Introduction
Melanoma is the most fatal type of skin cancer
(1); however, when it is diagnosed
at an early stage, there is a relatively high success rate of
treatment decreasing tumor size (2). However, melanoma diagnosed at an
advanced stage has been associated with poor prognosis. The
predominant therapeutic strategies for melanoma are chemo-and
immunotherapy, or radiation therapy (3), however, these strategies also affect
non-cancerous cells and result in side effects (4). Thus, elucidation of the molecular
mechanisms underlying melanoma initiation and progression is
required to develop effective therapeutic strategies against
tumorigenesis.
Hypoxia-inducible factor 1α (HIF-1α) activates the
transcription of genes that are involved in cancer initiation and
progression and associated with cell survival and cell invasion
(5–7). Overexpression of HIF-1α has been
associated with poor prognosis and high mortality rate in a number
of cancer types, including breast cancer (7–9). In
clinical applications, HIF-1α may be useful as a marker to indicate
tumor growth (10). It has been
reported that a decrease in HIF-1α reduces the capacity to release
vascular endothelial growth factor in hypoxia and resulted in
inhibition of solid tumor growth (11). Thus, inhibitors of HIF-1α may be
potential anticancer therapeutic agents.
MicroRNAs (miRNAs) are non-coding RNAs (length, ~22
nt) (12), they target
transcripts, particularly the 3′-untranslated region (UTR) to
regulate gene expression (13).
miRNAs have been indicated to be involved in a similar function in
other organisms (14–16). Current research into miRNA-based
anticancer therapeutic strategies aims to regulate and modulate
various cancer chemoprophylaxis agents (17). Thus, the development of a specific
and efficient target for miRNA molecules is key. Based on previous
studies (10,11), HIF-1α may be a potential target for
miRNA-based anticancer therapeutic strategies.
The present study focused on the expression profile
of miR-199a-5p in melanoma. The expression of miR-199a-5p and its
association with poor prognosis were detected and the suppressive
effects of miR-199a-5p on melanoma cells were demonstrated. The
downregulation of HIF-1α by miR-199a-5p was also demonstrated in
melanoma cells and the effect of miR-199a-5p on cell viability and
apoptosis was investigated. miR-199a-5p was demonstrated to
suppress melanoma proliferation in a nude mouse model. The present
results suggest that miR-199a-5p inhibited melanoma via targeting
of HIF-1α.
Materials and methods
Clinical samples
The clinical samples (cancer tissues and adjacent
tissues) from 25 patients, including 13 men and 12 women, were
collected from Xiangya Hospital, Central South University
(Changsha, China). The experiments were approved by the Xiangya
School of Medicine Research Ethics Committee (Changsha, China).
Informed consent was obtained according to the Declaration of
Helsinki. The 25 patients enrolled in the present study had not
undergone surgical removal of the lesion or any prior treatment.
Clinical characteristics of the patients is presented in Table I. The control samples were adjacent
non-cancerous tissue samples obtained from the same patients.
 | Table ICharacteristics of patients. |
Table I
Characteristics of patients.
| Variable | Data |
|---|
| Age (years) |
| Mean | 45 |
| Median | 47.62 |
| Range | 37–69 |
| Histological
grade |
| I | 10 |
| II | 6 |
| III | 9 |
| Tumor stage |
| I | 7 |
| II | 8 |
| III | 5 |
| IV | 5 |
Cell culture and transfection
B16 and HME1 melanoma cell lines were purchased from
the American Type Culture Collection (Manassas, VA, USA). The cells
were cultured in RPMI 1640 medium (Sigma-Aldrich, St. Louis, MO,
USA) supplemented with 10% fetal bovine serum (Invitrogen; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) in 5% CO2 at
37°C.
To understand the effect of miR-199a-5p on melanoma
cells, the cells were divided into groups: i) Control, ii) mimics
(100 nmol), and iii) inhibitors (100 nmol) with n=4 in each group.
The cells (1×106 cells/well) were plated into 6-well
plates and incubated at 37°C for 24 h in 5% CO2. The
cells were transfected using Lipofectamine 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.) according to the manufacturer's protocols.
The mimics and inhibitors were purchased from Sangon Biotech Co.,
Ltd. (Shanghai, China).
The RNA interference was performed as described
previously (18). Briefly, 100
nmol HIF-1α siRNA (sense, 5′-CUGAUGACCAGCAACUUGA-3′ and antisense,
5′-UCAAGUUGCUGGUCAUCAG-3′), or 100 nmol miR-199a-5p mimics, were
transfected into B16 cells using Lipofectamine 2000. Following 24
h, the cells were analyzed by flow cytometry, MTT and colony
formation assay.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNAs were isolated using TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's protocol, and were treated with DNase I (Sangon
Biotech Co., Ltd.) to remove contaminating genomic DNA. The purity
of the extracted RNA was assessed by measuring the optical density
ratio (A260/A280) using the NanoDrop 2000 spectrophotometer (Thermo
Fisher Scientific, Inc.). The expression level of miR-199a-5p was
determined using the TaqMan® microRNA Assays kit (cat.
no. 4324020; Applied Biosystems; Thermo Fisher Scientific, Inc.),
in which U6 served as an endogenous control. In order to determine
the expression level of HIF-1α, total RNA (1 μg) was reverse
transcribed into cDNA using the TaqMan® Reverse
Transcription kit (cat. no. N8080234; Applied Biosystems; Thermo
Fisher Scientific, Inc.). qPCR was performed using the
TaqMan® Universal PCR Master Mix (cat. no. 4324018;
Applied Biosystems; Thermo Fisher Scientific, Inc.) on the ABI 7500
thermal cycler (Applied Biosystems; Thermo Fisher Scientific,
Inc.). The primers were as follows: Sense,
5′-CCACAGGACAGTACAGGATG-3′ and antisense,
5′-TCAAGTCGTGCTGAATAATACC-3′ for HIF-1α and sense,
5′-AGGGCTGCTTTTAACTCTGGT-3′ and antisense,
5′-CCCCACTTGATTTTGGAGGGA-3′ for GAPDH (Sangon Biotech Co., Ltd.).
