Introduction
Organ transplantation is an effective treatment
strategy for end-stage organ failure. Despite improvements in
immunosuppressive strategies, allograft loss due to acute rejection
remains a significant problem. Clinical detection of graft injury
and rejection by repeated tissue biopsy is associated with certain
risks (1,2). Therefore, it is imperative to develop
non-invasive approaches for in vivo imaging to facilitate
the diagnosis and prognosis by evaluating the immune status and
function of transplanted organs. Nuclear medical imaging is a
reliable and highly sensitive technique allowing deep tissue
penetration (3,4). Currently, 18F-fluorodeoxyglucose
(18F-FDG) is the predominant radiolabel used to
visualize the metabolic activity of immune cells. Previous studies
have evaluated 18F-FDG in the assessment of acute
allograft rejection and the efficacy of immunosuppressive treatment
(5,6). Grabner et al (7) reported that
18F-FDG-labeled T lymphocytes accumulated in rat kidney
allografts undergoing acute rejection.
As immunosuppressive treatments inhibit or prevent
the proliferation and/or function of immune cells, including T
cells, B cells and macrophages (8–10),
the present study hypothesizes that the reliability of
18F-FDG is impaired following organ transplantation. The
aim of administering immunosuppressants is to impair the metabolic
activity of effector cells, which results in decreased
18F-FDG uptake. This was demonstrated in our previous
research, which revealed that the immunosuppressant rapamycin
significantly reduced 18F-FDG uptake and prevented
imaging of the graft (11). As an
inhibitor of the intracellular kinase, mammalian target of
rapamycin (mTOR), rapamycin is an immunosuppressive agent commonly
administered to transplant recipients. Rapamycin exerts various
effects on target cells, which include plasmacytoid dendritic cells
(12), effector T cells (13), regulatory T cells (14,15),
B cells (9) and sensitive tumor
cell lines (16). One consequence
of mTOR inhibition is glycometabolism reduction (17,18).
In addition, mTOR inhibition blocks the proliferation, activation
and trafficking of lymphocytes and inhibits the production of
cytotoxic antibodies. Finlay et al (19) demonstrated that the
phosphoinositide 3-kinase-mTOR signaling pathways determined the
expression of key lymph node homing receptors (CD62 L and CCR7) and
regulated lymphocyte trafficking. Therefore, whether
18F-FDG and 18F-FDG-labeled immune cells are
suitable for use in the imaging of rapamycin-treated allografts
remains to be elucidated.
In the present study, 18F-FDG-labeled
splenocytes (with/without rapamycin treatment: FR and FC,
respectively) were investigated for their potential application in
allograft in vivo imaging. 18F-FDG-labeled
splenocytes were transferred into non-obese diabetic/severe
combined immunodeficient (NOD/SCID) mice that had previously
received skin allografts. Tracking of transferred cells was
conducted using whole-body phosphor-autoradiography. The aim of
this study was to evaluate whether 18F-FDG-labeled
splenocyte accumulation is affected by rapamycin treatment.
Materials and methods
Ethical approval
The animal protocol was reviewed and approved by the
Institutional Animal Care and Use Committee at the School of
Medicine, Shandong University (Jinan, China).
Animal model
Female NOD/LtSz-PrKdcscid (NOD/SCID;
H-2d; n=10) mice, BALB/c (H-2d) mice and
C57BL/6 (H-2b; n=40) mice aged 6–8 weeks (weight, 18±2
g) were bred and maintained under defined flora conditions in
individually ventilated (HEPA-filtered air) sterile cages (19 days;
humidity, 50–60%). All mice were maintained under a 12-h light/dark
cycle with access to standard mice chow and sterilized water ad
libitum. Experiments were performed in accordance with national
animal protection guidelines. Full-thickness skin grafts from donor
C57BL/6 mice were harvested and transplanted onto the prepared
graft beds on the right shoulders of recipient NOD/SCID mice
(20). Surgeries were performed in
a sterile environment under anesthesia with 0.6% pentobarbital
sodium (0.1 ml/10 g body weight) by intraperitoneal (i.p.)
injection. Buprenorphine (Reckitt Benckiser Pharmaceuticals,
Richmond, Virginia, USA) was administered (0.1 mg/kg/BW i.p.) for 3
days after surgery to control wound pain. The wounds were then
dressed. Bandages were removed 14 days later and the recipients
with well-healed grafts were selected as the experimental subjects
for imaging at 30 days post-transplantation (n=20 per group).
