Introduction
Stroke caused by atherosclerosis remains a leading
cause of morbidity and mortality with over 15 million strokes
occurring every year in the world (1). The rapture of vulnerable
atherosclerotic plaque is the common reason in acute
cerebrovascular event, impacting prognostic implications and
eventually ending in death of patients (2). The progression of vulnerable plaques
rupture has not been clearly elucidated, and it tends to be treated
with routine clinical measures (3). Prognostic biomarker, which aid in
predicting the future course of plaque, may be used to identify
phenotype of plaques, especially the vulnerable one. Disease
progression biomarker can also serve as a marker of pharmacodynamic
effect, which have the potential to show the biological response
occurred in a patient who received an experimental therapeutic, may
help in assessing the efficacy of this treatment. Additionally,
inflammation in ischemic stroke plays an important role in
predicting the outcome in ischemic brain damage extent (4,5).
Therefore, this study investigated changes of both serum and
histological inflammatory markers in plaque of varying stages of
stroke patients in order to find a prognostic biomarker in serum
level which can accurately predicting the future course of
plaque.
It is well documented that increased plasma levels
of inflammatory markers such as interleukin (IL) which are
associated with a higher cardiovascular risk (6). It has already been described that
IL-6 has predictive value for future cardiovascular events and is
found to be expressed in atherosclerotic tissue and have
pro-inflammatory properties by activating and recruiting
inflammatory cells which can contribute to plaque instability
(7,8). By contrast, IL-10 is a potent
anti-inflammatory cytokine with macrophage deactivating properties
(9). Study has showed that serum
levels of anti-inflammatory marker IL-10 were downregulated in
patients with vulnerable plaques (7). However, the pattern of IL-6 and IL-10
on serum level changes in stroke patients of different stages and
the best utility for assessing the progression of plaque has not
been determined.
Kinins, a pro-inflammatory and vasoactive peptide,
can act through the activation of two G protein-coupled receptors,
denoted as B1 and B2 receptors (B1R and B2R) (10,11).
B1R is weakly expressed under physiological conditions, but is
significantly elevated during inflammatory conditions or after
tissue damage whereas B2R is constitutively expressed (12). B1R expression is more pronounced in
regions of plaques from human plaques tissue compared with regions
devoid of plaques, indicating that B1R is closely related to
atheromatous diseases (13).
Furthermore, our preliminary study has demonstrated that B1R is
upregulated in human symptomatic carotid atherosclerotic plaques
(14). A variety of studies have
attempted to explore inflammatory markers in plaques and its
relationship with predictive value for future cerebrovascular
events, but most of them failed to elaborate the dynamic changes of
these inflammatory markers in different stages of human
atherosclerotic plaques patients. However, an explicit study of
dynamic changes of these inflammatory markers may facilitate future
efforts towards early diagnosis of vulnerable plaques and
monitoring of disease progression.
The goal of the present study was to evaluate serum
levels of inflammation markers in plaques after stroke in different
stages of patients (<30, 30–90, 90–180 and >180 days) to
investigate the inflammatory markers as a potential disease
progression and prognostic biomarker in the process of plaque
remodeling and stability and then explore their possible
correlations with histological changes; the purpose was to provide
experimental cues for management of patients with carotid
atherosclerotic plaque using serum biomarkers.
Materials and methods
Subjects
All patients undergoing carotid endarterectomy (CEA)
in two participating hospitals are asked to participate in the
study. The Medical Ethics Committee of both hospitals approved the
study, and participants provided written informed consent. For the
current research questions we have studied the atherosclerotic
plaques from 111 consecutive patients (stroke [n=80], TIA [n=14]
and asymptomatic [n=17]) who were included between April 2014 and
June 2017. From all patients, baseline data were obtained by
extensive questionnaires including history of vascular disease,
cardiovascular risk factors, and medication use (Table I). All patients were reviewed by
the vascular surgeon or neurologist before CEA to assess the nature
and timing of clinical symptoms. Patients who suffered from stroke
were categorized into 4 different groups according to the time
between ischemic event and surgery (<30, 30–90, 90–180 and
>180 days). Results from asymptomatic patients served as
negative values in comparison with data obtained from patients
suffering from stroke, and TIA served as positive values.
 | Table I.Baseline characteristics of the study
population. |
Table I.
Baseline characteristics of the study
population.
|
| Stroke |
|
|
|
|---|
|
|
|
|
|
|
|---|
|
| <30 days | 30–90 days | 90–180 days | >180 days | TIA | Asymptomatic | P-value |
|---|
| N | 16 | 24 | 32 | 8 | 14 | 17 | – |
| Age, y, mean ±
SD | 66.0±5.8 | 65.2±6.8 | 68.3±7.2 | 67.8±5.3 | 68.1±6.3 | 68.1±5.6 | 0.50 |
| Male, % (n) | 68.8 (11) | 70.8 (17) | 68.8 (22) | 87.5 (7) | 85.7 (12) | 70.6 (12) | 0.77 |
| Hypertension, %
(n) | 68.8 (11) | 79.2 (19) | 71.9 (23) | 50.0 (4) | 71.4 (10) | 76.5 (13) | 0.73 |
| Diabetes, % (n) | 25.0 (4) | 29.1 (7) | 18.8 (6) | 12.5 (1) | 21.4 (3) | 29.4 (5) | 0.88 |
| Smoking, % (n) | 31.3 (5) | 50.0 (12) | 37.5 (12) | 25.0 (2) | 35.7 (5) | 35.3 (6) | 0.79 |
| Hyperlipidemia, %
(n) | 41.7 (10) | 33.3 (8) | 62.5 (20) | 50.0 (4) | 57.1 (8) | 64.7 (11) | 0.27 |
| Statin, % (n) | 75.0 (12) | 70.8 (17) | 65.6 (21) | 62.5 (5) | 50.0 (7) | 82.4 (14) | 0.78 |
| Aspirin, % (n) | 87.5 (14) | 79.2 (19) | 59.4 (19) | 87.5 (7) | 57.1 (8) | 52.9 (9) | 0.10 |
| Anticoagulants, %
(n) | 18.7 (3) | 8.3 (2) | 6.3 (2) | – | 14.3 (2) | 5.9 (1) | 0.63 |
| NSAID, % (n) | 6.3 (1) | 8.3 (2) | 12.5 (4) | 12.5 (1) | 28.6 (4) | 17.6 (3) | 0.51 |
| ACE-inhibitor, %
(n) | 50.0 (8) | 37.5 (9) | 31.3 (10) | 12.5 (1) | 64.3 (9) | 41.2 (7) | 0.17 |
Measurement of serum inflammatory
markers by ELISA
Blood sampling of control patients were drawn by
venipuncture one day before CEA. All serum samples was centrifuged
for 10 min at 2,000 × g and then stored at −80°C until later
analysis. The serum levels of the IL-6, B1R and IL-10 were measured
using commercially available ELISA kits according to the
manufacturer's protocol (Cusabio, Wuhan, China).