GAPDH served as an endogenous control. The cycling conditions were
as follows: 95°C for 10 min, followed by 40 cycles at 95°C for 15
sec and 60°C for 1 min. The relative mRNA expression was calculated
from three independent experiments using the 2−ΔΔCq
method (19). Melting curve
analyses were performed to assess whether the amplifications were
specific.
Western blot
The tested cells were lysed using
radioimmu-noprecipitation buffer (Beyotime Institute of
Biotechnology, Haimen, China) and the quantity of protein was
detected using a BCA kit (Beyotime Institute of Biotechnology). For
each sample, 15 μg total protein was separated on 12% sodium
dodecyl sulfate-polyacrylamide gel electrophoresis and transferred
onto polyvinyl difluoride membranes (EMD Millipore, Billerica, MA,
USA). Following blocking with 4% non-fat milk, the membranes were
incubated with rabbit anti-HIF-1α (1:1,000; cat. no. SAB2104366)
and anti-GAPDH (1:1,000; cat. no. SAB2100894) polyclonal antibodies
(Sigma-Aldrich), followed by incubation with horseradish
peroxidase-conjugated goat anti-rabbit immunoglobulin G (IgG)
secondary antibody (1:2,000; cat. no. A0545) and an enhanced
chemiluminescent substrate (ECL Plus, GE Healthcare Life Sciences,
Chalfont, UK). The protein bands were visualized using X-ray paper
(Kodak, Rochester, NY, USA)
MTT assay
The cells were plated into 96-well plates at
concentration of 5×103 cells/well. Following culture for
24 h, the cells were transfected with 100 nmol miR-199a-5p mimics
or 100 nmol miR-199a-5p inhibitors (n=3 for each group). Following
transfection, the cells were treated using MTT solution
(Sigma-Aldrich) according to the manufacturer's protocols. The
absorbance at a wavelength of 570 nm was measured on a DU 800
UV/Vis spectrophotometer (Beckman Coulter, Inc., Brea, CA,
USA).
Colony formation assay
For the colony formation assay, 1,000 cells/well
were plated into the 6-well plates and incubated for 7 days.
Following removal of the culture medium and fixing in 30%
formaldehyde (Sangon Biotech Co., Ltd.) for 15 min, the cells were
stained using 0.2% crystal violet (Sangon Biotech Co., Ltd.). The
colonies from three independent experiments were observed under an
optical microscope (Leica DMI6000B; Leica Microsystems GmbH,
Wetzlar, Germany).
Flow cytometry
Cells (1×104) from all groups were fixed
in 90% methanol for 2 h and resuspended in phosphate-buffered
saline with 0.1% bovine serum albumin, 0.05% Triton X-100 and 50
μg/ml RNase A (Sigma-Aldrich). Annexin V-fluorescein
isothiocyanate and propidium iodide (BioVision, Inc., Milpitas, CA,
USA) staining was performed according to the manufacturer's
protocols. The cell counting was performed on a BD FACSCalibur™
system analyzed using BD FACSDiva Software 6.0 (BD Biosciences,
Franklin Lakes, NJ, USA). Triplicate biological repeats were
measured.
Luciferase assay
Wild-type and mutant 3′-UTR of HIF-1α containing the
target sequence were synthesized by Sangon Biotech Co., Ltd. and
cloned into the pMIR-Report vector (Applied Biosystems; Thermo
Fisher Scientific, Inc.). The B16 cells were transfected with
mimics (50 and 100 nmol) and then co-transfected with wild-type and
mutant 3′-UTR of HIF-1α by Lipofectamine 2000. After 48 h, the
cells were lysed using FastBreak™ Cell Lysis Reagent (Promega
Corporation, Madison, WI, USA) and the cell lysates were incubated
with dual-luciferase reagents (Promega Corporation) and the
luciferase activities were assayed by Dual-Luciferase Reporter
assay system (Promega Corporation). The experiments were repeated
three times.
In vivo xenograft assay
A total of 12 female nu/nu mice (age, 6 weeks;
weight, 20.03±1.92 g) were obtained from the Xiangya School of
Medicine Research. The mice were maintained at 23°C under a 12-h
light/dark cycle with ad libitum access to food and water.
The mice were injected with 1×107 B16 cells (with or
without miR-199a-5p transfection of mimics) into to the posterior
flank. The volume [volume = (length × width2)/2] and
weight of the tumors were measured once a week. After 4 weeks, the
mice were sacrificed by cervical dislocation following
anesthetization with 10% chloral hydrate (4 ml/kg via
intraperitoneal injection; Sigma-Aldrich) and the xenograft tissues
were collected for immunohistochemical staining.
