. Thirdly, immunohistochemistry for cluster of
differentiation 10 (CD10) and cytokeratin 7 (CK7) was performed to
identify the tubular cells associated with clear cells
proliferation.
An algorithm is a finite sequence of instructions to
solve a problem (21), describes a
series of rigorous steps that were taken to obtain suitable tissue
blocks for analysis from VHL kidneys. For each case, the same
protocol was applied: Transforming the kidney into cuboids, cuboid
selection, creation of blocks, block selection, block of interest
selection, serial sectioning, followed by additional steps to allow
for 3d assessment.
Beyond analysis of pre-existent tubular cells, clear
cell proliferation events for CD10 and CK7 were also investigated
to study the faithfulness with which the clear cell immunophenotype
is reflective of its tubular origin. In addition, results were
associated with a recent study that identified distinct expression
patterns of HIF-1 alpha and HIF-2 alpha in human kidneys (22).
Materials and methods
Patients
The present study was reviewed and approved
(approval no. 609/2019) by the ethical committee of the Deanship of
Research at Jordan University of Science and Technology. The VHL
patients had died between the ages of 50 and 65 years (average 58.5
year). Tissues were collected during autopsy of four patients with
confirmed germline mutations of the VHL gene and established
clinical diagnosis of VHL disease at the National Institutes of
Health between January 2000 and December 2006 (designated as ‘VHL
kidneys’). Three patients were male and one female. Causes of death
were pneumonia in two cases, metastatic renal cell carcinoma, and
intracerebral hemorrhage. All patients had other VHL
disease-associated tumors including hemangioblastomas, epididymal
cystadenomas, endolymphatic sac tumors and microcystic adenomas of
the pancreas. Clinical data of patients are presented in Table I.
 | Table I.Clinical data of patients. |
Table I.
Clinical data of patients.
|
| Age, years/sex | VHL-related
pathology | Surgeries | Cause of death |
|---|
| Case 1 | 60/male | HBs cerebellum and
spinal cord | HB resection
cerebellum | Intracerebral
hemorrhage |
| Case 2 | 65/male | 3 cerebellar HBs,
renal cysts and RCCC, ECA | 1992 resection
cerebellar HB, Radiotherapy for brainstem HB | Hemorrhagic
pneumonia |
| Case 3 | 59/female | HBs cerebellum and
spinal cord, RCCC and cysts | 1990 spinal cord
HB | Cerebellar
hemorrhage |
|
|
|
| 1990 partial
nephrectomy |
|
|
|
|
| 1990 cerebellar
HB |
|
|
|
|
| 1992 spinal HB
resection |
|
|
|
|
| 1998 right hip
arthroplasty |
|
| Case 4 | 50/male | 2 HBs cerebellum,
multiple HBs spinal cord, RCC and cysts both kidneys, MCA pancreas,
ECA | Partial nephrectomy,
HB resection cerebellum | Sepsis, wide-spread
metastatic renal cell carcinoma |
All patients had provided informed consent to be
treated for VHL disease at NIH. This informed consent included
autopsy to be performed. Certain of the patients demanded autopsy
investigation for the purpose of improved understanding of this
disease. Dissection was performed under IRB 03N-0164.
Preliminary studies
Preliminary studies were performed to identify
reliable markers for the proximal and distal tubular systems on
unstained sections that were cut previously for 3D reconstruction
of paraffin blocks (21). Random
kidney sections were immunostained for CD138, CD10, epithelial
membrane antigen (EMA), E-cadherin (cat. no. GAO59; Dako; Agilent
Technologies, Inc.), CK AE1/AE3 and CK7. All antibodies were in a
‘ready to use’ formal. For CK AE1/AE3 and CK7, no secondary
antibody was used. For CD10 and CD138, EnVision-flex+ mouse link
was used (ready to use; cat. no. K800221-2; Agilent Technologies,
Inc.; 20 min incubation at room temperature). After
de-paraffinizing slides with 5-µm sections in xylene, for those
antibodies AE1/AE3 (cat. no. IR053), CD10 (cat. no. IR648), CD138
(cat. no. IR642), EMA (cat. no. IR629; all from Dako; Agilent
Technologies, Inc.) and CAIX (cat. no. 379R-18; Cell Marque) all
underwent antigen retrieval in a high pH buffer in the Dako PT
module (for 20 min at 99-degree Fahrenheit) followed by 20 min
incubation with the primary antibody at room temperature. Those for
CK7 (cat. no. IR619; Dako; Agilent Technologies, Inc.) and
E-cadherin were retrieved in low pH buffer in the PT module with 10
min primary antibody incubations. They were all visualized using
the Dako Flex-hrp system and developed with DAB. Staining results
were evaluated and best quality of staining was found for CD10 and
CK7. Both antibodies reliably and specifically identified the
proximal tubular system (CD10) and the distal tubular system (CK7)
(23–25). Selected sections were also
immunostained for Glut1 and vimentin. Tissue sections were first
treated with hydrogen peroxide blocking agent, followed by antigen
retrieval; low pH was applied for Glut1, Lab Vision RB-9052; high
pH was used for vimentin, DAKo/Agilent IR630. They were then
incubated with the primary antibody for 10 min at room temperature.
For Glut1 there was an incubation with a linker Flex+R, then both
were subjected to the detecting agent Flex-hrp (Agilent Flex kit)
for 10 min followed by the color development using DAB for 10
min.
All immunohistochemical studies were performed with
proper external controls additionally, internal controls stained
through the sections. All of controls were human samples of patient
cases.
Topographic origin of clear cell
proliferation
An algorithm for tissue procurement, processing and
3d analysis of tissue blocks of interest was recently described
(21). Having applied that
approach, access was gained to seven serially sectioned VHL kidney
blocks from 4 different patients as well as 3 serially sectioned
control blocks (kidneys of patients with sporadic RCCC). As
previously described, H&E-stained sections have been digitized
at 50 µ intervals (21).
