Introduction
Endometrial cancer is the most common invasive
malignant neoplasm of the female genital system, with an estimated
46,470 diagnosed cases and 8,120 deaths in 2011 in the United
States (1). The treatment of
endometrial cancer involves surgery combined with radiotherapy,
chemotherapy and biological treatment (2). Previous studies demonstrate high
loco-regional control in early-stage endometrial cancer treated
with adjuvant pelvic external radiotherapy. However, overall
survival remains largely unaffected (3). For patients with advanced stage
disease or recurrent disease, particularly when it is not amenable
to surgical resection, systemic chemotherapy is often required as
adjuvant treatment (4). Therefore,
it is beneficial to develop a novel therapeutic agent for patients
at risk of micrometastatic disease.
Currently, more and more attention is being paid to
phytochemicals and herbal extracts due to their inhibitory
abilities against cancer cell growth and metastasis, which are
suggested to improve the anticancer effect of standard treatments
as novel chemotherapeutic agents or adjuvants (5). Triptolide (TP), a highly oxygenated
diterpene isolated from Tripterygium wilfordii Hook. F, one
of the main active components in TWHF, has been used in traditional
Chinese medicine for centuries. It has been demonstrated to be
effective in patients with a variety of inflammatory and autoimmune
diseases (6). In recent studies,
TP has also been shown to have effective antiproliferative activity
and to induce apoptosis in a broad range of types of cancer in
vitro and in vivo, including colon, breast, prostate,
urothelial and hematological cancer (7–11).
However, the effect of TP on human endometrial cancer has not yet
been well described. Thus, we attempted to identify the mechanism
of TP-induced apoptosis in human endometrial cancer cells (HEC-1B)
in vitro. Previous studies from our group demonstrated that
TP was able to induce prominent growth inhibition and apoptosis in
HEC-1B cells (12–14).
This study aimed to investigate the exact targets
and molecular mechanisms of the pro-apoptosis action of TP on
HEC-1B cells. It was reported that TP was capable of inducing
apoptosis of HEC-1B cells via the p53-independent pathway, but not
the mitochondrial-mediated pathway. Our results suggested that TP
may be a useful anticancer agent in endometrial cancer.
Materials and methods
Cell culture and drugs
The human endometrial cancer cells (HEC-1B) were
obtained from the Chinese Academy of Sciences Shanghai cell bank.
The cell lines were cultured in DMEM (Gibco BRL, Carlsbad, CA,
USA), fetal bovine serum (FBS; Gibco BRL), 100 U/ml penicillin and
100 μg/ml streptomycin in a humidified atmosphere containing 5%
CO2 at 37°C. TP was obtained from National Institutes
for Food and Drug Control and dissolved in DMSO (Sigma, St. Louis,
MO, USA). The pan-caspase inhibitor z-VAD-fmk was obtained from
Beyotime (Jiangsu, China).
Cell proliferation assay
Cells were seeded in a 96-well dish at a density of
4×103 cells per well and incubated in DMEM containing
10% FBS for 24 h. After exposure to the drug for 24, 48 and 72 h,
the cells were washed with PBS and incubated in 100 μl DMEM
containing 10 μl Cell Counting Kit-8 (CCK-8; Dojindo, Japan)
solution for 90 min. The absorbance of each well was measured at a
wavelength of 450 nm. Five duplicate wells were used for each
measurement and experiments were repeated three times.
Annexin V/PI staining assay
Following treatment with TP for 48 h,
~1×105 cells/ml cells were harvested by 0.5%
Trypsin-EDTA (Gibco BRL), washed twice with cold PBS and
resuspended in 200 μl binding buffer. Cell suspension was added to
5 μl Annexin V-FITC (Becton Dickinson, Bedford, MA, USA) and 5 μl
propidium iodide (PI; Becton Dickinson), incubated for 15 min at
room temperature in the dark according to the manufacturer’s
instructions and the percentage of apoptotic cells in a 10,000-cell
cohort was analyzed by a FACSCalibur flow cytometer (Becton
Dickinson). The early and late apoptotic/necrotic cells were
determined as the percentage of Annexin
V+/PI− and Annexin
V+/PI+ cells.
Hoechst 33342 staining
The apoptotic effect of TP on HEC-1B cells was
analyzed by DNA staining. HEC-1B cells were placed in the wells of
a 24-well plate. After 24 h cell culture, the cells were treated
with TP for 48 h. The cells were washed with PBS three times and
fixed in 4% paraformaldehyde for 10 min and stained with Hoechst
33342 at a concentration of 5 μg/ml (Beyotime) in the dark at room
temperature for 30 min. After washing with PBS twice, the
fluorescent chromosomal DNA morphology was observed by fluorescence
microscopy with a 340-nm excitation filter. Apoptotic cells were
identified by morphology and condensation of the nuclei.
