Introduction
Osteoarthritis (OA) is a chronic disease
characterized by chondrocyte degeneration and cartilage matrix
structure degradation (1). The
pathological characteristics of OA include abnormal chondrocyte
metabolism, damage to the articular cartilage and excessive
degradation of collagen (2).
Furthermore, the leading causes of OA may be associated with
inflammation, oxidative stress, cellular damage and apoptosis of
chondrocytes and synoviocytes (3).
With aging populations and an accelerated pace of life, the
incidence of OA has not only significantly increased in those aged
>50 years (4), but even in the
younger population. Thus, the prevention and treatment of OA has
attracted increased attention worldwide.
In a healthy state, the extracellular matrix of
articular cartilage has a metabolic balance between synthesis and
degradation. However, when cartilage degeneration occurs, matrix
metalloproteinases (MMPs) degrade collagen and proteoglycan in the
cartilage by destroying their structures and disrupting the dynamic
balance between degradation and synthesis of the extracellular
matrix (5). The inflammatory
reaction serves a vital role in the pathogenesis underlying OA. For
instance, inflammatory mediators, such as interleukin (IL)-1β,
tumor necrosis factor (TNF)-α, IL-6, inducible nitric oxide
synthase and prostaglandin E2, participate in the development of OA
and lead to chondrocytes apoptosis and cartilage destruction
(6,7).
Isoflavone is a phytoestrogen found in soybeans that
has a chemical composition and biological effect similar to that of
estrogen (8). Genistein, which is
the major active component of isoflavone, alleviates osteoporosis
caused by decreases in estrogen levels in postmenopausal women, as
well as reduces fracture occurrence and cartilage degeneration
caused by the lack of estrogens (9). Previous studies have reported that
genistein not only restores the histomorphological structures of
cartilage but also corrects the abnormal synthesis of cartilage
matrix caused by estrogen deficiencies (10,11).
Despite these findings, the underlying mechanisms of action of
genistein on chondrocytes are yet to be elucidated. Previous
studies have revealed that genistein can be used to treat bladder
cancer and laryngeal cancer by regulating apoptosis (12,13).
Therefore, the aim of this present study was to investigate the
efficacy and mechanisms of action of genistein for the treatment of
inflammation induced OA by regulating chondrocyte apoptosis in
vitro and in vivo.
Materials and methods
Culturing and identification of human
chondrocytes
Human chondrocytes, CHON-001 (cat. no.
ATCC®CRL-2846TM), used in the present study were from
American Type Culture Collection. Cells were cultured at a
concentration of 5×105 cells in 25-cm2 flasks
with DMEM/F12 (cat. no. 1662222, Gibco; Thermo Fisher Scientific,
Inc.) containing 10% FBS (cat. no. 10099-141, Gibco; Thermo Fisher
Scientific, Inc.) and antibiotics (10% penicillin/streptomycin
solution) in an incubator at 37°C with 5% CO2.
Chondrocytes, which were identified using collagen II
immunohistochemical detection, were inoculated in a culture dishes
pre-coated with a coverslip and collected after 4 h, then washed
with PBS. The cells were fixed using 4% paraformaldehyde at room
temperature for 20 min, embedded with neutral resin at room
temperature for 20 min and sectioned at 18 µm. After being washed
three times with PBS for 5 min each time and incubated with 0.5%
Triton X-100 at room temperature for 20 min, chondrocytes were
treated with 3% H2O2 at room temperature for
15 min and blocked in 5% BSA (cat. no. mu30432, Bio-Swamp) at room
temperature for 20 min after dehydration. Subsequently,
chondrocytes were incubated with anti-collagen II antibodies
(rabbit; 1:100; cat. no. AF0135; Affinity Biosciences) at 4°C
overnight. The following day, sections were washed with PBS and
incubated with the secondary antibody [goat anti-rabbit horseradish
peroxidase (HRP); 1:50; cat. no. A0208; Beyotime Institute of
Biotechnology) for 50 min at 4°C. The sections were then washed
three times with PBS, with each wash lasting for 5 min, and were
stained with 100 µl 3,3-diaminobenzidine (cat. no. P0203; Beyotime
Institute of Biotechnology) at room temperature for 8 min, then
counter-stained with hematoxylin (cat. no. H9627, Sigma-Aldrich;
Merck KGaA) at room temperature for 25 sec. Images were captured
from each section at magnification ×100 and ×400, using an IX51
light microscope (Olympus Corporation).
OA cell model induced with IL-1β
Human chondrocytes were divided into normal
chondrocytes and those from the OA cell model. The OA cell model
was induced using IL-1β (cat. no. SRP6169; Sigma-Aldrich; Merck
KGaA) at the recommended concentration of 10 ng/ml (14). Chondrocytes were treated with IL-1β
at room temperature for 24 h.
Effect of genistein on chondrocytes in
the OA model
Human chondrocytes in the present study were divided
into six groups as follows: i) Group A, normal chondrocytes; ii)
group B, OA cell model (chondrocytes + IL-1β); iii) group C, OA
cell model (chondrocytes + IL-1β) + genistein (cat. no. 1288816,
Sigma-Aldrich; Merck KGaA) (25 µg/ml); iv) group D, OA cell model
(chondrocytes + IL-1β) + genistein (50 µg/ml); v) group E, OA cell
model (chondrocytes + IL-1β) + genistein (100 µg/ml); vi) and group
F, OA cell model (chondrocytes + IL-1β) + estradiol valerate (cat.
no. 1252003, Sigma-Aldrich; Merck KGaA) (100 µg/ml). After
treatment with IL-1β at room temperature for 24 h, chondrocytes in
groups B, C, D, E and F were incubated with different
concentrations of genistein and estradiol valerate as
aforementioned at room temperature for 72 h.
Detection of chondrocytes apoptosis
using flow cytometry
Chondrocyte apoptosis was assessed using flow
cytometry (FACSCalibur, BD Biosciences). The apoptotic rate was
calculated as the percentage of early + late apoptotic cells. A
total of 2×105 cells were seeded in 6-well plates.
Briefly, a cell apoptosis detection kit (cat. no. KGA108, Nanjing
KeyGen Biotech Co. Ltd.) was used: 500 µl binding buffer mixed with
5 µl Annexin V-FITC and 5 µl propidium iodide (PI) was added to
each well. Cells were then incubated at 25°C in darkness for 15
min. In the flow cytometry assay, green fluorescence indicated
Annexin V-FITC and red fluorescence indicated PI. The excitation
wavelength of FITC was 488 nm and was detected in the FL1 channel.
Red fluorescence was detected in the FL2 channel. The staining
profile was analyzed using Cell Quest Pro software, version 5.1 (BD
Biosciences). In the two-color dot plot, the x-axis represented FL1
and y-axis represented FL2. In total, 10,000 events were collected
for each sample.
