Globally, colorectal cancer (CRC) is the third most
commonly diagnosed malignant tumor and is the second leading cause
of cancer-associated mortality (1).
Overall, the incidence rate and mortality rate of CRC are rising
rapidly in several low-income and middle-income countries (2). Although the mortality rate of CRC
tends to be stable or declining in developed countries, it is still
higher than that in low-income and middle-income countries
(2). By 2030, the global CRC burden
is expected to increase by 60%, reaching >2.2 million new cases
and 1.1 million mortalities (3).
The majority of newly-diagnosed CRC cases are classified as a
sporadic form (4), and the
occurrence and development of CRC is a long-term process.
Conventional CRC begins with changes in the cell morphology of the
colonic epithelium, which proliferates uncontrollably to form
benign polyps. Gradually, it develops into a highly atypical
hyperplastic advanced adenoma, which causes a loss of epithelial
structure and function to form an invasive tumor (5,6).
A number of factors contribute to the formation of
CRC. Genetic susceptibility is a major driver of early CRC
occurrence. A study has demonstrated that CRC contains ≤80
mutations, of which <15 mutations are the driving force for
tumorigenesis (7). The probability
of developing CRC is also associated with personal features and
habits, such as age, gender, race/ethnicity, chronic disease
history, dietary factors, obesity, low physical activity, smoking
and intestinal microflora (4,8,9).
Chemotherapy based on 5-fluorouracil (5-FU) has been the main
treatment method for patients with CRC since the 1950s (10–12).
More chemotherapeutic drugs, such as oxaliplatin (L-OHP),
irinotecan and capecitabine, have been developed in recent years
and the emergence of monoclonal antibodies, such as bevacizumab and
cetuximab, have greatly advanced chemotherapy for CRC (13). However, even if the current response
rate to various systemic chemotherapy regimens reaches 50%, most
patients develop resistance within 3–12 months (14,15).
Drug resistance refers to the gradual decline in the response to
drugs during the treatment of various diseases (16). Resistance to chemotherapy drugs is a
major limitation in the use of chemotherapy (17). The failure of chemotherapy due to
cancer progression and resistance underlies the majority of
cancer-associated deaths (18).
Therefore, it is necessary to explore drug resistance mechanisms
and reversal strategies of CRC chemotherapy.
Previous studies have demonstrated that tumor stem
cells (CSCs), non-coding RNAs (ncRNAs) and disordered tumor
microenvironment (TME) contribute to the resistance of CRC
(19–22). Notably, Wnt/β-catenin signaling has
been reported to regulate the formation of CRC via these three
aspects (23,24). It is hypothesized that the
dysregulation of the Wnt/β-catenin signaling is related to
chemotherapy resistance in CRC. At present, numerous studies have
sustained this view (25–27), but there is no systematic summary to
the best of our knowledge. Therefore, the present authors have
systematically reviewed the reported studies on Wnt/β-catenin
signaling-mediated chemotherapy resistance of CRC, which may
provide clinical reference for the future.
The Wnt gene was first identified in mouse mammary
tumors in 1982 and was originally named the int1 gene (28). Subsequent investigation showed that
the int gene serves an important role in embryo growth and
development in mice, and its function is similar to the
Drosophila wingless gene (29). The int1 gene and wingless gene are
collectively referred to as the Wnt gene (29). The Wnt signaling pathway is one of
the key signaling pathways in the regulation of cell proliferation
and it serves a significant role in the pathological process of
malignant tumors (30–34). The Wnt gene is composed of various
glycoproteins, is a member of the coiled family of transmembrane
receptors and is the coreceptor for lipoprotein receptor-related
protein (LRP) family and other downstream components (35). There are currently 19 Wnt ligands in
mammals that function via autocrine and paracrine pathways
(36,37). These various Wnt ligands serve
different roles in the development of organisms and the aberrant
expression of Wnt ligand genes can lead to the occurrence and
development of different types of tumors (Table I) (78). Wnt ligands are divided into two
classes according to the different binding methods with downstream
receptors. One group binds to the Frizzled (Fzd) and low-density
lipoprotein-related receptor 5/6 (LRP/6) to activate canonical
β-catenin-dependent pathways. The other group binds Fzd protein to
activate the cyclic guanosine monophosphate protein and the
noncanonical Wnt pathway (79).