Real-time PCR
Total RNA was isolated from cells using TRIzol
reagent (Invitrogen, Carlsbad, CA, USA) after treatment with TP for
48 h. High-quality RNA, having a 28S/18S rRNA ratio of 1.5:2 was
used for further experimentation. Equal quantities of RNA (1000 ng)
from each sample were used as templates for cDNA synthesis with the
PrimeScript RT Master Mix (Takara Biotechnology, Dalian, China)
according to the manufacturer’s instructions. Real-time PCR assays
were carried out using Power SYBR Green PCR Master Mix kit (Applied
Biosystems, Carlsbad, CA, USA) in a total volume of 20 μl on the
ABI Prism 7300 Sequence Detection System (Applied Biosystems). The
primer sequences for bcl-2: forward, 5′-CGGTTCAGGTACTCAGTCATCC-3′;
reverse, 5′-GGTGGGGTCATGTGTGTGG-3′; bax: forward,
5′-CCCGAGAGGTCTTTTTCCGAG-3′; reverse, 5′-CCAGCCCATGATGGTTCTGAT-3′;
β-actin: forward, 5′-TTCTACAATGAGCTGCGTGTG-3′; reverse,
5′-CAGCCTGGATAGCAACGTACA-3′. The reaction parameters of bcl-2, bax
and β-actin were as follows: one cycle at 95°C for 30 sec and 40
cycles of denaturation at 95°C for 5 sec, annealing at 60°C for 30
sec. Standard curves were prepared for target and reference
(β-actin) genes.
Western blot analysis
Cells were treated with various concentrations of TP
for the indicated time in DMEM with 10% FBS. The cells were