For the present study, histologic review of
H&E-stained slides from 7 VHL kidney blocks (total of 140
slides) was performed, interrogating for structures of interest.
All H&E-stained slides were scanned (20–30 per case, taken at
depths of 50 µm) and 11 hybrid structures were identified. By
immunohistochemical analysis of consecutive sections, they were all
positive for CAIX. Structures of interest were defined as clear
cell proliferations that occurred in continuity with normal tubular
epithelium. ‘Hybrid’ structures of interest were defined as tubular
structures that contained multiple renal tubular cells as well as a
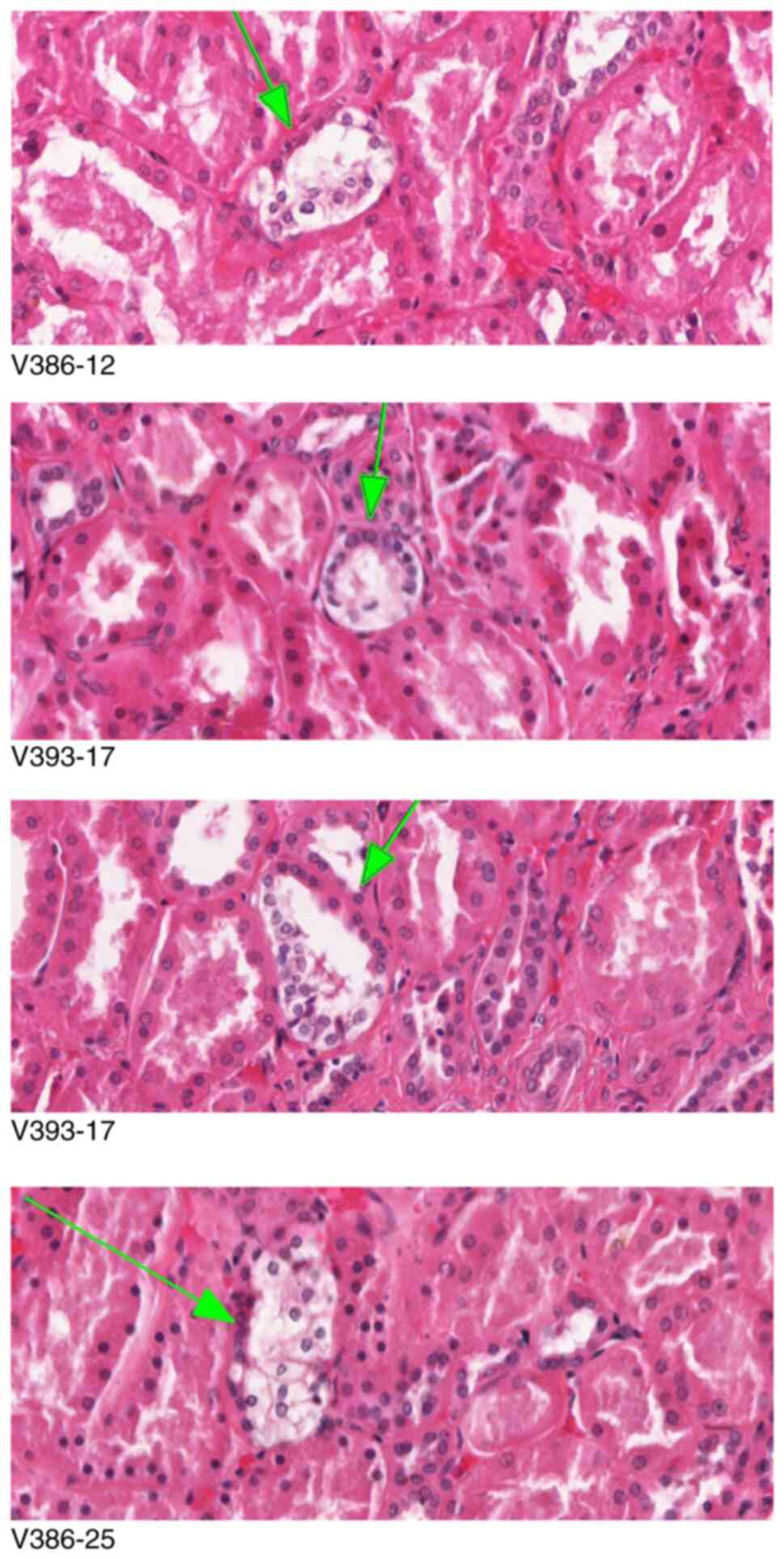
chain of multiple clear cells (Fig.
1). In those ‘hybrid’ structures of interest as defined above,
it was generally difficult to identify the tubular segment of
origin by histomorphology alone as there appeared to be distorting
effect on the tubular epithelium secondary to the clear cell
proliferation. In consecutive sections, immunohistochemistry was
performed for CK7 and CD10 for definitive identification of the
tubular segment. In addition, immunohistochemistry was performed
for CAIX for confirmation of VHL deficiency in the clear cell
proliferation event.
After identification of the topographic origin of
all ‘hybrid’ structures of interest discovered on H&E-stained
sections, all immunohistochemical preparations for CAIX were
re-examined, identifying 81 additional CAIX+ and three
CAIX-negative clear cell chains and clusters that were also
represented in consecutive sections, immunostained for CK7 and
CD10. The consecutive sections were analyzed for the tubular cells
associated with the CAIX+ clear cells.
Immunohistochemical characterization
of clear cell proliferations
Re-examination was performed of all 92 CAIX+ and
three CAIX-negative clear cell chains and clusters including
consecutive slides immunostained for CD10 and CK7. The purpose of
this analysis was to examine whether clear cells would be
immunoreactive for CD10 or CK7.