β-catenin is the central molecule in the canonical Wnt pathway that
controls the switch of the Wnt signaling pathway. Therefore, it is
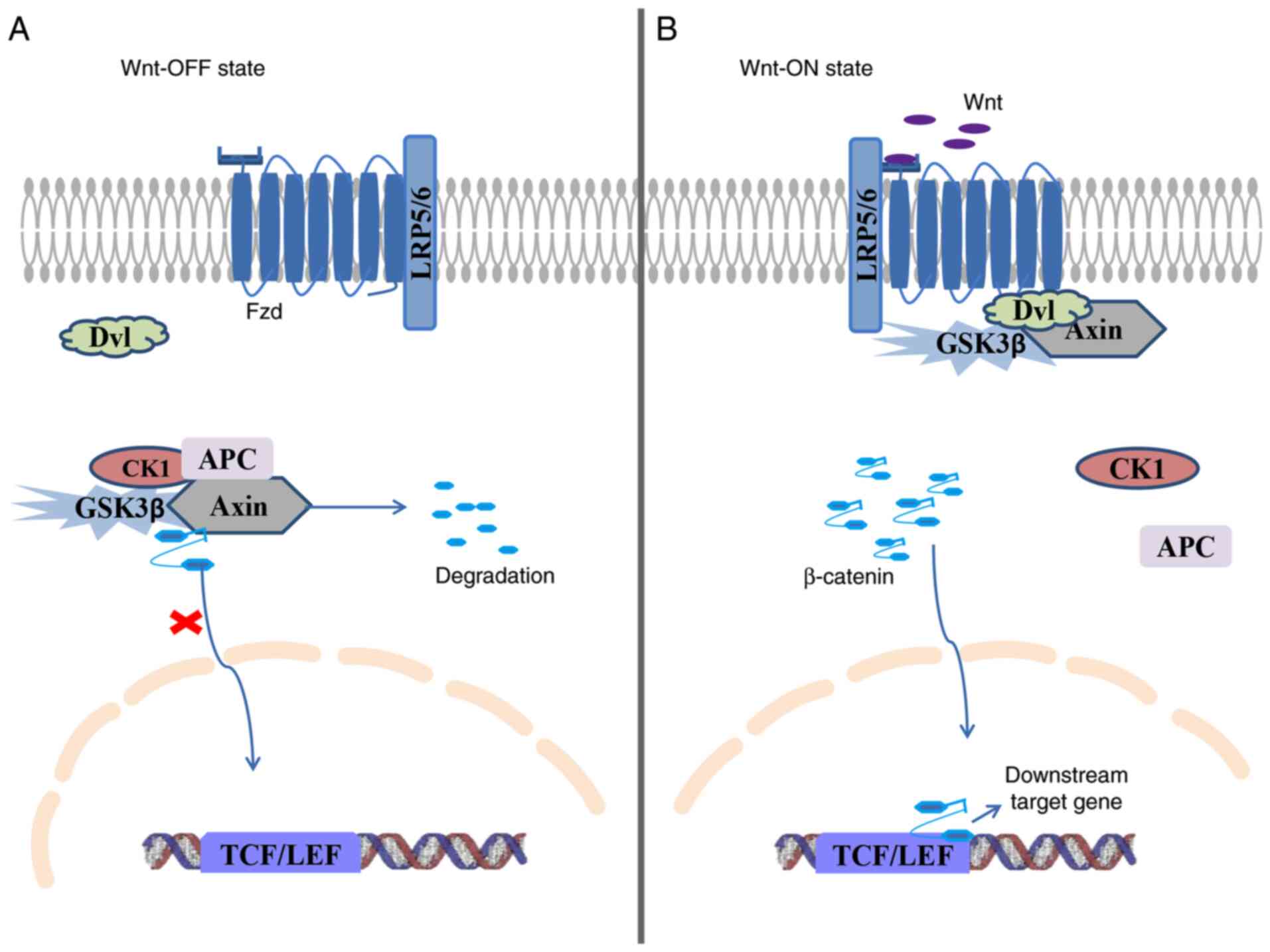
also called Wnt/β-catenin signaling (80). Wnt ligands do not bind to the
receptor in normal mature cells, and Wnt/β-catenin signaling is in
an ‘off’ state (81). Adenomatous
polyposis coli (APC) protein, framework protein Axin, glycogen
synthase kinase 3β (GSK3β) and casein kinase 1 (CK1) form a complex
that causes degradation of β-catenin (82). This complex degrades β-catenin,
which is phosphorylated, modified by ubiquitin and ultimately
degraded by the proteasome (83).
Eventually, the concentration of β-catenin is decreased, nuclear
translocation is suppressed, and downstream target genes, including
c-Myc, cyclin D1, survivin and porous metalloproteinase, cannot be
activated (83). When Wnt ligands
bind to transmembrane Fzd receptors and LRP5/6, CK1 and GSK3β are
attracted to LRP5/6 and function as phosphorylases of LRP5/6, which
prevents formation of the protein complexes that degrade β-catenin
(84). Continuously increasing
concentrations of free β-catenin enter the nucleus via the nuclear
pore membrane and bind to transcription factor/lymphocyte-enhancing
factor (TCF/LEF) (85). Binding
promotes the transcription of downstream target genes that affect
cell proliferation, apoptosis, stromal lysis and angiogenesis
(85). Wnt ligands bind to the
receptor, and Wnt/β-catenin signaling is in an ‘on’ state (81). The details are shown in Fig. 1.
The resistance of human tumors to anticancer drugs
is primarily due to the inherent chemical resistance of tumor
cells, generally attributed to gene mutation, gene amplification or
epigenetic changes, which affect absorption, metabolism or the
export of drugs by a single cell (19). CRC cells exhibit varied resistance
to different chemotherapy drugs, including 5-FU, L-OHP and
irinotecan, depending on enhanced intracellular metabolism,
upregulation or changes in intracellular targets, increased
dihydropyrimidine dehydrogenase and thymidine synthase activities,
upregulated levels of the diminished form glutathione or increased
nucleotide excision repair (86,87).
Resistance to capecitabine is accomplished via methylation of the
gene encoding thymidine phosphorylase and inactivation of
capecitabine (88). For the
checkpoint inhibitors, including ipilimumab, pembrolizumab and
nivolumab, tumors primarily achieve resistance via tumor mutation
and adaptation, decreased production or expression of neoantigens,
overexpression of indoleamine 2,3-dioxygenase and decreased
expression of phosphatase and tensin homolog (PTEN) (89). Ghadimi et al (90) reported that the Wnt transcription
factor TCF7L2 is overexpressed in 5-FU-resistant primary rectal
cancer. The stimulation of Wnt3a leads to the strong activation of
Wnt/β-catenin signaling in SW480, SW837 and LS1034 CRC cells
(91). At the same time, the
activity of TCF/LEF reporter gene is rapidly increased, which
results in resistance to 5-FU (91). The inhibition of β-catenin can avoid
the therapeutic resistance caused by enhanced TCF/LEF gene activity
(91). Another study also
demonstrated that the sensitivity of CRC cells to 5-FU can be
adjusted through Wnt/β-catenin signaling pathway (92). In addition, pharmacological or
genetic inhibition of β-catenin can change the chemical sensitivity
of SW480 and SW620 CRC cells to 5-FU and L-OHP by regulating the
Wnt/β-catenin-Jagged 2-p21 axis (93). Silencing of the T cell factor 4
(Tcf4) gene, which is a downstream effector of Wnt/β-catenin
signaling, sensitizes SW1874, SW1396, SW480 and SW-Sc CRC to L-OHP.
