Introduction
Ovarian cancer is a common type of malignant tumor
of the female reproductive organs, which is occurring with an
increasing incidence (1–4). However, epithelial ovarian cancer,
with the highest mortality rate of all types of gynecological
tumor, poses a serious threat to the lives of women. As a result of
ovarian embryonic development, the tissue anatomy and endocrine
function are complex. The tissue anatomy and endocrine function are
complex, the early symptoms of ovarian cancer are inconsistent and
there are no established primary prevention measures. Surgery
performed on patients with epithelial ovarian cancer identified
that the tumor, which was confined to the ovary, accounted for only
30% of the cancer, with the majority spreading to the uterus,
bilateral ovaries, omentum and pelvic organs (3). Neither surgery nor chemotherapy is
able to achieve a satisfactory outcome. In recent years, with the
continuous progression of scientific research, biological
therapeutic strategies for ovarian cancer have become a point of
interest, and certain studies have commenced clinical trials with
significant progress (5). Ovarian
cancer biotherapy includes gene therapy and immunotherapy. In
animal experiments and certain stage I, II and III clinical trials,
biotherapy has been effective, and may present as a novel treatment
modality following surgery, chemotherapy and radiotherapy (5,6).
In all organisms, the essential biological function
is the biological clock. Numerous complex biological functions
in vivo are controlled by regulation of the endogenous
biological clock, with aspects of behavior and physiology
demonstrating a 24-h rhythm (7–9).
Casein kinase 1 (CK1), three Period genes
(Per1, Per2 and Per3), two Cryptochrome
genes (Cry1 and Cry2), Clock and Bmal1
have been identified as core circadian clock genes (10,11).
The mammalian Per genes are key regulators of circadian
rhythm.
Previous studies found that biological clock rhythms
are associated with cell cycle regulation, and the circadian rhythm
system is involved in the cyclic processes of cell growth and
apoptosis (12–14). In addition, it has been reported
that changes in circadian rhythm are associated with human and
other mammalian tumor formation (15,16).
Further studies identified that knock down of Per2 impaired
the normal circadian rhythms of behavior and physiology, and
significantly increased the incidence of the tumor and
proliferative phenotype (17–19).
These findings demonstrate that Per2 is important in
carcinogenesis. In the study by Fu et al (18), Per2 expression was closely
associated with the occurrence of tumors, with the risk of tumor in
Per2 mutant mice being significantly higher than that of
wild-type mice. The Per2 mutant mice underwent gamma
irradiation and, compared with the wild-type mice, demonstrated
increased susceptibility to the gamma rays and a higher risk of
cancer (18). Therefore,
Per2 maintains normal biological rhythms; however, may also
be involved in tumor growth and apoptosis, exerting an inhibitory
effect on tumor growth.
As a malignant tumor of the female reproductive
system, ovarian tumors are closely associated with hormone
secretion. Furthermore, female hormone secretion is closely
associated with the circadian rhythm and sleep/wake cycles. At
present, and to the best of our knowledge, there are few studies
regarding the circadian gene, Per2 and ovarian cancer.
Therefore, in the present study, the changes of Per2 in
ovarian cancer tumor tissue samples at different stages and the
possible role of Per2 were analyzed. Nude mice xenograft
models of ovarian cancer were used, and Per2 was
overexpressed by exogenous infusion to detect its effect on tumor
growth and metastasis, and elucidate the potential underlying
mechanism.
Materials and methods
Specimen source
In order to investigate the change of Per2
during the development of ovarian cancer, archived
paraffin-embedded specimens of epithelial ovarian cancer, which had
been collected during January 2010 to December 2013 from the First
Hospital of Shanxi Medical University (Taiyuan, China), were
obtained. None of the patients had received chemotherapy or
immunotherapy prior to surgery. The clinical stage was established
according to The International Federation of Gynecology and
Obstetrics (FIGO) staging system (20); there were eight cases of malignant
epithelial ovarian tumor stage (malignant stage) I, eight cases of
malignant stage II, and six cases of malignant stage III. Six cases
of benign epithelial tumor served as the control group. Written
informed consent was obtained from the patients for the
paraffin-embedded specimens used in the current study and the
animal experiments were approved by the ethics review committee of
the First Hospital of Shanxi Medical University. The research was
conducted in strict accordance with the provided scheme and
relevant provisions of the medical ethics committee of Shanxi
Medical University.
Animal and reagents for in vivo
research
A total of 28 specific-pathogen-free (SPF), female
BALB/c nude mice (body weight, 13–15 g; age, 4 weeks) were obtained
from Shanghai Silaike Experimental Animals Co., Ltd. (Shanghai,
China). The SK-OV-3 human ovarian cancer cell line was obtained
from Peking Union Medical College (Beijing, China). The recombinant
plasmid, pcDNA3.1(+)-Per2 and empty plasmid,
pcDNA were obtained from Benyuan Zhengyang Gene Technology Co.,
Ltd. (Beijing, China). The Plasmid Extraction and Purification kit
was purchased from Hangzhou Bioer Technology Co., Ltd. (Beijing,
China) and, Invitrogen Lipofectamine 2000 was obtained from Thermo
Fisher Scientific, Inc. (Waltham, MA, USA).
Ethics statement
All surgical and animal care procedures were
approved by the Medical Ethics Committee of Shanxi Medical
University (Shanxi, China). All research was carried out in strict
accordance with the guidelines provided by the Medical Ethics
Committee.
Cell culture
SK-OV-3 cells were cultured in RPMI-1640 containing
15% fetal calf serum (Zhejiang Tianhang Biological Technology Co.,
Ltd., Zhejiang, China), and maintained in an atmosphere of 5%
CO2 at 37°C. Cells were cultured until the logarithmic
growth phase was reached. The cells were harvested, counted and the
cell number was adjusted to 2.5×107 cells/ml.
Establishment and grouping of the nude
mice xenograft models
The BALB/c nude mice were housed in SPF breeding
units. Each nude mice received a subcutaneous injection of 200
µl SK-OV-3 cell suspension (concentration,
2.5×107 cells/ml; ~5×106 cells) into the
central lateral region of the left axilla. The transplanted tumor
had developed after seven days and the tumor diameter was ~4–5 mm
14 days after the injection, which indicated successful
construction of the model. There were 24 successful mouse models,
which were randomly divided into a control group and two
experimental groups, the recombinant plasmid group and the empty
plasmid group, with eight mice per group.
Gene therapy
Following successful establishment of the animal
model, a vernier caliper was used for measurement of the
subcutaneous tumor size every 3 days. Gene therapy was initiated
when the tumor diameter reached 0.4 cm. Gene transfection was
performed using Lipofectamine 2000, which was directly injected
into the tumor and at the tumor edges by multi-point injection. The
treatment strategies were as follows: i) Control group, local tumor
injection once every three days, a total of five times, with 100
µl phosphate-buffered saline (PBS) per mouse; ii) empty
plasmid group, local tumor injection once every three days, a total
of five times, with 100 µl pcDNA3.1 (100 mg/l) per mouse, in
addition to a 25-µl injection of Lipofectamine 2000 for
transfection. iii) Per2 group, multi-point tumor injection
once every three days, a total of five times, of 100 µl
pcDNA3.1(+)-Per2 (100 mg/l) per mouse, in
addition to a 25-µl injection of Lipofectamine 2000 for
transfection. Every three days, vernier caliper measurements of the
mouse tumor length (L) and short diameter (W) were taken. Two weeks
after treatment cessation the nude mice were sacrificed by cervical
dislocation, and the tumor volume [V (mm3) = L ×
W2/2] and weight (g) were measured to calculate the
tumor inhibition rate (%) as follows: % = (control group mean tumor
weight - treatment group mean tumor weight) / control group mean
tumor weight × 100.