Results
Identification of topographic origin
of clear cell proliferation
A total of 11 ‘hybrid’ structures of interest (clear
cell proliferation adjacent to a tubular cell segment) were
identified on H&E-stained sections by light microscopy and
characterized by immunohistochemistry on consecutive sections
(Fig. 1). These ‘hybrid’
structures were identified in two out of three VHL kidneys. They
were identified in three different blocks on 4 different slides.
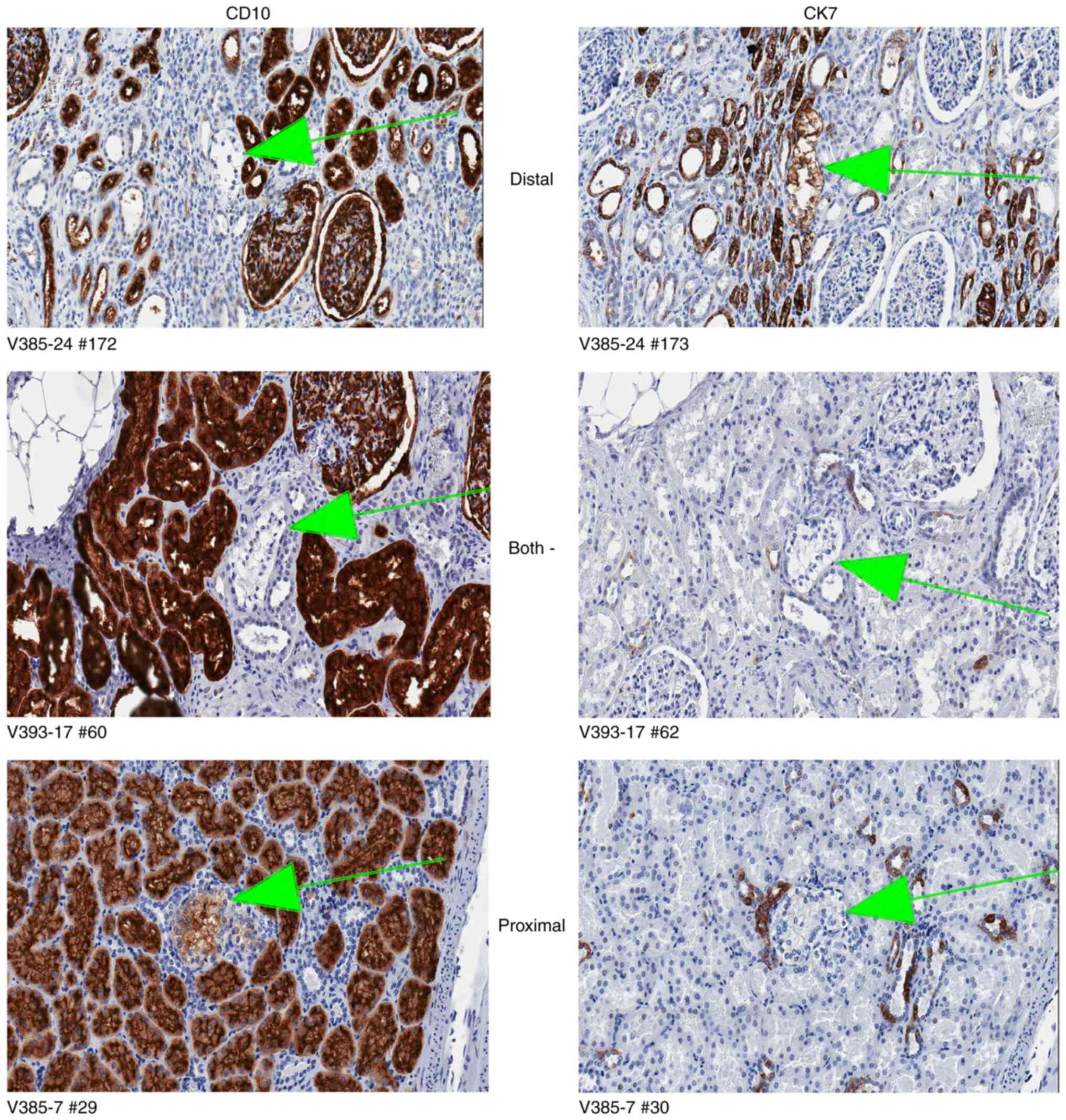
The renal tubular cells that were continuous with the clear cell
proliferation were consistently positive for CK7 and consistently
negative for CD10 (Fig. 2). The
clear cells were consistently positive for CAIX.
Additionally, identified in the CAIX-immunostained
sections were 81 CAIX+ clear cell chains or clusters. In total, 18
of these clear cell proliferations were identified in two out of
three VHL kidneys. They were identified in four different blocks on
5 different slides (Table II).
The renal tubular cells were consistently positive for CK7 and
consistently negative for CD10.
 | Table II.A total of 18 ‘hybrid’ structures of
interest discovered on carbonic anhydrase IX+ -stained sections in
two patients of four different blocks on five different slides. |
Table II.
A total of 18 ‘hybrid’ structures of
interest discovered on carbonic anhydrase IX+ -stained sections in
two patients of four different blocks on five different slides.
| Patient 1 | Patient 2 |
|---|
|
|
|---|
| Block 12 | Block 25 | Block 26 | Block 17 | Block 17 |
|---|
| Slide 150 | Slide 29 | Slide 26 | Slide 62 | Slide 205 |
| 2 | 5 | 2 | 7 | 2 |
Additionally, identified in the CAIX-immunostained
sections were three CAIX-negative clear cell chains or clusters
(Fig. 3), as previously identified
and associated with de-repression of HIF-2alpha (22). In contrast to those previously
described Schietke type II lesions, however, the CAIX-negative
clusters/chains in the present study showed only faint or no
immunoreactivity with anti-Glut1 or anti-vimentin (Fig. 3). Similar to Schietke type II
lesions, they were immunonegative for E-cadherin.
