Introduction
Arsenic trioxide (ATO) is the highly toxic main
ingredient of arsenic, which is used in Chinese medicine. For
instance, ATO has been used to successfully treat recurrent and
refractory acute promyelocytic leukemia (APL) since the 1970s
(1). The efficacy of ATO is
explained by its ability to induce apoptosis and the partial
differentiation of APL cells (2),
and understanding the mechanism of ATO against APL has enabled
additional breakthroughs in cancer treatment (2). Although well-established for its
therapeutic effects, QT interval (measure of the time between the
start of the Q wave and the end of the T wave in the hearts
electrical cycle) prolongation, action potential variation, torsade
de pointes and sudden cardiac death are associated with ATO
treatment and have hindered its application (3,4). The
major cause of cardiotoxicity is the increased oxidative stress
resulting from arsenic deposition (5), and this likely involves a variety of
mechanisms, including oxidative DNA damage, the production of
reactive oxygen species (ROS) in cardiomyocytes and arsenic
accumulation (6). Oxidative stress
is the most compelling of the numerous mechanisms underlying
ATO-induced toxicity, as it is a key driver in apoptosis and
myocardial injury (7,8). Therefore, antioxidative agents may
protect against ATO-induced cardiotoxicity. For example, the
antioxidant resveratrol protects against ATO-induced cardiotoxicity
by reducing oxidative stress (7).
The accumulation of ROS causes oxidative damage due
to an imbalance in the production and elimination of oxygen free
radicals in cells (9). Free
radicals can induce a series of oxidation reactions that cause
damage from the molecular to the organ level (10). Malondialdehyde (MDA), a product of
lipid peroxidation (LPO), can be used to reflect free radical
metabolism and the state of cells attacked by free radicals
(11). Superoxide dismutase (SOD)
is an oxygen free radical scavenger that protects cells by
neutralizing ROS, and SOD levels indirectly represent the ability
of the cell to protect itself from free radicals (12). When the production and
neutralization of free radicals are in dynamic equilibrium, tissue
is not susceptible to damage (13). LPO is caused by the combined effect
of unsaturated fatty acids and free radicals, resulting in an
imbalance between the unsaturated fatty acids and proteins in
membranes (14). This imbalance
reduces membrane fluidity and permeability, increases the influx of
Ca2+ and indirectly restricts the action of membrane
proteins, which leads to cellular swelling and Ca2+
overload (15).
Cytokines are inflammatory mediators involved in
immune reactions and pro-inflammatory cytokines, such as IL-1, IL-6
and TNF-α, contribute to the pathology and progression of numerous
diseases, such as numerous types of inflammatory disease and cancer
(16). NF-κB/p65 regulates genes
involved in inflammatory responses and can be induced in multiple
cell types (17). Moreover,
arsenic exposure likely increases the expression of inflammation
molecules, and as a transcriptional master regulator of
pro-inflammatory cytokines, NF-κB/p65 can be stimulated by ATO and
trigger inflammatory responses (18).
The primary mechanism underlying the adverse effects
of arsenic-induced cardiac injury may be associated with the
generation of ROS and ROS-induced apoptosis (19,20).
Oxidative stress is an important element of ATO-induced
mitochondria-mediated apoptosis (21). Apoptosis is a common mechanism of
cardiotoxicity, and ATO triggers apoptosis in cells (22). However, clinically suitable,
cardioprotective therapeutics do not protect against arsenic
toxicity.
Silent information regulator of transcription
(SirT1) is a NAD+-dependent histone deacetylase, which
regulates the proliferation, apoptosis, differentiation, aging and
metabolism of cells in a tissue-specific manner, as well as serves
an important role in the modulation of angiocardiopathy (23). The overexpression of SirT1, within
a certain range, counteracts myocardial hypertrophy and aging
(24). Furthermore, SirT1 exerts
pro-survival effects in a variety of cells and tissues due to its
antioxidative and anti-apoptotic properties (25), suggesting that SirT1 is a promising
mediator of the cardioprotective effects required to combat
ATO-induced cardiotoxicity.
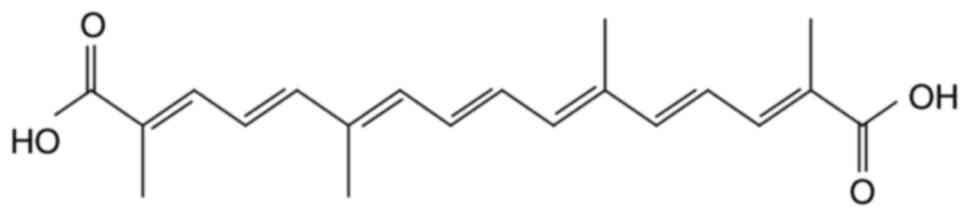
Crocetin (CRT;
C20H24O4; molecular weight, 328;
Fig. 1) is the critical ingredient
of saffron (Crocus sativus L) and possesses beneficial
pharmacological effects (26). In
Ayurvedic and other folk medicines, saffron is used for its
numerous pharmacological properties such as analgesia, prostration,
renoprotection, memory enhancement and anti-depression (27,28).
CRT can also reduce LPO in ischemia-reperfusion-induced oxidative
damage in male albino rats and scavenge free radicals (28). CRT is a natural carotenoid
dicarboxylic acid (29), which
possesses a diterpene and has a symmetrical structure with
alternating trans-double bonds in alkyl chains and four methyl and
carboxyl groups at both ends of the backbone (30,31).
CRT has beneficial cardiovascular effects, such as reducing
oxidative stress (32) and
inhibiting the development of insulin resistance (33), atherosclerosis (34), hypertension (35) and cardiac hypertrophy (36,37).
CRT also possesses a broad diversity of pharmacological properties,
including antioxidative and anti-inflammatory effects (38,39).
CRT significantly decreases LPO, enhances glutathione peroxidase
(GSH-Px) and SOD activity and improves the histopathology of the
myocardium in hypertrophic tissue (32). Moreover, CRT regulates myocardial
enzymes to reduce cardiac cytotoxicity and apoptosis levels
(32,40). However, it is yet to be elucidated
whether CRT attenuates ATO-induced cardiotoxicity and reduces
arsenic accumulation.
Based on the aforementioned findings, the present
study aimed to investigate the ameliorative effects and mechanism
of CRT on ATO-induced cardiotoxicity in rats. It was hypothesized
that CRT could protect against cardiotoxicity, and thus an
ATO-induced cardiotoxicity model was established to examine CRT.
Furthermore, oxidative stress, inflammatory cytokines and apoptotic
factors were evaluated in rats with cardiotoxicity.
Materials and methods
Experimental materials
CRT (98% purity) was purchased from Shanghai Yuanye
Bio-Technology Co., Ltd. ATO was purchased from Beijing Shuanglu
Pharmaceutical Co., Ltd. Sodium chloride solution was purchased
from Shijiazhuang No. 4 Pharmaceutical Co., Ltd. CRT and ATO were
dissolved in saline and used immediately after preparation. CRT
weas dissolved to a final concentration of 20 or 40 mg/kg, while
ATO was dissolved to a final concentration of 5 mg/kg.
Experimental animals
In total, 40 adult male Sprague-Dawley (SD) rats
(age, 6–8 weeks; weight 180–220 g) were obtained from the
Laboratory Animal Center of Hebei Medical University. Rats were
raised in rust-free cages at 22–25°C and 45–60% relative humidity
on a 12-h light-dark cycle with ad libitum intake of
granular rat chow and tap water. Veterinarians and researchers
monitored the animals health and behavior twice per day at 8:00 and
17:00. All animal experiments were approved by the Institutional
Animal Experimental Ethics Committee of the Hebei University of
Chinese Medicine.
Experimental model
After 7 days of adaptive feeding, the rats were
randomly separated into four groups (n=10): Control (Con), ATO,
CRT-L (ATO + low concentration of CRT treatment) and CRT-H (ATO +
high concentration of CRT treatment). The CRT-L and CRT-H groups
were given 20 and 40 mg/kg CRT, respectively, followed by 5 mg/kg
ATO 6 h later. The ATO group was given a CRT-matched amount of 0.9%
sodium chloride solution followed by 5 mg/kg of ATO 6 h later. The
Con group was given the same amount of 0.9% sodium chloride
solution followed by an ATO-matched amount of 0.9% sodium chloride
solution 6 h later. All treatments were delivered daily via
intraperitoneal injection for 10 days. After 10 days, the rats were
anesthetized with 1.0 g/kg ethyl carbamate intraperitoneally
(41,42). An electrocardiogram (ECG) was
performed in anesthetized rats. Then, the blood (5 ml) was
collected by exsanguination from the abdominal aorta (43), and the serum was isolated via
centrifugation at 1,500 × g for 10 min at 4°C and stored at −20°C
for further analyses.
The rats were euthanized with intraperitoneal
injection of sodium pentobarbital (200 mg/kg) (44). Then, hearts were quickly dissected
out from each animal and the blood was removed using cold
physiological saline. The heart tissue samples were fixed with 4%
paraformaldehyde solution at room temperature for 24 h or frozen in
liquid nitrogen, and then stored at −20°C until use.
Electrocardiogram measurement
After the final treatment, the rats were deprived of
food and water for 12 h. The rats were anesthetized, and then three
needle electrodes were attached to the right arm, left arm and left
leg of the rats (45), and the ECG
patterns were recorded using a RM6240BD Biological Signal
Collection system (Cheng Yi) to monitor changes in ECG heart rate
and J-point elevation.
Histopathological analysis
After rats were euthanized via overdose sodium
pentobarbital (200 mg/kg), the hearts were rapidly excised. Prior
to hematoxylin and eosin (H&E) staining, the apical myocardium
was fixed in 4% paraformaldehyde for 24 h at room temperature. The
heart tissues were removed from the fixing fluid, and were then
cleared, dehydrated, macerated and embedded in paraffin. The tissue
samples were sectioned at 5-µm thickness and were stained with
hematoxylin for 15 min and eosin for 5 min, both at room
temperature according to standard procedures (46). The samples were visualized under an
optical microscope at ×400 magnification (Leica DM4000B; Leica
Microsystems GmbH).
Detection of lactate dehydrogenase
(LDH) and creatine kinase (CK) activities
When the rats were anesthetized, blood (5 ml) was
collected by exsanguination from the abdominal aorta and serum was
isolated via centrifugation at 1,500 × g for 10 min at 4°C. Then,
diagnostic markers for CK (cat. no. A032) and LDH (cat. no. A020-1)
were measured using commercially available LDH and CK kits (Nanjing
Jiancheng Bioengineering Institute) according to the manufacturers
instructions.
