Introduction
Chronic obstructive pulmonary disease (COPD) is
expected to be the third leading cause of mortality and the fifth
leading cause of morbidity by the year 2020 (1). The disease is mainly characterized by
the presence of chronic airflow limitation that progresses slowly
over a period of years and is largely irreversible (2,3). In
China, it is becoming an increasingly common problem. A survey of
20,245 participants in seven regions of China conducted in 2007
indicated that the prevalence of COPD in adults aged over 40 years
was 8.2% (4). However, the disease
remains under-recognized and under-diagnosed, and we need to
further understand the pathogenesis, particularly in the earlier
mild and moderate stages of COPD. Although cigarette smoking is the
major risk factor for COPD, only a minority (20%) of smokers
develop the disease clinically (5). Hodge et al (6) revealed in their study that apoptosis
of airway epithelial cells and inflammation of the airway mucosa
persisted even after smoking cessation in patients with COPD.
Evidence suggests that the risk of developing COPD
is partially determined by genetic and environmental factors
(7). A number of candidate gene
studies have therefore been carried out in recent years. Family
studies and linkage analysis in early-onset COPD pedigrees have
highlighted a genetic predisposition (8–11),
and genome-wide association studies (GWAS) for COPD or pulmonary
function identified some susceptibility loci (12–16),
but with varying degrees of reproducibility. Conflicting results
may be due to ethnic differences and sample sizes.
The number of these studies carried out in Chinese
individuals is small. Past candidate gene studies may focus on a
single gene or on a few genes in combination, with these genes
identified based on prior knowledge or suspected mechanisms of
disease pathogenesis. Nonetheless, elucidating the genetics of
these disorders is severely hampered by genetic heterogeneity, the
low penetrance of individual disease alleles and the potential for
gene-gene and gene-environment interactions. It is probable that
groups of genes rather than single genes are involved in disease
development.
The aim of the present study was to investigate
candidate genes and haplotypes in susceptibility to COPD in a south
Han Chinese population.
Materials and methods
Subjects
A total of 200 male COPD patients visiting the
Department of Respiratory Disease of the Shanghai Ruijin Hospital,
China, between December 2008 and December 2009 were recruited. COPD
was diagnosed according to the criteria established by the
NHLBI/WHO Global Initiative for COPD (GOLD) (17). Criteria were as follows: age ≥40
years; chronic respiratory symptoms and signs, such as cough and
dyspnea; airflow limitation as indicated by forced expiratory
volume in 1 sec (FEV1)/forced vital capacity (FVC) <70% and FEV1
reversibility after inhalation of 400 μg salbutamol to
<12% of the pre-bronchodilator FEV1. Patients were excluded if
they had a diagnosis of asthma, lung cancer or radiographic
abnormalities suggestive of other significant respiratory diseases,
such as bronchiectasis or pulmonary tuberculosis.
A total of 250 control male subjects were enrolled
at the General Health Check-up Center in Shanghai No. 10 Hospital
during the same period. Their characteristics were mentioned in a
previous study (18). The cases
and control subjects were from an ethnic Chinese, southern Han
population who resided in Shanghai City or the surrounding regions,
and were matched for age, gender and smoking history. The study
protocol was approved by the medical ethics committee of Shanghai
Ruijin Hospital, School of Medicine, Shanghai Jiaotong University,
and all the participants gave written informed consent.
DNA extraction and genotyping of study
samples
We collected 4 ml of peripheral blood from each
participant for DNA preparation. Genomic DNA was extracted using a
Blood DNA Extraction kit (Tiangen Biotech, Co. Ltd., Beijing,
China). Any sample with a DNA concentration <10 ng/μl was
excluded.
In total, 54 single-nucleotide polymorphisms (SNPs)
were found in 23 genes associated with the development of COPD
and/or pulmonary function, as identified by publications of
previous GWAS and by searching the dbSNP database of NCBI (Table I). Genotyping was performed using
the Mass-Array™ Technology Platform of Sequenom Inc. (San Diego,
CA, USA). As a result of a quality control measure, we excluded 30
SNPs: the minor allele frequency (MAF) of 10 SNPs was <0.03;
these were rs8034191, rs17036052, rs17035960, rs11097901,
rs11728716, rs10516526, rs11727189, rs17036090, rs17331332 and
rs17036341. Eleven SNPs were not compatible in the same multiplex
PCR system; these were rs7710510, rs1042718, rs1042717, rs3753661,
rs3766934, rs1903003, rs7671167, rs1980057, rs11168048, rs7735184
and rs16865421. Nine SNPs showed deviation from the Hardy-Weinberg
equilibrium (HWE): rs2070600, rs2395730, rs6830970, rs13147758,
rs17019336, rs2035901, rs10498230, rs6712954 and rs6734100.
Therefore, 24 SNPs were selected for the investigation. The
sequence information of these 24 SNPs is shown in Table II.
 | Table ISummary of positive single-nucleotide
polymorphisms (SNPs) in the previous genome-wide association
studies. |
Table I
Summary of positive single-nucleotide
polymorphisms (SNPs) in the previous genome-wide association
studies.