Western blot analysis for
chondrocytes
Protein from chondrocytes was extracted using a
lysis buffer [containing 20 mM Tris-HCl (pH 7.4), 1 mM EDTA, 1%
TritonX-100, 1.5 M NaCl, 0.1% SDS and 1% phenylmethanesulfonyl
fluoride; cat. no. ST505; Beyotime Institute of Biotechnology] and
was preserved at −80°C after centrifugation at 16,000 × g for 10
min at 4°C. The protein concentration was determined using the
Bio-Rad DC protein assay(ChemiDoc XRS + system, Bio-Rad
Laboratories, Inc.). A total of 30 µg protein from each group was
analyzed using electrophoresis on 10% SDS-PAGE with a constant
voltage initially set at 80 V for 30 min, and then 120 V for 120
min. The proteins were transferred onto PVDF membranes in the
Bio-Rad TransBlot apparatus (Bio-Rad Laboratories, Inc.) at 100 V
for 120 min. Subsequently, the membrane was blocked with 5% non-fat
dry milk, which was dissolved in 0.15 M NaCl containing
Tris-buffered saline-0.2% Tween-20 (TBST) and 10 mM Tris-HCl, for 2
h at room temperature. The membrane was incubated overnight at 4°C
with the following primary antibodies: Aggrecan (rabbit; 1:1,000;
cat. no. DF7561; Affinity Biosciences), caspase 3 (rabbit; 1:1,000;
cat. no. AF6311; Affinity Biosciences), collagen II (rabbit;
1:1,000; cat. no. AF0135; Affinity Biosciences), estrogen receptor
α (ERα; rabbit; 1:500; cat. no. AF6058; Affinity Biosciences) and
GAPDH (rabbit; 1:1,000; cat. no. AP0063; Bioworld Technology,
Inc.). The following day, the membrane was washed with TBST three
times for 5 min each time at room temperature and was then
incubated with secondary antibodies (goat anti-rabbit HRP; 1:50;
cat. no. A0208; Beyotime Institute of Biotechnology) at room
temperature for 2 h. Subsequently, the membrane was incubated with
ECL chemiluminescence reagents (cat. no. NCI5079, Thermo Fisher
Scientific, Inc.) and exposed to Kodak X-OMAT films for detection
with ImageJ v1.8.0 (National Institutes of Health).
TNF-α levels in chondrocytes analyzed
using ELISA
To analyze the expression levels of TNF-α in
chondrocytes of each group, a TNF-α ELISA kit (cat. no. EHJ-10039;
Xiamen Huijia Biotechnology Co., Ltd.) was used according to the
manufacturer's protocol. Optical density values were determined
using a microplate reader at 450 nm and the concentration of TNF-α
was calculated using the linear regression equation
(y=0.004×-0.089, R2=0.9874).
OA model in Sprague-Dawley rats
All 40 pathogen-free male juvenile Sprague-Dawley
rats (age, 4 weeks; weight, 180–200 g) were provided by the
Shanghai Experimental Animal Center of the Chinese Academy of
Science [animal certificate no. SCXK (Shanghai 2007-0005)] and
housed at a constant temperature of 23±1°C, humidity of 40–70% and
under a 12-h light-dark cycle, with free access to water and food.
All animal studies, including the mice euthanasia procedure, were
performed in compliance with the regulations and guidelines of the
Zhejiang Chinese Medical University Institutional Animal Care and
Conducted according to the Association for Assessment and
Accreditation of Laboratory Animal Care (AAALAC) (15) and the Institutional Animal Care and
Use Committee (IACUC) (16)
guidelines. The present study was approved by Zhejiang Chinese
Medical University Institutional Animal Care.
The OA rat model used in this study was performed
based on a previous report (17).
Rats were anesthetized with 2.25% pentobarbital sodium (45 mg/kg,
intraperitoneally) and the medial knee joint of one of the limbs of
each rat was incised to expose an area of skin ~1×2 cm, thus
opening the knee joint cavity. Then, the medial and anterior
crucial ligaments were exposed and resected. Subsequently, the
medial meniscus was removed and sutured. On days 1–3 post-surgery,
rats were treated with an intramuscular injection of 1 ml
penicillin (40,000 U/ml). All 40 rats were randomly divided into a
normal group, OA group, genistein group and estradiol valerate
group (n=10 for each group). Rats in the OA, genistein and
estradiol valerate groups underwent OA model surgery as
aforementioned and were intragastrically administrated normal
saline (2 ml), genistein (20 mg/kg) (18) or estradiol valerate (0.8 mg/kg)
(19), respectively, each day for
6 weeks. Rats in the normal group were intragastrically
administrated normal saline (2 ml) each day for 6 weeks and housed
under the same conditions.
Paraffin-cut section of cartilage in
OA rats model
After 6 weeks post-surgery, all the rats were
sacrificed by dislocation of the neck and the cartilage of rats in
each group was removed to create paraffin-cut sections. Cartilage
tissues were fixed with formaldehyde-acetic acid-ethanol fixative
(90 ml 70% ethanol, 5 ml acetic acid and 5 ml formaldehyde) at room
temperature for 48 h, embedded in paraffin, and sectioned coronally
to 18 µm. Sections were dewaxed using xylene (cat. no. 10023418;
Sinopharm Chemical Reagent Co., Ltd.) after drying and placed in
100, 95, 85 and 75% ethanol respectively, each for 5 min at room
temperature, and finally soaked in distilled water. After staining
with Masson trichrome (cat. no. DC0032; Beijing Leagene Biotech
Co., Ltd.) and toluidine blue (cat. no. G1032; Servicebio, Inc.) at
room temperature for 20 min, the histopathological changes of
cartilage were viewed under an optical microscope at ×400
magnification.
Levels of TNF-α and IL-1β in synovial liquid
analyzed using ELISA. Synovial liquid (1 ml) was collected from
each rat and the concentration of TNF-α and IL-1β in the synovial
liquid was calculated using ELISA kits (cat. no. EHJ-10039 and
EHJ-10293, respectively; Xiamen Huijia Biotechnology Co., Ltd.)
according to the manufacturer's protocol.
Western blot analysis for articular
cartilages
The cartilage of rats in each group was removed with
a small scalpel and scissors for western blot analysis. Protein
from the articular cartilage was extracted using SDS lysis buffer
(cat. no. 10014118, Sinopharm Chemical Reagent Co., Ltd.), and the
protein expression levels of collagen II, aggrecan, caspase 3 and
ERα were detected using the aforementioned protocol.
Statistical analysis
All data were analyzed using SPSS 19.0 software (IBM
Corp.). Each experiment was repeated ≥3 times. Comparisons between
experimental groups were performed using one-way ANOVA and Tukey's
test. Data are presented as the mean ± SD. P<0.05 was considered
to indicate a statistically significant difference.