Cell isolation and labeling
Splenocytes were isolated from BALB/c mice as
previously described (21). The
cells were cultured in cell culture plates in the constant
temperature (37°C) incubator with 10 U/ml recombinant interleukin 2
(rIL-2) or 10 U/ml rIL-2 and 100 nM rapamycin (cat. no. M1768;
AbMole Bioscience, Inc., Houston, TX, USA) for 24 h. Cells were
centrifuged and washed twice at 400 × g for 6 min at 4°C with fresh
PBS and counted using a conventional Neubauer chamber. Splenocytes
(1×107) were then incubated with 15 MBq
18F-FDG (HTA Co., Ltd., Beijing, China) in 37°C
potassium-enriched physiological saline containing 100 IU/ml
insulin (Sigma-Aldrich, St. Louis, MO, USA) for 30 min (22). Splenocytes were collected by
centrifugation at 400 × g for 6 min at 4°C, and radioactivity in
the supernatant and pelleted cells was measured with a Wipe
Test/Well γ-Counter (Capintec, Inc., Ramsey, NJ, USA). Labeling
efficiency was defined as the ratio of radioactivity of cells to
overall radioactivity. To analyze labeling stability,
18F-FDG-labeled splenocytes (5×106) were
incubated in mouse blood plasma (total volume, 400 µl;
Shanghai Fanke Biotechnology Co., Ltd., Shanghai, China) for 5, 10,
30, 60 and 90 min. The ratios of radioactivity of cells to overall
radioactivity (the labeling stability) were calculated. Cells were
divided into two groups: 18F-FDG-labeled control (FC)
cells and 18F-FDG-labeled rapamycin-treated (FR)
cells.
Image acquisition: Dynamic whole-body
phosphor-autoradiography
Recipient mice were injected with 1×107
FC or FR cells via the tail vein. The imaging was performed under
isoflurane (Sigma-Aldrich) inhalation anesthesia (induction, 3%;
maintenance, 1.5%). Anesthetized mice were placed on the storage
phosphor screen plate with their backs to the plate, in subdued
light. The plate was exposed for 10 min. At cessation of exposure,
the plate was immediately covered with an opaque plastic sheet,
then transferred to the Cyclone Plusscanner (PerkinElmer Life
Sciences, Waltham, MA, USA). Dynamic in vivo whole-body
phosphor-autoradiography was performed at 30, 60 and 90 min
following injection of radio-labeled cells. Semi-quantitative
analysis was performed by manually drawing rectangular regions of
interest (n=5) within the graft at each time point. Digital light
units (DLU)/mm2 were obtained using OptiQuant™ image
analysis software 5.0 (PerkinElmer Life Sciences).
Histology
Portions of skin grafts were harvested, and stained
with hematoxylin and eosin for histologic evaluation according to a
previous study (20). The slides
were visualized with an inverted microscope (DMIRB; Leica
Microsystems GmbH, Wetzlar, Germany) at ×10 and ×20 magnifications.
Infiltrating splenocytes were quantified in five ×20 fields.
Statistical analysis
Data are presented as means ± SD (standard
deviation). Statistical analysis was performed using the paired
t-test by GraphPad Prism version 5 software (GraphPad
Software, Inc., La Jolla, CA, USA). P<0.05 was considered to
indicate a statistically significant difference. The non-parametric
Spearman's rho test was used to assess the association between the
accumulation of labeled splenocytes, as measured by radioactivity,
and the number of splenocytes, which was assessed
histologically.