| Gene | Gene description | Chromosome | SNP | MAF in HCa | Allele |
|---|
| ADAM19 (14) | Metallopeptidase
domain 19 | 5 | rs2277027 | 0.178 | A/C |
| | 5 | rs1422795 | 0.178 | A/G |
| ADCY2 (14) | Adenylate cyclase 2
(brain) | 5 | rs6555465 | 0.433 | A/G |
| | 5 |
rs7710510c | 0.432 | C/T |
| AGER (14) | Advanced
glycosylation end-product-specific receptor | 6 | rs2070600 | 0.289 | A/G |
| ARDB2 (36) | β2-adrenergic
receptor | 5 |
rs1042718c | 0.344 | A/C |
| | 5 |
rs1042717c | 0.322 | A/G |
| CHRNA3 (13) | Cholinergic
receptor nicotinic, α3 | 15 | rs1051730 | 0.033 | C/T |
| | 15 | rs8034191 | 0.011b | C/T |
| DAAM2 (14) | Dishevelled
activator of morphogenesis 2 | 6 | rs2395730 | 0.375 | A/G |
| EPHX1 (36) | Epoxide hydrolase
1, microsomal (xenobiotic) | 1 |
rs3753661c | 0.244 | G/T |
| | 1 |
rs3766934c | 0.244 | G/T |
| FAM13A (14) | Family with
sequence similarity 13, member A | 4 | rs2869967 | 0.478 | C/T |
| | 4 | rs6830970 | 0.444 | A/G |
| | 4 |
rs1903003c | 0.433 | C/T |
| | 4 |
rs7671167c | 0.488 | C/T |
| FLJ20184 (14) | Rho guanine
nucleotide exchange factor (GEF) 38 | 4 | rs17036052 | 0b | C/T |
| | 4 | rs17035960 | 0b | C/T |
| GPR126 (14) | G protein-coupled
receptor 126 | 6 | rs7776375 | 0.439 | A/G |
| | 6 | rs6937121 | 0.389 | G/T |
| | 6 | rs11155242 | 0.133 | A/C |
| | 6 | rs3817928 | 0.133 | A/G |
| GSTCD (14) | Glutathione
S-transferase, C-terminal domain containing | 4 | rs11097901 | 0b | C/T |
| | 4 | rs11728716 | 0.011b | A/G |
| | 4 | rs10516526 | 0b | A/G |
| HHIP (12,14,16) | Hedgehog
interacting protein | 4 | rs13147758 | 0.289 | A/G |
| | 4 | rs17019336 | 0.300 | A/T |
| | 4 | rs2353397 | 0.289 | C/T |
| | 4 | rs2035901 | 0.300 | A/G |
| | 4 | rs6537302 | 0.244 | A/T |
| | 4 | rs12504628 | 0.298 | C/T |
| | 4 | rs1032295 | 0.211 | T/G |
| | 4 |
rs1980057c | 0.300 | T/C |
| HTR4 (14,16) | 5-hydroxytryptamine
(serotonin) receptor 4 | 5 | rs3995090 | 0.256 | A/C |
| | 5 | rs6889822 | 0.333 | A/G |
| | 5 |
rs11168048c | 0.211 | C/T |
| | 5 |
rs7735184c | 0.267 | G/T |
| NT5DC1 (14) | 5′-nucleotidase
domain containing 1 | 6 | rs1052443 | 0.389 | A/C |
| INTS12 (14) | Integrator complex
subunit 12 | 4 | rs11727189 | 0b | G/T |
| | 4 | rs17036090 | 0b | C/T |
| NPNT (14) | Nephronectin | 4 | rs17331332 | 0b | A/G |
| | 4 | rs17036341 | 0b | C/G |
| PID1 (14) | Phosphotyrosine
interaction domain containing 1 | 2 | rs1435867 | 0.107 | C/T |
| | 2 | rs10498230 | 0.122 | C/T |
| PPT2 (34) | Palmitoyl-protein
thioesterase 2 | 6 | rs10947233 | 0.239 | G/T |
| PTCH1 (35) | Patched 1 | 9 | rs16909898 | 0.078 | A/G |
| | 9 | rs10512249 | 0.080 | C/T |
| SERPINE2 (36,37) | Serpin peptidase
inhibitor, clade E (nexin, plasminogen activator inhibitor type 1),
member 2 | 2 | rs7583463 | 0.292 | A/C |
| | 2 | rs6712954 | 0.086 | A/G |
| | 2 | rs6734100 | 0.189 | C/G |
| | 2 |
rs16865421c | 0.218 | A/G |
| THSD4 (16) | Thrombospondin,
type I, domain containing 4 | 15 | rs12899618 | 0.133 | A/G |
| TNS1 (16) | Tensin 1 | 2 | rs2571445 | 0.400 | C/T |
| TSPYL4 (14) | Testis-specific
protein Y encoded-like-4 | 6 | rs3749893 | 0.389 | A/G |
 | Table IISequence of 24 single-nucleotide
polymorphisms (SNPs) and their primers in multiplex PCR. |
Table II
Sequence of 24 single-nucleotide
polymorphisms (SNPs) and their primers in multiplex PCR.
| Gene | SNP_ID | Sequence | 1st-PCR Primer | 2nd-PCR Primer | UEP_SEQ |
|---|
| ADAM19 | rs2277027 |
GTGTCCTCAT[A/C]AAAAATTCAT |
ACGTTGGATGTTGGCTTGGCCATATGTGTC |
ACGTTGGATGGAAGAATTAAGGGCTGTGGC |
ccccGATCAAAATGTGTCCTCAT |
| rs1422795 |
GTCCTTTCTC[A/G]GTTGGAGGCGC |
ACGTTGGATGGCGTTGTCATGGTACTTCTG |
ACGTTGGATGCCATATTCTACCCTCTGGTC |
gCTACCCTCTGGTCCTTTCTC |
| ADCY2 | rs6555465 |
GGGATTCAGA[A/G]GCTGGATATG |
ACGTTGGATGACCTTCACCTTAGAGACAGC |
ACGTTGGATGCTGGGTGGCTTCCAATTTTC |
TATCCATGTCATATCCAGC |
| CHRNA3 | rs1051730 |
GCCCCAGGCTA[C/T]AAACACGACA |
ACGTTGGATGCAGCAGTTGTACTTGATGTC |