This sensitization effect may be due to different mechanisms,
including the Tcf4 motif in the ATP-binding cassette subfamily B
member 1 (ABCB1) promoter, defects in the nucleotide excision
repair or double strand break repair system after Tcf4-silencing
(87). Wnt inhibitors also improve
chemosensitivity (94,95). Among these inhibitors,
4-acetylantroquinol B, which is isolated from the mycelia of
Ganoderma camphora, negatively regulates the stem cell
maintenance signaling transduction pathway LGR5/Wnt/β-catenin and
is most effective in reducing stem-related chemical resistance
(96). The present study mainly
reviewed the molecular mechanisms of Wnt/β-catenin signaling
through CSCs, ncRNAs and TME that mediate the chemotherapy
resistance of CRC.
CSCs are cells that promote the development of
tumors, and have the ability to self-renew and have multiple
differentiation potentials (97,98).
CSCs have four known characteristics, including self-renewal,
differentiation, tumorigenic and specific surface markers. These
cells are responsible for tumor occurrence, development,
metastasis, recurrence and drug resistance (20,99).
CSCs are naturally chemoresistant. CSCs are functionally protected
in the tissue stem cell niche during chemotherapy (100). The CSCs niche is mainly composed
of fibroblasts and endothelial, mesenchymal and immune cells
(101,102). These adjacent cells promote the
molecular signaling pathways required for the maintenance and
survival of CSCs and trigger the endogenous drug resistance of CSCs
(103). In addition, the
extracellular matrix of niches can protect CSCs from the invasion
of therapeutic drugs (100).
Wnt/β-catenin signaling is a necessary pathway for
the initial activation, self-renewal and cloning ability of CSCs
(104). Fevr et al
(105) reported that
tissue-specific and inducible β-catenin gene ablation blocks
Wnt/β-catenin signaling and reduces the proliferation ability of
CSCs. Wnt/β-catenin signaling regulates the expression of surface
markers of CSCs (106,107). Leucine-rich repeat-containing
G-protein-coupled receptor 5 (LGR5) is a target gene of the Wnt
pathway and a marker of CSCs (107). Activation of Wnt/β-catenin
signaling increases the level of the CSC cell surface marker LGR5
in the CRC cell lines HCT116, SW480 and DLD1 and enriches gene
signatures associated with stemness and cancer relapse in CSCs
(108). LGR5-positive CSCs are
chemotherapy-resistant (109). The
rapid proliferation of CSCs may transform LGR5-negative cells into
LGR5-positive cells, which makes the cells enter a static state to
escape the toxicity of drugs (110). However, CSCs of the CRC cell lines
LoVo, HT29 and HCT116 also obtain drug resistance via the
upregulation of drug-resistant drug pumps mediated by LGR5
(107). As CSCs and normal stem
cells have very similar characteristics, most of these cells are in
the G0 phase of the cell cycle and express specific
ATP-binding cassette proteins (ABC transporter) (111). The ABC transporter is a drug pump
that mediates the outflow or uptake of a specific substrate. This
mechanism takes place at cell membranes (including plasma membrane,
endoplasmic reticulum, Golgi body, peroxisome and mitochondria)
(112). ABC transporters expel
numerous types of drugs from cancer cells and induce chemical
resistance in numerous solid tumors (113,114). ABCB1 was the first cloned human
ABC transporter (115). A study
has shown that ABC inhibitors can inhibit ABC transporters with
high potency and specificity and do not adversely affect the
pharmacokinetics of therapeutic drugs that can kill cancer cells
(116). NSC239225, as one of the
ABC transporter inhibitors, can inhibit ABCB1 to increase the
sensitivity of SW480TR CRC cells to some drugs, such as paclitaxel
(PTX), doxorubicin and mitoxantrone. Its inhibitory effect is
mainly achieved through stimulating ATP hydrolysis and directly
binding to the iodoarylazidoprazosin (IAAP)-specific substrate
binding site (117). Parguerenes I
and II, which also act as ABC transporter inhibitors, can repress
ABCB1 by modifying the extracellular substrate binding site of
ABCB1, thereby reducing the resistance of SW620 and SW620 Ad300 CRC
cells to PTX, Doxorubicin and vincristine (118). Studies have shown that
Wnt/β-catenin signaling is closely related to the ABC transporter
of CSCs (25,119). Inhibition of Wnt/β-catenin
signaling downregulates the expression of mRNA related to the ABC
transporter, which makes SW480 CRC cells more sensitive to PTX and
irinotecan (25). The ABCB1 level
of SW620/AD CRC cells is also positively correlated with
Wnt/β-catenin signaling transduction activity (120). Notably, Kugimiya et al
(121) demonstrated that the
downstream target gene of the Wnt/β-catenin signaling, c-Myc, makes
COLO-320 CRC cells resistant to the chemical 5-FU by regulating the
expression of ABCB5. This effect is primarily achieved by
c-Myc-mediated regulation of the ABC transporter gene expression
via binding to the upstream promoter (121). Wang et al (122) demonstrated that the transient
receptor potential channel short transient receptor potential
channel 5 (TRPC5)-induces an increase in [Ca2+],
promoting the transport of β-catenin to the nucleus, which serves
an important role in ABCB1-induced resistance to 5-FU in CRC cells.
Inhibition of TRPC5 using TRPC5-specific siRNA further inhibits the
Wnt/β-catenin signaling pathway, reduces the induction of ABCB1 and
reverses the resistance of HCT-8 and LoVo CRC cells to 5-FU
(122).
