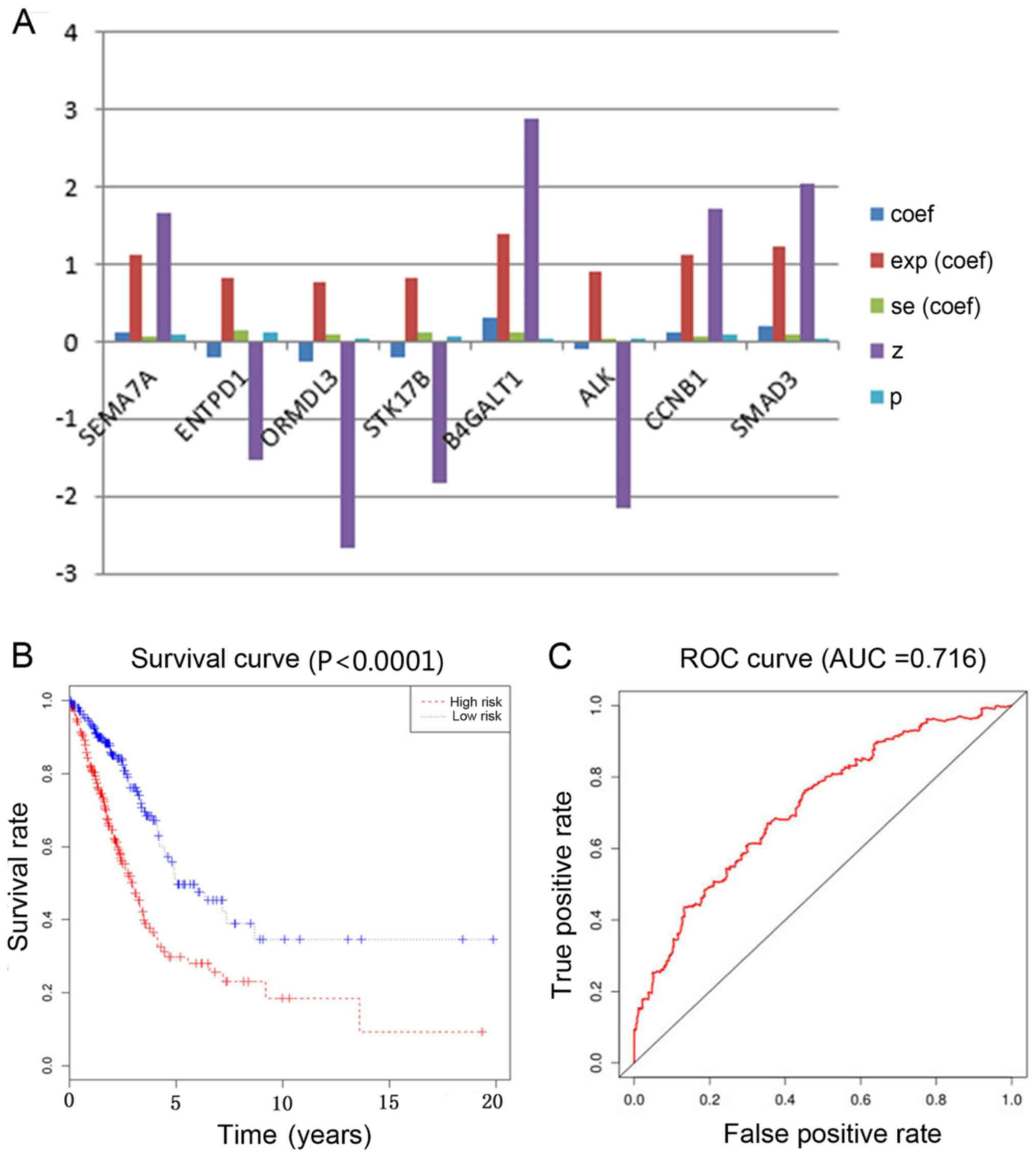
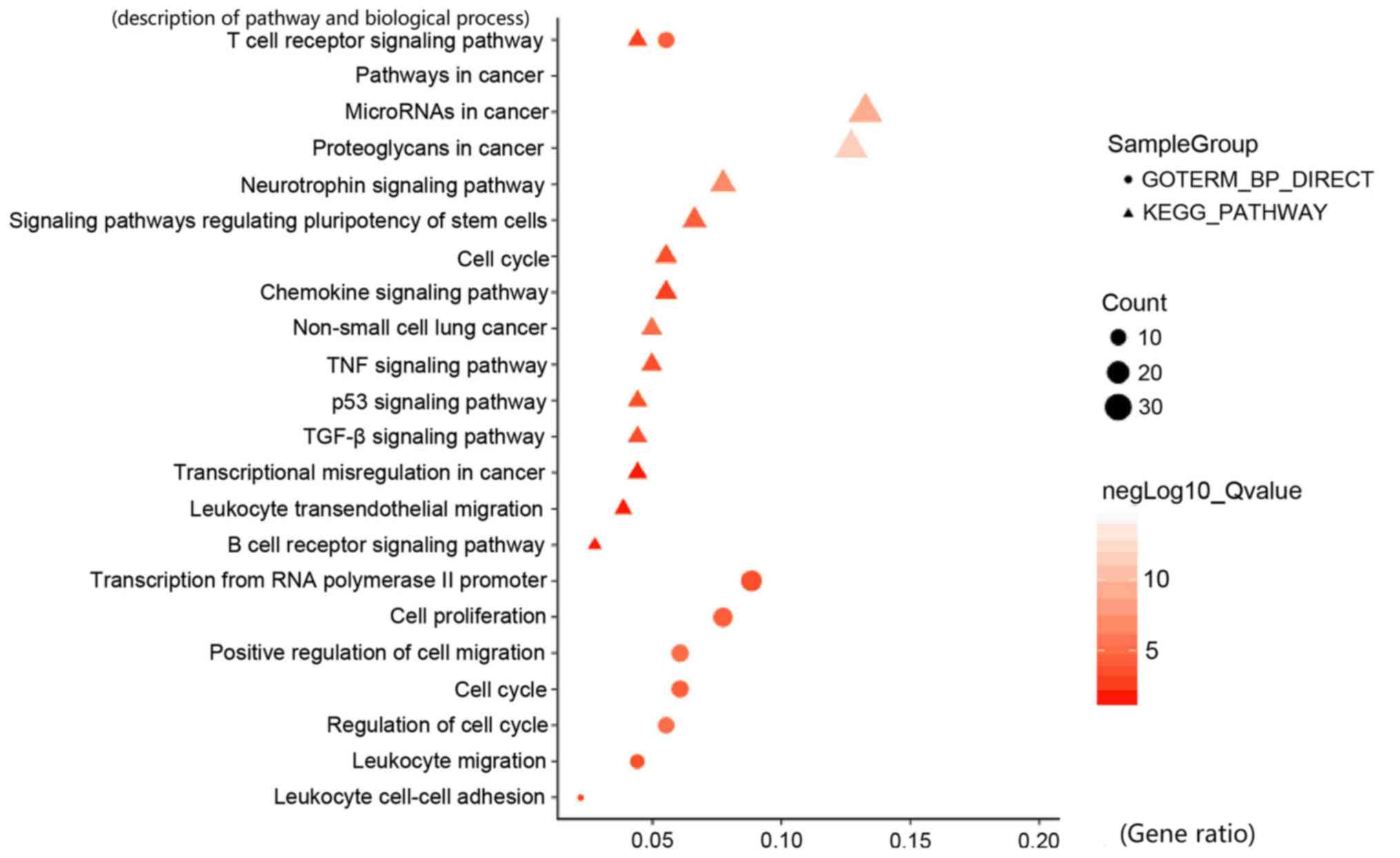
|
1
|
Sharma P, Wagner K, Wolchok JD and Allison
JP: Novel cancer immunotherapy agents with survival benefit: Recent
successes and next steps. Nat Rev Cancer. 11:3173–812. 2011.
View Article : Google Scholar
|
|
2
|
Pardoll DM: The blockade of immune
checkpoints in cancer immunotherapy. Nat Rev Cancer. 12:252–264.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Carter BW, Halpenny DF, Ginsberg MS,
Papadimitrakopoulou VA and de Groot PM: Immunotherapy in Non-small
cell lung cancer treatment: Current status and the role of imaging.
