Introduction
Alcoholic liver disease (ALD) is a major cause of
liver-associated morbidity and mortality worldwide (1). ALD encompasses a spectrum of
conditions, ranging from simple steatosis to chronic hepatitis with
fibrosis or cirrhosis (2).
Although the exact mechanisms of the pathogenesis of ALD are not
fully understood, is has been suggested that tight-junction protein
expression and intestinal barrier function have crucial roles in
the development of ALD (3).
Previous studies have indicated that alcohol increases intestinal
permeability to macromolecules and leads to an abnormal leakage of
bacterial endotoxins, thereby inducing alcohol-induced liver injury
(4,5). Numerous other studies have also
indicated that chronic alcohol consumption may accelerate the
progression of ALD via oxidative stress, increased intestinal
permeability and an elevated risk of endotoxemia (6,7). It
is now generally accepted that damaged intestinal epithelial
integrity and intestinal barrier dysfunction are the two
fundamental causes of increased intestinal permeability (8,9).
Therefore, an improved understanding of the mechanisms involved in
increased intestinal permeability induced by alcohol may lead to
the development of more effective prevention and treatment
strategies for ALD (10).
The intestinal epithelial barrier is mainly composed
of a monolayer of cells with intercellular tight junctions, a
complex three-dimensional structure and a thick mucosal gel layer
secreted by the mucous membrane; it also provides a dynamic and
regulated barrier to the extracellular flux of the lumina (11,12).
Tight junctions are essential in maintaining intestinal mucosal
integrity, which is able to effectively prevent bacteria,
endotoxins and other harmful substances from entering the blood
stream through the intestinal barrier (13). Previous studies have indicated that
altered expression of the tight junction-associated proteins,
zonula occludens-1 (ZO-l) and claudin-1, is associated with
increased intestinal permeability and a higher susceptibility to
ALD (14,15). However, no previous study
investigating whether alcohol is able to induce intestinal
epithelial barrier dysfunction and affect the expression of these
tight junction-associated proteins is available. The present study
used an alcohol-treated Caco-2 intestinal epithelial cell monolayer
in vitro model to observe the effects of alcohol on
intestinal epithelial barrier permeability and the expression of
tight junction-associated proteins.
Materials and methods
Culture of Caco-2 cells
The human colon adenocarcinoma Caco-2 cell line was
obtained from the American Type Culture Collection (ATCC;
Rockville, MD, USA). Caco-2 cells were cultured in Dulbecco’s
modified Eagle’s medium (DMEM; Gibco-BRL, Grand Island, NY, USA)
supplemented with 0.1 mmol/l non-essential amino acids, 10 mmol/l
HEPES, 4.5 mg/ml glucose, 100 U/ml penicillin, 100 U/ml
streptomycin, 4 mM glutamine and 10% fetal bovine serum (FBS;
Hyclone, Logan, UT, USA), and incubated in a humidified atmosphere
(95% air, 5% CO2) at 37°C.
Establishment of an in vitro model of the
intestinal epithelial cell barrier
To establish an in vitro model of the
intestinal epithelial barrier, the Caco-2 cells were plated on
Transwell filters (Corning, Inc., Corning, NY, USA) and regularly
monitored visually using an inverted microscope (Olympus, Tokyo,
Japan) and through epithelial resistance measurements.
Epithelial resistance measurement in
Ussing chambers
Firstly, the abdomen was carefully incised from the
midline and a 1- to 1.5-cm sample of the small intestine was
dissected out. Then, an exposed area of 0.126 cm2 was
utilized to mount the sample vertically in Ussing chambers.
Following that, Krebs-Ringer bicarbonate solution (in mM: 128 NaCl,
5.1 KCl, 1.4 CaCl2, 1.3 MgCl2, 21
NaHCO3, 1.3 KH2PO4 and 10
NaH2PO4, pH 7.4) gassed with 95% O2–5%
CO2 was used to bath the tissues. Following a 15-min
equilibration period, an EVC 4000 Precision V/I clamp device (World
Precision Instruments, Sarasota, FL, USA) was used for the
measurement of the transepithelial electrical voltage and current
per 5 min until 30 min. The calculation of epithelial resistance
was achieved using Ohm’s law (R=V/I). The experiment was repeated
at least three times using a different tissue sample each time.
Viability assay
The Caco-2 cells were seeded on flat-bottomed,
96-well tissue culture plates. Following incubation with alcohol
(1, 2.5, 5, 7.5 and 10%) for 4 h, the cells were incubated with 100
μl MTT [5 mg/ml solution in phosphate-buffered saline (PBS);
Sigma-Aldrich, Taufkirchen, Germany] for 1 h at 37°C. The culture
media was removed, and 150 μl dimethylsulfoxide was added to each
well. The absorbance of the resulting colored solution was measured
at 570 nm with a microplate reader (Wallac Victor 2; Perkin-Elmer,
Waltham, MA, USA). In this assay, the yellow MTT solution is
converted into a blue formazan dye within the mitochondria and
deposited intracellularly. The intensity of the blue stain is then
quantitatively assessed by spectrometry and used as a measure of
cell viability.