Results
Collagen II immunohistochemical
identification of human chondrocytes
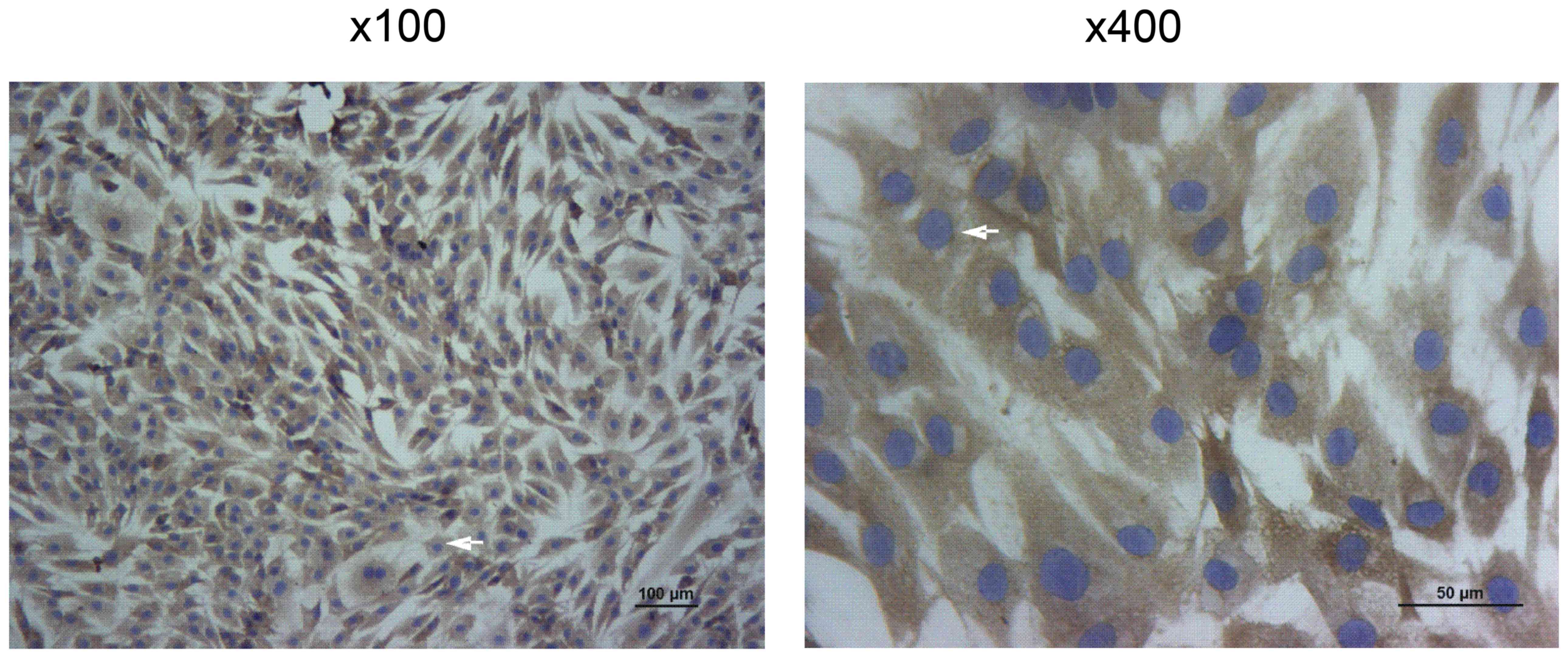
The collagen II immunohistochemical identification
of human chondrocytes is presented in Fig. 1. Collagen II is specifically
secreted by chondrocytes and can be accurately used to localize
chondrocytes (20).
After being counterstained with hematoxylin,
collagen II was labelled brown and the cell nucleus was stained
blue-purple. Collagen II immunohistochemical staining identified
the heterochromatism of cultured chondrocytes, which were confirmed
to be human chondrocytes (Fig.
1).
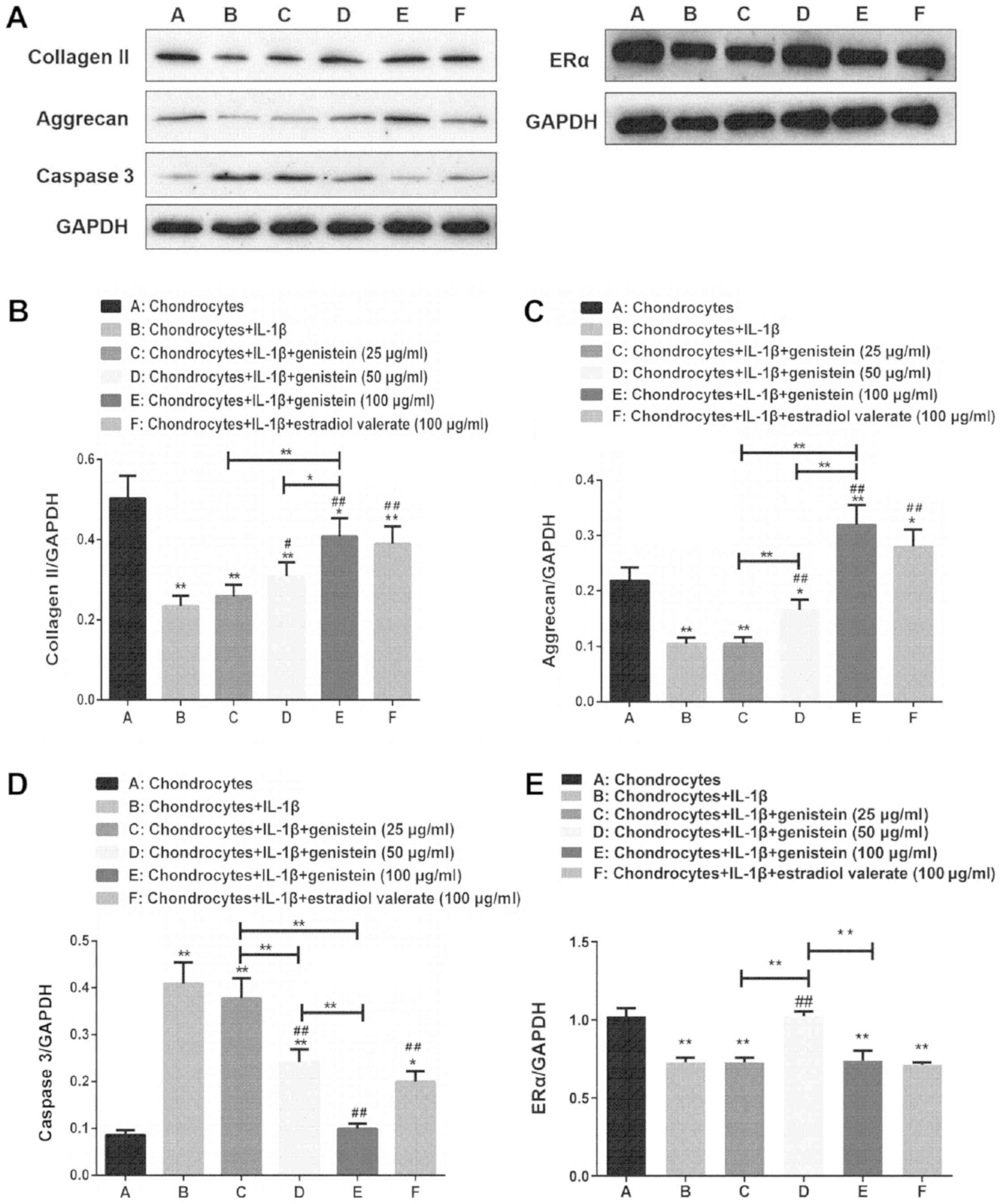
Expression levels of collagen II,
aggrecan, caspase 3 and ERα of chondrocytes in the OA cell
model
The protein expression levels of collagen II,
aggrecan, caspase 3 and ERα of chondrocytes in the OA model treated
with varying concentrations of genistein and estradiol valerate
were detected using western blotting (Fig. 2A). Compared with normal
chondrocytes in group A, the expression levels of collagen II
(Fig. 2B), aggrecan (Fig. 2C) and ERα (Fig. 2E) decreased in group B (all
P<0.01), while those of caspase 3 increased (Fig. 2D; P<0.01). Compared with group
B, the expression levels of collagen II and aggrecan increased in
groups D and E (P<0.01 and P<0.05), while the expression of
caspase 3 decreased in a dose-dependent manner (both P<0.01).
Furthermore, compared with group B, the expression of ERα increased
in group D (P<0.01). Compared with group E, the expression
levels of collagen II and aggrecan decreased in groups C and D
(P<0.01 and P<0.05), while the expression of caspase 3
increased in groups C and D (P<0.01), and the expression of ERα
increased in group D (P<0.01).