Histological analysis
The atherosclerotic lesions were dissected into 5 mm
segments. Segment was fixed in formaldehyde 4% and then embedded in
paraffin. The segment having the greatest plaque area was used to
assess the plaque phenotype for histological analyses. For
measuring changes in plaque composition in histology, 4 µm thick
sections were cut from each paraffin block and stained with
hematoxylin and eosin (H&E) for histology and Picro-sirius red
for collagen. For immunohistochemical analyses, deparaffinized
sections were incubated with 1% H2O2 in
methanol for 3 min to eliminate endogenous peroxidase activity.
After blocking with 10% normal serum, the sections were incubated
for 1 h at room temperature with the primary antibodies with CD68
antibody for macrophages (1:300, GB13067-M-1, Servicebio, WuHan,
China); IL-6 (1:500, GB1117, Servicebio) and IL-10 (1:100,
sc-32815, Santa Cruz Biotechnology, Santa Cruz, CA, USA) and B1R
antibody (1:100, Bs-8675R, Bioss, WuHan, China). After washes in
phosphate-buffered saline for three times, the sections were
incubated with biotinylated secondary antibodies for 1 h and then
with the avidin-biotinylated horseradish peroxidase complex (ABC
Elite kit; Vector Laboratories Inc., Burlingame, CA, USA) for 30
min. Counterstaining was performed on representative sections with
hematoxylin, and then the sections were subject to image analysis
(Leica TCS SP2; Leica, Wetzlar, Germany) and analyzed using the
Image Pro Plus 6.0 software package (Media Cybernetics Inc.,
Rockville, MD, USA) to calculate the percentage of positive area in
each microscopic view at 200× magnification in plaques.
Statistical analysis
All results were expressed as means ± SD.
Statistical analyses were performed using GraphPad Prism 6.0
(GraphPad Software, Inc., La Jolla, CA, USA). For statistical
analyses of baseline characteristics of patients, the one-factor
ANOVA test was used for continuous variables and the χ2
test for categorical variables. For analysis of serum and
histological data, ANOVA followed by Tukey multiple comparison
tests (α=0.05) was used to calculate the difference between any two
groups of patients. Spearman's correlation analysis was used to
predict the association between serum and histological parameters
in patients. Receiver operating curve was constructed to identify
the cutoff point. P<0.05 was considered to indicate a
statistically significant difference.
Results
Clinical and histological
characteristics
The study population includes 17 asymptomatic, 14
TIA and 80 patients with stroke of different stages (<30, 30–90,
90–180 and >180 days). Baseline characteristics for all patients
are documented in Table I. There
were no statistically significant differences among different
groups with respect to clinical characteristics, risk factors or
drug use.
H&E, Picro-sirius red and CD68 staining were
used to reveal the plaque histological characteristics in stroke
patients of <30, 30–90, 90–180 and >180 days; typical images
are showed in Fig. 1. Compared
with asymptomatic group, plaques from stroke patients showed
intense macrophage infiltration, intraplaque hemorrhage and large
lipid core size at stroke of <30 days; and then declined at
stroke of 30–90 days; this decline was notably at stroke of 90–180
days and to basal levels at >180 days (ANOVA, all P<0.05). By
contrast, smooth muscle cells (SMC) content increased progressively
in plaques of stroke from <30 to >180 days (ANOVA,
P<0.05). However, no significant difference was found in stroke
of <30 days and TIA groups; stroke of >180 days and
asymptomatic groups (all P>0.05). These results suggest that
stroke patients of <30 and 30–90 days presented the early stage
of vulnerable plaque, whereas stroke patients of 90–180 and >180
days exhibited the process of vulnerable plaques remodeling to
stable plaques.