Reagents
A High Pure FFPE RNA extraction kit (Comwin Biotech
Co., Ltd., Beijing, China) was used to extract RNA from
paraffin-embedded tissue. In addition, a SYBR Green Real-Time
polymerase chain reaction (PCR) kit (Roche Diagnostics,
Indianapolis, IN, USA) was used to perform qPCR.
Per2 RNA expression levels in tumor
tissue and tumor metastasis-associated gene, MTA-1 and suppressor
gene, nm23-H1 in mouse tumors, as detected by reverse
transcription-quantitative PCR (RT-qPCR)
The RT conditions were as follows: RNA (11
µl; 1 µg) and 1 µl random primers (0.2
µg/ml) were incubated at 65°C for 5 min; 4 µl 5X
Buffer, 3 µl dNTP (10 mmol/l), l µl RNA enzyme
inhibitor (20 U/µl) and l µl reverse transcriptase
(20 U/µl) were added [from a RevertAid First Strand cDNA
Synthesis Kit (Thermo Fisher Scientific, Inc., Pittsburgh, PA,
USA)] and incubated at 25°C for 10 min, followed by incubation at
42°C for 1 h and at 72°C for 15 min. The qPCR reaction system was
as follows: Fast Start Universal SYBR Green Master (ROX; 10
µl; Roche Diagnostics), 0.5 µl upstream primer (15
µM), 0.5 µl downstream primer (15 µM), 2
µl cDNA, 7 µl DNAse- and RNase-free water, with a
total volume of 20 µl. The primer sequence for the
Per2 gene (GenBank® gene sequence coding
AF036893) was as follows: Forward,
5′-GCAGGCTCCACCATGAATGGATACGTGGACTTCT-3′ and reverse,
5′-CAAGAAAGCTGGGTGTTACGTCTGGGCCTCTATCCT-3′. The primer sequence for
MTA-1 was as follows: Forward, 5′-CGCTCAAGTCCTACCTGGAG-3′
and reverse, 5′-TGGTACCGGTTTCCTACTCG-3′. The primer sequence for
nm23-H1 was as follows: Forward, 5′-ACCTTCATTGCGATCAAACC-3′
and reverse, 5′-GGCCCTGAGTGCATGTATTT-3′. The primer sequence for
GAPDH was as follows: Forward, 5′-AGAGCTACGAGCTGCCTGAC-3′ and
reverse, 5′-AGCACTGTGTTGGCGTACAG-3′. All primer sequences were
designed and synthesized by Sangon Biotech Co., Ltd. The PCR
reaction conditions were as follows: 94°C initial denaturation (10
min); 94°C denaturation for 15 sec and 60°C annealing and
elongation for 60 sec, for 40 cycles. Quantitative PCR was
conducted to obtain the Cq value. The difference between the Cq of
the sample determination gene, Per2, and the Cq of the
internal control gene, β-actin, (ΔCq) was calculated as follows:
ΔCq = CqPer2 − Cqβ-actin and ΔΔCq was
obtained by subtracting the ΔCqexperimental group from
the ΔCqcontrol group. To determine the expression of
Per2 in the experimental group compared with the normal
control group (for multiple sample variation), 2−ΔΔCq
was calculated (21).
Western blot analysis of Per2, MTA-1,
nm23-H1, PI3K and PKB kinase protein expression levels
The tumor tissue protein was extracted from
paraffin-embedded tissue (Per2) or nude mice with ovarian
cancer xenografts (MTA-1, nm23-H1, PI3K and PKB),
using RIPA buffer (Beyotime Institute of Biotechnology, Haimen,
China) and the concentration was determined using a bicinchoninic
acid assay (Pierce Biotechnology, Inc., Rockford, IL, USA).
Proteins (at 10 µg/µl) were separated on a 12% sodium
dodecyl sulfate-polyacrylamide gel by electrophoresis, and
transferred onto a polyvinylidene fluoride membrane (Sigma-Aldrich,
St. Louis, MO, USA). The membranes were incubated with antibodies
to visualize the proteins. These were metastasis-associated gene 1
(MTA-1; cat. no. sc-9445; goat polyclonal), non-metastasis protein
23-H1 (nm23-H1; cat. no. sc-343; rabbit polyclonal), Per2
(cat. no. sc-25363; rabbit polyclonal), phosphatidylinositol
3-kinase (PI3K; cat. no. sc-8010; mouse monoclonal), protein kinase
B (PKB) kinase (cat. no. sc-17766) (all 1:500 dilution) and β-actin
(cat. no. sc-47778; mouse monoclonal; reference gene; dilution,
1:1,000) antibodies. Membranes were then incubated with the
secondary antibodies bovine anti-goat immunoglobulin G
(IgG)-horseradish peroxidase (HRP) (cat. no. sc-2378) and bovine
anti-rabbit IgG-HRP (cat. no. sc-2379) (both used at a 1:2,000
dilution). All antibodies were all purchased from Santa Cruz
Biotechnology, Inc. (Heidelberg, Germany). Proteins were detected
with enhanced chemiluminescence (ECL) using Pierce ECL Western
Blotting substrate (Pierce Biotechnology, Inc.). Analysis of the
immune response and exposure were conducted using Image Lab imaging
software version 5.2 (Bio-Rad Laboratories, Inc., Hercules, CA,
USA). This experiment was conducted five times.
Statistical analysis
SPSS version 16.0 statistical software (SPSS, Inc.,
Chicago, IL, USA) was used for statistical analysis. Multi-group
comparisons were performed using analysis of variance, following a
Kolmogorov-Smirnov normality test. Bonferroni's test was used for
comparisons between two groups. Data are presented as mean ±
standard deviation. A P-value <0.05 was considered to indicate a
statistically significant difference.
Results
Per2 expression level varies at different
tumor stages
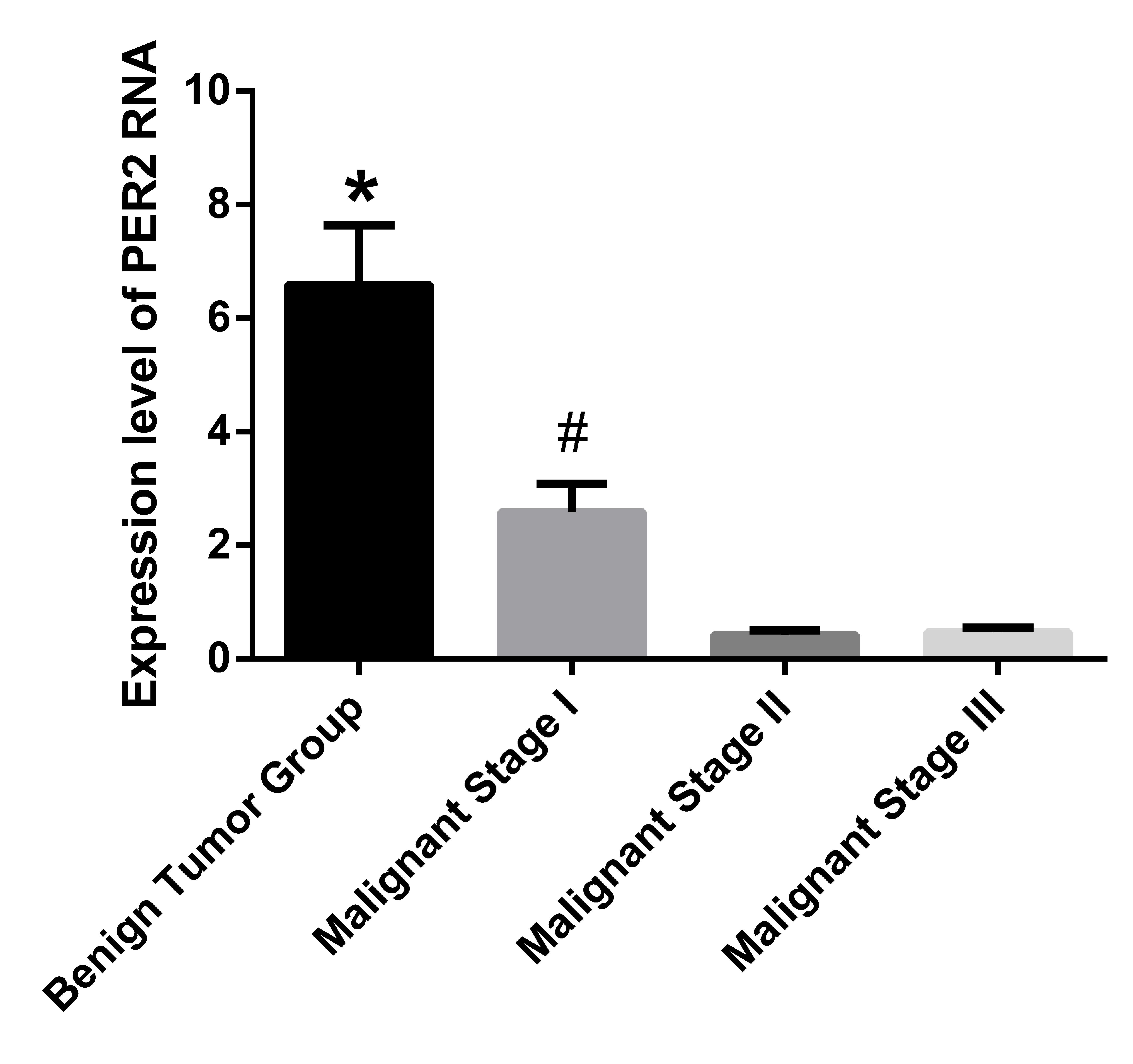
The expression of Per2 RNA in tumor tissue
samples was detected by RT-qPCR. The expression levels of
Per2 RNA in the benign tumor group were identified to be
significantly greater when compared with the three other groups
(P<0.001). Furthermore, the malignant stage I group demonstrated
significantly higher Per2 RNA expression levels versus the
malignant stage II and III groups (P<0.001). No significant
difference was identified between the malignant stage II and III
groups. The RNA expression level of Per2 in the benign tumor
group was ~14 times that of malignant stage II group and ~15 times
that of the malignant stage III group (Fig. 1).