Results
Mouse model and radiolabeling of
splenocytes
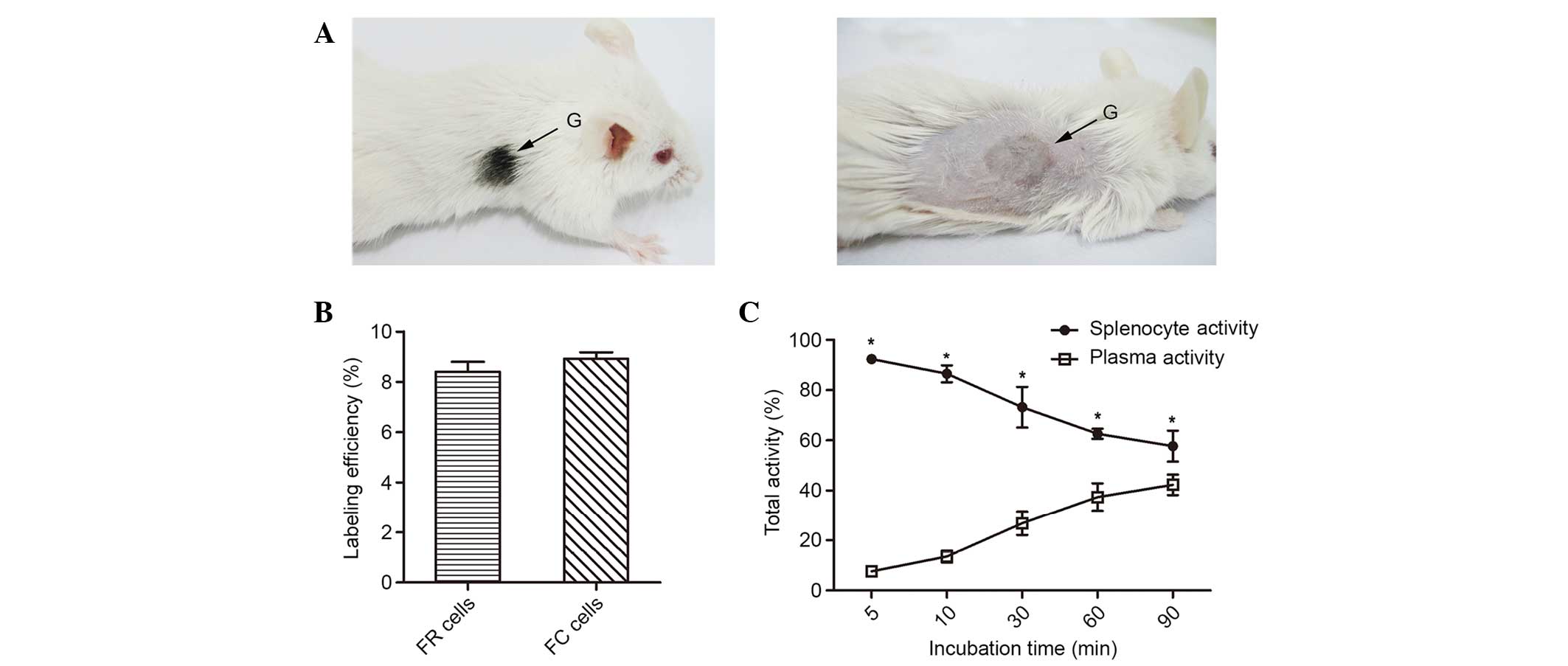
A representative C57BL/6 skin graft on the right
shoulder of a NOD/SCID mouse is presented in Fig. 1A. Although rapamycin reduces the
glycometabolism function of host immune cells during the rejection
response, no significant difference was identified in labeling
efficiency between the FC and FR cells (Fig. 1B). In the FC cells, labeling
stability was evaluated and the percentage of labeled cells slowly
decreased in vitro over time, as retention of
18F-FDG in the cells declined from 92.4±1.7% at 5 min to
73.2±8.1% at 30 min, 62.6±2.0% at 60 min and 57.7±6.2% at 90 min
(Fig. 1C).