Detection of ROS
The frozen cardiac tissues were used to analyze ROS
generation reflected in the fluorescence intensity of
dihydroethidium (DHE; cat. no. KGAF019; Nanjing KeyGen Biotech Co.,
Ltd.). After the animals were euthanized by overdose of sodium
pentobarbital, the hearts were quickly excised, embedded in the
optimal cutting temperature (OCT) compound and flash-frozen in
liquid nitrogen. The unfixed frozen samples were sectioned
(thickness, 10 µm) at −20°C (47).
After fixing with 50% OTC at −80°C for 2 h, the sections were
washed three times with PBS for 5 min each time and DHE (50 µM) was
applied to each tissue section, which was incubated in a
light-protected humidified chamber at 37°C for 30 min. Sections
were washed three times with PBS for 5 min each time (48). Subsequently, the sections were
sealed with neutral balsam (cat. no. 10004160; Sinopharm Chemical
Reagent Co., Ltd.), and observed under a fluorescence microscope
(magnification, ×400; Olympus Corporation) (49). ROS production (red staining) was
quantified using Image-Pro Plus 6.0 software (Media Cybernetics,
Inc.).
Detection of catalase (CAT), SOD,
GSH-Px and MDA
Blood (5 ml) samples were collected from the
abdominal aorta. Serum was isolated via centrifugation at 1,500 × g
for 10 min at 4°C and the enzymatic activities of CAT (cat. no.
A007-1), SOD (cat. no. A001-1) and GSH-Px (cat. no. A005-1), as
well as the concentration of MDA (cat. no. A003-1) were determined
using colorimetric commercially available kits (Nanjing Jiancheng
Bioengineering Institute) according to the manufacturers
instructions.
Immunohistochemistry
Immunohistochemistry was performed according to the
methods previously described (50–52).
After the heart of the rat was removed, samples were fixed in 4%
paraformaldehyde for 24 h at room temperature and embedded in
paraffin. Then, the paraffin-embedded tissue sections (4-µm thick)
were deparaffinized, rehydrated in a descending series of ethanol
(100, 95, 90 and 80%) and immersed in retrieval solution. Sections
were incubated with 3% methanol-H2O2 for
20–30 min at 37°C to eliminate endogenous peroxidase activity.
Non-specific staining was blocked with 5% Ig blocking reagent and
5% serum (Shanghai Regal Biological Technology Development Co.,
Ltd.) for 15 min at 37°C after rinsing. Sections were incubated
with primary antibodies against IL-1 (1:100; cat. no. 16765-1-AP;
ProteinTech Group, Inc.), IL-6 (1:100; cat. no. 21865-1-AP;
ProteinTech Group, Inc.), Bax (1:100; cat. no. 50599-2-Ig;
ProteinTech Group, Inc.), TNF-α (1:100; cat. no. 60291-1-Ig;
ProteinTech Group, Inc.), Bcl-2 (1:100; cat. no. 26593-1-AP;
ProteinTech Group, Inc.) and p65 (1:100; cat. no. 10745-1-AP;
ProteinTech Group, Inc.) at 4°C overnight. The following day, the
sections were maintained at room temperature and washed with PBS
three times for 5 min each time. Next, the sections were incubated
with HRP-conjugated anti-rabbit and anti-mouse secondary antibodies
(1:200; cat. nos. PV-6001 and PV-6002; OriGene Technologies, Inc.)
at room temperature for 20 min and then washed in PBS three times
(5 min each time). The 3,3diaminobenzidine (DAB) dye solution was
added onto the sections and incubated for 5–10 sec at room
temperature, at which point the reaction was stopped using water
washes three times. Color development was induced with a DAB kit
(cat. no. ZLI-9019; OriGene Technologies, Inc.) and counterstained
with hematoxylin for 2 min at room temperature. The slides were
subsequently dehydrated with an ascending gradient ethanol series
(30, 50, 70, 80, 90, 95 and 100%) for 3 min each at room
temperature. Xylene was used to make the sections transparent and
these were then sealed. The slices were visualized using a digital
light microscope (magnification, ×400) and the resulting images
were analyzed using Image-Pro 6.0 software (Media Cybernetics,
Inc.).
Western blotting
Frozen heart tissues were removed, homogenized and
lysed in ice-cold cell lysis buffer (cat. no. 9803; Cell Signaling
Technology, Inc.). The proteins were extracted from different
issues and quantified using the BCA assay. Protein samples (50 µg)
were subjected to SDS-PAGE and transferred to PVDF membranes (EMD
Millipore) (51). The membranes
were blocked with 5% skim milk in TBS-0.1% Tween-20 (TBST) buffer
at room temperature for 1 h. The cell membranes were incubated
overnight at 4°C with primary antibody in blocking buffer,
including anti-SirT1 (cat. no. 60303-1-Ig; 1:1,000; ProteinTech
Group, Inc.) and anti-β-actin (cat. no. TA-09; 1:1,000; OriGene
Technologies, Inc.). Next, the membranes were washed three times
with TBST and then incubated with horseradish peroxidase-conjugated
secondary anti-rabbit and anti-mouse antibodies (cat. nos. 7074 and
7076; 1:5,000; Cell Signaling Technology, Inc.) for 1 h at room
temperature. Membranes were washed three times and proteins were
visualized using the ECL Detection system (TransGen Biotech Co.,
Ltd.) (53), and imaged using
Tanon-1600 Gel Image Analysis system (Tanon Science and Technology
Co., Ltd.). Bands were quantified using Tanon Gis 1D software
(Tanon Science and Technology Co., Ltd.) (49).
Statistical analysis
All data were statistically analyzed using SPSS 20.0
software (IBM Corp.) and graphs were created using Origin Pro 9.1
software (Europa Science Ltd.). A single-factor ANOVA was used for
multigroup comparisons followed by Tukeys test. Data are presented
as the mean ± SEM. Each experiment was repeated ≥3 times. P<0.05
was considered to indicate a statistically significant
difference.
Results
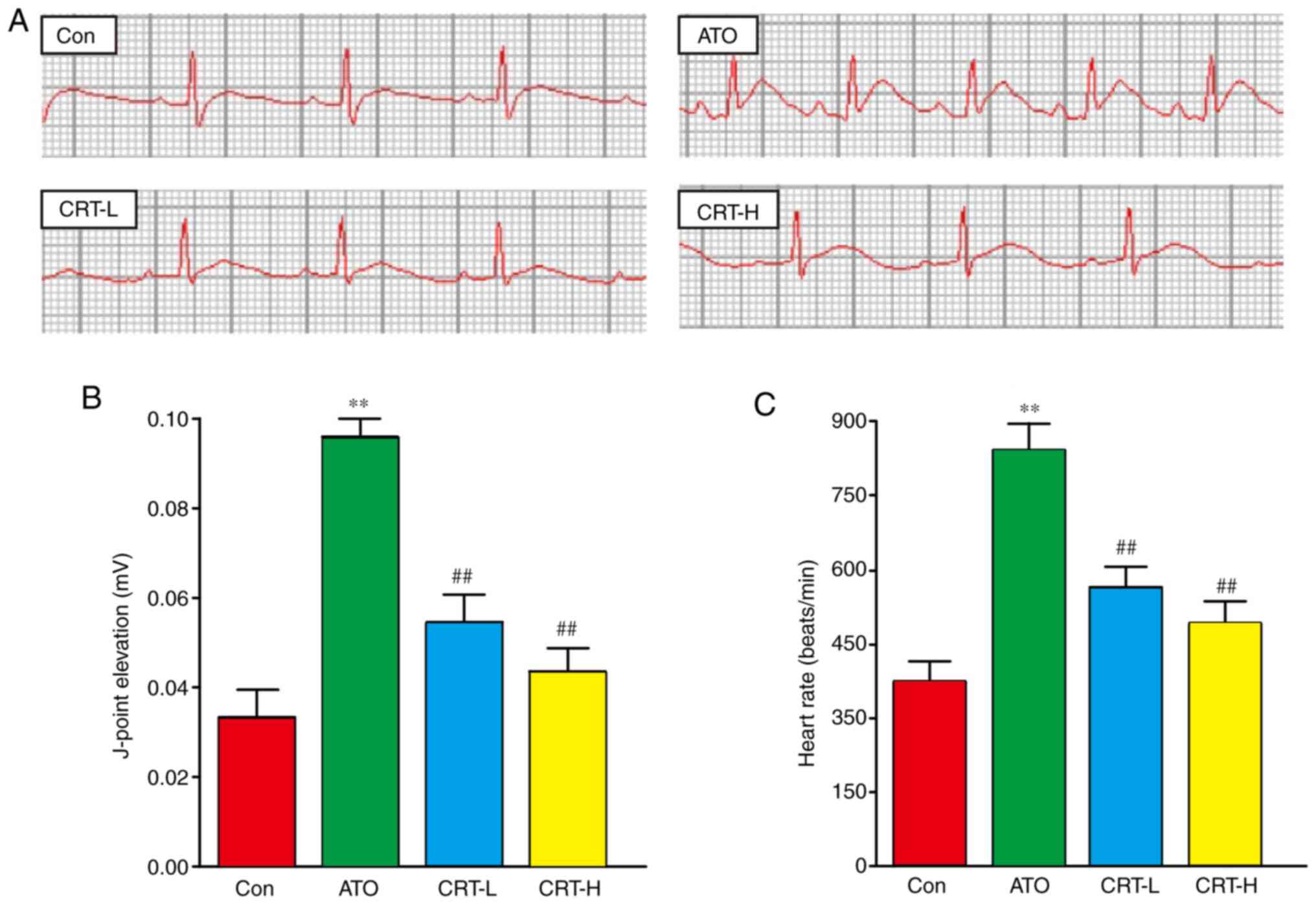
Effect of CRT on ECG
Sample tracings of ECG from the experimental animals
are presented in Fig. 2A. Compared
with the Con group, the heart rate and J-point of the rats in the
ATO group were significantly increased (P<0.01), suggesting that
the experimental myocardial injury model was successfully
established (Fig. 2B and C).
Moreover, these two indexes were significantly lower in the CRT-L
and CRT-H groups compared with the ATO group (P<0.01).
Effects of CRT on histopathology
The results of the histopathological examination of
the myocardial tissues are presented in Fig. 3. The H&E-stained heart sections
from the Con group had healthy cardiomyocyte structures. However,
the sections from the ATO-induced cardiotoxicity group contained
extensive necrosis, inflammatory cell infiltration, cytoplasmic
vacuolization, slight fiber swelling, soft interstitial edema and
myofiber loss. Compared with the Con group, the ATO caused a
significant increase in the morphological changes (P<0.01;
Fig. 3B). It was found that
treatment with CRT-L and CRT-H significantly improved these signs
of ATO-induced cardiotoxicity (P<0.01; Fig. 3B), suggesting that CRT exerted a
potent protective effect in this model of cardiac injury.