Cell viability was also assessed by measuring the
release of cytosolic enzymes. The electrical resistance of Caco-2
cell monolayers cultured on Transwell filters was assessed using a
Millicell-ERS instrument (Millipore, Bedford, MA, USA). The
electrical resistance was expressed in units of Ω•cm2
using the surface area of the Transwell insert.
Fluorescent yellow (40 μg/ml; Sigma-Aldrich) in
serum-free DMEM was added to the upper chamber of the Transwell
system. Following incubation with alcohol for 0, 20, 40, 60, 120 or
180 min, the media from the lower chamber was collected. The
absorbance was assessed using a fluorescence spectrophotometer
(excitation wavelength, 427 nm; emission wavelength, 536 nm), and
the concentration of fluorescent yellow was calculated based on the
standard curve. The fluorescent yellow flux rate (%) was equal to
the fluorescent yellow concentration in the lower chamber/the
fluorescent yellow concentration in the upper chamber. Lactate
dehydrogenase (LDH) was added into the medium. The Caco-2 cells
were incubated with the varying concentrations of alcohol for 4 h.
LDH assessment was performed in 250 μl-aliquots using an LDH kit
(Doles reagents, Goiânia, GO, Brazil).
Assessment of intestinal epithelial
barrier permeability
Transepithelial electrical resistance (TEER) and the
fluorescent yellow flux rate were assessed to estimate the effects
of alcohol on the paracellular permeability in the Caco-2 cell
monolayers. The electrical resistance of the Caco-2 cell monolayers
cultured on Transwell filters was assessed using a Millicell-ERS
instrument (Millipore, Bedford, MA, USA). The electrical resistance
was expressed in units of Ω•cm2 using the surface area
of the Transwell insert. Fluorescent yellow (40 μg/ml;
Sigma-Aldrich) in serum-free DMEM was added to the upper chamber of
the Transwell system. Following incubation with alcohol for the
varying times indicated, the media from the lower chamber was
collected. The absorbance was assessed using a fluorescence
spectrophotometer (excitation wavelength, 427 nm; emission
wavelength, 536 nm), and the fluorescent yellow concentration was
calculated based on the standard curve. The fluorescent yellow flux
rate (%) was equal to the fluorescent yellow concentration in the
lower chamber/the fluorescent yellow concentration in the upper
chamber.
Western blot analysis
The Caco-2 cells were washed with Dulbecco’s PBS
(D-PBS) containing 0.1 mM ethylenediamine tetraacetic acid (EDTA)
three times without calcium and magnesium. The Caco-2 cells were
then homogenized in 1 ml lysis buffer A [(Shanghai Pik-day,
Shanghai, China) 2 mM EDTA, 10 mM ethylene glycol tetraacetic acid,
0.4% NaF, 20 mM Tris-HCl, protease inhibitor cocktail, phosphatase
inhibitor and 1% Triton X-100 (pH 7.5)] at 4°C. Samples were
centrifuged at 14,000 × g for 30 min, and the supernatant was
transferred to a separate tube and collected as the soluble
fraction. Buffer A (150 μl) with 1% sodium dodecyl sulfate (SDS) at
4°C was then added to the pellet. The pellet’s structure was
disrupted with an ultrasonic crusher. The samples were then
centrifuged at 14,000 × g for 30 min at 4°C. The supernatant was
collected as the insoluble fraction. Equal amounts of protein
(40–50 μg) were separated by SDS-PAGE and processed for
immunoblotting with antibodies for ZO-1 (diluted 1:1,000; Santa
Cruz Biotechnology, Inc., Santa Cruz, CA, USA) and claudin-1
(diluted 1:100; Santa Cruz Biotechnology, Inc.). The protein bands
were scanned using ChemiImager 5500 V2.03 software (Alpha US Inc.,
Miami, FL, USA), and the integrated density values (IDVs) were
calculated using a computerized image analysis system (Fluor Chen
2.0; Olympus, Yokohama, Japan) and normalized to that of
β-actin.
Immunofluorescence
The Caco-2 cell monolayers grown on glass coverslips
were fixed with 4% paraformaldehyde and permeabilized with 0.5%
Triton X-100. Following blocking with 2% bovine serum albumin in
PBS, the cells were incubated with rabbit anti-ZO-1 (diluted 1:50;
Santa Cruz Biotechnology, Inc.) and rabbit anti-claudin-1 (diluted
1:100; Zymed, South San Francisco, CA) for visualization of the
distribution of ZO-1 and claudin-1. The glass slides were analyzed
using immunofluorescence microscopy (Olympus, Tokyo, Japan).
Statistical analysis
Experiments were repeated at least three times.