ACGTTGGATGCTCAAGGACTATTGGGAGAG |
ATCATCAAAGCCCCAGGCTA |
| FAM13A | rs2869967 |
CTGGAAGGGT[C/T]AAGGGATGTATT |
ACGTTGGATGAAGAGCAGAGCTCTTGGAAC |
ACGTTGGATGTGAGCCCCCATTTTCTAACC |
tacCCCAGGATCCTGAATACATCCCTT |
| GPR126 | rs11155242 |
TTCTGATTCA[A/C]AATGTTTGTT |
ACGTTGGATGGGCTACTTTCTATCCATTTC |
ACGTTGGATGAGCTGTTCAAAGCTTTCTGC |
TAATGCATTATTCAACAAACATT |
| rs7776375 |
CCCCCAACAC[A/G]GGTACCAAAA |
ACGTTGGATGACTTTGCACCGTGTGGTAAC |
ACGTTGGATGGTCAAATTAATGATCCCCCC |
attcATGATCCCCCCCAACAC |
| rs6937121 |
AATTTGTCCA[G/T]TCCTTTTAAC |
ACGTTGGATGCAGTAAAATTCTGATAGCC |
ACGTTGGATGGAAAGATTGGCACAACTGTC |
gCAAATCTATTATCTAATTTGTCCA |
| rs3817928 |
GATGACCCACCA[A/G]TTCAGTCCCTC |
ACGTTGGATGGGTGCCAAGGTAAAGAGATG |
ACGTTGGATGTGCAGCGTGTAATGTCTCAG |
ttATGTTTCACTTGTGAGGGACTGAA |
| HHIP | rs6537302 |
CCATCATCTA[A/T]CATACTAACA |
ACGTTGGATGGTGAAGTGCTTTGTAAGCTG |
ACGTTGGATGGCATATTTTCCTTCTTCCCC |
cccCCCCTCCTTTTTATGTTAGTATG |
| rs12504628 |
CATTACCCCA[C/T]TAAAGGTA |
ACGTTGGATGCCCAGAAAATAAGAGTCTGC |
ACGTTGGATGGGATTTGAGATTTAGAGTGCC |
TTAGAGTGCCATTACCCCA |
| rs1032295 |
TGGAGGAGAG[T/G]TGTATCAAGT |
ACGTTGGATGTTTGCCCAGAGCTGCTAAAG |
ACGTTGGATGTGGCAAAGAACATCTGGGAG |
ttCAGTGTTTGGAGGAGAG |
| rs2353397 |
CACCATTATT[C/T]GTATTCTTGT |
ACGTTGGATGGCTACCATAGCTGTTATATAC |
ACGTTGGATGGGTACAGCTGCAAATAGCTC |
aatcGCTCATTTCCACCATTATT |
| HTR4 | rs3995090 |
TCCTGTAGCT[A/C]ATCTCCATGA |
ACGTTGGATGAAACTCAGCCTCTTCTTCCC |
ACGTTGGATGGGAAGTGGCCATATATATTC |
atgCAACATAGACATATCATGGAGAT |
| rs6889822 |
TAGTGCACAA[A/G]GGGTAGCCAA |
ACGTTGGATGTGGGAGATCAAGAGTGAAGG |
ACGTTGGATGCCATTCACTCAAATAGCAGG |
cTTGTTGAATTTTAGTGCACAA |
| NT5DC1 | rs1052443 |
ACTGCTTGCC[A/C]ATCAGTTTGT |
ACGTTGGATGGCCTATAGAGAAGTTGGGTC |
ACGTTGGATGGTCTGTGAACAGGTACATGG |
aaAGGTACATGGTACAACAAACTGAT |
| PID1 | rs1435867 |
ATATTTCCTT[C/T]CTGATTTTAC |
ACGTTGGATGACAGTGACTCATCAAAGCTC |
ACGTTGGATGGAGTTGGGGAGAACAGATAG |
ccccTGCTAAACTTGTATATTTCCTT |
| PPT2 | rs10947233 |
GGCTGGATTT[G/T]GTCCTTTGGTC |
ACGTTGGATGAGAGGTTGGCAAACTGTGAC |
ACGTTGGATGAATGAATGTGCCTGTGTTCC |
AGTAGCAGGCTGGATTT |
| PTCH1 | rs16909898 |
GAAGACAGGA[A/G]GAGCCTTAAG |
ACGTTGGATGGAAGCAATCTGATGAACTCC |
ACGTTGGATGCCAAGGTAATCTGCCACAAC |
TCTGCCACAACTTAAGGCTC |
| rs10512249 |
TGGTGGTTGA[C/T]CTCTCACTGG |
ACGTTGGATGTCAGCCCAAAGAGAATGTGC |
ACGTTGGATGGGTGTCCTTTTCTCGTTATG |
TTATGCTTTGGTGGTTGA |
| SERPINE2 | rs7583463 |
GTTCTATTAA[A/C]CCTCACTGTA |
ACGTTGGATGCCTTATGAAAGCACATGGAG |
ACGTTGGATGACTGAAAACACACGTGTGTC |
TATGGATGAAAGTACAGTGAGG |
| THSD4 | rs12899618 |
GAGCCCTGAT[A/G]AAAGAAAAAT |
ACGTTGGATGCCCAATACTCTGGCTGGAAT |
ACGTTGGATGACAGTGTCCTGTGTGCTATG |
ccTATTGCTTTTATGAGCCCTGAT |
| TNS1 | rs2571445 |
TGGCTTCGGC[A/C/G/T]GGCGGGCCAT |
ACGTTGGATGCAGCCATGCTGGGATTGATG |
ACGTTGGATGAACAGTGGGCACCAACACTC |
ccCTCCCCCTAGTCCTGGCTTCGGC |
| TSPYL-4 | rs3749893 |
AGAAAACATC[a/G]ACTTACATTT |
ACGTTGGATGCCTAAGTCCTTGAATTCACC |
ACGTTGGATGGGTCACTCCTAGGATAATTG |
cgAGGATAATTGAGAGAAAACATC |
Genotyping by multiplex PCR
Genotyping was performed by multiplex PCR, which was
a variant of PCR enabling the simultaneous amplification of
numerous targets of interest in one reaction using more than one
pair of primers (19). We used
Mass-array Assay Design 2.0 software to design multiplex primers:
1st-PCR primer, 2nd-PCR primer and UEP primer for each SNP; primers
of the 24 SNPs are shown in Table
II.
Statistical analysis
P-values for genotype and allele frequencies were
obtained using the χ2 test with SPSS 13.0 software
(P<0.05). We excluded the SNPs in which MAF was <0.03. The
relative risk associated was estimated as an odds ratio (OR) with a
95% confidence interval (CI), as analyzed by the Woolf method. Each
SNP was tested for deviation from HWE (http://ihg2.helmholtz-muenchen.de/cgi-bin/hw/hwa1.pl).
SNPs were excluded from the analysis if they were out of HWE
(P≤0.05). Haplotype frequencies and linkage disequilibrium (LD)
analysis were evaluated using the Phase and Haploview software.
