Introduction
Pelvic organ prolapse (POP) is a common symptom of
pelvic floor disorders that is characterized by loss of support to
the uterus, bladder and bowel, leading to their descent from their
normal anatomic position towards or through the vagina (1). Pelvic floor disorders affect a
substantial proportion of women in the USA, where the incidence
increases with age (2). In total,
≥75% women receiving routine gynecological care exhibit some levels
of prolapse (2). The risk of POP
increases with age, reaching a peak in individuals aged 60–69 years
(3). A sample study previously
revealed that women in the 60–70 years age group experience higher
levels of distress from POP compared with those in younger women
regardless of the stage of prolapse (4). Pelvic organs are connected and are
supported by the levator ani muscle complex, cardinal and
uterosacral ligament (USL), endopelvic fascia and the pelvis itself
(3). A connective tissue network
forms the footing of the pelvic structures, where fibroblastic
cells produce the extracellular matrix (ECM) proteins, including
collagen I/III and fibronectin, which are necessary for sustaining
the mechanical integrity of the normal pelvic structure (5). Any disruptions or malfunction in this
cell-based support and suspension network will lead to the
weakening of the pelvic floor, leading to prolapse (6,7). In
summary, the pathogenic mechanism of POP mainly results from the
loss of anatomical support during aging.
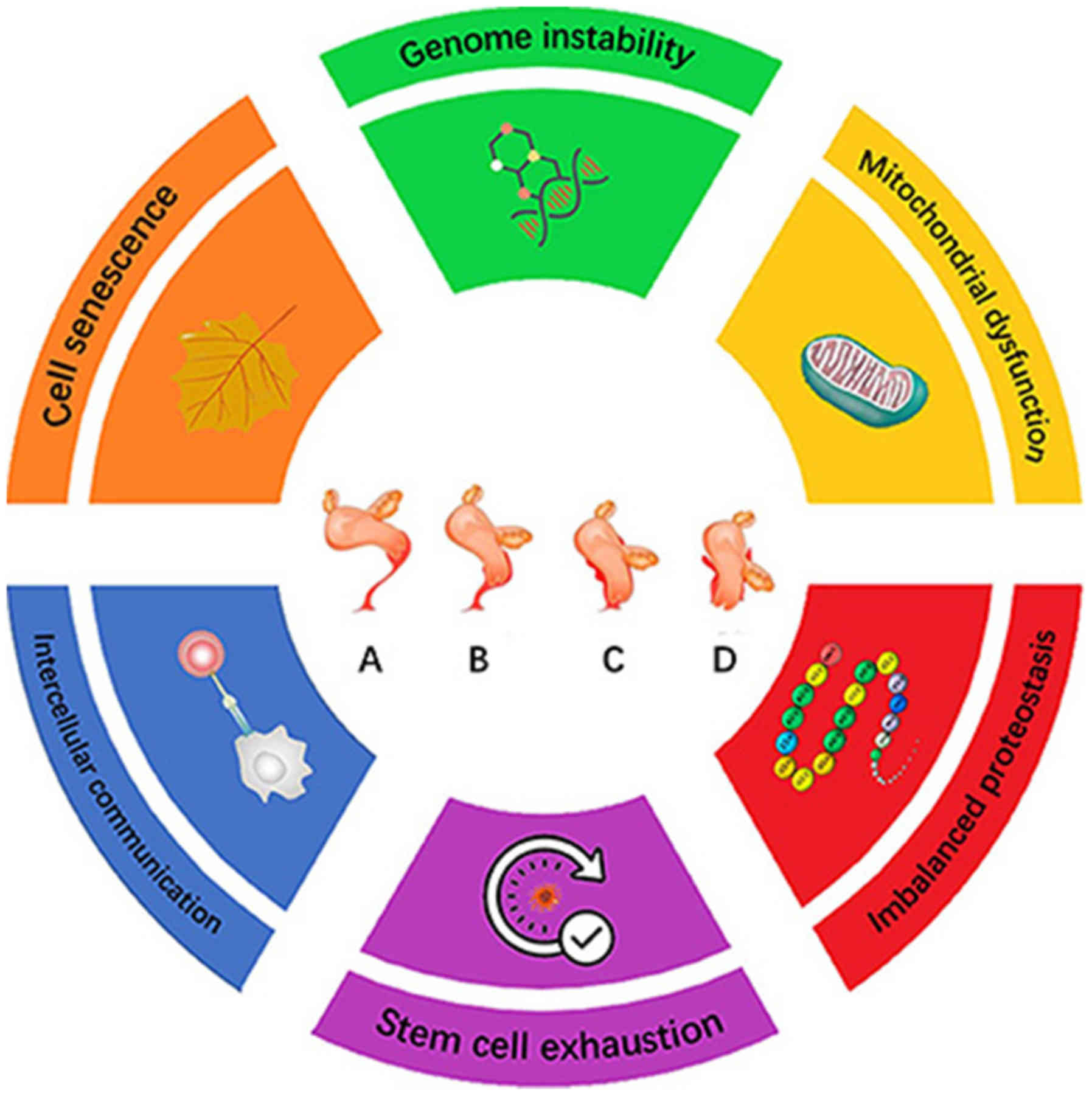
On a cellular basis, general cell senescence is the
predominant process driving aging. There are a number of phenotypes
associated with cellular aging, including genomic instability,
telomere attrition, epigenetic alterations, loss of proteostasis,
dysregulated nutrient sensing, mitochondrial dysfunction, cellular
senescence, stem cell exhaustion and altered intercellular
communication (Fig. 1) (8). In the present review, the roles of
cellular senescence in POP, in addition to the possible senescence
biomarkers reported from previous POP studies, will be discussed.
It is hoped that this information can provide novel insights into
the pathogenesis of POP.
Increases in oxidative stress causes
fibroblast cell damage in the pelvic tissue
Genetic alterations in resident cells in the pelvic
tissues serve as the main hallmark of the aging process (8). The integrity and stability of DNA are
constantly under threat from both exogenous and endogenous sources,
including those provided by oxidative stress and enzymes associated
with DNA, causing a variety of genetic lesions, including oxidative
DNA replication errors, double-strand breaks and chromosome
aberrations (9).