In vivo imaging
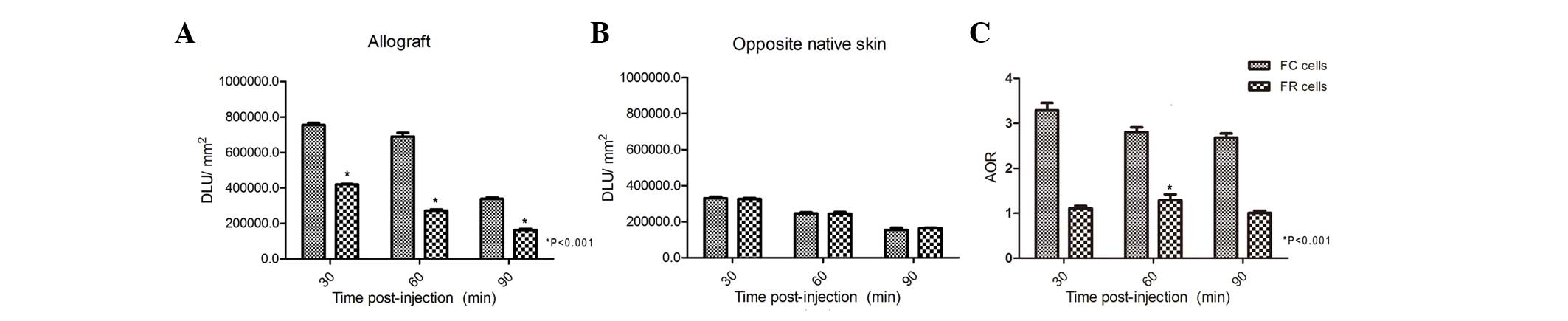
Whole-body phosphor-autoradiography of allografts
revealed significantly reduced radioactivity (DLU/mm2)
in the transplanted skin area (allograft) of mice that received FR
cells compared with those that received FC cells, at 30, 60 and 90
min (Fig. 2A). The opposite native
skin in all mice exhibited consistently low radioactivity (Fig. 2B). The peak of allograft to
opposite native skin ratio (AOR) was significantly lower in the FR
cell group (1.29±0.02 at 60 min) compared with the FC cell group
(3.29±0.17 at 30 min; P<0.001) (Fig. 2C). Whole-body
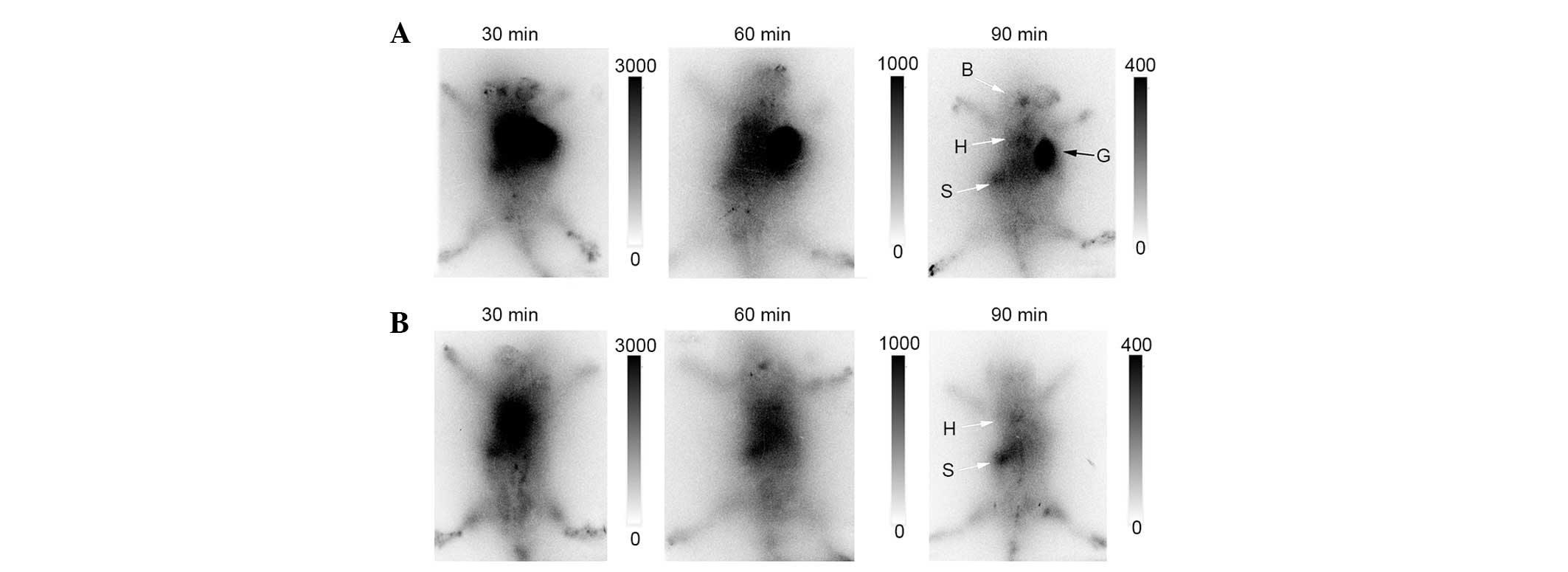
phosphor-autoradiography revealed a clearly elevated radioactive
signal in the allograft area 30 min following injection, which
indicated a significant accumulation of FC cells in skin allografts
(Fig. 3A). High-uptake organs
included the brain, heart and spleen. This may be due to
lymphocytes homing to the spleen and non-specific uptake of free
18F-FDG dissociated from FC cells. However, images of FR
cells did not enable graft visualization at any time point
(Fig. 3B). These results confirmed
that the migration of FR cells to graft areas was notably
impaired.
