Introduction
Osteoarthritis (OA) is a common chronic degenerative
joint disease and is a leading cause of pain and disability in the
adult population. In Asian countries, the incidence of OA in
individuals aged >65 years, may increase from 6.8% in 2008 to
16.2% in 2040 (1). Risk factors of
OA may be divided into person-level factors (age, gender, obesity,
genetics and diet) and joint-level factors (injury, malalignment
and abnormal loading of the joints), which interact in a complex
manner (2). It is characterized by
bone remodeling, synovium inflammation and cartilage loss (3). Although the management of OA has been
diverse, including pharmacological therapy treatment options,
surgical interventions and orthopedic procedures (4,5),
there are no effective drug treatments that are able to reverse
disease progression (6,7). Therefore, the development of new
therapeutic strategies that are effective and safe for OA treatment
are required.
OA pathogenesis is complex and involves the
interaction of numerous factors, and an increasing number of
studies have suggested that inflammation serves a key role in the
pathogenesis of OA (8–10). Chondrocytes secrete
pro-inflammatory cytokines, such as interleukin-1β (IL-1β) and
tumor necrosis factor-α (TNF-α), that may contribute to the
progression of OA (11). IL-1β was
reported to enhance the production of matrix metalloproteinases
(MMPs) and to inhibit the synthesis of extracellular matrix (ECM),
thus contributing the progression of OA (12,13).
Carvacrol is a monoterpenic phenol that is present
in Origanum vulgare (oregano) and Thymus vulgaris
(thyme), which has been demonstrated to possess a spectrum of
pharmacological activities, including antioxidative, analgesic,
antihepatotoxic, antimicrobial and antitumoral (14,15).
In addition, a previous study confirmed its anti-inflammatory
property. For example, carvacrol was reported to inhibit the levels
of inflammatory cytokines and the expression of inducible nitric
oxide synthase (iNOS) and cyclooxygenase (COX)-2 in ischemic
cortical tissues (16). However,
the effects and underlying mechanism of carvacrol on chondrocytes
in OA remain unknown. The present study aimed to investigate the
protective effects of carvacrol against inflammation in
IL-1β-stimulated human chondrocytes, and the results demonstrated
that carvacrol pretreatment inhibited IL-1β-induced nitric oxide
(NO) and prostaglandin E2 (PGE2) production, and reduced the
expression levels of iNOS, COX-2 and MMPs in human OA chondrocytes
by suppressing the activation of the NF-κB signaling pathway. Thus,
carvacrol may provide a potential therapeutic function for the
treatment of OA.
Materials and methods
Normal human articular cartilage
chondrocyte culture and treatment
Normal human articular cartilage was obtained from
eight patients (4 men, 4 women; age 24–41 years) after death or
from trauma in Tianjin Hospital (Tianjin, China). The normal donors
were significantly younger whose articular cartilage had no
degenerative changes. Written informed consent was obtained from
the patients according to the terms of the Ethics Committee of
Tianjin Hospital. Chondrocytes were isolated from cartilage as
previously described (17).
Briefly, cartilage fragments were digested with 0.25% trypsin for
15 min and incubated with 0.2% (v/v) collagenase for 4 h at 37°C.
The resulting cells were maintained in Dulbecco's modified Eagle's
medium (DMEM) supplemented with 10% fetal bovine serum (both from
HyClone; GE Healthcare Life Sciences, Logan, UT, USA), 100 U/ml
penicillin and 100 µg/ml streptomycin (Sigma-Aldrich; Merck KGaA,
Darmstadt, Germany) at 37°C and 5% CO2 in a humidified
incubator.
Human chondrocytes (1×105 cells/well)
were pretreated with various concentrations of carvacrol (0, 1, 5
and 10 µg/ml; Sigma-Aldrich; Merck KGaA) for 2 h and then
co-incubated in the absence or presence of IL-1β (10 ng/ml;
Sigma-Aldrich; Merck KGaA) for 24 h at 37°C.
Cell viability assay
Cell viability was measured by the MTT assay.
Briefly, following treatments, human chondrocytes (1×105
cells/well) were incubated with MTT solution (5 mg/ml;
Sigma-Aldrich; Merck KGaA) at 37°C for 4 h; subsequently, the
purple formazan crystals were dissolved using dimethyl sulfoxide by
shaking at room temperature for 10 min. Spectrophotometric
absorbance was measured at 570 nm using a multifunctional
microplate reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
All experiments were repeated at least three times.
Measurement of NO and PGE2 levels
Nitrite levels in the culture medium were detected
by the Griess reaction as previously described (18). The level of PGE2 was evaluated
using a ELISA kit (cat no. KHL1701; Invitrogen; Thermo Fisher
Scientific, Inc., Waltham, MA, USA), according to the manufacture's
protocol. All experiments were repeated at least three times.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from human chondrocytes
(1×106 cells/well) using TRIzol reagent (Invitrogen;
Thermo Fisher Scientific, Inc.), according to the manufacturer's
instructions. First-strand cDNA was synthesized from total RNA (1
µg) using the PrimeScript RT Reagent kit with gDNA Eraser (Takara
Bio, Inc., Otsu, Japan). Subsequently, a 7500 Real-Time PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.) was used to
conduct RT-qPCR using SYBR Premix Ex Taq II (Takara Bio, Inc.).
