Introduction
During pregnancy, the lungs of the fetus do not
exchange oxygen (O2) and carbon dioxide
(CO2), and therefore require a minimal blood supply.
When the baby is born, it breathes by itself. The lungs dilate,
decreasing the pulmonary vascular resistance and left atrium
pressure considerably. However, certain newborns are unable to
decrease their pulmonary pressure due to prenatal or postnatal
factors (1).
The hypoxic level of blood that perfuses the fetal
lungs is perhaps the major determinant of the degree of
vasoconstriction of the pulmonary arterioles. Muscle fibers in
pulmonary arterioles are highly sensitive to oxygen levels and
changes in pH, contract under acidotic and hypoxic conditions, and
vasodilate when there are changes to the partial pressure of oxygen
(PaO2) and pH levels (2). Pulmonary vascular disease may affect
the pulmonary capillaries and/or arterioles responsible for
pulmonary arterial hypertension, or affect the left ventricle,
leading to pulmonary venous hypertension. Pulmonary arterial
hypertension is characterized by dysfunction of endothelial cells
and proliferation of intimal and smooth muscle cells. Pulmonary
vascular resistance and pressure has a deleterious effect on the
right ventricle (RV), with a resulting decrease in cardiac output
and right heart failure (HF), accompanied by the patient having a
decreased ability to exercise and in the most serious cases,
patient mortality (3–6).
In addition, it is likely that a number of
epigenetic changes are involved in the regulation of the perinatal
pulmonary circulation (7–9). Factors involved in pulmonary
hypertension (PH) include hypoxia inducible factor 1α (HIF-1α)
(10), transforming growth factor
β (TGF-β) (11), tumor necrosis
factor α (TNF-α) (12–14), soluble endoglin and placental
growth factor (PlGF) (15),
angiopoetin-2 (Ang-2) (16) and α
smooth muscle actin (α-SMA) protein (17).
Furthermore, pulmonary arteriole vasodilatation
during birth is generally inversely correlated with the muscular
tunic thickness at the end of the gestational period. However, the
muscular tunic is reabsorbed postnatally, a process that occurs
during the first 14 days of life. Occasionally, this reabsorption
may take a considerably longer period of time. This timeframe may
have a negative effect on the necessary adjustments to the
postnatal circulation, as the thickness of the muscular tunic
limits the lumen diameter, rendering pulmonary arterioles more
reactive to stimuli that trigger vasoconstriction (18,19),
which may negatively affect the therapeutic outcome.
Pulmonary arterial hypertension, however, is a
condition that may ultimately trigger right HF. Prostaglandin
I2-targeted drugs are currently used for vascular
endothelial proliferation, expansion of pulmonary vessels and to
inhibit pulmonary vascular remodeling (20). Endothelin receptor antagonists
serve a role in the expansion of pulmonary vessels,
anti-proliferation and endothelial function improvements.
Phosphodiesterase type 5 (PDE5) inhibitors prevent the general
expression of PDE5 in PH by increasing the levels of cGMP in
vascular smooth muscle cells. cGMP triggers dilation of vascular
smooth muscle, leading to an expansion of the pulmonary artery
lumen and a decrease in pulmonary vascular resistance, thereby
decreasing pulmonary arterial pressure (PAP). A previous study has
demonstrated that arginine vasopressin (AVP) is released from the
posterior pituitary gland when there is a decrease in the
circulating blood volume (21,22).
AVP may induce vasopressin type-2 receptor (V2R) expression, while
previous data has suggested that the long-term administration of
V2R antagonists has beneficial actions on cardiac hemodynamics, and
improves myocardial fibrosis and left ventricular (LV) remodeling
together with a diuretic effect in left HF (18,19).
However, the prognostic effect of PH and RV dysfunction in LV
function is of critical importance in determining precisely the
cause and severity of PH prior to making decisions concerning
treatment. Therefore, a comprehensive diagnostic strategy is
required, but targeted HF treatments (medical/interventional)
remain limited (3). Therefore, the
goal of treatment is to delay or prevent the progression of PH.
Although its incidence is not high at present, it is an easily
misdiagnosed disease. The incidence of PH in the newborn is
0.43–6.6 per 1,000 live offspring and most commonly occurs in
preterm newborns (23–25). In adults, the average age of onset
is 36 years, with 75% of patients within the 20–40 years age
category. The onset of PH in adulthood may be associated with the
hypoxic intrauterine environment (26,27).
A possibility is that the expression pattern of molecular markers
associated with PH and hypoxia, and associated factors induced by
hypoxia in utero, remain unchanged in adulthood.
The objectives of the present study were to identify
the physiological roles of factors associated with pulmonary artery
hypertension including V2R, HIF-1α, HIF-2α, TGF-β, PlGF and other
angiogenesis factors. To explore the mechanism of PH induction for
diagnostic and therapeutic possibilities, the expression of these
factors was measured in newborns and in later months (corresponding
to adulthood in the human), utilizing a rat model in which maternal
rats lived in a hypoxic environment during their pregnancy.
Materials and methods
Animals
All experiments on rats were conducted according to
the guidelines for the Humane Treatment of Laboratory Animals
(Ministry of Science and Technology of the People's Republic of
China, Policy. No. 2006 398) (28)
and additional approval for the present study was waived by the
Ethics Committee of the Affiliated People's Hospital of Jiangsu
University (Zhenjiang, China). A total of 20 3-month old female
Sprague Dawley rats, kindly provided by the Shanghai Experimental
Animal Center (Shanghai, China), and were housed under standard
laboratory conditions (22±2°C, 60% relative humidity, 12/12 h
light/dark cycle and access to food and water ad libitum).
The rats were mated and vaginal smears taken each morning were
examined. Day zero of a pregnancy duration of 22 days was denoted
when the presence of sperm was observed. During each rat's
pregnancy and period of lactation, all experimental animals had
ad libitum access to standard laboratory water and food. On
day 7 of pregnancy, the animals were assigned randomly into control
(normoxic) and experimental (hypoxic) groups (Fig. 1).
Hypoxic treatment of maternal
rats
From days 7 to 21 of pregnancy, the rats were
allocated to the control or hypoxic groups. The rats in the hypoxic
group were placed in a hypoxic chamber in which the oxygen supply
was reduced to 10±1% by continuous infusion of nitrogen gas and
compressed air mixture, while expired CO2 was eliminated
by circulating the atmosphere through soda lime. The water
contained in the expired gas was trapped in a chilled glass tank. A
portable oxygen analyzer (S-450; IST-AIM) was calibrated daily and
used to monitor the oxygen concentration of the chamber as
described previously (29).
Blood gases and the pH of arterial blood were
measured in aliquots of blood withdrawn after 1 h exposure to
hypoxia from a cannula positioned in the left femoral artery.
Measurements were performed on 3 randomly selected rats using a
Rapidlab 850 analyzer (Bayer AG) that automatically measured blood
gas levels. After 4 h of hypoxia, the pregnant rats were released
from the hypoxia chamber to normal room air. A total of 10 rats
(controls) were placed in an identical environment under normoxic
conditions. The rats allocated to the hypoxia group were exposed to
a hypoxic environment twice a day for 4 h in the morning and 4 h in
the afternoon, for a total of 2 weeks, with a break for 1 day after
6 days (Fig. 1).
Measurement of pulmonary artery
pressure and the RV hypertrophy index
Following anoxic treatment for 1 and 3 weeks,
maternal rats were anesthetized with an intraperitoneal injection
of 50 mg/kg pentobarbital sodium (30,31).