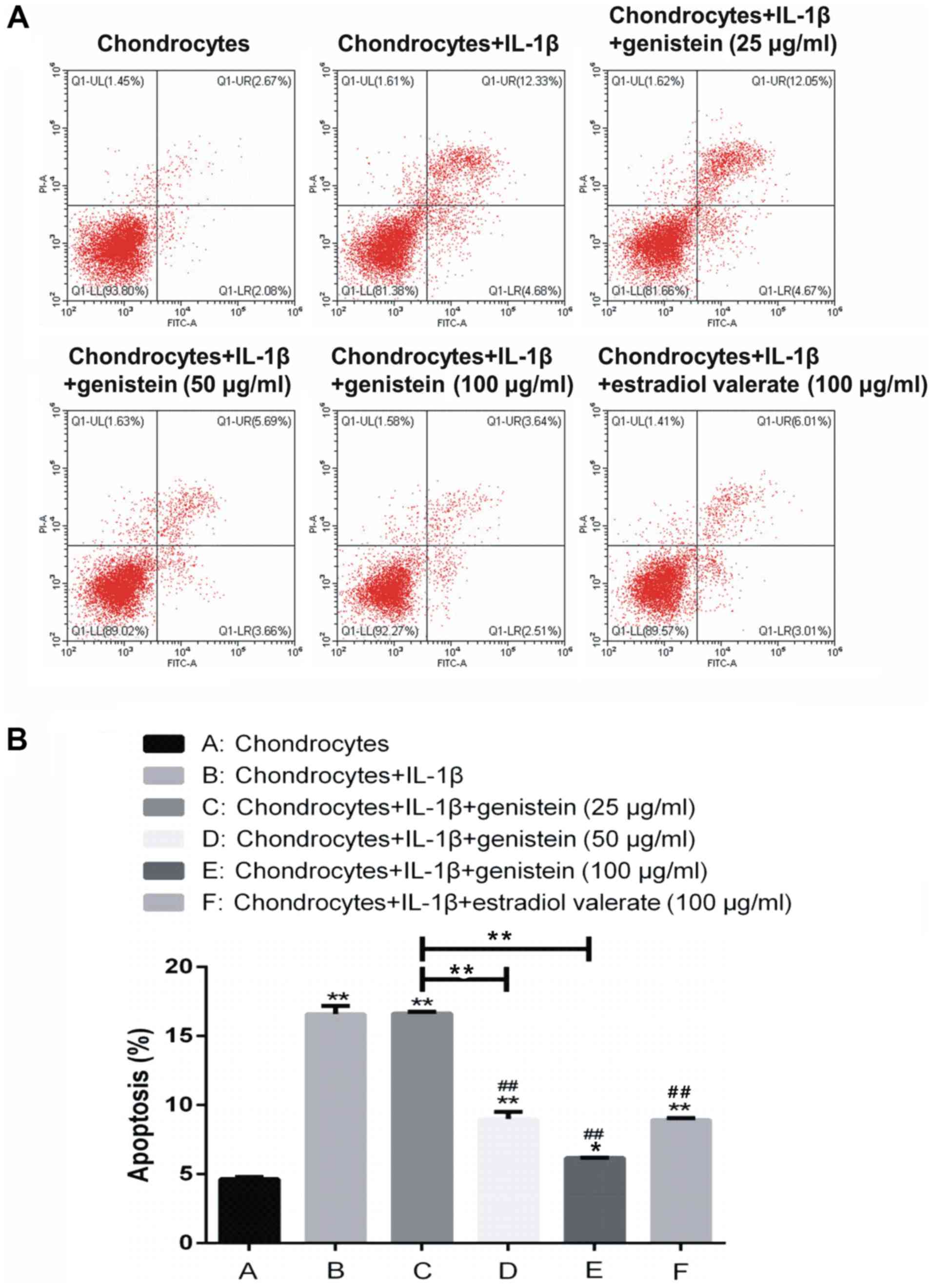
Effect of different concentrations of
genistein on chondrocyte apoptosis
The extent of chondrocytes apoptosis was detected
using flow cytometry (Fig. 3).
Compared with group A, the rate of apoptosis in groups B, C, D, E
and F was significantly increased (P<0.01 and P<0.05). In
addition, compared with group B, there was no significant
difference in the apoptotic rate of group C with 25 µg/ml genistein
(P>0.05); while in groups D, E and F, the rate of apoptosis was
significantly decreased (P<0.01). Thus, the results indicated
that IL-1β promoted chondrocyte apoptosis, but genistein reversed
this effect in a dose-dependent manner.
Effect of various concentrations of
genistein on the level of TNF-α in chondrocytes
TNF-α plays an important role in the development of
OA (21). The levels of TNF-α in
chondrocytes of each group were measured using ELISA. Compared with
group A, the levels of TNF-α were higher in groups B, C, D and F
(P<0.01; Fig. 4). However, the
levels of TNF-α in groups C, D, E and F, with different
concentrations of genistein, were significantly decreased compared
with group B (P<0.01). Compared with group C, the levels of
TNF-α significantly decreased in group D and E (P<0.01).
 | Figure 4.As the concentration of genistein
increases, the levels of TNF-α in the chondrocytes of the OA model
gradually decrease. Compared with group A, the levels of TNF-α were
higher in groups B, C, D and F, as detected using ELISA. However,
the expression levels of TNF-α in groups C, D, E and F, treated
with various concentrations of genistein and estradiol valerate,
significantly decreased compared with group B. Data are presented
as the mean ± SD (n=10). **P<0.01 vs. group A or as indicated;
##P<0.01 vs. B. OA, osteoarthritis; TNF, tumor
necrosis factor; IL, interleukin. |
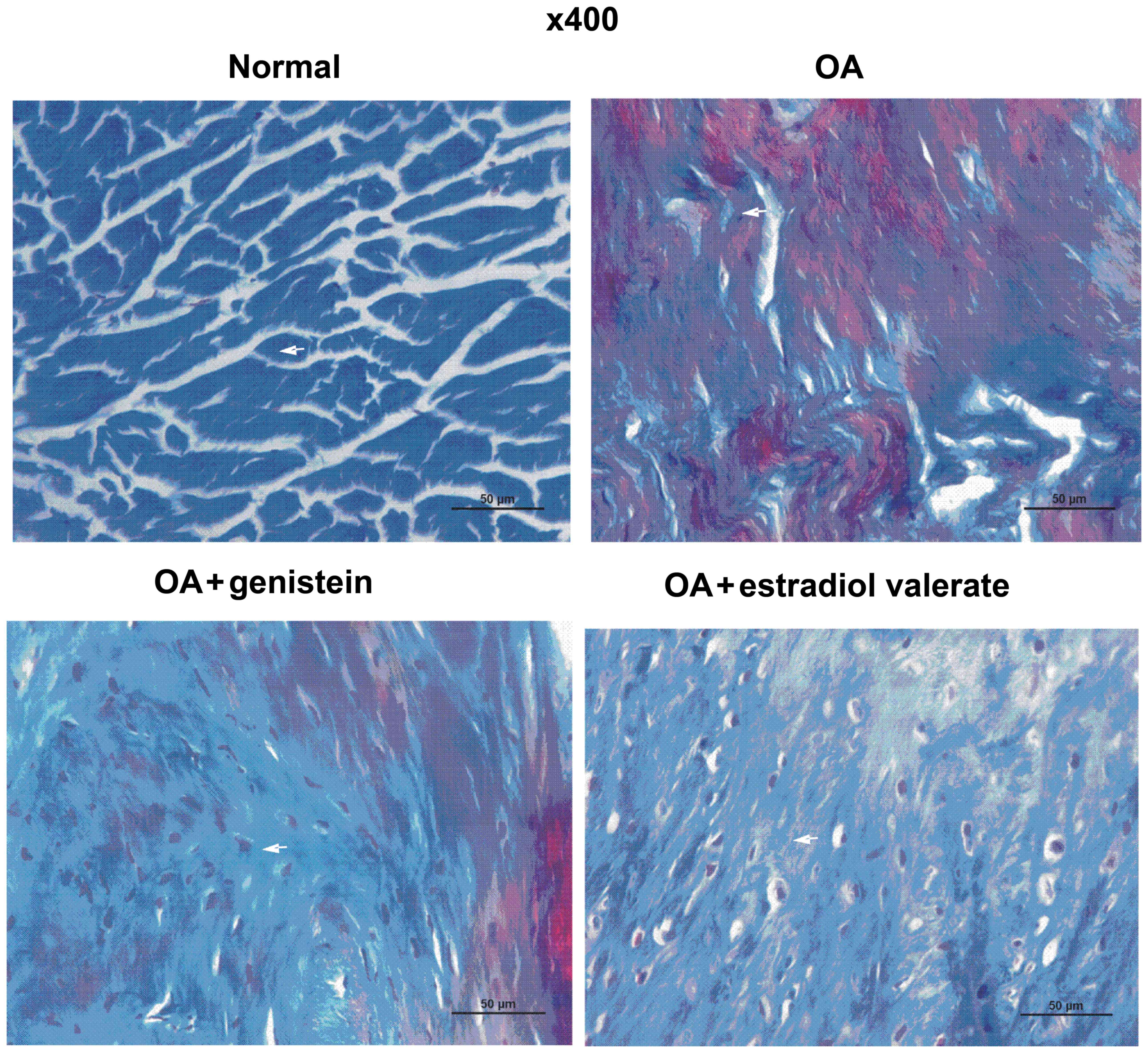
Effect of genistein on the collagen
content in cartilage of OA model rats
The Masson staining of the articular cartilage in
each group of the OA model rats is presented in Fig. 5. Collagenous fibers were closely
arranged and were stained blue in the normal group. Compared with
the normal group, collagenous fibers in the OA group were
disordered and locally broken, while the articular cartilage was
degraded with a decreased collagen content. Furthermore, compared
with the OA group, collagenous fibers in the genistein group were
more closely arranged and had an increased collagen content, which
reduced the cartilage degradation. In the estradiol valerate group,
the staining results were similar to the genistein group, although
the collagenous fibers were more closely arranged and collagen
content was increased compared with the OA group.