Results
Study population characteristics
The study population characteristics for those
subjects with successful genotyping are shown in Table III. Due to a lack of certain
data, the case group comprised 160 subjects and the control group
177 subjects. The two groups were matched for age, gender and
percentage of smokers. FEV1 and FVC of the case group were
significantly decreased compared to the control group
(P<0.05).
 | Table IIIDemographics of chronic obstructive
pulmonary disease patients and control subjects. |
Table III
Demographics of chronic obstructive
pulmonary disease patients and control subjects.
| Case | Control |
|---|
| No. | 160 | 177 |
| Age (years) | 54.00±13.28 | 52.00±4.84 |
| BMI
(kg/m2) | 21.62±3.77 | 23.81±2.54 |
| FEV1 (L) | 1.03±0.56a | 2.88±0.63 |
| FVC (L) | 2.10±0.92a | 3.35±0.90 |
| FEV1/FVC (%) | 51.00±14.02a | 80.00±10.03 |
| Smoking
percentage | 75.50 | 74.71 |
Result of genotyping
As a result of a quality control measure, a total of
24 SNPs were finally compared between the case and control groups.
The frequencies of the SNP [rs3749893 of testis-specific protein Y
encoded-like 4 (TSPYL-4) gene] G allele and SNP [rs1052443 of
5′-nucleotidase domain containing 1 (NT5DC1) gene] A allele were
significantly higher in the cases studies compared to the control
subjects (P=0.032, P<0.05, OR=0.692, 95% CI 0.495–0.970;
P=0.0205, P<0.05, OR=0.670, 95% CI 0.477–0.941, respectively).
The details are shown in Table
IV.
 | Table IVAllele frequencies and genotypes of
the candidate single-nucleotide polymorphisms (SNPs) in the case
and control groups. |
Table IV
Allele frequencies and genotypes of
the candidate single-nucleotide polymorphisms (SNPs) in the case
and control groups.
| SNP | Gene | SNP_ID | Case group | Control group | Case (%) | Control (%) | Allele P-value | OR (95% CI) |
|---|
|
|
|---|
| a | b | c | d | HWE | a | b | c | d | HWE |
|---|
| 1 | ADAM19 | rs2277027 | 115 | 40 | 5 | 160 | Yes | 132 | 40 | 5 | 177 | Yes | 15.63 | 14.12 | 0.580 | 1.13
(0.736–1.722) |
| 2 | ADAM19 | rs1422795 | 112 | 41 | 5 | 158 | Yes | 129 | 38 | 5 | 172 | Yes | 16.14 | 13.95 | 0.430 | 1.19
(0.774–1.820) |
| 3 | ADCY2 | rs6555465 | 45 | 69 | 41 | 155 | Yes | 50 | 94 | 33 | 177 | Yes | 48.71 | 45.20 | 0.370 | 1.15
(0.848–1.563) |
| 4 | CHRNA3 | rs1051730 | 149 | 10 | 1 | 160 | Yes | 164 | 10 | 0 | 174 | Yes | 3.75 | 2.87 | 0.530 | 1.32
(0.561–3.091) |
| 5 | FAM13A | rs2869967 | 40 | 87 | 29 | 156 | Yes | 46 | 93 | 33 | 172 | Yes | 53.53 | 47.97 | 0.150 | 1.25
(0.919–1.698) |
| 6 | GPR126 | rs7776375 | 70 | 69 | 21 | 160 | Yes | 69 | 87 | 20 | 176 | Yes | 34.69 | 36.08 | 0.710 | 0.94
(0.685–1.292) |
| 7 | GPR126 | rs11155242 | 135 | 23 | 2 | 160 | Yes | 144 | 31 | 2 | 177 | Yes | 8.44 | 9.89 | 0.520 | 0.84
(0.496–1.422) |
| 8 | GPR126 | rs6937121 | 64 | 71 | 21 | 156 | Yes | 64 | 85 | 22 | 171 | Yes | 36.22 | 37.72 | 0.690 | 0.94