Immunohistochemical staining assay
The xenograft tissue samples were harvested from all
mice and then fixed in 10% formalin and paraffin-embedded (both
Sangon Biotech Co., Ltd.), prior to cutting into 6-μm
sections. Following deparaffinzation using xylene (Sangon Biotech
Co., Ltd.), the tissues were incubated with rabbit anti-HIF-1α and
anti-GAPDH polyclonal antibodies (1:200), followed by incubation
with horseradish peroxidase-conjugated goat anti-rabbit IgG
secondary antibody (1:1,000) and and diaminobenzidine (Beyotime
Institute of Biotechnology). Subsequently, the tissue sections were
visualized using the Leica DMI6000B microscope. The experiments
were repeated three times.
Statistical analysis
All the values are presented as the mean ± standard
deviation. Statistical analyses were performed using SPSS 17.0
software (SPSS, Inc., Chicago, IL, USA). One-way analysis of
variance was used to determine differences among groups. P≤0.05 was
considered to indicate a statistically significant difference. If
the F ratios exceeded the critical value (P≤0.05), the
Newman-Keul's post-hoc test was performed to compare the
groups.
Results
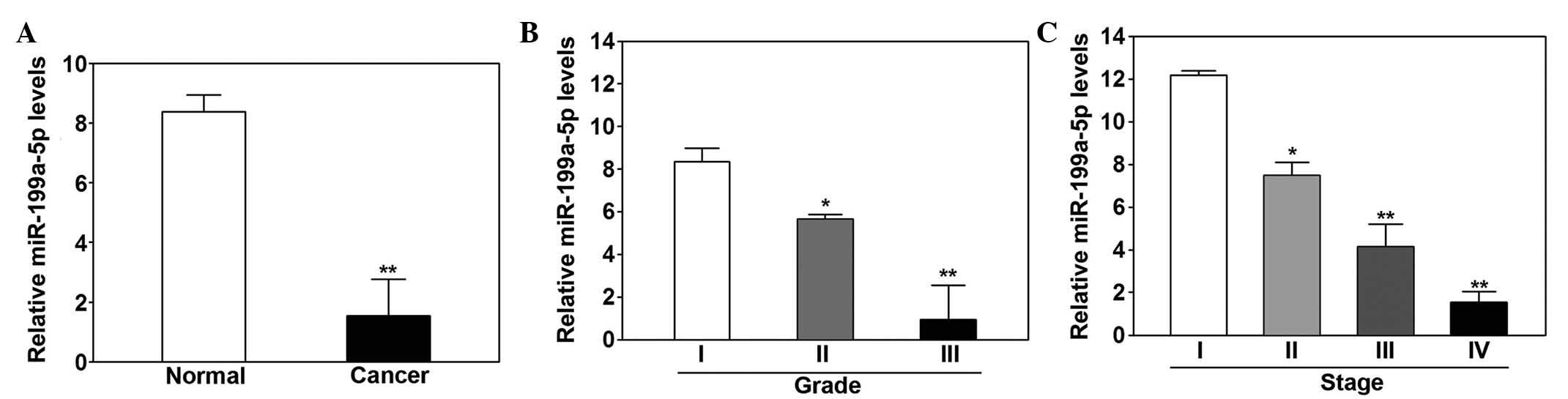
Decreased expression of miR-199a-5p was
demonstrated in melanoma tissue samples
The expression levels of miR-199a-5p were
significantly lower in melanoma tissue samples compared with the
