Introduction
Cervical cancer is one of the most prevalent female
cancers (1), and is responsible
for significant morbidity and mortality worldwide (2). A lack of effective treatment programs
is a primary reason for this; therefore, novel therapeutic agents
are required. Plants are being investigated for their use in
chemotherapy, due to their availability, cost and lack of
side-effects (3).
Berberine is an isoquinoline alkaloid derived from
the Chinese herb Huang Lian (4),
which is commonly used for the treatment of gastrointestinal
complaints, diarrhea and other conditions. Previous studies have
suggested that berberine exerts significant anticancer activities
against various cancer cell types, including human breast cancer
(1,5), lung cancer (6), colon cancer (7), uterine leiomyoma (8), multiple myeloma (9), osteosarcoma (10), prostate cancer (11,12),
cervical cancer (13,14), nasopharyngeal carcinoma (15,16),
hepatocellular carcinoma (17–19),
gastric carcinoma (20) and murine
melanoma (21).
Berberine has been reported to suppress human
papilloma virus (HPV) transcription and downstream signaling to
induce growth arrest and apoptosis in SiHa and HPV18-positive
cervical cancer cells via the modulation of activator protein 1
activity (22). In addition,
berberine may reverse epithelial-to-mesenchymal transition, and
inhibit metastasis and tumor-induced angiogenesis in SiHa cells
(13). In Ca Ski human cervical
cancer cells, berberine has been reported to enhance apoptosis via
an increase in the ratio of p53 and B-cell lymphoma 2
(Bcl-2)-associated X protein (Bax)/Bcl-2, increased reactive oxygen
species and calcium levels, disrupted mitochondrial membrane
potential and increased caspase-3 activity, as mediated by GADD153
(23). Although berberine has been
demonstrated to possess anticancer activities, the underlying
mechanisms by which it exerts these effects remain to be fully
elucidated. In addition, the effects of berberine on HeLa229 cells
have not been reported. Therefore, the present study aimed to
investigate the effects of berberine hydrochloride on cell
proliferation, apoptosis and associated gene expression in HeLa229
human cervical cancer cells.
Materials and methods
Materials
HeLa229 human cervical carcinoma cells were obtained
from the China Center for Type Culture Collection (Wuhan, China).
Berberine hydrochloride was purchased from Xi'an Guanyu Bio-Tech
Co., Ltd. (Xi'an, China). Fetal calf serum (FCS) was purchased from
Hangzhou Sijiqing Biological Engineering Materials Co., Ltd.
(Hangzhou, China). Trypsin and
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
were purchased from Amresco, LLC (Cleveland, OH, USA). Penicillin,
streptomycin and dimethyl sulfoxide (DMSO) were obtained from
Sigma-Aldrich; Merck Millipore (Darmstadt, Germany). The Annexin
V-Fluorescein Isothiocyanate (FITC)/Propidium Iodide (PI) Apoptosis
Detection kit was from BioVision, Inc. (Milpitas, CA, USA). All
other chemicals and solvents used were of the highest purity grade
available.
Cell culture and treatment
Cells were cultured in RPMI-1640 medium,
supplemented with 10% heat-inactivated FCS, 100 IU/ml penicillin
and 100 μg/ml streptomycin at 37°C in a humidified
atmosphere containing 5% CO2.
MTT assay
Cells in the exponential growth phase were
harvested, adjusted to 2×104 cells/ml and seeded in
96-well plates (200 μl/well). Following a 24-h incubation at
37°C, the medium was removed and berberine hydrochloride was added
to wells in a final concentration range of 3.362–215.168
μmol/l. The plate was incubated for a further 72 h,
following which 20 μl 5 mg/ml MTT reagent was added to
wells. Subsequent to a 4-h incubation at 37°C, formazan crystals
formed by live cells were dissolved with 150 μl DMSO and
absorbance was measured at 490 nm using a microplate reader
(DG5033A; Nanjing Huadong Electronics Group Medical Equipment Co.,
Ltd., Nanjing, China). Viability was determined using the following
formula: % of growth = (optical density of treated cells/optical
density of untreated cells) × 100. The half maximal inhibitory
concentration (IC50) values were calculated as the
concentration of drug required to inhibit 50% proliferation
compared with untreated cells.
Detection of apoptosis-microscopy
Experiments were conducted as described previously
(24,25), using an Annexin V-FITC/PI Apoptosis
Detection kit. Cells at a density of 1.5×105 cells/ml
were incubated with 26.896 or 107.584 μmol/l berberine
hydrochloride at 37°C for 48 h. Adherent and floating cells were
harvested, washed twice with PBS and suspended in 500 μl of
1X Binding Buffer. Annexin V-FITC (5 μl) and 10 μl PI
were added and cells were vortexed and incubated for 5 min in the
dark. Cells were visualized immediately using an inverted
fluorescence biological microscope XD-101 (Nanjing Jiangnan
Photovoltaic Group Co., Ltd., Nanjing, China).