A catheter was introduced into the RV and the pulmonary artery via
the right external jugular vein. A pressure sensor connected to the
multi-guide physiological recorder was used to make measurements of
the pulmonary artery and to compute the mean PAP. Then, the animals
were sacrificed by exsanguination. In the neonates (7 days old),
0.5–1 ml blood was removed. In the 3- and 6-month-old rats, 8–10 ml
blood was removed. The animals were then decapitated, and the heart
and lung tissues were separated and the blood in the heart was
rinsed away with PBS buffer. The atrium, aorta and pulmonary artery
were cut off along the atrioventricular canal. The RV free wall was
separated along the anterior interventricular sulcus.
Offspring selection and grouping
From each litter, 1 pup was randomly selected and
the heart, lungs, liver, kidney and brain tissues were dissected
and weighed between 3 and 12 h after birth. The size of the litter
was decreased to 8 experimental animals to ensure that the
offspring had equal access to nutrients. The organs from 1 randomly
selected pup in each litter were weighed after 28 days. The
offspring of 10 control and 10 hypoxia litters were randomly
assigned into 6 groups of 3 rats: Maternal; normoxia; and hypoxia
rat groups, at 7 days, 3 months and 6 months after birth. In total,
20 rats, including 2 extra rats, were used in the experiments. The
tissues of 10 rats in each group, which were resected at the
indicated time points, were immediately cut into small pieces for
freezing. Different sections were used for RNA extraction, protein
extraction and hematoxylin & eosin (H&E) and Masson's
staining following slicing. For each experiment, 3 samples were
used from different rats and each sample was measured 3 times. The
offspring rats (female and male) were randomly placed in each group
with at least 1 female rat and 1 male rat in one group without any
differences in appearance (color or size).
Extraction of RNA, reverse
transcription and quantitative (q)PCR to identify target
factors
The frozen lung tissue of rats exposed to hypoxic
conditions (7 days, 3 months and 6 months after birth) was kept in
1 ml TRIzol® in a clean homogenate tube. The tissue was
homogenized for 20 sec and centrifuged at 13,400 × g for 10 min at
room temperature. Subsequently, the isolated supernatant was
transferred into fresh 1.5 ml centrifuge tubes and 200 µl
chloroform and 600 µl isopropyl alcohol were added. Following
centrifugation (13,400 × g, 10 min, 4°C), the precipitate was
washed in ethanol (75%) and the extracted RNA was subsequently
dissolved in diethylpyrocarbonate-treated water.
A SuperScript II reverse transcriptase kit
(Invitrogen; Thermo Fisher Scientific, Inc.) was used to measure
total RNA. qPCR was performed using SYBR Green Supermix and Premix
Ex Taq™ kits (Takara Biotechnology Co., Ltd.) and an iCycler system
(Bio-Rad Laboratories, Inc.). A quantity of 2 µl was then mixed
with 12.5 µl SYBR Green reaction master mix, 0.5 µl forward and 0.5
µl reverse primers. The thermocycler conditions were as follows:
Initial heating at 95°C for 10 min; then 40 cycles of denaturation:
95°C for 15 sec; annealing and elongation: 60°C for 45 sec without
final extension. Melt curve analysis: 95°C for 15 Sec; 60°C for 1
min; 95°C for 15 Sec; 60°C for 15 Sec. The sequences of forward and
reverse primers used are provided in Table I.
 | Table I.Primer sequences of the indicated
target genes. |
Table I.
Primer sequences of the indicated
target genes.
| Genes | Primer
sequences |
|---|
| HIF-1α
(NM_001313919.1) | F:
5′TCTCGGCGAAGCAAAGAGTC3′ |
|
| R:
5′AGCCATCTAGGGCTTTCAGATAA3′ |
| HIF-2α
(NM_010137.3) | F:
5′GAGGAAGGAGAAATCCCGTGA3′ |
|
| R:
5′TATGTGTCCGAAGGAAGCTGA3′ |
| V1R
(NM_016847.2) | F:
5′-TCGTGGTGGCCGTGCTGGGT-3′ |
|
| R:
5′-ACCGCCAGGTCTGCCAGGCT-3′ |
| V2R
(NM_001276298.1) | F:
5′ATCCTGGTGTCTACCACGTCT3′ |
|
| R:
5′GTTCAGCCCGGACTAACAGC3′ |
| PlGF
(NM_008838.1) | F:
5′TCCTTCTTCGTGGACAACTTCT3′ |
|
| R:
5′GTACGACAAGAGATTGACAGTGG3′ |
| TGF-β
(NM_011577.2) | F:
5′CCACCTGCAAGACCATCGAC3′ |
|
| R:
5′CTGGCGAGCCTTAGTTTGGAC3′ |
| Ang-2
(NM_007426.4) | F:
5′CAGCCACGGTCAACAACTC3′ |
|
| R:
5′CTTCTTTACGGATAGCAACCGAG3′ |
| TNF-α
(NM_001278601.1) | F:
5′GTGCTCAGAGCTTTCAAC3′ |
|
| R:
5′ACTCTCCCTTTGCAGAAC3′ |
| α-SMA
(NM_007392.3) | F:
5′CCCAGACATCAGGGAGTAATGG3′ |
|
| R:
5′TCTATCGGATACTTCAGCGTCA3′ |
| GAPDH
(NM_001289726.1) | F:
5′CTGCCCAGAACATCATCC3′ |
|
| R:
5′CTCAGATGCCTGCTTCAC3′ |
The levels of gene expression were normalized to
GAPDH, which was used as the housekeeping gene. Each reaction was
performed in triplicate and qPCR was carried out with the aid of a
sequence detection system (ABI 7300; Applied Biosystems; Thermo
Fisher Scientific, Inc.). The data are presented as means ±
standard deviation (SD) from at least 3 separate measurements.
Expression levels, again normalized to GAPDH expression, were
determined using the 2−ΔΔCq method (32).
Western blotting
The frozen lung tissue was cut into small fragments.
Then 150–250 µl lysis buffer (cat. no. R0010, Beijing Solarbio
Science & Technology Co., Ltd.) containing protease and
phosphatase inhibitors was added to a 20 mg tissue sample, which
was subsequently thoroughly homogenized. The homogenate was spun at
12,000 × g for 15 min at 4°C, and the separated supernatant protein
concentration measured using the BCA assay (Thermo Fisher
Scientific, Inc.). Equal quantities of protein (10 µg) were
separated in 8% denaturing polyacrylamide gel and then electro
transferred onto a nitrocellulose membrane (cat. no. 1620115;
Bio-Rad Laboratories, Inc.). Following blocking at room overnight
at 4°C in TBS-T (containing fat-free milk 5%), membranes were
incubated with the following primary antibodies: Mouse anti-HIF-1α
(1:500, cat. no. Ab113642; Abcam); rabbit anti-HIF-2α (1:1,000,
cat. no. Ab109616; Abcam); rabbit anti-α-SMA (1:300, cat. no.
Ab5694; Abcam); rabbit anti-Ang-2 (1:1,000, cat. no. Ab180820;
Abcam); rabbit anti-TNF-α (1:1,000, cat. no. Ab6671; Abcam); rabbit
anti-TGF-β (1:400, cat. no. Ab92486, Abcam); mouse anti-PlGF
(1:800, cat. no. RM0010-8F09; Novus Biologicals, LLC); rabbit
anti-V2R (1:500, cat. no. ab176488; Abcam); and rabbit anti-GAPDH
(1:2,000, cat. no. 5174; Cell Signaling Technology, Inc.),
overnight at 4°C. Then the membranes were washed by TBS-T for 5 min
three times at room temperature. They were then incubated with
horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG
(1:1,000, cat. no. A0208) and goat anti-mouse IgG (1:1,000, cat.
no. A0216) (both from Beyotime Institute of Biotechnology)
secondary antibodies for 1 h at 37°C, and bands were measured using
an ECL detection system (GE Healthcare Life Sciences).