In terms of OS, reactive oxygen species (ROS) or OS
levels are regulated by the balance between ROS production and
elimination. Aging processes have been frequently associated with
defective anti-oxidative systems. Kim et al (10) found that the levels of OS markers
8-hydroxy-2′-deoxyguanosine and 4-hydroxy-2-nonenal were increased
in the USLs of patients with POP. OS activated a downstream
effector of AKT, initiating the PI3K/AKT signaling pathway to
promote fibroblast apoptosis in pelvic tissues and the reduction of
type I collagen (11). A previous
study reported that the antioxidant gene inducer nuclear factor
erythroid-2-related factor 2 (Nrf2) counteracted ROS production.
Mice deficient in Nrf2 displayed higher stress urinary incontinence
indices and increased levels of OS-induced apoptosis in vivo
(12). In addition, higher levels
of Nrf2 expression alleviated trauma-induced abnormalities in the
anterior vaginal wall. Lower levels of Nrf2 and excessive OS
induced the loss of fibroblasts and type I collagen in the pelvic
tissue, which in turn weakened the force of pelvic supportive
tissues (13).
Aside from aberrant redox signaling caused by
senescence, endogenous ROS can also mediate DNA damage, revealing
another mechanism associated with prolapse as a result of aging.
The accumulation of oxidative products inhibits cell proliferation
(14). Advanced glycation end
products (AGEs) are products of non-enzymatic glycation and
oxidation that frequently accumulate during aging. Several studies
have previously revealed that AGEs inhibited human vaginal
fibroblast proliferation in a number of POP cases, where they
decreased the expression of collagen whilst increasing that of
matrix metalloproteinase (MMP). These agents adjust the
intracellular conditions by regulating the balance between MMPs and
tissue inhibitors of metalloproteinases (TIMPs) in addition to gene
expression (15). Following AGE
treatment in vitro, increased expression of MMPs and reduced
levels of TIMPs resulted in the rapid degradation of type I
collagen and elastin, along with the subsequent loss of force in
the supportive tissues (16). In
senescent pelvic tissues, receptor of AGEs, p38 MAPK and NF-κB
signaling pathways partially mediated the effects of AGE treatment
on fibroblasts from patients with POP by inhibiting collagen
synthesis whilst upregulating MMP-1 expression (17). Thus, the aforementioned studies
suggested that the imbalanced senescent ROS/OS system may induce
the accumulation of AGEs and suppress Nrf2 signaling, thereby
inhibiting collagen/elastin synthesis through various molecular
pathways.
Mitochondrial dysfunction in
fibroblasts
Mitochondrial dysfunction and the decline in
respiratory chain activity have been demonstrated to be a major
characteristic of aging in numerous types of tissues, such as skin
and nervous tissues (18).
Dysfunctional mitochondria exhibit reduced respiratory capacity,
imbalanced ROS levels, an increased number of mitochondrial DNA
(mtDNA) mutations and altered mitochondrial biogenesis. mtDNA is
highly sensitive to oxidative damage due to its proximity to the
site of ROS production, and lack of standard chromatin structure or
repair mechanism (19).
Mitochondrial fusion protein-2 (Mfn2) is a conserved
dynamin-like GTPase protein localized to the mitochondrial outer
membrane and regulates mitochondrial fusion (20). Mfn2 can also localize to the
endoplasmic reticulum (ER) where it can mediate fusion between the
mitochondria and ER (21,22). Previous studies demonstrated that
the expression levels of Mfn2 were significantly higher in cultured
fibroblasts from the elderly POP group, with reduced modulation of
types I and III procollagen (23,24).
Concomitantly, cyclin-dependent kinase (CDK) 2, ERK1/2 and Raf-1
expression were also reduced, while that of the cell cycling signal
phosphoprotein 21 wild-type p53 activating fragment (p21CIP1/WAF1)
was adversely increased (23).
Senescent cells in aging pelvic tissues represent a mitochondrial
dysfunction. Via the Raf-ERK axis, cells such as mesenchymal stem
cells suffer from rapid cell cycle arrest and functionally impaired
fibroblasts secrete less extracellular matrix proteins, including
collagen. In addition, dysfunctional mitochondria in the senescent
fibroblasts of pelvic tissues overexpress Mfn2, resulting in the
loss of procollagens via the Raf-ERK axis and rapid cell cycle
arrest (19,23).
General cellular senescent phenotypes
associated with the pathogenesis of POP
Senescence is a cellular response which is
characterized by stable growth arrest and phenotypic adjustments,
such as the expression of proinflammatory factors (25–27).
Molecules such as transforming growth factor-β (TGF-β) are
deactivated during fibroblast cell senescence in pelvic tissues
(28). Senescent fibroblasts
demonstrate distinctive mRNA expression profiles that involve a
wide variety of cytokines, chemokines, proteases and growth factors
(9).
When undergoing senescence, fibroblasts experience
destruction in specific DNA sequences and genes, shortening of the
telomere and the activation of the tumor suppressor p53. The p53
pathway upregulates p21CIP1/WAF1 signaling through the NF-κB
pathway. Meanwhile, senescence-mediated stimuli CDK4/6 activates
the p16INK4A pathway, which operates in concert with p21CIP1/WAF1
in arresting cell cycle progression at the G1 phrase (29–31).
Senescent fibroblasts accumulate in the pelvic tissue, where they
exhaust the tissue of proliferative and renewable stem cells over
time, interfering with the homeostatic and regenerative capacity of
the pelvic tissues. With this reduction in active fibroblasts,
pelvic tissues are deprived of the collagen necessary to support
the pelvic organs (28).