Detection of apoptosis-flow
cytometry
Cells at a density of 1.5×105 cells/ml
were incubated with 42.93 or 107.584 μmol/l berberine
hydrochloride at 37°C for 24, 48 and 72 h. Apoptosis was detected
using the Annexin V-FITC/PI Apoptosis Detection kit, as
aforementioned. Cells were analyzed immediately by flow cytometry
using an FC 500 (Beckman Coulter, Inc., Brea, CA, USA).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Cells (1.5×105 cells/ml) were incubated
with 21.465, 42.93 or 107.584 μmol/l berberine hydrochloride
for 48 h. Total RNA was prepared using TRIzol® reagent
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
according to the manufacturer's protocol and was reverse
transcribed using RevertAid™ Moloney Murine Leukemia Virus Reverse
Transcriptase and oligo (dT) primers (Fermentas; Thermo Fisher
Scientific, Inc., Pittsburgh, PA, USA). qPCR was performed on the
resulting cDNA using an ABI 7900HT Fast Real Time PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.) and
SYBR® Green Real Time PCR Master mix (Toyobo Co., Ltd.,
Osaka, Japan). The reaction mixture volume was 25 μl,
including 11.2 μl PCR water, 2.5 μl SYBR®
Green Real Time PCR Master mix, 0.5 μl forward primer (10
μM), 0.5 μl reverse primer (10 μM) and 0.3
μl cDNA. Primers were synthesized by Shanghai Generay
Biotech Co., Ltd. (Shanghai, China), and sequences are presented in
Table I. The cycling conditions
were as follows: An initial denaturation step at 94°C for 7 min,
followed by 35 cycles of denaturation at 94°C for 30 sec, annealing
at 63°C for 30 sec and extension at 72°C for 20 sec. Results were
analyzed to determine the PCR cycle number that generated the first
fluorescence signal over a threshold [quantification cycle (Cq), 10
standard deviations (SDs) over the mean fluorescence generated
during the baseline cycles], following which the ΔΔCq method was
used to measure relative gene expression (29). Expression of the analyzed genes
were normalized to the endogenous reference gene, β-actin.
 | Table IPrimer sequences for reverse
transcription-quantitative polymerase chain reaction. |
Table I
Primer sequences for reverse
transcription-quantitative polymerase chain reaction.
| Gene | Sequence (5′-3′)
| Product size
(bp) | Reference |
|---|
| Forward | Reverse |
|---|
| β-actin |
GTACCCTGGCATTGCCGACA |
GGACTCGTCATACTCCTGCTTGCT | 181 | 26 |
| p53 |
GCCCACTTCACCGTACTAA |
TGGTTTCAAGGCCAGATGT | 153 | 25 |
| Bcl-2 |
GGGAGGATTGTGGCCTTCTT |
TCATCCACAGGGCGATGTT | 99 | 27 |
| Cox-2 |
CACCCATGTCAAAACCGAGG |
CCGGTGTTGAGCAGTTTTCTC | 103 | 28 |
Statistical analysis
All experiments were performed in triplicate. Data
are expressed as the mean ± SD. Data were analyzed in SPSS version
16.0 (SPSS, Inc., Chicago, IL, USA), using one-way analysis of
variance followed by the least significant difference test to
compare treatment and control groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
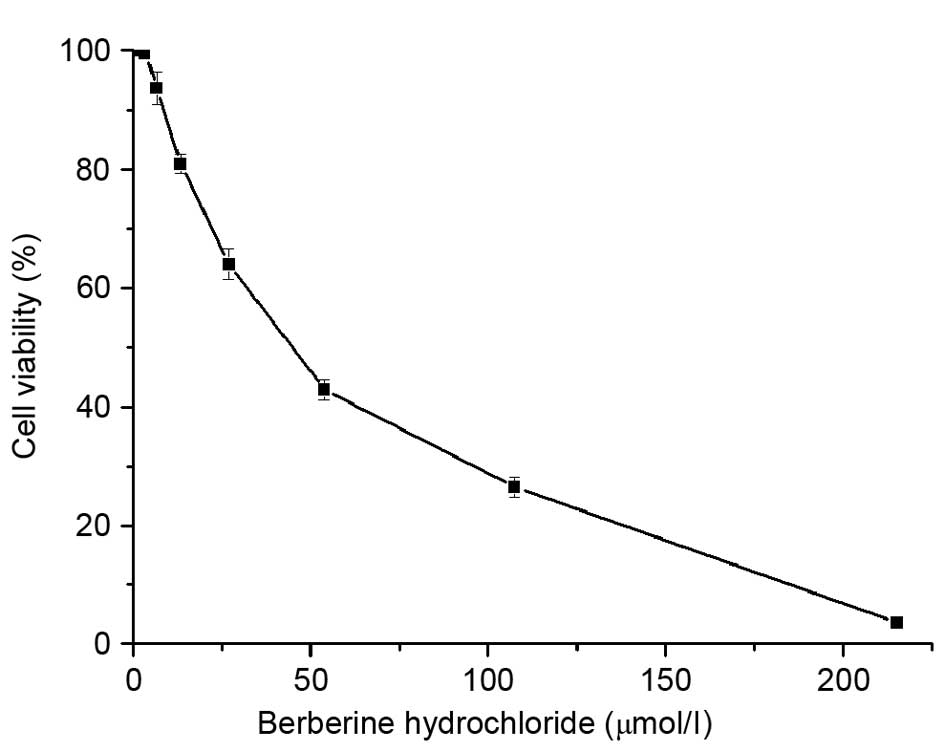
Berberine hydrochloride reduces cell
viability
The effects of berberine hydrochloride on the
viability of HeLa229 human cervical carcinoma cells were evaluated
using an MTT assay (Fig. 1). The
IC50 for HeLa229 cells at 72 h was 42.93 μmol/l.