Primers used in qPCR are listed as follows: iNOS forward,
5′-TTTCCAAGACACACTTCACCA-3′ and reverse,
5′-ATCTCCTTTGTTACCGCTTCC−3′; COX-2 forward,
5′-GAGAGATGTATCCTCCCACAGTCA-3′ and reverse,
5′-GACCAGGCACCAGACCAAAG-3′; MMP-3 forward,
5′-GCATTGGCTGAGTGAAAGAGACTGTATC-3′ and reverse,
5′-ATGATGAACGATGGACAGATGA-3′; MMP-13 forward,
5′-AGTAGTTCCAAAGGCTACAACTTGTTT-3′ and reverse,
5′-GGAGTGGTCAAGCCCTAAGGA-3′; GAPDH forward, 5′-CTGGGCTACACTGAGCA-3′
and reverse, 5′-AAGTGGTCGTTGAGGGCAATG−3′. GAPDH was used as a
reference gene. The PCR amplification cycles were performed as
follows: 30 sec at 95°C, followed by 40 cycles of 5 sec at 95°C and
30 sec at 60°C. The 2−ΔΔCt method (19) was used to calculate relative
changes in gene expression. All experiments were repeated at least
three times.
Western blot analysis
Human chondrocytes (1×106 cells/well)
were lysed in radioimmunoprecipitation assay buffer supplemented
with protease and phosphatase inhibitor mixtures (Sigma-Aldrich;
Merck KGaA). Protein concentrations were determined using a
Bradford assay (Bio-Rad Laboratories, Inc.). Protein lysates (30
µg) were separated by 10% SDS-PAGE and transferred to
polyvinylidene fluoride membranes (both from Bio-Rad Laboratories,
Inc.). Membranes were blocked in 5% non-fat milk for 2 h at room
temperature and then incubated with rabbit anti-human iNOS (1:500;
ab3523), COX-2 (1:500; ab52237), MMP-3 (1:500; ab53015), MMP-13
(1:3,000; ab39012), NF-κB p65 (1:50,000; ab32536) and IκBα primary
antibodies (1:1,000; ab32518; all from Abcam, Cambridge, UK)
overnight at 4°C. Following washing with TBS containing 0.1%
Tween-20 (TBST; Sigma-Aldrich; Merck KGaA), membranes were
incubated with horseradish peroxidase-conjugated secondary goat
anti-rabbit immunoglobulin G antibodies (1:1,000; sc-2922; Santa
Cruz Biotechnology, Inc., Dallas, TX, USA) at room temperature for
1 h. Membranes were washed with TBST buffer, and immunoreactivity
was detected with Enhanced Chemiluminescence reagent (GE Healthcare
Life Sciences) and quantified by the Quantity One (Bio-Rad
Laboratories, Inc.) version 5.2 software. β-actin was used as the
internal control. All experiments were repeated at least three
times.
Statistical analysis
Statistical analyses were performed using SPSS 13.0
software (SPSS, Inc., Chicago, IL, USA). Data are expressed as the
mean ± standard deviation. One-way analysis of variance followed by
Newman-Keuls post-hoc test was used for the statistical comparison
of multiple groups. Results from two groups were evaluated using
Student's t-test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Effects of carvacrol on human OA
chondrocyte viability
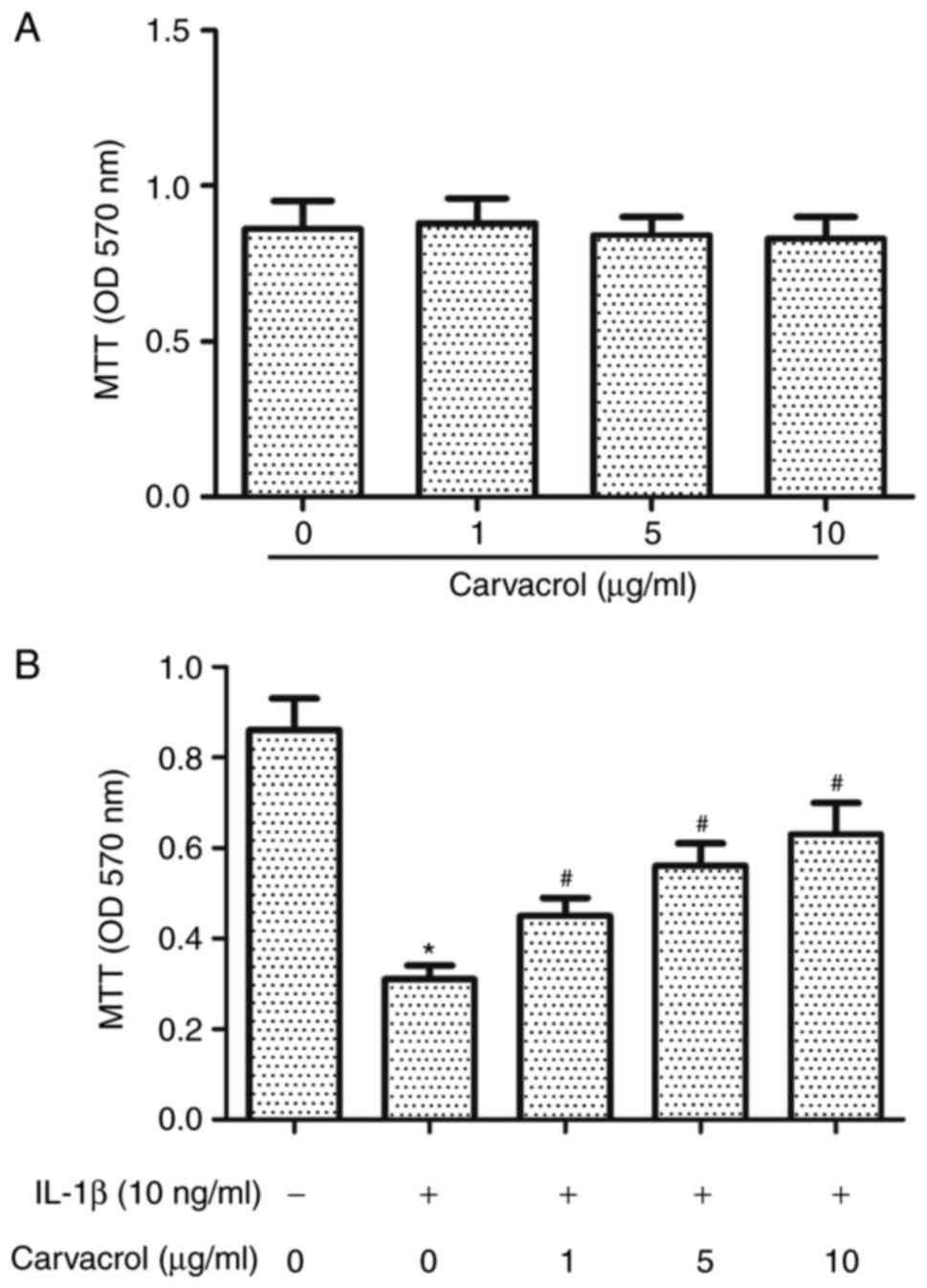
Carvacrol cytotoxicity on chondrocyte viability was
examined by MTT assay. Compared with untreated chondrocytes, the
various treatments with carvacrol at concentrations between 1 and
10 µg/ml did not significantly affect cell viability (Fig. 1A). Treatment with IL-1β (10 ng/ml)
significantly reduced cell viability (Fig. 1B); whereas, pretreatment with
carvacrol reversed the effects of IL-1β in a
concentration-dependent manner (Fig.