collected in ice-cold PBS and prepared in RIPA buffer with PMSF
(Beyotime) for 30 min at 4°C. The protein concentrations of the
cell lysates were boiled with gel-loading buffer at 100°C for 5
min. An equal amount of protein was electrophoresed on 10–15%
SDS-polyacrylamide gels and transferred onto PVDF membranes
(Millipore, Billerica, MA, USA). Membranes were blocked with 5%
nonfat milk in Tris-buffered saline with 0.1% Tween-20 (TBST) for 2
h at room temperature and then incubated with the appropriate
primary antibodies including caspase-3/9 (Cell Signaling, Danvers,
MA, USA), p53 (Bioworld, St. Louis, MO, USA) and bcl-2/bax (Santa
Cruz Biotechnology Inc., Santa Cruz, CA, USA) in PBST overnight at
4°C. After washing three times in PBST, the membranes were
incubated with horseradish peroxidase-conjugated appropriate
secondary antibodies for 90 min at room temperature. The blots were
revealed by enhanced chemiluminescene (ECL; Millipore) according to
the manufacturer’s instructions.
Statistical analysis
Values are expressed as the means ± SD. Data were
analyzed by a one-way analysis of variance (ANOVA). The difference
was considered statistically significant if P<0.05. All analyses
were performed using GraphPad Prism 5 software version. All
statistical tests were three-sided.
Results
Effect of TP on the growth of HEC-1B cell
lines