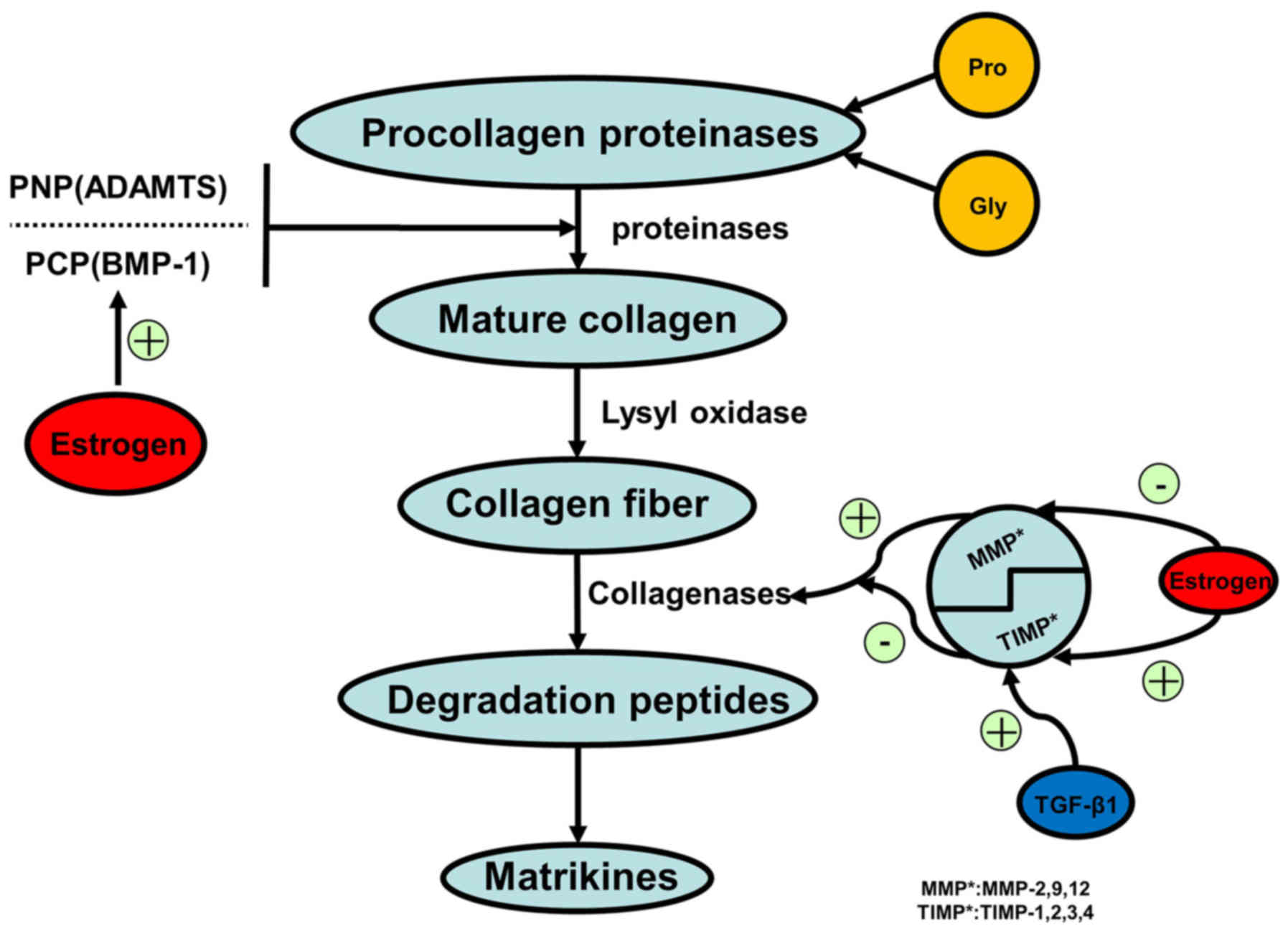
Procollagen types I and III are precursors to
collagen I/III depending on the isoform of the active MMP.
Senescent cells express reduced levels of TGF-β, which is an MMP
regulator that suppresses the activity of MMP-2/9 (28,32).
USL collagen fibers constitute 80% of the connective tissues in the
USL and are considered to be an essential component of the pelvic
supporting structure (33). Loss
of collagen I/III contributes to the weakened tensile force of the
connective tissue. Specimens obtained from elderly patients with
POP showed a significant reduction in TGF-β1 expression compared
with that in younger POP groups (34). As a result of the lower expression
levels of TGF-β associated with senescence in pelvic tissue
fibroblasts, MMPs are activated to degrade collagen I/III in the
pelvic supportive tissues. (Figs.
2 and 3).
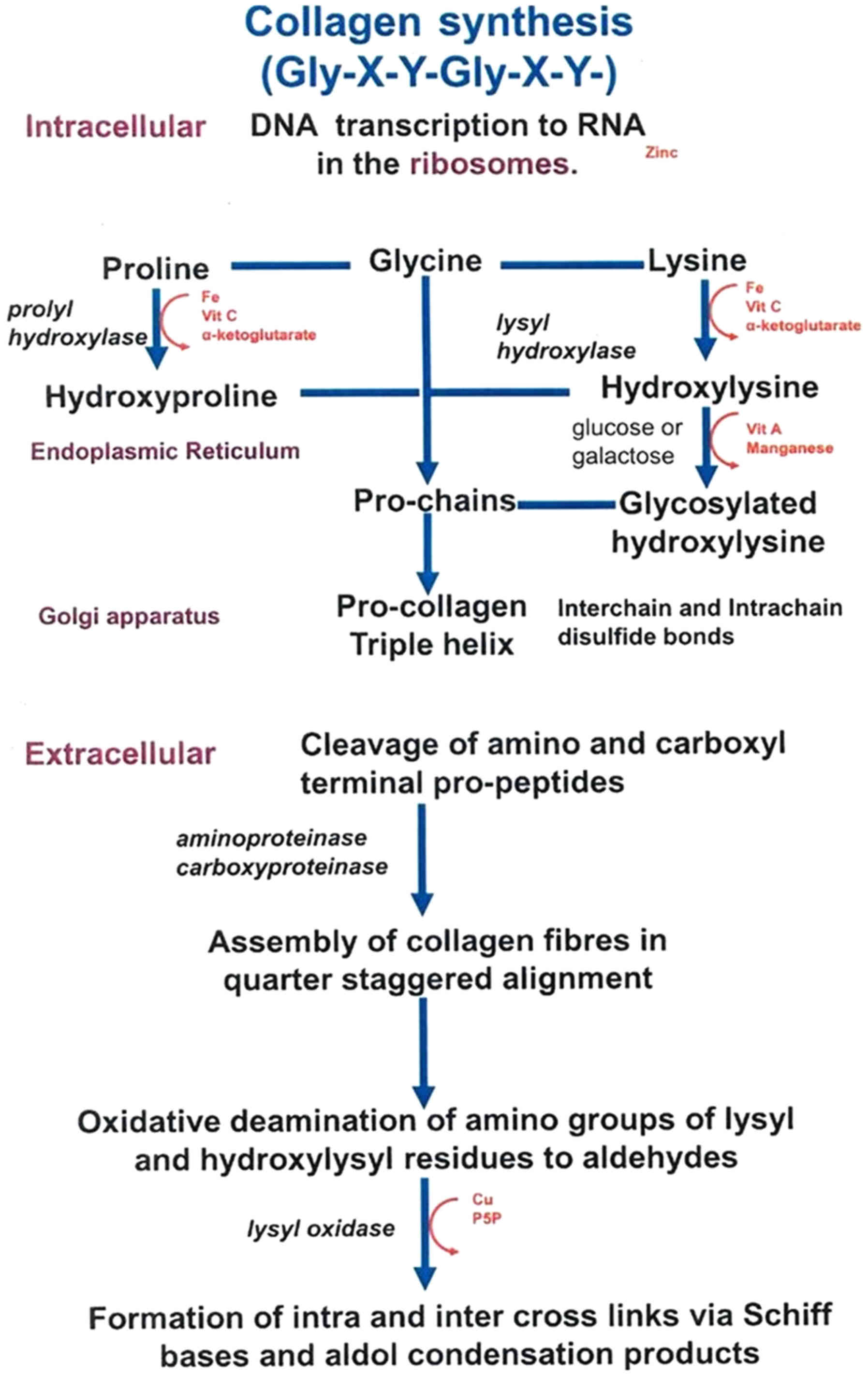
 | Figure 2.Collagen synthesis. Synthesis of
collagen in human cells, such as myofibroblasts, fibroblasts,
cardiomyocytes and inflammatory cells. Amino acids synthesize the
procollagen that transforms to mature collagen via proteinase
activity. Lysyl oxidase continuously catalyzes to form collagen
fibers. During degradation, collagenases break down collagen fibers
to peptides and matrikines. The lysyl oxidase family, and
BMP-1-ADAMTS and MMP-TIMP complexes play an essential role in
mature collagen synthesis and collagen fiber degradation processes.
MMP, matrix metalloprotein; TIMP, tissue inhibitor of
metalloproteinase; BMP-1, bone morphogenetic protein-1; ADAMTS, a
disintegrin and metalloproteinase with thrombospondin motifs;
TGF-β, transforming growth factor β. |
Additionally, a previous study with other tissue
dysfunctions such as prolapsus uteri previously demonstrated that
the levels of the NF-κB pathway-related p53 protein and mRNA are
decreased in elderly prolapse groups, suggesting a higher
proliferative capacity of prolapsus fibroblasts (35). Accompanied with the reduced
expression of p53 and p21, an intrinsic perturbation of cell cycle
progression occurs in the fibroblasts (36). The activation of senescent cell
defensive system induces the loss of fibroblasts in pelvic tissues
through apoptosis, necroptosis and autophagic cell death.
Previous studies with a number of
senescence-associated secretory phenotypes suggested that cell
senescence correlates strongly with the pathogenesis of POP
(11,16,23,28).
In dermal fibroblasts, an in vivo experiment has confirmed
the increased secretion of the cysteine-rich angiogenic inducer
protein 61, also known as cellular communication network factor 1
(CCN1) (37). CCN1 is a matrix
protein that stimulates the secretion of proinflammatory cytokines