Berberine hydrochloride inhibited HeLa229 cells in a dose-dependent
manner. Cell viability following treatment with 3.362, 6.724,
53.791 and 215.164 μmol/l berberine hydrochloride treatment
was 99.56, 93.61, 42.85 and 3.61%, respectively. The results
demonstrated that HeLa229 cell viability was reduced following a
72-h incubation with berberine hydrochloride.
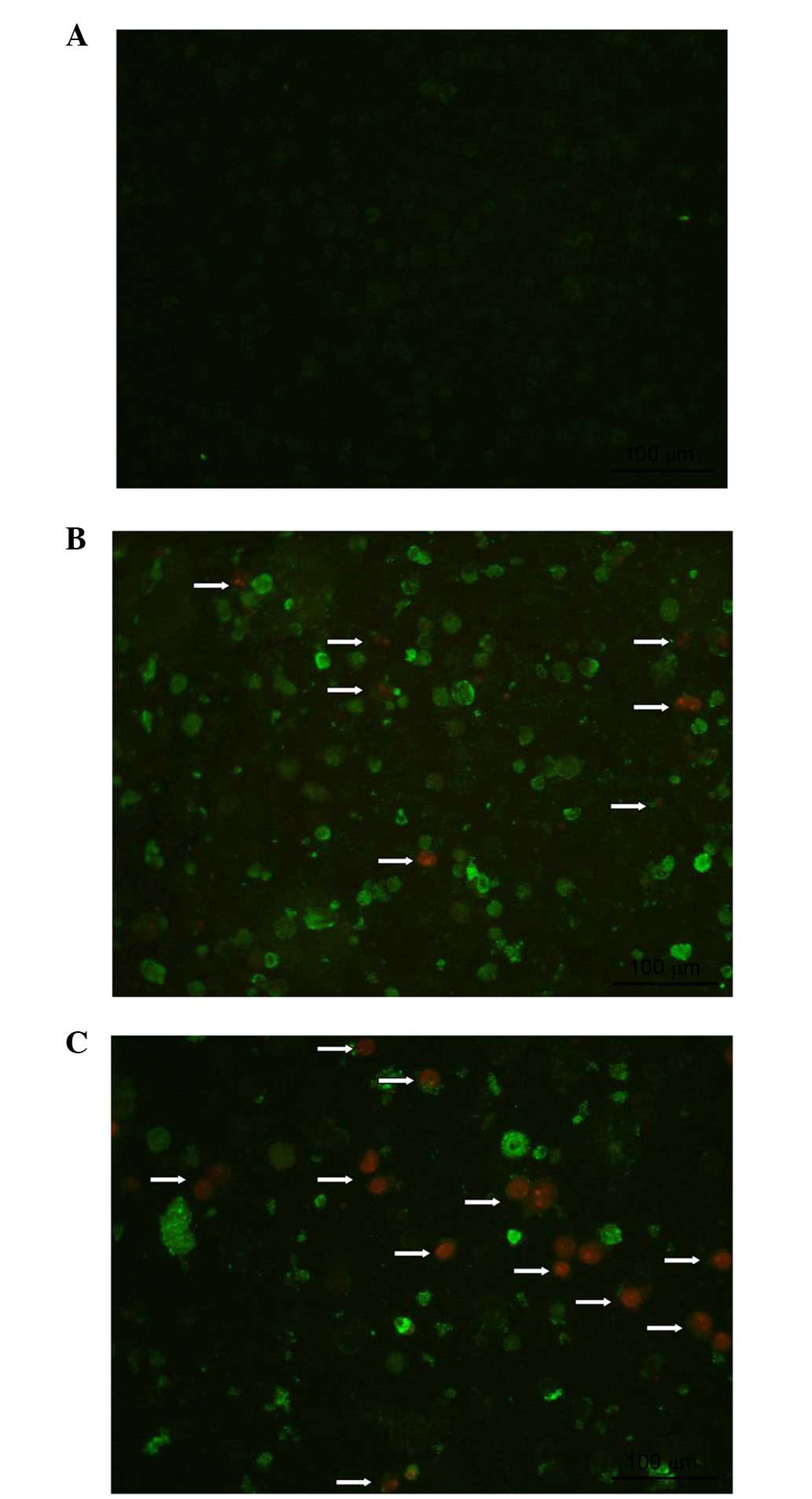
Berberine hydrochloride induces apoptosis
of HeLa229 cells
Apoptosis of HeLa229 cells was detected using the
Annexin V-FITC/Propidium Iodide Apoptosis Detection kit. As
presented in Fig. 2, apoptotic
cells could be observed clearly by fluorescence microscopy. The
cell membranes of early and late apoptotic cells were FITC-positive
(green), whereas late apoptotic cells additionally had PI-positive
(red) nuclei accompanied by condensed chromatin and apoptotic
bodies. Increased numbers of late apoptotic cells were observed
following treatment with 107.584 μmol/l, compared with
26.896 μmol/l berberine hydrochloride.