Histology
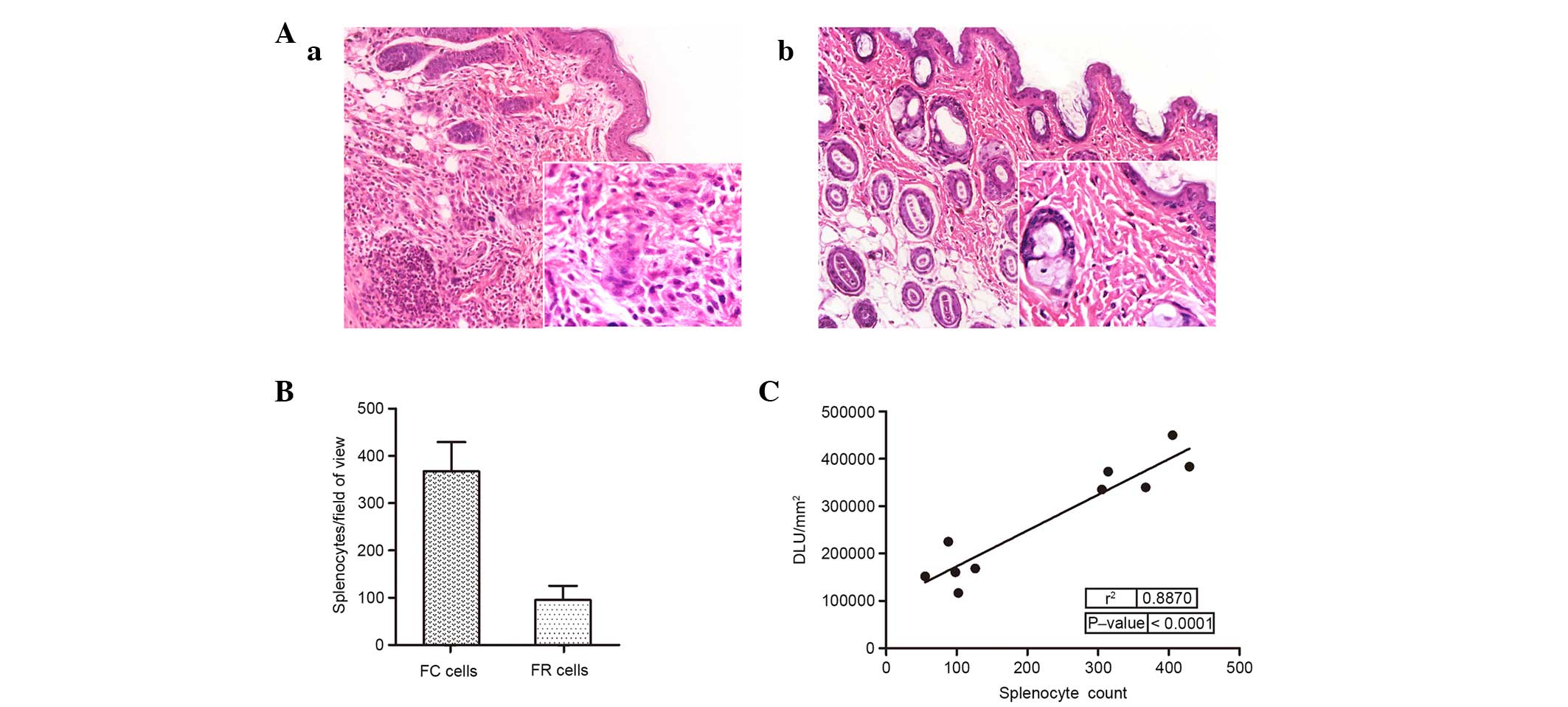
Representative images of allograft sections were
stained with hematoxylin and eosin to estimate lymphocytic
infiltration, and are presented in Fig. 4A. As exhibited in Fig. 4B, significant infiltration of
lymphocytes was absent in the grafts of mice injected with FR cells
(P<0.001). The number of splenocytes per field of view was then
correlated with radioactivity of the graft (Fig. 4C), and revealed to be significantly
correlated (r2= 0.887; P<0.0001).