Continuous variables are expressed as the mean ± standard
deviation. Categorical data are presented as frequencies and
percentages. Differences between the groups were compared using the
two-tailed, non-paired Student’s t-test or one-way analysis of
variance for continuous variables, where appropriate. Comparisons
of categorical variables between the groups were performed using
the χ2 test. All tests of statistical significance were
two-sided, with P<0.05 being considered to indicate a
statistically significant difference. The statistical analyses were
performed using SPSS 17.0 (SPSS, Inc., Chicago, IL, USA).
Results
Effects of alcohol on Caco-2 cell
viability
The Caco-2 cells were initially treated for 3 h with
varying concentrations (1, 2.5, 5, 7.5 and 10%) of alcohol, and
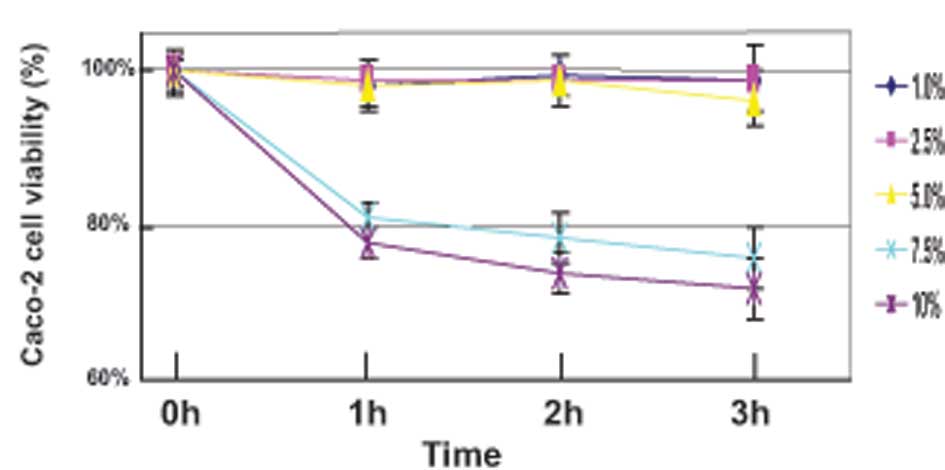
cell viability was evaluated using the MTT and LDH assays. The MTT
assay results showed that the cell viability was not altered at
alcohol concentrations of <5% (Fig.
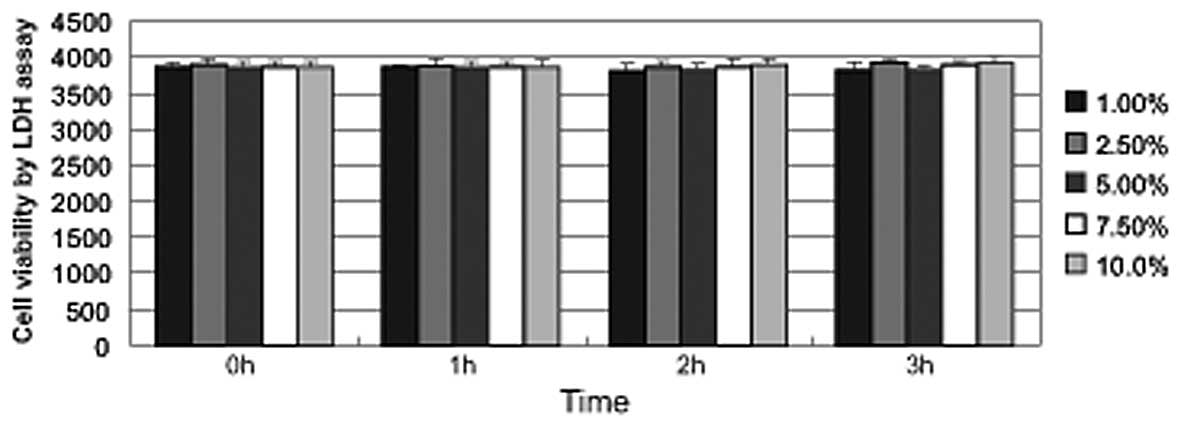
1). Furthermore, the LDH assay indicated that alcohol did not
increase the release of the cytosolic enzyme LDH into the media at
concentrations of ≤10% (Fig. 2).
Alcohol concentrations of >5% caused cell shedding but not cell
fragmentation. At an alcohol concentration of 10%, the ratio of
cell shedding was up to 51.67±3.36%. Therefore, subsequent
experiments were performed using alcohol concentrations of
1–5%.