and MMPs. They also induce fibroblast senescence to limit tissue
fibrosis during wound healing (38). A previous study also reported
elevated CCN1 expression in elderly patients with POP, with a
greater extent of fibroblast and collagen loss, suggesting that
general fibroblast senescence results in the loss of structural
support (Huang et al, unpublished data).
Imbalanced proteostasis in fibroblasts of
pelvic tissues
Impaired protein synthesis and dysregulation of
protein volume have demonstrated a close correlation with diseases
associated with aging such as POP (39). Proteins are regulated by a
sequenced mechanism to preserve stability and functionality, in a
process known as proteostasis. Proteostasis encompasses mechanisms
that preserve the stability of correctly folded proteins (40), where proteolytic systems remove
damaged proteins (41–43). The unstable proteolytic systems
during cellular senescence results in the decline of the proteasome
(44).
Imbalanced proteostasis in elderly patients with POP
presents as marked reductions in estrogen, leading to lower levels
of mature enzymes, including the lysyl oxidase family of enzymes, a
disintegrin and metalloproteinase with thrombospondin motifs and
bone morphogenetic protein-1 (BMP1) (45,46).
Estrogen treatment on post-menopausal patients with POP resulted in
the enhanced expression of collagens and elastin through regulation
of the BMP1 and MMP/TIMP complexes to improve the wound healing
process, in turn leading to the preservation of fibroblasts and
supportive collagens (47). MMP-1,
MMP-2 and MMP-3 showed a reduction in expression along with an
increase in TIMP1/4 expression after estrogen treatment in elderly
patients with POP (47).
Observations from local estrogen therapy indicates the essential
role of proteostasis during senescence within the pelvic tissue
environment, including the balance between BMP1, MMP/TIMP and
procollagen/collagen. In this regard, estrogen serves to balance
the protein levels, limit undesirable ECM degradation and to
strengthen the vaginal ECM supporting structures.
Muscle dysfunction is also one of the causes of
organ prolapse in the pelvis (6).
In a similar manner, heat-shock protein 72 (hsp72) induced the
intrinsic preservation of muscle function and postponed the
development of dystrophic muscle changes. Hsp72 is highly expressed
in patients with POP together with other molecules, where they
ensure the correct folding of newly generated proteins, especially
collagen and actin, in addition to the refolding of damaged
proteins to maintain the force of pelvic tissues (48). Dysfunctional proteins accumulate in
muscle cells as the Hsp family of proteins, autophagy-lysosome and
ubiquitin-proteasome systems decline with aging, resulting in
muscle dysfunction (49).