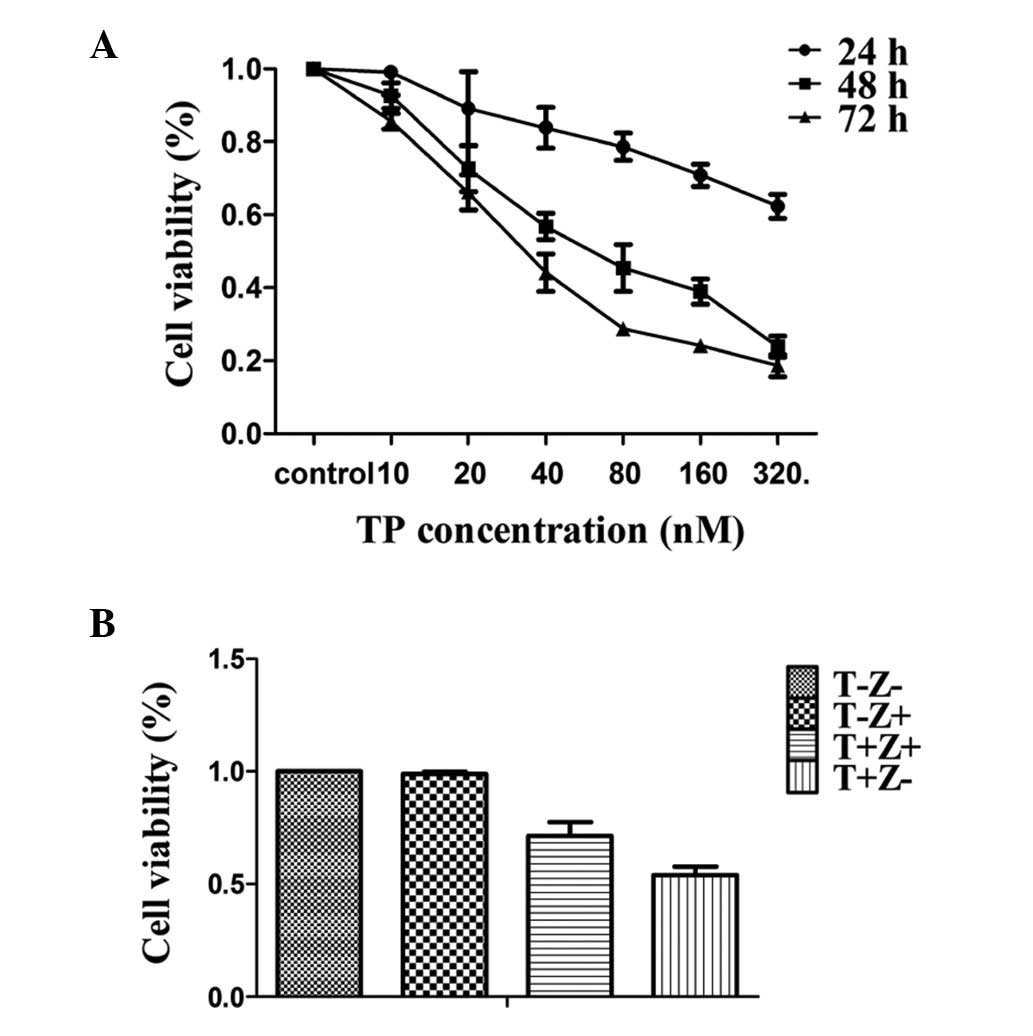
In our current study, we examined the effect of TP
on the human endometrial cancer cell line HEC-1B by CCK-8 assay.
Cells were treated with various concentrations of TP for 24, 48 and
72 h. Fig. 1A shows the
dose-dependent effect of TP after time-dependent exposure.
Following 48 h of treatment, the majority of HEC-1B cell lines were
reduced by TP therapy, indicating the existence of cell death
induced by TP. To further question the role of caspase activation
in TP-induced apoptosis, we treated HEC-1B cells with the
pan-caspase inhibitor z-VAD-fmk (20 μM) prior to TP treatment.
Pretreatment with the pan-caspase inhibitor z-VAD-fmk reduced
TP-induced apoptosis (Fig.
1B).
TP induces apoptosis in HEC-1B cells
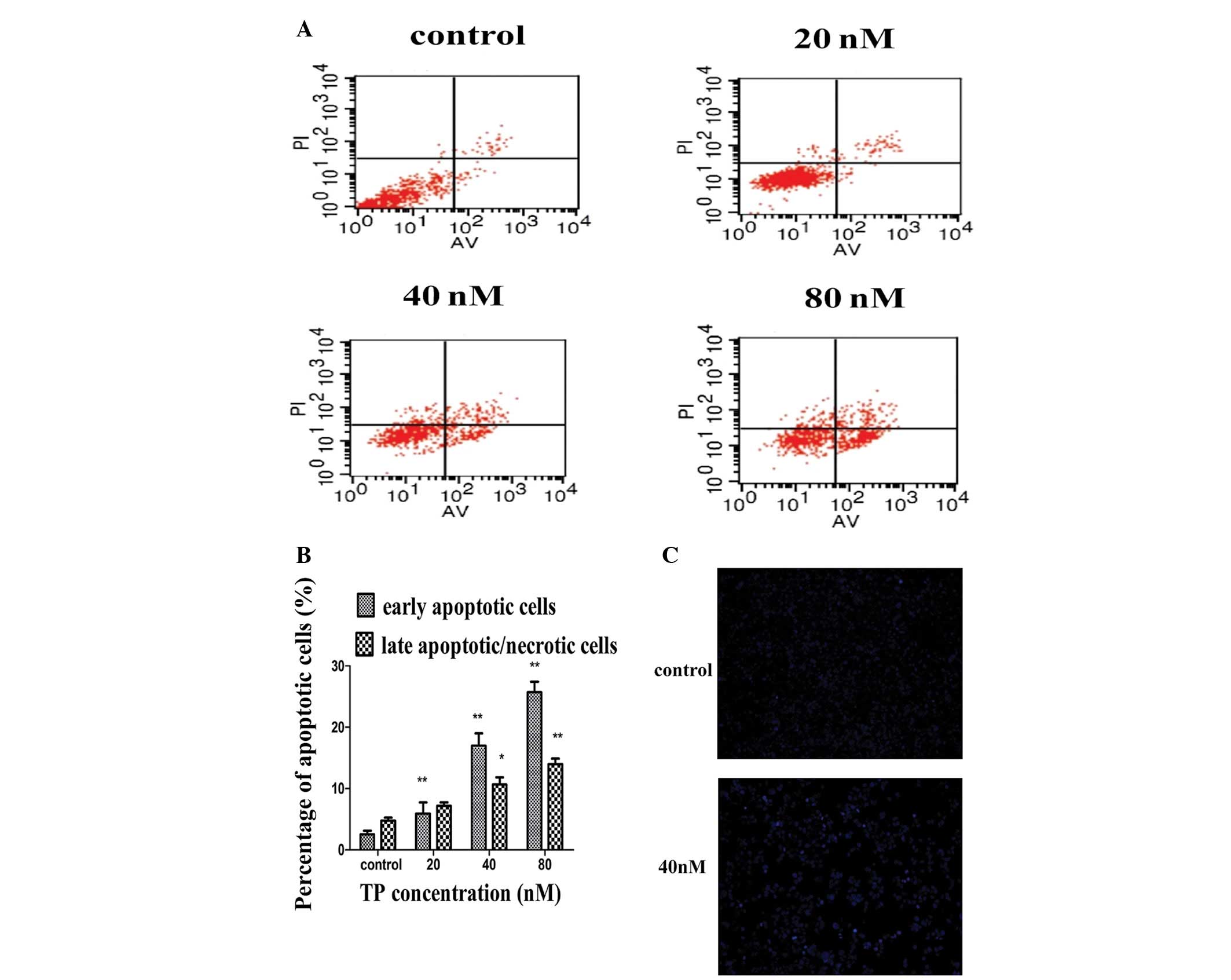
To understand the mechanisms of TP-induced cell
death in endometrial cancer cells, we examined the ability of TP to
induce apoptosis using Annexin V/PI by flow cytometry analyses. As
shown in Fig. 2A, 80 nM TP for 48
h resulted in apoptosis in a large percentage of cells. This assay
divided apoptotic cells into early and late apoptosis/necrosis. We
observed that treatment with various concentrations of TP for 48 h
resulted in a concentration-dependent increase in both early and
late apoptotic/necrotic cells. The percentage of early apoptotic