Effects of alcohol on paracellular
permeability
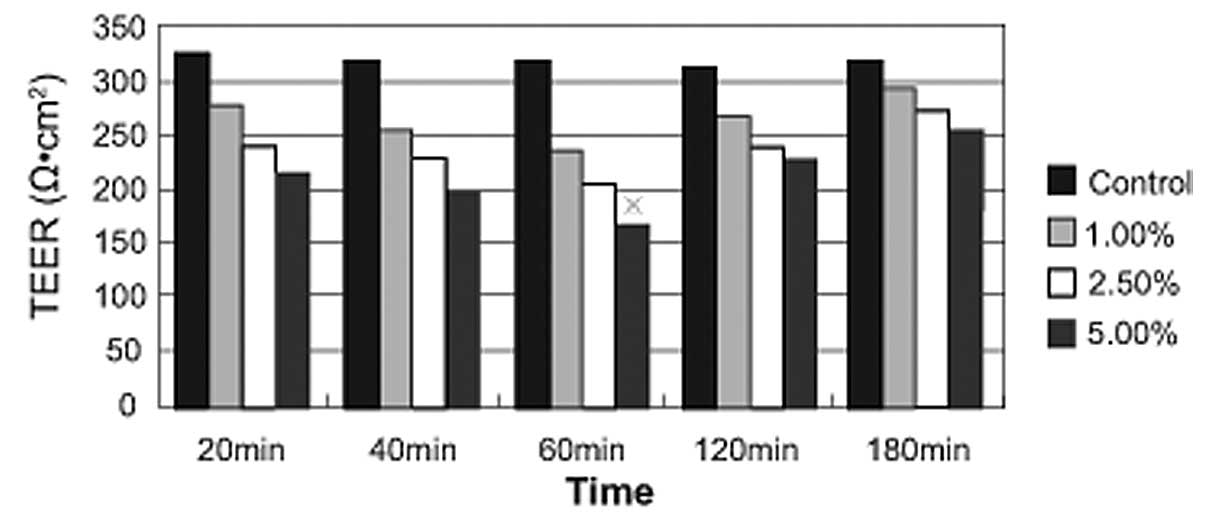
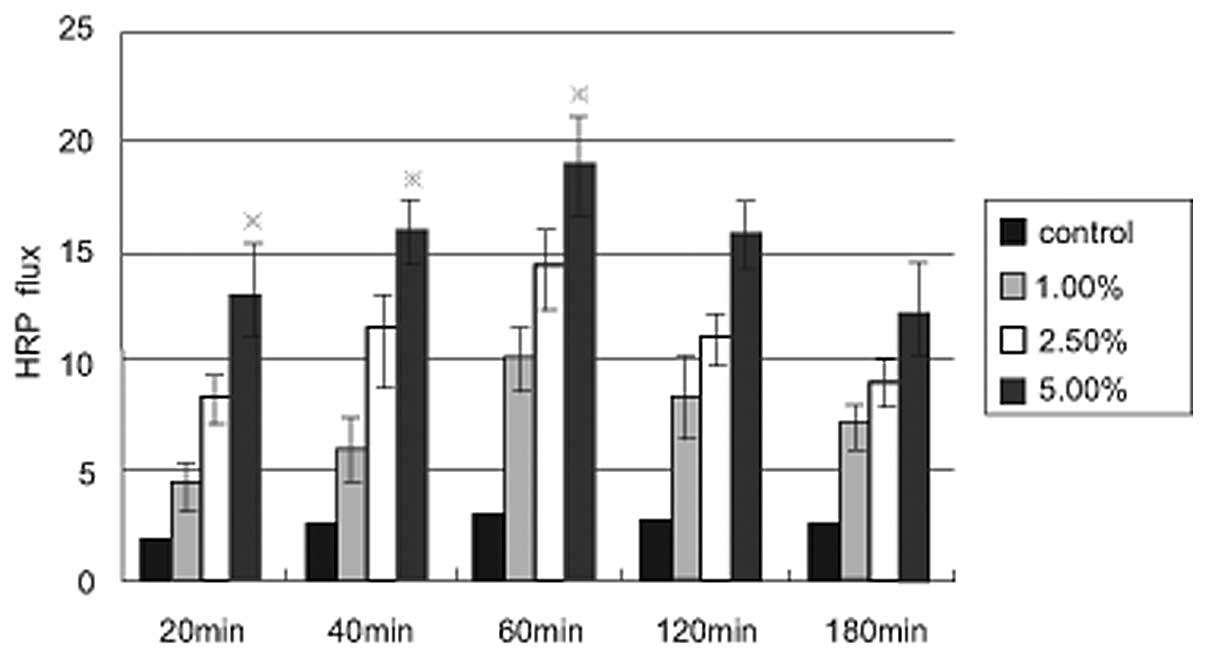
As shown in Fig. 3,
5% alcohol induced a significant
time-dependent decrease in transepithelial electrical resistance
(TEER), with the lowest value obtained after 60 min of alcohol
treatment. A time-dependent increase in the fluorescent yellow flux
rate as a result of alcohol treatment was observed, with the
maximum flux rate occurring at 60 min (Fig. 4). The aforementioned results
indicated that the decreased TEER value was associated with an
increase in the fluorescent yellow flux rate.
Effects of ethanol on the expression of
ZO-1
The Caco-2 cells in the alcohol treatment groups
were incubated with 5% alcohol for 0, 20, 40, 60, 120 and 180 min.