J Thorac Imaging. 32:300–312. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Di Giacomo AM, Calabrò L, Danielli R,
Fonsatti E, Bertocci E, Pesce I, Fazio C, Cutaia O, Giannarelli D,
Miracco C, et al: Long-term survival and immunological parameters
in metastatic melanoma; patients who responded to ipilimumab 10
mg/kg within an expanded access; programme. Cancer Immunol
Immunother. 62:1021–1028. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Prieto PA, Yang JC, Sherry RM, Hughes MS,
Kammula US, White DE, Levy CL, Rosenberg SA and Phan GQ: CTLA-4
blockade with ipilimumab: Long-term follow-up of 177 patients with
metastatic melanoma. Clin Cancer Res. 18:2039–2047. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu XS and Mardis ER: Applications of
immunogenomics to cancer. Cell. 168:600–612. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yang IV, Konigsberg I, Macphail K, Li L,
Davidson EJ, Mroz PM, Hamzeh N, Gillespie M, Silveira LJ, Fingerlin
TE and Maier LA: DNA methylation changes in lung immune cells are
associated with granulomatous lung disease. Am J Respir Cell Mol
Biol. 60:96–105. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gudrun B, Rowley MJ, Kuciński J, Zhu Y,
Amies I and Wierzbicki AT: RNA-directed DNA methylation requires
stepwise binding of silencing factors to long non-coding RNA. Plant
J Cell Mol Biol. 79:181–191. 2014. View Article : Google Scholar
|
|
9
|
Hui Z, Shangwei N, Xiang L, Yuyun L, Wei W
and Xia L: A novel reannotation strategy for dissecting DNA
methylation patterns of human long intergenic non-coding RNAs in
cancers. Nucleic Acids Res. 42:82582014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bohne F, Langer D, Martiné U, Eider CS,
Cencic R, Begemann M, Elbracht M, Bülow L, Eggermann T, Zechner U,
et al: Kaiso mediates human ICR1 methylation maintenance and H19
transcriptional fine regulation. Clin Epigenetics. 8:472016.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vennin C, Spruyt N, Robin YM, Chassat T,
Le Bourhis X and Adriaenssens E: The long non-coding RNA 91H
increases aggressive phenotype of breast cancer cells and
up-regulates H19/IGF2 expression through epigenetic modifications.
Cancer Lett. 385:198–206. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yu F, Chen B, Dong P and Zheng J: HOTAIR
epigenetically modulates PTEN expression via MicroRNA-29b: A novel
mechanism in regulation of liver fibrosis. Mol Ther. 25:205–217.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gregory MC and Loeb DM: Hypoxia-sensitive
epigenetic regulation of an antisense-oriented lncRNA controls WT1
expression in myeloid leukemia cells. PLoS One. 10:e01198372015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tianyi G, Bangshun H, Yuqin P, Yeqiong X,
Rui L, Qiwen D, Huilin S and Shukui W: Long non-coding RNA 91H
contributes to the occurrence and progression of esophageal
squamous cell carcinoma by inhibiting IGF2 expression. Chinese J
Clin Lab Sci. 54:359–367. 2015.
|
|
15
|
Ruscio AD, Ebralidze AK, Benoukraf T,
Amabile G, Goff LA, Terragni J, Figueroa ME, Pontes LLDF,
Alberich-Jorda M, Zhang P, et al: DNMT1-interacting RNAs block
gene-specific DNA methylation. Nature. 503:371–376. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Salmena L, Poliseno L, Tay Y, Kats L and
Pandolfi PP: ceRNA hypothesis: The Rosetta stone of a hidden RNA
language? Cell. 146:353–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li B and Dewey CN: RSEM: Accurate
transcript quantification from RNA-Seq data with or without a
reference genome. BMC Bioinformatics. 12:323. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhi H, Li X, Wang P, Gao Y, Gao B, Zhou D,
Zhang Y, Guo M, Yue M, Shen W, et al: Lnc2Meth: A manually curated
database of regulatory relationships between long non-coding RNAs
and DNA methylation associated with human disease. Nucleic Acids
Res. 46:D133–D138. 2017. View Article : Google Scholar
|
|
19
|
Love MI, Wolfgang H and Simon A: Moderated
estimation of fold change and dispersion for RNA-seq data with
DESeq2. Genome Biol. 15:5502014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Li B, Severson E, Pignon JC, Zhao H, Li T,
Novak J, Peng J, Hui S, Aster JC, Rodig S, et al: Comprehensive
analyses of tumor immunity: Implications for cancer immunotherapy.
Genome Biol. 17:1742016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li B and Li JZ: A general framework for
analyzing tumor subclonality using SNP array and DNA sequencing
data. Genome Biol. 15:4732014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chao C, Kay G, Judith B, Dandan Z, Elliot
G, Li J and Chunyu L: Removing batch effects in analysis of
expression microarray data: An evaluation of six batch adjustment
methods. PLoS One. 6:e172382011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ashwini J, Marks DS and Erik L: miRcode: A
map of putative microRNA target sites in the long non-coding
transcriptome. Bioinformatics. 28:2062–2063. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42((Database Issue)): D92–D97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Agarwal V, Bell GW, Nam JW and Bartel DP:
Predicting effective microRNA target sites in mammalian mRNAs.