cells was larger than that of late apoptotic/necrotic cells
(Fig. 2B). Hoechst 33342 staining
revealed that there were significant morphological changes in the
nuclear chromatin of the HEC-1B cells after treatment with 40 nM TP
for 48 h. Compared with the normal morphological nuclei in the
control group, condensed chromatin or fragmented nuclei were
observed in the treated cells (Fig.
2C).
TP regulates the expression of bcl-2
family and p53 proteins in HEC-1B cells
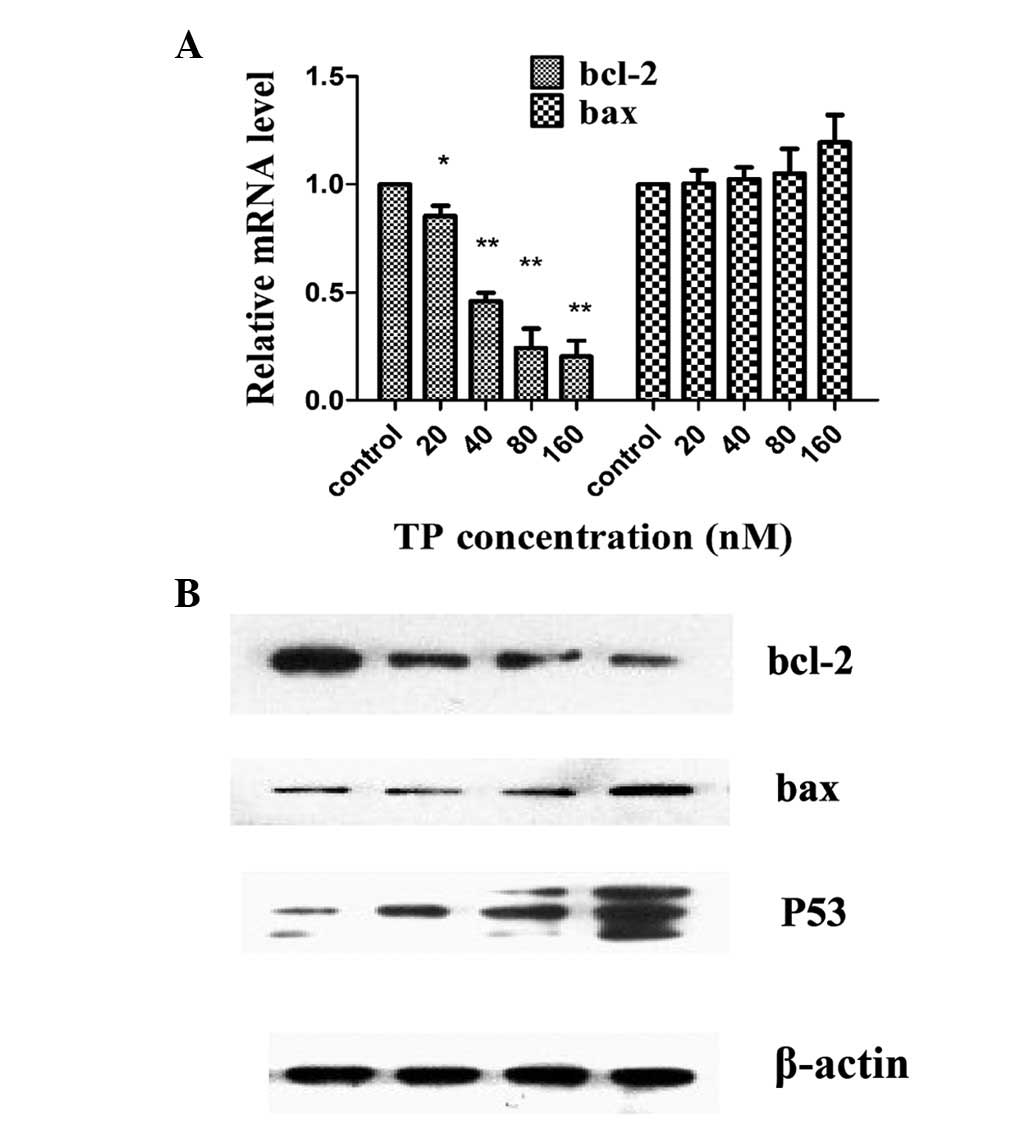
We tested the effects of TP on the expression of the
anti-apoptotic protein bcl-2 and the pro-apoptotic protein bax
using real-time PCR in HEC-1B cells. As shown in Fig. 3A, TP reduced the mRNA levels of
bcl-2 in a dose-dependent manner, while no significant changes in
the expression levels of bax were observed. Furthermore, similar
results were observed using western blotting: the protein
expression of bcl-2 was reduced, whereas bax protein expression
remained mostly constant with no significant changes. Bax protein
was slightly reduced in 80 nM TP, accompanied by a marked enhanced
p53 protein expression (Fig.
3B).
Expression of caspase-3 and caspase-9
proteins
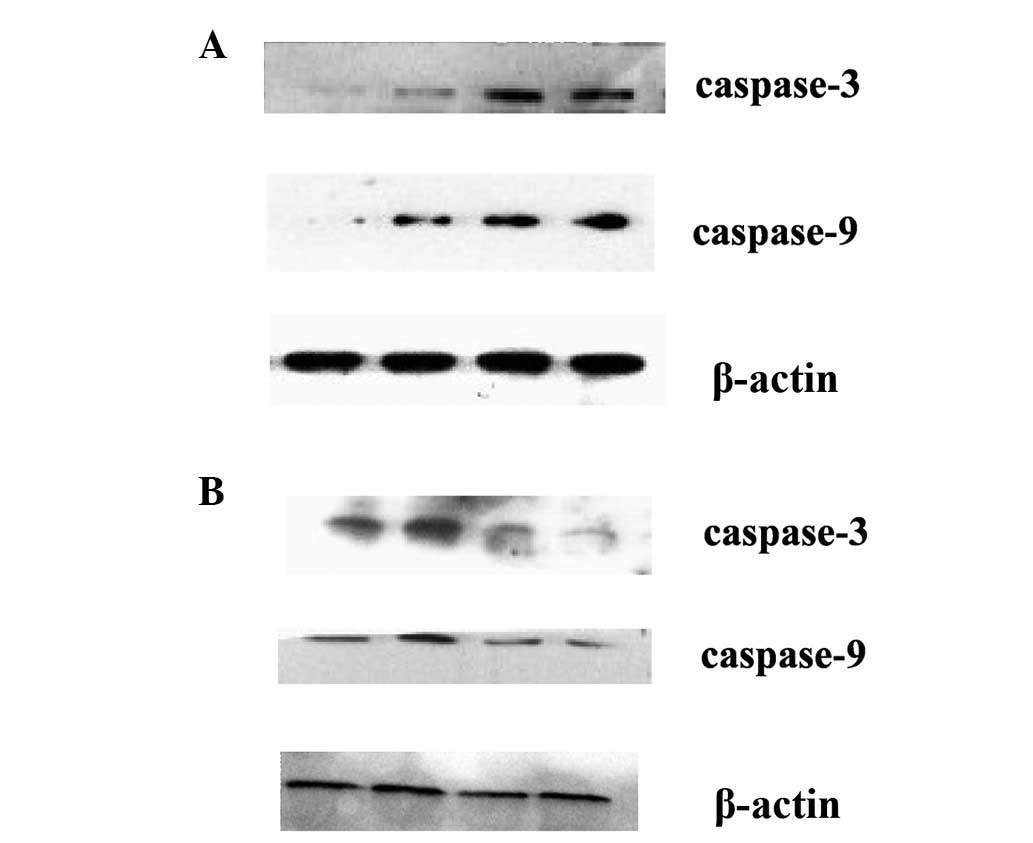
Time- and dose-dependent effects of TP on the
expression of caspase-3 and caspase-9 were examined in our study.