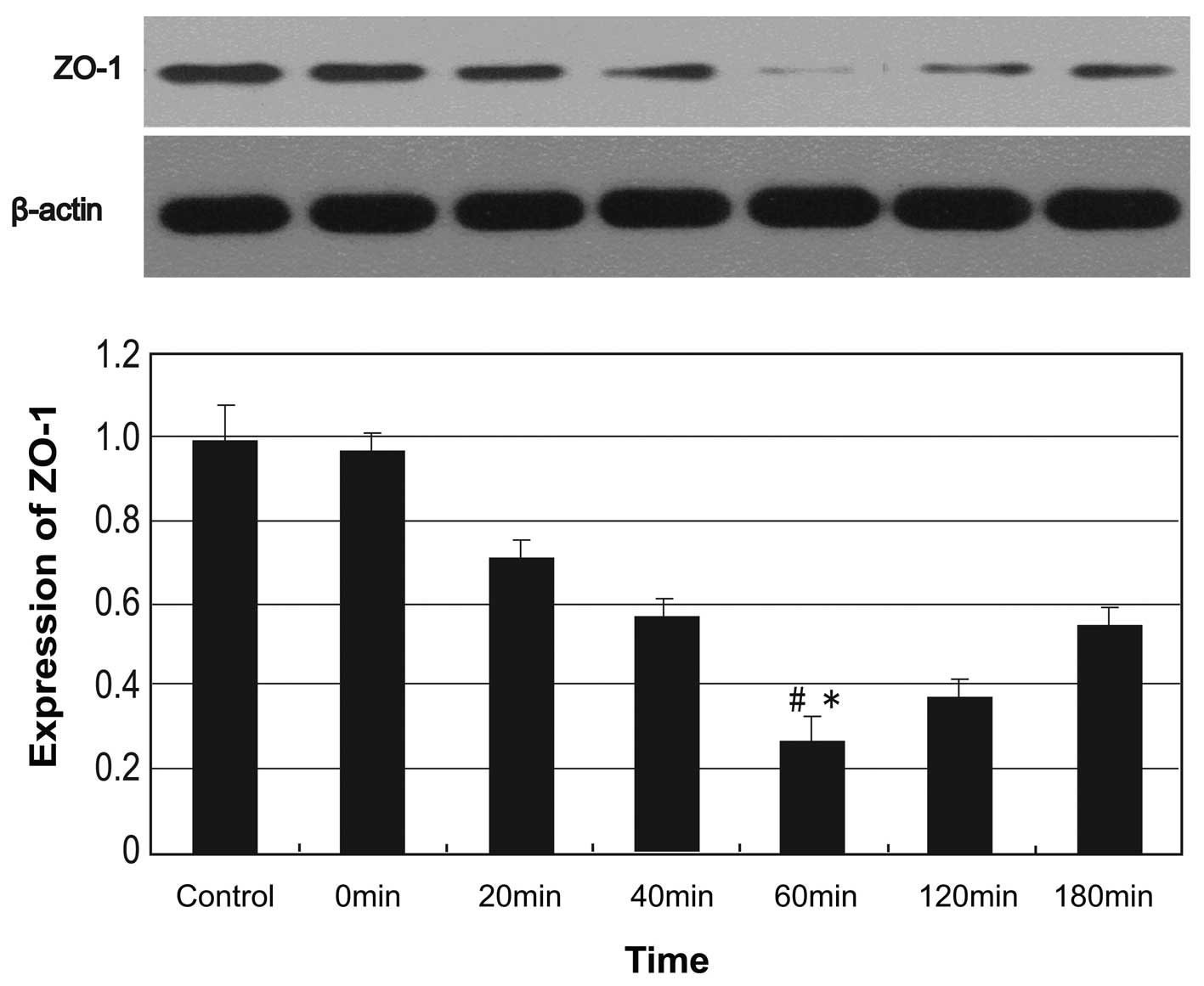
As shown in Fig. 5, the expression
levels of the tight junction-associated protein, ZO-1, exhibited a
progressive decline following 20 min of incubation, reached a
minimum level at 60 min and exhibited an increasing trend after 60
min of incubation. Significant differences in ZO-1 expression
between the control and alcohol groups were observed at 60 min of
incubation (P<0.05).