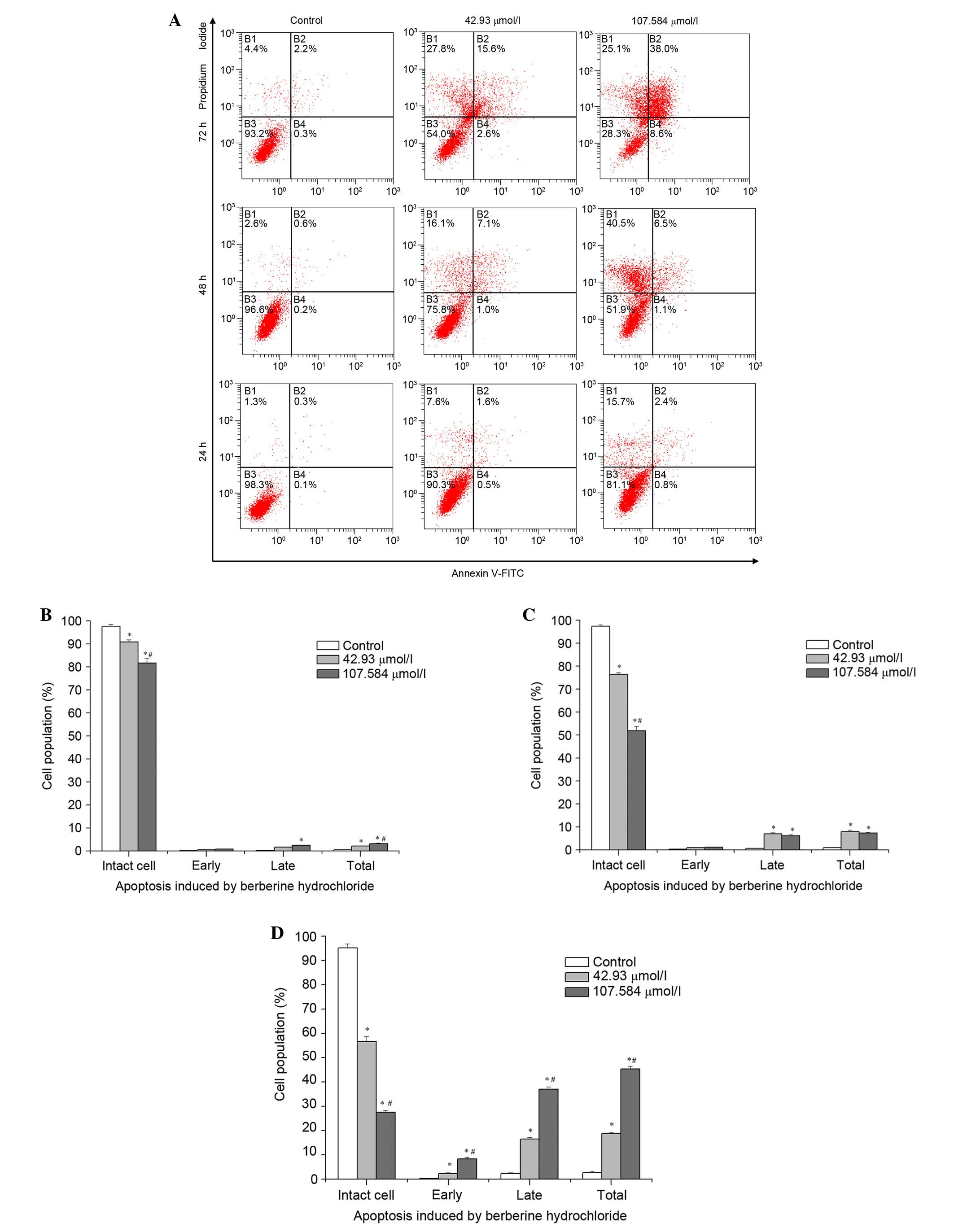
As presented in Fig.
3, no significant differences were observed in the proportions
of early apoptotic cells following treatment with 42.93 or 107.584
μmol/l berberine hydrochloride for 24 or 48 h, compared with
untreated control cells; or in the proportions of late apoptotic
cells following treatment with 42.93 μmol/l berberine
hydrochloride for 24 h, compared with control cells. Significant
early apoptosis was observed following treatment with 42.93 or
107.584 μmol/l berberine hydrochloride for 72 h, at 2.37 and
8.37% of cells, respectively (all P<0.001). The percentage of
late apoptotic cells treated with 42.93 μmol/l berberine
hydrochloride for 48 h was greater than for cells treated with
107.584 μmol/l berberine hydrochloride; however, this was
reversed at 72 h, at 16.43 and 37%, respectively. The percentage of
total apoptotic cells increased markedly from 7.3% in the 42.93
μmol/l treatment group at 48 h to 45.37% in the 107.584
μmol/l treatment group for 72 h. Significant differences
were observed in the proportions of intact cells (non-apoptotic
live cells) at 24, 48 and 72 h between the three groups (all
P<0.001). In addition, compared with control groups, significant
differences were detected in the proportions of early and late
apoptotic cells at 72 h [all P<0.001, except early apoptotic
cells of the 42.93 μmol/l treatment group (P=0.007)], and in
the percentage of total apoptotic cells at 24 (42.93 μmol/l,
P=0.001; 107.584 μmol/l, P<0.001), 48 and 72 h (all
P<0.001). Compared with the 42.93 μmol/l berberine
hydrochloride treatment group, the 107.584 μmol/l berberine
hydrochloride treatment group revealed significant differences
(P<0.001) in the percentage of intact cells at all time points
and early and late apoptotic cells at 72 h, and in the percentage
of total apoptotic cells at 24 h (P=0.014) and 72 h (P<0.001).