1B). The highest inhibition was observed with 10 µg/ml
carvacrol treatment. This concentration of carvacrol was used in
the following experiment.
Carvacrol inhibits IL-1β-induced NO
and PGE2 production in OA chondrocytes
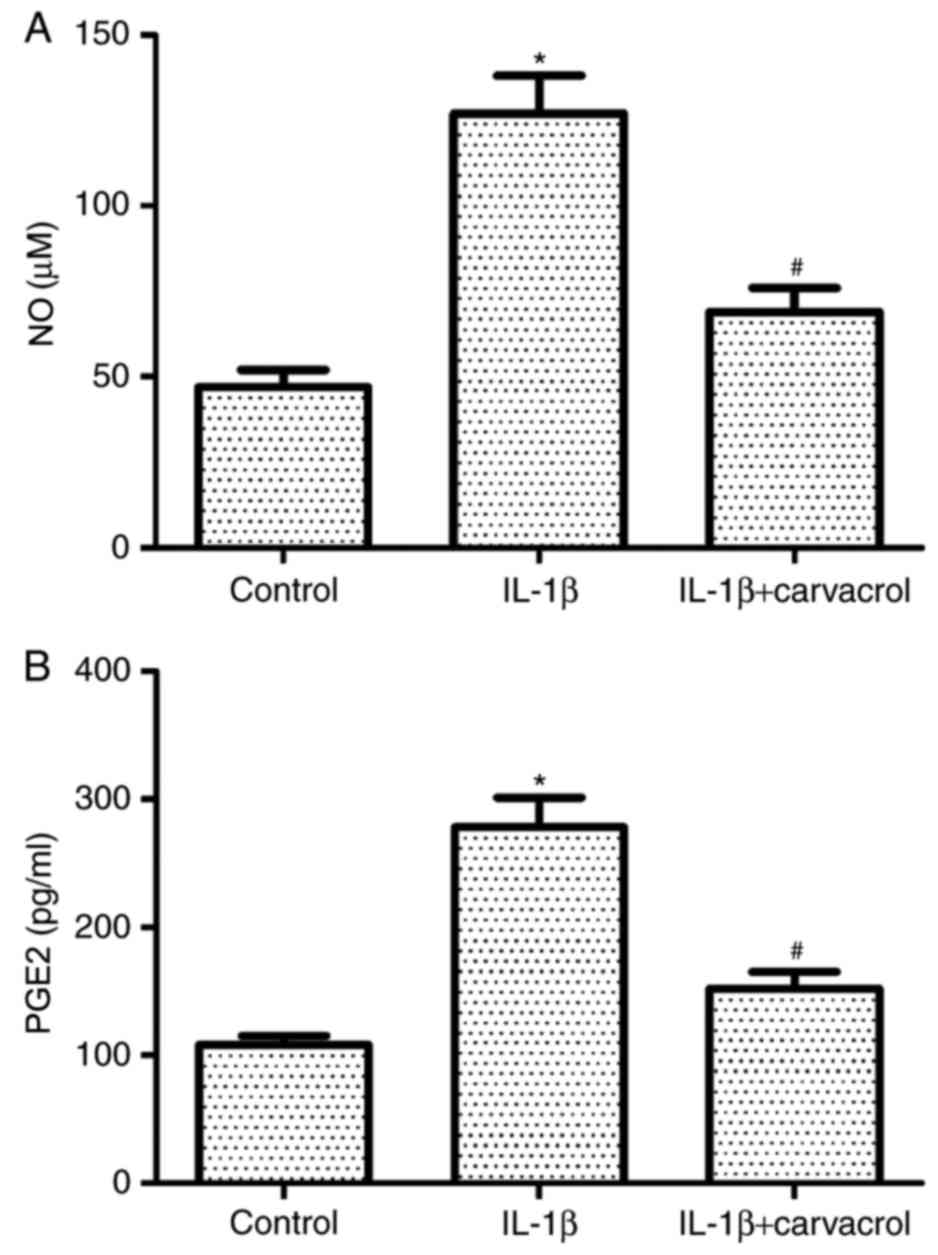
The effects of carvacrol on NO and PGE2 production
in IL-1β-induced chondrocytes were also investigated. IL-1β
treatment significantly induced the production of NO and PGE2 in OA
chondrocytes (Fig. 2), and these
increased levels of expression were significantly inhibited in
cells co-treated with carvacrol.