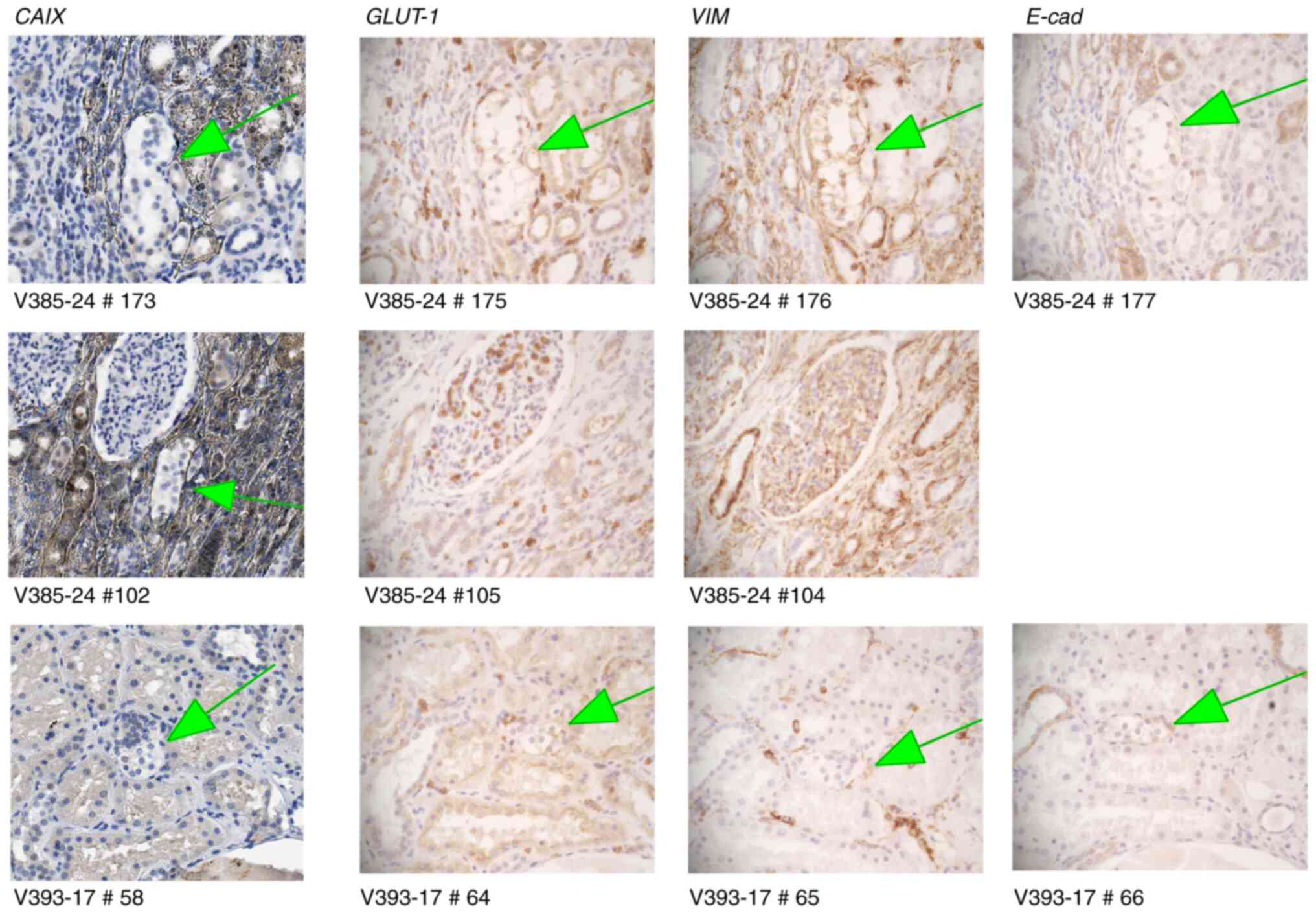 | Figure 3.CAIX-negative clear cell
proliferations. Upper row demonstrated weak immunoreactivity with
anti-Glut1 (magnification, ×200) and anti-vimentin (magnification,
×200) and negative reactivity with anti-E-cadherin (20X). Middle
row demonstrated CAIX-negative clear cell cluster that has
disappeared on consecutive sections, and therefore cannot be
further characterized (magnification, ×200). Lower row shows clear
cell cluster, negative for CAIX (magnification, ×200), Glut1
(magnification, ×200), vimentin (magnification, ×200) and
E-cadherin (magnification, ×200). CAIX, carbonic anhydrase IX. |
In total, 29 distal tubules containing clear cell
proliferation were therefore identified, all with CAIX+. A proximal
tubule containing clear cell proliferation was not detected,
suggesting that clear cell proliferation uniformly occurred in the
distal tubular system.
The ninety-five clear cell proliferation events
stained variably for either CD10 or CK7. A total of 39 events were
negative for both CK7 and CD10 (41.0%), 4 events were positive for
CD10 (4.3%), and 52 events were positive for CK7 (54.7%) (Fig. 4). No event was positive for both
CD10 and CK7 (Table III). Of the
3 CAIX-negative proliferation events, two were strongly positive
for CK7, and one was weakly positive for CK7; none was positive for
CD10.