adjacent normal tissue samples (P<0.01, Fig. 1A). Low expression levels of
miR-199a-5p were associated with higher histological grade
(P=0.031; Fig. 1B), and more
advanced tumor stage (P=0.042; Fig.
1C).
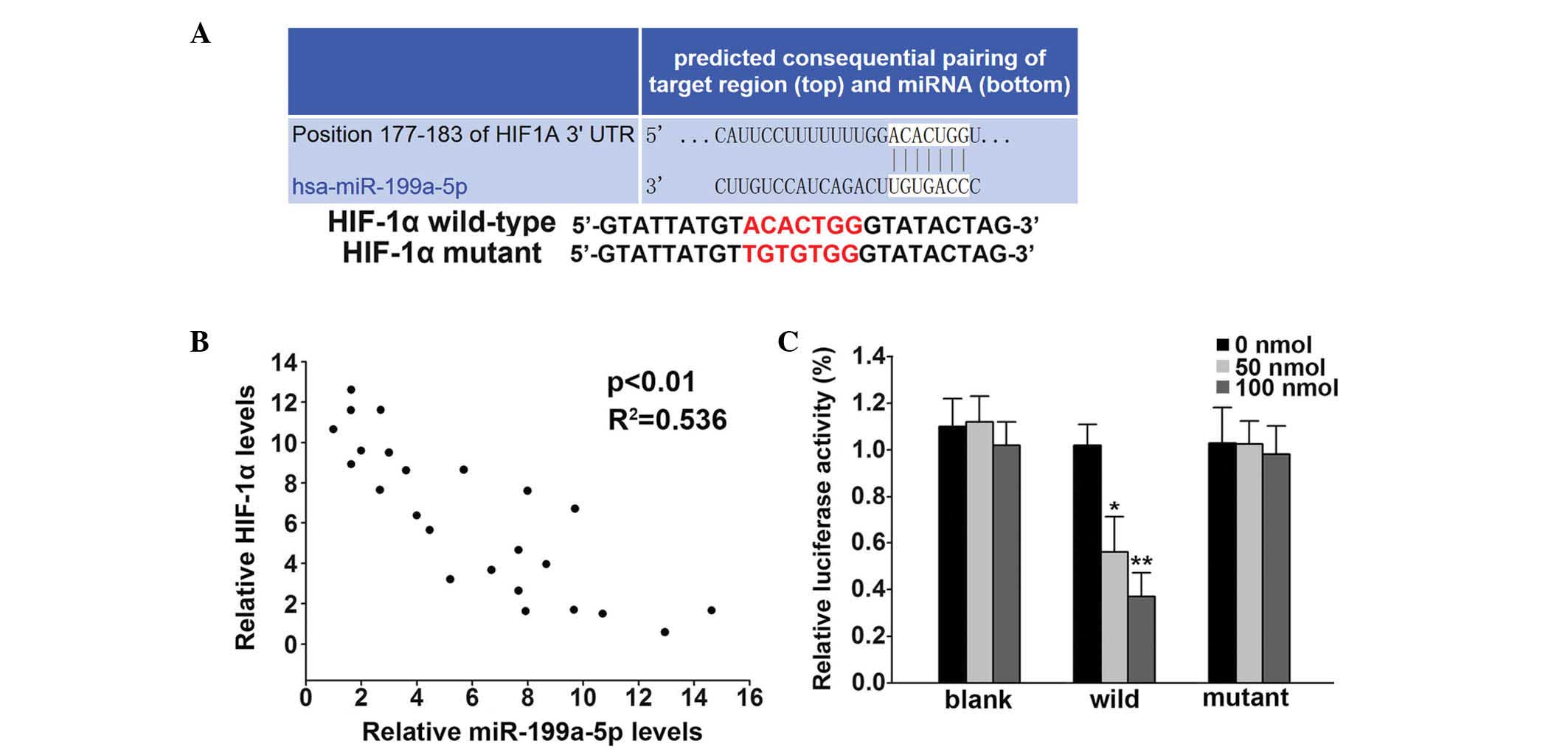
miR-199a-5p directly targeted the 3′-UTR
of HIF-1α
Targetscan software (www.targetscan.org) was used to predict the target of
miR-199a-5p; it was identified as the 3′-UTR of HIF-1α (Fig. 2A). The association between
miR-199a-5p and HIF-1α was analyzed in patients. The mRNA
expression level of HIF-1α was negatively correlated with
miR-199a-5p (P<0.001, R2= 0.536; Fig. 2B). Using the dual-luciferase
reporter system, the direct regulation of miR-199a-5p on 3′-UTR of
HIF-1α was confirmed. The 50 and 100 nmol miR-199a-5p transfection
of mimics significantly decreased the luciferase activities by 44%
(P<0.05) and 73% (P<0.01), respectively, in the wild-type
group (Fig. 2C) while no marked
difference was detected in the mutant group (Fig. 2C).
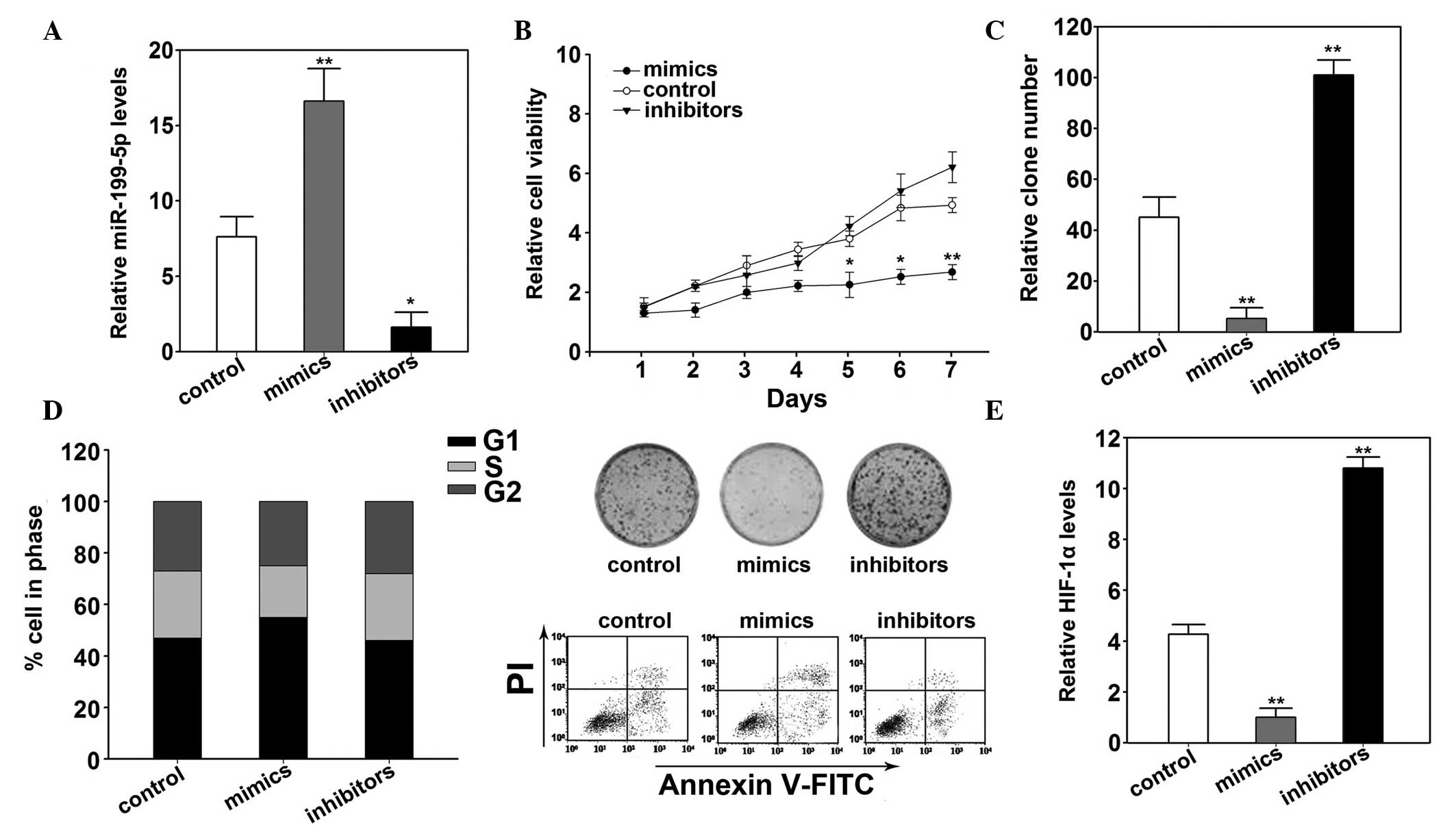
miR-199a-5p suppressed cell proliferation
of B16 cells by targeting HIF-1α
The effects exerted by miR-199a-5p on B16 cells were
detected by transfection with miR-199a-5p mimics and inhibitor. The
results of the RT-qPCR demonstrated that groups transfected with
miR-199a-5p mimics and inhibitors groups showed significantly
increased (P<0.01) and decreased (P<0.05) miR-199a-5p
expression levels, respectively (Fig.