Obstacles and outlook
The present review presented some findings on the
underlying mechanism of fibroblast senescence on the pathogenesis
of POP. Thus far, available interventions for patients with POP
include surgical, non-surgical and physical interventions, which
are mainly focused on the physical conditions of the supportive
structures, where the ligaments are overhung or the introitus is
sealed (50). These interventions
may gradually lead to either palindromia or other adverse effects
such as postoperative dysuria and urethral injury long term
(50). Therapies that focus on
reversing the senescent process solve the issue from the root and
will not do harm to the important structures in the pelvis. The
mechanism of POP pathogenesis requires further research in
patients. In the present article, novel assessments on drug
therapy, biomaterial supplement and stem cell treatment on POP are
proposed. Estrogen serves a protective role in maintaining the
pelvic tissue microenvironment in women, where the mechanism of
estrogen affecting the synthesis of fibroblast is as
aforementioned. Estrogen shock therapy can be used as an effective
therapeutic option for POP. In a similar manner, in the biomaterial
assessment on patients with POP, TGF-β can be used in addition to
the partially absorbable material donut pessaries, using the newly
proposed biomaterial caffeic acid conjugated with peptide as an
alternative to reduce the process of senescence, whilst adjusting
the microenvironment in pelvic tissues (51).
During the process of aging, several types of stem
cells lose their quiescence, leading to excessive proliferation and
their regenerative potential (52). Notable examples include age-related
hematopoietic stem cell-induced anemia and the aging mesenchymal
stem cell (MSC)-induced reduction in fracture repair (8). In terms of stem cell therapy, MSCs
show a multi-directive differentiation ability, whose loss of
regenerative capacity lead to the impaired ability to facilitate
tissue repair, thereby increasing morbidity (53). In terms of aging, taking advantage
of the differentiation ability of bone marrow-derived MSCs and
their secretory factors, which are beneficial for tissue repair and
elastin regeneration (54,55) is now being considered as another
alternative for vaginal prolapse to reactivate the stem cells
(56). MSCs are not only able to
transdifferentiate into various cell types present in the pelvic
tissues and maintain collagen secretion with neovascularization but
they can also deliver bioactive molecules to sites of injury to
promote fibroblast proliferation and collagen synthesis (57). Although there remains a lack of
sufficient studies on the possibility of senescent therapy in POP,
a recent review shed light on a potential stem cell therapy
strategy. The stem cells can be combined with mesh, which can be
inserted into the bladder neck for POP treatment by inducing
vascular endothelial growth factor, platelet-derived growth
factor-β and TGF-β, which have been reported to delay the
senescence process and improve tissue repairs (51).
Conclusion
Knowledge on the pathogenesis of POP remains
multifactorial, which can be mainly attributed to the general aging
of the individual and overall senescence of the fibroblasts in
pelvic tissues. This causes the exhaustion or apoptosis of the
fibroblasts in the pelvic floor, reducing its supportive ability
and integrity. The main five hallmarks of POP associated with aging
were reviewed primarily based on a previous review on the hallmarks
of general aging. Genomic instability and imbalanced proteostasis
are likely to be the primary triggers of aging-related prolapse.
Mitochondrial dysfunction in the progression of senescence may
consequentially become the integrated cause of POP. Cell
senescence, one facet of the aging process, is a collective
phenotype caused by a complex network of signaling pathways.
Mechanisms causing fibroblast senescence, including gene
instability, mitochondrial dysfunction and altered
senescence-symbolized phenotype, provide a basis for
senescence-related POP pathological mechanisms (Table I) (16,17,39,42,43,49,50,58–77).
Nevertheless, with an improved understanding of the effect of
senescence on POP, currently available clinical treatments could
potentially be applied in conjunction with estrogen treatment and
stem cell-based material with the aim of reversing fibroblast
senescence in patients with POP in the future.
 | Table I.Evidence of the possible genes or
biomarkers in patients with POP, compared with normal dermal
fibroblast. |
Table I.
Evidence of the possible genes or
biomarkers in patients with POP, compared with normal dermal
fibroblast.
| Types of aging | Evidence in
vitro culture | Evidence ex
vivo from patients with POP | Evidence in
vivo from the dermal fibroblasts in the elderly |
|---|
| Genome
instability |
| HOXA11,
HOXA13, ESR1 and ESR2 | Y (49) | Y (62) | ND |
| γH2AX
foci | Y (50) | Y (17) | Y (16) |
| Mutant
NER | Y (59) | ND | ND |
|
Replicative activity | Y (60) | ND | ND |
|
Telomere shortening | Y (61,62) | Y (63) | ND |
|
Telomere damage | Y (50) | ND | Y (16) |
| Mitochondrial
dysfunction |
| mtDNA
mutations | Y (64,65) | ND | ND |
| Altered
fusion | ND | Y (67) | ND |
|
Increasing mitogenesis | Y (69,70) | ND |
|
| Cell
senescence |
|
p16INK4A | Y (71) | ND | Y (66,67) |
|
p21 | Y (72,73) | Y (78) | ND |
|
p53 | ND | Y (78) | ND |
|
SASP | Y (74) | Y (74) | ND |
|
SAHF | Y (75,76) | ND | Y (68) |
| γH2AX
foci | Y (50) | ND | Y (16) |
| Imbalanced
proteostasis |
|
Chaperon dysfunction | ND | Y (78) | ND |
|
Proteasome activity | Y (42,77) | ND | ND |
|
Decreased autophagy | ND | ND | ND |
|
Increased MMP secretion | Y (43) | Y (39) | Y (39) |
| Stem cell
exhaustion |
|
Regeneration potential | ND | ND | ND |
Acknowledgements
Not applicable.
Funding
The present study was supported by The Foundation of
Sichuan Provincial Science and Technology Program (grant nos.