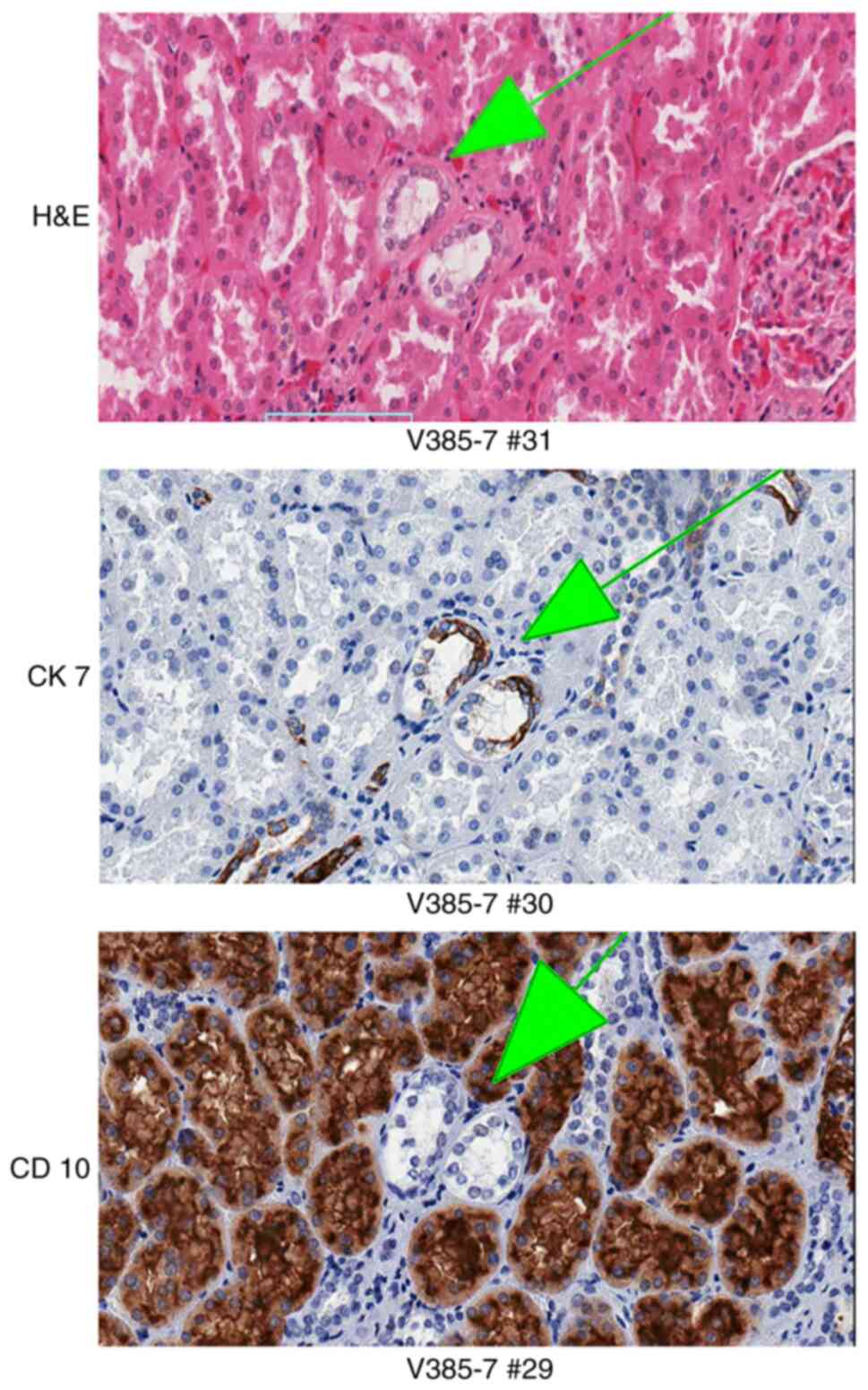 | Figure 4.Clear cell proliferation events are
variably positive for CK7 and CD10. The designation at the bottom
of each picture identifies the tissue block and the number of the
serial section. Upper panel, clear cell proliferation, positive for
CK7 (magnification, ×200) and negative for CD10 (magnification,
×200) by immunohistochemistry (arrows). Middle panel, clear cell
proliferation, negative for both CK7 (magnification, ×200) and CD10
(magnification, ×200) (arrows). Bottom, clear cell proliferation,
positive for CD10 (magnification, ×200) and negative for CK7
(magnification, ×200) (arrows). CK7, cytokeratin 7; CD10, cluster
of differentiation 10. |
 | Table III.Total number of lesions were analyzed
according to H&E and immunohistochemical staining. |
Table III.
Total number of lesions were analyzed
according to H&E and immunohistochemical staining.
| CAIX-stained
lesions | Hybrid structure of
interest | Clear cell
immunophenotype |
|---|
|
|
|
|---|
| CAIX-positive | CAIX-negative |
H&E-stained | CAIX-stained | CK7-positive | CD10-positive | Both CK7/CD10
negative | Both CK7/CD10
positive |
|---|
| 92 | 3 | 11 | 18 | 52 | 4 | 39 | 0 |
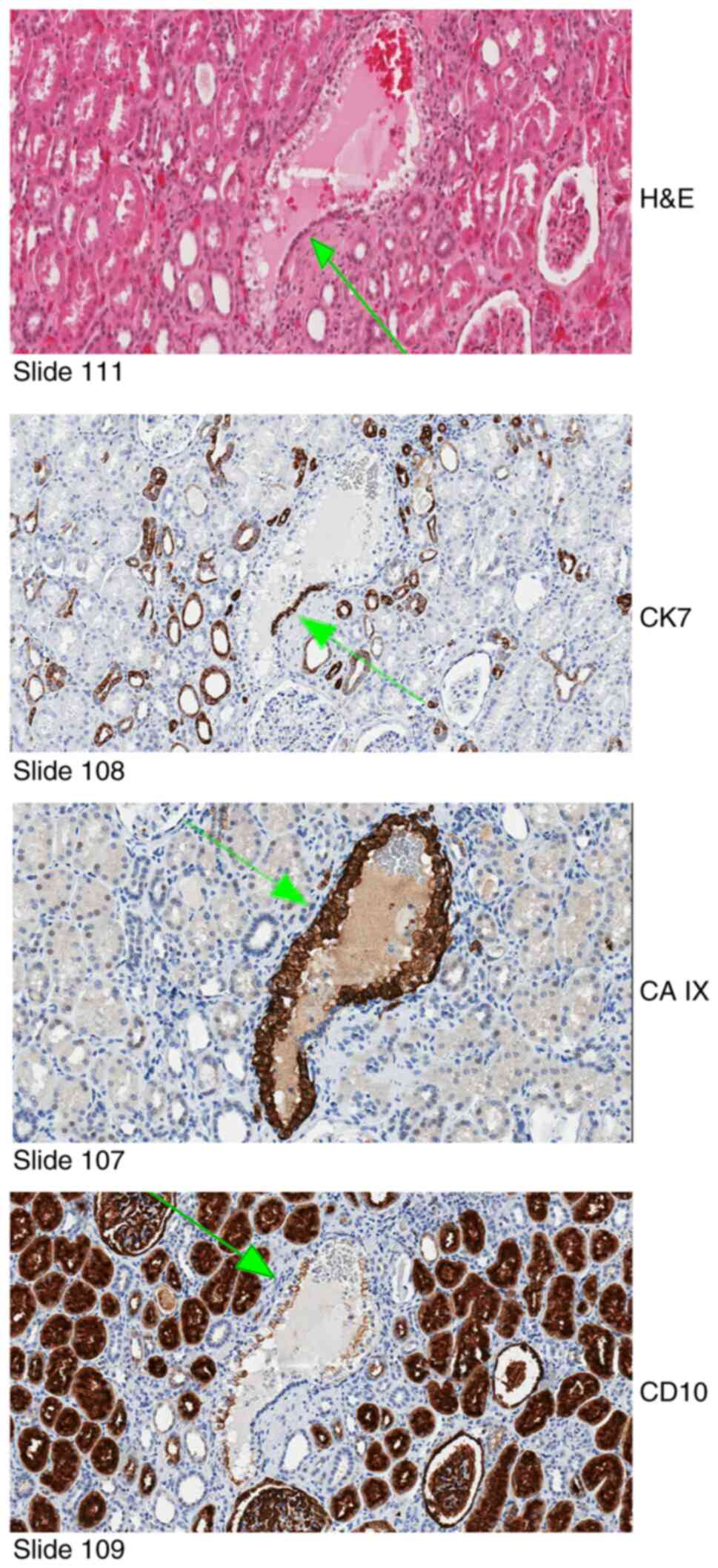
Additionally, a single pathologic event in the VHL
kidney was identified on H&E-stained slide #111 (this lesion
has multiple adherent clear cells that replace about 80% of the
tubular circumference and 20% are normal tubular cells), then three
consecutive serial sections were used for immunohistochemical
studies (Fig. 5).
Immunohistochemistry for CK7 (slide #108) identified the normal
tubular cells as distal tubular cells. Immunohistochemical analysis
for CAIX (slide #107) confirmed VHL deficiency of the clear cell
proliferation. Immunohistochemical analysis for CD10 (slide #109)
was positive in the clear cell proliferation, suggestive of
proximal tubular differentiation in VHL-deficient clear cells.
Discussion
It is challenging to histologically study minute
cellular proliferation with standard histologic techniques. An
obvious limitation of routine histology is that a section
represents only a very small amount of tissue, while most of that
tissue remains in the tissue block. Once a minute-sized cellular
proliferation event is identified in a tissue section, it cannot be
further characterized in consecutive sections as the structure of
interest is lost when additional sections are performed from that
block.
To allow for the creation of serial sections of
minute pathologic events, an algorithm was recently developed for
3-dimensional histologic analysis (21). This approach is rewarding in
tissues that contain a multiplicity of independent pathologic
events, as it has been shown in kidney tissue of VHL kidney.