Increased glycolysis is also an important cause of
CSC drug resistance. The stem cell niche is an anoxic functional
chamber that induces CSCs to reprogram for glycolysis (123). This effect promotes the expression
of genes involved in apoptosis resistance, which enables the cells
to survive in a hostile environment and avoid the influence of
chemotherapy (123). Abnormal
activation of Wnt/β-catenin signaling transduction is observed in a
number of types of human cancer, which promotes glycolysis via the
upregulation of solute carrier family 2, facilitated glucose
transporter member 1 expression through its target gene c-Myc
(124). The role of Wnt/β-catenin
signaling transduction in promoting glycolysis is related to drug
resistance (125)
lncRNAs also affect the chemosensitivity of CRC by
regulating the Wnt/β-catenin signaling (145–157). Han et al (145) used reverse
transcription-quantitative PCR and functional testing of CRC
tissues and cell lines and identified that lncRNA CRNDE activates
the downstream targets β-catenin and TCF4 via binding to
miR-181a-5p, which causes resistance to 5-FU and L-OHP. Xiao et
al (146) demonstrated that
lncRNA HOTAIR knockout and mir-203a-3p overexpression inhibited the
Wnt/β-catenin signaling pathway, thereby inhibiting cell
proliferation and reducing chemoresistance. Another study confirmed
that lncRNA H19 increases proliferation via activation of the
Wnt/β-catenin signaling, which promotes the resistance of HT-29 CRC
to methotrexate (147).
CRC-related lncRNA CCAL is another key regulator of CRC
progression. Clinical data has demonstrated that patients with CRC
with high CCAL expression have shorter overall survival rates, and
promotes the resistance of CRC cells to L-OHP (148). A subsequent study showed that the
CCAL promoter region possesses reduced methylation and increased
acetylation in patients with CRC, which promotes its expression.
Upregulated CCAL activates Wnt/β-catenin signaling via inhibition
of activating enhancer-binding protein 2α, which upregulates
MDR1P-gp and induces MDR (149).
Previous studies of chemical resistance primarily
focused on the tumor cells themselves, but TME has also received
attention (150,151). Various cytokines secreted in the
tumor microenvironment, including those from cancer-associated
fibroblasts (CAFs), immune cells, inflammatory factors and
chemokines, may interact with Wnt/β-catenin signaling to cause a
heterogeneous distribution of β-catenin in cells (152–154). Clear colocalization between CAFs
and tumor cells expressing nuclear β-catenin is observed in primary
CRC samples (27). These findings
indicate a close relationship between drug resistance and the tumor
environment, especially CAFs (27).
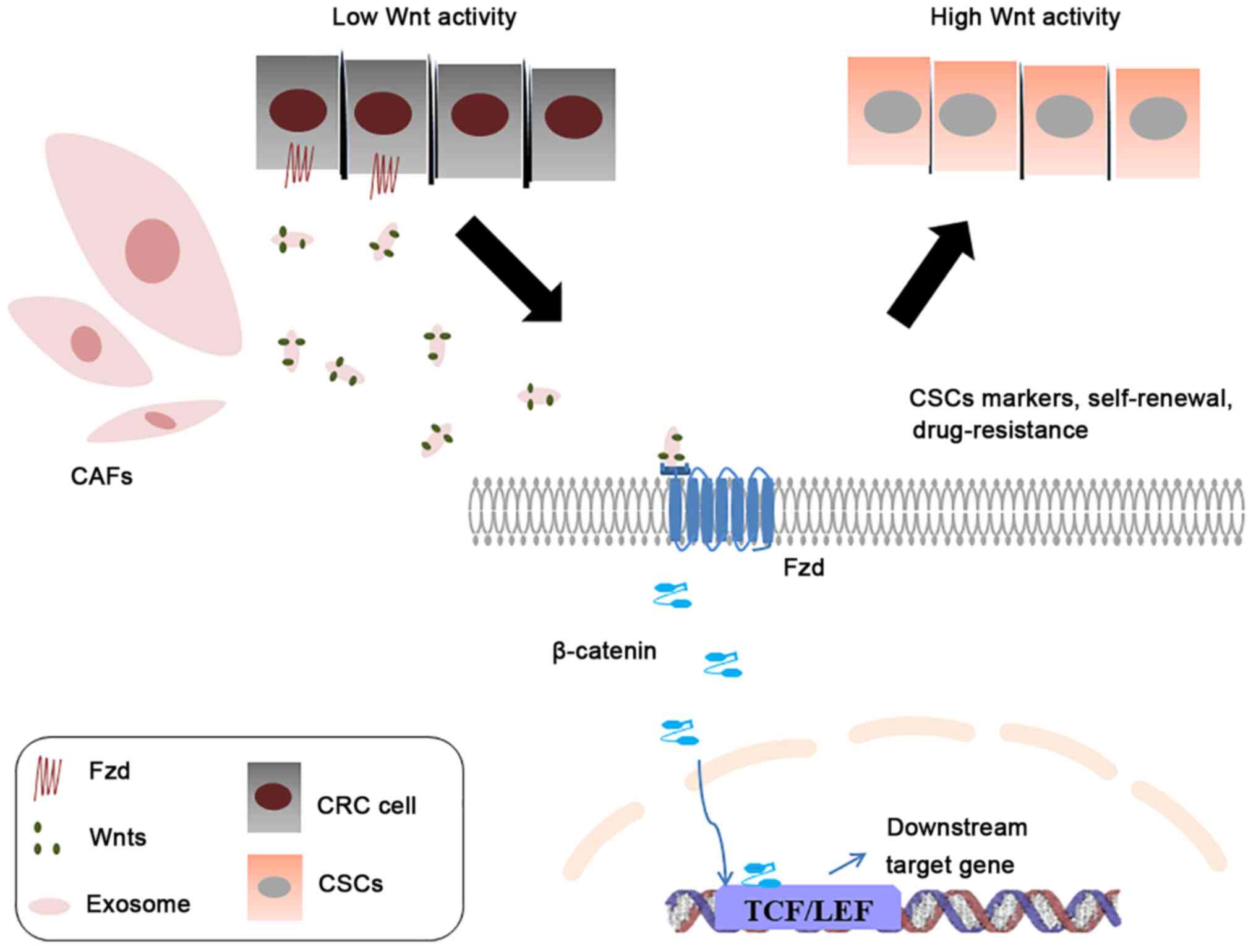
A study has demonstrated that exosomes are ideal carriers for the
delivery of insoluble hydrophobic Wnt proteins (155). CAF-derived exosomes contribute to
the secretion of Wnt ligands, promote the phenotypic recovery of
differentiated CRC cells and the function of CSCs characteristics
by carrying Wnt ligands. These ligands activate Wnt/β-catenin
signaling to regulate Wnt activity (27). All of these actions contribute to
drug resistance (Fig. 2). Hu et
al (156) treated human SW480,
SW620 and LoVo CRC cells with CAF-conditioned medium (CAFs-CM). The
results showed that CAF secretes exosomes into CRC cells, which
leads to a significant increase in miR-92a-3p levels in CRC cells.