Discussion
Currently, the lack of effective parameters to
assess allograft function and rejection status limits the success
of treatment. Lymphocyte migration, a crucial element during the
development of the acute alloimmuneresponse, mediates the immune
response during acute allograft rejection; therefore, lymphocytes
present as attractive biomarkers and targets for graft imaging.
Thus, the tracing of lymphocyte trafficking has been evaluated for
allograft monitoring and even prediction of allograft survival
(23,24). However, the use of
immunosuppressants following clinical organ transplantation
severely impairs the proliferation and trafficking of lymphocytes,
and it therefore remains to be elucidated whether imaging of
lymphocytes has clinical applications. In the present study,
C57BL/6 skin was transplanted onto NOD/SCID recipient mice, which
then received FC or FR BABL/c splenocytes 30 days later. NOD/SCID
recipient mice, used to reduce the severity of the immunologically
mediated surgical injury, are deficient in T and B lymphocytes and
have impaired natural killer cells and complement function. The
fully mismatched allografts do not undergo rejection; therefore,
the trafficking of transferred cells could be monitored more
accurately (25).
The results of the present study revealed that
rapamycin had minimal impact on the uptake of 18F-FDG by
naive lymphocytes. No significant difference in labeling efficiency
and stability was identified between FC and FR cells. FC cell
accumulation was observed in the allograft area with high
radioactivity and AORs at all time points; however, FR cell
accumulation in allografts was never clearly imaged. Histological
analysis was performed and confirmed reduced FR cell accumulation
in allografts. The present study employs molecular imaging to
extend the results of a previous study in which 18F-FDG
was determined to be unsuitable as a rapamycin-treated graft
biotracer (11). In the present
study, the accumulation of FR cells in the allograft was barely
visible when compared with FC cells. Furthermore, a significant
correlation was observed between allograft radioactivity and
splenocyte infiltration, which was determined by histology.
There are several limitations of the present study.
The rapamycin dose was selected according to a previous study,
which revealed it to be effective at controlling lymphocyte
trafficking (26). This was
confirmed by the results of the present study; however, no
alternative doses were evaluated. In addition, to avoid
interference by the recipient's immune response, labeled BALB/c
lymphocytes were transferred into NOD/SCID, rather than BALB/c,
recipients. This may not be reflective of the physiological
situation.
Despite increasing knowledge of the rejection
process and tolerance induction, elements of these processes have
not been extensively investigated as imaging targets. In acute
rejection, the non-specific accumulation of
18F-FDG-labeled immune cells at sites of inflammation
following surgery impedes the accurate assessment of the immune
response (27,28). Clinically, immunosuppressive
treatments have been shown to inhibit immune cell migration, which
rendered postoperative detection of grafts impossible (29). Thus, the use of labeled lymphocytes
in in vivo imaging remains controversial. The results of the
present study suggest that splenocyte trafficking was impaired by
the mTOR inhibitor, rapamycin and is therefore not a suitable
method for monitoring patients receiving this particular
immunosuppressant. Alternative imaging strategies are required,
which will enable determination of graft function and immune status
in the presence of immunosuppressive therapy.
Acknowledgments
The present study was supported by a grant from the
National Natural Science Foundation of China (grant no.
81371601).