Densitometric analysis was performed using Image-Pro Plus 6.0
software (Media Cybernetics, Inc.) to quantify the band expression
levels, which were expressed as the band density to total GAPDH
ratio.
H&E staining
The frozen lung tissues, which had been stored at
−80°C, were placed at −20°C for 30 min and then sliced into
5–7-µm-thick sections using a freezing microtome (Leica
Microsystems GmbH). The sections were carefully attached to the
slides and immobilized in acetone at 4°C for 15 min. The sections
were then washed with distilled water and placed for 5 min in
hematoxylin solution (cat. no. 714094, BaSO Biotech Co., Ltd.) at
room temperature for staining, and then washed with ammonia water.
After a few seconds, the sections were washed with running water
for 15 min and dehydrated in 70 and 90% alcohol for 10 min each at
room temperature. Sections were then stained with ethanol eosin
staining solution (cat. no. BA4099, BaSO Biotech Co., Ltd.) at room
temperature for 1–2 min. The stained sections were dehydrated in
99.7% ethanol and the slides were vitrified in dimethyl benzene
twice for 3 min. The samples were placed in a 65°C oven for 15 min
after mounting the slides in neutral balsam and viewed using a CX41
light microscope at magnification ×400 (Olympus Corporation).
Masson's trichrome staining
Each section was attached to a slide coated with
poly-l-lysine and then immobilized for 20 min in 4% acetone
solution and washed 3 times with distilled water for 3 min. Then
the Masson's Trichrome Staining Kit (cat. no. DC0032; Leagene
Biotech Co., Ltd.) was used according to the manufacturer's
protocol. The section was subsequently stained with freshly
prepared Weigert iron hematoxylin solution (hematoxylin
solution/ferric chloride solution=1:1) at room temperature for 5–10
min and then differentiated with acidic ethanol differentiation
solution. Following washing, the section was treated with Masson
blue solution and then stained with aniline blue counterstain
solution at room temperature for 1–2 min after washing the section
with weak acid working fluid and phosphate molybdenum acid.
Finally, the sections were mounted in neutral balsam after
vitrifying them in dimethylbenzene 3 times and viewed with a CX41
light microscope at magnification ×400 (Olympus Corporation).
Percentage of pulmonary vascular wall
thickness to vascular diameter
H&E-stained sections from mouse lung tissues
were observed using a light microscope at magnification, ×400. A
total of 5 small and medium-sized arteries with relatively round
cross sections were selected. The vascular wall thickness/vascular
diameter (WT%) was determined using Image-Pro Plus 6.0 software
(Media Cybernetics, Inc) and the average values were
calculated.
Immunohistochemistry staining
The sections were attached to slides coated with
poly-l-lysine. Then the sections were placed in 3%
H2O2 and incubated in a wet box for 10 min to
block endogenous peroxidase activity. Sections were then washed 3
times in 0.02 M PBS for 3 min and primary antibodies
(anti-rabbit-α-SMA; 1:200, cat. no. Ab5694; Abcam) were added.
Sections were incubated in a wet box and stored at room temperature
for 1 h or overnight at 4°C. Then they were washed 3 times in 0.02
M PBS for 3 min and HRP-conjugated secondary antibodies (1:50, cat.
no. D-3004, Shanghai Long Island Biotec Co., Ltd.) were applied.
They were incubated for 20–30 min at room temperature, washed with
PBS, and then stained using a DAB kit (Shanghai Long Island Biotec.
Co., Ltd; Mindray Medical), and counterstained with hematoxylin
(BaSO Biotech Co., Ltd) for 3 min. They were differentiated in 1%
hydrochloric acid alcohol, vitrified in 100% dimethyl benzene,
mounted in neutral balsam and observed using a CX41 light
microscope (Olympus Corporation).
Statistical analysis
SPSS for Windows v.16.0 (SPSS Inc.) was used for
statistical analysis of the data. Data are presented as the mean ±
SD. A Student's t-test was used to analyze the arterial blood gas
levels [PH, arterial CO2 partial pressure
(PaCO2), PaO2 and SaO2], body
weight and artery pressure. Comparisons between the relative levels
of mRNA and protein of target genes, the ratio of vascular wall
thickness and vascular diameter and positive counts of α-SMA
staining between the control and hypoxia groups in different
treatment days were measured by two-way ANOVA and Bonferroni post
hoc test. All tests were two-sided and P<0.05 was considered to
indicate a statistically significant difference.
Results
Characteristics of offspring
Firstly, the levels of PaCO2,
PaO2, SaO2 and pH in the blood of maternal
pregnant rats were determined after 1 h of hypoxia treatment
(Table II). The results showed
that hypoxia treatment significantly affected the concentrations of
PaO2 and SaO2 (Table II).
 | Table II.Arterial blood gas levels in maternal
pregnant control rats and hypoxic pregnant rats after hypoxia
treatment for 1 h. |
Table II.
Arterial blood gas levels in maternal
pregnant control rats and hypoxic pregnant rats after hypoxia
treatment for 1 h.
| Parameters | Control (n=3) | Hypoxia (n=3) | P-value |
|---|
| PH |
7.42±0.15 | 7.33±0.18 | 0.369 |
| PaCO2,
kPa |
5.67±1.43 | 7.38±2.12 | 0.133 |
| PaO2,
kPa | 10.98±1.22 | 6.58±2.13 | 0.001 |
| SaO2,
% | 94.50±2.30 | 70.50±14.20 | 0.002 |
It was confirmed that 3 weeks of intermittent
hypoxia promoted the development of typical PH and RV hypertrophy
in the rats. These results indicated that the hypoxia PH rat model
was successfully established (data not shown). In the newborns from
the hypoxic rat group, significant differences in body weights of
the newborns on day 21 (~20% lighter) compared with the newborns
from control rats were observed (Table III). Significant differences in
RV pressure (Rvp) and PAP were also observed; these values in the
offspring from hypoxic rats were decreased by approximately
one-half of those in the offspring from the control group. However,
the ratios of lung weight to body weight between the groups were
not statistically different.
 | Table III.Effect of maternal hypoxia on weights
and artery pressure of newborns. |
Table III.