These results suggest that berberine hydrochloride induced
apoptosis of HeLa229 cells in a dose- and time-dependent
manner.
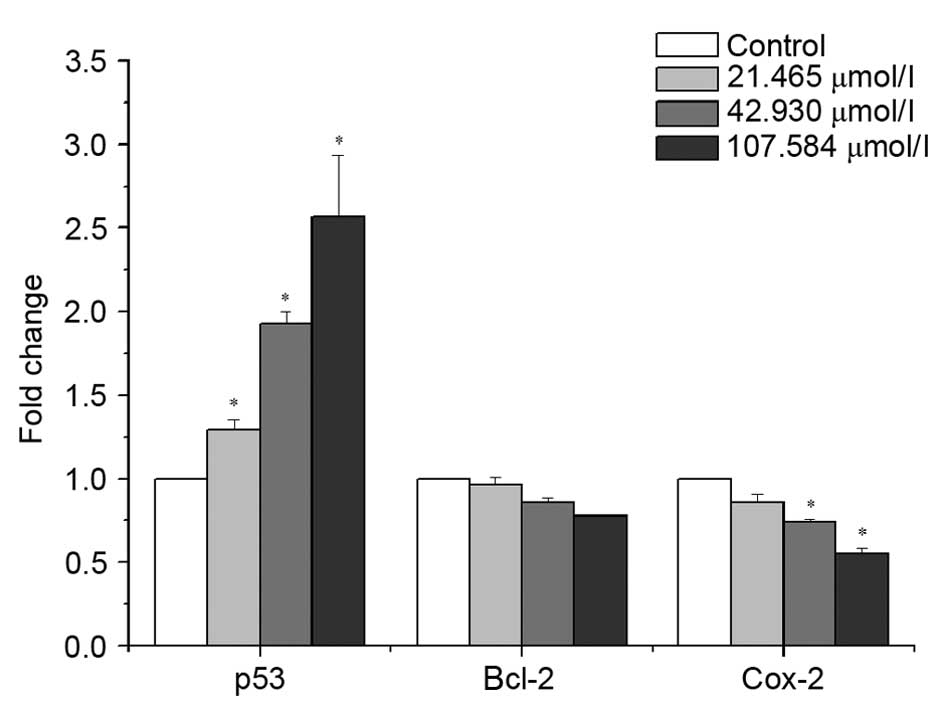
mRNA expression levels in berberine
hydrochloride-treated cells
p53, Bcl-2 and cox-2 mRNA expression levels in
HeLa229 cells were assessed by RT-qPCR, following treatment with
21.465, 42.93 or 107.584 μmol/l berberine hydrochloride for
48 h (Fig. 4). Berberine
hydrochloride upregulated mRNA expression levels of p53, whereas
mRNA expression levels of Bcl-2 and cox-2 were downregulated in a
dose-dependent manner. mRNA expression levels of p53 increased from
1.287- to 2.57-fold relative to control, whereas mRNA expression
levels of cox-2 decreased from 0.856-to 0.545-fold, and Bcl-2
decreased from 0.962- to 0.775-fold. Significant differences were
observed in p53 mRNA expression levels between treated (21.465
μmol/l, P=0.025; 42.93 μmol/l, P<0.001; 107.584
μmol/l, P<0.001) and untreated control cells, and in
cox-2 mRNA expression levels between cells treated with 42.93
(P=0.039) or 107.584 (P=0.002) μmol/l berberine
hydrochloride and control cells.
Discussion
Berberine is a naturally-occurring isoquinoline
alkaloid, which exerts antitumor effects on numerous cancer types
(5,30–38)
and is non-toxic to normal cells (22). However, the effects of berberine on
the HeLa229 human cervical carcinoma cell line remain unclear.
The results of the present study suggested that
treatment with berberine hydrochloride for 72 h significantly
decreased the viability of HeLa229 cells. Annexin V and PI staining
demonstrated that berberine hydrochloride treatment resulted in
apoptosis of HeLa229 cells. Apoptosis is tightly regulated by anti-
and proapoptotic effector molecules (39) and is caused by the activation of
caspases. Two separate pathways (extrinsic and intrinsic) of
caspase activation have been described (40). p53 is a critical regulator of
apoptosis (41), initiating the
intrinsic pathway via the transcriptional activation of Bcl-2
family members (42). The Bcl-2
family consists of three major groups, which differ in regions of
Bcl-2 homology (BH domains) and function: Multidomain
anti-apoptotic (including Bcl-2), multidomain proapoptotic and
BH3-only proapoptotic (43).