Importantly, this approach not only allows for more detailed
characterization of small pathologic events, but also for their
quantification.
The limitations of the present study include the low
number of cases examined; however, VHL is a rare syndrome with a
reported incidence varying from 1/39,000 to 1/91,000 (26). Our way around the low number of
cases was to examine as numerous sections as possible from the
tissue blocks obtained by performing multiple level sections,
analyzing them in consecutive order, tracking all clear cell
changes, and correlating the findings to immunohistochemical
findings. All immunohistochemical studies were performed with
proper external controls-additionally, internal controls stained
through the sections. Immunogenicity can be lost over time (given
that the tissue was collected years ago). This could be a
theoretical explanation for weak Glut1 and vimentin staining in our
Schietke type II lesions; however, all our immunostained sections
displayed effective internal control. Therefore, there may be more
than just two types of early pathogenetic lesions in the VHL
kidney, and further investigation is warranted.
An additional limitation is the lack of statistical
and morphometric analysis to determine the size distribution of
lesions and their classification as arising from proximal or distal
tubes. Still, it is strongly considered that morphometric analysis
is extremely relevant for the study of the development of
clustering clear cells. In the present study, however, structures
composed of pre-existent tubular cells and a rim of proliferating
clear cells were analyzed. In our view, morphometric analysis is
only relevant at a later stage of clear cell proliferation. Our
current knowledge, however, is limited regarding the evidence for
differentiation into benign and malignant processes at far earlier
microscopic stages (15,22). More work on the earliest
proliferative changes in the VHL kidney is required.
VHL kidneys reveal an abundance of independent
pathologic events, predominantly characterized by intratubular
proliferation of VHL-deficient clear cells (21). As there is evidence for a
protracted growth from single VHL-deficient clear cells to larger
clear cell clusters, it is reasonable to hypothesize that they
represent potential cancer precursor structures (21).
While single cell clear cell changes or ‘small
chains’ were virtually innumerable, there are exact counts of large
chains and cell clusters (21). To
address our specific question of tubule of origin, the pathologic
event needed to fulfill additional criteria. As
immunohistochemistry was performed on serial sections, the lesion
needed to reveal multiple normal tubular cells adherent to multiple
clear cells.