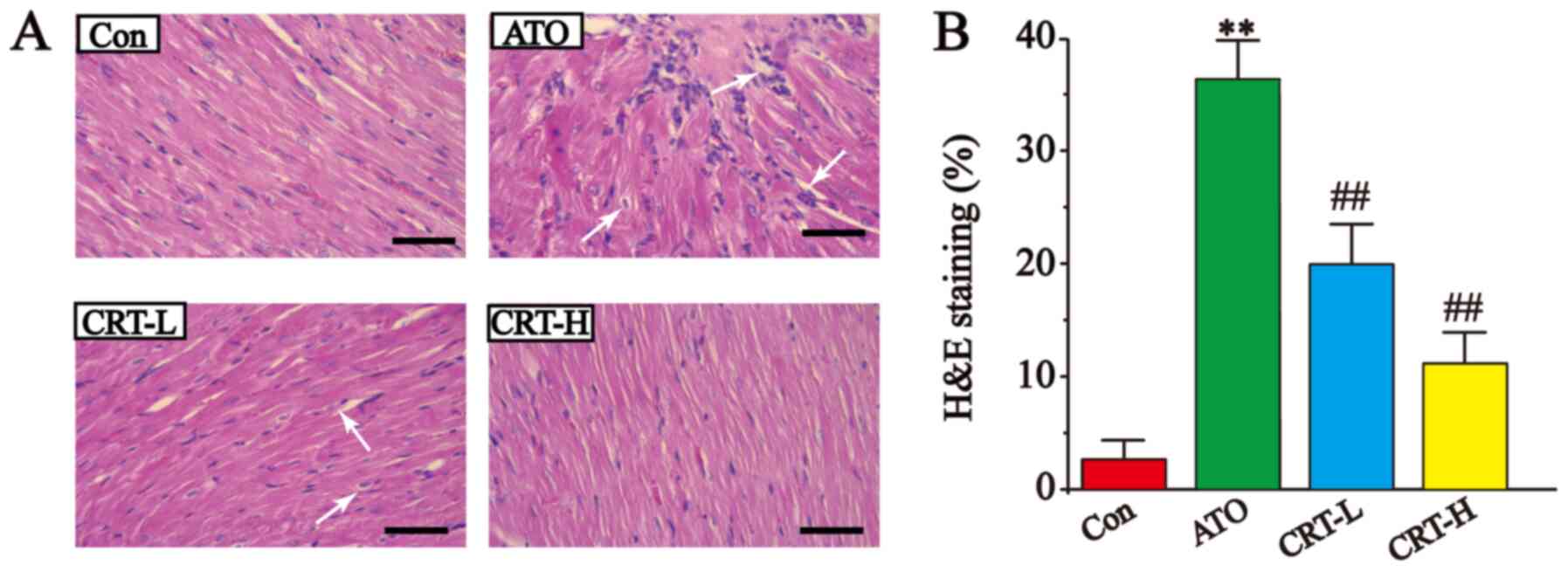 | Figure 3.Effects of CRT on H&E staining in
ATO-induced cardiotoxicity in rats. (A) Myocardial tissue obtained
from the Con, ATO, CRT-L and CRT-H groups. Histopathological
changes are indicated by white arrows. (B) Pathological damage
scores of the heart tissue in the Con, ATO, CRT-L and CRT-H groups.
Scale bar, 50 µm. Data are presented as the mean ± SEM for each
group, n=10. **P<0.01 vs. Con group; ##P<0.01 vs.
ATO group. CRT, crocetin; Con, control; ATO, arsenic trioxide;
CRT-L, ATO + low concentration of CRT treatment group; CRT-H, ATO +
high concentration of CRT treatment group; H&E, hematoxylin and
eosin. |
Effect of CRT on CK and LDH
activities
Serum CK and LDH activities were significantly
increased in the ATO group (P<0.01; Fig. 4A and B) compared with the Con
group. Relative to the ATO group, CRT treatment resulted in a
significant decrease in CK and LDH activities at 20 mg/kg/day
(P<0.01) and at 40 mg/kg/day (P<0.01).
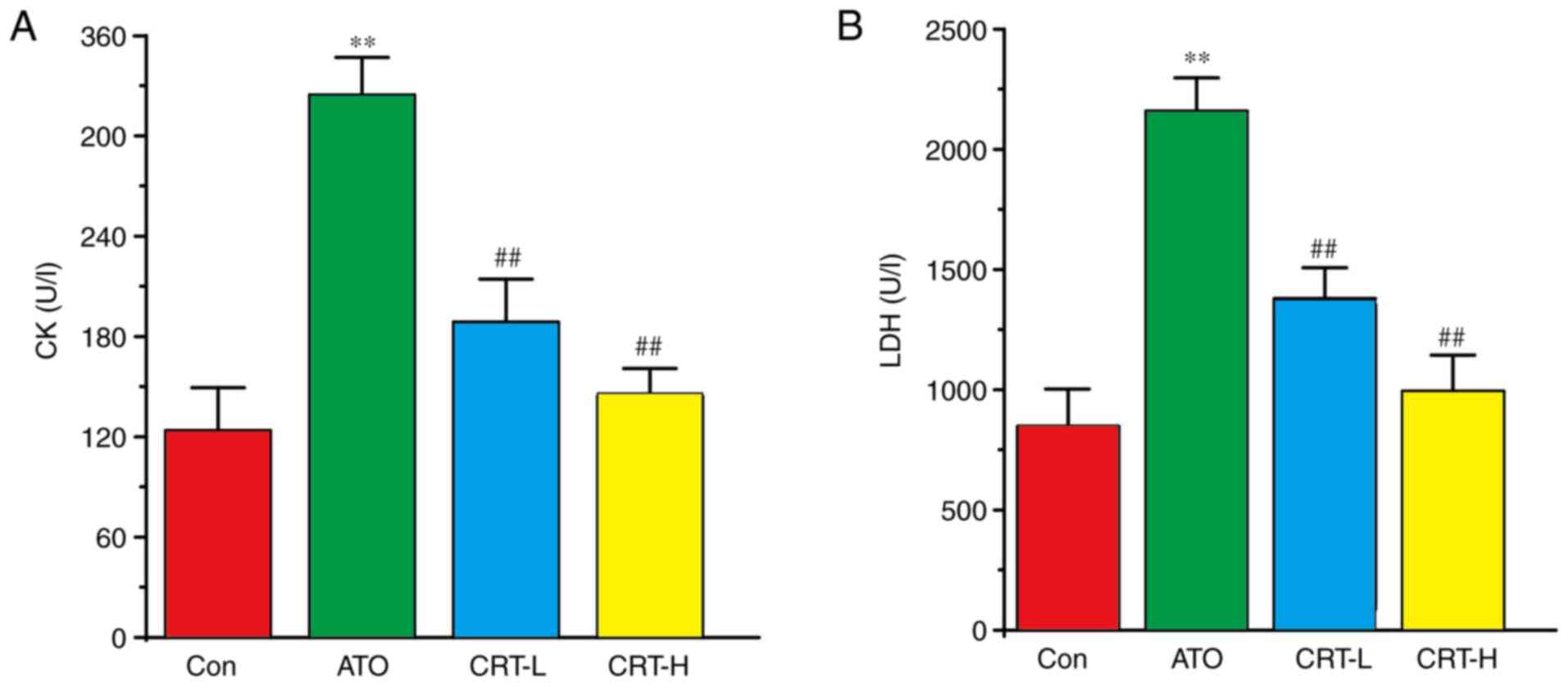 | Figure 4.Effects of CRT on CK and LDH
activities in ATO-induced cardiotoxicity in rats. Serum was
collected from the Con, ATO, CRT-L and CRT-H groups and assayed for
(A) CK and (B) LDH enzyme activities using CK and LDH kits. Data
are presented as the mean ± SEM for each group, n=10. **P<0.01
vs. Con group; ##P<0.01 vs. ATO group. CRT, crocetin;
Con, control; ATO, arsenic trioxide; CRT-L, ATO + low concentration
of CRT treatment group; CRT-H, ATO + high concentration of CRT
treatment group; CK, creatine kinase; LDH, lactate
dehydrogenase. |
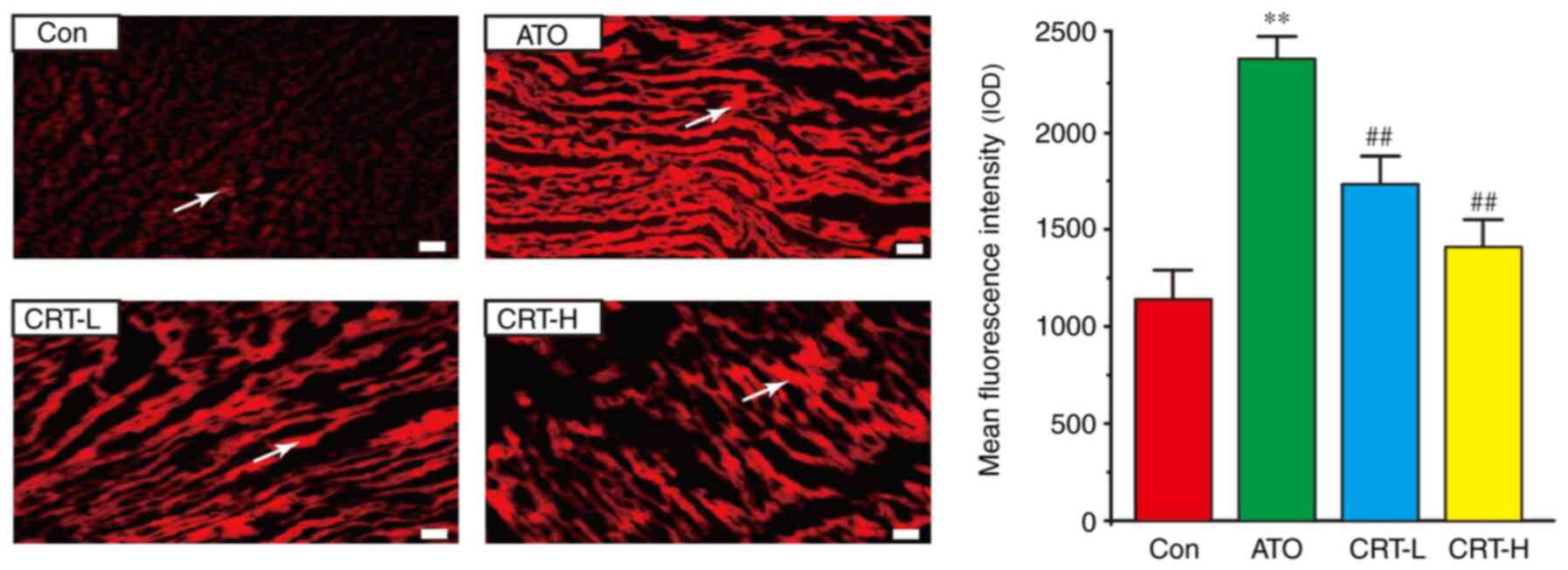
Effects of CRT on ROS release
A significant increase in ROS generation (red) was
observed in the rat hearts from the ATO group in comparison with
the Con group (Fig. 5). However,
CRT preconditioning partially eliminated these changes (P<0.01),
and ROS generation was markedly lower in the CRT-H group compared
with the CRT-L group.