2019YFH0147 and 2019YFH0158), Chengdu technological innovation
research and development project (grant no. 2018-YF05-00195-SN),
West China Second University Hospital Xinya fund (grant no. kx111)
and 1.3.5 project for disciplines of excellence, West China
Hospital, Sichuan University (grant no. ZYJC18016).
Availability of data and materials
The datasets generated and/or analyzed during the
current study are available in the NCBI repository, https://www.ncbi.nlm.nih.gov.
Authors' contributions
LH, ZZ and JW conceived and designed the review,
tables and figures, and drafted the manuscript. YM, JW and WL
researched the literature and critically revised the article for
important intellectual content. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Iglesia CB and Smithling KR: Pelvic organ
prolapse. Am Fam Physician. 96:179–185. 2017.PubMed/NCBI
|
|
2
|
Nygaard I, Barber MD, Burgio KL, Kenton K,
Meikle S, Schaffer J, Spino C, Whitehead WE, Wu J and Brody DJ;
Pelvic floor disorders network, : Prevalence of symptomatic pelvic
floor disorders in US women. JAMA. 300:1311–1316. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jelovsek JE, Maher C and Barber MD: Pelvic
organ prolapse. Lancet. 369:1027–1038. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kinman CL, Lemieux CA, Agrawal A, Gaskins
JT, Meriwether KV and Francis SL: The relationship between age and
pelvic organ prolapse bother. Int Urogynecol J Pelvic Floor
Dysfunct. 28:751–755. 2017. View Article : Google Scholar
|
|
5
|
Sun B, Zhou L, Wen Y, Wang C, Baer TM,
Pera RR and Chen B: Proliferative behavior of vaginal fibroblasts
from women with pelvic organ prolapse. Eur J Obstet Gynecol Reprod
Biol. 183:1–4. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Richardson AC, Lyon JB and Williams NL: A
new look at pelvic relaxation. Am J Obstet Gynecol. 126:568–573.
1976. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jackson SR, Avery NC, Tarlton JF, Eckford
SD, Abrams P and Bailey AJ: Changes in metabolism of collagen in
genitourinary prolapse. Lancet. 347:1658–1661. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
López-Otín C, Blasco MA, Partridge L,
Serrano M and Kroemer G: The hallmarks of aging. Cell.
153:1194–1217. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tigges J, Krutmann J, Fritsche E,
Haendeler J, Schaal H, Fischer JW, Kalfalah F, Reinke H,
Reifenberger G, Stühler K, et al: The hallmarks of fibroblast
ageing. Mech Ageing Dev. 138:26–44. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim EJ, Chung N, Park SH, Lee KH, Kim SW,
Kim JY, Bai SW and Jeon MJ: Involvement of oxidative stress and
mitochondrial apoptosis in the pathogenesis of pelvic organ
prolapse. J Urol. 189:588–594. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li BS, Guo WJ, Hong L, Liu YD, Liu C, Hong
SS, Wu DB and Min J: Role of mechanical strain-activated PI3K/Akt
signaling pathway in pelvic organ prolapse. Mol Med Rep.
14:243–253. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tian X, Wang F, Luo Y, Ma S, Zhang N, Sun
Y, You C, Tang G, Li S, Gong Y, et al: Protective role of nuclear
factor-erythroid 2-related factor 2 against radiation-induced lung
injury and inflammation. Front Oncol. 8:5422018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang W, Sun Z, Yang B and Wang Q:
Nrf2-knockout protects from intestinal injuries in C57BL/6J mice
following abdominal irradiation with γ rays. Int J Mol Sci.
18:E16562017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Møller P, Løhr M, Folkmann JK, Mikkelsen L
and Loft S: Aging and oxidatively damaged nuclear DNA in animal
organs. Free Radic Biol Med. 48:1275–1285. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gkogkolou P and Böhm M: Advanced glycation
end products: key players in skin aging? Dermatoendocrinol.
4:259–270. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Willett TL, Pasquale J and Grynpas MD:
Collagen modifications in postmenopausal osteoporosis: Advanced
glycation end products may affect bone volume, structure and
quality. Curr Osteoporos Rep. 12:329–337. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen YS, Wang XJ, Feng W and Hua KQ:
Advanced glycation end products decrease collagen I levels in
fibroblasts from the vaginal wall of patients with POP via the
RAGE, MAPK and NF-κB pathways. Int J Mol Med. 40:987–998. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nunnari J and Suomalainen A: Mitochondria:
In sickness and in health. Cell. 148:1145–1159. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kim SJ, Cheresh P, Jablonski RP, Williams
DB and Kamp DW: The role of mitochondrial DNA in mediating alveolar
epithelial cell apoptosis and pulmonary fibrosis. Int J Mol Sci.
16:21486–21519. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen H, Detmer SA, Ewald AJ, Griffin EE,
Fraser SE and Chan DC: Mitofusins Mfn1 and Mfn2 coordinately
regulate mitochondrial fusion and are essential for embryonic
development. J Cell Biol. 160:189–200. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
de Brito OM and Scorrano L: Mitofusin 2
tethers endoplasmic reticulum to mitochondria. Nature. 456:605–610.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sebastián D, Hernández-Alvarez MI, Segalés
J, Sorianello E, Muñoz JP, Sala D, Waget A, Liesa M, Paz JC,
Gopalacharyulu P, et al: Mitofusin 2 (Mfn2) links mitochondrial and
endoplasmic reticulum function with insulin signaling and is
essential for normal glucose homeostasis. Proc Natl Acad Sci USA.
109:5523–5528. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang X, Wang X, Zhou Y, Peng C, Chen H and
Lu Y: Mitofusin2 regulates the proliferation and function of
fibroblasts: The possible mechanisms underlying pelvic organ
prolapse development. Mol Med Rep. 20:2859–2866. 2019.PubMed/NCBI
|
|
24
|
Lu Y, Chen HY, Wang XQ and Wang JX:
Correlations between Mitofusin 2 expression in fibroblasts and
pelvic organ prolapse: An in vitro study. Chin Med J (Engl).
130:2951–2959. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
McHugh D and Gil J: Senescence and aging:
Causes, consequences, and therapeutic avenues. J Cell Biol.
217:65–77. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Burton DG and Krizhanovsky V:
Physiological and pathological consequences of cellular senescence.