3A). Furthermore, overexpression of miR-199a-5p inhibited cell
viability (P=0.012; Fig. 3B). The
colony formation was also decreased by miR-199a-5p overexpression
(P=0.031; Fig. 3C). Apoptosis was
investigated using flow cytometry, the miR-199a-5p mimics group
indicated increased apoptosis compared with the control (P=0.031;
Fig. 3D). Compared with the
control groups, miR-199a-5p overexpression arrested the cell cycle
into G1 phase (P= 0.011; Fig. 3D). The expression of HIF-1α was
decreased following transfection of miR-199a-5p mimics (P=0.014;
Fig. 3E).
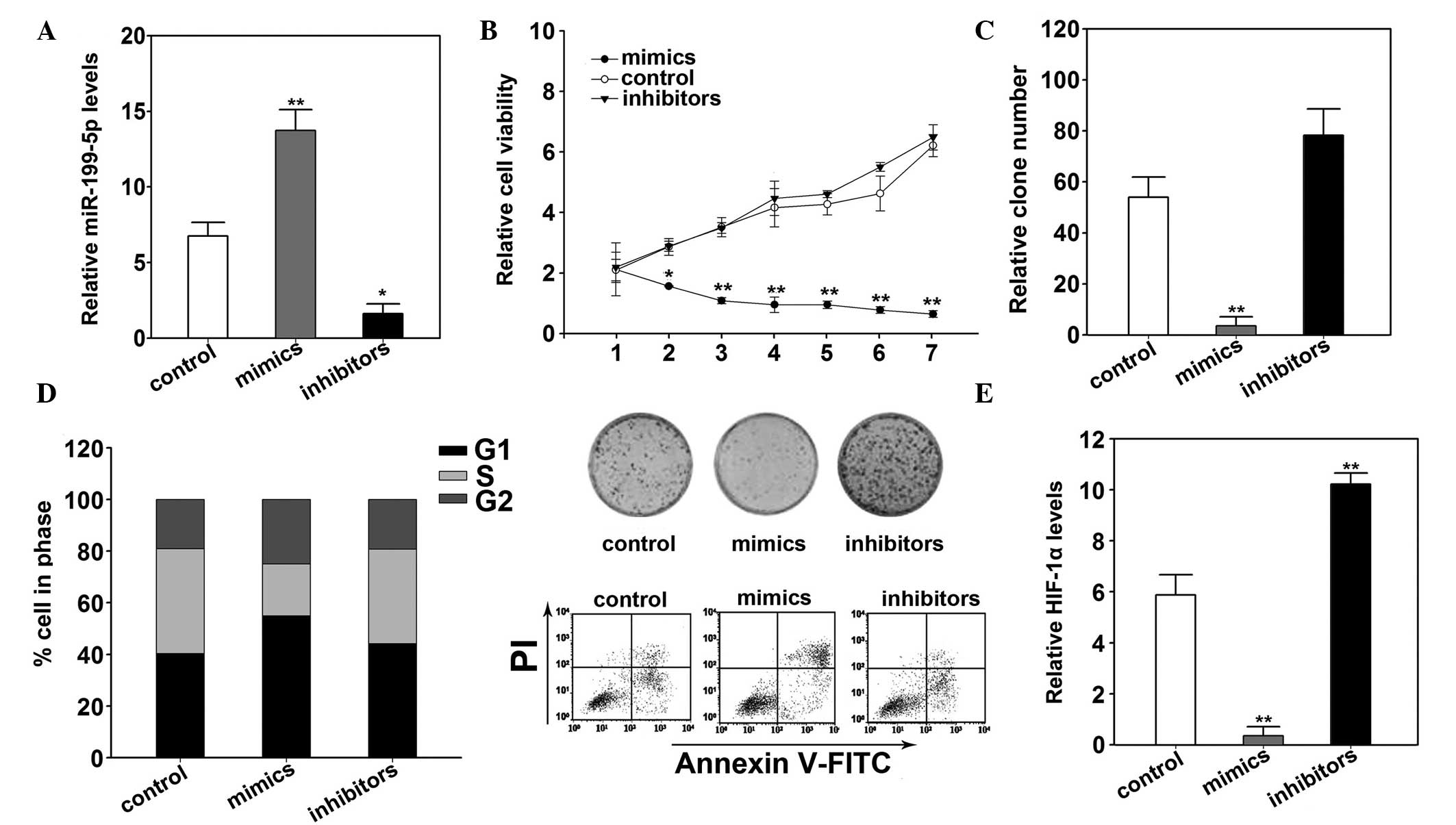
miR-199a-5p suppressed cell proliferation
of HME1 cells via targeting HIF-1α
Similar effects of miR-199a-5p on HME1 were
observed. The expression levels of miR-199a-5p increased
significantly following transfection of mimics (P<0.05; Fig. 4A). The transfection also
significantly inhibited the cell viability (P=0.023; Fig. 4B). Overexpression of miR-199a-5p
also decreased the colony formation in HME1 cells (P=0.019;
Fig. 4C). Flow cytometry analysis
demonstrated increased apoptosis with overexpression of miR-199a-5p
(Fig. 4D). The cells were arrested
in G1 phase significantly more than the control cells
(P=0.022, Fig. 4D). In addition,
miR-199a-5p mimics decreased HIF-1α mRNA and protein expression
levels (P=0.032; Fig. 4E).