Elife. 4:e050052015. View Article : Google Scholar
|
|
26
|
Chou CH, Shrestha S, Yang CD, Chang NW,
Lin YL, Liao KW, Huang WC, Sun TH, Tu SJ, Lee WH, et al: miRTarBase
update 2018: A resource for experimentally validated
microRNA-target interactions. Nucleic Acids Res. 46:D296–D302.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wong N and Wang X: miRDB: An online
resource for microRNA target prediction and functional annotations.
Nucleic Acids Res. 43((Database Issue)): D146–D152. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gill RD: Censoring and stochastic
integrals. Statistica Neerlandica. 34:124. 1980. View Article : Google Scholar
|
|
30
|
Fox J: Cox proportional-hazards regression
for survival data. See Also. 371–372. 2012.
|
|
31
|
Huang DW, Sherman BT and Lempicki RA:
Bioinformatics enrichment tools: Paths toward the comprehensive
functional analysis of large gene lists. Nucleic Acids Res.
37:1–13. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang W, Cui Q, Qu W, Ding X, Jiang D and
Liu H: TRIM58/cg26157385 methylation is associated with eight
prognostic genes in lung squamous cell carcinoma. Oncol Rep.
40:206–216. 2018.PubMed/NCBI
|
|
33
|
Nakayama K, Rahman MT, Rahman M, Nakamura
K, Ishikawa M, Katagiri H, Sato E, Ishibashi T, Iida K, Ishikawa N
and Kyo S: CCNE1amplification is associated with aggressive
potential in endometrioid endometrial carcinomas. Int J Oncol.
48:506–516. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yao Y, Luo J, Sun Q, Xu T, Sun S, Chen M,
Lin X, Qian Q, Zhang Y, Cao L, et al: HOXC13 promotes proliferation
of lung adenocarcinoma via modulation of CCND1 and CCNE1. Am J
Cancer Res. 7:1820–1834. 2017.PubMed/NCBI
|
|
35
|
Han Z, Zhang Y, Yang Q, Liu B, Wu J, Zhang
Y, Yang C and Jiang Y: miR-497 and miR-34a retard lung cancer
growth by co-inhibiting cyclin E1 (CCNE1). Oncotarget.
6:13149–13163. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ji X, Qian J, Rahman SMJ, Siska PJ, Zou Y,
Harris BK, Hoeksema MD, Trenary IA, Chen H, Eisenberg R, et al: xCT
(SLC7A11)-mediated metabolic reprogramming promotes non-small cell
lung cancer progression. Oncogene. 37:5007–5019. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Simner C, Novakovic B, Lillycrop KA, Bell
CG, Harvey NC, Cooper C, Saffery R, Lewis RM and Cleal JK: DNA
methylation of amino acid transporter genes in the human placenta.
Placenta. 60:64–73. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cohen AS, Khalil FK, Welsh EA, Schabath
MB, Enkemann SA, Davis A, Zhou JM, Boulware DC, Kim J, Haura EB and
Morse DL: Cell-surface marker discovery for lung cancer.
Oncotarget. 8:113373–113402. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ben L, Jinli Q, Fangxiu X, Yan G, Yu W,
Herbert Y and Biyun Q: MiR-195 suppresses non-small cell lung
cancer by targeting CHEK1. Oncotarget. 6:9445–9456. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lakomy R, Sana J, Hankeova S, Fadrus P,
Kren L, Lzicarova E, Svoboda M, Dolezelova H, Smrcka M, Vyzula R,
et al: MiR-195, miR-196b, miR-181c, miR-21 expression levels and
O-6-methylguanine-DNA methyltransferase methylation status are
associated with clinical outcome in glioblastoma patients. Cancer
Sci. 102:2186–2190. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang Y, Hu Y, Wu G, Yang Y, Tang Y, Zhang
W, Wang K, Liu Y, Wang X and Li T: Long noncoding RNA PCAT-14
induces proliferation and invasion by hepatocellular carcinoma
cells by inducing methylation of miR-372. Oncotarget.
8:34429–34441. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lai JH, She TF, Juang YM, Tsay YG, Huang
AH, Yu SL, Chen JJW and Lai CC: Comparative proteomic profiling of
human lung adenocarcinoma cells (CL 1-0) expressing miR-372.
Electrophoresis. 33:675–688. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sun H and Gao D: Propofol suppresses
growth, migration and invasion of A549 cells by down-regulation of
miR-372. BMC Cancer. 18:12522018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Chen Z, Li JL, Lin S, Cao C, Gimbrone NT,
Yang R, Fu DA, Carper MB, Haura EB, Schabath MB, et al:
cAMP/CREB-regulated LINC00473 marks LKB1-inactivated lung cancer
and mediates tumor growth. J Clin Invest. 126:2267–2279. 2016.
View Article : Google Scholar : PubMed/NCBI
|