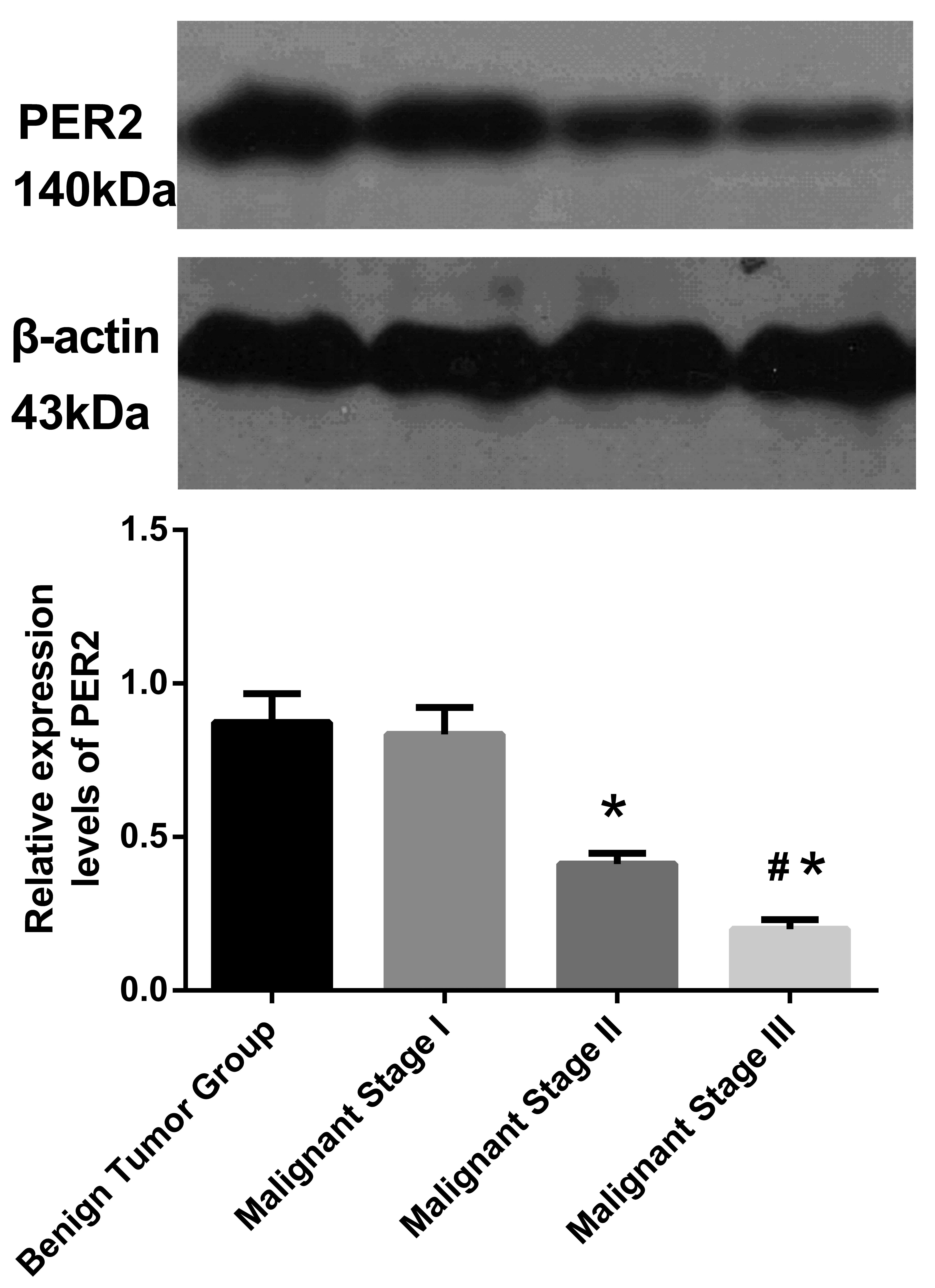
Per2 protein expression in tumor tissue
samples was detected by western blotting
The expression levels of Per2 at different
malignant tumor stages were varied; as the degree of tumor
malignancy increased, the Per2 expression gradually
decreased (single factor variance analysis between the four groups:
F=1,785; P<0.001). No significant difference was identified
between the benign tumor group and the malignant stage I group. A
significant difference was observed between the malignant stage I
group, and malignant stage II and III groups (P<0.05), in
addition, the difference between malignant stage II and III was
significant (P<0.05; Fig.
2).