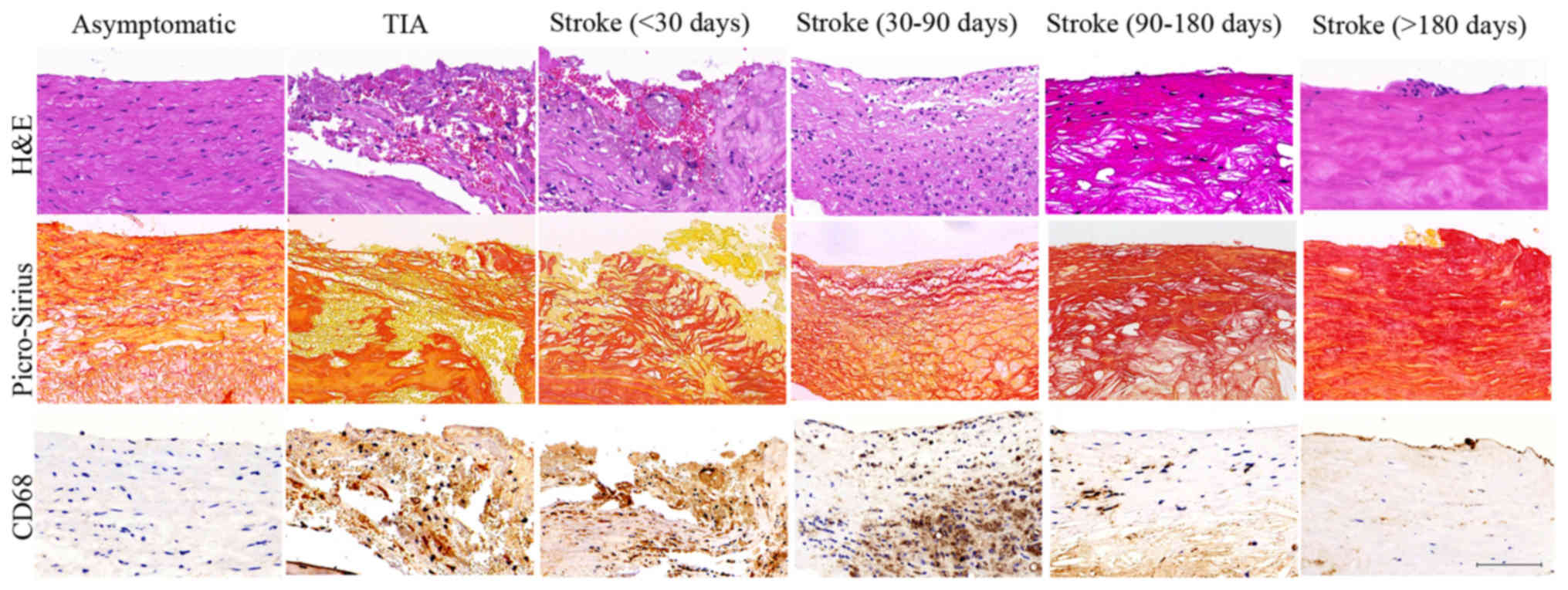 | Figure 1.Histological characteristics of
plaques. Representative images of plaques in asymptomatic, TIA,
stroke patients of <30, 30–90, 90–180 and >180 days. H&E
staining showed inflammatory cell infiltrations (TIA, Stroke <30
d and Stroke 30–90 d groups) and intraplaque hemorrhage (TIA,
Stroke <30 d groups) in the vulnerable plaques and intact cap
and predominantly fibrous tissue in the stable plaques
(Asymptomatic, Stroke 90–180 d and Stroke >180 d groups).
Picro-Sirius Red staining showed the large cholesterol crystals in
the lipid core in vulnerable plaques (TIA, Stroke <30 d and
Stroke 30–90 d groups) and collagen fibrous tissue in stable
plaques (Asymptomatic, Stroke 90–180 d and Stroke >180 d
groups). CD 68 staining showed histological appearance of a plaque
with substantial macrophage infiltration in the vulnerable plaques
(TIA, Stroke <30 d and Stroke 30–90 d groups) and minor
macrophage infiltration in stable plaques (Asymptomatic, Stroke
90–180 d and Stroke >180 d groups). Scale bar: 100 µm. |
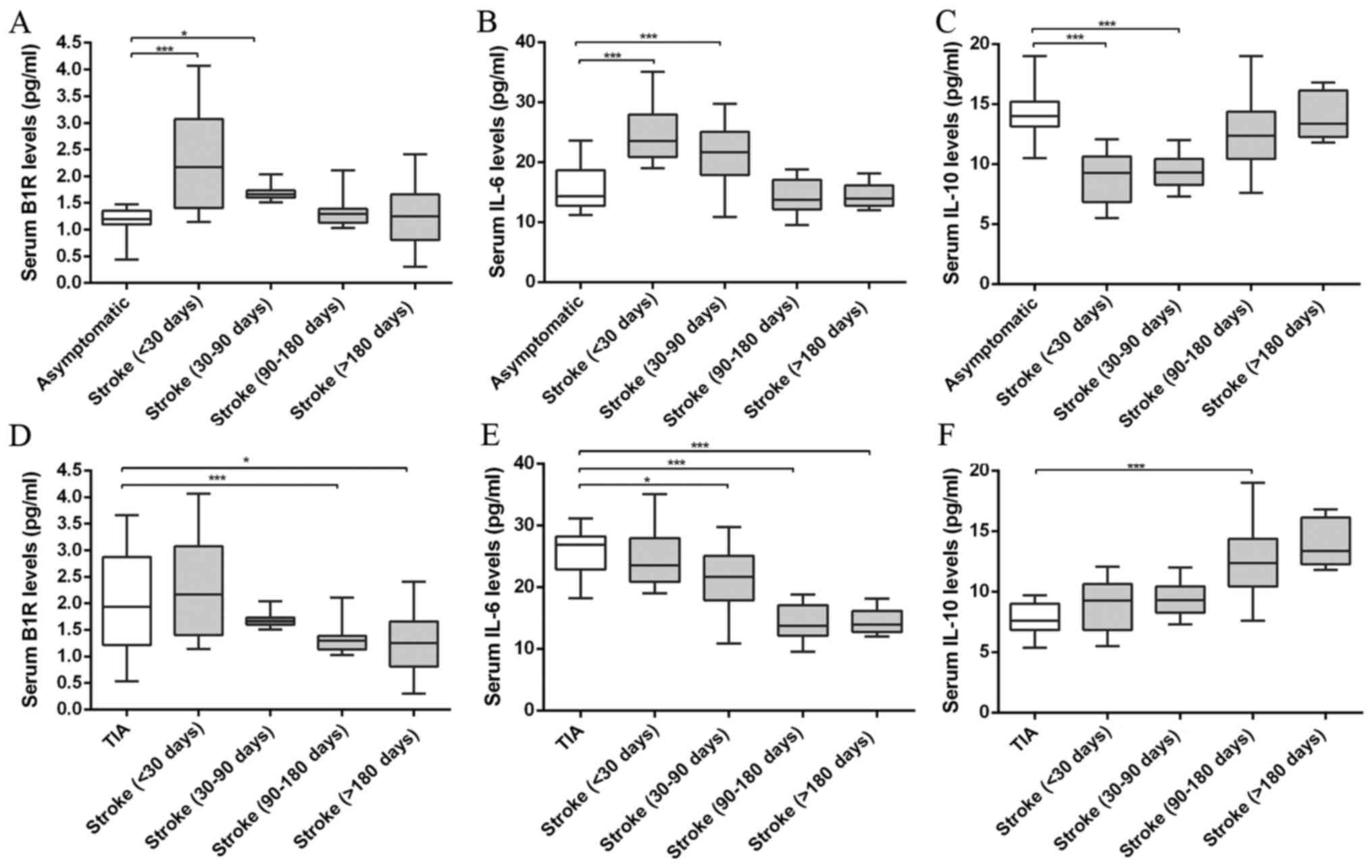
Serum levels of inflammatory
markers
The ELISA was used to measure the inflammatory
marker levels of stroke patients of <30, 30–90, 90–180 and
>180 days, and the results are showed in Fig. 2. After analysis and comparison the
serum inflammatory marker levels, we found the B1R levels were
significantly higher in the vulnerable plaques of the TIA and
stroke patients of <30, 30–90 days than that of asymptomatic and
stroke patients of 90–180 and >180 days (ANOVA, P<0.001). In
addition, compared with asymptomatic group, stroke patients showed
B1R level increase at <30 days and this increased B1R level
tended to decline after stroke from 30–90 to >180 days (ANOVA,
P<0.001). The B1R level decreased approximately two-fold in
stroke patients of >180 days compared to that of <30 days.
Likewise, the IL-6 levels showed a similar pattern in stroke
patients from <30 to >180 days (ANOVA, P<0.001). IL-6
levels were significantly higher in the vulnerable plaques of the
TIA and stroke patients of <30, 30–90 days than that of