Inhibition of miR-199a-5p suppressed
tumor growth in vivo
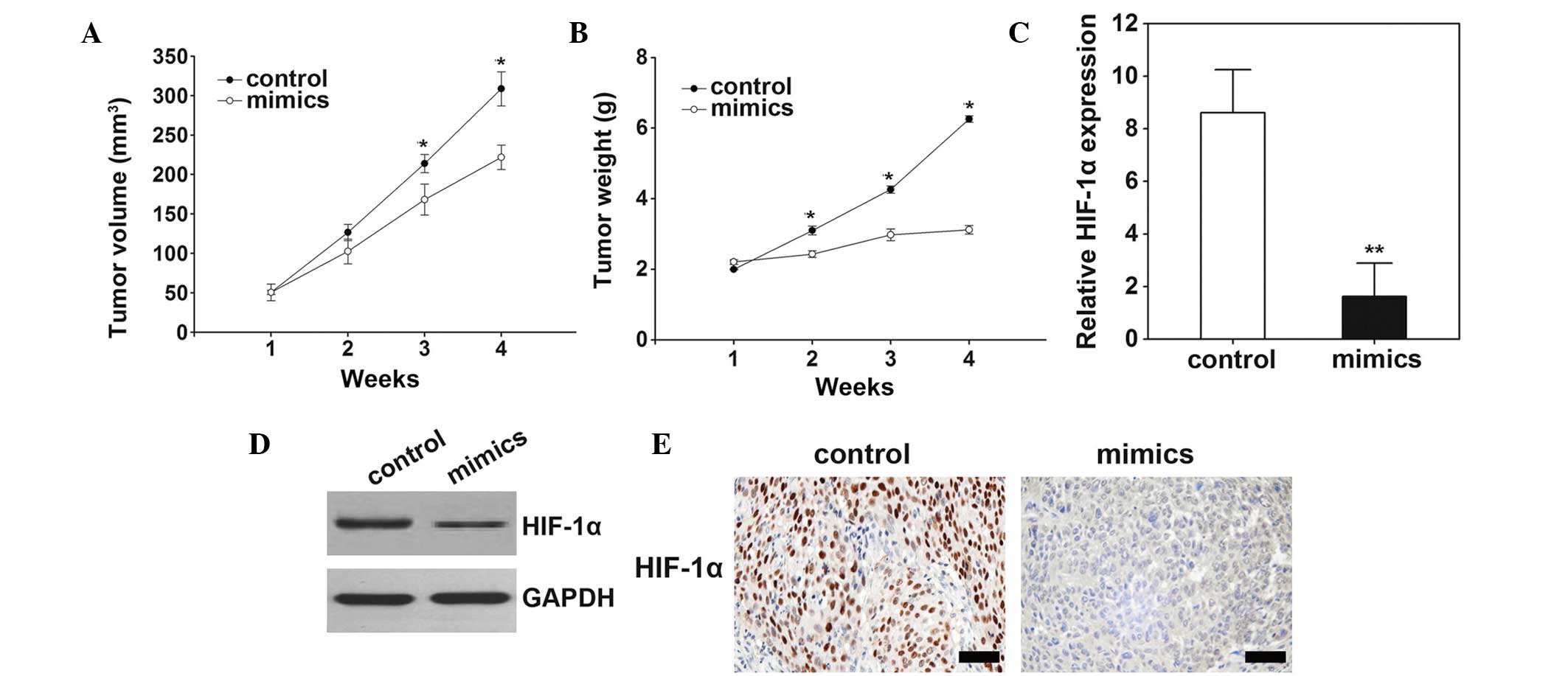
The effects of miR-199a-5p on tumor growth were
observed in the nude mice model. The results demonstrated that
following treatment with miR-199a-5p mimics, the tumor volume (P=
0.012; Fig. 5A) and weight
(P=0.023; Fig. 5B) significantly
increased. Consistently with in vitro study, overexpression
of miR-199a-5p decreased HIF-1α mRNA and protein expression levels
in tumor tissue samples (Fig.