(0.682–1.289) |
| 9 | GPR126 | rs3817928 | 130 | 28 | 2 | 160 | Yes | 137 | 35 | 2 | 174 | Yes | 10.00 | 11.21 | 0.610 | 0.88
(0.537–1.443) |
| 10 | HHIP | rs12504628 | 79 | 63 | 15 | 157 | Yes | 90 | 63 | 19 | 172 | Yes | 29.62 | 29.36 | 0.940 | 1.01
(0.724–1.416) |
| 11 | HHIP | rs1032295 | 98 | 51 | 8 | 157 | Yes | 101 | 63 | 9 | 173 | Yes | 21.34 | 23.41 | 0.520 | 0.89
(0.615–1.281) |
| 12 | HHIP | rs2353397 | 82 | 66 | 12 | 160 | Yes | 97 | 69 | 10 | 176 | Yes | 28.10 | 25.30 | 0.410 | 1.16
(0.821–1.628) |
| 13 | HHIP | rs6537302 | 97 | 49 | 9 | 155 | Yes | 96 | 69 | 12 | 177 | Yes | 21.61 | 26.27 | 0.160 | 0.77
(0.540–1.108) |
| 14 | HTR4 | rs3995090 | 78 | 70 | 12 | 160 | Yes | 86 | 80 | 11 | 177 | Yes | 29.38 | 28.81 | 0.870 | 1.03
(0.737–1.433) |
| 15 | HTR4 | rs6889822 | 60 | 79 | 16 | 155 | Yes | 72 | 88 | 17 | 177 | Yes | 35.81 | 34.46 | 0.720 | 1.06
(0.771–1.460) |
| 16 | NT5DC1 | rs1052443 | 85 | 64 | 7 | 156 | Yes | 78 | 75 | 20 | 173 | Yes | 25.00 | 33.24 |
0.021* | 0.67
(0.477–0.941) |
| 17 | PID1 | rs1435867 | 126 | 29 | 5 | 160 | Yes | 132 | 41 | 4 | 177 | Yes | 12.19 | 13.84 | 0.520 | 0.86
(0.550–1.356) |
| 18 | PPT2 | rs10947233 | 101 | 52 | 7 | 160 | Yes | 98 | 64 | 9 | 171 | Yes | 20.63 | 24.43 | 0.240 | 0.80
(0.558–1.158) |
| 19 | PTCH1 | rs16909898 | 138 | 21 | 1 | 160 | Yes | 150 | 25 | 2 | 177 | Yes | 7.19 | 8.19 | 0.630 | 0.87
(0.491–1.534) |
| 20 | PTCH1 | rs10512249 | 133 | 21 | 1 | 155 | Yes | 149 | 26 | 2 | 177 | Yes | 7.42 | 8.47 | 0.620 | 0.87
(0.491–1.524) |
| 21 | serpine2 | rs7583463 | 73 | 67 | 16 | 156 | Yes | 92 | 70 | 10 | 172 | Yes | 31.73 | 26.16 | 0.120 | 1.31
(0.935–1.840) |
| 22 | THSD4 | rs12899618 | 121 | 32 | 2 | 155 | Yes | 135 | 41 | 1 | 177 | Yes | 11.61 | 12.15 | 0.830 | 0.95
(0.593–1.523) |
| 23 | TNS1 | rs2571445 | 51 | 69 | 33 | 153 | Yes | 61 | 73 | 35 | 169 | Yes | 44.12 | 42.31 | 0.640 | 1.08
(0.788–1.471) |
| 24 | TSPYL4 | rs3749893 | 86 | 67 | 7 | 160 | Yes | 79 | 77 | 19 | 175 | Yes | 25.31 | 32.86 |
0.032* | 0.69
(0.495–0.970) |
Linkage disequilibrium and haplotype
analysis
Using Haploview, haplotype blocks were constructed
separately according to the confidence interval method of Gabriel
et al (20) for the cases
and controls. This method uses both an estimate of d’ and a measure
of its precision (confidence bounds) to construct haplotype blocks
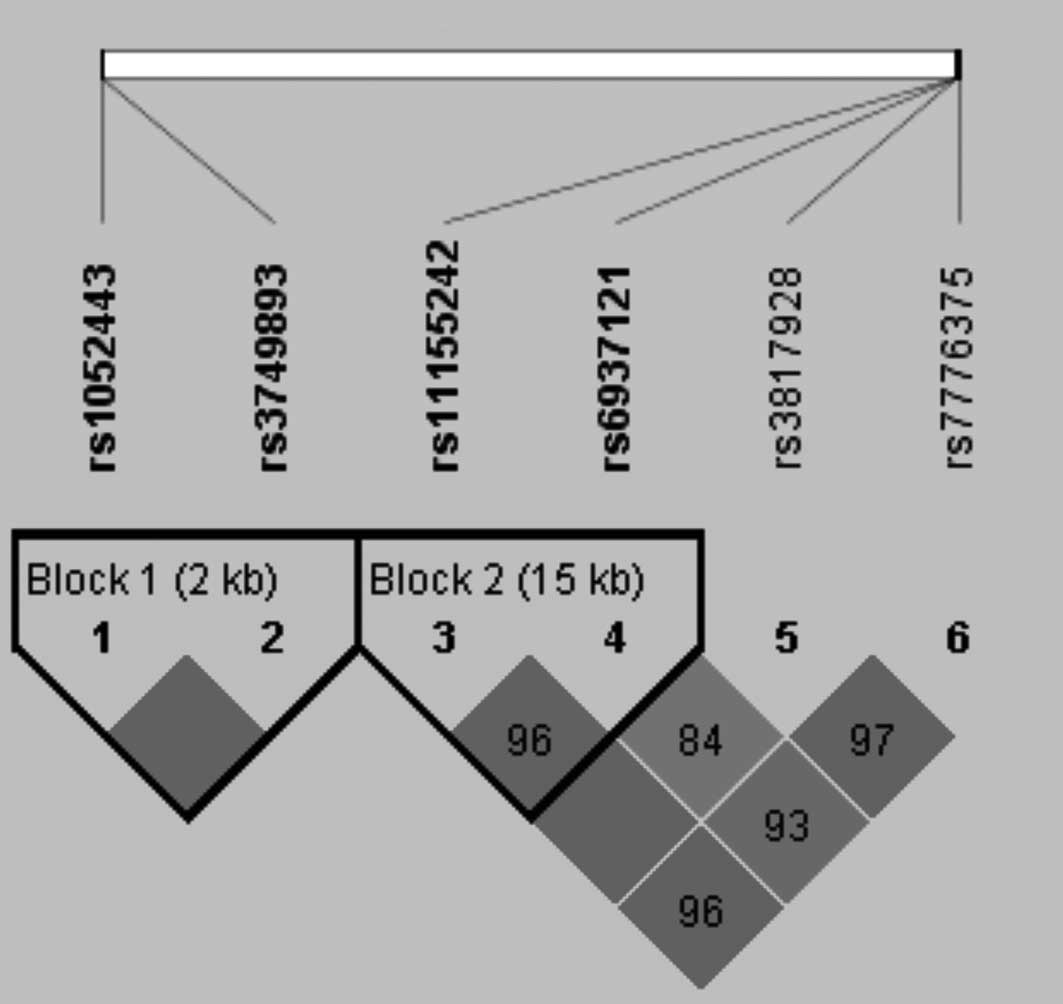
(Fig. 1). Blocks with pairwise
d’<1 have actual d’ values in the squares. Although the
estimated pairwise LD between a number of the SNPs was high, the
precision of the estimates was not sufficiently high to fulfill the
criteria for the construction of haplotype blocks utilising all the
SNPs. Two blocks of SNPs (rs1052443 and rs3749893; rs11155242 and
rs6937121) were identified that had sufficient precision to allow
construction of a haplotype block.