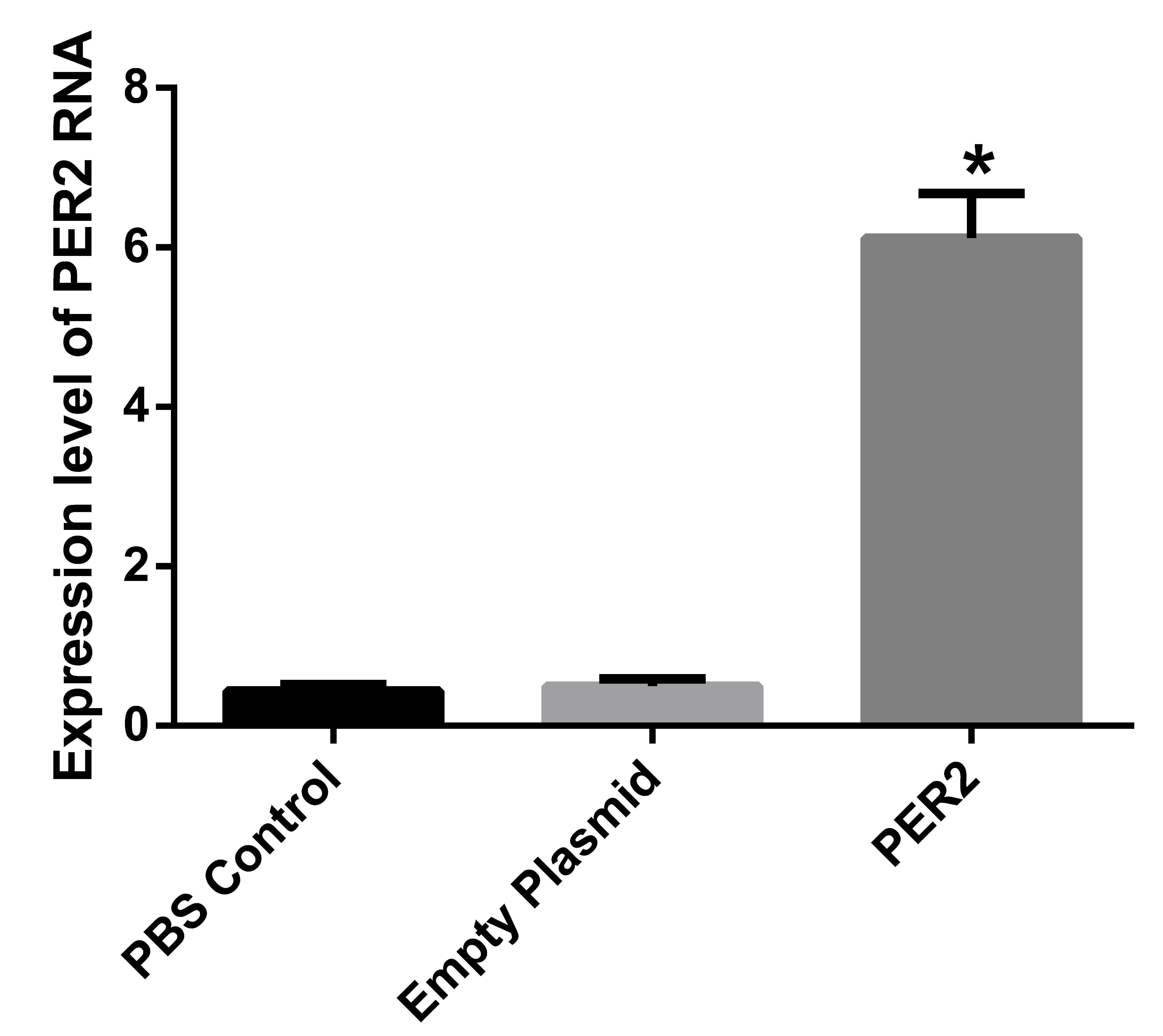
Per2 overexpression exerts inhibitory
effects on ovarian cancer in nude mice xenograft models
Per2 was transfected and successfully
expressed in tumor tissue samples. In order to confirm that the
liposome-mediated Per2 had been converted into a functional
Per2 gene in the body, a specific gene was designed and the
expression of Per2 protein was detected by western blot
analysis.
No statistically significant difference was
identified between the PBS control group and the empty plasmid
group; however, the Per2 expression level in the transfected
group was significantly greater compared with the other two groups,
(P<0.01). The Per2 expression level was ~11 times greater
compared with the other two groups (Fig. 3).
Comparison between tumor growth and tumor
inhibition
Three mice from the PBS control group and two mice
from the empty plasmid group exhibited ascites. The mice with
ascites lost weight, consumed less water and presented with
cancer-associated cachexia. Furthermore, axillary and inguinal
regions in the control and empty plasmid group mice demonstrated
varying lymph node sizes and metastasis, and a range of sizes of
satellite foci were apparent surrounding the transplanted tumor. In
the treatment group overexpressing Per2, ascites and
metastasis were not obvious, and visible satellite foci were
apparent in one of the mice (data not shown).
The interaction between experiment duration and
group was determined and the results are presented in Table I (F=22.72; P<0.001). The tumor
volumes were observed to increase in a time-dependent manner. Upon
cessation of treatment, the mean tumor weight of mice from the
recombinant Per2 plasmid group was significantly less than
that of the control and empty plasmid groups (F=26.70;
P<0.001).
 | Table IChange in tumor volume prior to and
following treatment. |
Table I
Change in tumor volume prior to and
following treatment.
| Group | Volume
(mm3)
|
|---|
| Initial | Final |
|---|