asymptomatic and stroke patients of 90–180 and >180 days (ANOVA,
P<0.001). By contrast, the IL-10 was significantly reduced in
the vulnerable plaques of the TIA and stroke patients of <30,
30–90 days compared with that of asymptomatic and stroke patients
of 90–180 and >180 days (ANOVA, P<0.001). The IL-10 levels
were gradually upregulated in stroke patients in spatiotemporal
course (ANOVA, P<0.001). However, no significant differences
were found in serum B1R, IL-6 and IL-10 levels between TIA and
stroke groups of <30 days; asymptomatic and stroke groups of
>180 days (all P>0.05).
All results indicate that stroke patients at <30,
30–90 days showed unstable plaque phenotype with increased
pro-inflammatory marker IL-6 and B1R levels and decreased
anti-inflammatory marker IL-10 level; and then further remodeled to
more stable plaques with decreased IL-6, B1R levels and increased
IL-10 level at 90–180 and >180 days.
Histological analysis of inflammatory
markers
Immuno-histochemical analysis was used to stain
these inflammation markers to confirm histological changes in
plaques; typical images are shown in Fig. 3. The B1R and IL-6 staining appeared
in plaques of different groups, and percentages of positive area in
each microscopic view were markedly increased in plaques obtained
from TIA and stroke patients of <30 and 30–90 days compared with
asymptomatic and stroke patients of 90–180 and >180 days (ANOVA,
P<0.001). Stroke plaques showed the initial increases of B1R and
IL-6 contents at <30 days, and subsequent reduction from 30–90
to >180 days (both P<0.001). By contrast, the IL-10 content
was significantly reduced in the vulnerable plaques of the TIA and
stroke patients of <30, 30–90 days compared with that of
asymptomatic and stroke patients of 90–180 and >180 days (ANOVA,
P<0.001). In stroke plaques, we showed a noticeable increase in
IL-10 contents from <30 to >180 days (ANOVA, P<0.001).
However, no significant differences were found in the contents of
B1R, IL-6 and IL-10 in plaques from stroke patients of <30 days
and TIA groups; stroke patients of >180 days compared to
asymptomatic group.