As the two SNPs (rs3749893 and rs1052443) are
situated on chromosome 6, we constructed the TSPYL-4 and NT5DC1
haplotypes of cases and controls. Haplotypes with frequencies
>2% were selected for the analysis. No significant difference
was observed between the two groups (Table V).
 | Table VResults of the haplotype analysis in
the case and control groups. |
Table V
Results of the haplotype analysis in
the case and control groups.
| Chromosome | Haplotype | Percentage (%) | P-value | OR (95% CI) |
|---|
|
|
| | |
|---|
| | | Case | Control | | |
|---|
| 6 | 0 | 2 | 53.750 | 44.820 | 0.10152 | 0.69891
(0.455–1.074) |
| 1 | 2 | 21.620 | 21.240 | 0.93233 | 0.97768
(0.581–1.646) |
| 1 | 0 | 33.548 | 25.007 | 0.08464 | 0.66051
(0.412–1.060) |
Genotype of TSPYL-4 and NT5DC1 SNPs, and
pulmonary function
The SNPs of TSPYL-4 and NT5DC1 were associated with
the development of COPD according to the results of our study,
while COPD is characterized by an airflow limitation that is not
fully reversible. We also investigated the relationship between the
TSPYL-4 and NT5DC1 gene polymorphisms, and the pulmonary function
(FEV1, FVC and FEV1/FVC) was then investigated using ANOVA.
In COPD patients, for rs3749893 of TSPYL-4, the mean
FEV1/FVC levels were significantly higher in AA carriers than in AG
carriers (57.00±2.16 vs. 51.99±12.27, P=0.043, P<0.05) and
FEV1/FVC levels were also significantly higher in AG carriers than
in GG carriers (51.99±12.27 vs. 47.09±12.8, P=0.016, P<0.05).
For rs1052443 of NT5DC1, CC carriers were associated with
significantly higher FEV1/FVC levels compared to CA carriers
(57.00±2.16 vs. 52.09±12.36, P=0.037, P<0.05) and CA carriers
had significantly higher FEV1/FVC levels than AA carriers
(52.09±12.36 vs. 46.83±12.65, P=0.011, P<0.05). It appears that
the rs3749893 A allele of TSPYL-4 and the rs1052443 C allele of
NT5DC1 are associated with a protective effect against the
deterioration of pulmonary function in our COPD patients. In the
control group, similar trends were observed, but these did not
reach the level of significance. The details are shown in Table VI.
 | Table VIUnivariate analysis of variance
comparing mean FEV1/FVC levels according to the different genotypes
of the TSPYL-4 and NT5DC1 single-nucleotide polymorphisms. |
Table VI
Univariate analysis of variance
comparing mean FEV1/FVC levels according to the different genotypes
of the TSPYL-4 and NT5DC1 single-nucleotide polymorphisms.
| rs no. | Genotype | Subject (n) | FEV1/FVCa | P-value |
|---|
| COPD | rs3749893 | GG | 84 | 47.09±12.8 | Reference |
| | AG | 67 | 51.99±12.27c | 0.016 |
| | AA | 7 | 57.00±2.16b | 0.043 |
| rs1052443 | AA | 85 | 46.83±12.65 | Reference |
| | CA | 64 | 52.09±12.36e | 0.011 |
| | CC | 7 | 57.00±2.16d | 0.037 |
| Control | rs3749893 | GG | 79 | 87.77±10.54 | Reference |
| | AG | 77 | 88.89±9.71 | 0.272 |
| | AA | 19 | 91.17±9.67 | 0.513 |
| rs1052443 | AA | 78 | 87.91±10.54 | Reference |
| | CA | 75 | 88.79±9.83 | 0.200 |
| | CC | 20 | 91.17±9.42 | 0.588 |
Discussion
The present study identified that SNP rs3749893 of
TSPYL-4 and rs1052443 of NT5DC1 genes was significantly associated
with susceptibility to COPD in a south Han Chinese population. In
addition, the two SNPs constitute a haploview block. Recently,
hundreds of GWAS were published, involving a number of diseases,
such as asthma, obesity, diabetes and mental illness (21–26).
GWAS for COPD were also carried out and related SNPs were reported;
however, most of the studies were performed in Caucasians, not in
Asians. In our study, we succeeded in replicating these SNPs in a
southern Han Chinese population and found that two SNPs are
associated with susceptibility to COPD.
We also demonstrated that the two gene polymorphisms
played a significant role in pulmonary function (FEV1/FVC). The
rs3749893 A allele of TSPYL-4 and the rs1052443 C allele of NT5DC1
are associated with a protective effect against the deterioration
of pulmonary function in our COPD patients. Our finding is similar
to that of the study by Hancock et al (14). Those authors conducted a
meta-analysis of GWAS, which revealed that the two gene
polymorphisms were associated with pulmonary function. However,
their study mainly referred to ethnicities other than Chinese.
The TSPYL gene is significantly homologous to TSPY,
which is expressed in the normal germ cells of fetal and adult
testes and ectopically in tumor germ cells; designated as TSPY-like
(TSPYL). TSPYL was assigned as a new member of the TSPY-SET-NAP1L1
family (27), which includes
TSPYL1, TSPYL2, TSPYL3, TSPYL4, TSPYL5 and TSPYL6 (28). Human TSPYL is mapped to chromosome
6, and murine TSPYL to chromosome 10 (27). The TSPYL gene lacks introns and
contains a coding region of 1,314 bases. The mRNA is approximately
3,200 bases in length, and the mature TSPYL protein is 437 aa
(29). Expression of TSPYL was
observed in all tissues, as well as at early onset during
development. Vogel et al (27) investigated its expression in
different tissues by northern blot analysis and RT-PCR. TSPYL is
transcribed in all probed murine tissues, including the ovary and
liver from females, as well as testes, spleen, brain, kidney,
prostate, lung, liver and heart from males.
The functions of the TSPYL gene in the pathogenesis
of COPD are far from being sufficiently studied. TSPYL may play a
role in development by altering the regulation of specific
developmental genes and contributing to region-specific chromatin
remodeling (29). TSPYL is the
putative gene for gonadoblastoma. The expression of TSPYL4 in human
lung tissue and its function remain unclear. Published studies on
the TSPYL4 gene are limited. Other members, such as TSPYL1, were
found to be associated with the 46,XY disorder of sex development,
male infertility and sudden infant death with dysgenesis of the
testes syndrome (SIDDT) (30).
TSPYL5 is one of the frequent targets of epigenetic silencing in
primary glial tumors (31). In
their study, Jung et al (32) reported that this gene is important
in the development of gastric cancer, indicating that it is one of
the potent tumor suppressor genes associated with DNA methylation.