Abbreviations:
|
18F-FDG
|
18F-fluorodeoxyglucose
|
|
FC cells
|
18F-FDG-labeled
splenocytes
|
|
FR cells
|
18F-FDG-labeled
rapamycin-treated splenocytes
|
|
mTOR
|
mammalian target of rapamycin
|
|
DLU
|
digital light units
|
|
AOR
|
allograft to opposite native skin
ratio
|
References
|
1
|
Baraldi-Junkins C, Levin HR, Kasper EK,
Rayburn BK, Herskowitz A and Baughman KL: Complications of
endo-myocardial biopsy in heart transplant patients. J Heart Lung
Tranplant. 12:63–67. 1993.
|
|
2
|
Mueller TF, Reeve J, Jhangri GS, Mengel M,
Jacaj Z, Cairo L, Obeidat M, Todd G, Moore R, Famulski KS, et al:
The transcriptome of the implant biopsy identifies donor kidneys at
increased risk of delayed graft function. Am J Transplant. 8:78–85.
2008.
|
|
3
|
Virostko J, Henske J, Vinet L, Lamprianou
S, Dai C, Radhika A, Baldwin RM, Ansari MS, Hefti F, Skovronsky D,
et al: Multimodal image coregistration and inducible selective cell
ablation to evaluate imaging ligands. Proc Natl Acad Sci USA.
108:20719–20724. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Signore A, Mather SJ, Piaggio G, Malviya G
and Dierckx RA: Molecular imaging of inflammation/infection:
Nuclear medicine and optical imaging agents and methods. Chem Rev.
110:3112–3145. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tsuji AB, Morita M, Li XK, Sogawa C, Sudo
H, Sugyo A, Fujino M, Sugioka A, Koizumi M and Saga T: 18F-FDG PET
for semiquantitative evaluation of acute allograft rejection and
immunosuppressive therapy efficacy in rat models of liver
transplantation. J Nucl Med. 50:827–830. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Reuter S, Schnöckel U, Edemir B, Schröter
R, Kentrup D, Pavenstädt H, Schober O, Schlatter E, Gabriëls G and
Schäfers M: Potential of noninvasive serial assessment of acute
renal allograft rejection by 18F-FDG PET to monitor treatment
efficiency. J Nucl Med. 51:1644–1652. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Grabner A, Kentrup D, Edemir B, Sirin Y,
Pavenstädt H, Schlatter E, Schober O, Schäfers M, Schnöckel U and
Reuter S: PET with F-18-FDG-labeled T lymphocytes for diagnosis of
acute rat renal allograft rejection. J Nucl Med. 54:1147–1153.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kimura T, Hasegawa T, Nakai H, Azuma T,
Usui N, Sasaki T and Okada A: FTY720 reduces T-cell recruitment
into murine intestinal allograft and prevents activation of
graft-infiltrating cells. Transplantation. 75:1469–1474. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Heidt S, Roelen DL, Eijsink C, van Kooten
C, Claas FH and Mulder A: Effects of immunosuppressive drugs on
purified human B cells: Evidence supporting the use of MMF and
rapamycin. Transplantation. 86:1292–1300. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Brown FG, Nikolic-Paterson DJ, Metz C,
Bucala R, Atkins RC and Lan HY: Up-regulation of macrophage
migration inhibitory factor in acute renal allograft rejection in
the rat. Clin Exp Immunol. 118:329–336. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sun H, Yang G, Liang T, Zhang C, Song J,
Han J and Hou G: Non-invasive imaging of allogeneic transplanted
skin graft by 131I-anti-TLR5 mAb. J Cell Mol Med. 18:2437–2444.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cao W, Manicassamy S, Tang H, Kasturi SP,
Pirani A, Murthy N and Pulendran B: Toll-like receptor-mediated
induction of type I interferon in plasmacytoid dendritic cells
requires the rapamycin-sensitive PI (3)K-mTOR-p70S6K pathway. Nat
Immunol. 9:1157–1164. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Strauss L, Czystowska M, Szajnik M,
Mandapathil M and Whiteside TL: Differential responses of human
regulatory T cells (Treg) and effector T cells to rapamycin. PLoS
One. 4:e59942009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sauer S, Bruno L, Hertweck A, Finlay D,
Leleu M, Spivakov M, Knight ZA, Cobb BS, Cantrell D, O'Connor E, et
al: T cell receptor signaling controls Foxp3 expression via PI3K,
Akt, and mTOR. Proc Natl Acad Sci USA. 105:7797–7802. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Battaglia M, Stabilini A and Roncarolo MG:
Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells.