Effects of CRT on SOD, GSH-Px, MDA and
CAT
Compared with the Con group, the ATO group had
increased myocardial MDA activity (P<0.01), but decreased SOD,
GSH-Px and CAT activities (P<0.01) (Fig. 6A-D). Furthermore, CRT
administration (20 and 40 mg/kg/day) led to a dose-dependent
increase in SOD, GSH-Px and CAT activities (P<0.01 or
P<0.05), along with a concomitant decrease in MDA content
compared with the ATO group (P<0.01 or P<0.05). Therefore,
CRT attenuated the oxidative stress associated with ATO-induced
cardiotoxicity in a dose-dependent manner (P<0.01).
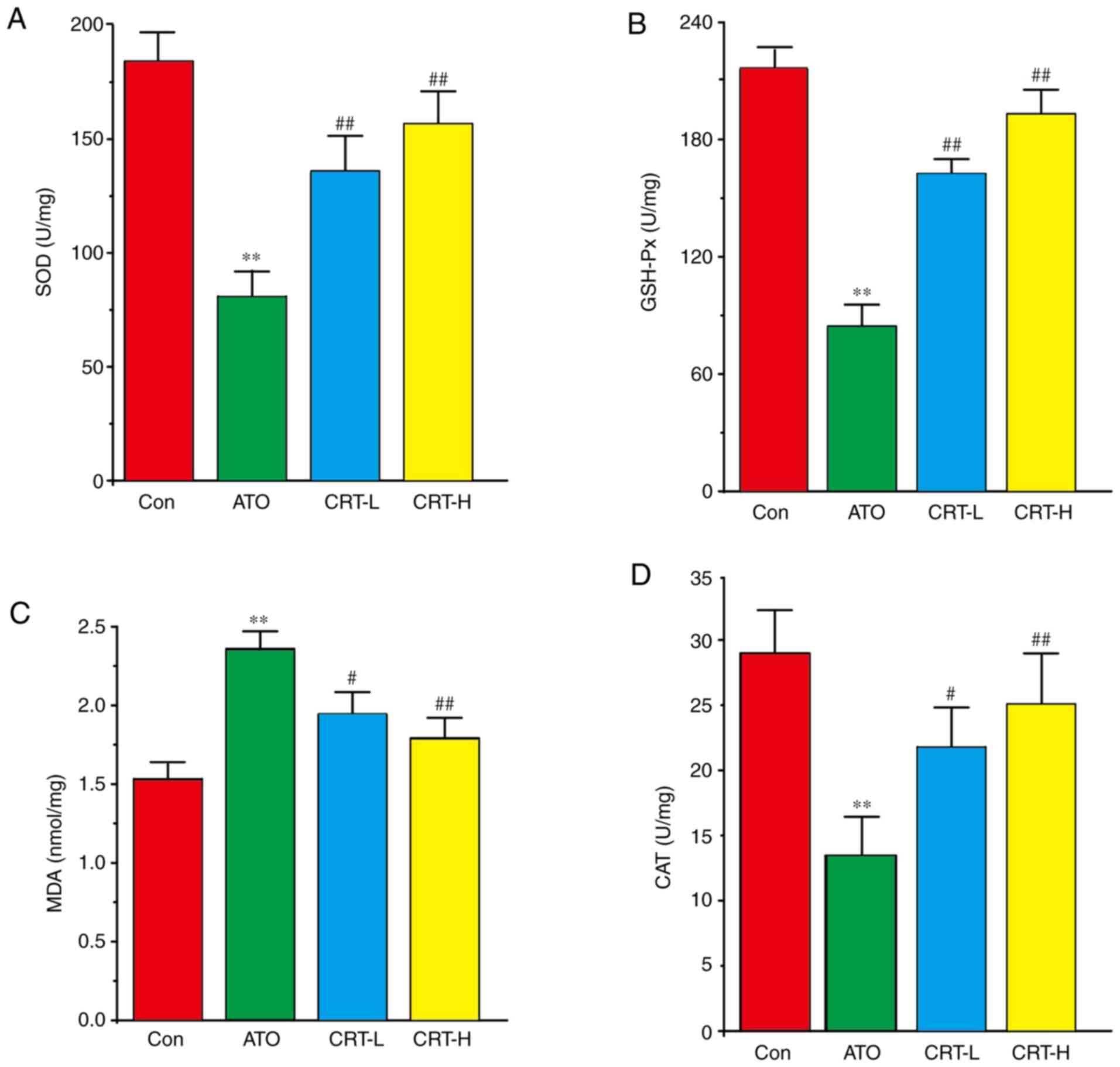 | Figure 6.Effects of CRT on SOD, GSH-Px, MDA
and CAT levels in ATO-induced cardiotoxicity in rats. Serum was
analyzed for (A) SOD, (B) GSH-Px, (C) MDA and (D) CAT in the Con,
ATO, CRT-L and CRT-H groups. Data are presented as the mean ± SEM
for each group, n=10. **P<0.01 vs. Con group;
#P<0.05, ##P<0.01 vs. ATO group. CRT,
crocetin; Con, control; ATO, arsenic trioxide; CRT-L, ATO + low
concentration of CRT treatment group; CRT-H, ATO + high
concentration of CRT treatment group; SOD, superoxide dismutase;
GSH-Px, glutathione peroxidase; MDA, malondialdehyde; CAT,
catalase. |
Effects of CRT on IL-1, IL-6 and TNF-α
expression levels
The expression levels of IL-1, IL-6 and TNF-α in
cardiac tissue were assessed using immunohistochemistry (Fig. 7A-F). IL-1, IL-6, and TNF-α
expression was low in the Con group (Fig. 7A-F) and increased after ATO
treatment (P<0.01). The immunoreactive area was also more
widespread in the ATO group compared with the Con group
(P<0.01). CRT treatment (20 and 40 mg/kg) significantly
prevented the large increased expression of IL-1, IL-6, and TNF-α
and restricted their prevalence. These findings demonstrated that
CRT attenuated the expression levels of IL-1, IL-6 and TNF-α in a
dose-dependent manner.
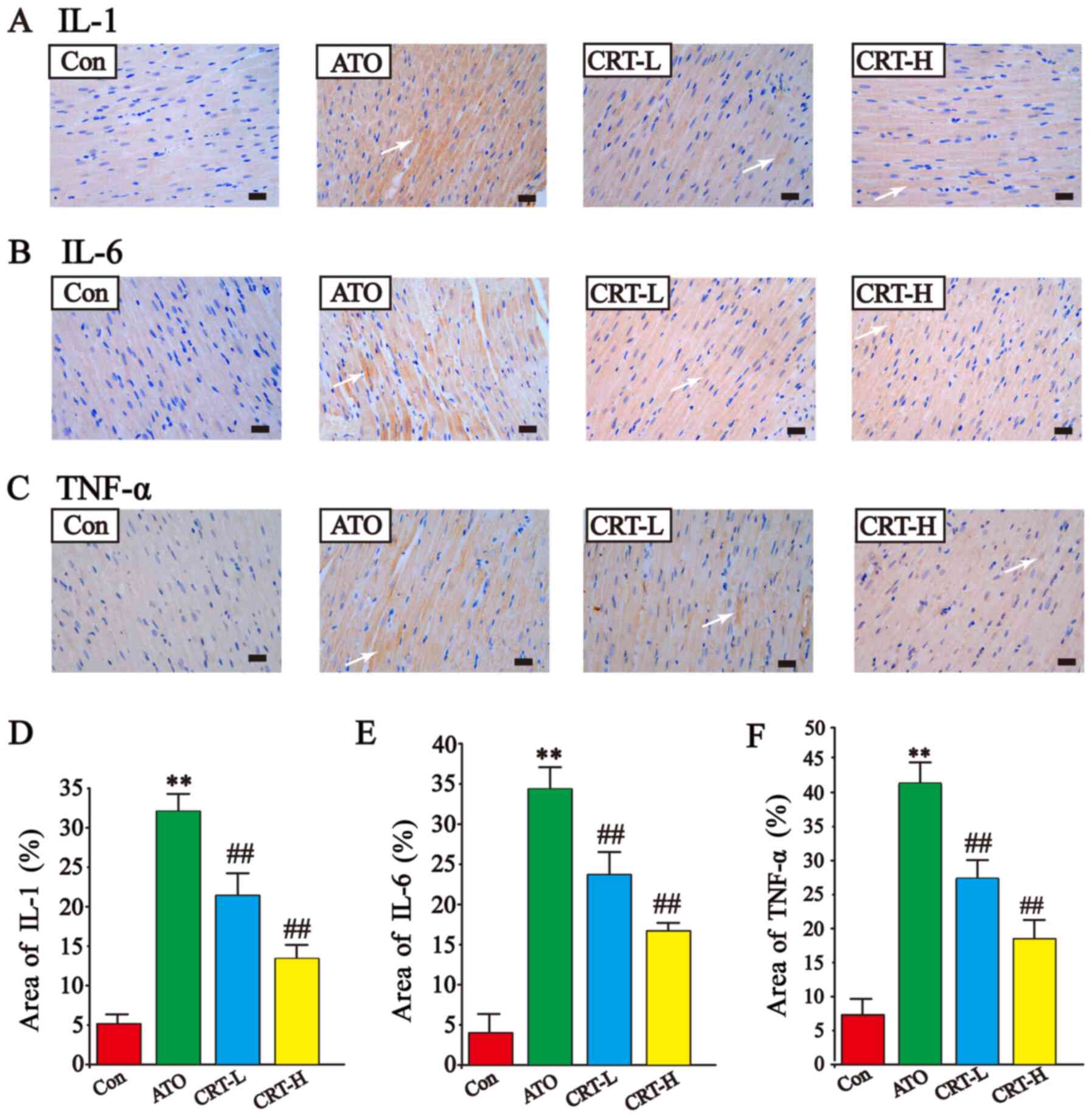 | Figure 7.Effects of CRT on the expression
levels of IL-1, IL-6 and TNF-α in ATO-treated rats detected via
immunohistochemistry. Anatomical location of (A) IL-1 expression,
(B) IL-6 expression and (C) TNF-α expression. Immunoreactive IL-1,
IL-6 and TNF-α areas are indicated by white arrows. Quantification
of the % area of (D) IL-1 expression, (E) IL-6 expression and (F)
TNF-α expression. Scale bar, 50 µm; magnification, ×400. Data are
presented as the mean ± SEM for each group, n=10. **P<0.01 vs.