Kim et al (33) were the
first to show that the TSPYL5 gene is partly involved in cell
growth and resistance to cytotoxic agents via regulation of the
cell level of the P21WAF/Cip1 and PTEN/AKT pathways.
NT5DC1 shows significant structural homology to
several established members of the haloacid dehydrogenase (HAD)
super-family, particularly phosphoserine phosphatase (PSP). The
catalytic mechanism of NT5DC1 is also closely correlated with that
of PSPs (34,35). Its expression and role in human
lung tissue remain to be elucidated.
Limitations of our study are missing data, which may
make it difficult to classify patients according to lung function
severity, smoking index and family history. This lack of data may
have resulted in bias towards the null, although we used strict
criteria for both the case patients and control subjects to
minimize this possibility.
In conclusion, these are the first reported SNPs in
TSPYL-4 and NT5DC1 associated with COPD in a southern Han Chinese
population. The two gene polymorphisms are crucial in pulmonary
function (FEV1/FVC). The rs3749893 A allele of TSPYL-4 and the
rs1052443 C allele of NT5DC1 are associated with a protective
effect against the deterioration of pulmonary function. SNP
rs3749893 of TSPYL-4 and rs1052443 of NT5DC1 constitute a haplotype
block, which transmits as a whole unit. However, the expression and
function of TSPYL-4 and NT5DC1 genes in human pulmonary tissue
remain to be elucidated by further experimentation.
Acknowledgements
This study was funded by the 11th Chinese National
Five-year Development Plan (2008BAI52B00). The authors thank
Shaojun Yin and Changhui Wang, Shanghai No. 10 Hospital, Xiaoyan
Jin, Changning District Central Hospital, and Boying Zhang, Luwan
District Central Hospital, and acknowledge the help of the
recruited volunteers. They also thank Yanyan Song from the
Department of Biostatistics, Shanghai Jiao Tong University, School
of Medicine. They also gratefully acknowledge the numerous sample
donors for making this study possible.
References
|
1
|
Murray CJ and Lopez AD: Alternative
projections of mortality and disability by cause 1990–2020: global
burden of disease study. Lancet. 349:1498–1504. 1997.PubMed/NCBI
|
|
2
|
Anthonisen NR, Connett JE, Kiley JP,
Altose MD, Bailey WC, Buist AS, Conway WA Jr, Enright PL, Kanner
RE, O’Hara P, et al: Effects of smoking intervention and the use of
an inhaled anticholinergic bronchodilator on the rate of decline of
FEV1. The lung health study. JAMA. 272:1497–1505. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siafakas NM, Vermeire P, Pride NB,
Paoletti P, Gibson J, Howard P, Yernault JC, Decramer M,
Higenbottam T, Postma DS, et al: Optimal assessment and management
of chronic obstructive pulmonary disease (COPD). The European
Respiratory Society Task Force. Eur Respir J. 8:1398–1420. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang X, Li L, Xiao J, Jin C, Huang K, Kang
X, Wu X and Lv F: Association of ADAM33 gene polymorphisms with
COPD in a northeastern Chinese population. BMC Med Genet.
10:132–138. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cigarette smoking and health. American
Thoracic Society. Am J Respir Crit Care Med. 153:861–865. 1996.
View Article : Google Scholar
|
|
6
|
Hodge S, Hodge G, Holmes M and Reynolds
PN: Increased airway epithelial and T-cell apoptosis in COPD
remains despite smoking cessation. Eur Respir J. 25:447–454. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Silverman EK: Progress in chronic
obstructive pulmonary disease genetics. Proc Am Thorac Soc.
3:405–408. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Silverman EK, Chapman HA, Drazen JM, Weiss
ST, Rosner B, Campbell EJ, O’Donnell WJ, Reilly JJ, Ginns L,
Mentzer S, Wain J and Speizer FE: Genetic epidemiology of severe,
early-onset chronic obstructive pulmonary disease. Risk to
relatives for airflow obstruction and chronic bronchitis. Am J
Respir Crit Care Med. 157:1770–1778. 1998. View Article : Google Scholar
|
|
9
|
Silverman EK, Mosley JD, Palmer LJ, Barth
M, Senter JM, Brown A, Drazen JM, Kwiatkowski DJ, Chapman HA,
Campbell EJ, et al: Genome-wide linkage analysis of severe,
early-onset chronic obstructive pulmonary disease: airflow
obstruction and chronic bronchitis phenotypes. Hum Mol Genet.
11:623–632. 2002. View Article : Google Scholar
|
|
10
|
DeMeo DL, Celedón JC, Lange C, Reilly JJ,
Chapman HA, Sylvia JS, Speizer FE, Weiss ST and Silverman EK:
Genome-wide linkage of forced mid-expiratory flow in chronic
obstructive pulmonary disease. Am J Respir Crit Care Med.
170:1294–1301. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Silverman EK, Palmer LJ, Mosley JD, Barth
M, Senter JM, Brown A, Drazen JM, Kwiatkowski DJ, Chapman HA,
Campbell EJ, et al: Genomewide linkage analysis of quantitative
spirometric phenotypes in severe early-onset chronic obstructive
pulmonary disease. Am J Hum Genet. 70:1229–1239. 2002. View Article : Google Scholar
|
|
12
|
Wilk JB, Chen TH, Gottlieb DJ, Walter RE,
Nagle MW, Brandler BJ, Myers RH, Borecki IB, Silverman EK, Weiss ST
and O’Connor GT: A genome-wide association study of pulmonary
function measures in the Framingham Heart Study. PLoS Genet.
5:e10004292009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pillai SG, Ge D, Zhu G, Kong X, Shianna
KV, Need AC, Feng S, Hersh CP, Bakke P, Gulsvik A, et al: A
genome-wide association study in chronic obstructive pulmonary
disease (COPD): identification of two major susceptibility loci.