Carvacrol inhibits IL-1β-induced iNOS
and COX-2 expression in OA chondrocytes
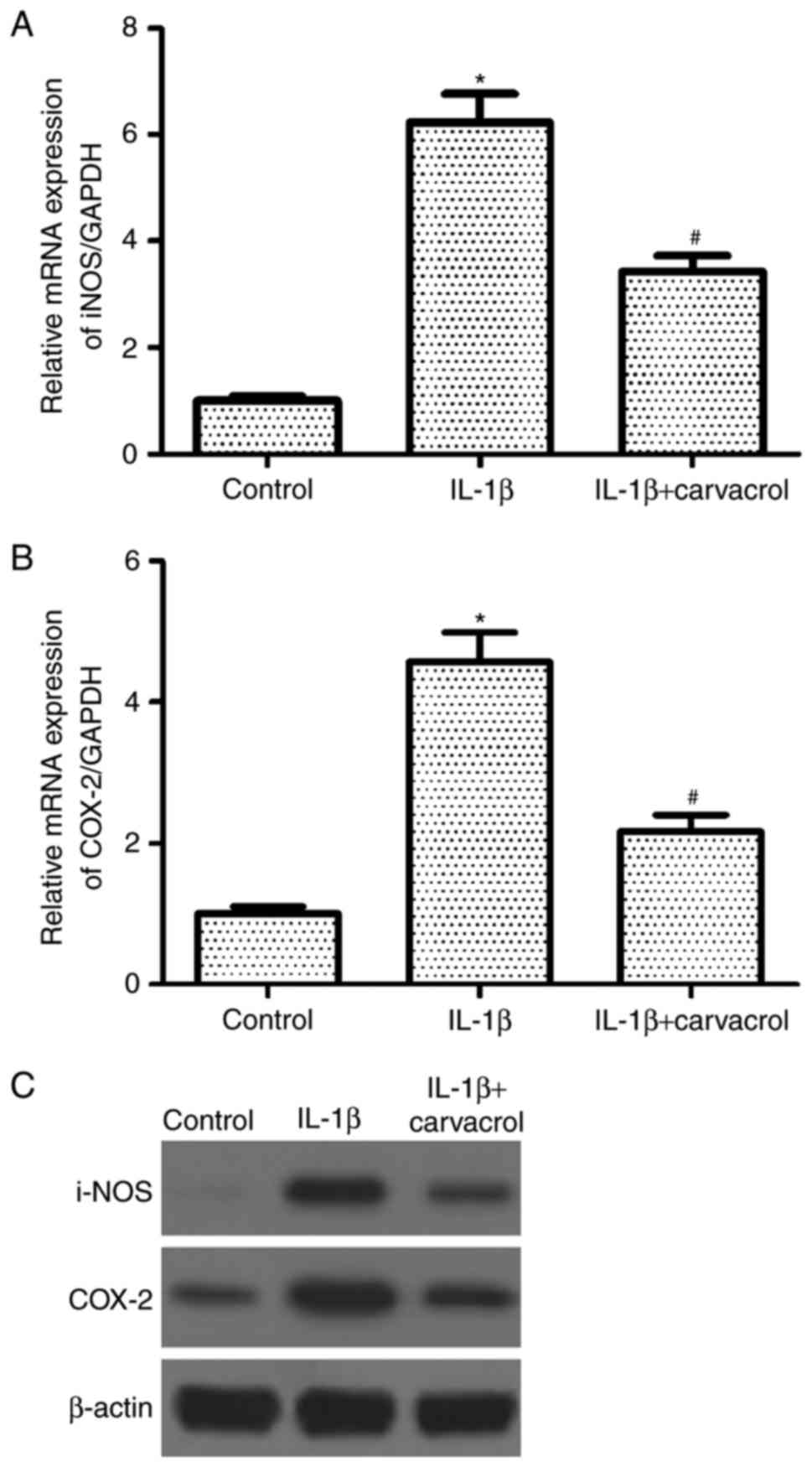
Western blot analysis was used to determine the
effects of carvacrol on iNOS and COX-2 expression in human
chondrocytes stimulated with IL-1β. The mRNA and protein expression
levels of iNOS and COX-2 were markedly increased following IL-1β
incubation compared with untreated controls (Fig. 3). By contrast, chondrocytes that
were co-treated with carvacrol exhibited a notable decrease in iNOS
and COX-2 expressions compared with IL-1β-treated OA
chondrocytes.