Con group; ##P<0.01 vs. ATO group. CRT, crocetin;
Con, control; ATO, arsenic trioxide; CRT-L, ATO + low concentration
of CRT treatment group; CRT-H, ATO + high concentration of CRT
treatment group. |
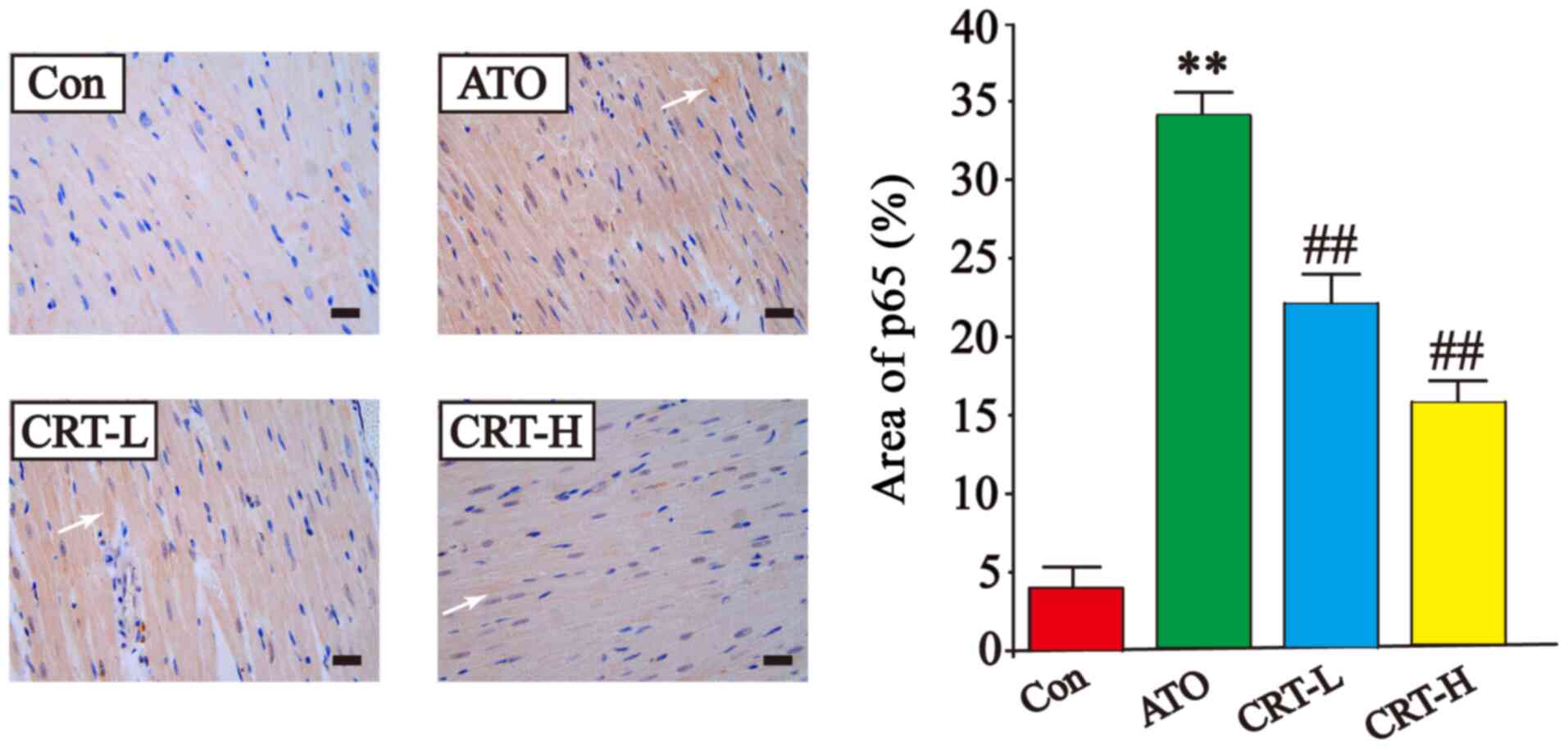
Effects of CRT on p65 expression
The expression of p65 was higher in the ATO group
compared with the Con group (P<0.01), while treatment with CRT
decreased p65 expression compared with the ATO group (P<0.01).
These results demonstrated that CRT treatment could prevent the
cascade of intracellular signaling (Fig. 8).
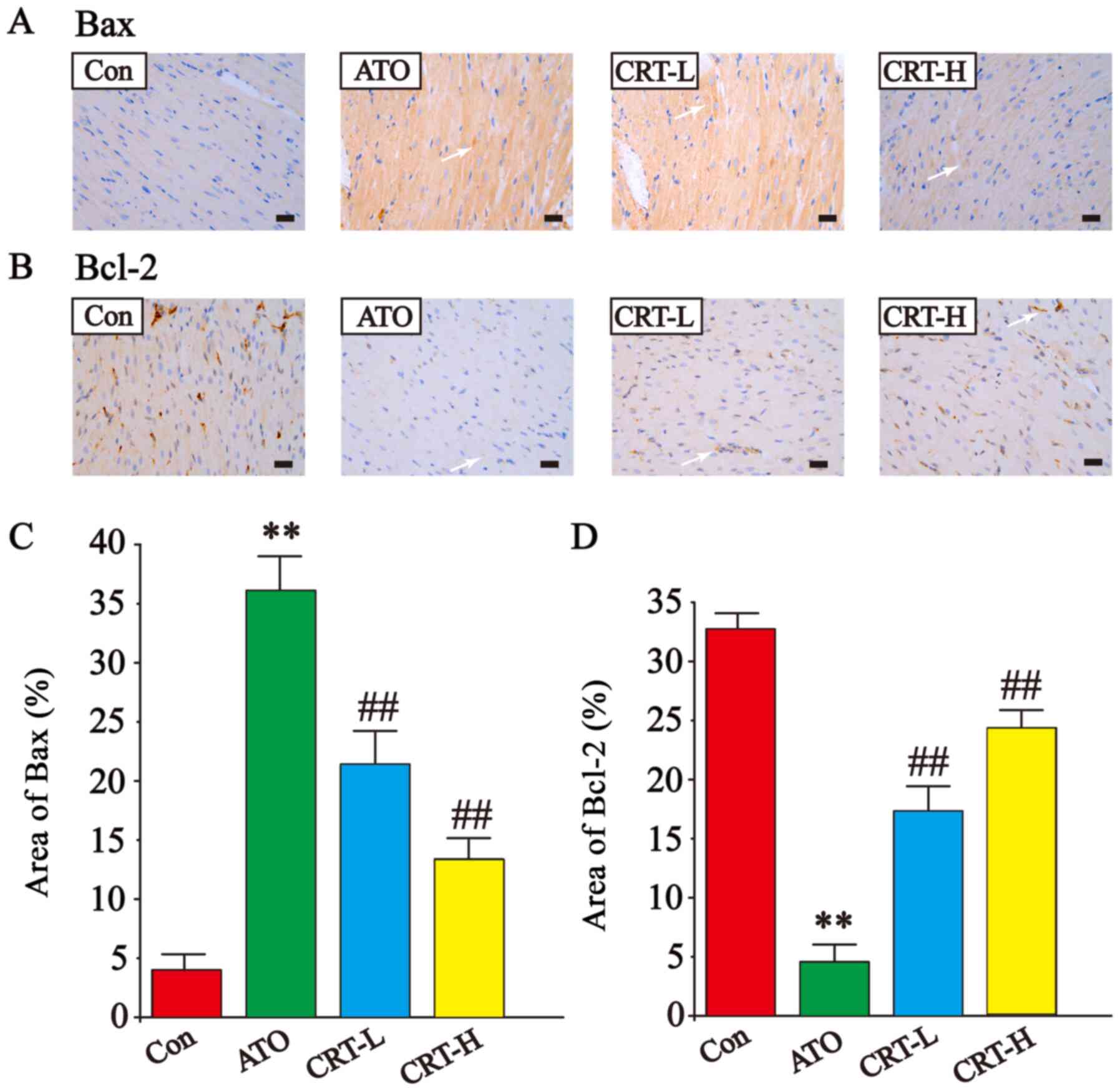
Effects of CRT on Bax and Bcl-2
expression
The expression levels of Bax and Bcl-2 were also
detected via immunohistochemistry of cardiac tissue (Fig. 9A-D). The Bax-immunoreactive area
(Fig. 9A) was increased and
Bcl-2-immunoreactive area (Fig.
9B) was decreased in ATO-treated sections compared with Con
group (P<0.01; Fig. 9C and D).
However, treatment with CRT (20 and 40 mg/kg) suppressed Bax
upregulation (P<0.01) and increased Bcl-2 expression (P<0.01)
compared with the ATO group. Thus, it was indicated that CRT
reduced the expression of Bax and increased Bcl-2 expression.
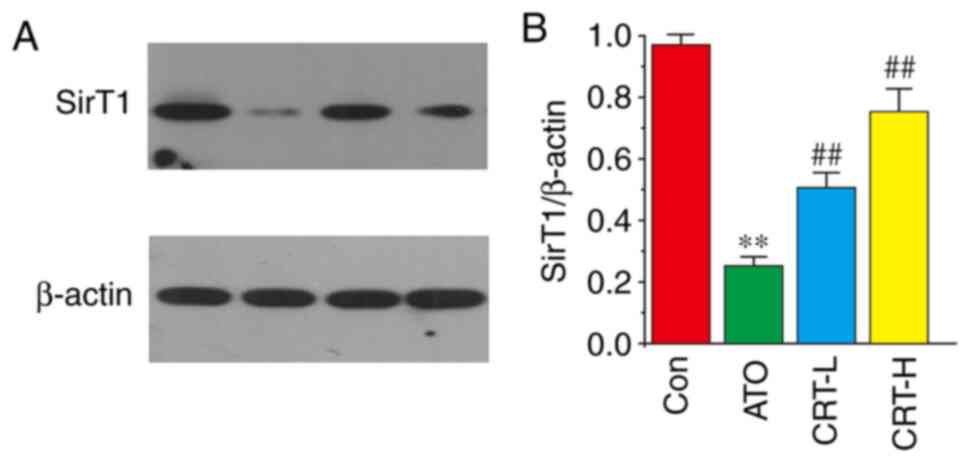
Effects of CRT on SirT1
expression
The effects of CRT on SirT1 protein expression was
evaluated via western blot analysis (Fig. 10A). The ATO group had decreased
SirT1 expression compared with the Con group (P<0.01; Fig. 10B). Furthermore, SirT1 expression
was significantly increased in the CRT-L and CRT-H groups relative
to the ATO group. It was demonstrated that CRT treatment caused
dose-dependent increases in SirT1 expression. Collectively, these
results indicated that SirT1 may be involved in ATO-induced
cardiotoxicity and that the protective effects of CRT could be
mediated by SirT1.