Multicellular aggregations of VHL-inactivated clear
cells were previously identified in the distal tubular system only
(15). Using an independent
approach, clear cell proliferation was also found to occur
exclusively in the distal tubular system. Beyond the previous
study, all analyzed microscopic structures were proven to be
confined to the tubular system, as their extensions were documented
in three dimensions.
As transcription factor HIF if currently under
evaluation as a putative therapeutic target, differentiation
between CAIX+ lesions (HIF1 alpha+; Schietke type I) and
CAIX-negative lesions (HIF2 alpha+; Schietke type II) is of
additional interest (22). In the
present study, CAIX-immunostained slides numerically revealed far
more type I than type II lesions. Large ‘hybrid’ structures,
originally identified in H&E-stained sections consistently
showed CAIX+ clear cell proliferations. Upon examination of the
entire slide, however, smaller CAIX-negative clear cell
proliferations were also detected. By immunohistochemical analysis
for CK7 and CD10, the CAIX-negative and CAIX+ clear cell
proliferations were not distinct from each other. The relatively
low number of CAIX-negative proliferation events compared with
CAIX+ proliferation events in the present study may be a random
event, as the purpose of this study was not quantitative
differentiation between type I and type II lesions. Intuitively, a
predominance of type I lesions would be consistent with benign
cystic growth and only few type II lesions with more aggressive
potential. Our current knowledge, however, is extremely limited.
While benign cystic growth may transform into malignancy (27) in the VHL kidney, evidence for
differentiation into benign and malignant processes at far earlier
microscopic stages has been provided (15,22).
Further research on earliest proliferative changes in the VHL
kidney is required.
Furthermore, first evidence was provided that
immunohistochemical reactivity of early proliferating clear cells
can markedly differ from that of the tubule of origin, and
therefore may not allow to predict the origin of the clear cells. A
total of 52/95 clear cell proliferations (54.7%) were positive for
CK7, and therefore congruent with their site of origin. By
contrast, 39/95 clear cell proliferations (41.0%) were negative for
both CK7 and CD10. Most remarkably, however, 4/95 clear cell
proliferations (4.3%) were positive for proximal marker CD10,
although the clear cell proliferation occurred in the distal
tubular system. The results therefore demonstrated that the
immunophenotype of VHL-deficient clear cells in the kidney does not
necessarily reflect their site of origin. While more investigation
is necessary, this result is consistent with the clear cell to
represent an intratubular progenitor cell the immunophenotype of
which may relate to its state of differentiation. VHL deficiency of
the clear cell and upregulation of hypoxia-associated proteins may
further contribute to aberrant immunophenotype. Multiple
immunohistochemical studies have produced highly variable results
in regard to shared immunoreactivity of RCCC clear cells with the
proximal and distal renal tubular system (17). The present study demonstrated that
variability of immunohistochemical reaction is already evident in
earliest stages of clear cell proliferation.
In summary, our results confirmed and expanded
previous findings that proliferation of VHL-inactivated clear cells
is confined to the distal renal tubules. Earliest clear cell
proliferations reveal a diverse immunophenotype, likely in analogy
to frank renal cancer.
Acknowledgements
The authors would like to acknowledge the Sandusky
laboratory (Indiana University School of Medicine) for digitization
of slides; Dr Carrie L. Philips (Indiana University School of
Medicine, Department of Pathology) for providing information on
markers and reviewing selected images; Miss Lee-Ann Baldridge
(Indiana University School of Medicine, Histology Research
Technician, Department of Pathology) for assistance with additional
IHC stains and Mr Haitham Al-Gharaibeh (Internal Design Studio,
Amman-Jordan) for assistance with image documentation.
Funding
The present study was partially supported by the Deanship of
Research of Jordan University of Science of Technology (grant no.
609/2019), and by the Department of Pathology, University of
Indiana Medical School.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CJT performed immunohistochemical staining and
participated in writing parts of the manuscript. SBS assisted with
additional immunohistochemistry and drafting the new manuscript.
NSA-G participated in study execution, data interpretation and
manuscript writing. AOV participated in study design, study
execution, data interpretation and manuscript writing. All authors
read and approved the final version of the manuscript.
Ethics approval and consent to
participate
The present study was reviewed and approved
(approval no. 609/2019) by the ethical committee of the Deanship of
Research at Jordan University of Science and Technology. The VHL
tissues were derived from autopsy patients. Autopsies were
performed between 2001 and 2005. All tissues of interest were
submitted to the Surgical Neurology Branch Tumor Bank Repository.
The Tumor Bank contains specimens routinely procured at the time of
surgery or autopsy under IRB Protocol 03N-0164. Written informed
consent was provided by all patients for being treated for VHL
disease at NIH. This informed consent included autopsy. Dissection
was performed under IRB 03N-0164.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
VHL kidney
|
kidney of von Hippel-Lindau disease
patient
|
|
RCCC
|
renal clear cell carcinoma
|
|
EMA
|
epithelial membrane antigen
|
|
THP
|
Tamm-Horsfall distal tubule
protein
|
|
CAIX
|
carbonic anhydrase IX
|
|
H&E
|
hematoxylin and eosin
|
|
CK7
|
cytokeratin 7
|
|
CD10
|
cluster of differentiation 10
|
|
EMA
|
epithelial membrane antigen
|
|
Glut1
|
glucose transporter protein 1
|
|
HIF
|
hypoxia-inducible factor
|
References
|
1
|
Lonser RR, Glenn GM, Walther M, Chew EY,
Libutti SK, Linehan WM and Oldfield EH: Von Hippel-Lindau disease.
Lancet. 361:2059–2067. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Glasker S, Vergauwen E, Koch CA, Kutikov A
and Vortmeyer AO: Von Hippel-Lindau disease: Current challenges and
future propects. Onco Targets Ther. 13:5669–5690. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Braunstein H and Adelman JU: Histochemical
study of the enzymatic activity of human neoplasms. II.