Cell Mol Life Sci. 71:4373–4386. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kirkland JL and Tchkonia T: Cellular
senescence: A translational perspective. EBioMedicine. 21:21–28.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen B and Yeh J: Alterations in
connective tissue metabolism in stress incontinence and prolapse. J
Urol. 186:1768–1772. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Alcorta DA, Xiong Y, Phelps D, Hannon G,
Beach D and Barrett JC: Involvement of the cyclin-dependent kinase
inhibitor p16 (INK4a) in replicative senescence of normal human
fibroblasts. Proc Natl Acad Sci USA. 93:13742–13747. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Takahashi A, Ohtani N, Yamakoshi K, Iida
S, Tahara H, Nakayama K, Nakayama KI, Ide T, Saya H and Hara E:
Mitogenic signalling and the p16INK4a-Rb pathway cooperate to
enforce irreversible cellular senescence. Nat Cell Biol.
8:1291–1297. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Beauséjour CM, Krtolica A, Galimi F,
Narita M, Lowe SW, Yaswen P and Campisi J: Reversal of human
cellular senescence: Roles of the p53 and p16 pathways. EMBO J.
22:4212–4222. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sampson N, Berger P and Zenzmaier C: Redox
signaling as a therapeutic target to inhibit myofibroblast
activation in degenerative fibrotic disease. BioMed Res Int.
2014:1317372014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cole EE, Leu PB, Gomelsky A, Revelo P,
Shappell H, Scarpero HM and Dmochowski RR: Histopathological
evaluation of the uterosacral ligament: Is this a dependable
structure for pelvic reconstruction? BJU Int. 97:345–348. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu C, Wang Y, Li BS, Yang Q, Tang JM, Min
J, Hong SS, Guo WJ and Hong L: Role of transforming growth factor β
1 in the pathogenesis of pelvic organ prolapse: A potential
therapeutic target. Int J Mol Med. 40:347–356. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yamamoto K, Yamamoto M, Akazawa K, Tajima
S, Wakimoto H and Aoyagi M: Decrease in elastin gene expression and
protein synthesis in fibroblasts derived from cardinal ligaments of
patients with prolapsus uteri. Cell Biol Int. 21:605–611. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yamamoto M, Aoyagi M, Akazawa K, Tajima S
and Yamamoto K: Decrease in p53 protein in cultured cardinal
ligament fibroblasts from patients with prolapsus uteri. Cell Biol
Int. 22:31–40. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Quan T, Qin Z, Robichaud P, Voorhees JJ
and Fisher GJ: CCN1 contributes to skin connective tissue aging by
inducing age-associated secretory phenotype in human skin dermal
fibroblasts. J Cell Commun Signal. 5:201–207. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jun JI and Lau LF: The matricellular
protein CCN1 induces fibroblast senescence and restricts fibrosis
in cutaneous wound healing. Nat Cell Biol. 12:676–685. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Powers ET, Morimoto RI, Dillin A, Kelly JW
and Balch WE: Biological and chemical approaches to diseases of
proteostasis deficiency. Annu Rev Biochem. 78:959–991. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hartl FU, Bracher A and Hayer-Hartl M:
Molecular chaperones in protein folding and proteostasis. Nature.
475:324–332. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Brennan M, Bhatti H, Nerusu KC,
Bhagavathula N, Kang S, Fisher GJ, Varani J and Voorhees JJ: Matrix
metalloproteinase-1 is the major collagenolytic enzyme responsible
for collagen damage in UV-irradiated human skin. Photochem
Photobiol. 78:43–48. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Cuervo AM, Bergamini E, Brunk UT, Dröge W,
Ffrench M and Terman A: Autophagy and aging: The importance of
maintaining ‘clean’ cells. Autophagy. 1:131–140. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mizushima N, Levine B, Cuervo AM and
Klionsky DJ: Autophagy fights disease through cellular
self-digestion. Nature. 451:1069–1075. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bulteau AL, Moreau M, Nizard C and Friguet
B: Proteasome and photoaging: The effects of UV irradiation. Ann N
Y Acad Sci. 1100:280–290. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Pereira L, D'Alessio M, Ramirez F, Lynch
JR, Sykes B, Pangilinan T and Bonadio J: Genomic organization of
the sequence coding for fibrillin, the defective gene product in
Marfan syndrome. Hum Mol Genet. 2:17621993. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhang H, Hu W and Ramirez F: Developmental
expression of fibrillin genes suggests heterogeneity of
extracellular microfibrils. J Cell Biol. 129:1165–1176. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Tyagi T, Alarab M, Leong Y, Lye S and
Shynlova O: Local oestrogen therapy modulates extracellular matrix
and immune response in the vaginal tissue of post-menopausal women
with severe pelvic organ prolapse. J Cell Mol Med. 23:2907–2919.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Calamini B and Morimoto RI: Protein
homeostasis as a therapeutic target for diseases of protein
conformation. Curr Top Med Chem. 12:2623–2640. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Tomaru U, Takahashi S, Ishizu A, Miyatake
Y, Gohda A, Suzuki S, Ono A, Ohara J, Baba T, Murata S, et al:
Decreased proteasomal activity causes age-related phenotypes and
promotes the development of metabolic abnormalities. Am J Pathol.
180:963–972. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Coolen AWM, Troost S, Mol BWJ, Roovers
JPWR and Bongers MY: Primary treatment of pelvic organ prolapse:
Pessary use versus prolapse surgery. Int Urogynecol J Pelvic Floor
Dysfunct. 29:99–107. 2018. View Article : Google Scholar
|
|
51
|
Cheng J, Zhao ZW, Wen JR, Wang L, Huang
LW, Yang YL, Zhao FN, Xiao JY, Fang F, Wu J, et al: Status,
challenges, and future prospects of stem cell therapy in pelvic
floor disorders. World J Clin Cases. 8:1400–1413. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Shaw AC, Joshi S, Greenwood H, Panda A and
Lord JM: Aging of the innate immune system. Curr Opin Immunol.