Berberine hydrochloride may upregulate the expression levels of
p53, triggering the intrinsic pathway of apoptosis via
downregulation of Bcl-2 expression levels. This would result in
release of cytochrome c in the mitochondrial membrane and
activation of caspase-9, resulting in apoptosis (44). Cox-2 is a target for anticancer
therapy (45), which is involved
in the extrinsic pathway. Its expression increases as cells become
cancerous (46), and it is
associated with the stimulation of angiogenesis, and tumor growth,
invasion and metastasis (47–49).
In the present study, treatment with berberine hydrochloride
increased the expression of p53 and decreased the expression of
Bcl-2 and cox-2, in a dose-dependent manner. These results are
consistent with a previous study, which demonstrated that berberine
induced apoptosis via a significant decrease in the Bcl-2/Bax
ratio, and the upregulation of Fas, Fas ligand, tumor necrosis
factor (TNF)-α, TNF receptor-associated factor 1 and p53 in HeLa
cells (14).
In conclusion, the results of the present study
suggested that berberine hydrochloride may exhibit significant
cytotoxicity against HeLa229 cells. At the lowest concentration
assessed (3.362 μmol/l), the inhibition of HeLa229 cells by
berberine hydrochloride was <1% (0.44%); however, inhibition
increased to >96% (96.39%) at the maximum concentration examined
(215.168 μmol/l). Berberine hydrochloride induced typical
characteristics of apoptosis in HeLa229 cells, including nuclear
condensation, nuclear fragmentation and the formation of apoptotic
bodies. In addition, 42.93 and 107.584 μmol/l berberine
hydrochloride induced apoptosis in a time-dependent manner.
Berberine hydrochloride induced apoptosis in HeLa229 cells via the
activation of the extrinsic and intrinsic pathways, involving the
upregulation of p53 mRNA expression levels and the downregulation
of Bcl-2 and cox-2 mRNA expression levels. Therefore, berberine
appears to be a potential therapeutic agent for the treatment of
cervical cancer.
Acknowledgments
The present study was supported by a grant from the
Science and Technology Research Project of Hubei Provincial
Department of Education (grant no. Q20092504) and a grant from the
Discipline Groups Construction of Food New-type Industrialization
of Hubei University of Arts and Science (grant no. XKQ08321).
References
|
1
|
Xie J, Xu Y, Huang X, Chen Y, Fu J, Xi M
and Wang L: Berberine-induced apoptosis in human breast cancer
cells is mediated by reactive oxygen species generation and
mitochondrial-related apoptotic pathway. Tumour Biol. 36:1279–1288.
2015. View Article : Google Scholar
|
|
2
|
Parkin DM: The global health burden of
infection-associated cancers in the year 2002. Int J Cancer.
118:3030–3044. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Engel N, Oppermann C, Falodun A and Kragl
U: Proliferative effects of five traditional Nigerian medicinal
plant extracts on human breast and bone cancer cell lines. J
Ethnopharmacol. 137:1003–1010. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sun Y, Xun K, Wang Y and Chen X: A
systematic review of the anticancer properties of berberine, a
natural product from Chinese herbs. Anticancer Drugs. 20:757–769.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Li X, Zhao SJ, Shi HL, Qiu SP, Xie JQ, Wu
H, Zhang BB, Wang ZT, Yuan JY and Wu XJ: Berberine hydrochloride
IL-8 dependently inhibits invasion and IL-8-independently promotes
cell apoptosis in MDA-MB-231 cells. Oncol Rep. 32:2777–2788.
2014.PubMed/NCBI
|
|
6
|
Xi S, Chuang K, Fang K, Lee Y, Chung J and
Chuang Y: Effect of berberine on activity and mRNA expression of
N-acetyltransferase in human lung cancer cell line A549. J Tradit
Chin Med. 34:302–308. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Guamán Ortiz LM, Tillhon M, Parks M, Dutto
I, Prosperi E, Savio M, Arcamone AG, Buzzetti F, Lombardi P and
Scovassi AI: Multiple effects of berberine derivatives on colon
cancer cells. Biomed Res Int. 2014:9245852014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wu HL, Chuang TY, Al-Hendy A, Diamond MP,
Azziz R and Chen YH: Berberine inhibits the proliferation of human
uterine leiomyoma cells. Fertil Steril. 103:1098–1106. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Qing Y, Hu H, Liu Y, Feng T, Meng W, Jiang
L, Sun Y and Yao Y: Berberine induces apoptosis in human multiple
myeloma cell line U266 through hypomethylation of p53 promoter.