Effect of maternal hypoxia on weights
and artery pressure of newborns.
| Parameters | Control | Hypoxia | P-value |
|---|
| Body weight, g | 5.86±0.31 | 4.87±0.51 | <0.01 |
| Average Rvp,
kPa | 2.56±0.28 | 5.05±0.65 | <0.05 |
| Average PAP,
kPa | 2.86±0.57 | 5.38±0.65 | <0.05 |
| Lung weight to body
weight, % | 2.21±0.63 | 2.03±0.59 | >0.05 |
Changes in epigenetic factors involved
in the control of the pulmonary circulation of the neonate
The mRNA expression levels of HIF-1α, HIF-2α, TGF-β,
TNF-α, PlGF, Ang-2, α-SMA, V1R and V2R in the lung tissues of
newborns, and 3-month- and 6-month-old rats were measured using
RT-qPCR (Fig. 2). The expression
levels of all of these mRNAs were increased in rats that had been
exposed to a hypoxic environment. This may be due to the activation
of HIF induced by long-term hypoxia treatment and the activation of
angiogenic factors regulated by HIF. However, the expression of
these factors were mostly normalized 3 months after birth. Only the
levels of V2R and Ang-2 exhibited significant differences between
the two groups at 3 months after birth, with increased expression
levels in the hypoxia groups. The transcription levels of the other
mRNAs were all significant increased between in the hypoxia
compared to the control group at 7 days after birth. As the
increase in V2R mRNA transcription levels was sustained in the
hypoxia group at 6 months after birth, V2R may be an indicator for
PH in postnatal rats. However, the transcription levels of several
other mRNAs were also significantly different between the hypoxia
and control groups at 6 months after birth. In particular, HIF-1α,