5C–E).
Discussion
A previous study has demonstrated that HIF-1 was
significantly increased in malignant tumor samples (20). HIF-1 consists of HIF-1α,
oxygen-responsive, and HIF-1β, constitutive, subunits (21). The induced expression of HIF-1 by
hypoxia resulted in the proliferation of malignant melanoma cells
(22). Furthermore, HIF-1
knockdown suppressed the proliferation of melanoma cells, which
suggested that the inhibition of HIF-1 is a potent therapy for
melanoma (22). An effective
treatment on targeting HIF-1 remains to be developed in other
cancer types. The present study demonstrates that HIF-1α was a
direct target of miR-199a-5p and that overexpression of miR-199a-5p
reduced melanoma proliferation suggesting this may be a potential
therapeutic strategy for melanoma.
Research into miRNA as a therapeutic strategy has
developed in recent years (23–25).
However, only miR-122 as a therapeutic agent for hepatitis C virus
has entered phase 2 clinical trials (26). However, the development of miRNAs
as critical regulators in human disease has shown rapid progress
and may result in a novel class of therapeutic agents. In melanoma,
miRNAs may be identified as oncomiRs or tumor suppressors (27). The results from the present study
indicated that miR-199a-5p is a tumor suppressor, which suppressed
proliferation in the cells via direct targeting of HIF-1α. The
expression levels of miR-199a-5p also inhibited the effects of
HIF-1α in cancer cells. To the best of our knowledge, the present
study is the first to demonstrate the target of miR-199a-5p in
malignant melanoma.
Consistent with the results from the present study,
miR-199a-5p was demonstrated to be a tumor suppressor in aggressive
human cancer cells (28,29). Decreased expression of miR-199a-5p
increased cell invasion via downregulating the expression of
discoidin domain receptor tyrosine kinase 1 in human hepatocellular
carcinoma (29). Furthermore,
miR-199a-5p is highly expressed in Brahma-related gene 1
(Brm)-deficient tumor cell lines, however, this is reduced in
Brm-expressing tumor cells (28).
The luciferase assay demonstrated that miR-199a-5p directly
targeted the Brm subunit of SWI/sucrose non-fermentable and
generate a double-negative feedback loop in human cancers (28). In another previous study,
miR-199a-5p was demonstrated to target HIF-1α and sirtuin 1, during
hypoxia preconditioning antagonistically to the AKT and
beta-adrenergic signaling pathways in neonatal cardiac myocytes,
which may lead to suppression of cancer cell proliferation
(30). The results from previous
studies and the current study suggest that miR-199a-5p inhibits
tumor cell proliferation via targeting multiple genes, further
research is required in the future.
The cell cycle controls cell proliferation and tumor
aggression during tumor growth. The present study demonstrated that
low expression of miR-199a-5p is associated with tumor growth. A
previous study indicates that miRNAs may activate their target
genes in arrested cells by repressing their targets (16). Thus, miR-199a-5p may affect cell
cycle dynamics that induce accumulation of cells at the
G2/M phase in the MDA-MB-231 breast cancer line
(31). However, the effect was not
observed in the MCF7 breast cancer cell line (31). The present study indicated that
overexpression of miR-199a-5p arrested the cell cycle in the
G1 phase, suggesting an underlying molecular mechanism
of miR-199a-5p in suppressing proliferation of malignant
melanoma.
In conclusion, the present study observed low
expression of miR-199a-5p was demonstrated malignant melanoma.
Overexpression of miR-199a-5p inhibited melanoma cells in
vitro by arresting the cells in G1 phase.
Furthermore, HIF-1α was identified as a target of miR-199a-5p and
the results from the present study suggest that overexpression of
miR-199a-5p may be a potential therapeutic strategy for
melanoma.
Acknowledgments
The present study was supported by the National
Natural Science Foundation of China (grant no. 81450061), the
Natural Science Foundation of Hunan Province (grant no. 13JJ2011)
and the Programs of Hunan Province Department of Science and
Technology (grant no. 2014SK3075).
References
|
1
|
Goldstraw P, Ball D, Jett JR, Le Chevalier
T, Lim E, Nicholson AG and Shepherd FA: Non-small-cell lung cancer.
Lancet. 378:1727–1740. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Whitson BA, Groth SS, Duval SJ, Swanson SJ
and Maddaus MA: Surgery for melanoma: A systematic review of the
video-assisted thoracoscopic surgery versus thoracotomy approaches
to lobectomy. Ann Thorac Surg. 86:2008–2016. 2008. View Article : Google Scholar
|
|
3
|
Garbe C, Eigentler TK, Keilholz U,
Hauschild A and Kirkwood JM: Systematic review of medical treatment
in melanoma: Current status and future prospects. Oncologist.
16:5–24. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Le Chevalier T, Arriagada R, Quoix E,
Ruffie P, Martin M, Tarayre M, Lacombe-Terrier MJ, Douillard JY and
Laplanche A: Radiotherapy alone versus combined chemotherapy and
radiotherapy in nonresectable melanoma: First analysis of a
randomized trial in 353 patients. J Natl Cancer Inst. 83:417–423.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Semenza GL: Hypoxia-inducible factor 1:
Master regulator of O2 homeostasis. Curr Opin Genet Dev.
8:588–594. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Iyer NV, Kotch LE, Agani F, Leung SW,
Laughner E, Wenger RH, Gassmann M, Gearhart JD, Lawler AM, Yu AY
and Semenza GL: Cellular and developmental control of O2
homeostasis by hypoxia-inducible factor 1alpha. Genes Dev.