Effect of genistein on the acid
glycosaminoglycan content in the cartilage of the OA model
rats
The toluidine blue staining of articular cartilage
from each group of the OA model rats is presented in Fig. 6. Toluidine blue is a basic dye,
which turns blue after combining with acid glycosaminoglycan
(22). Acid glycosaminoglycans
were abundantly identified in the cartilage matrix of the normal
group rats, demonstrating metachromasia. Furthermore, the acid
glycosaminoglycan content in the cartilage matrix was markedly
decreased in the OA group but notably increased in the genistein
group. The acid glycosaminoglycan content in the estradiol valerate
group rats increased compared with the OA group but was lower
compared with the normal group.
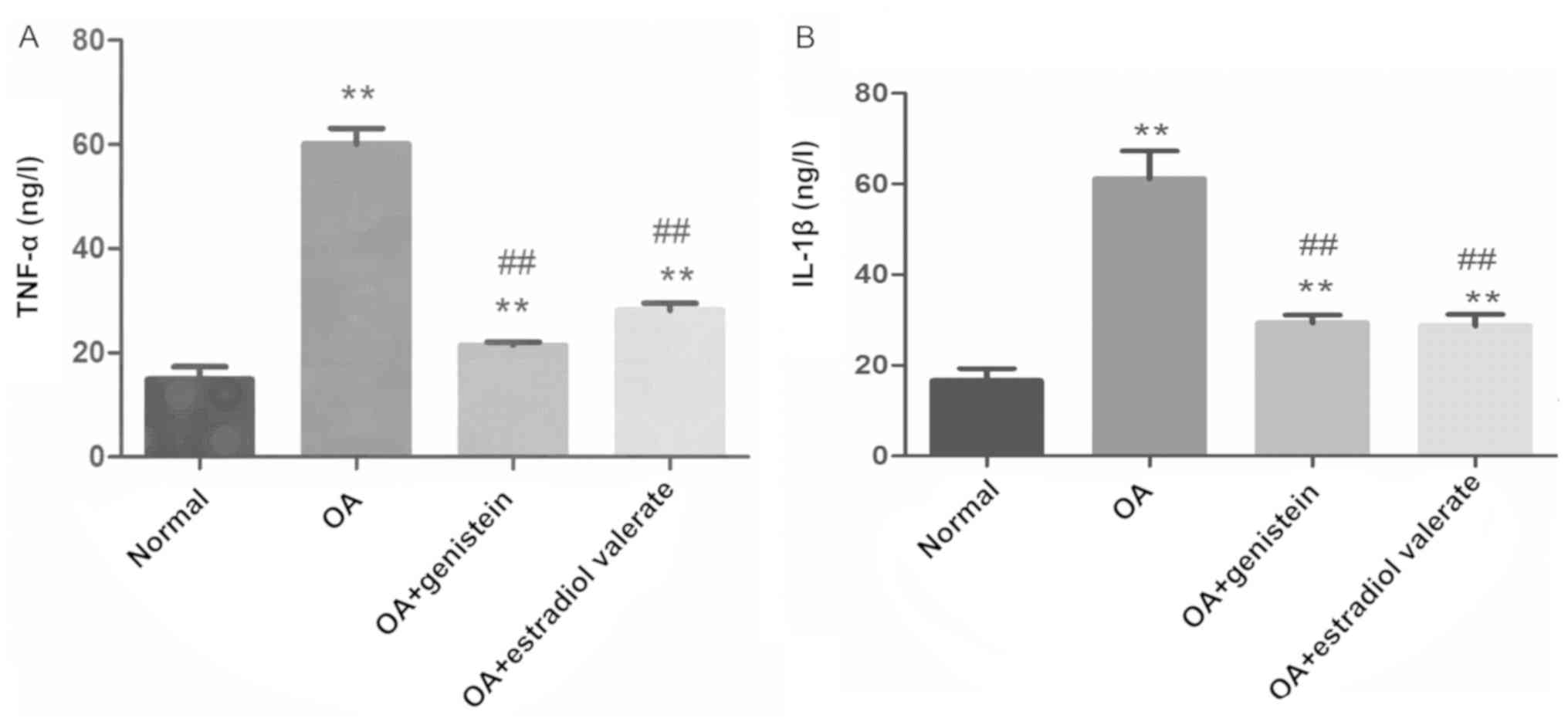
Effect of genistein on the levels of
TNF-α and IL-1β in the synovial fluid of the OA model rats
The levels of TNF-α and IL-1β in the synovial fluid
of each group were detected using ELISA (Fig. 7). Compared with the normal group,
levels of TNF-α and IL-1β in the OA group were significantly
increased (both P<0.01). However, in the genistein and estradiol
valerate treated groups, TNF-α and IL-1β levels were significantly
decreased compared with the OA group (all P<0.01).