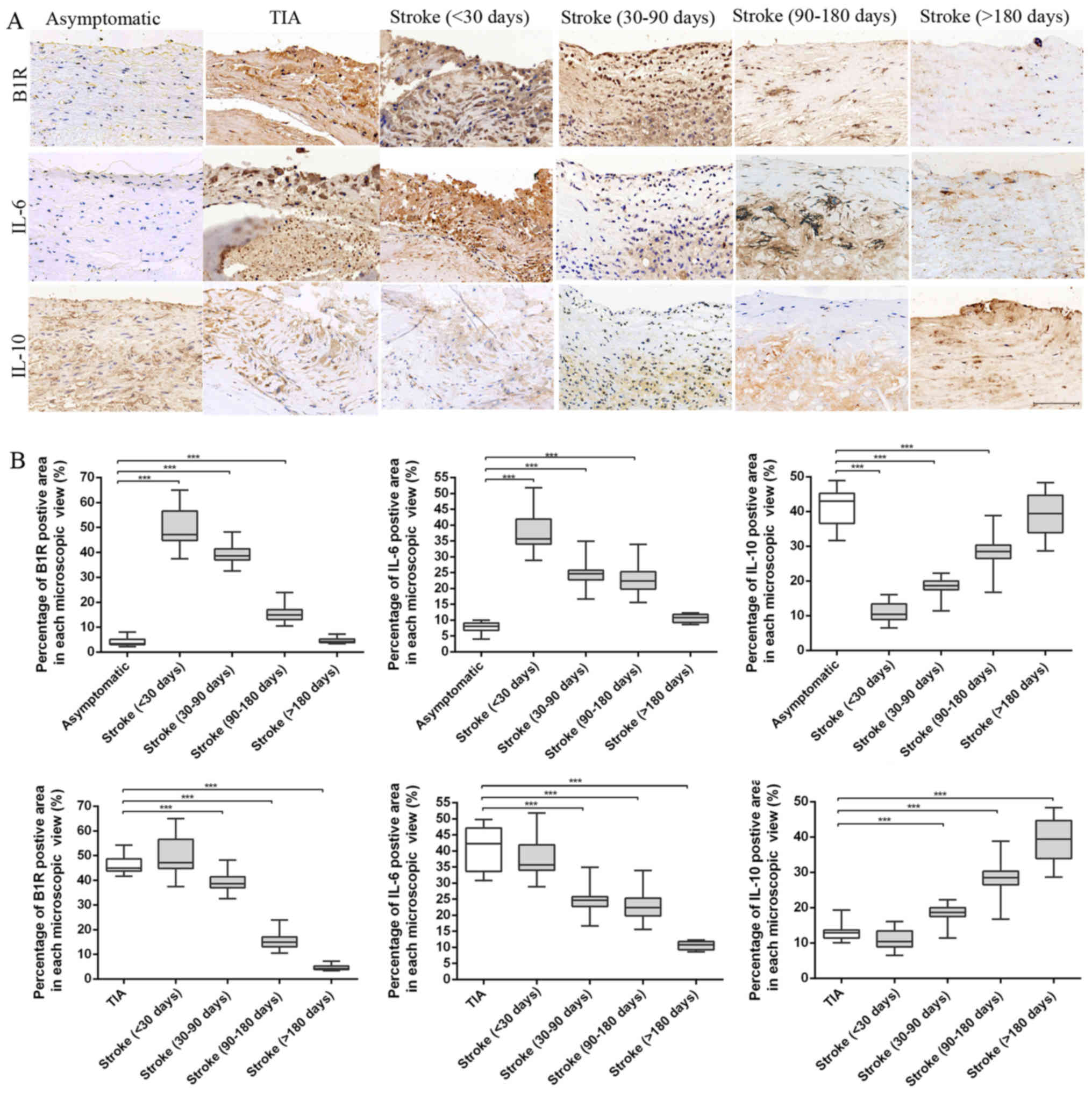 | Figure 3.Immunohistochemical results. (A)
Representative immunohistochemical images of B1R, IL-6 and IL-10 in
plaques of asymptomatic, TIA and stroke patients of <30, 30–90,
90–180 and >180 days. (B) Quantification of immunohistochemistry
analysis of B1R, IL-6 and IL-10 staining of section from
asymptomatic, TIA and stroke patients of <30, 30–90, 90–180 and
>180 days. Percentage of positive staining area in each
microscopic view (at 200x) showed that stroke plaques experienced
initial increases of B1R and IL-6 contents at <30 days, and
subsequent reduction from 30–90 to >180 days. However, a
noticeable increase in IL-10 contents was found in stroke plaques
from <30 to >180 days. (***P<0.001 vs. asymptomatic or TIA
group). Scale bar: 100 µm. |
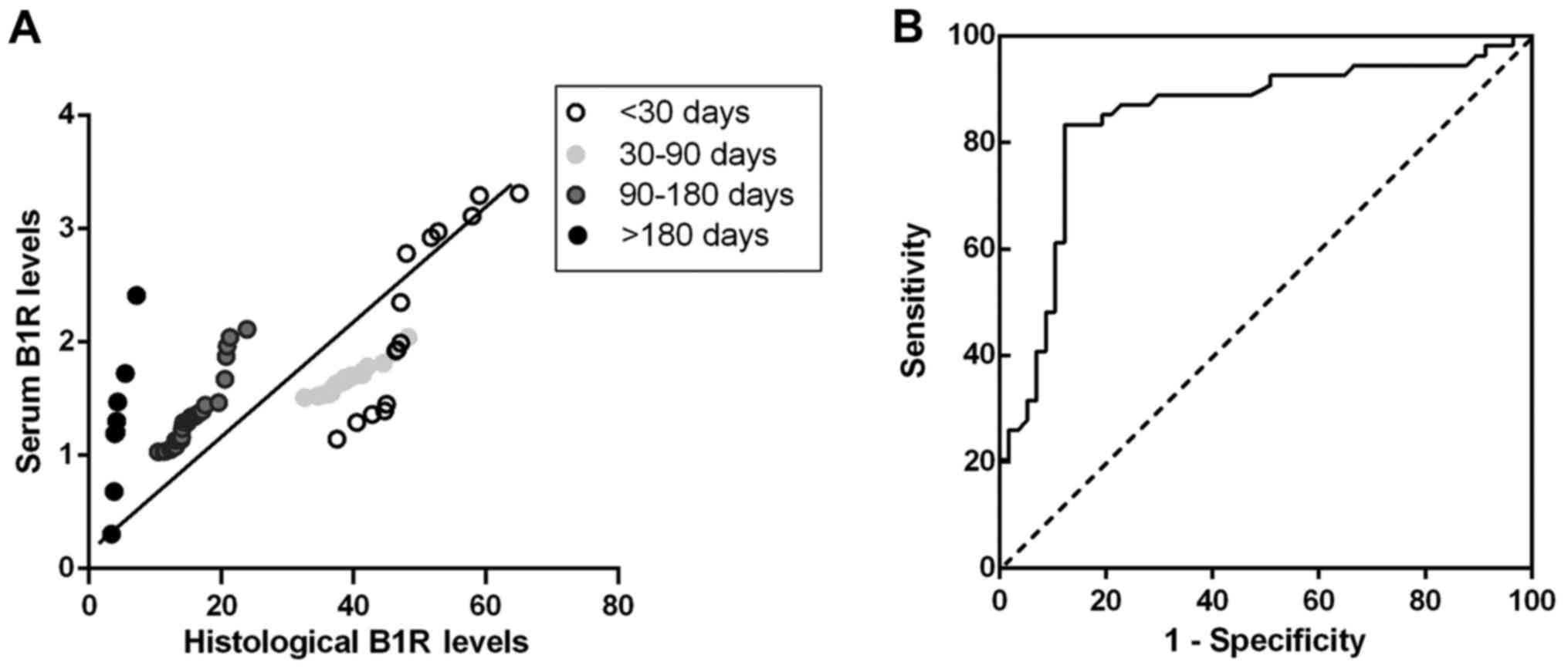
Correlation analysis between serum and
histological inflammatory marker levels
Correlation analysis between serum inflammation
marker and histological levels were performed in the plaques from
stroke, TIA and asymptomatic patients to test whether B1R, IL-6 and
IL-10 in serum level can indicate plaque instability. Spearman
correlation test showed that there was a close and highly
significant relationship between serum level of B1R with
histological staining of B1R in stroke patients (r=0.54,
P<0.001) (Fig. 4A), TIA
(r=0.81, P<0.001) and asymptomatic patients (r=0.61,
P<0.001), suggesting of a notable correlation between changes in
serum B1R level and changes in plaques in the progression of
atherosclerotic plaques. However, serum IL-6, IL-10 levels have
non-significant correlations with histological levels in plaques of
stroke (r=0.15, P>0.05; r=0.13, P>0.05), TIA (r= −0.29,
P>0.05; r=0.02, P>0.05) and asymptomatic patients (r=0.08,
P>0.05; r=0.01, P>0.05). These results suggest that patients
with carotid atherosclerotic plaques share uniform correlations
between serum and histological levels of B1R in the remodeling
progression of unstable plaques.
Diagnostic efficiency of serum B1R
level on carotid plaque
From our correlation analysis, it seems that serum
B1R level had significantly positive and uniform correlation with
histological level in plaque which is closely associated with the
progression and instability of plaque. Receiver-operating curve of
serum B1R level was used for predicting vulnerable plaques of
patients. Because stroke patients of different stages represented
dynamic changes of vulnerable plaques, only the data from stroke
patients were included in the analysis. After stroke, the area
under the curve was 0.85 (95% confidence interval: 0.77 to 0.93;
P<0.001). A B1R cutoff level of 1.49 pg/ml distinguished
vulnerable plaques from stable plaques with 83.3% sensitivity and