PLoS Genet. 5:e10004212009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hancock DB, Eijgelsheim M, Wilk JB, Gharib
SA, Loehr LR, Marciante KD, Franceschini N, van Durme YM, Chen TH,
Barr RG, et al: Meta-analyses of genome-wide association studies
identify multiple loci associated with pulmonary function. Nat
Genet. 42:45–52. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cho MH, Boutaoui N, Klanderman BJ, Sylvia
JS, Ziniti JP, Hersh CP, DeMeo DL, Hunninghake GM, Litonjua AA,
Sparrow D, et al: Variants in FAM13A are associated with chronic
obstructive pulmonary disease. Nat Genet. 42:200–202. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Repapi E, Sayers I, Wain LV, Burton PR,
Johnson T, Obeidat M, Zhao JH, Ramasamy A, Zhai G, Vitart V, et al:
Genome-wide association study identifies five loci associated with
lung function. Nat Genet. 42:36–44. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rabe KF, Hurd S, Anzueto A, Barnes PJ,
Buist SA, Calverley P, Fukuchi Y, Jenkins C, Rodriguez-Roisin R,
van Weel C and Zielinski J: Global Initiative for Chronic
Obstructive Lung Disease: Global strategy for the diagnosis,
management, and prevention of chronic obstructive pulmonary
disease: GOLD executive summary. Am J Respir Crit Care Med.
176:532–555. 2007. View Article : Google Scholar
|
|
18
|
Gong Y, Fan L, Wan H, Shi Y, Shi G, Feng
Y, Liu J, Ni L, Pan C and Zhang R: Lack of association between the
TGF-β(1) gene and development of COPD in Asians: a case-control
study and meta-analysis. Lung. 189:213–223. 2011.
|
|
19
|
Edwards MC and Gibbs RA: Multiplex PCR:
advantages, development, and applications. Genome Res. 3:S65–S75.
1994. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gabriel SB, Schaffner SF, Nguyen H, Moore
JM, Roy J, Blumenstiel B, Higgins J, DeFelice M, Lochner A, Faggart
M, et al: The structure of haplotype blocks in the human genome.
Science. 296:2225–2229. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Klein RJ, Zeiss C, Chew EY, Tsai JY,
Sackler RS, Haynes C, Henning AK, SanGiovanni JP, Mane SM, Mayne
ST, et al: Complement factor H polymorphism in age-related macular
degeneration. Science. 308:385–389. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cantor RM, Lange K and Sinsheimer JS:
Prioritizing GWAS results: a review of statistical methods and
recommendations for their application. Am J Hum Genet. 86:6–22.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Melen E, Himes BE, Brehm JM, Weiss ST and
Lasky-Su J: Limited evidence for shared genetic factors between
asthma and obesity based on known candidate genes. Am J Respir Crit
Care Med. 181:A13052010.
|
|
24
|
Frayling TM: Genome-wide association
studies provide new insights into type 2 diabetes aetiology. Nat
Rev Genet. 8:657–662. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hinney A, Nguyen TT, Scherag A, Friedel S,
Brönner G, Müller TD, Grallert H, Illig T, Wichmann HE, Rief W,
Schäfer H and Hebebrand J: Genome wide association (GWA) study for
early onset extreme obesity supports the role of fat mass and
obesity associated gene (FTO) variants. PLoS One. 2:e13612007.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
The Psychiatric GWAS Consortium Steering
Committee. A framework for interpreting genome-wide association
studies of psychiatric disorders. Mol Psychiatry. 14:10–17. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vogel T, Dittrich O, Mehraein Y, Dechend
F, Schnieders F and Schmidtke J: Murine and human TSPYL genes:
novel members of the TSPY-SET-NAP1L1 family. Cytogenet Cell Genet.
81:265–270. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
von Lindern M, van Baal V, Wiegant J, Raap
A, Hagemeijer A and Grosveld G: Can, a putative oncogene associated
with myeloid leukemogenesis, may be activated by fusion of its 3′
half to different genes: characterization of the set gene. Mol Cell
Biol. 12:3346–3355. 1992.PubMed/NCBI
|
|
29
|
Puffenberger EG, Hu-Lince D, Parod JM,
Craig DW, Dobrin SE, Conway AR, Donarum EA, Strauss KA, Dunckley T,
Cardenas JF, et al: Mapping of sudden infant death with dysgenesis
of the testes syndrome (SIDDT) by a SNP genome scan and
indentification of TSPYL loss of function. Proc Natl Acad Sci USA.
101:11689–11694. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vinci G, Brauner R, Tar A, et al:
Mutations in the TSPYL1 gene associated with 46,XY disorder of sex
development and male infertility. Fertil Steril. 92:1347–1350.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kim TY, Zhong S, Fields CR, Fields RC, Kim
JH and Robertson KD: Epigenomic profiling reveals novel and
frequent targets of aberrant DNA methylation-mediated silencing in
malignant glioma. Cancer Res. 66:7490–7501. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jung Y, Park J, Bang YJ and Kim TY: Gene
silencing of TSPYL5 mediated by aberrant promoter methylation in
gastric cancers. Lab Invest. 88:153–160. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kim EJ, Lee SY, Kim TR, Choi SI, Cho EW,
Kim KC and Kim IJ: TSPYL5 is involved in cell growth and the
resistance to radiation in A549 cells via the regulation of
p21(WAF1/Cip1) and PTEN/AKT pathway. Biochem Biophys Res Commun.
392:448–453. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bitto E, Bingman CA, Wesenberg GE, McCoy
JG and Phillips GN Jr: Structure of pyrimidine 5′-nucleotidase type
1. Insight into mechanism of action and inhibition during lead
poisoning. J Biol Chem. 281:20521–20529. 2006.
|
|
35
|
Kim WJ, Hersh CP, DeMeo DL, Reilly JJ and
Silverman EK: Genetic association analysis of COPD candidate genes
with bronchodilator responsiveness. Respir Med. 103:552–557. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhu G, Warren L, Aponte J, Gulsvik A,
Bakke P, Anderson WH, Lomas DA, Silverman EK and Pillai SG;
International COPD Genetics Network (ICGN) Investigators. The
SERPINE2 gene is associated with chronic obstructive pulmonary
disease in two large population. Am J Respir Crit Care Med.
176:167–173. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
DeMeo D, Mariani T, Lange C, Lake S,
Litonjua A, Celedón J, Reilly J, Chapman HA, Sparrow D, Spira A, et
al: The SERPINE2 gene is associated with chronic obstructive
pulmonary disease. Proc Am Thorac Soc. 3:5022006. View Article : Google Scholar : PubMed/NCBI
|