The increased expression of miR-92a-3p activates the Wnt/β-catenin
pathway and inhibits mitochondrial apoptosis via direct inhibition
of the tumor suppressor gene F-box/WD repeat-containing protein 7
and apoptosis regulator 1, which promotes the resistance of CRC
cells to 5-FU/L-OHP (157).
Similarly, periodin secreted by fibroblasts also activates
Wnt/β-catenin signaling, which promotes differentiated CRC cells to
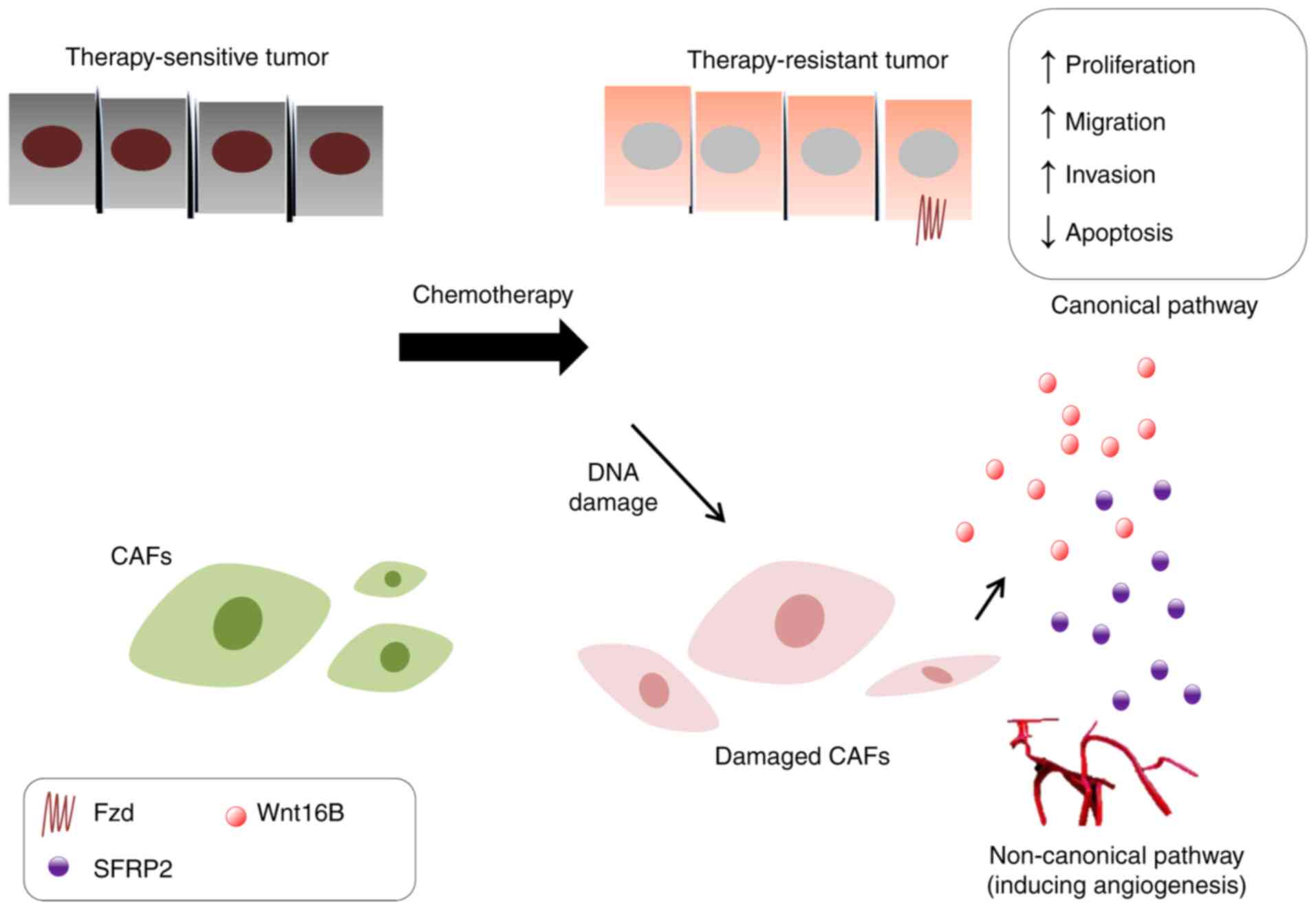
restore CSCs characteristics and functions (158). DNA damage caused by chemotherapy
promotes CAFs to produce numerous soluble factors, including Wnt16B
and stable free radical polymerization 2 (SFRP2) (159). Wnt16B promotes tumor growth via
activation of the canonical pathway in cancer cells, which reduces
treatment sensitivity (159).