As shown in Fig. 4A, TP treatment
resulted in significantly increased expression of caspase-3 and
caspase-9 in HEC-1B cells. Furthermore, to assess the role of
caspase-3/9 in TP-induced apoptosis in HEC-1B cells, we pretreated
HEC-1B cells with the pan-caspase inhibitor z-VAD-fmk (20 μM). The
protein levels of caspase-3 and caspase-9 were reduced by
pretreatment with pan-caspase inhibitor z-VAD-fmk, although they
were still higher than the control (Fig. 4B).
Discussion
In our present study, we demonstrated that TP, a
compound purified from a traditional Chinese herb, induced
effective growth inhibition and cell apoptosis in human endometrial
cancer HEC-1B cells in a dose- and time-dependent manner.
Furthermore, the Annexin V/PI staining assay demonstrated that TP
induced apoptosis, but not necrosis, in HEC-1B cells. Apoptotic
cells lose asymmetry of membrane phospholipids, which induces the
phosphatidylserine (PS) translocation from the inner leaflet of the
plasma membrane to the outer layer (PS also appears on the surface
of necrotic cells), and can bind with Annexin V (15). PI is a standard flow cytometric
viability probe for staining the cellular DNA of late
apoptotic/necrotic cells with compromised cell membranes, whereas
it excluded normal or early apoptotic cells with intact membranes
(16). It is considered that
viable cells are Annexin V− and PI−, early
apoptotic cells are Annexin V+ and PI−, and
late apoptotic/necrotic cells are Annexin V+ and
PI+. Annexin V may be used in combination with PI to
detect apoptotic cells and discriminate between apoptosis and
necrosis (17). In the present
study, the Annexin V/PI assay showed that TP treatment increased
the proportion of early apoptotic HEC-1B cells. Apoptosis is
characterized by chromatin condensation, which was observed by
Hoechst 33342 staining in HEC-1B cells with TP treatment at a
concentration of 40 nM. We showed that TP markedly and
significantly increased the number of apoptotic cells in
endometrial cancer cell lines, which suggested that TP may be
useful as an effective therapeutic reagent for endometrial
cancer.
TP has been shown to promote apoptosis by inducing
mitochondrial damage and decreasing the expression of
anti-apoptotic proteins in a number of types of tumor cells
(18). This is the first study to
demonstrate involvement of the mitochondrial pathway in TP-induced
apoptosis in human endometrial cancer HEC-1B cell lines. There are
two major intracellular apoptosis signaling pathways, the
mitochondrial pathway and the death receptor pathway. The
mitochondrial pathway is regulated by members of the bcl-2 protein
family, which can be subdivided into anti-apoptotic proteins, such
as bcl-2 and bcl-xl, and pro-apoptotic proteins, such as bax and
bad (19). The bcl-2 family is an
important group of proteins that was first found to alter the
permeability of the outer mitochondrial membrane to regulate the
release of apoptotic proteins from the mitochondria (20). Bcl-2 is the first protein involved
in the regulation of apoptosis to be molecularly characterized. It
does not rely on cell division to prevent programmed cell
apoptosis. Bax translocation from the cytosol to the mitochondria
induces cytochrome c release, whereas overexpression of the
anti-apoptotic protein bcl-2 may inhibit the activity of the
pro-apoptotic protein bax by inhibiting the translocation of bax to
the mitochondria and causing aberrant proliferation and abnormal
cell survival (16,21). Clearly, the ratio of bcl-2/bax
expression is crucial for controlling cell apoptosis (22). In our study, the protein level of
bcl-2 was significantly decreased, while the protein level of bax
was unchanged by real-time PCR and western blotting. The alteration
of the bcl-2/bax ratio may be in favor of apoptosis in HEC-1B
cells.
A key event in apoptosis is the release of
mitochondrial intermembrane space proteins to the cytosol, such as
cytochrome c. Furthermore, the release of cytochrome c from the
mitochondria into the cytosol to activate caspase frequently
results in activation of the mitochondrial pathway (23). Among the variety of cellular
proteins that play a role in the induction of apoptosis, caspases
are required for the execution of apoptotic stimuli, inducing cell
death (24,25). We observed that TP exposure
potently induced apoptosis through increasing the expression of
caspase-3/9, which was associated with reduced bcl-2. Furthermore,
the caspase inhibitor z-VAD-fmk blocked TP-induced cell death,
indicating that TP-induced cell death in HEC-1B cells is
caspase-dependent. A mitochondrial breach is often triggered by
caspases rather than being required for caspase activation
(26). Therefore, the
mitochondrial apoptosis pathway plays a critical role in TP-induced
cell death, in accordance with other cancer cell lines undergoing
TP treatment (27).
There are several survival mechanisms that enable
cancer cells to escape the cell death induced by chemotherapeutic
drugs. Mutant p53 is one of particular significance (28). As p53-mediated pathways are used to
induce growth arrest or apoptosis by a number of chemotherapeutic
drugs, it has been suggested that dysfunction of p53 may partially
explain the resistance of endometrial cancer to drug treatment.