Carvacrol inhibits IL-1β-induced MMP-3
and MMP-13 expression in OA chondrocytes
A number of studies have demonstrated that MMPs
serve crucial roles in the initiation and progression of OA
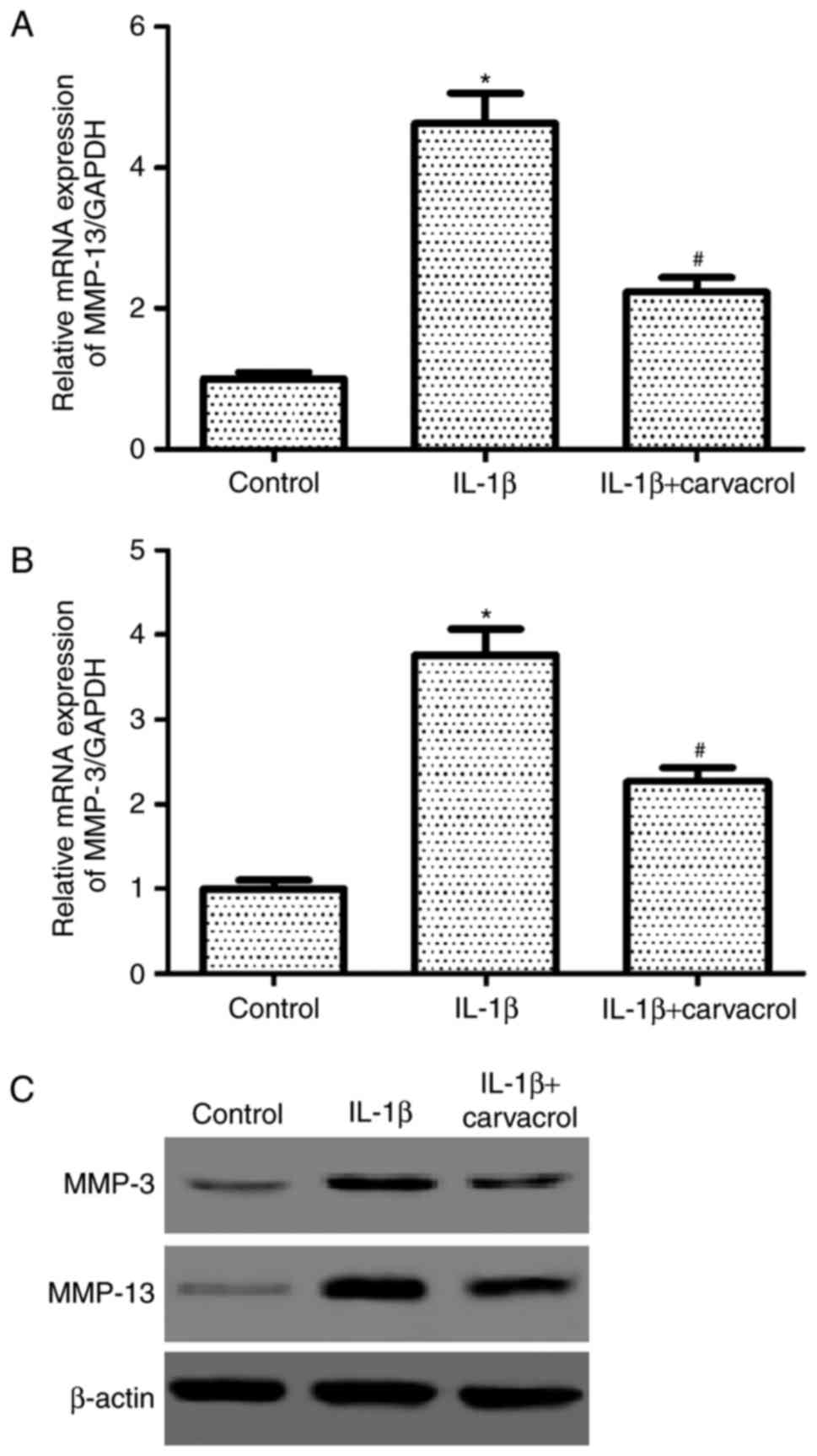
(20–22). Therefore, the effects of carvacrol
on MMP-3 and MMP-13 expression in IL-1β-induced chondrocytes were
examined. RT-qPCR analysis results demonstrated that the mRNA
expression levels of MMP-13 and MMP-3 generated in IL-1β-induced
chondrocytes increased significantly compared with controls
(Fig. 4A and B, respectively).
However, pretreatment with carvacrol greatly inhibited these
IL-1β-induced effects. Similarly, western blot analysis
demonstrated that carvacrol was able to suppress IL-1β-induced
MMP-3 and MMP-13 protein expression in OA chondrocytes (Fig. 4C).
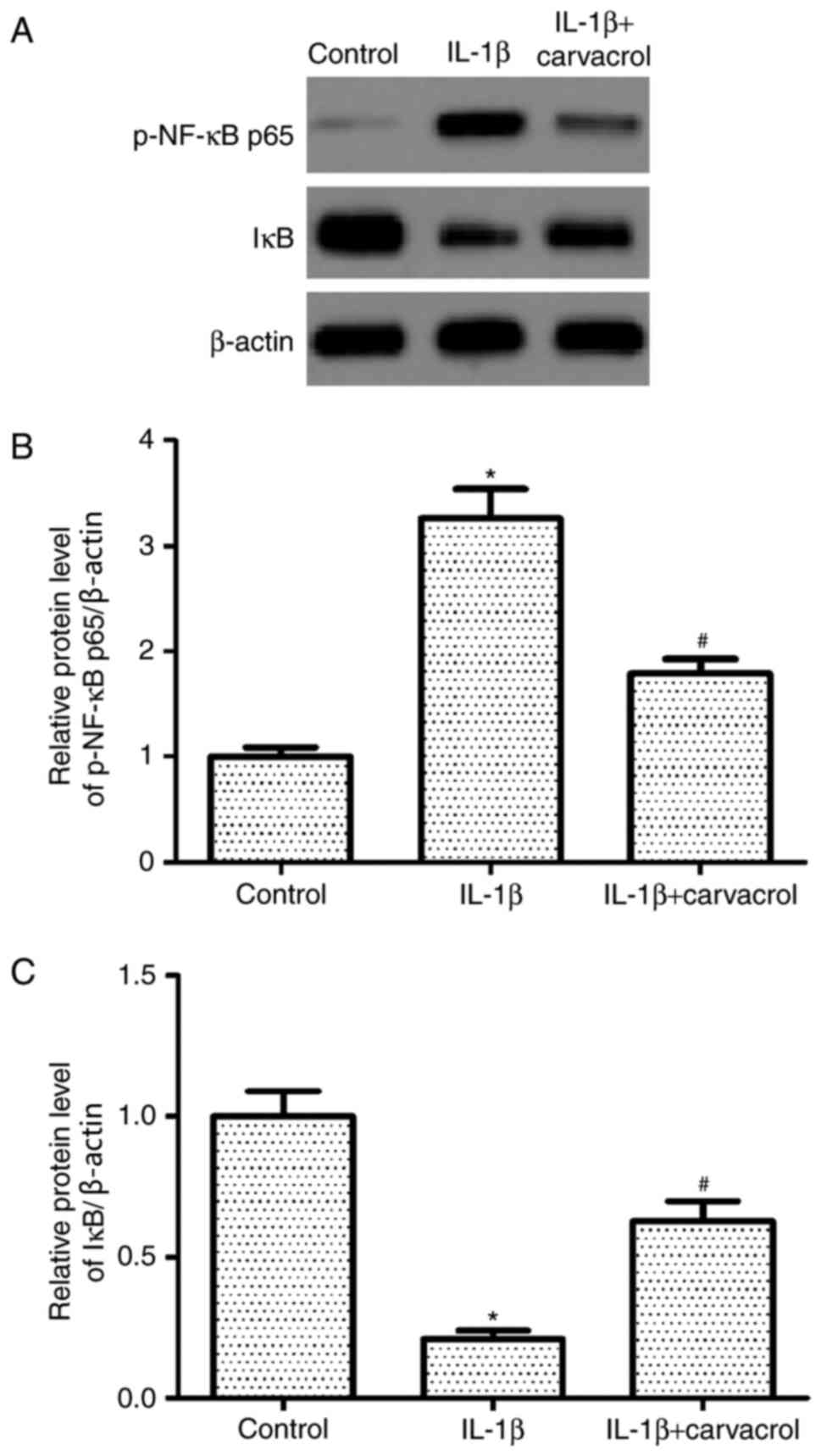
Carvacrol inhibits the activation of
nuclear factor (NF)-κB signaling pathway in chondrocytes
Activation of NF-κB signaling pathway has been
reported to participate in inflammation in OA (23). Therefore, the effects of carvacrol
on NF-κB activation in human chondrocytes stimulated with IL-1β
were investigated. Western blot analysis data revealed that IL-1β
treatment significantly increased the protein expression level of
phosphorylated-NF-κB p65 and reduced the protein expression level
of IκBα in chondrocytes compared with untreated chondrocytes
(Fig. 5). Notably, co-treatment
with carvacrol significantly decreased the IL-1β-induced expression
of NF-κB in chondrocytes and increased the protein expression level
of IκBα.
Discussion
A number of previous studies have reported that
carvacrol possesses anti-inflammatory effects (24–27).
Results from the present study indicated that carvacrol inhibited
NO and PGE2 production, as well as decreased iNOS and COX-2
expression. Carvacrol was also demonstrated to suppress the protein
expression levels of MMP-3 and MMP-13 in IL-1β-stimulated human OA
chondrocytes. Furthermore, carvacrol suppressed the activation of
NF-κB signaling pathway in IL-1β-induced human chondrocytes.
IL-1β treatment has been widely used to mimic the
microenvironment of OA in in vitro studies (28–30);
in the present study, IL-1β-induced human OA chondrocytes were used
as a model to investigate the protective effects of carvacrol on
human chondrocytes, and the results suggested that pretreatment
with carvacrol was able to reverse IL-1β-reduced cell
viability.
NO has been demonstrated to serve a pivotal role in
the development of OA (31). It is
produced by iNOS in several types of cells, including chondrocytes
(32). PGE2 is an inflammatory
mediator that is elevated by COX-2 (33). In addition, previous studies have