22:507–513. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kirkwood TB: Understanding the odd science
of aging. Cell. 120:437–447. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Jin M, Wu Y, Wang J, Ye W, Wang L, Yin P,
Liu W, Pan C and Hua X: MicroRNA-29 facilitates transplantation of
bone marrow-derived mesenchymal stem cells to alleviate pelvic
floor dysfunction by repressing elastin. Stem Cell Res Ther.
7:1672016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Jin M, Chen Y, Zhou Y, Mei Y, Liu W, Pan C
and Hua X: Transplantation of bone marrow-derived mesenchymal stem
cells expressing elastin alleviates pelvic floor dysfunction. Stem
Cell Res Ther. 7:512016. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Ulrich D, Edwards SL, Su K, Tan KS, White
JF, Ramshaw JA, Lo C, Rosamilia A, Werkmeister JA and Gargett CE:
Human endometrial mesenchymal stem cells modulate the tissue
response and mechanical behavior of polyamide mesh implants for
pelvic organ prolapse repair. Tissue Eng Part A. 20:785–798.
2014.PubMed/NCBI
|
|
57
|
El Agha E, Kramann R, Schneider RK, Li X,
Seeger W, Humphreys BD and Bellusci S: Mesenchymal stem cells in
fibrotic disease. Cell Stem Cell. 21:166–177. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Ohshima S: Centrosome aberrations
associated with cellular senescence and p53 localization at
supernumerary centrosomes. Oxid Med Cell Longev. 2012:2175942012.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Fumagalli M, Rossiello F, Clerici M,
Barozzi S, Cittaro D, Kaplunov JM, Bucci G, Dobreva M, Matti V,
Beausejour CM, et al: Telomeric DNA damage is irreparable and
causes persistent DNA-damage-response activation. Nat Cell Biol.
14:355–365. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Garm C, Moreno-Villanueva M, Bürkle A,
Petersen I, Bohr VA, Christensen K and Stevnsner T: Age and gender
effects on DNA strand break repair in peripheral blood mononuclear
cells. Aging Cell. 12:58–66. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
De Cecco M, Criscione SW, Peckham EJ,
Hillenmeyer S, Hamm EA, Manivannan J, Peterson AL, Kreiling JA,
Neretti N and Sedivy JM: Genomes of replicatively senescent cells
undergo global epigenetic changes leading to gene silencing and
activation of transposable elements. Aging Cell. 12:247–256. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Harley CB, Futcher AB and Greider CW:
Telomeres shorten during ageing of human fibroblasts. Nature.
345:458–460. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Sedelnikova OA, Horikawa I, Zimonjic DB,
Popescu NC, Bonner WM and Barrett JC: Senescing human cells and
ageing mice accumulate DNA lesions with unrepairable double-strand
breaks. Nat Cell Biol. 6:168–170. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Hoeijmakers JH: DNA damage, aging, and
cancer. N Engl J Med. 361:1475–1485. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Herbig U, Ferreira M, Condel L, Carey D
and Sedivy JM: Cellular senescence in aging primates. Science.
311:12572006. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Krutmann J and Schroeder P: Role of
mitochondria in photoaging of human skin: The defective powerhouse
model. J Investig Dermatol Symp Proc. 14:44–49. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ressler S, Bartkova J, Niederegger H,
Bartek J, Scharffetter-Kochanek K, Jansen-Dürr P and Wlaschek M:
p16INK4A is a robust in vivo biomarker of cellular aging in human
skin. Aging Cell. 5:379–389. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Jeyapalan JC, Ferreira M, Sedivy JM and
Herbig U: Accumulation of senescent cells in mitotic tissue of
aging primates. Mech Ageing Dev. 128:36–44. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Kreiling JA, Tamamori-Adachi M, Sexton AN,
Jeyapalan JC, Munoz-Najar U, Peterson AL, Manivannan J, Rogers ES,
Pchelintsev NA, Adams PD, et al: Age-associated increase in
heterochromatic marks in murine and primate tissues. Aging Cell.
10:292–304. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Hayflick L: The biology of human aging.
Adv Pathobiol. 7:80–99. 1980.PubMed/NCBI
|
|
71
|
Lee HC, Yin PH, Chi CW and Wei YH:
Increase in mitochondrial mass in human fibroblasts under oxidative
stress and during replicative cell senescence. J Biomed Sci.
9:517–526. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
d'Adda di Fagagna F, Reaper PM,
Clay-Farrace L, Fiegler H, Carr P, Von Zglinicki T, Saretzki G,
Carter NP and Jackson SP: A DNA damage checkpoint response in
telomere-initiated senescence. Nature. 426:194–198. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Passos JF, Nelson G, Wang C, Richter T,
Simillion C, Proctor CJ, Miwa S, Olijslagers S, Hallinan J, Wipat
A, et al: Feedback between p21 and reactive oxygen production is
necessary for cell senescence. Mol Syst Biol. 6:3472010. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Herbig U, Jobling WA, Chen BP, Chen DJ and
Sedivy JM: Telomere shortening triggers senescence of human cells
through a pathway involving ATM, p53, and p21(CIP1), but not
p16(INK4a). Mol Cell. 14:501–513. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Coppé JP, Desprez PY, Krtolica A and
Campisi J: The senescence-associated secretory phenotype: The dark
side of tumor suppression. Annu Rev Pathol. 5:99–118. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Narita M, Nũnez S, Heard E, Narita M, Lin
AW, Hearn SA, Spector DL, Hannon GJ and Lowe SW: Rb-mediated
heterochromatin formation and silencing of E2F target genes during
cellular senescence. Cell. 113:703–716. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Gabriel B, Denschlag D, Göbel H, Fittkow
C, Werner M, Gitsch G and Watermann D: Uterosacral ligament in
postmenopausal women with or without pelvic organ prolapse. Int
Urogynecol J Pelvic Floor Dysfunct. 16:475–479. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Bump RC, Mattiasson A, Bø K, Brubaker LP,
DeLancey JO, Klarskov P, Shull BL and Smith AR: The standardization
of terminology of female pelvic organ prolapse and pelvic floor
dysfunction. Am J Obstet Gynecol. 175:10–17. 1996. View Article : Google Scholar : PubMed/NCBI
|