| PBS control | 72.56±6.24 | 845.53±110.00 |
| Empty plasmid | 72.49±7.91 |
835.32±105.68a |
| Per2 | 71.24±5.73 |
486.38±70.12b |
Following treatment the mean tumor weights of nude
mice were 4.73±0.95, 4.55±1.06 and 2.7±0.52 g, in the control,
empty plasmid and Per2 groups, respectively. The tumor
weights of mice in the Per2 group were significantly lighter
than those of the control and empty plasmid groups (P<0.05). In
addition, the inhibitory rate of the Per2 group was 42.9%, a
significant increase compared with the other two groups (Table II).
 | Table IIMean tumor weight and tumor
inhibition rate. |
Table II
Mean tumor weight and tumor
inhibition rate.
| Group | Tumor weight after
transfection, g | Tumor inhibition
rate, % |
|---|
| PBS control | 4.73±0.95 | 0.0 |
| Empty plasmid | 4.55±1.06 | 3.8 |
| Per2 | 2.7±0.52 | 42.9a |
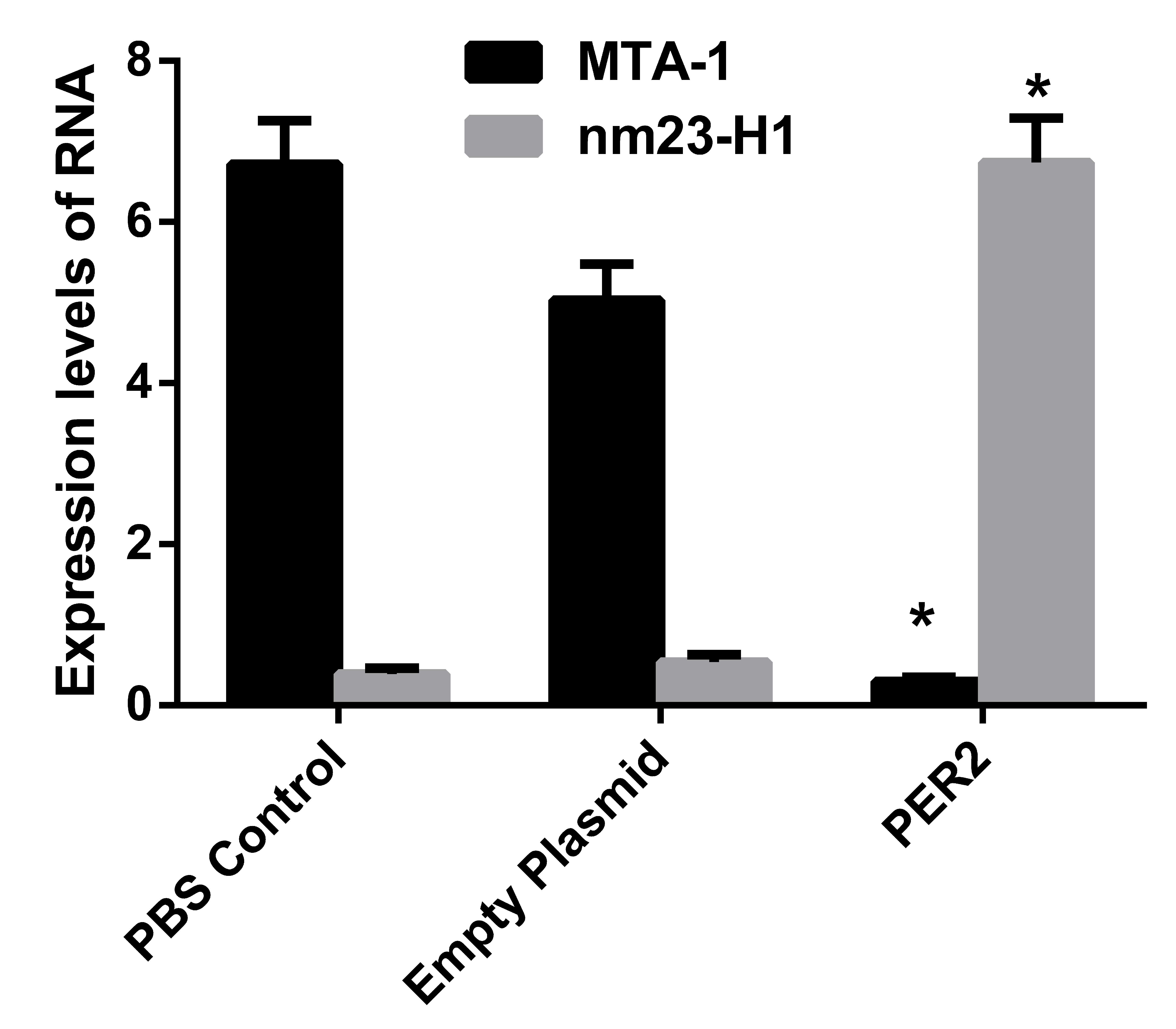
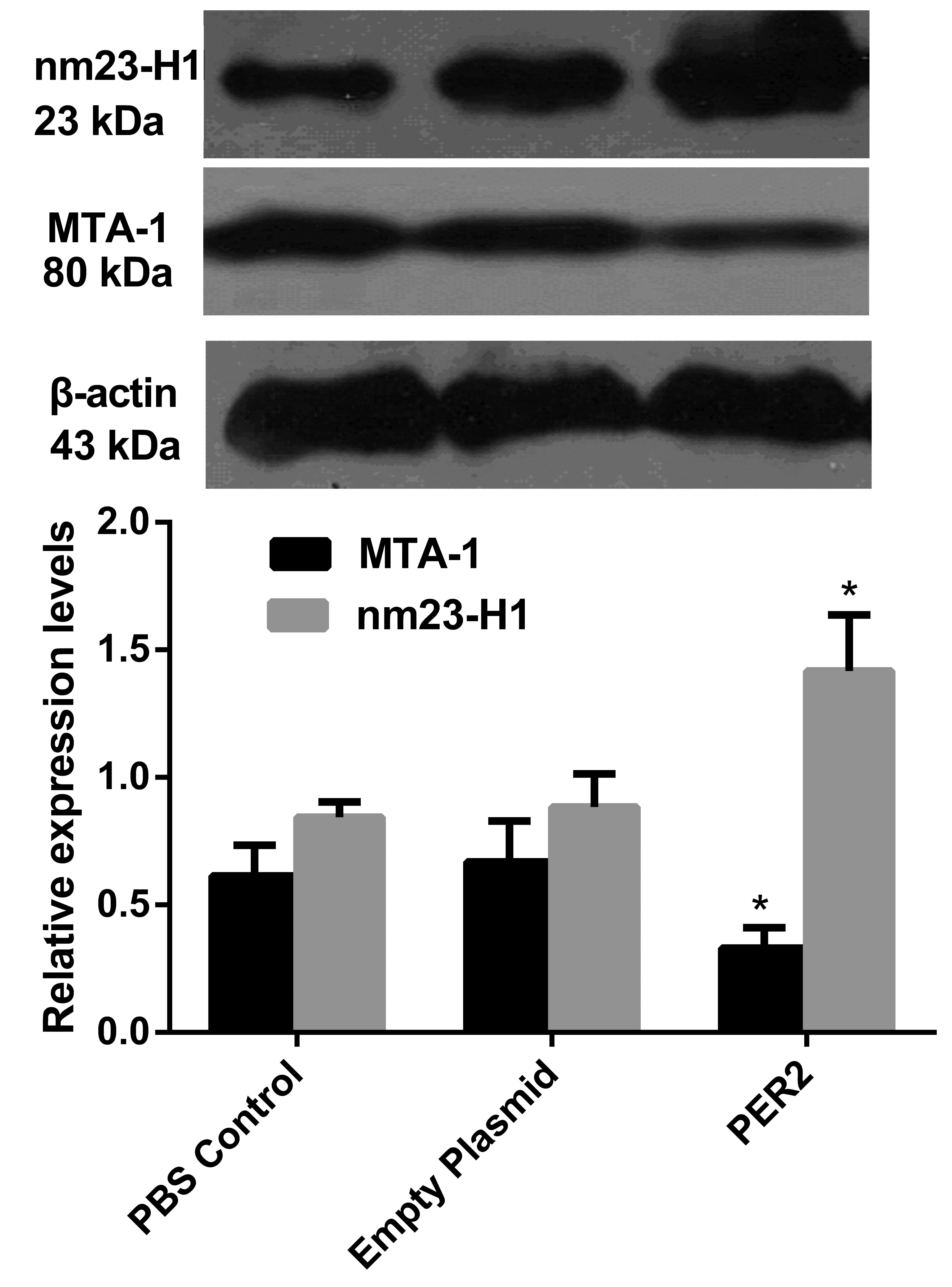
RT-qPCR detection of MTA-1 and nm23H1 RNA
expression levels
No significant difference was identified between the
control and the empty plasmid groups; however, the Per2
transfected group was significantly different when compared with
the other two groups (P<0.001). In the Per2
overexpression group, the MTA-1 RNA expression was
significantly decreased (~1/10 of the RNA expression of the PBS
control group). However, the RNA expression of nm23-H1 was
increased by ~12 times when compared with the control group
(Fig. 4).
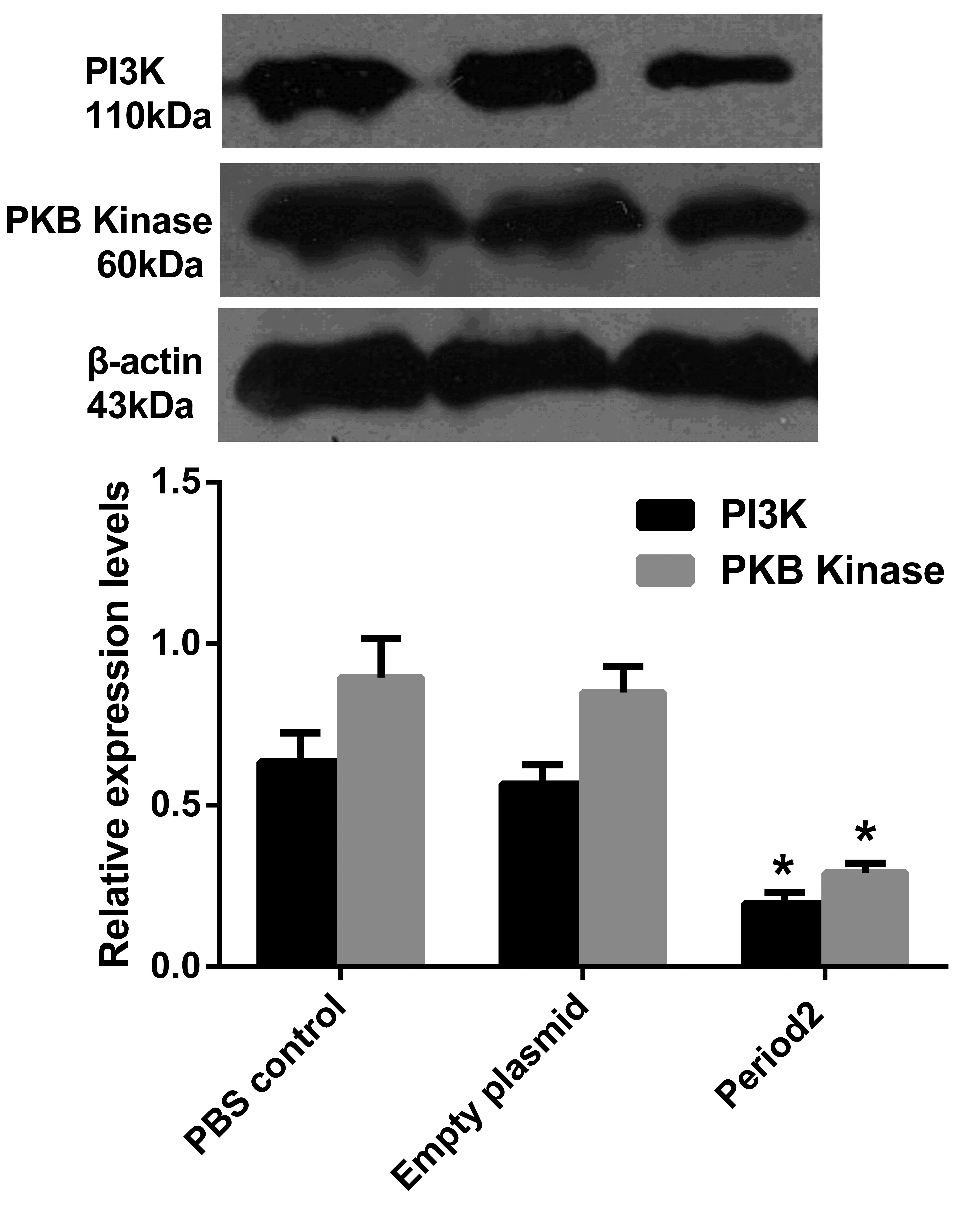
Western blot detection of MTA-1 and
nm23-H1 proteins, and protein expression levels of PI3K/PKB pathway
marker genes, PI3K and PKB kinase
In the Per2 group, MTA-1 protein expression
was significantly downregulated (P<0.05 vs. the other two
groups), while the expression level of nm23-H1 was significantly
increased (P<0.05; Fig. 5). The
PI3K/PKB signaling pathway marker genes, PI3K and PKB kinase,
demonstrated significantly reduced expression levels when compared
with the PBS control and empty plasmid groups (P<0.05; Fig. 6).