reported that IL-1β was able to induce iNOS and COX-2 expression in
chondrocytes, which led to elevated production of NO and PGE2,
respectively (34,35). The present study observed that
carvacrol treatment inhibited NO and PGE2 production, as well as
decreased iNOS and COX-2 expression in IL-1β-stimulated human OA
chondrocytes. These results are in agreement with previous studies,
which reported that carvacrol significantly downregulated the
expression levels of TNF-α, IL-6, iNOS and COX-2 in
D-galactosamine-induced hepatotoxic rats (36).
An increasing number of studies have indicated that
MMPs may also be involved in the progression of OA (22,37,38).
For example, MMP-3 was reported to induce inflammation by
activating various pro-MMPs and the cleavage of extracellular
components (39). MMP-13 serves a
crucial role in the degradation of collagens, proteoglycans and
other ECM macromolecules in cartilage (40). Additional studies have demonstrated
that IL-1β was able to upregulate the expression of MMPs in
chondrocytes (41–43). The present study observed that
carvacrol co-treatment suppressed IL-1β-induced MMP-3 and MMP-13
protein expression in OA chondrocytes. These results suggested that
carvacrol exhibited chondroprotective activity by downregulating
MMP expression in vitro.
The NF-κB signaling pathway serves an important role
in OA pathogenesis (44–46). Stimulation by inflammatory
mediators such as IL-1β leads to the phosphorylation and
degradation of the inhibitory subunit, which allows the active
NF-κB complex to translocate into nucleus and induced the
expression of various inflammation-related genes that regulate the
synthesis of cytokines, chemokines and adhesion molecules (47). It was reported previously that the
NF-κB inhibitor, pyrrolidine dithiocarbamate, decreased
IL-1β-induced MMP-3 and MMP-13 production in human chondrocytes
(44). A recent study using
ischemic cortical tissues confirmed that carvacrol treatment was
able to suppress the ischemia/reperfusion-induced increase in
nuclear NF-κB p65 protein expression in (16). Similarly, results from the present
study revealed that pretreatment with carvacrol significantly
inhibited IL-1β-induced NF-κB activation in OA chondrocytes. These
data suggested that carvacrol may inhibit IL-1β-induced
inflammation in chondrocytes by suppressing the activation of the
NF-κB signaling pathway.
In conclusion, the present results demonstrated that
carvacrol pretreatment was able to inhibit IL-1β-induced NO and
PGE2 production, as well as reduced the expression levels of iNOS,
COX-2, MMPs in human OA chondrocytes by suppressing the activation
of NF-κB signaling pathway. Thus, carvacrol may provide a potential
therapeutic function for the treatment of OA.