87.7% specificity (Fig. 4B).
Discussion
To provide basic evidence regarding the serum
markers in patients with carotid atherosclerotic plaque, we studied
the serum inflammation biomarkers and atherosclerotic plaque
composition in stroke patients of varying stages. We found the
remodeling progression of vulnerable atherosclerotic plaques over
time after stroke with a significant decrease in macrophage content
and increase in smooth muscle cells. By comparing inflammation
markers in serum levels, vulnerable plaques were distinguished from
stable ones before histological changes. The serum biomarkers of
IL-6 and B1R tended to decline while IL-10 tended to increase over
time after stroke. And, the histological levels of BIR, IL-6 and
IL-10 showed similar dynamic patterns as serum level changes. In
addition, serum B1R levels were significantly associated with
histological levels in all patients with carotid atherosclerotic
plaque. These results suggest that serum B1R changes can reveal
unstable plaques and indicate progression of plaques after ischemic
cerebrovascular events. To the best of our knowledge, this is the
first study that details the dynamic changes of inflammation
biomarkers in both serum and histological levels after stroke and
explores their utility as prognostic biomarker in development of
carotid atherosclerotic plaques.
Growing evidence has shown that atherosclerotic
carotid plaques from symptomatic patients reveal a vulnerable
phenotype early after acute ischemic event (15) and then remodel into stable ones
over time (8). This period often
involves several pathophysiological variables and complex
interactions, including local and systematic inflammation
alterations (16). Elucidation of
the inflammatory alterations can facilitate early vulnerable
plaques diagnosis. However, most patients already have late-stage
plaque rupture at diagnosis and then developed an acute ischemic
event because of the prior absence of featured clinical signs;
thus, it is impractical to explore vulnerable plaque-related
pathophysiological changes in patients. Plaques obtained from
stroke patients of different stages facilitate the study of a
serial of pathological changes in the progression of vulnerable
plaques. In this study, plaques from stroke patients showed a
significant reduction of macrophage content and a clear trend of
increasing numbers of smooth muscle cells from <30 to >180
days. However, plaque from stroke of <30 and 30–90 days showed
unstable phenotype and then tended to stable ones at stroke of
90–180 and >180 days. Thus, based on these findings, plaques
from stroke patients of four stages used in this study can mimic
the progression of vulnerable plaques in clinical.