Ang-2, a-SMA mRNAs, and to a certain extent PlGF mRNAs, were
expressed at significantly increased levels in pups from normoxic
maternal rats compared with pups from hypoxic rats. Compared with
their respective levels at birth, these mRNAs in the control group
were all increased at 6 months. The protein expression levels,
determined by western blotting, confirmed the mRNA expression
results (Fig. 3). For example, the
protein expression level of V2R in the lung tissue of the
experimental group was 3.2-fold higher compared with the level in
the control group, probably due to exposure to hypoxia in
utero.
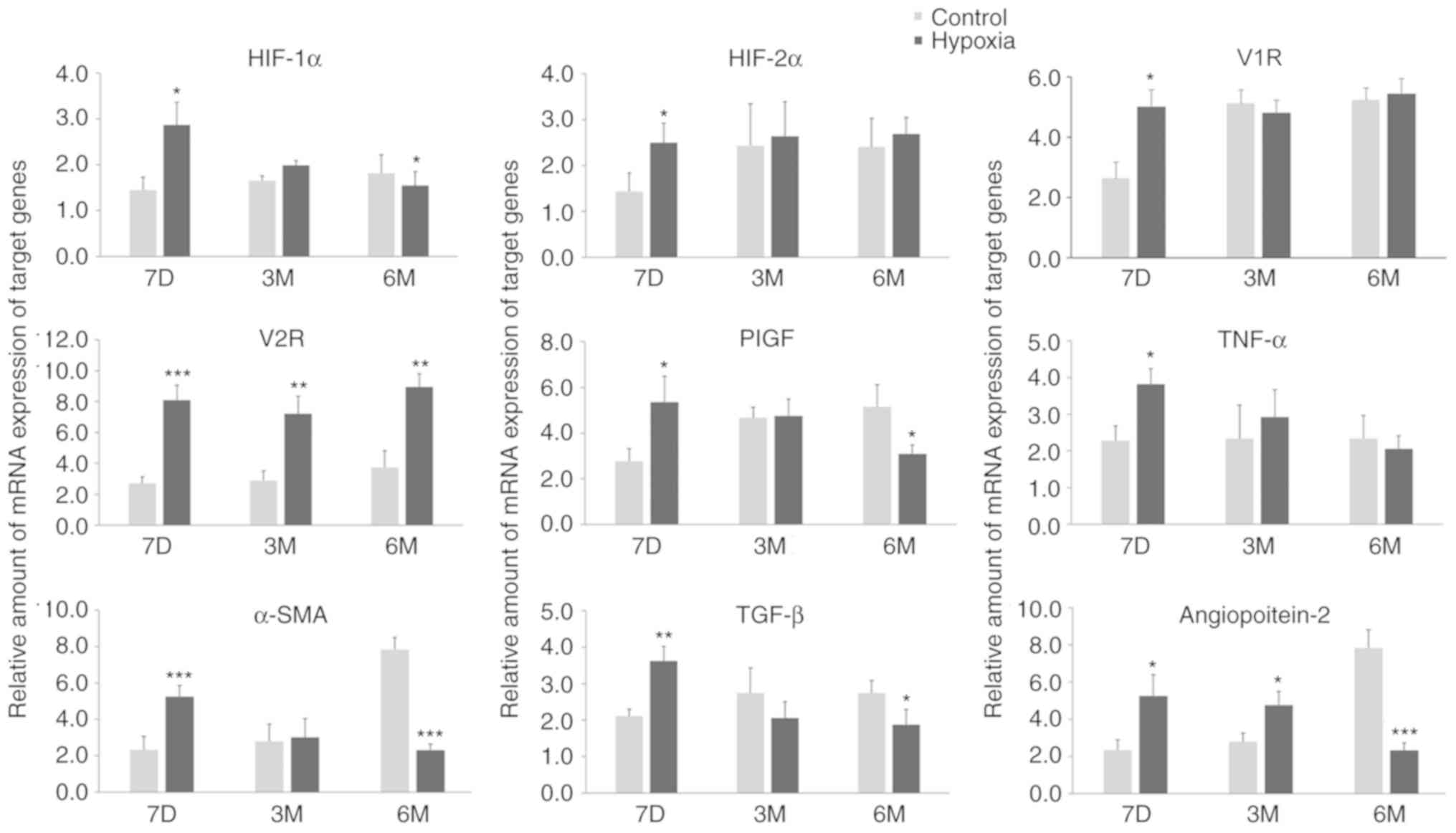 | Figure 2.Maternal rats were exposed to
continuous hypoxia from days 7–21 during pregnancy and the relative
levels of mRNA of target genes in lung tissues of pups at 7D, 3M
and 6M after birth were measured. n=3. Samples were each measured 3
times. Data are presented as mean ± standard deviation. A two-way
ANOVA and Bonferroni post hoc test were used to analyze the data
between the control and hypoxia groups. *P<0.05, **P<0.01 and
***P<0.001 vs. control. HIF-1α, hypoxia-inducible factor-1α;
HIF-2α, hypoxia-inducible factor-2α; V1R, vasopressin type-1
receptor; V2R, vasopressin type-2 receptor; PlGF, placental growth
factor; TGF-β, Transforming growth factor-β 1; TNF-α, tumor
necrosis factor-α; α-SMA, α-smooth muscle aorta; 7D, 7 days; 3M, 3
months; 6M, 6 months. |
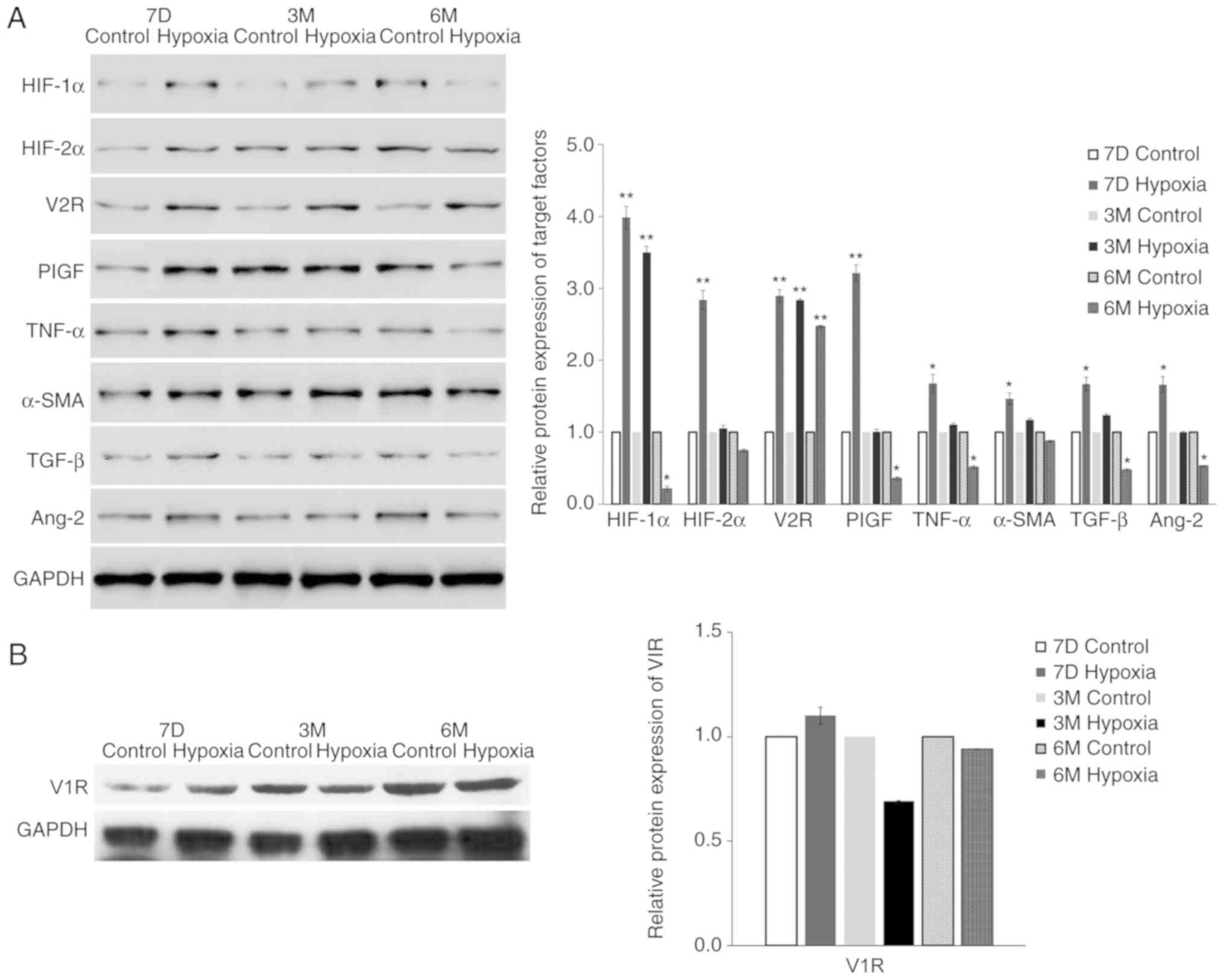 | Figure 3.Maternal rats were exposed to
continuous hypoxia from days 7–21 during pregnancy and the protein
levels of (A) indicated proteins and the (B) target gene V1R in