Cell Biol Int. 38:563–570. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhu Y, Ma N, Li HX, Tian L, Ba YF and Hao
B: Berberine induces apoptosis and DNA damage in MG-63 human
osteosarcoma cells. Mol Med Rep. 10:1734–1738. 2014.PubMed/NCBI
|
|
11
|
Zhang Q, Zhang C, Yang X, Yang B, Wang J,
Kang Y, Wang Z, Li D, Huang G, Ma Z, et al: Berberine inhibits the
expression of hypoxia induction factor-1alpha and increases the
radiosensitivity of prostate cancer. Diagn Pathol. 9:982014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang LY, Wu YL, Gao XH and Guo F:
Mitochondrial protein cyclophilin-D-mediated programmed necrosis
attributes to berberine-induced cytotoxicity in cultured prostate
cancer cells. Biochem Biophys Res Commun. 450:697–703. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chu SC, Yu CC, Hsu LS, Chen KS, Su MY and
Chen PN: Berberine reverses epithelial-to-mesenchymal transition
and inhibits metastasis and tumor-induced angiogenesis in human
cervical cancer cells. Mol Pharmacol. 86:609–623. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lu B, Hu M, Liu K and Peng J: Cytotoxicity
of berberine on human cervical carcinoma HeLa cells through
mitochondria, death receptor and MAPK pathways, and in-silico
drug-target prediction. Toxicol In Vitro. 24:1482–1490. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li CH, Wu DF, Ding H, Zhao Y, Zhou KY and
Xu DF: Berberine hydrochloride impact on physiological processes
and modulation of twist levels in nasopharyngeal carcinoma CNE-1
cells. Asian Pac J Cancer Prev. 15:1851–1857. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang C, Yang X, Zhang Q, Yang B, Xu L,
Qin Q, Zhu H, Liu J, Cai J, Tao G, et al: Berberine radiosensitizes
human nasopharyngeal carcinoma by suppressing hypoxia-inducible
factor-1α expression. Acta Otolaryngol. 134:185–192. 2014.
View Article : Google Scholar
|
|
17
|
Wang N, Zhu M, Wang X, Tan HY, Tsao SW and
Feng Y: Berberine-induced tumor suppressor p53 up-regulation gets
involved in the regulatory network of MIR-23a in hepatocellular
carcinoma. Biochim Biophys Acta. 1839:849–857. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang L, Wei D, Han X, Zhang W, Fan C,
Zhang J, Mo C, Yang M, Li J, Wang Z, et al: The combinational
effect of vincristine and berberine on growth inhibition and
apoptosis induction in hepatoma cells. J Cell Biochem. 115:721–730.
2014. View Article : Google Scholar
|
|
19
|
Guo N, Yan A, Gao X, Chen Y, He X, Hu Z,
Mi M, Tang X and Gou X: Berberine sensitizes rapamycin-mediated
human hepatoma cell death in vitro. Mol Med Rep. 10:3132–3138.
2014.PubMed/NCBI
|
|
20
|
Zhang XZ, Wang L, Liu DW, Tang GY and
Zhang HY: Synergistic inhibitory effect of berberine and d-limonene
on human gastric carcinoma cell line MGC803. J Med Food.
17:955–962. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mittal A, Tabasum S and Singh RP:
Berberine in combination with doxorubicin suppresses growth of
murine melanoma B16F10 cells in culture and xenograft.
Phytomedicine. 21:340–347. 2014. View Article : Google Scholar
|
|
22
|
Mahata S, Bharti AC, Shukla S, Tyagi A,
Husain SA and Das BC: Berberine modulates AP-1 activity to suppress
HPV transcription and downstream signaling to induce growth arrest
and apoptosis in cervical cancer cells. Mol Cancer. 10:392011.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lin JP, Yang JS, Chang NW, Chiu TH, Su CC,
Lu KW, Ho YT, Yeh CC, Mei-Dueyang, Lin HJ and Chung JG: GADD153
mediates berberine-induced apoptosis in human cervical cancer Ca
ski cells. Anticancer Res. 27:3379–3386. 2007.PubMed/NCBI
|
|
24
|
Abu Bakar MF, Mohamad M, Rahmat A, Burr SA
and Fry JR: Cytotoxicity, cell cycle arrest, and apoptosis in
breast cancer cell lines exposed to an extract of the seed kernel
of Mangifera pajang (bambangan). Food Chem Toxicol. 48:1688–1697.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tian Z, An N, Zhou B, Xiao P, Kohane IS
and Wu E: Cytotoxic diarylheptanoid induces cell cycle arrest and
apoptosis via increasing ATF3 and stabilizing p53 in SH-SY5Y cells.
Cancer Chemother Pharmacol. 63:1131–1139. 2009. View Article : Google Scholar
|
|
26
|
Itoh M, Murata T, Suzuki T, Shindoh M,
Nakajima K, Imai K and Yoshida K: Requirement of STAT3 activation
for maximal collagenase-1 (MMP-1) induction by epidermal growth
factor and malignant characteristics in T24 bladder cancer cells.
Oncogene. 25:1195–1204. 2006. View Article : Google Scholar
|
|
27
|
Jiang N, Zhou LQ and Zhang XY:
Downregulation of the nucleosome-binding protein 1 (NSBP1) gene can
inhibit the in vitro and in vivo proliferation of prostate cancer
cells. Asian J Androl. 12:709–717. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhou A, Scoggin S, Gaynor RB and Williams
NS: Identification of NF-kappa B-regulated genes induced by
TNFalpha utilizing expression profiling and RNA interference.
Oncogene. 22:2054–2064. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lapillonne H, Konopleva M, Tsao T, Gold D,
McQueen T, Sutherland RL, Madden T and Andreeff M: Activation of
peroxisome proliferator-activated receptor gamma by a novel
synthetic triterpenoid 2-cyano-3, 12-dioxooleana-1, 9-dien-28-oic
acid induces growth arrest and apoptosis in breast cancer cells.