Discussion
In North America, the number of mortalities
associated with ovarian cancer is greater than the number of deaths
from all other gynecological malignancies (1,2). As
the early clinical manifestations of ovarian cancer are not typical
and patients do not exhibit obvious discomfort, diagnosis is often
late, with symptom such as ascites already apparent, which leads to
tumor metastasis. Although the combined treatment of cytoreductive
surgery and chemotherapy for ovarian cancer patients initially
demonstrates a positive effect, the majority of these patients
experience recurrence. The increasing resistance of patients to
chemotherapeutic agents, caused by genetic instability of tumor
cells and a high mutation rate, is the main cause of the high
recurrence rate of ovarian cancer, resulting in it becoming an
incurable disease. Novel treatment methods, such as gene, immune,
interventional and anti-angiogenesis therapy are currently being
investigated.
Ovarian cancer is associated with cell proliferation
and differentiation of oncogenes and tumor suppressor genes. It is
also associated with multi-stage interactions, such as mutation and
amplification of ErbB, c-Myc and KRAS oncogenes, and/or of tumor
suppressor gene, RB, p53 and p16. The loss of function of
these genes may lead to the development of tumors. Ovarian cancer
is closely associated with the female endocrine system, and the
female endocrine system is regulated by circadian rhythm genes;
when the rhythm is disrupted, this may cause disorders of the
female endocrine system, which may be associated with the
occurrence of tumors.
The clock gene, Per2 is important in
generating and maintaining circadian rhythms, and is involved in
tumor suppression and responding to DNA damage. Per2, via
the regulation of oncogenes, tumor suppressor gene expression, and
the expression duration of cell cycle regulation-associated genes,
inhibits vascular endothelial growth factor (VEGF) agonist activity
and, therefore, inhibits tumor development.
Previous studies identified reduced Per2
expression levels in human colorectal cancer, and that Per2
expression levels were closely associated with patient age, tumor
histological grade, invasion depth, lymph node metastasis and TNM
staging (22). Recent studies
demonstrated that the Per2 protein was expressed in healthy lung
tissue samples and non-small cell lung cancer (NSCLC) tissues;
however, the expression in NSCLC tissue was significantly reduced.
Furthermore, expression of the Per2 protein was found to be
associated with the degree of NSCLC differentiation and TNM stage.
A lower degree of tumor differentiation and later TNM stage was
observed with a reduced expression level of Per2 protein, which
demonstrated that the abnormal expression of Per2 in patients with
NSCLC was associated with the occurrence and development of NSCLC
(23). Yang et al (24) demonstrated that downregulation of
Per2 expression accelerated tumor growth, and doubled the
diurnal amplitude of the tumor growth rhythm, in an in vitro
study. Koyanagi et al (25)
indicated that Per2 inhibited VEGF agonist activity, which
was induced by tumor cell hypoxia, thereby inhibiting tumor
angiogenesis.
MTA-1 was discovered in 1993, demonstrating
high expression levels with high metastatic potential in rat breast
cancer cells (26). Subsequently,
it was confirmed that MTA-1 is involved in the invasion and
metastasis of tumors, and is involved in regulating a variety of
other malignant tumor processes, including cell proliferation,
angiogenesis and resistance to chemotherapy (26–30).
Furthermore, MTA-1 is associated with high recurrence and a
poor prognosis, and eventually causes cancer. In previous studies,
MTA-1 was closely associated with angiogenesis, lymph node
metastasis and colorectal and gastric cancers (26,31).
In addition, in breast cancer studies, MTA-1 was correlated
with the tumor stage, angiogenesis and recurrence (32,33).
In a study regarding small cell lung cancer, it was found that
compared with stage I tumors, the MTA-1 expression level in
stage II-IV tumors was significantly increased, and was associated
with lymph node metastasis (34).
Recently, MTA-1 was identified to be associated with
invasive ability, metastasis and a later stage, in oral squamous
cell carcinoma (35).
nm23 was the first gene to be identified as a
tumor metastasis suppressor gene and two types of nm23 gene
are present in humans: nm23-H1 and nm23-H2. A close
association between nm23-H1 and tumor metastasis has been
identified (36). Mutations or low
expression levels of nm23-H1 contribute to tumor metastasis,
and its regulation, proliferation and differentiation. Luo et
al (37) compared 69 cases of
cervical carcinoma and 20 cases of healthy cervical tissue, and the
positive expression of nm23 was identified to be negatively
correlated with the clinical stage, pathological grade and lymph
node metastasis (P<0.01). No association was identified between
nm23 expression and the pathological type or patient age. It
was hypothesized that reduced levels of nm23 expression may
exert a significant effect in cervical carcinogenesis, invasion and
transfer, and previous studies have confirmed the association
between nm23-H1, and the invasive ability and metastasis of
cervical cancer (38). Wu et
al (39) demonstrated that the
positive expression of nm23-H1 in endometrial carcinoma
tissue samples was significantly lower than that of adjacent tissue
samples and a healthy uterine mucosa (P<0.05); the decreased
expression of nm23-H1 in lymph node metastasis (P<0.05)
was not identified to be associated with age, clinical stage or
histological grade. Another study demonstrated that the expression
of nm23-H1 mRNA was negatively correlated with prostate
cancer bone metastasis and with microvessel density (40).
The PI3K/PKB signaling pathway is a downstream
signal transduction pathway involving numerous growth factors and
cytokines. The PI3K/PKB signaling pathway is involved in
anti-apoptotic activity, and promotes cell proliferation, migration
and cancerous transformation processes. Abnormal changes in this
pathway, such as increased expression of key oncogenic genes or
decreased expression of tumor suppressor genes, occur in certain
types of malignant tumor tissue and are closely correlated with
tumor progression (41). The
structural changes in key molecules of PI3K/PKB, such as pll0α,
p85α, PKB and phosphatase and tensin homolog-encoding genes, are
associated with cell transformation, and these genes have been
proven to be oncogenes or tumor suppressors, dependent on the gene.