lung tissues of pups at 7D, 3M and 6M after birth were measured by
western blot analysis. The expression of GAPDH was used for
normalization. n=3. Samples were each measured 3 times. Data are
presented as mean ± standard deviation. A two-way ANOVA and
Bonferroni post hoc test were used to analyze the data between the
control and hypoxia groups. *P<0.05 and **P<0.01 vs. control.
HIF-1α, hypoxia-inducible factor-1α; HIF-2α, hypoxia-inducible
factor-2α; V1R, vasopressin type-1 receptor; V2R, vasopressin
type-2 receptor; PlGF, placental growth factor; TGF-β, Transforming
growth factor-β 1; TNF-α, tumor necrosis factor-α; α-SMA, α-smooth
muscle aorta; 7D, 7 days; 3M, 3 months; 6M, 6 months. |
Blood vessels in the lungs of
offspring rats are damaged following fetal hypoxia
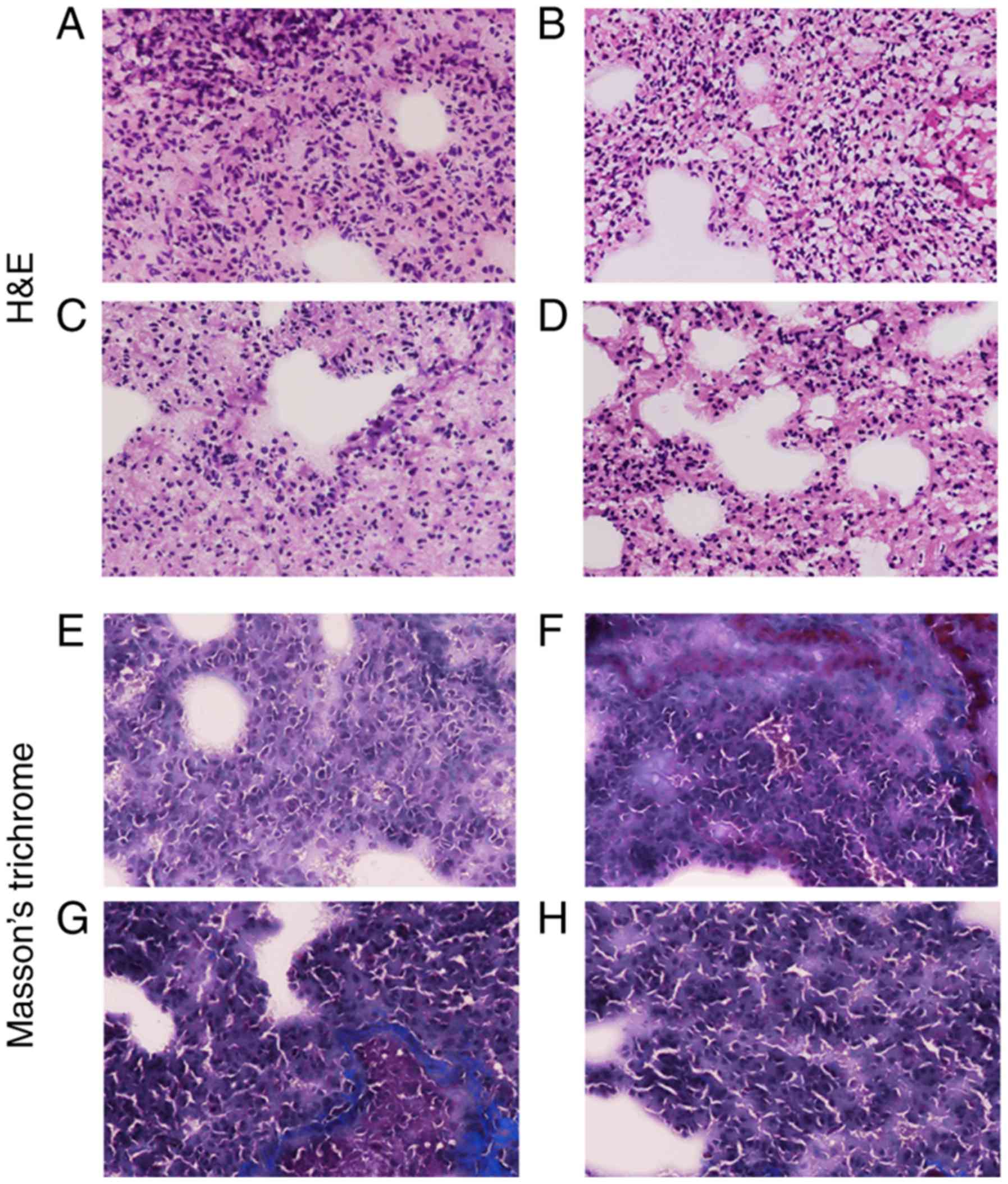
H&E (Fig. 4A-D)
and Masson's (Fig. 4E-H) staining
protocols of the tissue sections demonstrated that the lung tissues
from hypoxia rats were significantly damaged due to the long-term
hypoxia treatment, and the lung weight of newborns was
significantly affected (Table
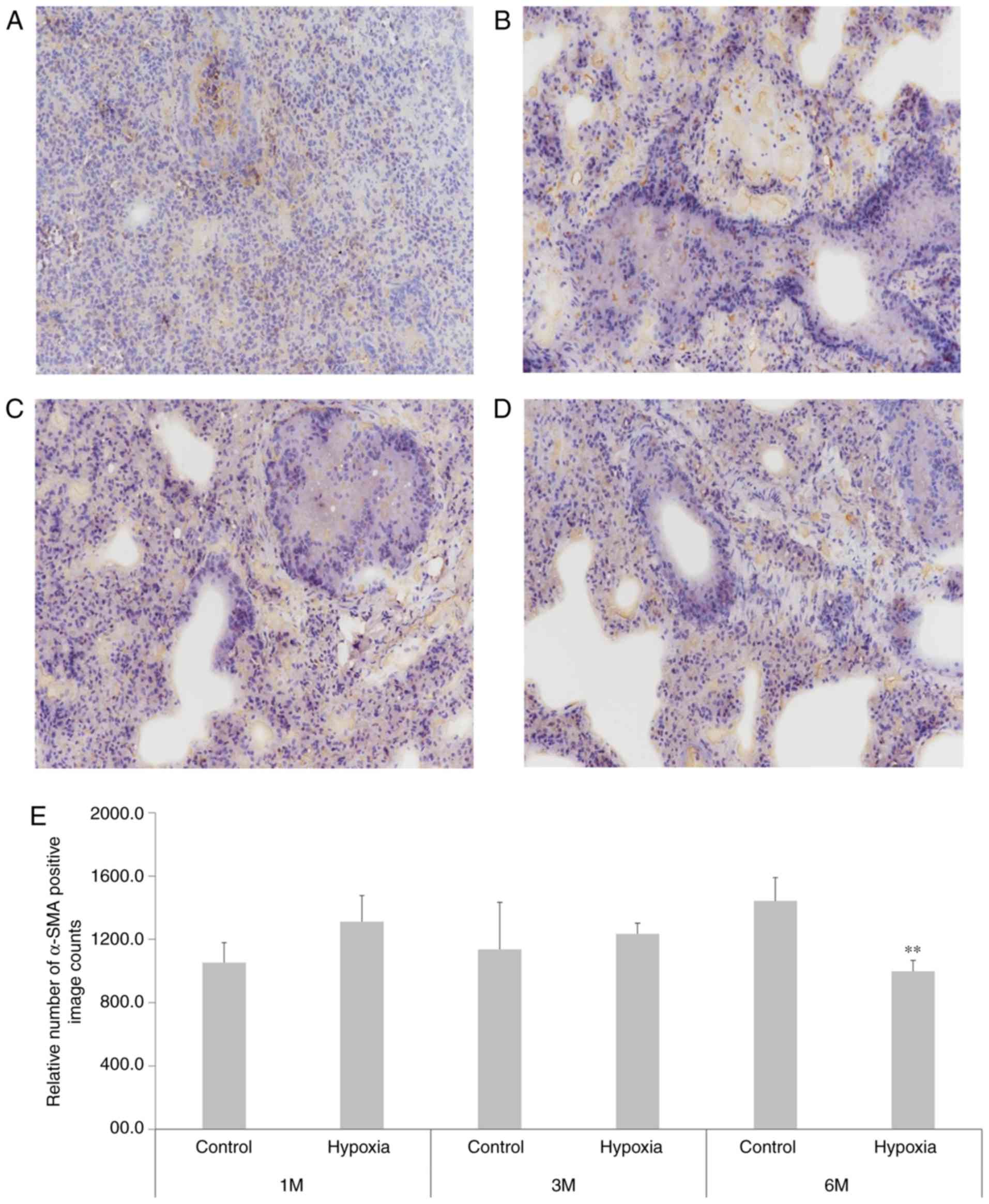
III). α-SMA immunohistochemical staining (Fig. 5) indicated that at 1 month after
birth, the positive count of α-SMA staining was increased in the
lung tissue of rats in the hypoxia group compared with the
controls. However, at 6 months, the positive count of α-SMA
staining decreased in the hypoxia rat group and inversely increased
in the control group.
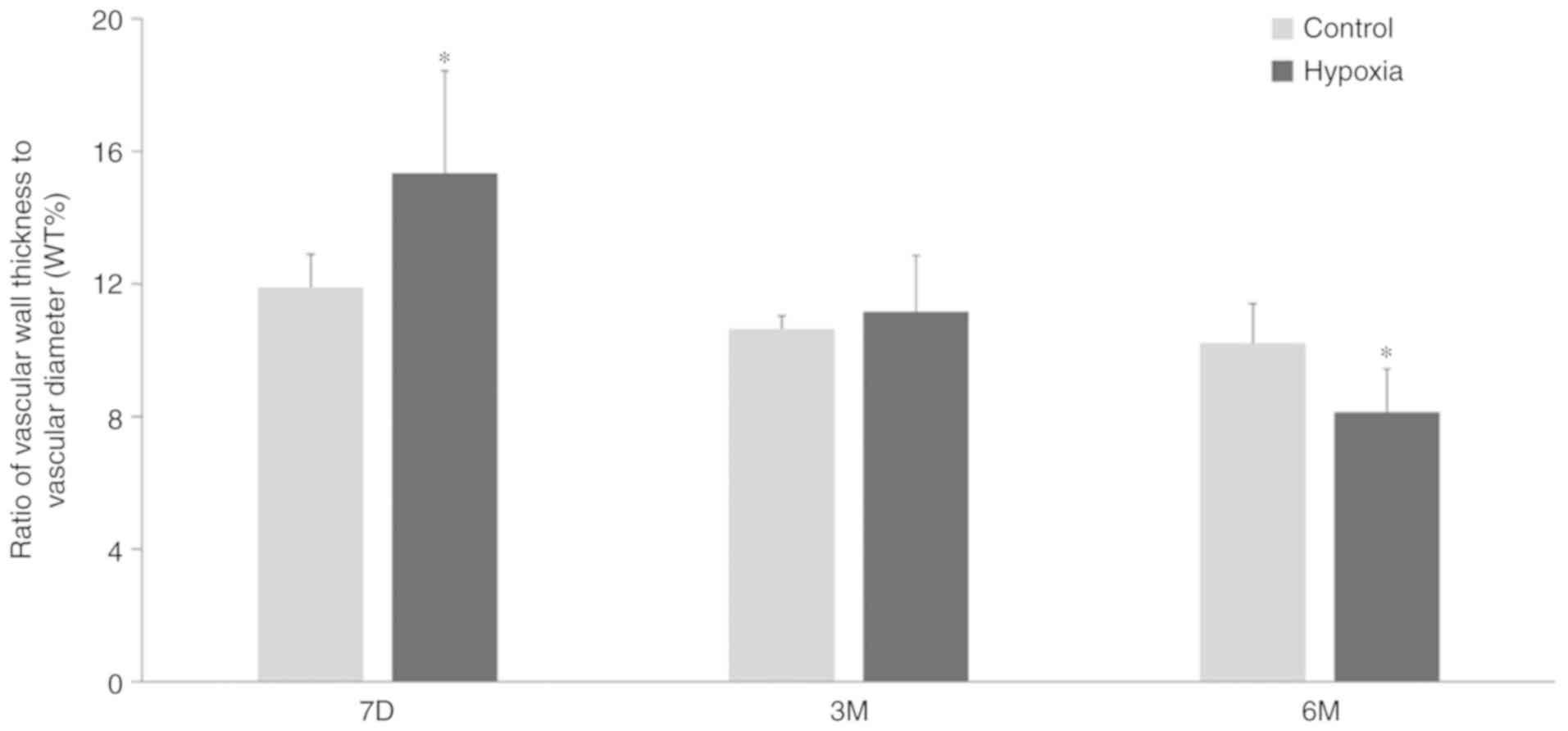
Changes in WT% in offspring exposed to
hypoxia
The WT% was measured using sections stained with
H&E and Masson's stain (Fig.