Cancer Res. 63:5926–5939. 2003.PubMed/NCBI
|
|
30
|
Chidambara Murthy KN, Jayaprakasha GK and
Patil BS: The natural alkaloid berberine targets multiple pathways
to induce cell death in cultured human colon cancer cells. Eur J
Pharmacol. 688:14–21. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hur JM, Hyun MS, Lim SY, Lee WY and Kim D:
The combination of berberine and irradiation enhances anti-cancer
effects via activation of p38 MAPK pathway and ROS generation in
human hepatoma cells. J Cell Biochem. 107:955–964. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kim JB, Yu JH, Ko E, Lee KW, Song AK, Park
SY, Shin I, Han W and Noh DY: The alkaloid Berberine inhibits the
growth of Anoikis-resistant MCF-7 and MDA-MB-231 breast cancer cell
lines by inducing cell cycle arrest. Phytomedicine. 17:436–440.
2010. View Article : Google Scholar
|
|
33
|
Wang J, Liu Q and Yang Q:
Radiosensitization effects of berberine on human breast cancer
cells. Int J Mol Med. 30:1166–1172. 2012.PubMed/NCBI
|
|
34
|
Chou HC, Lu YC, Cheng CS, Chen YW, Lyu PC,
Lin CW, Timms JF and Chan HL: Proteomic and redox-proteomic
analysis of berberine-induced cytotoxicity in breast cancer cells.
J Proteomics. 75:3158–3176. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zheng F, Tang Q, Wu J, Zhao S, Liang Z, Li
L, Wu W and Hann S: p38α MAPK-mediated induction and interaction of
FOXO3a and p53 contribute to the inhibited-growth and
induced-apoptosis of human lung adenocarcinoma cells by berberine.
J Exp Clin Cancer Res. 33:362014. View Article : Google Scholar
|
|
36
|
Ma X, Zhou J, Zhang CX, Li XY, Li N, Ju
RJ, Shi JF, Sun MG, Zhao WY, Mu LM, et al: Modulation of
drug-resistant membrane and apoptosis proteins of breast cancer
stem cells by targeting berberine liposomes. Biomaterials.
34:4452–4465. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Qi HW, Xin LY, Xu X, Ji XX and Fan LH:
Epithelial-to-mesenchymal transition markers to predict response of
Berberine in suppressing lung cancer invasion and metastasis. J
Transl Med. 12:222014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Scordino A, Campisi A, Grasso R, Bonfanti
R, Gulino M, Iauk L, Parenti R and Musumeci F: Delayed luminescence
to monitor programmed cell death induced by berberine on thyroid
cancer cells. J Biomed Opt. 19:1170052014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Weyhenmeyer B, Murphy AC, Prehn JH and
Murphy BM: Targeting the anti-apoptotic bcl-2 family members for
the treatment of cancer. Exp Oncol. 34:192–199. 2012.PubMed/NCBI
|
|
40
|
Green DR: Apoptotic pathways: Paper wraps
stone blunts scissors. Cell. 102:1–4. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Žegura B, Gajski G, Štraser A and
Garaj-Vrhovac V: Cylindrospermopsin induced DNA damage and
alteration in the expression of genes involved in the response to
DNA damage, apoptosis and oxidative stress. Toxicon. 58:471–479.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Vogelstein B, Lane D and Levine AJ:
Surfing the p53 network. Nature. 408:307–310. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Certo M, Del Gaizo Moore V, Nishino M, Wei
G, Korsmeyer S, Armstrong SA and Letai A: Mitochondria primed by
death signals determine cellular addiction to antiapoptotic BCL-2
family members. Cancer Cell. 9:351–365. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kim R, Tanabe K, Uchida Y, Emi M, Inoue H
and Toge T: Current status of the molecular mechanisms of
anticancer drug-induced apoptosis. The contribution of
molecular-level analysis to cancer chemotherapy. Cancer Chemother
Pharmacol. 50:343–352. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang C, Yan W, Li B, Xu B, Gong Y, Chu F,
Zhang Y, Yao Q, Wang P and Lei H: A New Ligustrazine
Derivative-Selective Cytotoxicity by Suppression of NF-kB/p65 and
COX-2 expression on human hepatoma cells. Part 3. Int J Mol Sci.
16:16401–16413. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Breinig M, Schirmacher P and Kern MA:
Cyclooxygenase-2 (COX-2)-a therapeutic target in liver cancer? Curr
Pharm Des. 13:3305–3315. 2007. View Article : Google Scholar
|
|
47
|
Tang TC, Poon RT, Lau CP, Xie D and Fan
ST: Tumor cyclooxygenase-2 levels correlate with tumor invasiveness
in human hepatocellular carcinoma. World J Gastroenterol.
11:1896–1902. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhong B, Cai X, Chennamaneni S, Yi X, Liu
L, Pink JJ, Dowlati A, Xu Y, Zhou A and Su B: From COX-2 inhibitor
nimesulide to potent anti-cancer agent: Synthesis, in vitro, in
vivo, and pharmacokinetic evaluation. Eur J Med Chem. 47:432–444.
2012. View Article : Google Scholar
|
|
49
|
Mazhar D, Gillmore R and Waxman J: COX and
cancer. QJM. 98:711–718. 2005. View Article : Google Scholar : PubMed/NCBI
|