Using ELISA, we showed noticeable increases in serum
inflammatory markers of IL-6 and B1R in stroke patients from <30
to 180 days. Conversely, IL-10 reduced in stroke patients from
<30 to >180 days in serum levels. There are several causes
explaining the inflammatory changes of plaques observed after
stroke. Because circulating biomarkers reliably predict the
vascular events, the serum inflammatory biomarkers as surrogates
that could reflect processes associated with atherosclerotic
disease progression (17). IL-6
has pro-inflammatory properties by activating and recruiting
inflammatory cells and has been reported to be higher in local
plaques from patients (18). IL-6
are also reported elevated in patients with acute coronary
syndromes and may contribute to the exacerbation of atherosclerosis
(19,20). By contrast, IL-10, an
anti-inflammatory marker can control inflammation by inhibiting the
synthesis of several cytokines produced by macrophages (21). Kamaly et al also showed
controlled-release polymeric nanoparticles incorporating the
anti-inflammatory cytokine IL-10 can be an effective treatment for
atherosclerotic plaques in advanced lesions of high fat-fed
low-density lipoprotein receptor-/-mice (22). However, it must be noted that the
dynamic changes of inflammatory biomarkers on both serum and
histological levels from stroke patients with different stages of
plaques compared with asymptomatic controls were found in the
present study. Thus, it is required to learn the dynamic changes in
inflammatory biomarkers when using serum biomarkers to monitor
progression in vulnerable plaques.
The most striking and important aspect of our
findings is that serum B1R change in stroke patients over time,
increasing as disease progresses. Kinins are pro-inflammatory
peptides that are released during tissue injury (23). The effects of kinins are mediated
by B1 and B2 receptor (B1R and B2R) and comprise induction of edema
formation and release of pro-inflammatory mediators (24). Austinat et al reported that
B1R knockout mice developed significantly smaller brain infarctions
and less neurological deficits compared to wild-type controls
(25). In our study, it is true
that serum and histological B1R levels were highest in stroke
patients of <30 days compared with other groups. Besides
detailing dynamic changes in serum and histological inflammatory
markers, we also explored the association of serum changes with
histological in patients with carotid atherosclerotic plaques. From
our correlation analysis, it seems that the serum levels of B1R
remains closely related with histological levels even in patients
with asymptomatic plaques. In this respect, B1R is currently the
potential biochemical biomarker in serum of atherosclerosis disease
progression. Since patients comfort and compliance are important
considerations, obtaining serum for B1R quantification may be more
practical than the more invasive option of surgery for plaques.
Thus, B1R may have utility as a potential pharmacodynamic biomarker
insofar as showing that an experimental therapeutic that the
changes of B1R in serum would provide evidence of underlying
biological effect of treatment.
We acknowledge that our study has several
limitations. First, drug use for patients associated with the
different time intervals may determine possible confounding
effects. However, no differences between the groups have been
observed, indicating that this confounder could not explain the
results. Second, our findings are based on a small number of
different samples and our data need confirmation in larger studies
in other populations at risk. However, because nowadays patients
are being operated much faster after the event, it is hard to
recruit larger number of samples to confirm in this study. Third,
although HE staining was used as a gold standard for the diagnosis
of vulnerable plaques, other techniques such as MRI or
18F-FDG-PET/CT can be used to further monitor the
unstable plaque in these patients (26,27).
In summary, carotid atherosclerosis is a progression
with the upregulation of pro-inflammatory factors and the
downregulation of anti-inflammatory factors leading to plaque
destabilization, which may eventually result in acute
cerebrovascular events. This study demonstrates that the remodeling
and progression of vulnerable plaques after stroke is significantly
associated with the decrease of serum inflammatory factors IL-6 and
B1R and an increase of anti-inflammatory marker-IL-10 levels. In
particular, serum B1R represent the most promising potential
pharmacodynamic biomarkers available today that are suitable for
further investigation in the context of future clinical trials.
Acknowledgements
The present study work was supported by funding from
the National Natural Science Foundation of China (81371435;
81671299), the Hunan Natural Science Foundation (2016JC2057) and
the Xiangya Hospital foundation (xywm2015I32).
References
|
1
|
Rothstein L and Jickling GC: Ischemic
stroke biomarkers in blood. Biomark Med. 7:37–47. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mauriello A, Servadei F, Sangiorgi G,
Anemona L, Giacobbi E, Liotti D and Spagnoli LG: Asymptomatic
carotid plaque rupture with unexpected thrombosis over a
non-canonical vulnerable lesion. Atherosclerosis. 218:356–362.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Stefanadis C, Antoniou CK, Tsiachris D and
Pietri P: Coronary atherosclerotic vulnerable plaque: Current
perspectives. J Am Heart Assoc. 6:pii: e0055432017. View Article : Google Scholar
|
|
4
|
Tuttolomondo A, Pecoraro R, Casuccio A, Di
Raimondo D, Buttà C, Clemente G, Corte V Della, Guggino G, Arnao V,
Maida C, et al: Peripheral frequency of CD4+ CD28- cells in acute
ischemic stroke: Relationship with stroke subtype and severity
markers. Medicine (Baltimore). 94:e8132015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tuttolomondo A, Pedone C, Pinto A, Di
Raimondo D, Fernandez P, Di Sciacca R and Licata G: Gruppo Italiano
di Farmacoepidemiologia dell'Anziano (GIFA) researchers: Predictors
of outcome in acute ischemic cerebrovascular syndromes: The GIFA
study. Int J Cardiol. 125:391–396. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ammirati E, Moroni F, Norata GD, Magnoni M
and Camici PG: Markers of inflammation associated with plaque
progression and instability in patients with carotid
atherosclerosis. Mediators Inflamm. 2015:7183292015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shindo A, Tanemura H, Yata K, Hamada K,
Shibata M, Umeda Y, Asakura F, Toma N, Sakaida H, Fujisawa T, et
al: Inflammatory biomarkers in atherosclerosis: Pentraxin 3 can
become a novel marker of plaque vulnerability. PLoS One.