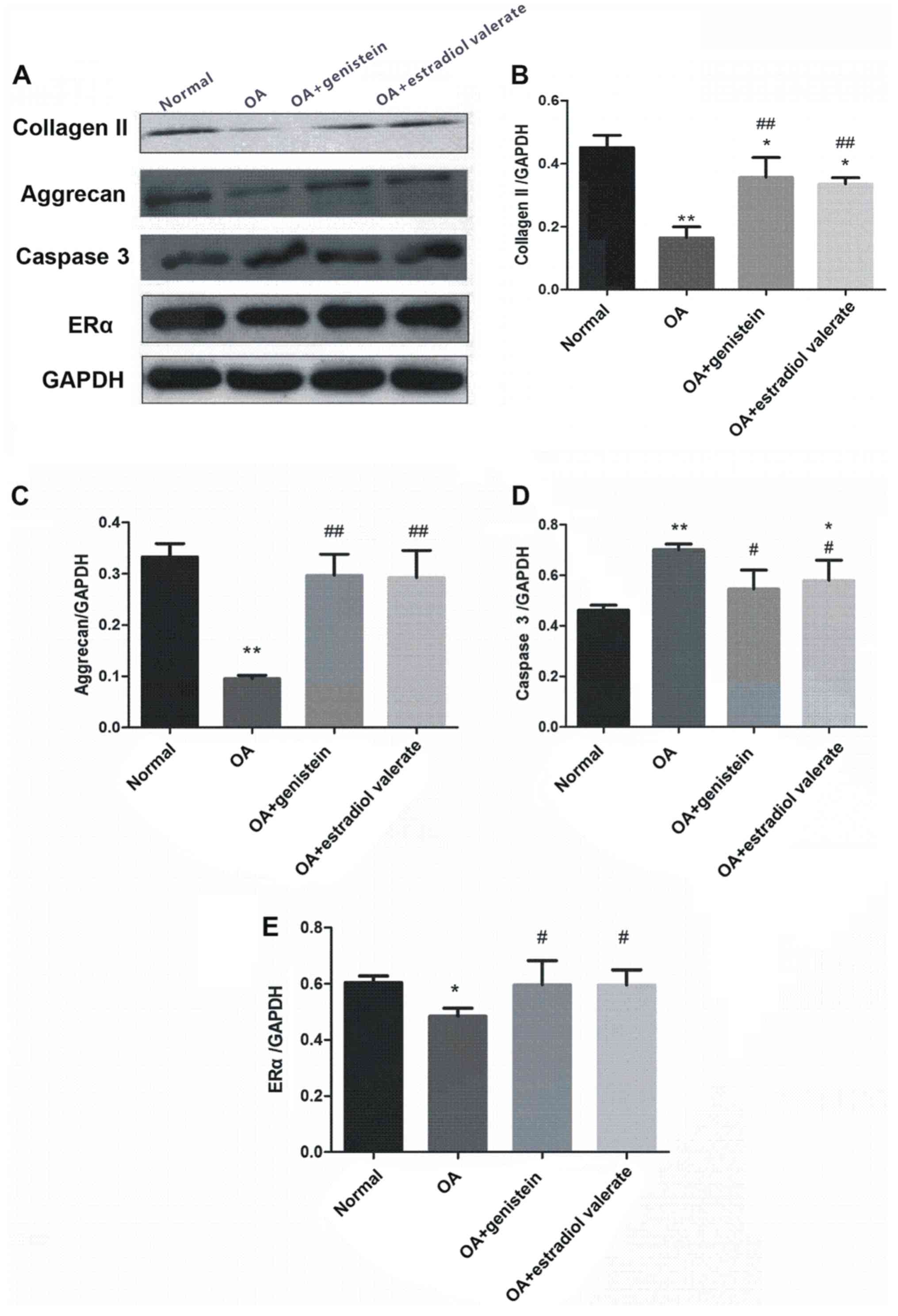
Expression levels of collagen II,
aggrecan, caspase 3 and ERα in the articular cartilage in the OA
model rats
The expression levels of collagen II, aggrecan,
caspase 3 and ERα from the articular cartilage in the OA model rats
treated with genistein and estradiol valerate were measured using
western blotting (Fig. 8A).
Compared with the normal group, protein expression levels of
collagen II (Fig. 8B; P<0.01),
aggrecan (Fig. 8C; P<0.01) and
ERα (Fig. 8E; P<0.05) were
decreased in the OA group, while caspase 3 expression was increased
(Fig. 8D; P<0.01). Moreover,
compared with the OA group, the expression levels of collagen II
(P<0.01), aggrecan (P<0.01) and ERα (P<0.05) were
increased in the genistein and estradiol valerate groups, but the
expression of caspase 3 was decreased (P<0.05).
Discussion
Currently, the repair and treatment of cartilage
degeneration in OA remains challenging and requires further
investigation. In the early and mid-term stages of OA, cartilage
repair promoting drugs are the most extensively used treatment in
the clinic (23). Chondroitin
sulfate, hyaluronic acid and glucosamine polysaccharides, which are
most commonly used, function under the basic treatment principle of
promoting the formation of cartilage matrix components and slowing
collagen frame disintegration, thus inhibiting the local
inflammatory response of the joint and constructing a molecular
barrier that has a chemical protective role to reduce chondrocyte
apoptosis (24–26). However, it has been reported that
these drugs can only temporarily relieve pain, while the local site
is mainly composed of fibrocartilage and the outcome remains
degeneration and necrosis in the local cartilage (27).
Related biological proteins, such as serum
adiponectin, leptin and resistin, released from the articular
cartilage and synovium can trigger an intra-articular inflammatory
reaction, which is closely related to cartilage degeneration in OA
(28,29). Chondrocytes are the main target of
inflammatory mediators, particularly IL-1β and TNF-α (30). Inflammatory mediators activate the
p38 mitogen-activated protein kinase, ERK1/2, JNK and NF-κB
signaling pathways in chondrocytes by binding to receptors on the
superficial surface of the articular cartilage and inducing the
release of MMPs to degrade collagen and proteoglycan in cartilage
(31,32). Furthermore, inflammatory mediators,
such as IL-1β and TNF-α, promote gene transcription of Fas, Fas
ligand and TNF receptor 1, thus accelerating the release of
cytochrome c from mitochondria and activating the caspase gene to
induce upregulation of apoptosis in chondrocyte (33).
Phytoestrogens, which are extracted from plants, are
highly safe and reliable. Previous studies have reported that the
chemical structure and pharmacological activity of phytoestrogens
are similar to that of estrogens (34–36).
The estrogen-like effects of phytoestrogens in human and mammalian
cells are generated from the combination between phytoestrogens and
ERs (37). Isoflavones, natural
organic compounds found in traditional Chinese herbal medicine, are
a type of phytoestrogens that have a notable effect on the
regulation of ER (38). The
chondrocyte is a target cell of estrogen and there are ERs on the
surface of chondrocytes (39).
Moreover, estrogen regulates chondrocyte metabolism in the
treatment of OA by inhibiting the release of MMPs and promoting
cartilage matrix formation (40).
Genistein, which is the main component of isoflavone extracted from
soybean, has antitumor, neuroprotective, bone
metabolism-regulating, anti-osteoporosis, anti-inflammatory and
antioxidant effects (41–43).
In the present study, it was found that genistein
significantly inhibited chondrocyte apoptosis induced by IL-1β,
decreased the levels of TNF-α and promoted the expression levels of
collagen II and aggrecan in chondrocytes, in a dose-dependent
manner. In addition, these data were further demonstrated in the OA
rat model. Genistein increased the collagen and acid
glycosaminoglycan content, promoted the expression levels of
collagen II and aggrecan, decreased the expression of caspase 3 in
cartilage and downgraded the levels of TNF-α and IL-1β, thus
alleviating cartilage degradation. Therefore, these data indicated
that genistein reduced the release of TNF-α and IL-1β in OA,
downregulated the apoptotic-related protein expression and
decreased chondrocyte apoptosis, thus reducing cartilage
degradation.