Blood. 105:4743–4748. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hidalgo M and Rowinsky EK: The
rapamycin-sensitive signal transduction pathway as a target for
cancer therapy. Oncogene. 19:6680–6686. 2000. View Article : Google Scholar
|
|
17
|
Wullschleger S, Loewith R and Hall MN: TOR
signaling in growth and metabolism. Cell. 124:471–484. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Teutonico A, Schena PF and Di Paolo S:
Glucose metabolism in renal transplant recipients: Effect of
calcineurin inhibitor withdrawal and conversion to sirolimus. J Am
Soc Nephrol. 16:3128–3135. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Finlay D and Cantrell D: Phosphoinositide
3-kinase and the mammalian target of rapamycin pathways control T
cell migration. Ann N Y Acad Sci. 1183:149–157. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hou G, Valujskikh A, Bayer J, Stavitsky
AB, Metz C and Heeger PS: In vivo blockade of macrophage migration
inhibitory factor prevents skin graft destruction after indirect
allorecognition. Transplantation. 72:1890–1897. 2001. View Article : Google Scholar
|
|
21
|
Fantini MC, Dominitzki S, Rizzo A, Neurath
MF and Becker C: In vitro generation of CD4+ CD25+ regulatory cells
from murine naive T cells. Nat Protoc. 2:1789–1794. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Meier R, Piert M, Piontek G, Rudelius M,
Oostendorp RA, Senekowitsch-Schmidtke R, Henning TD, Wels WS,
Uherek C, Rummeny EJ and Daldrup-Link HE: Tracking of [18F]
FDG-labeled natural killer cells to HER2/neu-positive tumors. Nucl
Med Biol. 35:579–588. 2008. View Article : Google Scholar
|
|
23
|
Butler KS, Lovato DM, Adolphi NL, Belfon
R, Fegan DL, Monson TC, Hathaway HJ, Huber DL, Tessier TE, Bryant
HC, et al: Development of antibody-tagged nanoparticles for
detection of transplant rejection using biomagnetic sensors. Cell
Transplant. 22:1943–1954. 2013. View Article : Google Scholar
|
|
24
|
Tanaka M, Swijnenburg RJ, Gunawan F, Cao
YA, Yang Y, Caffarelli AD, de Bruin JL, Contag CH and Robbins RC:
In vivo visualization of cardiac allograft rejection and
trafficking passenger leukocytes using bioluminescence imaging.
Circulation. 112(9 Suppl): I105–I110. 2005.PubMed/NCBI
|
|
25
|
Greiner DL, Shultz LD, Yates J, Appel MC,
Perdrizet G, Hesselton RM, Schweitzer I, Beamer WG, Shultz KL,
Pelsue SC, et al: Improved engraftment of human spleen cells in
NOD/LtSz-scid/scid mice as compared with C.B-17-scid/scid mice. Am
J Pathol. 146:888–902. 1995.PubMed/NCBI
|
|
26
|
Valmori D, Tosello V, Souleimanian NE,
Godefroy E, Scotto L, Wang Y and Ayyoub M: Rapamycin-mediated
enrichment of T cells with regulatory activity in stimulated CD4+ T
cell cultures is not due to the selective expansion of naturally
occurring regulatory T cells but to the induction of regulatory
functions in conventional CD4+ T cells. J Immunol. 177:944–949.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ishimori T, Saga T, Mamede M, Kobayashi H,
Higashi T, Nakamoto Y, Sato N and Konishi J: Increased (18)F-FDG
uptake in a model of inflammation: Concanavalin A-mediated
lymphocyte activation. J Nucl Med. 43:658–663. 2002.PubMed/NCBI
|
|
28
|
Pellegrino D, Bonab AA, Dragotakes SC,
Pitman JT, Mariani G and Carter EA: Inflammation and infection:
Imaging properties of 18F-FDG-labeled white blood cells versus
18F-FDG. J Nucl Med. 46:1522–1530. 2005.PubMed/NCBI
|
|
29
|
Chen DL, Wang X, Yamamoto S, Carpenter D,
Engle JT, Li W, Lin X, Kreisel D, Krupnick AS, Huang HJ and Gelman
AE: Increased T cell glucose uptake reflects acute rejection in
lung grafts. Am J Transplant. 13:2540–2549. 2013. View Article : Google Scholar : PubMed/NCBI
|