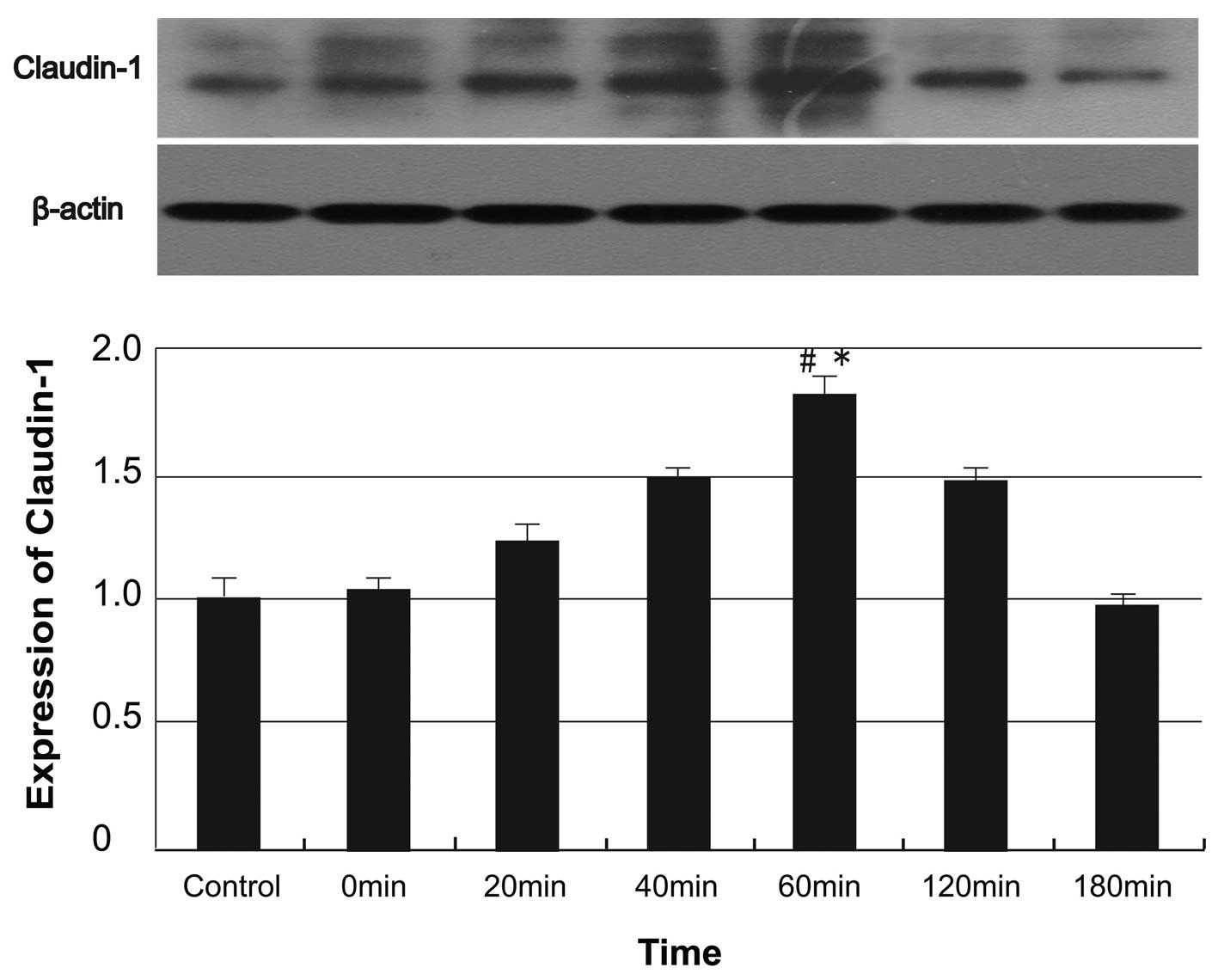
Effects of alcohol on the expression of
claudin-1
Expression of the claudin-1 protein in the
alcohol-treated Caco-2 cells showed a progressive increase
following 60 min of incubation, reached its maximum level at 60 min
and then showed a decreasing trend following 60 min of incubation
(Fig. 6). Significant differences
in claudin-1 expression were identified among the control and
alcohol-treated groups at 60 min (P<0.05).
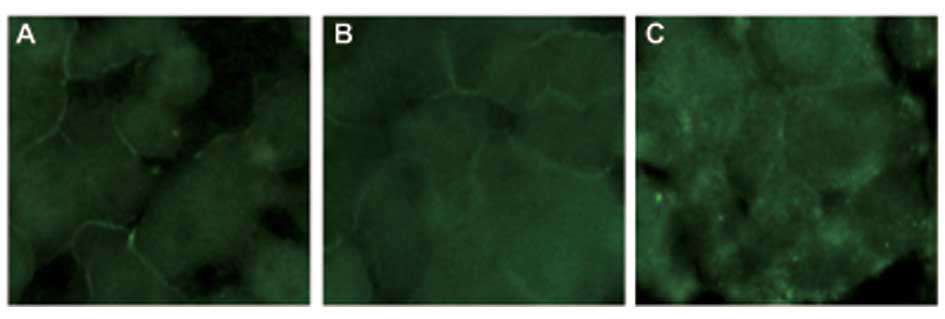
Effects of alcohol on the distribution of
ZO-1 and claudin-1
The distribution of ZO-1 and claudin-1 was assessed
using immunofluorescence microscopy (Fig. 7). Alcohol was shown to induce the
discontinuous distribution of ZO-1 and claudin-1 at the cellular
membrane.