ERs are expressed in articular cartilage, and
participate in cartilage growth and absorption (44). ERs are also associated with the
occurrence and development of OA and include three subtypes, ERα,
ERβ and ERγ (45,46). According to previous research, ERα
serves an important role in cartilage formation and is vital for
the maintenance of cartilage homeostasis (47). Levels of estrogen affect the
expression and function of ERs in the articular cartilage. For
instance, decreased levels of estrogen downregulate the expression
levels of the ER gene, while estrogen supplementation can delay the
development of OA (48). Estrogen
replacement therapy also relieves cartilage injury, inhibits the
catabolic activity of proteases within the chondrocyte
extracellular matrix and can treat OA (49). In the present in vitro
study, the expression of ERα increased significantly in group D
following treatment with 50 µg/ml genistein. In addition, the
expression of ERα in the genistein and estradiol valerate groups
were significantly enhanced compared with the OA group in
vivo study. These preliminary results suggested that genistein
upregulated the expression levels of ER in chondrocytes when
treating cells for OA. However, the sex of the experimental animals
and the polymorphism presented in the ER genotype may affect the
expression and function of ER (50). Therefore, additional studies
investigating the regulation of ER by genistein for the treatment
of OA should be performed. Moreover, a novel clinical treatment for
OA aiming to increase the expression of ER levels would be
beneficial.
In conclusion, the present study demonstrated that
genistein decreased the release of TNF-α and IL-1β in the OA model,
thus reducing chondrocyte apoptosis and slowing cartilage
degeneration. The current findings also suggested that genistein
could be used as a suitable drug to treat OA by preventing
cartilage degeneration. Moreover, it was indicated that the
underlying mechanism of action was related to inflammatory
mediators that reduced chondrocyte apoptosis. However, further
studies should be performed to investigate the underlying mechanism
of action for genistein in treating OA.
Acknowledgements
The authors would like to thank Professor Changxing
Wang and Dr Weidong Wang from The Second Affiliated Hospital of
Zhejiang Chinese Medical University for their technical
assistance.
Funding
The present study was supported by the Natural
Science Foundation of China (grant no. 81803876), Science and
Technology Program of Zhejiang province (grant no. 2018C37107) and
Medical Science and Technology Project of Zhejiang province (grant
no. 2016KYA153).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JH designed the study. YZ, QML, PTG, YH and ZCY
performed the experiments. YZ and QML reviewed and edited the
manuscript. All authors wrote, read and approved the manuscript and
agree to be accountable for all aspects of the research in ensuring
that the accuracy or integrity of any part of the work are
appropriately investigated and resolved.
Ethics approval and consent to
participate
The present study was approved by Zhejiang Chinese
Medical University Institutional Animal Care and conducted
according to the AAALAC and the IACUC guidelines.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Guilak F, Nims RJ, Dicks A, Wu CL and
Meulenbelt I: Osteoarthritis as a disease of the cartilage
pericellular matrix. Matrix Biol 71–72. 40–50. 2018. View Article : Google Scholar
|
|
2
|
Yang X, Guan Y, Tian S, Wang Y, Sun K and
Chen Q: Mechanical and IL-1β responsive miR-365 contributes to
osteoarthritis development by targeting histone deacetylase 4. Int
J Mol Sci. 17:4362016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Whitworth DJ and Banks TA: Stem cell
therapies for treating osteoarthritis: Prescient or premature? Vet
J. 202:416–424. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Singer SP, Dammerer D, Krismer M and
Liebensteiner MC: Maximum lifetime body mass index is the
appropriate predictor of knee and hip osteoarthritis. Arch Orthop
Trauma Surg. 138:99–103. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Baker M, Brook BS and Owen MR:
Mathematical modelling of cytokines, MMPs and fibronectin fragments
in osteoarthritic cartilage. J Math Biol. 75:985–1024. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kaneva MK, Kerrigan MJ, Grieco P, Curley
GP, Locke IC and Getting SJ: Chondroprotective and
anti-inflammatory role of melanocortin peptides in TNF-α activated
human C-20/A4 chondrocytes. Br J Pharmacol. 167:67–79. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yao ZZ, Hu AX and Liu XS: DUSP19 regulates
IL-1β-induced apoptosis and MMPs expression in rat chondrocytes
through JAK2/STAT3 signaling pathway. Biomed Pharmacother.
96:1209–1215. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Johnson KA, Vemuri S, Alsahafi S, Castillo
R and Cheriyath V: Glycone-rich Soy Isoflavone Extracts Promote
Estrogen Receptor Positive Breast Cancer Cell Growth. Nutr Cancer.
68:622–633. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu FC, Wang CC, Lu JW, Lee CH, Chen SC,
Ho YJ and Peng YJ: Chondroprotective effects of genistein against
osteoarthritis induced joint inflammation. Nutrients. 11:E11802019.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Oliviero F, Scanu A, Zamudio-Cuevas Y,
Punzi L and Spinella P: Anti-inflammatory effects of polyphenols in
arthritis. J Sci Food Agric. 98:1653–1659. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pie JE, Park JH, Park YH, Ryu YM, Kim KN,
Suh SW, Becker KG, Cho-Chung YS and Kim MK: Effect of genistein on
the expression of bone metabolism genes in ovariectomized mice
using a cDNA microarray. J Nutr Biochem. 17:157–164. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang Y, Wang H, Zhang W, Shao C, Xu P, Shi
CH, Shi JG, Li YM, Fu Q, Xue W, et al: Genistein sensitizes bladder
cancer cells to HCPT treatment in vitro and in vivo via
ATM/NF-κB/IKK pathway-induced apoptosis. PLoS One. 8:e501752013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ma CH, Zhang YX, Tang LH, Yang XJ, Cui WM,
Han CC and Ji WY: MicroRNA-1469, a p53-responsive microRNA promotes
Genistein induced apoptosis by targeting Mcl1 in human laryngeal
cancer cells. Biomed Pharmacother. 106:665–671. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Du G, Song Y, Wei L, Li L, Wang X, Xu Q,
Zhan H, Cao Y, Zheng Y and Ding D: Osthole inhibits proliferation
and induces catabolism in rat chondrocytes and cartilage tissue.
Cell Physiol Biochem. 36:2480–2493. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Newcomer CE: The evolution and adoption of
standards used by AAALAC. J Am Assoc Lab Anim Sci. 51:293–297.
2012.PubMed/NCBI
|
|
16
|
Couto M and Cates C: Laboratory Guidelines
for Animal Care. Methods Mol Biol. 1920:407–430. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou X, Zhang L, Guo X, Liu G, Wang G and
Fu S: A macaca fascicularis knee osteoarthritis model developed by
modified hulth combined with joint scratches. Med Sci Monit.
24:3393–3404. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
King TJ, Shandala T, Lee AM, Foster BK,
Chen KM, Howe PR and Xian CJ: Potential effects of phytoestrogen
genistein in modulating acute methotrexate chemotherapy-induced
osteoclastogenesis and bone damage in rats. Int J Mol Sci.
16:18293–18311. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang W, Cui G, Jin B, Wang K, Chen X, Sun
Y, Qin L and Bai W: Estradiol Valerate and Remifemin ameliorate
ovariectomy-induced decrease in a serotonin dorsal raphe-preoptic
hypothalamus pathway in rats. Ann Anat. 208:31–39. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rejtarová O, Hejna P, Soukup T and Kuchar
M: Age and sexually dimorphic changes in costal cartilages. A
preliminary microscopic study. Forensic Sci Int. 193:72–78. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li ZC, Han N, Li X, Li G, Liu YZ, Sun GX,
Wang Y, Chen GT and Li GF: Decreased expression of microRNA-130a
correlates with TNF-α in the development of osteoarthritis. Int J
Clin Exp Pathol. 8:2555–2564. 2015.PubMed/NCBI
|
|
22
|
Bergholt NL, Lysdahl H, Lind M and
Foldager CB: A standardized method of applying toluidine blue
metachromatic staining for assessment of chondrogenesis. Cartilage.