Histogenesis of renal cell carcinoma. Cancer. 19:935–938. 1966.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Holthöfer H, Miettinen A, Paasivuo R,
Lehto VP, Linder E, Alfthan O and Virtanen I: Cellular origin and
differentiation of renal carcinomas. A fluorescence microscopic
study with kidney-specific antibodies, antiintermediate filament
antibodies, and lectins. Lab Invest. 49:317–326. 1983.PubMed/NCBI
|
|
5
|
Wallace AC and Nairn RC: Renal tubular
antigens in kidney tumors. Cancer. 29:977–981. 1972. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gröne HJ, Weber K, Helmchen U and Osborn
M: Villin-a marker of brush border differentiation and cellular
origin in human renal cell carcinoma. Am J Pathol. 124:294–302.
1986.PubMed/NCBI
|
|
7
|
Kageyama Y, Sasaki S, Yamamura Y, Oshima H
and Ikawa Y: Water channel protein subtype suggests the origin of
renal cell carcinoma. J Urol. 156:291–295. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Motzer RJ, Bander NH and Nanus DM:
Renal-cell carcinoma. N Engl J Med. 335:865–875. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Aizawa S, Kikuchi Y, Suzuki M and Furusato
M: Renal cell carcinoma of lower nephron origin. Acta Pathol Jpn.
37:567–574. 1987.PubMed/NCBI
|
|
10
|
Cao Y, Karsten U, Zerban H and Bannasch P:
Expression of MUC1, Thomsen-Friedenreich-related antigens, and
cytokeratin 19 in human renal cell carcinomas and tubular clear
cell lesions. Virchows Arch. 436:119–126. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nogueira E, Klimek F, Weber E and Bannasch
P: Collecting duct origin of rat renal clear cell tumors. Virchows
Arch B Cell Pathol Incl Mol Pathol. 57:275–283. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cohen C, McCue PA and Derose PB:
Histogenesis of renal cell carcinoma and renal oncocytoma. An
immunohistochemical study. Cancer. 62:1946–1951. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Droz D, Zachar D, Charbit L, Gogusev J,
Chrétein Y and Iris L: Expression of the human nephron
differentiation molecules in renal cell carcinomas. Am J Pathol.
137:895–905. 1990.PubMed/NCBI
|
|
14
|
Markovic-Lipkovski J, Brasanac D,
Todorovic V, Müller CA and Müller GA: Immunomorphological
characteristics of renal cell carcinoma. Histol Histopathol.
10:651–659. 1995.PubMed/NCBI
|
|
15
|
Mandriota SJ, Turner KJ, Davies DR, Murray
PG, Morgan NV, Sowter HM, Wykoff CC, Maher ER, Harris AL, Ratcliffe
PJ and Maxwell PH: HIF activation identifies early lesions in VHL
kidneys: Evidence for site-specific tumor suppressor function in
the nephron. Cancer Cell. 1:459–468. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bobulescu IA, Pop LM, Mani C, Turner K,
Rivera C, Khatoon S, Kairamkonda S, Hannan R and Palle K: Renal
lipid metabolism abnormalities in obesity and clear cell renal cell
carcinoma. Metabolites. 11:6082021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Frew IJ and Moch H: A clearer view of the
molecular complexity of clear cell renal cell carcinoma. Annu Rev
Pathol. 10:263–289. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Turajlic S, Larkin J and Swanton C:
SnapShot: Renal cell carcinoma. Cell. 163:1556. e12015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Büttner F, Winter S, Rausch S, Reustle A,
Kruck S, Junker K, Stenzl A, Agaimy A, Hartmann A, Bedke J, et al:
Survival prediction of clear cell renal cell carcinoma based on
gene expression similarity to the proximal tubule of the nephron.
Eur Urol. 68:1016–1020. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sangoi AR, Karamchandani J, Kim J, Pai RK
and McKenney JK: The use of immunohistochemistry in the diagnosis
of metastatic clear cell renal cell carcinoma: A review of PAX-8,
PAX-2, hKIM-1, RCCma, and CD10. Adv Anat Pathol. 17:377–393. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mubarak M, Al-Gharaibeh N, Sommaruga S, Li
J and Vortmeyer AO: Histological tracking into the third dimension:
Evolution of early tumorigenesis in VHL kidney. J Kidney Cancer
VHL. 8:5–14. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Schietke RE, Hackenbeck T, Tran M, Günther
R, Klanke B, Warnecke CL, Knaup KX, Shukla D, Rosenberger C,
Koesters R, et al: Renal tubular HIF-2α expression requires VHL
inactivation and causes fibrosis and cysts. PLoS One. 7:e310342012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Holm-Nielsen P and Pallesen G: Expression
of segment-specific antigens in the human nephron and in renal
epithelial tumors. APMIS Suppl. 4:48–55. 1988.PubMed/NCBI
|
|
24
|
Skinnider BF, Folpe AL, Hennigar RA, Lim
SD, Cohen C, Tamboli P, Young A, de Peralta-Venturina M and Amin
MB: Distribution of cytokeratins and vimentin in adult renal
neoplasms and normal renal tissue: Potential utility of a
cytokeratin antibody panel in the differential diagnosis of renal
tumors. Am J Surg Pathol. 29:747–754. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wilkerson ML, Lin F, Liu H and Cheng L:
The application of immunohistochemical biomarkers in urologic
surgical pathology. Arch Pathol Lab Med. 138:1643–1665. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
van der Horst-Schrivers ANA, Sluiter WJ,
Kruizinga RC, van Leeuwaarde RS, Giles R, Olderode-Berends MJW and
Links TP: The incidence of consecutive manifestations in von
Hippel-Lindau disease. Fam Cancer. 18:369–376. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Solomon D and Schwartz A: Renal pathology
in von Hippel-Lindau disease. Hum Pathol. 19:1072–1079. 1988.
View Article : Google Scholar : PubMed/NCBI
|