4). Compared with the control group, the WT% value of 7-day old
pups in the hypoxia group was significantly increased compared with
age-matched pups in the control group. However, as postnatal
adjustment to normoxic conditions progressed, the vascular wall of
the pups exposed to hypoxia became considerably thinner and the
vascular diameter became larger (Fig.
6). The WT% at 3 months after birth in the hypoxic animals was
equivalent to that of the control pups. At 6 months, the value
decreased further, to below the level in the control pups. By
contrast, the WT% values of the normoxia control pups showed no
significant changes at 7 days, 3 months and 6 months after birth,
demonstrating that the WT% values of the pups exposed to hypoxia
treatment during gestation were significantly smaller compared with
those of the control pups at 6 months after birth.
Discussion
In the present study, persistent PH demonstrated to
be caused in newborn rats by the exposure of the mother to a
hypoxic environment for 3 weeks during pregnancy. PH is a complex
disease reflected in physiological (vasoconstriction) or structural
(arteriole remodeling and a decrease in pulmonary vessel density)
changes. As the developmental stage of rats at birth is roughly
equivalent to humans in utero at 3 months, the rat
hypothalamus and pituitary gland at birth and up to 7 days after
are still under development, which are critical central factors
involved in blood pressure regulation of the animals. The present
study identified that the expression of several
angiogenesis-associated factors, including HIF-1α, HIF-2α, TGF-β,
TNF-α, PlGF, α-SMA, Ang-2 and V2R, were affected to different
extents. Among these factors, which were all increased at birth,
only V2R expression remained increased in pups exposed to hypoxia
in utero until 6 months of age, compared with the control
pups. The V2R expression levels in their lung tissue at both 3
months and 6 months was at least 2-fold greater compared with those
of normoxic control pups. These results suggest that intrauterine
hypoxia may be responsible for the decreased expression levels of
vascular factors in lung tissue and the decrease in the number of
pulmonary vessels, as well as thinning of the pulmonary vascular
walls, as observed in the tissue sections of 6-month-old offspring
exposed to hypoxia in utero.
Activation of V2R in cancer cells leads to a release
of factors that decrease their metastatic potential (33,34).
Previous studies have demonstrated that long-term administration of
the selective competitive V2R and kidney V1R receptor antagonist
tolvaptan elicited positive effects on cardiac hemodynamics, and
moderated LV remodeling and myocardial fibrosis in association with
diuretic therapy (35,36). Therefore, we hypothesized that
continuous chronic V2R expression negatively affected the pulmonary
vasculature in the experimental model of the present study.
HIF-1α has been demonstrated to be a vital mediator
in PH and to alleviate PH when knocked down by RNA interference in
adult rats (10). In addition to
HIF-1α, data concerning HIF-2α, a HIF-1α homologue, has revealed
that heterozygous deletion of HIF-2α can attenuate PH induced by
hypoxia in mice (23), which
suggests that HIF-2α also contributes to the development of hypoxic
PH. The present study identified that the levels of HIF-1α and 2α
were raised in hypoxia pups at birth; however, they were decreased
compared with control levels after 3 months. At 6 months, both
HIF-1α and HIF-2α levels were significantly decreased, suggesting
that it is unlikely that activation of HIF-1α and 2α is the primary
cause of PH. It was also observed that V2R, which is closely
related to HF, was highly expressed in lung tissues of hypoxia rats
following birth. Therefore, we hypothesized that V2R is not only an
indicator of the potential risk of PH in infants suffering from
hypoxia, but may actually be directly involved in PH and could be
considered a target for treatment. Goto et al (21) reported that tolvaptan may
contribute to RV remodeling in a hypoxic rat model and had a
detrimental effect on pulmonary heart hypertension. However, their
model used the vascular endothelial growth factor receptor
inhibitor SU5416 in combination with hypoxia to induce PH on
6-week-old male Sprague-Dawley rats, and then evaluated the
effects, which is unsuitable for a comparison with the results of
the present study, which analyzed the effects of hypoxic conditions
during pregnancy on the offspring of rats without additional
treatments. Conversely, a few previous studies observed that
tolvaptan was efficient in treating patients with right heart
failure due to PH (37,38).
A limitation of the present study is that hypoxia
related parameters, such as blood vessel growth pattern, and
pulmonary hemodynamic changes were not measured in the pregnant
rats after hypoxia, which might have provided additional valuable
information for the prediction of PH in their offspring.
In conclusion, the present study identified that the
V2R expression levels in newborn rats induced by maternal exposure
to hypoxia were sustained to significantly increased levels
compared with the pups not exposed to maternal hypoxia. V2R may be
a useful marker for the diagnosis and prognosis of PH, and for the
risk of right heart failure in fetuses suffering from hypoxia
during pregnancy.
Acknowledgements
Not applicable.
Funding
Funding for the present study was provided by grants
from the National Natural Science Foundation of China (grant nos.
81370181 and 81770089) and Zhenjiang Key Research and Development
Program of China (grant no. SH2018077).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
KH was responsible for the conception and design of
the study. HD, YL, PL, HH, LZ, JY and YX were responsible for
acquisition and analysis of the data. MX, HH and LZ were
responsible for the statistical analysis. HD, KH, HH, PL and MX
drafted the manuscript; YL, LZ, JY and YX revised the manuscript
critically. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All experiments on rats were conducted according to
the guidelines for the Humane Treatment of Laboratory Animals
(Ministry of Science and Technology of the People's Republic of
China, Policy No. 2006 398) and further approval for this study was
waived by the ethics committee of the Affiliated People's Hospital
of Jiangsu University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mathew B and Lakshminrusimha S: Persistent
pulmonary hypertension in the newborn. Children (Basel).
4:632017.
|
|
2
|
Rudolph AM and Yuan S: Response of the
pulmonary vasculature to hypoxia and H+ ion
concentration changes. J Clin Invest. 45:399–411. 1966. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tabima DM, Frizzell S and Gladwin MT:
Reactive oxygen and nitrogen species in pulmonary hypertension.
Free Radic Biol Med. 52:1970–1986. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Galiè N, Hoeper MM, Humbert M, Torbicki A,
Vachiery JL, Barbera JA, Beghetti M, Corris P, Gaine S, Gibbs JS,
et al: Guidelines for the diagnosis and treatment of pulmonary
hypertension: The task force for the diagnosis and treatment of
pulmonary hypertension of the European society of cardiology (ESC)
and the European respiratory society (ERS), endorsed by the
international society of heart and lung transplantation (ISHLT).
Eur Heart J. 30:2493–2537. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Simonneau G, Gatzoulis MA, Adatia I,
Celermajer D, Denton C, Ghofrani A, Gomez Sanchez MA, Krishna Kumar
R, Landzberg M, Machado RF, et al: Updated clinical classification
of pulmonary hypertension. J Am Coll Cardiol. 62:D34–D41. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rosenkranz S: Pulmonary hypertension 2015:
Current definitions, terminology, and novel treatment options. Clin
Res Cardiol. 104:197–207. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Delaney C and Cornfield DN: Risk factors
for persistent pulmonary hypertension of the newborn. Pulm Circ.