12:149–162. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhong H, De Marzo AM, Laughner E, Lim M,
Hilton DA, Zagzag D, Buechler P, Isaacs WB, Semenza GL and Simons
JW: Overexpression of hypoxia-inducible factor 1alpha in common
human cancers and their metastases. Cancer Res. 59:5830–5835.
1999.PubMed/NCBI
|
|
8
|
Greijer AE and van der Wall E: The role of
hypoxia inducible factor 1 (HIF-1) in hypoxia induced apoptosis. J
Clin Pathol. 57:1009–1014. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Semenza GL: Defining the role of
hypoxia-inducible factor 1 in cancer biology and therapeutics.
Oncogene. 29:625–634. 2010. View Article : Google Scholar :
|
|
10
|
Birner P, Schindl M, Obermair A, Plank C,
Breitenecker G and Oberhuber G: Overexpression of hypoxia-inducible
factor 1alpha is a marker for an unfavorable prognosis in
early-stage invasive cervical cancer. Cancer Res. 60:4693–4696.
2000.PubMed/NCBI
|
|
11
|
Ryan HE, Poloni M, Mc Nulty W, Elson D,
Gassmann M, Arbeit JM and Johnson RS: Hypoxia-inducible
factor-1alpha is a positive factor in solid tumor growth. Cancer
Res. 60:4010–4015. 2000.PubMed/NCBI
|
|
12
|
Eddy SR: Non-coding RNA genes and the
modern RNA world. Nat Rev Genet. 2:919–929. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
He L and Hannon GJ: MicroRNAs: Small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Krek A, Grün D, Poy MN, Wolf R, Rosenberg
L, Epstein EJ, MacMenamin P, da Piedade I, Gunsalus KC, Stoffel M
and Rajewsky N: Combinatorial microRNA target predictions. Nat
Genet. 37:495–500. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Vasudevan S, Tong Y and Steitz JA:
Switching from repression to activation: MicroRNAs can up-regulate
translation. Science. 318:1931–1934. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yiannakopoulou E: Targeting epigenetic
mechanisms and microRNAs by aspirin and other non steroidal
anti-inflammatory agents-implications for cancer treatment and
chemoprevention. Cell Oncol (Dordr). 37:167–178. 2014. View Article : Google Scholar
|
|
18
|
Aprelikova O, Chandramouli GV, Wood M,
Vasselli JR, Riss J, Maranchie JK, Linehan WM and Barrett JC:
Regulation of HIF prolyl hydroxylases by hypoxia-inducible factors.
J Cell Biochem. 92:491–501. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
20
|
Semenza GL: Targeting HIF-1 for cancer
therapy. Nat Rev Cancer. 3:721–732. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Semenza GL: Hypoxia-inducible factor 1
(HIF-1) pathway. Sci STKE. 2007:cm82007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kuphal S, Winklmeier A, Warnecke C and
Bosserhoff AK: Constitutive HIF-1 activity in malignant melanoma.
Eur J Cancer. 46:1159–1169. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kota J, Chivukula RR, O'Donnell KA,
Wentzel EA, Montgomery CL, Hwang HW, Chang TC, Vivekanandan P,
Torbenson M, Clark KR, et al: Therapeutic microRNA delivery
suppresses tumorigenesis in a murine liver cancer model. Cell.
137:1005–1017. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Garzon R, Marcucci G and Croce CM:
Targeting microRNAs in cancer: Rationale, strategies and
challenges. Nat Rev Drug Discov. 9:775–789. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bader AG, Brown D and Winkler M: The
promise of microRNA replacement therapy. Cancer Res. 70:7027–7030.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lanford RE, Hildebrandt-Eriksen ES, Petri
A, Persson R, Lindow M, Munk ME, Kauppinen S and Ørum H:
Therapeutic silencing of microRNA-122 in primates with chronic
hepatitis C virus infection. Science. 327:198–201. 2010. View Article : Google Scholar
|
|
27
|
Cowland JB, Hother C and Grønbaek K:
MicroRNAs and cancer. APMIS. 115:1090–1106. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sakurai K, Furukawa C, Haraguchi T, Inada
K, Shiogama K, Tagawa T, Fujita S, Ueno Y, Ogata A, Ito M, et al:
MicroRNAs miR-199a-5p and-3p target the Brm subunit of SWI/SNF to
generate a double-negative feedback loop in a variety of human
cancers. Cancer Res. 71:1680–1689. 2011. View Article : Google Scholar
|
|
29
|
Shen Q, Cicinnati VR, Zhang X, Iacob S,
Weber F, Sotiropoulos GC, Radtke A, Lu M, Paul A, Gerken G and
Beckebaum S: Role of microRNA-199a-5p and discoidin domain receptor
1 in human hepatocellular carcinoma invasion. Mol Cancer.
9:2272010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rane S, He M, Sayed D, Yan L, Vatner D and
Abdellatif M: An antagonism between the AKT and beta-adrenergic
signaling pathways mediated through their reciprocal effects on
miR-199a-5p. Cell Signal. 22:1054–1062. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yi H, Liang B, Jia J, Liang N, Xu H, Ju G,
Ma S and Liu X: Differential roles of miR-199a-5p in
radiation-induced autophagy in breast cancer cells. FEBS Lett.
587:436–443. 2013. View Article : Google Scholar : PubMed/NCBI
|