Previous studies have shown that TP promotes the apoptosis of tumor
cells via the p53-dependent pathway (10,29)
or p53-independent pathway (30,31).
In human tumors, the reactivation of p53 to induce apoptosis of
abnormal cells depends on the downstream pro-apoptotic protein bax
(32). In the present study, we
found that TP was capable of upregulating the level of p53 without
changing the expression of bax, indicating that the p53 pathway was
not effectively activated. We therefore hypothesize that TP using
p53-independent pathways may be most efficacious against
endometrial cancer. In addition, the expression of bcl-2 by tumors
may induce a variety of antitumor agents to protect cancer cells
from programmed cell death in order to confer resistance to
chemotherapy (33). Our results
showed that TP led to increased levels of p53 with downregulation
of the expression of bcl-2, while there was no change in the level
of bax expression, indicating that TP is an effective novel
therapeutic agent to resolve the aberrant tumor growth and drug
resistance in human endometrial cancer.
In conclusion, our data revealed that TP induces
human endometrial cancer HEC-1B cell apoptosis via the
mitochondrial pathway, not the p53-dependent pathway. The current
study demonstrated that TP was effective as a single agent in
inducing apoptosis of human endometrial cancer cell lines and
further suggested the potential of TP combining with other
chemotherapeutic agents or radiotherapy for the treatment of
advanced endometrial cancer.
Acknowledgements
This study is supported by a grant from the Jiangsu
Provincial Department of Sciences and Technology [BS2006072 (Wu)],
and by the Traditional Chinese Medicine Scientific Research Project
by the Jiangsu Province Administration of Traditional Chinese
Medicine [No. LZ13234 (Wu); HZ07029 (Sun)] and Project supported by
the Research Foundation of Jiangsu Cancer Hospital [No. ZS201202
(Wu)].
References
|
1
|
D’Andrilli G, Bovicelli A, Paggi MG and
Giordano A: New insights in endometrial carcinogenesis. J Cell
Physiol. 227:2842–2846. 2012.PubMed/NCBI
|
|
2
|
Amant F, Moerman P, Neven P, Timmerman D,
Van Limbergen E and Vergote L: Endometrial cancer. Lancet.
366:491–505. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hogberg T, Signorelli M, de Oliveira CF,
et al: Sequential adjuvant chemotherapy and radiotherapy in
endometrial cancer - results from two randomised studies. Eur J
Cancer. 46:2422–2431. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Horan TC, Zompa MA, Kim CTSKK, et al:
Description of the cytotoxic effect of a novel drug
abietyl-isothiocyanate on endometrial cancer cell lines. Invest New
Drug. 30:1460–1470. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen YW, Lin GJ, Chia WT, Lin CK, Chuang
YP and Sytwu HK: Triptolide exerts anti-tumor effect on oral cancer
and KB cells in vitro and in vivo. Oral Oncol. 45:562–568. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Leuenroth SJ and Crews CM:
Triptolide-induced transcriptional arrest is associated with
changes in nuclear substructure. Cancer Res. 68:5257–5266. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Johnson SM, Wang XF and Evers BM:
Triptolide inhibits proliferation and migration of colon cancer
cells by inhibition of cell cycle regulators and cytokine
receptors. J Surg Res. 168:197–205. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kang DW, Lee JY, Oh DH, et al:
Triptolide-induced suppression of phospholipase D expression
inhibits proliferation of MDA-MB-231 breast cancer cells. Exp Mol
Med. 41:678–685. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li W, Liu Y, Yu Y, et al: MAPKs are not
involved in triptolide-induced cell growth inhibition and apoptosis
in prostate cancer cell lines with different p53 status. Planta
Med. 77:27–31. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Matsui Y, Watanabe J, Ikegawa M, Kamoto T,
Ogawa O and Nishiyama H: Cancer-specific enhancement of
cisplatin-induced cytotoxicity with triptolide through an
interaction of inactivated glycogen synthase kinase-3β with p53.
Oncogene. 27:4603–4614. 2008.PubMed/NCBI
|
|
11
|
Meng HT, Zhu L, Ni WM, You LS, Jin J and
Qian WB: Triptolide inhibits the proliferation of cells from
lymphocytic leukemic cell lines in association with downregulation
of NF-κB activity and miR-16-1*. Acta Pharmacol Sin. 32:503–511.
2011.PubMed/NCBI
|
|
12
|
Yu C, Zhi-hua S, Qiang W and Jian Z:
Effects of tirptolide on endometrial cancer in human HEC-1B cells.
Prog Obstet Gynecol. 18:282–285. 2009.
|
|
13
|
Jian Z, Zhi-hua S, Qiang W and Yu C:
Growth inhibition effect of triptolide on human endometrial cancer
cells in vivo. Acta Universitatis Medicinalis Nanjing. 29:775–778.