SFRP2 acts as a synergistic effector that further enhances the drug
resistance of Wnt16B/β-catenin (160). SFRP2 also participates in the
non-canonical pathway, including angiogenesis, via activation of
calcineurin/nuclear factor of activated T cells, cytoplasmic 3
signaling in endothelial cells, which indirectly promotes tumor
development (Fig. 3) (160). Cancer-associated CAFs in CRC cells
upregulate Wnt signaling-related genes, T-lymphoma infiltration and
metastasis-inducing protein 1, and ultimately enhance the
resistance of CRC by increasing the expression of tumor stem cells
(161). BCL-9 serves a key role in
promoting chemoresistance via the Wnt signaling pathway (162). Hypoxia in the TME leads to the
upregulation of the key Wnt coactivator BCL-9 in a
hypoxia-inducible factor-1α/2α-related manner (163). There is crosstalk between Wnt
signaling and the hypoxia signaling pathway. This crosstalk
synergistically acts on the development of CRC resistance (163).
A number of Wnt inhibitors avoid resistance to drug
recognition and work in conjunction with current clinical
front-line drugs for CRC. Several studies are focused on Wnt
inhibitors in 5-FU resistance (56,94,173).
Coumarin Esculetin (EST) reduces the release of E-cadherin,
vimentin, β-catenin, c-Myc, cyclin D1, Wnt3a and VEGF, which
inhibit Wnt/β-catenin signaling (174). In vitro and in vivo
experiments have shown that EST combined with 5-FU enhances the
sensitivity of HT-29, SW480, HCT-116 and Caco-2 CRC cells to 5-FU
(174). Similarly, the use of the
multikinase inhibitor regorafenib increases miR-34a levels and
reverses 5-FU resistance and the cancer-initiating cell phenotype
by degrading Wnt/β-catenin in HCT-116R and DLD-1R CRC cells
(175). In vitro
experimental results showed that the inhibition of the
Wnt/β-catenin signaling cascade using the tankyrase inhibitor
XAV939 overcomes the resistance of CRC cells carrying short APCs to
5-FU (176). The upregulation of
guanylate-binding protein-1 enhances the killing effect of PTX in
PTX-sensitive CRC cells and PTX-resistant CRC cells via inhibition
of Wnt/β-catenin signaling in the CRC cell lines DLD-1, HT29, DiFi,
T84 and HCT116 (177). Wu et
al (178) reported that the
synergistic use of cinnamaldehyde and L-OHP inhibits
hypoxia-activated Wnt/β-catenin signaling, reverses EMT, actives
CSC and diminishes the occurrence of L-OHP resistance. Patients
with CRC with KRAS mutations are not sensitive to cetuximab and
panitumumab (179). The potent and
selective Wnt/β-catenin inhibitor KYA1797K activates GSK3β and
degrades small β-catenin and Ras molecules to increase the
sensitivity of tumors bearing KRAS mutations to cetuximab and
panitumumab (180). These results
indicate that Wnt signaling leads to chemoresistance in CRC. These
studies highlight that the use of Wnt inhibitors affects the
chemical sensitivity of cells to other drugs, which provides new
approaches for the clinical treatment of CRC.
Activation of the checkpoint kinase 1 (CHK1) pathway
enhances the drug sensitivity of CRC. He et al (181) performed microarray analysis on
CRC-resistant cells and reported that Wnt signaling activation
leads to 5-FU resistance via inhibition of the CHK1 pathway in TP53
wild-type cells, such as HCT-8. In addition, period circadian
protein homolog 3 and dishevelled-3 are common members of the
Wnt/β-catenin pathway and the Notch signaling pathway, which are
involved in chemoresistance (182). Experimental inhibition or
enhancement of the expression of these genes act on the
Wnt/β-catenin signaling and Notch signaling pathways simultaneously
to improve drug sensitivity (182,183). These findings highlight the fact
that the Wnt/β-catenin signaling pathway and other signaling
pathways exhibit crosstalk, synergistic and antagonistic effects in
the occurrence of CRC resistance. Common members between these
different signaling pathways should be identified as targets to
overcome the occurrence of CRC resistance.
CRC is one of the most common malignant tumors in
humans, and the survival rate remains low (1). Treatment resistance in CRC remains an
unsolved problem (17). Generally,
the chemical resistance mechanism of CRC is closely associated with
CSCs, ncRNAs and the TME (19–22).
Wnt/β-catenin signaling maintains the natural chemical resistance
of CSCs and improves drug resistance via the promotion of ABC
transporter and glycolysis in CSCs cells (25,119).
The interaction between Wnt/β-catenin signaling and ncRNAs
regulates the cell cycle and the expression of cancer-related genes
(26,131,132). The TME enhances Wnt/β-catenin
signaling activity (150).
Wnt/β-catenin signaling also mediates tumor immune escape in the
TME (151). Therefore, examining
the role of Wnt/β-catenin signaling in depth has great potential
for therapeutic intervention. More studies should focus on the
mechanism of CRC resistance, and robust preclinical drug testing of
Wnt inhibitors as a single drug or in combination with CRC is
required.
Not applicable.
This study was supported by grants from the Natural
Science Foundation of Jiangxi Province (grant no. 20202BABL206089)
and the Natural Science Foundation of Department of Education of
Jiangxi Province (grant no. GJJ190072).
Not applicable.
GXZ, ZZS, LC and WJD collected the related
literature and drafted the manuscript. QFY and DG participated in
the design of the review and drafted the manuscript. All authors
read and approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Keum N and Giovannucci E: Global burden of
colorectal cancer: Emerging trends, risk factors and prevention
strategies. Nat Rev Gastroenterol Hepatol. 16:713–732. 2019.
View Article : Google Scholar
|
|
2
|
Rawla P, Sunkara T and Barsouk A:
Epidemiology of colorectal cancer: Incidence, mortality, survival,
and risk factors. Prz Gastroenterol. 14:89–103. 2019.
|
|
3
|
Arnold M, Sierra MS, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global patterns and trends in
colorectal cancer incidence and mortality. Gut. 66:683–691. 2017.