Discussion
ATO has a beneficial effect on recurrent and
refractory APL (54). However, ATO
causes cardiotoxicity, characterized by a drop in antioxidant
enzymes that serve as makers of ATO-induced myocardial injury and
necrosis (7). Moreover, oxidative
insults occur in the hearts of SD rats due to increased formation
of ROS, leading to severe histological alterations, including
cardiomyocyte necrosis and myocardial hemorrhage (55). In the initiation and succession of
ATO-induced myocardial injury, the overproduction of ROS and
subsequent oxidative stress exerts a crucial role (3). However, the cardiovascular effects of
CRT dampen oxidative stress and inhibit the development of insulin
resistance, atherosclerosis, hypertension, cardiac hypertrophy and
other related diseases (56). The
present results suggested that CRT attenuates ATO-induced
cardiotoxicity and reduces arsenic accumulation.
CRT is a short carbon chain carotenoid
(apocarotenoid of C20) with a carboxyl group at each end
(Fig. 1). CRT has a potential
therapeutic benefit, including its cardioprotective,
hepatoprotective, neuroprotective, memory-enhancing,
anti-inflammatory, anti-depressant and anxiolytic effects (57,58).
Pharmacokinetic studies have reported that after oral
administration, CRT is rapidly absorbed by intestinal cells via
passive diffusion (29,59–61)
and is then distributed into the liver, heart, lungs, kidneys,
spleen, adipose tissue and central nervous system (59,61).
This absorbed CRT is present in an intact form, and as
monoglucuronide and diglucuronide conjugates in plasma (61), and the half-life of CRT is 2.5–2.9
h (62). No serious adverse events
have observed in concentrations up to 40 mg/kg of orally
administered CRT in rat pharmacokinetic experiments (62). The doses used in the present study
are based on previous experiments (62,63),
and no toxic side effects were evident in the current
experiments.
ECG is a sensitive method for detecting cardiac
abnormalities and identifying altered cardiac electrical activity
(64). An increase in J-point and
heart rate was observed in ATO-treated rats in the present study,
which may be due to ATO-induced myocardial injury; the increase in
J-point and heart rate was decreased in the CRT group, indicative
of its protective effect.
In the present study, ATO treatment caused CK and
LDH activities to increase, which is indicative of disrupted
cardiomyocyte structure and damaged cell membranes (64,65).
Thus, the present data suggested that CRT treatment (20 and 40
mg/kg) preserved the integrity and functionality of
cardiomyocytes.
Arsenic cardiotoxicity affects several physiological
processes including cardiac repolarization, intracellular
Ca2+ overload, increased Ca2+ currents and
elevated intracellular ROS (66,67).
Exposure to inorganic arsenic leads to cellular oxidative stress by
generating ROS (68).
Cardiovascular diseases such as ischemia-reperfusion injury,
endothelial dysfunction and atherosclerosis are associated with the
release of intracellular ROS (69,70).
In the present study, the effects of cardiovascular treatments were
assessed in rats using an ATO-induced cardiotoxicity model.
Accumulating evidence suggests that oxidative stress
serves an important role in ATO-induced cardiac damage (71). MDA, a typical product of LPO, is
formed by the oxidation of polyunsaturated fatty acids caused by
ROS and serves as a directional biomarker of oxidative stress that
indirectly reflects the degree of cardiomyocyte damage (72). In the present study, ATO raised MDA
levels in the myocardial tissue, while CRT protected against
arsenic cardiotoxicity, possibly by inhibiting LPO in cardiac
tissue. GSH-Px is the major endogenous antioxidative in the body,
and its decrease can cause significant damage to cells (73,74).
SOD and CAT are antioxidant enzymes whose reinforced activity
inhibits oxidative stress and delays the succession of the
ATO-induced cardiotoxicity mediated decline in arsenic accumulation
(73). Furthermore, CAT, SOD and
GSH-Px serve a key role in the regulation of and response to
intracellular oxidation (75). In
the present study, treatment with CRT increased the levels of CAT,
SOD and GSH-Px in myocardial tissue. Thus, it was suggested that
CRT relieved ATO-induced oxidative stress, indicated by increased
CAT, SOD and GSH-Px activities and simultaneously decreased MDA
levels.
In the present study, the mechanism underlying the
myocardial protection effect of CRT was investigated by analyzing
the expression levels of pro-inflammatory markers. ATO induced a
significant increase in myocardial TNF-α expression, which was
larger compared with the increase observed in IL-1 and IL-6
expression levels. Moreover, ATO induced an increase in p65
expression. Therefore, the anti-inflammatory activity of CRT was
demonstrated by its ability to reduce IL-1, IL-6, TNF-α and p65
expression levels, which is consistent with previous studies that
reported elevated Bax and reduced Bcl-2 expression after ATO
treatment (76–78).
The increased expression of Bax in the ATO group and
simultaneous decrease in Bcl-2 expression indicated that apoptosis
occurred in the current rat model of cardiotoxicity. Furthermore,
CRT decreased the number of apoptotic cells, illustrated here as
changes to these apoptosis-related transcripts. These findings are
consistent with another previous study (79).
In the present study, western blotting results
identified that SirT1 protein expression was decreased in
ATO-induced myocardial tissue. However, this lower expression was
increased after CRT treatment, which concurs with the results of
previous studies, whereby cardiotoxicity was ameliorated through
increasing the SirT1 protein expression levels (80,81).
Taken together, the present results suggested that treatment with
CRT enhanced SirT1 activity, thus counteracting the cardiomyocyte
apoptosis caused by ATO.
Several previous studies have demonstrated that ATO
could induce morphological changes in the myocardium, such as
necrosis, swelling and edema (55,82).
The morphological alterations to the myocardium of ATO-treated rats
were significantly ameliorated by CRT administration. The present
histopathological observations provided evidence of the protective
effect of CRT against ATO-induced cardiomyocyte necrosis. It was
identified that CRT pretreatment prior to ATO administration
efficiently prevented histopathological alterations, which would be
particularly beneficial against chemotherapy-induced
cardiotoxicity.
In conclusion, the present results suggested that
CRT protected cardiomyocytes from ATO-induced cardiotoxicity. The
cardioprotective effect of CRT is attributed to the reduction of
myocardial oxidative stress, inhibition of inflammation and
suppression of apoptosis. Collectively, the present results
indicated that pretreatment with CRT ameliorated ATO-induced
myocardial damage. Therefore, CRT may have extensive clinical
application value in treating ATO-induced cardiotoxicity.
Acknowledgements
Not applicable.
Funding
The present stuy was supported by the Research
Foundation of Administration of Traditional Chinese Medicine of
Hebei Province, China (grant no. 2020188).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors contributions
XC, YG, LC and ZZ designed the study and conducted
the experiments. BZ, JL and JS performed the formal experiment and
acquired the data. BZ and YL analyzed the data. ZZ, XH and JZ
interpreted the data. ZZ and JL drafted the manuscript. JL, LC and
XH revised the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
All animal experiments were approved by the
Institutional Animal Experimental Ethics Committee of Hebei
University of Chinese Medicine. All experimental procedures
complied with the protocols and ethical regulations provided by the
Institutional Animal Experimental Ethics Committee of Hebei
University of Chinese Medicine.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ATO
|
arsenic trioxide
|
|
CAT
|
catalase
|
|
CK
|
creatine kinase
|
|
Con
|
control group
|
|
CRT
|
crocetin
|
|
CRT-L
|
ATO + low concentration of CRT
treatment group
|
|
CRT-H
|
ATO + high concentration of CRT
treatment group
|
|
ECG
|
electrocardiogram
|
|
GSH-Px
|
glutathione peroxidase
|
|
H&E
|
hematoxylin and eosin
|
|
LDH
|
lactate dehydrogenase
|
|
MDA
|
malondialdehyde
|
|
LPO
|
lipid peroxidation
|
|
ROS
|
reactive oxygen species
|
|
SirT1
|
silent information regulator of
transcription 1
|
|
SOD
|
superoxide dismutase
|
References
|
1
|
Rao Y, Li R and Zhang D: A drug from
poison: How the therapeutic effect of arsenic trioxide on acute
promyelocytic leukemia was discovered. Sci China Life Sci.
56:495–502. 2013.PubMed/NCBI
|
|
2
|
Sfaxi I, Charradi K, Limam F, El May MV
and Aouani E: Grape seed and skin extract protects against arsenic
trioxide induced oxidative stress in rat heart. Can J Physiol
Pharmacol. 94:168–176. 2016.PubMed/NCBI
|
|
3
|
Yu X, Wang Z, Shu Z, Li Z, Ning Y, Yun K,
Bai H, Liu R and Liu W: Effect and mechanism of Sorbus
pohuashanensis (Hante) Hedl. Flavonoids protect against arsenic
trioxide-induced cardiotoxicity. Biomed Pharmacother. 88:1–10.
2017.PubMed/NCBI
|
|
4
|
Raghu KG and Cherian OL: Characterization
of cytotoxicity induced by arsenic trioxide (a potent anti-APL
drug) in rat cardiac myocytes. J Trace Elem Med Biol. 23:61–68.
2009.PubMed/NCBI
|
|
5
|
Varghese MV, Abhilash M, Paul MV, Alex M
and Nair RH: Omega-3 fatty acid protects against arsenic
trioxide-induced cardiotoxicity in vitro and in vivo. Cardiovasc
Toxicol. 17:109–119. 2017.PubMed/NCBI
|
|
6
|
Manna P, Sinha M and Sil PC:
Arsenic-induced oxidative myocardial injury: Protective role of
arjunolic acid. Arch Toxicol. 82:137–149. 2008.PubMed/NCBI
|
|
7
|
Zhang W, Guo C, Gao R, Ge M, Zhu Y and
Zhang Z: The protective role of resveratrol against arsenic
trioxide-induced cardiotoxicity. Evid Based Complement Alternat
Med. 2013:4078392013.PubMed/NCBI
|
|
8
|
Abeyrathna P and Su Y: The critical role
of Akt in cardiovascular function. Vascul Pharmacol. 74:38–48.
2015.PubMed/NCBI
|
|
9
|
Wang Z, Yu J, Wu J, Qi F, Wang H, Wang Z
and Xu Z: Scutellarin protects cardiomyocyte ischemia-reperfusion
injury by reducing apoptosis and oxidative stress. Life Sci.