2009.
|
|
14
|
Xiao-hao H, Zhi-hua S and Qiang W: The
effect of triptolide on proliferation and apoptosis signal
transduction pathway-PI3K/PKB in human endometrial carcinoma cell
line. Acta Universitatis Medicinalis Nanjing. 30:910–913. 2010.
|
|
15
|
Baskic D, Popovic S, Ristic P and
Arsenijevic N: Analysis of cycloheximide-induced apoptosis in human
leukocytes: fluorescence microscopy using annexin V/propidium
iodide versus acridin orange/ethidium bromide. Cell Biol Int.
30:924–932. 2006. View Article : Google Scholar
|
|
16
|
Tong JS, Zhang QH, Huang X, et al:
Icaritin causes sustained ERK1/2 activation and induces apoptosis
in human endometrial cancer cells. Plos One. 6:e167812011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wilkins RC, Kutzner BC, Truong M,
Sanchez-Dardon J and McLean JRN: Analysis of radiation-induced
apoptosis in human lymphocytes: flow cytometry using Annexin V and
propidium iodide versus the neutral comet assay. Cytometry.
48:14–19. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang JS, Chung JG, Wu PP, et al:
Triptolide induces apoptosis in human adrenal cancer NCI-H295 cells
through a mitochondrial-dependent pathway. Oncol Rep. 25:551–557.
2011.PubMed/NCBI
|
|
19
|
Cotter TG: Apoptosis and cancer: the
genesis of a research field. Nat Rev Cancer. 9:501–507. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sasi N, Hwang M, Jaboin J, Csiki I and Lu
B: Regulated cell death pathways: new twists in modulation of BCL2
family function. Mol Cancer Ther. 8:1421–1429. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xu B, Guo XX, Mathew S, Armesilla AL,
Cassidy J, Darling JL and Wang WG: Triptolide simultaneously
induces reactive oxygen species, inhibits NF-κB activity and
sensitizes 5-fluorouracil in colorectal cancer cell lines. Cancer
Lett. 291:200–208. 2010.PubMed/NCBI
|
|
22
|
Antonsson B: Bax and other pro-apoptotic
Bcl-2 family ‘killer-proteins’ and their victim the mitochondrion.
Cell Tissue Res. 306:347–361. 2001.
|
|
23
|
Kuhar M, Sen S and Singh N: Role of
mitochondria in quercetin-enhanced chemotherapeutic response in
human non-small cell lung carcinoma H-520 cells. Anticancer Res.
26:1297–1303. 2006.PubMed/NCBI
|
|
24
|
Kim MJ, Lee TH, Kim SH, Choi YJ, Heo J and
Kim YH: Triptolide inactivates Akt and induces caspase-dependent
death in cervical cancer cells via the mitochondrial pathway. Int J
Oncol. 37:1177–1185. 2010.PubMed/NCBI
|
|
25
|
Yang M, Huang J, Pan HZ and Jin J:
Triptolide overcomes dexamethasone resistance and enhanced
PS-341-induced apoptosis via PI3k/Akt/NF-κB pathways in human
multiple myeloma cells. Int J Mol Med. 22:489–496. 2008.PubMed/NCBI
|
|
26
|
Cory S and Adams JM: The bcl2 family:
regulators of the cellular life-or-death switch. Nat Rev Cancer.
2:647–656. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Phillips PA, Dudeja V, McCarroll JA, et
al: Triptolide induces pancreatic cancer cell death via inhibition
of heat shock protein 70. Cancer Res. 67:9407–9416. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhu W, Hu H, Qiu P and Yan GM: Triptolide
induces apoptosis in human anaplastic thyroid carcinoma cells by a
p53-independent but NF-κB-related mechanism. Oncol Rep.
22:1397–1401. 2009.PubMed/NCBI
|
|
29
|
Yao JC, Jiang ZZ, Duan WG, et al:
Involvement of mitochondrial pathway in triptolide-induced
cytotoxicity in human normal liver L-02 cells. Biol Pharm Bull.
31:592–597. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wan CK, Wang C, Cheung HY, Yang M and Fong
WF: Triptolide induces Bcl-2 cleavage and mitochondria dependent
apoptosis in p53-deficient HL-60 cells. Cancer Lett. 241:31–41.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Carter BZ, Mak DH, Schober WD, et al:
Triptolide sensitizes AML cells to TRAIL-induced apoptosis via
decrease of XIAP and p53-mediated increase of DR5. Blood.
111:3742–3750. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Amundson SA, Myers TG and Fornace AJ Jr:
Roles for p53 in growth arrest and apoptosis: putting on the brakes
after genotoxic stress. Oncogene. 17:3287–3299. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mano Y, Kikuchi Y, Yamamoto K, et al:
Bcl-2 as a predictor of chemosensitivity and prognosis in primary
epithelial ovarian cancer. Eur J Cancer. 35:1214–1219. 1999.
View Article : Google Scholar : PubMed/NCBI
|