View Article : Google Scholar
|
|
4
|
Marmol I, Sanchez-de-Diego C, Pradilla
Dieste A, Cerrada E and Rodriguez Yoldi MJ: Colorectal carcinoma: A
general overview and future perspectives in colorectal cancer. Int
J Mol Sci. 18:1972017. View Article : Google Scholar
|
|
5
|
Calvert PM and Frucht H: The genetics of
colorectal cancer. Ann Intern Med. 137:603–612. 2002. View Article : Google Scholar
|
|
6
|
Angarita FA, Feinberg AE, Feinberg SM,
Riddell RH and McCart JA: Management of complex polyps of the colon
and rectum. Int J Colorectal Dis. 33:115–129. 2018. View Article : Google Scholar
|
|
7
|
Blank A, Roberts DE II, Dawson H, Zlobec I
and Lugli A: Tumor heterogeneity in primary colorectal cancer and
corresponding metastases. Does the apple fall far from the tree?
Front Med (Lausanne). 5:2342018.
|
|
8
|
Dahmus JD, Kotler DL, Kastenberg DM and
Kistler CA: The gut microbiome and colorectal cancer: A review of
bacterial pathogenesis. J Gastrointest Oncol. 9:769–777. 2018.
View Article : Google Scholar
|
|
9
|
Jayasekara H, English DR, Haydon A, Hodge
AM, Lynch BM, Rosty C, Williamson EJ, Clendenning M, Southey MC,
Jenkins MA, et al: Associations of alcohol intake, smoking,
physical activity and obesity with survival following colorectal
cancer diagnosis by stage, anatomic site and tumor molecular
subtype. Int J Cancer. 142:238–250. 2018. View Article : Google Scholar
|
|
10
|
Mehta A and Patel BM: Therapeutic
opportunities in colon cancer: Focus on phosphodiesterase
inhibitors. Life Sci. 230:150–161. 2019. View Article : Google Scholar
|
|
11
|
Salonga D, Danenberg KD, Johnson M,
Metzger R, Groshen S, Tsao-Wei DD, Lenz HJ, Leichman CG, Leichman
L, Diasio RB and Danenberg PV: Colorectal tumors responding to
5-fluorouracil have low gene expression levels of dihydropyrimidine
dehydrogenase, thymidylate synthase, and thymidine phosphorylase.
Clin Cancer Res. 6:1322–1327. 2000.
|
|
12
|
Showalter SL, Showalter TN, Witkiewicz A,
Havens R, Kennedy EP, Hucl T, Kern SE, Yeo CJ and Brody JR:
Evaluating the drug-target relationship between thymidylate
synthase expression and tumor response to 5-fluorouracil. Is it
time to move forward? Cancer Biol Ther. 7:986–994. 2008. View Article : Google Scholar
|
|
13
|
Yaffee P, Osipov A, Tan C, Tuli R and
Hendifar A: Review of systemic therapies for locally advanced and
metastatic rectal cancer. J Gastrointest Oncol. 6:185–200.
2015.
|
|
14
|
Cunningham D, Humblet Y, Siena S, Khayat
D, Bleiberg H, Santoro A, Bets D, Mueser M, Harstrick A, Verslype
C, et al: Cetuximab monotherapy and cetuximab plus irinotecan in
irinotecan-refractory metastatic colorectal cancer. N Engl J Med.
351:337–345. 2004. View Article : Google Scholar
|
|
15
|
Van Cutsem E, Peeters M, Siena S, Humblet
Y, Hendlisz A, Neyns B, Canon JL, Van Laethem JL, Maurel J,
Richardson G, et al: Open-label phase III trial of panitumumab plus
best supportive care compared with best supportive care alone in
patients with chemotherapy-refractory metastatic colorectal cancer.
J Clin Oncol. 25:1658–1664. 2007. View Article : Google Scholar
|
|
16
|
Hu T, Li Z, Gao CY and Cho CH: Mechanisms
of drug resistance in colon cancer and its therapeutic strategies.
World J Gastroenterol. 22:6876–6889. 2016. View Article : Google Scholar
|
|
17
|
Li YJ, Lei YH, Yao N, Wang CR, Hu N, Ye
WC, Zhang DM and Chen ZS: Autophagy and multidrug resisitance in
cancer. Chin J Cancer. 36:522017. View Article : Google Scholar
|
|
18
|
Thomas H and Coley HM: Overcoming
multidrug resistance in cancer: An update on the clinical strategy
of inhibiting p-glycoprotein. Cancer Control. 10:159–165. 2003.
View Article : Google Scholar
|
|
19
|
Tredan O, Galmarini CM, Patel K and
Tannock IF: Drug resistance and the solid tumor microenvironment. J
Natl Cancer Inst. 99:1441–1454. 2007. View Article : Google Scholar
|
|
20
|
Wang MY, Qiu YH, Cai ML, Zhang CH, Wang
XW, Liu H, Chen Y, Zhao WL, Liu JB and Shao RG: Role and molecular
mechanism of stem cells in colorectal cancer initiation. J Drug
Target. 28:1–10. 2020. View Article : Google Scholar
|
|
21
|
Liu X, Fu Q, Du Y, Yang Y and Cho WC:
MicroRNA as regulators of cancer stem cells and chemoresistance in
colorectal cancer. Curr Cancer Drug Targets. 16:738–754. 2016.
View Article : Google Scholar
|
|
22
|
Fanale D, Barraco N, Listi A, Bazan V and
Russo A: Non-coding RNAs functioning in colorectal cancer stem
cells. Adv Exp Med Biol. 937:93–108. 2016. View Article : Google Scholar
|
|
23
|
Rahmani F, Avan A, Hashemy SI and
Hassanian SM: Role of Wnt/beta-catenin signaling regulatory
microRNAs in the pathogenesis of colorectal cancer. J Cell Physiol.
233:811–817. 2018. View Article : Google Scholar
|
|
24
|
Das PK, Islam F and Lam AK: The roles of
cancer stem cells and therapy resistance in colorectal carcinoma.