Discussion
Experimental and clinical studies have revealed
evidence that damaged intestinal epithelial integrity and
intestinal barrier dysfunction are the two fundamental causes of
increased intestinal permeability (8,9).
Intestinal epithelial barrier integrity has a significant role in
preventing bacterial translocation by altering pathogenetic
mechanisms (16). Alcohol-induced
intestinal barrier dysfunction may lead to increased intestinal
permeability (17). However, the
specific action sites at which alcohol may increase intestinal
epithelial barrier permeability are not fully understood.
The Caco-2 cell line, originally derived from a
human colon adenocarcinoma, resembles small intestinal enterocytes
and spontaneously undergoes typical enterocytic differentiation
(18). The Caco-2 monolayer cells
exhibit a well-differentiated brush border containing tight
junctions on the apical surface and have been extensively used to
study intestinal epithelial barrier functions (19). The present study used an
alcohol-treated Caco-2 intestinal epithelial cell monolayer in
vitro model to investigate whether alcohol is able to induce
intestinal barrier dysfunction and decrease expression of the tight
junction-associated proteins, ZO-1 and claudin-1. The effects of
alcohol on Caco-2 cell viability were assessed using the MTT assay,
and the LDH concentration in supernatant fluid was assessed at 450
nm using a microplate reader. The MTT assay indicated that cell
viability was not altered at alcohol concentrations of <5%,
while concentrations of ≥7.5% led to a ≥20% reduction in Caco-2
cell viability. However, alcohol concentrations of 1–10% had
virtually no impact on the LDH concentration in the supernatant
fluid, and the Caco-2 cell membrane showed no evidence of
morphological damage. Based on these results, an alcohol
concentration of 5% was defined as the maximum concentration at
which ethanol has no significant effect on Caco-2 cell viability
and the molecular structure of tight junctions.
Smaller molecules penetrate enterocytes through a
transcellular route via a large number of transmembrane channels
and carriers, whereas larger molecules are transported across
enterocytes through a paracellular pathway via intercellular tight
junctions (20,21). The permeability of the paracellular
pathway is the molecular basis of the substance selectivity of
epithelial cells (22). In the
present study, the TEER and fluorescent yellow flux rate were
utilized to estimate the effects of alcohol on the paracellular
permeability of the Caco-2 cell monolayers. The flow of ions in the
paracellular gap was represented by the TEER values. The results
showed that alcohol induced a significant time-dependent decrease
in the TEER, with the lowest value obtained at 60 min of alcohol
treatment. A significant correlation was present between the
decrease in the TEER value and the increase in the fluorescent
yellow flux rate. A time-dependent increase in the fluorescent
yellow flux rate as a result of the alcohol treatment was also
observed. An increase in the TEER value and fluorescent yellow
transmittance rate was observed within 60 min of incubation,
indicating that the effects of alcohol on the paracellular
permeability in the Caco-2 cell monolayers may be a transient
process. Furthermore, western blot analysis was performed to detect
the effects of alcohol on the expression of the tight
junction-associated proteins, ZO-l and claudin-1. ZO-1, also known
as tight junction protein 1, is an important member of the
membrane-associated guanylate kinase homologs (MAGUK) family,
having binding domains to adherens, tight junction proteins and the
actin cytoskeleton (23).
Claudin-1, a protein responsible for controlling cell-to-cell
adhesion, is an integral component of tight junctions, regulating
the paracellular and transcellular transport of solutes across
human epithelia and endothelia (24). The present study demonstrated that
ZO-l expression exhibited a progressive decline at 20 min and
reached its minimum level at 60 min, with an increasing trend
observed following 60 min of incubation, and claudin-1 expression
showed an inverse correlation. Alterations in the expression of the
ZO-l and claudin-1 proteins showed consistent trends with changes
in the TEER value and the fluorescent yellow transmittance rate in
the Caco-2 cells, indicating that ZO-l and claudin-1 have important
roles in alcohol-induced intestinal barrier dysfunction.
In conclusion, the results of the present study
indicated that alcohol increases the permeability of the intestinal
epithelial barrier in a dose- and time-dependent manner. Alcohol
induces a change in the expression of the tight junction-associated
proteins, ZO-1 and claudin-1, which are two major sites of alcohol
action, thus increasing intestinal epithelial barrier
permeability.
Acknowledgements
This study was funded by the Scientific Research of
the First Hospital of China Medical University (fsfh1313).