References
|
1
|
Miller ME, Rejeski WJ, Messier SP and
Loeser RF: Modifiers of change in physical functioning in older
adults with knee pain. The Observational Arthritis Study in Seniors
(OASIS). Arthritis Rheum. 45:331–339. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Palazzo C, Nguyen C, Lefevre-Colau MM,
Rannou F and Poiraudeau S: Risk factors and burden of
osteoarthritis. Ann Phys Rehabil Med. 59:134–138. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Loeser RF, Goldring SR, Scanzello CR and
Goldring MB: Osteoarthritis: A disease of the joint as an organ.
Arthritis Rheum. 64:1697–1707. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Pulsatelli L, Addimanda O, Brusi V,
Pavloska B and Meliconi R: New findings in osteoarthritis
pathogenesis: Therapeutic implications. Ther Adv Chronic Dis.
4:23–43. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Thysen S, Luyten FP and Lories RJ:
Targets, models and challenges in osteoarthritis research. Dis
Model Mech. 8:17–30. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lawrence RC, Felson DT, Helmick CG, Arnold
LM, Choi H, Deyo RA, Gabriel S, Hirsch R, Hochberg MC, Hunder GG,
et al: Estimates of the prevalence of arthritis and other rheumatic
conditions in the United States: Part II. Arthritis Rheum.
58:26–35. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cheng DS and Visco CJ: Pharmaceutical
therapy for osteoarthritis. PM R. 4 5 Suppl:S82–S88. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pelletier JP, Martel-Pelletier J and
Abramson SB: Osteoarthritis, an inflammatory disease: Potential
implication for the selection of new therapeutic targets. Arthritis
Rheum. 44:1237–1247. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Goldring MB and Otero M: Inflammation in
osteoarthritis. Curr Opin Rheum. 23:471–478. 2011. View Article : Google Scholar
|
|
10
|
Haywood L, McWilliams DF, Pearson CI, Gill
SE, Ganesan A, Wilson D and Walsh DA: Inflammation and angiogenesis
in osteoarthritis. Arthritis Rheum. 48:2173–2177. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
López-Armada MJ, Caramés B, Lires-Deán M,
Cillero-Pastor B, Ruiz-Romero C, Galdo F and Blanco FJ: Cytokines,
tumor necrosis factor-alpha and interleukin-1beta, differentially
regulate apoptosis in osteoarthritis cultured human chondrocytes.
Osteoarthritis Cartilage. 14:660–669. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mengshol JA, Vincenti MP, Coon CI,
Barchowsky A and Brinckerhoff CE: Interleukin-1 induction of
collagenase 3 (matrix metalloproteinase 13) gene expression in
chondrocytes requires p38, c-jun N-terminal kinase, and nuclear
factor kappaB: Differential regulation of collagenase 1 and
collagenase 3. Arthritis Rheum. 43:801–811. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pujol JP, Chadjichristos C, Legendre F,
Bauge C, Beauchef G, Andriamanalijaona R, Galera P and Boumediene
K: Interleukin-1 and transforming growth factor-beta1 as crucial
factors in osteoarthritic cartilage metabolism. Connect Tissue Res.
49:293–297. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Melusova M, Slamenova D, Kozics K, Jantova
S and Horvathova E: Carvacrol and rosemary essential oil manifest
cytotoxic, DNA-protective and pro-apoptotic effect having no effect
on DNA repair. Neoplasma. 61:690–699. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bakır M, Geyikoglu F, Colak S, Turkez H,
Bakır TO and Hosseinigouzdagani M: The carvacrol ameliorates acute
pancreatitis-induced liver injury via antioxidant response.
Cytotechnology. 68:1131–1146. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Z, Hua C, Pan X, Fu X and Wu W:
Carvacrol exerts neuroprotective effects via suppression of the
inflammatory response in middle cerebral artery occlusion rats.
Inflammation. 39:1566–1572. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Akhtar N, Rasheed Z, Ramamurthy S,
Anbazhagan AN, Voss FR and Haqqi TM: MicroRNA-27b regulates the
expression of matrix metalloproteinase 13 in human osteoarthritis
chondrocytes. Arthritis Rheum. 62:1361–1371. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Marcu KB, Otero M, Olivotto E, Borzi RM
and Goldring MB: NF-kappaB signaling: Multiple angles to target OA.
Curr Drug Targets. 11:599–613. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Martel-Pelletier J, Boileau C, Pelletier
JP and Roughley PJ: Cartilage in normal and osteoarthritis
conditions. Best Pract Res Clin Rheumatol. 22:351–384. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Aida Y, Maeno M, Suzuki N, Shiratsuchi H,
Motohashi M and Matsumura H: The effect of IL-1beta on the
expression of matrix metalloproteinases and tissue inhibitors of
matrix metalloproteinases in human chondrocytes. Life Sci.
77:3210–3221. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Li H, Li L, Min J, Yang H, Xu X, Yuan Y
and Wang D: Levels of metalloproteinase (MMP-3, MMP-9), NF-kappaB
ligand (RANKL), and nitric oxide (NO) in peripheral blood of
osteoarthritis (OA) patients. Clin Lab. 58:755–762. 2012.PubMed/NCBI
|
|
23
|
Marcu KB, Otero M, Olivotto E, Borzi RM
and Goldring MB: NF-kappaB signaling: Multiple angles to target OA.