10:370–374. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang W, Ouyang H, Dass CR and Xu J:
Current research on pharmacologic and regenerative therapies for
osteoarthritis. Bone Res. 4:150402016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang XX and Cai L: Expression level of
proteoglycan, collagen and type II collagen in osteoarthritis rat
model is promoted and degradation of cartilage is prevented by
glucosamine methyl ester. Eur Rev Med Pharmacol Sci. 22:3609–3616.
2018.PubMed/NCBI
|
|
25
|
Bishnoi M, Jain A, Hurkat P and Jain SK:
Chondroitin sulphate: A focus on osteoarthritis. Glycoconj J.
33:693–705. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Barreto RB, Sadigursky D, de Rezende MU
and Hernandez AJ: Effect of hyaluronic acid on chondrocyte
apoptosis. Acta Ortop Bras. 23:90–93. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mantovani V, Maccari F and Volpi N:
Chondroitin sulfate and glucosamine as disease modifying anti-
osteoarthritis drugs (DMOADs). Curr Med Chem. 23:1139–1151. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bonnet CS, Williams AS, Gilbert SJ, Harvey
AK, Evans BA and Mason DJ: AMPA/kainate glutamate receptors
contribute to inflammation, degeneration and pain related behaviour
in inflammatory stages of arthritis. Ann Rheum Dis. 74:242–251.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
de Boer TN, van Spil WE, Huisman AM, Polak
AA, Bijlsma JW, Lafeber FP and Mastbergen SC: Serum adipokines in
osteoarthritis; comparison with controls and relationship with
local parameters of synovial inflammation and cartilage damage.
Osteoarthritis Cartilage. 20:846–853. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kobayashi M, Squires GR, Mousa A, Tanzer
M, Zukor DJ, Antoniou J, Feige U and Poole AR: Role of
interleukin-1 and tumor necrosis factor alpha in matrix degradation
of human osteoarthritic cartilage. Arthritis Rheum. 52:128–135.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Santiago B, Baleux F, Palao G,
Gutiérrez-Cañas I, Ramírez JC, Arenzana-Seisdedos F and Pablos JL:
CXCL12 is displayed by rheumatoid endothelial cells through its
basic amino-terminal motif on heparan sulfate proteoglycans.
Arthritis Res Ther. 8:R432006. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chiu YC, Yang RS, Hsieh KH, Fong YC, Way
TD, Lee TS, Wu HC, Fu WM and Tang CH: Stromal cell-derived factor-1
induces matrix metalloprotease-13 expression in human chondrocytes.
Mol Pharmacol. 72:695–703. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Dondelinger Y, Darding M, Bertrand MJ and
Walczak H: Poly-ubiquitination in TNFR1-mediated necroptosis. Cell
Mol Life Sci. 73:2165–2176. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vitale DC, Piazza C, Melilli B, Drago F
and Salomone S: Isoflavones: Estrogenic activity, biological effect
and bioavailability. Eur J Drug Metab Pharmacokinet. 38:15–25.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Basu P and Maier C: Phytoestrogens and
breast cancer: In vitro anticancer activities of isoflavones,
lignans, coumestans, stilbenes and their analogs and derivatives.
Biomed Pharmacother. 107:1648–1666. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Landete JM, Arqués J, Medina M, Gaya P, de
Las Rivas B and Muñoz R: Bioactivation of Phytoestrogens:
Intestinal Bacteria and Health. Crit Rev Food Sci Nutr.
56:1826–1843. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Nanashima N, Horie K and Maeda H:
Phytoestrogenic activity of blackcurrant anthocyanins is partially
mediated through estrogen receptor beta. Molecules. 23:E742017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chinigarzadeh A, Karim K, Muniandy S and
Salleh N: Isoflavone genistein inhibits estrogen-induced chloride
and bicarbonate secretory mechanisms in the uterus in rats. J
Biochem Mol Toxicol. 31:e218782017. View Article : Google Scholar
|
|
39
|
Schwartz N, Verma A, Bivens CB, Schwartz Z
and Boyan BD: Rapid steroid hormone actions via membrane receptors.
Biochim Biophys Acta. 1863:2289–2298. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Parikka V, Lehenkari P, Sassi ML, Halleen
J, Risteli J, Härkönen P and Väänänen HK: Estrogen reduces the
depth of resorption pits by disturbing the organic bone matrix
degradation activity of mature osteoclasts. Endocrinology.
142:5371–5378. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang ZL, Sun JY, Wang DN, Xie YH, Wang SW
and Zhao WM: Pharmacological studies of the large-scaled purified
genistein from Huaijiao (Sophora japonica-Leguminosae) on
anti-osteoporosis. Phytomedicine. 13:718–723. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ji G, Yang Q, Hao J, Guo L, Chen X, Hu J,
Leng L and Jiang Z: Anti-inflammatory effect of genistein on
non-alcoholic steatohepatitis rats induced by high fat diet and its
potential mechanisms. Int Immunopharmacol. 11:762–768. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Honndorf VS, Wiehr S, Rolle AM, Schmitt J,
Kreft L, Quintanilla-Martinez L, Kohlhofer U, Reischl G, Maurer A,
Boldt K, et al: Preclinical evaluation of the anti-tumor effects of
the natural isoflavone genistein in two xenograft mouse models
monitored by [18F]FDG, [18F]FLT, and [64Cu]NODAGA-cetuximab small
animal PET. Oncotarget. 7:28247–28261. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Xu K, Sha Y, Wang S, Chi Q, Liu Y, Wang C
and Yang L: Effects of Bakuchiol on chondrocyte proliferation via
the PI3K-Akt and ERK1/2 pathways mediated by the estrogen receptor
for promotion of the regeneration of knee articular cartilage
defects. Cell Prolif. 52:e126662019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fytili P, Giannatou E, Papanikolaou V,
Stripeli F, Karachalios T, Malizos K and Tsezou A: Association of
repeat polymorphisms in the estrogen receptors alpha, beta, and
androgen receptor genes with knee osteoarthritis. Clin Genet.
68:268–277. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Son YO and Chun JS: Estrogen-related
receptor γ is a novel catabolic regulator of osteoarthritis
pathogenesis. BMB Rep. 51:165–166. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Tian L, Su Z, Ma X, Wang F and Guo Y:
Inhibition of miR-203 ameliorates osteoarthritis cartilage
degradation in the postmenopausal rat model: Involvement of
Estrogen Receptor α. Hum Gene Ther Clin Dev. 30:160–168. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Xu X, Li X, Liang Y, Ou Y, Huang J, Xiong
J, Duan L and Wang D: Estrogen modulates cartilage and subchondral
bone remodeling in an ovariectomized rat model of postmenopausal
osteoarthritis. Med Sci Monit. 25:3146–3153. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Liang Y, Duan L, Xiong J, Zhu W, Liu Q and
Wang D, Liu W, Li Z and Wang D: E2 regulates MMP-13 via targeting
miR-140 in IL-1β-induced extracellular matrix degradation in human
chondrocytes. Arthritis Res Ther. 18:1052016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wang Q, Yan XB, Sun QQ, Hu AM, Liu HL and
Yin YW: Genetic polymorphism of the estrogen receptor alpha gene
and susceptibility to osteoarthritis: Evidence based on 15,022
subjects. Curr Med Res Opin. 31:1047–1055. 2015. View Article : Google Scholar : PubMed/NCBI
|