157:200–207. 2016.PubMed/NCBI
|
|
10
|
Galano A: Free radicals induced oxidative
stress at a molecular level: The current status, challenges and
perspectives of computational chemistry based protocols. J Mex Chem
Soc. 59:231–262. 2015.
|
|
11
|
Zhou SS, He F, Chen AH, Hao PY and Song
XD: Suppression of rat frizzled-2 attenuates
hypoxia/reoxygenation-induced Ca2+ accumulation in rat
H9c2 cells. Exp Cell Res. 318:1480–1491. 2012.PubMed/NCBI
|
|
12
|
Andrades ME, Ritter C and Dal-Pizzol F:
The role of free radicals in sepsis development. Front Biosci
(Elite Ed). 1:277–287. 2009.PubMed/NCBI
|
|
13
|
Baird AM, OByrne KJ and Gray SG: Reactive
oxygen species and reactive nitrogen species in epigenetic
modifications. Biochem Soc Trans. 437–455. 2012.
|
|
14
|
Edson KZ and Rettie AE: CYP4
enzymes as potential drug targets: Focus on enzyme multiplicity,
inducers and inhibitors, and therapeutic modulation of
20-hydroxyeicosatetraenoic acid (20-HETE) synthase and fatty acid
ω-hydroxylase activities. Curr Top Med Chem. 13:1429–1440.
2013.PubMed/NCBI
|
|
15
|
Nakayama H, Chen X, Baines CP, Klevitsky
R, Zhang X, Zhang H, Jaleel N, Chua BH, Hewett TE, Robbins J, et
al: Ca2+- and Mitochondrial-dependent cardiomyocyte
necrosis as a primary mediator of heart failure. J Clin Invest.
117:2431–2444. 2007.PubMed/NCBI
|
|
16
|
Zhang J, Zhang Y, Wang W, Li C and Zhang
Z: Double-sided personality: Effects of arsenic trioxide on
inflammation. Inflammation. 41:1128–1134. 2018.PubMed/NCBI
|
|
17
|
Gasparini C and Feldmann M: NF-κB as a
target for modulating inflammatory responses. Curr Pharm Des.
18:5735–5745. 2012.PubMed/NCBI
|
|
18
|
Li S, Wang Y, Zhao H, He Y and Xing M:
NF-κB-mediated inflammation correlates with calcium overload under
arsenic trioxide-induced myocardial damage in Gallus gallus.
Chemosphere. 185:618–627. 2017.PubMed/NCBI
|
|
19
|
Saad SY, Alkharfy KM and Arafah MM:
Cardiotoxic effects of arsenic trioxide/imatinib mesilate
combination in rats. J Pharm Pharmacol. 58:567–573. 2006.PubMed/NCBI
|
|
20
|
Sepand MR, Razavi-Azarkhiavi K, Omidi A,
Zirak MR, Sabzevari S, Kazemi AR and Sabzevari O: Effect of
acetyl-l-carnitine on antioxidant status, lipid peroxidation, and
oxidative damage of arsenic in rat. Biol Trace Elem Res.
171:107–115. 2016.PubMed/NCBI
|
|
21
|
Miao X, Tang Z, Wang Y, Su G, Sun W, Wei
W, Li W, Miao L, Cai L, Tan Y and Liu Q: Metallothionein prevention
of arsenic trioxide-induced cardiac cell death is associated with
its inhibition of Mitogen-activated protein kinases activation in
vitro and in vivo. Toxicol Lett. 220:277–285. 2013.PubMed/NCBI
|
|
22
|
Zhao X, Feng T, Chen H, Shan H, Zhang Y,
Lu Y and Yang B: Arsenic trioxide-induced apoptosis in H9c2
cardiomyocytes: Implications in cardiotoxicity. Basic Clin
Pharmacol Toxicol. 102:419–425. 2008.PubMed/NCBI
|
|
23
|
Testai L, Piragine E, Piano I, Flori L and
Calderone V: The citrus flavonoid naringenin protects the
myocardium from ageing-dependent dysfunction: Potential role of
SIRT1. Oxid Med Cell Longev. 2020:46502072020.PubMed/NCBI
|
|
24
|
Pirola RC and Elmslie RG: Exchange
transfusion and liver perfusion in the treatment of acute hepatic
coma. Med J Aust. 1:891–894. 1968.PubMed/NCBI
|
|
25
|
Mu W, Zhang Q, Tang X, Fu W, Zheng W, Lu
Y, Li H, Wei Y, Li L, She Z, et al: Overexpression of a
dominant-negative mutant of SIRT1 in mouse heart causes
cardiomyocyte apoptosis and Early-onset heart failure. Sci China
Life Sci. 57:915–924. 2014.PubMed/NCBI
|
|
26
|
Zhang W, Li Y and Ge Z: Cardiaprotective
effect of crocetin by attenuating apoptosis in isoproterenol
induced myocardial infarction rat model. Biomed Pharmacother.
93:376–382. 2017.PubMed/NCBI
|
|
27
|
Batarseh YS, Bharate SS, Kumar V, Kumar A,
Vishwakarma RA, Bharate SB and Kaddoumi A: Crocus sativus
extract tightens the Blood-brain barrier, reduces amyloid beta load
and related toxicity in 5XFAD mice. ACS Chem Neurosci. 8:1756–1766.
2017.PubMed/NCBI
|
|
28
|
Hosseinzadeh H, Sadeghnia HR, Ziaee T and
Danaee A: Protective effect of aqueous saffron extract (Crocus
sativus L.) and crocin, its active constituent, on renal
ischemia-reperfusion-induced oxidative damage in rats. J Pharm
Pharm Sci. 8:387–393. 2005.PubMed/NCBI
|
|
29
|
Umigai N, Murakami K, Ulit MV, Antonio LS,
Shirotori M, Morikawa H and Nakano T: The pharmacokinetic profile
of crocetin in healthy adult human volunteers after a single oral
administration. Phytomedicine. 18:575–578. 2011.PubMed/NCBI
|
|
30
|
Gao K, Liu F, Chen X, Chen M, Deng Q, Zou
X and Guo H: Crocetin protects against fulminant hepatic failure
induced by lipopolysaccharide/D-galactosamine by decreasing
apoptosis, inflammation and oxidative stress in a rat model. Exp
Ther Med. 18:3775–3782. 2019.PubMed/NCBI
|
|
31
|
Bolhassani A, Khavari A and Bathaie SZ:
Saffron and natural carotenoids: Biochemical activities and
anti-tumor effects. Biochim Biophys Acta. 1845:20–30.
2014.PubMed/NCBI
|
|
32
|
Shen XC and Qian ZY: Effects of crocetin
on antioxidant enzymatic activities in cardiac hypertrophy induced
by norepinephrine in rats. Pharmazie. 61:348–352. 2006.PubMed/NCBI
|
|
33
|
Yang L, Qian Z, Ji H, Yang R, Wang Y, Xi
L, Sheng L, Zhao B and Zhang X: Inhibitory effect on protein kinase
Ctheta by Crocetin attenuates palmitate-induced insulin
insensitivity in 3T3-L1 adipocytes. Eur J Pharmacol. 642:47–55.
2010.PubMed/NCBI
|
|
34
|
Diao SL, Sun JW, Ma BX, Li XM and Wang D:
Influence of crocetin on high-cholesterol diet induced
atherosclerosis in rats via anti-oxidant activity together with
inhibition of inflammatory response and p38 MAPK signaling pathway.
Saudi J Biol Sci. 25:493–499. 2018.PubMed/NCBI
|
|
35
|
Mancini A, Serrano-Díaz J, Nava E,
DAlessandro AM, Alonso GL, Carmona M and Llorens S: Crocetin, a
carotenoid derived from saffron (Crocus sativus L.),
improves acetylcholine-induced vascular relaxation in hypertension.
J Vasc Res. 51:393–404. 2014.PubMed/NCBI
|
|
36
|
Cai J, Yi FF, Bian ZY, Shen DF, Yang L,
Yan L, Tang QZ, Yang XC and Li H: Crocetin protects against cardiac
hypertrophy by blocking MEK-ERK1/2 signalling pathway. J Cell Mol
Med. 13:909–925. 2009.PubMed/NCBI
|
|
37
|
Shen XC and Qian ZY: Effect of crocetin on
cardiac hypertrophy induced by overloading pressure in rats. Acta
Pharm Sin. 39:172–175. 2004.
|
|
38
|
Li CY, Huang WF, Wang QL, Wang F, Cai E,
Hu B, Du JC, Wang J, Chen R, Cai XJ, et al: Crocetin induces
cytotoxicity in colon cancer cells via p53-independent mechanisms.
Asian Pac J Cancer Prev. 13:3757–3761. 2012.PubMed/NCBI
|
|
39
|
Kim SH, Lee JM, Kim SC, Park CB and Lee
PC: Proposed cytotoxic mechanisms of the saffron carotenoids crocin
and crocetin on cancer cell lines. Biochem Cell Biol. 92:105–111.
2014.PubMed/NCBI
|
|
40
|
Yang M, Mao G, Ouyang L, Shi C, Hu P and
Huang S: Crocetin alleviates myocardial ischemia/reperfusion injury
by regulating inflammation and the unfolded protein response. Mol
Med Rep. 21:641–648. 2020.PubMed/NCBI
|
|
41
|
Zhang D: Comparison of the effect of
different anesthetics in five hundred rats. Lab Anim Sci. 1:19–20.
2007.
|
|
42
|
Zhao H, Haitao LI, Dingwei GU, Suxin LI
and Fang Y: Effects of oral spironolactone on TNF-α expression of
myocardial tissues in spontaneous hypertension rats. Tianjin Med J.
40:64–66. 2012.
|
|
43
|
Wang T, Sun X, Cui H, Liu K and Zhao J:
The peptide compound urantide regulates collagen metabolism in
atherosclerotic rat hearts and inhibits the JAK2/STAT3 pathway. Mol
Med Rep. 21:1097–1106. 2020.PubMed/NCBI
|
|
44
|
Knuckles TL, Buntz JG, Paffett M, Channell
M, Harmon M, Cherng T, Lucas SN, McDonald JD, Kanagy NL and Campen
MJ: Formation of vascular s-nitrosothiols and plasma
nitrates/nitrites following inhalation of diesel emissions. J
Toxicol Environ Health A. 74:828–837. 2011.PubMed/NCBI
|
|
45
|
Song Q, Chu X, Zhang X, Bao Y, Zhang Y,
Guo H, Liu Y, Liu H, Zhang J, Zhang Y and Chu L: Mechanisms
underlying the cardioprotective effect of Salvianic acid A against
isoproterenol-induced myocardial ischemia injury in rats: Possible
involvement of L-type calcium channels and myocardial
contractility. J Ethnopharmacol. 189:157–164. 2016.PubMed/NCBI
|
|
46
|
Han X, Li M, Zhao Z, Zhang Y, Zhang J,
Zhang X, Zhang Y, Guan S and Chu L: Mechanisms underlying the
cardio-protection of total ginsenosides against myocardial ischemia
in rats in vivo and in vitro: Possible involvement of L-type
Ca2+ channels, contractility and Ca2+
homeostasis. J Pharmacol Sci. 139:240–248. 2019.PubMed/NCBI
|
|
47
|
Lee T, Chen C and Chang N: Cardiac
sympathetic hyperinnervation in deoxycorticosterone acetate-salt
hypertensive rats. Clin Sci (Lond). 123:445–457. 2012.PubMed/NCBI
|
|
48
|
Li R, Liu Y, Xie J, Huang X, Zhang L, Liu
H and Li L: Sirt3 mediates the protective effect of hydrogen in
inhibiting ROS-induced retinal senescence. Free Radic Biol Med.