Curr Drug Targets. 11:599–613. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Landa P, Kokoska L, Pribylova M, Vanek T
and Marsik P: In vitro anti-inflammatory activity of carvacrol:
Inhibitory effect on COX-2 catalyzed prostaglandin E(2)
biosynthesis. Arch Pharm Res. 32:75–78. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Silva FV, Guimarães AG, Silva ER,
Sousa-Neto BP, Machado FD, Quintans-Júnior LJ, Arcanjo DD, Oliveira
FA and Oliveira RC: Anti-inflammatory and anti-ulcer activities of
carvacrol, a monoterpene present in the essential oil of oregano. J
Med Food. 15:984–991. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lima Mda S, Quintans-Júnior LJ, de Santana
WA, Martins Kaneto C, Pereira Soares MB and Villarreal CF:
Anti-inflammatory effects of carvacrol: Evidence for a key role of
interleukin-10. Eur J Pharmacol. 699:112–117. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Arigesavan K and Sudhandiran G: Carvacrol
exhibits anti-oxidant and anti-inflammatory effects against 1,
2-dimethyl hydrazine plus dextran sodium sulfate induced
inflammation associated carcinogenicity in the colon of Fischer 344
rats. Biochem Biophys Res Commun. 461:314–320. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Vincenti MP and Brinckerhoff CE: Early
response genes induced in chondrocytes stimulated with the
inflammatory cytokine interleukin-1beta. Arthritis Res. 3:381–388.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chen WP, Tang JL, Bao JP, Hu PF, Shi ZL
and Wu LD: Anti-arthritic effects of chlorogenic acid in
interleukin-1β-induced rabbit chondrocytes and a rabbit
osteoarthritis model. Int Immunopharmacol. 11:23–28. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Han G, Shao H, Zhu X, Wang G, Liu F, Wang
F, Ling P and Zhang T: The protective effect of xanthan gum on
interleukin-1β induced rabbit chondrocytes. Carbohydr Polym.
89:870–875. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Nakagawa S, Arai Y, Mazda O, Kishida T,
Takahashi KA, Sakao K, Saito M, Honjo K, Imanishi J and Kubo T:
N-acetylcysteine prevents nitric oxide-induced chondrocyte
apoptosis and cartilage degeneration in an experimental model of
osteoarthritis. J Orthop Res. 28:156–163. 2010.PubMed/NCBI
|
|
32
|
Aktan F: iNOS-mediated nitric oxide
production and its regulation. Life Sci. 75:639–653. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Abramson SB: The role of COX-2 produced by
cartilage in arthritis. Osteoarthritis Cartilage. 7:380–381. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ying X, Chen X, Cheng S, Shen Y, Peng L
and Xu HZ: Piperine inhibits IL-β induced expression of
inflammatory mediators in human osteoarthritis chondrocyte. Int
Immunopharmacol. 17:293–299. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chowdhury TT, Bader DL and Lee DA: Dynamic
compression counteracts IL-1beta induced iNOS and COX-2 activity by
human chondrocytes cultured in agarose constructs. Biorheology.
43:413–429. 2006.PubMed/NCBI
|
|
36
|
Aristatile B, Al-Assaf AH and Pugalendi
KV: Carvacrol suppresses the expression of inflammatory marker
genes in D-galactosamine-hepatotoxic rats. Asian Pac J Trop Med.
6:205–211. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Dreier R, Grässel S, Fuchs S, Schaumburger
J and Bruckner P: Pro-MMP-9 is a specific macrophage product and is
activated by osteoarthritic chondrocytes via MMP-3 or a
MT1-MMP/MMP-13 cascade. Exp Cell Res. 297:303–312. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Takaishi H, Kimura T, Dalal S, Okada Y and
D'Armiento J: Joint diseases and matrix metalloproteinases: A role
for MMP-13. Curr Pharm Biotechnol. 9:47–54. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fosang AJ, Last K, Knäuper V, Murphy G and
Neame PJ: Degradation of cartilage aggrecan by collagenase-3
(MMP-13). FEBS Lett. 380:17–20. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Johansson N, Saarialho-Kere U, Airola K,
Herva R, Nissinen L, Westermarck J, Vuorio E, Heino J and Kähäri
VM: Collagenase-3 (MMP-13) is expressed by hypertrophic
chondrocytes, periosteal cells, and osteoblasts during human fetal
bone development. Dev Dyn. 208:387–397. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ahmed S, Wang N, Hafeez BB, Cheruvu VK and
Haqqi TM: Punica granatum L. extract inhibits IL-1beta-Induced
expression of matrix metalloproteinases by inhibiting the
activation of MAP kinases and NF-kappaB in human chondrocytes in
vitro. J Nutr. 135:2096–2102. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Aida Y, Maeno M, Suzuki N, Shiratsuchi H,
Motohashi M and Matsumura H: The effect of IL-1beta on the
expression of matrix metalloproteinases and tissue inhibitors of
matrix metalloproteinases in human chondrocytes. Life Sci.
77:3210–3221. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Woodell-May J, Matuska A, Oyster M, Welch
Z, O'Shaughnessey K and Hoeppner J: Autologous protein solution
inhibits MMP-13 production by IL-1β and TNFα-stimulated human
articular chondrocytes. J Orthop Res. 29:1320–1326. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liacini A, Sylvester J, Li WQ and
Zafarullah M: Inhibition of interleukin-1-stimulated MAP kinases,
activating protein-1 (AP-1) and nuclear factor kappa B (NF-kappaB)
transcription factors down-regulates matrix metalloproteinase gene
expression in articular chondrocytes. Matrix Biol. 21:251–262.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Saklatvala J: Inflammatory signaling in
cartilage: MAPK and NF-kappaB pathways in chondrocytes and the use
of inhibitors for research into pathogenesis and therapy of
osteoarthritis. Curr Drug Targets. 8:305–313. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Roman-Blas JA and Jimenez SA: NF-kappaB as
a potential therapeutic target in osteoarthritis and rheumatoid
arthritis. Osteoarthritis Cartilage. 14:839–848. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wehling N, Palmer GD, Pilapil C, Liu F,
Wells JW, Müller PE, Evans CH and Porter RM: Interleukin-1beta and
tumor necrosis factor alpha inhibit chondrogenesis by human
mesenchymal stem cells through NF-kappaB-dependent pathways.
Arthritis Rheum. 60:801–812. 2009. View Article : Google Scholar : PubMed/NCBI
|