The PI3K/PKB signaling pathway is involved in cell signal
transduction to regulate apoptosis; previous studies demonstrated
that the upregulation of phosphorylated-PKB inhibits apoptosis and
is a primary regulatory mechanism of autophagy. PKB-deficient mouse
embryonic fibroblast cells demonstrate a reduced rate of
progression from the G2/M to the G1 phase,
indicating that the activation of PKB causes cells to divide
rapidly (42). In addition, the
PI3K/PKB signaling pathway also affects glucose metabolism and
promotes the growth of tumor cells (43). The PI3K/PKB signaling pathway
inhibits cell apoptosis, promotes cell proliferation and is
associated with tumor occurrence, whilst also affecting migration
and invasion of tumor cells, and promoting tumor progression. In a
previous study of cell proliferation and migration, the PI3K
inhibitor LY294002 eliminated the polar localization of PKB, and
reduced the speed of cell migration by >60%, indicating that
cell migration is dependent on the PI3K/PKB signaling pathway
(44).
The present study demonstrated that the expression
of Per2 in tumor tissue samples varied with each stage of
malignant ovarian cancer, when compared with the benign ovarian
tissue samples. Furthermore, increases in the degree of tumor
differentiation and the TNM stage were associated with a gradual
decrease in the expression level of Per2, indicating that
the circadian gene is closely associated with the occurrence and
development of ovarian cancer. In addition, it was hypothesized
that Per2 deletion or mutation leads to tumor progression
and metastasis.
In order to verify the possible role of Per2
in ovarian cancer inhibition, a Per2 recombinant plasmid was
transfected into ovarian cancer cells from ovarian xenografts in
nude mice. The overexpression of Per2 in the transplantation
tumor was stable, and continuous high expression was observed for
4–6 weeks, which was adequate time to allow growth inhibition of
the tumor. The tumor growth rate and tumor volume were
significantly reduced when compared with the PBS control group, and
the tumor inhibition rate increased significantly. Tumor metastasis
and the rate of ascites formation was significantly lower than in
the PBS control group, with no adverse effects (such as those that
are observed with chemotherapy) on survival in the experimental
mice. The local and continuous Per2 expression resulted in a
significant decrease in MTA-1 expression, which is highly
associated with solid tumor metastasis, and indicates that the
tumor metastasis inhibition mechanism of Per2 may act by
inhibiting MTA-1 expression. In addition, previous studies
revealed that the grade and lymph node metastasis in cervical
cancer cases were closely and negatively associated with
nm23-H1 expression (45–47).
In the present study, the local and continuous elevated expression
of Per2 induced the expression of nm23-H1, which was
significantly increased when compared with the PBS control group.
Notably, the occurrence and development of tumors is closely
associated with the PI3K/PKB signal transduction pathway, which has
an important role. The local and continuously elevated expression
of Per2 inhibits PKB activation, thus inducing apoptosis of
tumor cells, inhibiting the expression of its downstream oncogenes
and, therefore, inhibiting tumor growth. However, it must be noted
that the use of Per2 gene therapy alone for the treatment of
a malignant tumor only inhibits cancer progression and does not
completely eradicate it. It is proposed that combining Per2
gene therapy with traditional methods of tumor therapy, such as
chemotherapy, may be more efficacious. The expression of
Per2 is hypothesized to increase the sensitivity of tumor
cells to chemotherapeutic agents via the PI3K/PKB signaling pathway
downstream genes, which enhances the therapeutic agent efficacy.
Thus, reducing the required dose of chemotherapeutic agent and
reducing the side effects.
In conclusion, the present study indicates that
Per2 is closely and negatively associated with the
occurrence and development of ovarian cancer. Per2
expression, and the clinical stage and TNM development of ovarian
cancer were identified to be correlated. Furthermore, Per2
may promote expression of the tumor metastasis suppressor gene,
nm23-H1 and inhibit MTA-1 expression, and is
hypothesized to exert its antitumor effects via the PI3K/PKB
signaling pathway and its downstream genes. To the best of our
knowledge, this is the first study regarding expression of the
circadian gene, Per2 in ovarian cancer. However, the
specific underlying mechanisms require further investigation.
References
|
1
|
Nasioudis D, Sisti G, Kanninen TT, Holcomb
K, Di Tommaso M, Fambrini M and Witkin SS: Epidemiology and
outcomes of squamous ovarian carcinoma; a population-based study.
Gynecol Oncol. 2016. View Article : Google Scholar
|
|
2
|
Kosary CL: FIGO stage, histology,
histologic grade, age and race as prognostic factors in determining
survival for cancers of the female gynecological system: An
analysis of 1973–87 SEER cases of cancers of the endometrium,
cervix, ovary, vulva, and vagina. Semin Surg Oncol. 10:31–46. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jiang J, Xie W and Cao J: Current
situation of the therapies of middle-advanced ovarian cancer. Zhong
Liu Yao Xue. 3:416–421. 2013.In Chinese.
|
|
4
|
He JR, Gao X and Ren ZF: Global Incidence
Patterns of Female Breast and Ovarian Cancers. China Cancer.
3:169–172. 2009.
|
|
5
|
Lou JY, Peng ZL, Zheng Y, Wang H, He B and
Wang HJ: Research on human ovarian cancer cell MDR1 gene silenced
by siRNA. Sichuan Da Xue Xue Bao Yi Xue Ban. 38:753–755. 2007.In
Chinese. PubMed/NCBI
|
|
6
|
Menendez L, Walker D, Matyunina LV,
Dickerson EB, Bowen NJ, Polavarapu N, Benigno BB and McDonald JF:
Identification of candidate methylation-responsive genes in ovarian
cancer. Mol Cancer. 6:102007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tei H, Okamura H, Shigeyoshi Y, Fukuhara
C, Ozawa R, Hirose M and Sakaki Y: Circadian oscillation of a
mammalian homologue of the Drosophila period gene. Nature.
389:512–516. 1997. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shigeyoshi Y, Taguchi K, Yamamoto S,
Takekida S, Yan L, Tei H, Moriya T, Shibata S, Loros JJ, Dunlap JC
and Okamura H: Light-induced resetting of a mammalian circadian
clock is associated with rapid induction of the mPer1 transcript.
Cell. 91:1043–1053. 1997. View Article : Google Scholar
|
|
9
|
Jin X, Shearman LP, Weaver DR, Zylka MJ,
de Vries GJ and Reppert SM: A molecular mechanism regulating
rhythmic output from the suprachiasmatic circadian clock. Cell.
96:57–68. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Reppert SM and Weaver DR: Molecular
analysis of mammalian circadian rhythms. Annu Rev Physiol.
63:647–676. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Young MW and Kay SA: Time zones: A
comparative genetics of circadian clocks. Nat Rev Genet. 2:702–715.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bjarnason GA and Jordan R: Circadian
variation of cell proliferation and cell cycle protein expression
in man: Clinical implications. Prog Cell Cycle Res. 4:193–206.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Matsuo T, Yamaguchi S, Mitsui S, Emi A,
Shimoda F and Okamura H: Control mechanism of the circadian clock
for timing of cell division in vivo. Science. 302:255–259. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Panda S, Antoch MP, Miller BH, Su AI,
Schook AB, Straume M, Schultz PG, Kay SA, Takahashi JS and
Hogenesch JB: Coordinated transcription of key pathways in the
mouse by the circadian clock. Cell. 109:307–320. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Barbason H, Herens C, Robaye B, Milis G,
Sulon J, Bouzahzah B and VanCantfort J: Importance of cell kinetics
rhythmicity for the control of cell proliferation and
carcinogenesis in rat liver (review). In Vivo. 9:539–548.
1995.PubMed/NCBI
|
|
16
|
Hansen J: Increased breast cancer risk
among women who work predominantly at night. Epidemiology.