9:e1000452014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Peeters W, Hellings WE, de Kleijn DP, de
Vries JP, Moll FL, Vink A and Pasterkamp G: Carotid atherosclerotic
plaques stabilize after stroke: Insights into the natural process
of atherosclerotic plaque stabilization. Arterioscler Thromb Vasc
Biol. 29:128–133. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Roquilly A, Lejus C and Asehnoune K: Brain
injury, immunity and infections. Ann Fr Anesth Reanim. 31:e97–e100.
2012.(In French). View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Golias C, Charalabopoulos A, Stagikas D,
Charalabopoulos K and Batistatou A: The kinin system-bradykinin:
Biological effects and clinical implications. Multiple role of the
kinin system-bradykinin. Hippokratia. 11:124–128. 2007.PubMed/NCBI
|
|
11
|
Leeb-Lundberg LM, Marceau F, Müller-Esterl
W, Pettibone DJ and Zuraw BL: International union of pharmacology.
XLV. Classification of the kinin receptor family: From molecular
mechanisms to pathophysiological consequences. Pharmacol Rev.
57:27–77. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Medeiros R, Cabrini DA, Ferreira J,
Fernandes ES, Mori MA, Pesquero JB, Bader M, Avellar MC, Campos MM
and Calixto JB: Bradykinin B1 receptor expression induced by tissue
damage in the rat portal vein: A critical role for
mitogen-activated protein kinase and nuclear factor-kappaB
signaling pathways. Circ Res. 94:1375–1382. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Duchene J, Cayla C, Vessillier S, Scotland
R, Yamashiro K, Lecomte F, Syed I, Vo P, Marrelli A, Pitzalis C, et
al: Laminar shear stress regulates endothelial kinin B1 receptor
expression and function: Potential implication in atherogenesis.
Arterioscler Thromb Vasc Biol. 29:1757–1763. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Guo Y, Liu T, Li X, Zhang M, Shi L and Liu
H: Expression of the genes encoding kinin receptors are increased
in human carotid atherosclerotic plaques. Biomed Rep. 3:398–402.
2015.PubMed/NCBI
|
|
15
|
Hatsukami TS, Ferguson MS, Beach KW,
Gordon D, Detmer P, Burns D, Alpers C and Strandness DE Jr: Carotid
plaque morphology and clinical events. Stroke. 28:95–100. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Marnane M, Prendeville S, McDonnell C,
Noone I, Barry M, Crowe M, Mulligan N and Kelly PJ: Plaque
inflammation and unstable morphology are associated with early
stroke recurrence in symptomatic carotid stenosis. Stroke.
45:801–806. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen YC, Huang AL, Kyaw TS, Bobik A and
Peter K: Atherosclerotic plaque rupture: Identifying the straw that
breaks the camel's back. Arterioscler Thromb Vasc Biol. 36:e63–e72.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lindmark E, Diderholm E, Wallentin L and
Siegbahn A: Relationship between interleukin 6 and mortality in
patients with unstable coronary artery disease: Effects of an early
invasive or noninvasive strategy. JAMA. 286:2107–2113. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Poredos P, Spirkoska A, Lezaic L, Mijovski
MB and Jezovnik MK: Patients with an inflamed atherosclerotic
plaque have increased levels of circulating inflammatory markers. J
Atheroscler Thromb. 24:39–46. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Schieffer B, Selle T, Hilfiker A,
Hilfiker-Kleiner D, Grote K, Tietge UJ, Trautwein C, Luchtefeld M,
Schmittkamp C, Heeneman S, et al: Impact of interleukin-6 on plaque
development and morphology in experimental atherosclerosis.
Circulation. 110:3493–3500. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ramakrishnan V, Akram HR and Ahmed SS:
Genetic predisposition of IL-10 promoter polymorphisms with risk of
multiple sclerosis: A meta-analysis. J Neuroimmunol. 306:11–18.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kamaly N, Fredman G, Fojas JJ, Subramanian
M, Choi WI, Zepeda K, Vilos C, Yu M, Gadde S, Wu J, et al: Targeted
interleukin-10 nanotherapeutics developed with a microfluidic chip
enhance resolution of inflammation in advanced atherosclerosis. ACS
Nano. 10:5280–5292. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kuhr F, Lowry J, Zhang Y, Brovkovych V and
Skidgel RA: Differential regulation of inducible and endothelial
nitric oxide synthase by kinin B1 and B2 receptors. Neuropeptides.
44:145–154. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kahn R, Mossberg M, Ståhl AL, Johansson K,
Lindman I Lopatko, Heijl C, Segelmark M, Mörgelin M, Leeb-Lundberg
LM and Karpman D: Microvesicle transfer of kinin B1-receptors is a
novel inflammatory mechanism in vasculitis. Kidney Int. 91:96–105.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Austinat M, Braeuninger S, Pesquero JB,
Brede M, Bader M, Stoll G, Renné T and Kleinschnitz C: Blockade of
bradykinin receptor B1 but not bradykinin receptor B2 provides
protection from cerebral infarction and brain edema. Stroke.
40:285–293. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chan JM, Monaco C, Wylezinska-Arridge M,
Tremoleda JL and Gibbs RG: Imaging of the vulnerable carotid
plaque: Biological targeting of inflammation in atherosclerosis
using iron oxide particles and MRI. Eur J Vasc Endovasc Surg.
47:462–469. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Rudd JH, Narula J, Strauss HW, Virmani R,
Machac J, Klimas M, Tahara N, Fuster V, Warburton EA, Fayad ZA and
Tawakol AA: Imaging atherosclerotic plaque inflammation by
fluorodeoxyglucose with positron emission tomography: Ready for
prime time? J Am Coll Cardiol. 55:2527–2535. 2010. View Article : Google Scholar : PubMed/NCBI
|