Abbreviations:
|
ALD
|
alcoholic liver disease
|
|
ZO-l
|
zonula occludens-1
|
|
TEER
|
transepithelial electrical
resistance
|
|
LDH
|
lactate dehydrogenase
|
Referenes
|
1
|
Bataller R and Gao B: Dissecting the role
of CB1 receptors on chronic liver diseases. Gut. 62:957–958. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Li YM, Fan JG, Wang BY, et al: Guidelines
for the diagnosis and management of alcoholic liver disease: update
2010: (published in Chinese on Chinese Journal of Hepatology 2010;
18: 167–170). J Dig Dis. 12:45–50. 2011.PubMed/NCBI
|
|
3
|
Rao RK, Seth A and Sheth P: Recent
Advances in Alcoholic Liver Disease I. Role of intestinal
permeability and endotoxemia in alcoholic liver disease. Am J
Physiol Gastrointest Liver Physiol. 286:G881–G884. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brun P, Castagliuolo I, Di Leo V, et al:
Increased intestinal permeability in obese mice: new evidence in
the pathogenesis of nonalcoholic steatohepatitis. Am J Physiol
Gastrointest Liver Physiol. 292:G518–G525. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Purohit V, Bode JC, Bode C, et al:
Alcohol, intestinal bacterial growth, intestinal permeability to
endotoxin, and medical consequences: summary of a symposium.
Alcohol. 42:349–361. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Parlesak A, Schäfer C, Schütz T, Bode JC
and Bode C: Increased intestinal permeability to macromolecules and
endotoxemia in patients with chronic alcohol abuse in different
stages of alcohol-induced liver disease. J Hepatol. 32:742–747.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zima T and Kalousová M: Oxidative stress
and signal transduction pathways in alcoholic liver disease.
Alcohol Clin Exp Res. 29(11 Suppl): 110S–115S. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Turner JR: Intestinal mucosal barrier
function in health and disease. Nat Rev Immunol. 9:799–809. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yu J, Liu F, Yin P, et al: Involvement of
oxidative stress and mitogen-activated protein kinase signaling
pathways in heat stress-induced injury in the rat small intestine.
Stress. 16:99–113. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Banan A, Keshavarzian A, Zhang L, et al:
NF-kappaB activation as a key mechanism in ethanol-induced
disruption of the F-actin cytoskeleton and monolayer barrier
integrity in intestinal epithelium. Alcohol. 41:447–460. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Colgan SP and Taylor CT: Hypoxia: an alarm
signal during intestinal inflammation. Nat Rev Gastroenterol
Hepatol. 7:281–287. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kong J, Zhang Z, Musch MW, et al: Novel
role of the vitamin D receptor in maintaining the integrity of the
intestinal mucosal barrier. Am J Physiol Gastrointest Liver
Physiol. 294:G208–G216. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ulluwishewa D, Anderson RC, McNabb WC,
Moughan PJ, Wells JM and Roy NC: Regulation of tight junction
permeability by intestinal bacteria and dietary components. J Nutr.
141:769–776. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Blaskewicz CD, Pudney J and Anderson DJ:
Structure and function of intercellular junctions in human cervical
and vaginal mucosal epithelia. Biol Reprod. 85:97–104. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Groschwitz KR and Hogan SP: Intestinal
barrier function: molecular regulation and disease pathogenesis. J
Allergy Clin Immunol. 124:3–22. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Balzan S, de Almeida Quadros C, de Cleva
R, Zilberstein B and Cecconello I: Bacterial translocation:
overview of mechanisms and clinical impact. J Gastroenterol
Hepatol. 22:464–471. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Marchiando AM, Graham WV and Turner JR:
Epithelial barriers in homeostasis and disease. Annu Rev Pathol.
5:119–144. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fogh J, Fogh JM and Orfeo T: One hundred
and twenty-seven cultured human tumor cell lines producing tumors
in nude mice. J Natl Cancer Inst. 59:221–226. 1977.PubMed/NCBI
|
|
19
|
Fischer A, Gluth M, Weege F, et al:
Glucocorticoids regulate barrier function and claudin expression in
intestinal epithelial cells via MKP-1. Am J Physiol Gastrointest
Liver Physiol. 306:G218–G228. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Colegio OR, Van Itallie CM, McCrea HJ,
Rahner C and Anderson JM: Claudins create charge-selective channels
in the paracellular pathway between epithelial cells. Am J Physiol
Cell Physiol. 283:C142–C147. 2002. View Article : Google Scholar
|
|
21
|
Ding LA and Li JS: Gut in diseases:
physiological elements and their clinical significance. World J
Gastroenterol. 9:2385–2389. 2003.PubMed/NCBI
|
|
22
|
Muto S, Hata M, Taniguchi J, et al:
Claudin-2-deficient mice are defective in the leaky and
cation-selective paracellular permeability properties of renal
proximal tubules. Proc Natl Acad Sci USA. 107:8011–8016. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sobarzo CM, Lustig L, Ponzio R, Suescun MO
and Denduchis B: Effects of di(2-ethylhexyl) phthalate on gap and
tight junction protein expression in the testis of prepubertal
rats. Microsc Res Tech. 72:868–877. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Krämer F, White K, Kubbies M, Swisshelm K
and Weber BH: Genomic organization of claudin-1 and its assessment
in hereditary and sporadic breast cancer. Hum Genet. 107:249–256.
2000.PubMed/NCBI
|