12:74–77. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lee CC: Tumor suppression by the mammalian
Period genes. Cancer Causes Control. 17:525–530. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fu L, Pelicano H, Liu J, Huang P and Lee
C: The circadian gene Period2 plays an important role in tumor
suppression and DNA damage response in vivo. Cell. 111:41–50. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Gery S, Komatsu N, Baldjyan L, Yu A, Koo D
and Koeffler HP: The circadian gene per1 plays an important role in
cell growth and DNA damage control in human cancer cells. Mol Cell.
22:375–382. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pecorelli S, Benedet JL, Creasman WT and
Shepherd JH; FIGO staging of gynecologic cancer; 1994–1997 FIGO
committee on Gynecologic Oncology: International Federation of
Gynecology and Obstetrics. Int J Gynaecol Obstet. 65:243–249. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pfaffl MW: A new mathematical model for
relative quantification in real-time RT-PCR. Nucleic Acids Res.
29:e452001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Karantanos T, Theodoropoulos G, Pektasides
D and Gazouli M: Clock genes: Their role in colorectal cancer.
World J Gastroenterol. 20:1986–1992. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chi C, He ZF, Liu Y, Lin XM and Sun CC:
Expression and clinical significance of circadian gene Per2 in
non-small cell lung cancer. Zhonghua Zhong Liu Za Zhi. 35:129–131.
2013.In Chinese. PubMed/NCBI
|
|
24
|
Yang X, Wood PA, Oh EY, Du-Quiton J,
Ansell CM and Hrushesky WJ: Down regulation of circadian clock gene
Period 2 accelerates breast cancer growth by altering its daily
growth rhythm. Breast Cancer Res Treat. 117:423–431. 2009.
View Article : Google Scholar
|
|
25
|
Koyanagi S, Kuramoto Y, Nakagawa H,
Aramaki H, Ohdo S, Soeda S and Shimeno H: A molecular mechanism
regulating circadian expression of vascular endothelial growth
factor in tumor cells. Cancer Res. 63:7277–7283. 2003.PubMed/NCBI
|
|
26
|
Toh Y, Oki E, Oda S, Tokunaga E, Ohno S,
Maehara Y, Nicolson GL and Sugimachi K: Overexpression of the MTA1
gene in gastrointestinal carcinomas: Correlation with invasion and
metastasis. Int J Cancer. 74:459–463. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Talukder AH, Mishra SK, Mandal M,
Balasenthil S, Mehta S, Sahin AA, Barnes CJ and Kumar R: MTA1
interacts with MAT1, a cyclin-dependent kinase-activating kinase
complex ring finger factor, and regulates estrogen receptor
transactivation functions. J Biol Chem. 278:11676–11685. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Avtanski DB, Nagalingam A, Kuppusamy P,
Bonner MY, Arbiser JL, Saxena NK and Sharma D: Honokiol abrogates
leptin-induced tumor progression by inhibiting Wnt1-MTA1-β-catenin
signaling axis in a microRNA-34a dependent manner. Oncotarget.
6:16396–16410. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Marzook H, Deivendran S, Kumar R and
Pillai MR: Role of MTA1 in head and neck cancers. Cancer Metastasis
Rev. 33:953–964. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bruning A, Blankenstein T, Jückstock J and
Mylonas I: Function and regulation of MTA1 and MTA3 in malignancies
of the female reproductive system. Cancer Metastasis Rev.
33:943–951. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Deng X, Du L, Wang C, Yang Y, Li J, Liu H,
Zhang J, Wang L, Zhang X, Li W, et al: Close association of
metastasis-associated protein 1 overexpression with increased
angiogenesis and poor survival in patients with histologically
node-negative gastric cancer. World J Surg. 37:792–798. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Martin MD, Hilsenbeck SG, Mohsin SK, Hopp
TA, Clark GM, Osborne CK, Allred DC and O'Connell P: Breast tumors
that overexpress nuclear metastasis-associated 1 (MTA1) protein
have high recurrence risks but enhanced responses to systemic
therapies. Breast Cancer Res Treat. 95:7–12. 2006. View Article : Google Scholar
|
|
33
|
Cheng CW, Liu YF, Yu JC, Wang HW, Ding SL,
Hsiung CN, Hsu HM, Shieh JC, Wu PE and Shen CY: Prognostic
significance of cyclin D1, β-catenin, and MTA1 in patients with
invasive ductal carcinoma of the breast. Ann Surg Oncol.
19:4129–4139. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang H, Zhu X, Li N, Li D, Sha Z, Zheng X
and Wang H: miR-125a-3p targets MTA1 to suppress NSCLC cell
proliferation, migration, and invasion. Acta Biochim Biophys Sin.
47:496–503. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kawasaki G, Yanamoto S, Yoshitomi I,
Yamada S and Mizuno A: Overexpression of metastasis-associated MTA1
in oral squamous cell carcinomas: Correlation with metastasis and
invasion. Int J Oral Maxillofac Surg. 37:1039–1046. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gilles AM, Presecan E, Vonica A and Lascu
I: Nucleoside diphosphate kinase from human erythrocytes.
Structural characterization of the two polypeptide chains
responsible for heterogeneity of the hexameric enzyme. J Biol Chem.
266:8784–8789. 1991.PubMed/NCBI
|
|
37
|
Luo S, Wang X and Sun X: The expression
and clinical significance of p16 and nm23 in cervical carcinoma.
Zhong Guo Shi Yong Fu Ke Yu Chan Ke. 7:421–422. 2004.In
Chinese.
|
|
38
|
Utrera-Barillas D, Salcedo-Vargas M,
Gariglio-Vidal P, Hernández-Hernández DM, Gutiérrez-Delgado F and
Benítez-Bribiesca L: H-ras and Nm23-H1 gene expression and
proteolytic activity in squamous cell carcinoma of the uterine
cervix. Arch Med Res. 31:172–181. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wu W, Xu H and Deng Z: Expression of
nm23H1 and E-cadherin protein in 39 cases of endometrial carcinoma.
Zhong Liu Xue Zazhi. 4:322–323. 2006.
|
|
40
|
Ding GF, Li JC and Xu YF: Study on the
correlationship between the expression of nm23H1mRNA, TGF-beta1mRNA
and tumor metastases, survival rate with prostate cancer. Fen Zi Xi
Bao Sheng Wu Xue Bao. 39:544–552. 2006.In Chinese.
|
|
41
|
Nicholson KM and Anderson NG: The protein
kinase B/Akt signalling pathway in human malignancy. Cell Signal.
14:381–395. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Li Q and Zhu GD: Targeting
serine/threonine protein kinase B/Akt and cell-cycle checkpoint
kinases for treating cancer. Curr Top Med Chem. 2:939–971. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Elstrom RL, Bauer DE, Buzzai M, Karnauskas
R, Harris MH, Plas DR, Zhuang H, Cinalli RM, Alavi A, Rudin CM and
Thompso CB: Akt stimulates aerobic glycolysis in cancer cells.
Cancer Res. 64:3892–3899. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kim D, Kim S, Koh H, Yoon SO, Chung AS,
Cho KS and Chung J: Akt/PKB promotes cancer cell invasion via
increased motility and metalloproteinase production. FASEB J.
15:1953–1962. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Huang Y, Cai S and Yu S: Relationship
between nm23-H1 expression and lymph node metastasis and prognosis
in cervical cancer. Zhonghua Fu Chan Ke Za Zhi. 32:718–721. 1997.In
Chinese.
|
|
46
|
Utrera-Barillas D, Salcedo-Vargas M,
Gariglio-Vidal P, Hernández-Hernández DM, Gutiérrez-Delgado F and
Benítez-Bribiesca F: H-ras and nm23-H1 gene expression and
proteolytic activity in squamous cell carcinoma of the uterine
cervix. Arch Med Res. 31:172–181. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Tong Y, Yung LY and Wong YH: Metastasis
suppressors nm23H1 and nm23H2 differentially regulate neoplastic
transformation and tumorigenesis. Cancer Lett. 361:207–217. 2015.
View Article : Google Scholar : PubMed/NCBI
|