2:15–20. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Storme L, Aubry E, Rakza T, Houeijeh A,
Debarge V, Tourneux P, Deruelle P and Pennaforte T; French
Congenital Diaphragmatic Hernia Study G, : Pathophysiology of
persistent pulmonary hypertension of the newborn: Impact of the
perinatal environment. Arch Cardiovasc Dis. 106:169–177. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xu XF, Ma XL, Shen Z, Wu XL, Cheng F and
Du LZ: Epigenetic regulation of the endothelial nitric oxide
synthase gene in persistent pulmonary hypertension of the newborn
rat. J Hypertens. 28:2227–2235. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li Y, Shi B, Huang L, Wang X, Yu X, Guo B
and Ren W: Suppression of the expression of hypoxia-inducible
factor-1alpha by RNA interference alleviates hypoxia-induced
pulmonary hypertension in adult rats. Int J Mol Med. 38:1786–1794.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ambalavanan N, Nicola T, Hagood J, Bulger
A, Serra R, Murphy-Ullrich J, Oparil S and Chen YF: Transforming
growth factor-beta signaling mediates hypoxia-induced pulmonary
arterial remodeling and inhibition of alveolar development in
newborn mouse lung. Am J Physiol Lung Cell Mol Physiol.
295:L86–L95. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Özpelit E, Akdeniz B, Özpelit ME, Tas S,
Bozkurt S, Tertemiz KC, Sevinc C and Badak Ö: Prognostic value of
neutrophil-to-lymphocyte ratio in pulmonary arterial hypertension.
J Int Med Res. 43:661–671. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Seyfarth HJ, Sack U, Gessner C and Wirtz
H: Angiogenin, bFGF and VEGF: angiogenic markers in breath
condensate of patients with pulmonary hypertension. Pneumologie.
69:207–211. 2015.(In German). PubMed/NCBI
|
|
14
|
Rameh V and Kossaify A: Role of biomarkers
in the diagnosis, risk assessment, and management of pulmonary
hypertension. Biomark Insights. 11:85–89. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Muttukrishna S, Swer M, Suri S, Jamil A,
Calleja-Agius J, Gangooly S, Ludlow H, Jurkovic D and Jauniaux E:
Soluble flt-1 and PlGF: New markers of early pregnancy loss? PLoS
One. 6:e180412011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cohen SS, Powers BR, Lerch-Gaggl A, Teng
RJ and Konduri GG: Impaired cerebral angiogenesis in the fetal lamb
model of persistent pulmonary hypertension. Int J Dev Neurosci.
38:113–118. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu XF, Gu WZ, Wu XL, Li RY and Du LZ:
Fetal pulmonary vascular remodeling in a rat model induced by
hypoxia and indomethacin. J Matern Fetal Neonatal Med. 24:172–182.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Haworth SG and Reid L: Persistent fetal
circulation: Newly recognized structural features. J Pediatr.
88:614–620. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Haworth SG: Pulmonary vascular remodeling
in neonatal pulmonary hypertension. State of the art. Chest.
93:133S–138S. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pluchart H, Khouri C, Blaise S, Roustit M
and Cracowski JL: Targeting the prostacyclin pathway: Beyond
pulmonary arterial hypertension. Trends Pharmacol Sci. 38:512–523.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Goto I, Dohi K, Ogihara Y, Okamoto R,
Yamada N, Mitani Y and Ito M: Detrimental impact of vasopressin V2
receptor antagonism in a su5416/hypoxia/normoxia-exposed rat model
of pulmonary arterial hypertension. Circul J. 80:989–997. 2016.
View Article : Google Scholar
|
|
22
|
Ikeda T, Iwanaga Y, Watanabe H, Morooka H,
Akahoshi Y, Fujiki H and Miyazaki S: Effects of long-term blockade
of vasopressin receptor types 1a and 2 on cardiac and renal damage
in a rat model of hypertensive heart failure. J Cardiovasc
Pharmacol. 66:487–496. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stayer SA and Liu Y: Pulmonary
hypertension of the newborn. Best Pract Res Clin Anaesthesiol.
24:375–386. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Teng RJ and Wu TJ: Persistent pulmonary
hypertension of the newborn. J Formos Med Assoc. 112:177–184. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Roofthooft MT, Elema A, Bergman KA and
Berger RM: Patient characteristics in persistent pulmonary
hypertension of the newborn. Pulm Med. 2011:8581542011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Niermeyer S: Cardiopulmonary transition in
the high altitude infant. High Alt Med Biol. 4:225–239. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Niermeyer S: Going to high altitude with a
newborn infant. High Alt Med Biol. 8:117–123. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Guidance on the Care of Laboratory
Animals: (2006) No. 398, Ministry of Science and Technology.
2006.
|
|
29
|
Wang Z, Huang Z, Lu G, Lin L and Ferrari
M: Hypoxia during pregnancy in rats leads to early morphological
changes of atherosclerosis in adult offspring. Am J Physiol Heart
Circ Physiol. 296:H1321–H1328. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Canadian Council on Animal Care. CCAC
guidelines on, . Euthanasia of animals used in science, 2010.
http://www.ccac.ca/Documents/Standards/Guidelines/Euthanasia.pdfFebruary
21–2016
|
|
31
|
Albert Einstein College of Medicine
Institute for Animal Studies. Recommended Methods of Anesthesia,
Analgesia, and Euthanasia for Laboratory Animal Species. 2014.
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pifano M, Garona J, Capobianco CS,
Gonzalez N, Alonso DF and Ripoll GV: Peptide agonists of
vasopressin V2 receptor reduce expression of neuroendocrine markers
and tumor growth in human lung and prostate tumor cells. Front
Oncol. 7:112017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ripoll GV, Garona J, Hermo GA, Gomez DE
and Alonso DF: Effects of the synthetic vasopressin analog
desmopressin in a mouse model of colon cancer. Anticancer Res.
30:5049–5054. 2010.PubMed/NCBI
|
|
35
|
Morooka H, Iwanaga Y, Tamaki Y, Takase T,
Akahoshi Y, Nakano Y, Fujiki H and Miyazaki S: Chronic
administration of oral vasopressin type 2 receptor antagonist
tolvaptan exerts both myocardial and renal protective effects in
rats with hypertensive heart failure. Circ Heart Fail. 5:484–492.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yamazaki T, Izumi Y, Nakamura Y, Yamashita
N, Fujiki H, Osada-Oka M, Shiota M, Hanatani A, Shimada K, Iwao H
and Yoshiyama M: Tolvaptan improves left ventricular dysfunction
after myocardial infarction in rats. Circ Heart Fail. 5:794–802.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tamura Y, Kimura M, Takei M, Ono T, Kuwana
M, Satoh T, Fukuda K and Humbert M: Oral vasopressin receptor
antagonist tolvaptan in right heart failure due to pulmonary
hypertension. Eur Respir J. 46:283–286. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Joko Y, Ikemura N, Miyata K, Shiraishi Y,
Tanaka H, Yoshida T, Ikegami Y, Fuse J, Sakamoto M and Momiyama Y:
Efficacy of tolvaptan in a patient with right-sided heart failure
and renal dysfunction refractory to diuretic therapy. J Cardiol
Cases. 9:226–229. 2014. View Article : Google Scholar : PubMed/NCBI
|