135:116–124. 2019.PubMed/NCBI
|
|
49
|
Jin W, Xue Y, Xue Y, Han X, Song Q, Zhang
J, Li Z, Cheng J, Guan S, Sun S and Chu L: Tannic acid ameliorates
arsenic Trioxide-induced nephrotoxicity, contribution of NF-κB and
Nrf2 pathways. Biomed. Pharmacother. 126:1100472020.
|
|
50
|
Meister AL, Doheny KK and Travagli RA:
Necrotizing enterocolitis attenuates developmental heart rate
variability increases in newborn rats. Neurogastroenterol Motil.
31:e134842019.PubMed/NCBI
|
|
51
|
Jin W, Zhang Y, Xue Y, Han X, Zhang X, Ma
Z, Sun S, Chu X, Cheng J, Guan S, et al: Crocin attenuates
isoprenaline-induced myocardial fibrosis by targeting TLR4/NF-κB
signaling: Connecting oxidative stress, inflammation, and
apoptosis. Naunyn Schmiedebergs Arch Pharmacol. 393:13–23.
2020.PubMed/NCBI
|
|
52
|
Chu L, Li P, Song T, Han X, Zhang X, Song
Q, Liu T, Zhang Y and Zhang J: Protective effects of tannic acid on
pressure overload-induced cardiac hypertrophy and underlying
mechanisms in rats. J Pharm Pharmacol. 69:1191–1207.
2017.PubMed/NCBI
|
|
53
|
Sun Q, Jia N, Wang W, Jin H, Xu J and Hu
H: Activation of SIRT1 by curcumin blocks the neurotoxicity of
amyloid-β 25–35 in rat cortical neurons. Biochem Biophys Res
Commun. 448:89–94. 2014.PubMed/NCBI
|
|
54
|
Tallman MS: Treatment of relapsed or
refractory acute promyelocytic leukemia. Best Pract Res Clin
Haematol. 20:57–65. 2007.PubMed/NCBI
|
|
55
|
Sun TL, Liu Z, Qi ZJ, Huang YP, Gao XQ and
Zhang YY: (−)-Epigallocatechin-3-gallate (EGCG) attenuates
arsenic-induced cardiotoxicity in rats. Food Chem Toxicol.
93:102–110. 2016.PubMed/NCBI
|
|
56
|
Hashemi M and Hosseinzadeh H: A
comprehensive review on biological activities and toxicology of
crocetin. Food Chem Toxicol. 130:44–60. 2019.PubMed/NCBI
|
|
57
|
Liang X and Qian Z: Pharmacological
properties of crocetin and crocin (digentiobiosyl ester of
crocetin) from saffron. Nat Prod Commun. 1:65–75. 2006.
|
|
58
|
Hosseini A, Razavi BM and Hosseinzadeh H:
Pharmacokinetic properties of saffron and its active components.
Eur J Drug Metab Pharmacokinet. 43:383–390. 2018.PubMed/NCBI
|
|
59
|
Xi L, Qian Z, Du P and Fu J:
Pharmacokinetic properties of crocin (crocetin digentiobiose ester)
following oral administration in rats. Phytomedicine. 14:633–636.
2007.PubMed/NCBI
|
|
60
|
Asai A, Nakano T, Takahashi M and Nagao A:
Orally administered crocetin and crocins are absorbed into blood
plasma as crocetin and its glucuronide conjugates in mice. J Agric
Food Chem. 53:7302–7306. 2005.PubMed/NCBI
|
|
61
|
Zhang Y, Fei F, Zhen L, Zhu X, Wang J, Li
S, Geng J, Sun R, Yu X, Chen T, et al: Sensitive analysis and
simultaneous assessment of pharmacokinetic properties of crocin and
crocetin after oral administration in rats. J Chromatogr B Analyt
Technol Biomed Life Sci. 1044-1045:1–7. 2017.PubMed/NCBI
|
|
62
|
Farkhondeh T, Samarghandian S, Samini F
and Sanati AR: Protective effects of crocetin on depression-like
behavior induced by immobilization in rat. CNS Neurol Disord Drug
Targets. 17:361–369. 2018.PubMed/NCBI
|
|
63
|
Danesi R, Tacca MD and Soldani G:
Measurement of the SαT segment as the most reliable
electrocardiogram parameter for the assessment of
adriamycin-induced cardiotoxicity in the rat. J Pharmacol Methods.
16:251–259. 1986.PubMed/NCBI
|
|
64
|
Kumari P, Saifi MA, Khurana A and Godugu
C: Cardioprotective effects of nanoceria in a murine model of
cardiac remodeling. J Trace Elem Med Biol. 50:198–208.
2018.PubMed/NCBI
|
|
65
|
Tang Y, Wang M, Le X, Meng J, Huang L, Yu
P, Chen J and Wu P: Antioxidant and cardioprotective effects of
Danshensu (3-(3, 4-dihydroxyphenyl)-2-hydroxy-propanoic acid from
Salvia miltiorrhiza) on isoproterenol-induced myocardial
hypertrophy in rats. Phytomedicine. 18:1024–1030. 2011.PubMed/NCBI
|
|
66
|
Shan H, Zhang Y, Cai B, Chen X, Fan Y,
Yang L, Chen X, Liang H, Zhang Y, Song X, et al: Upregulation of
microRNA-1 and microRNA-133 contributes to arsenic-induced cardiac
electrical remodeling. Int J Cardiol. 167:2798–2805.
2013.PubMed/NCBI
|
|
67
|
Drolet B, Simard C and Roden DM: Unusual
effects of a QT-prolonging drug, arsenic trioxide, on cardiac
potassium currents. Circulation. 109:26–29. 2004.PubMed/NCBI
|
|
68
|
Aposhian HV and Aposhian MM: Arsenic
toxicology: Five questions. Chem Res Toxicol. 19:1–15.
2006.PubMed/NCBI
|
|
69
|
Hwang JT, Kwon DY, Park OJ and Kim MS:
Resveratrol protects ROS-induced cell death by activating AMPK in
H9c2 cardiac muscle cells. Genes Nutr. 2:323–326. 2008.PubMed/NCBI
|
|
70
|
Oudit GY, Trivieri MG, Khaper N, Husain T,
Wilson GJ, Liu P, Sole MJ and Backx PH: Taurine supplementation
reduces oxidative stress and improves cardiovascular function in an
Iron-overload murine model. Circulation. 109:1877–1885.
2004.PubMed/NCBI
|
|
71
|
Li SW, Sun X, He Y, Guo Y, Zhao HJ, Hou ZJ
and Xing MW: Assessment of arsenic trioxide in the heart of Gallus
gallus: Alterations of oxidative damage parameters, inflammatory
cytokines, and cardiac enzymes. Environ Sci Pollut Res Int.
24:5781–5790. 2017.PubMed/NCBI
|
|
72
|
Kowalczuk K and Stryjecka-Zimmer M: The
influence of oxidative stress on the level of malondialdehyde (MDA)
in different areas of the rabbit brain. Ann Univ Mariae Curie
Sklodowska Med. 57:160–164. 2002.PubMed/NCBI
|
|
73
|
Djordjevic A, Spasic S, Jovanovic-Galovic
A, Djordjevic R and Grubor-Lajsic G: Oxidative stress in diabetic
pregnancy: SOD, CAT and GSH-Px activity and lipid peroxidation
products. J Matern Fetal Neonatal Med. 16:367–372. 2004.PubMed/NCBI
|
|
74
|
Alexa ID and Jerca L: The role of
oxidative stress in the etiology of Pre-eclampsia: Changes at the
GSH and GSH-Px levels in normal pregnancy and pre-eclampsia. Rev
Med Chir Soc Med Nat Iasi. 100:131–135. 1996.(In Romanian).
PubMed/NCBI
|
|
75
|
Espinosa J, Pérez JM, López-Olvera JR,
Ráez-Bravo A, Cano-Manuel FJ, Fandos P, Soriguer RC, Granados JE
and Romero D: Evaluation of oxidant/antioxidant balance in Iberian
ibex (Capra pyrenaica) experimentally infested with
Sarcoptes scabiei. Vet Parasitol. 242:63–70. 2017.PubMed/NCBI
|
|
76
|
Yang R, Yang L, Shen X, Cheng W, Zhao B,
Ali KH, Qian Z and Ji H: Suppression of NF-κB pathway by crocetin
contributes to attenuation of lipopolysaccharide-induced acute lung
injury in mice. Eur J Pharmacol. 674:391–396. 2012.PubMed/NCBI
|
|
77
|
Wang Y, Yu W, Shi C and Hu P: Crocetin
attenuates sepsis-induced cardiac dysfunction regulation of
inflammatory response and mitochondrial function. Front Physiol.
11:5142020.PubMed/NCBI
|
|
78
|
Song L, Kang C, Sun Y, Huang WR, Liu W and
Qian ZY: Crocetin inhibits lipopolysaccharide-induced inflammatory
response in human umbilical vein endothelial cells. Cell Physiol
Biochem. 40:443–452. 2016.PubMed/NCBI
|
|
79
|
Mantawy EM, El-Bakly WM, Esmat A, Badr AM
and El-Demerdash E: Chrysin alleviates acute doxorubicin
cardiotoxicity in rats via suppression of oxidative stress,
inflammation and apoptosis. Eur J Pharmacol. 728:107–118.
2014.PubMed/NCBI
|
|
80
|
Liu MH, Shan J, Li J, Zhang Y and Lin XL:
Resveratrol inhibits Doxorubicin-induced cardiotoxicity via sirtuin
1 activation in H9c2 cardiomyocytes. Exp Ther Med. 12:1113–1118.
2016.PubMed/NCBI
|
|
81
|
Sun Z, Lu W, Lin N, Lin H, Zhang J, Ni T,
Meng L, Zhang C and Guo H: Dihydromyricetin alleviates
doxorubicin-induced cardiotoxicity by inhibiting NLRP3 inflammasome
through activation of SIRT1. Biochem Pharmacol.
175:1138882020.PubMed/NCBI
|
|
82
|
Binu P, Priya N, Abhilash S, Vineetha R
and Nair R: Studies on curative efficacy of monoterpene eugenol on
anti-leukemic drug arsenic trioxide induced cardiotoxicity. Biomed
Pharmacother. 91:559–566. 2017.PubMed/NCBI
|