Cells. 9:13922020. View Article : Google Scholar
|
|
25
|
Chikazawa N, Tanaka H, Tasaka T, Nakamura
M, Tanaka M, Onishi H and Katano M: Inhibition of Wnt signaling
pathway decreases chemotherapy-resistant side-population colon
cancer cells. Anticancer Res. 30:2041–2048. 2010.
|
|
26
|
Shi L, Xi J, Xu X, Peng B and Zhang B:
MiR-148a suppressed cell invasion and migration via targeting
WNT10b and modulating β-catenin signaling in cisplatin-resistant
colorectal cancer cells. Biomed Pharmacother. 109:902–909. 2019.
View Article : Google Scholar
|
|
27
|
Hu YB, Yan C, Mu L, Mi YL, Zhao H, Hu H,
Li XL, Tao DD, Wu YQ, Gong JP and Qin JC: Exosomal Wnt-induced
dedifferentiation of colorectal cancer cells contributes to
chemotherapy resistance. Oncogene. 38:1951–1965. 2019. View Article : Google Scholar
|
|
28
|
Nusse R and Varmus HE: Many tumors induced
by the mouse mammary tumor virus contain a provirus integrated in
the same region of the host genome. Cell. 31:99–109. 1982.
View Article : Google Scholar
|
|
29
|
Rijsewijk F, Schuermann M, Wagenaar E,
Parren P, Weigel D and Nusse R: The Drosophila homolog of the mouse
mammary oncogene int-1 is identical to the segment polarity gene
wingless. Cell. 50:649–657. 1987. View Article : Google Scholar
|
|
30
|
Kahn M: Can we safely target the WNT
pathway? Nat Rev Drug Discov. 13:513–532. 2014. View Article : Google Scholar
|
|
31
|
Zeng ZY, Zhou YH, Zhang WL, Xiong W, Fan
SQ, Li XL, Luo XM, Wu MH, Yang YX, Huang C, et al: Gene expression
profiling of nasopharyngeal carcinoma reveals the abnormally
regulated Wnt signaling pathway. Hum Pathol. 38:120–133. 2007.
View Article : Google Scholar
|
|
32
|
Pate KT, Stringari C, Sprowl-Tanio S, Wang
K, TeSlaa T, Hoverter NP, McQuade MM, Garner C, Digman MA, Teitell
MA, et al: Wnt signaling directs a metabolic program of glycolysis
and angiogenesis in colon cancer. EMBO J. 33:1454–1473. 2014.
View Article : Google Scholar
|
|
33
|
Polakis P: Wnt signaling in cancer. Cold
Spring Harb Perspect Biol. 4:a0080522012. View Article : Google Scholar
|
|
34
|
Tammela T, Sanchez-Rivera FJ, Cetinbas NM,
Wu K, Joshi NS, Helenius K, Park Y, Azimi R, Kerper NR, Wesselhoeft
RA, et al: A Wnt-producing niche drives proliferative potential and
progression in lung adenocarcinoma. Nature. 545:355–359. 2017.
View Article : Google Scholar
|
|
35
|
Lee SY, Jeon HM, Ju MK, Kim CH, Yoon G,
Han SI, Park HG and Kang HS: Wnt/Snail signaling regulates
cytochrome C oxidase and glucose metabolism. Cancer Res.
72:3607–3617. 2012. View Article : Google Scholar
|
|
36
|
Willert K, Brown JD, Danenberg E, Duncan
AW, Weissman IL, Reya T, Yates JR III and Nusse R: Wnt proteins are
lipid-modified and can act as stem cell growth factors. Nature.
423:448–452. 2003. View Article : Google Scholar
|
|
37
|
Takada R, Satomi Y, Kurata T, Ueno N,
Norioka S, Kondoh H, Takao T and Takada S: Monounsaturated fatty
acid modification of Wnt protein: Its role in Wnt secretion. Dev
Cell. 11:791–801. 2006. View Article : Google Scholar
|
|
38
|
He B, You L, Uematsu K, Xu Z, Lee AY,
Matsangou M, McCormick F and Jablons DM: A monoclonal antibody
against Wnt-1 induces apoptosis in human cancer cells. Neoplasia.
6:7–14. 2004. View Article : Google Scholar
|
|
39
|
Chen G, Shukeir N, Potti A, Sircar K,
Aprikian A, Goltzman D and Rabbani SA: Up-regulation of Wnt-1 and
beta-catenin production in patients with advanced metastatic
prostate carcinoma: Potential pathogenetic and prognostic
implications. Cancer. 101:1345–1356. 2004. View Article : Google Scholar
|
|
40
|
Babaei K, Khaksar R, Zeinali T, Hemmati H,
Bandegi A, Samidoust P, Ashoobi MT, Hashemian H, Delpasand K,
Talebinasab F, et al: Epigenetic profiling of MUTYH, KLF6, WNT1 and
KLF4 genes in carcinogenesis and tumorigenesis of colorectal
cancer. Biomedicine (Taipei). 9:222019. View Article : Google Scholar
|
|
41
|
Jia S, Qu T, Feng M, Ji K, Li Z, Jiang W
and Ji J: Association of Wnt1-inducible signaling pathway protein-1
with the proliferation, migration and invasion in gastric cancer
cells. Tumour Biol. 39:10104283176997552017. View Article : Google Scholar
|
|
42
|
Bodnar L, Stanczak A, Cierniak S, Smoter
M, Cichowicz M, Kozlowski W, Szczylik C, Wieczorek M and
Lamparska-Przybysz M: Wnt/β-catenin pathway as a potential
prognostic and predictive marker in patients with advanced ovarian
cancer. J Ovarian Res. 7:162014. View Article : Google Scholar
|
|
43
|
Huang CB, Ma RJ, Xu Y, Li N, Li ZX, Yue J,
Li HX, Guo Y and Qi D: Wnt2 promotes non-small cell lung cancer
progression by activating WNT/β-catenin pathway. Am J Cancer Res.
5:1032–1046. 2015.
|
|
44
|
Katoh M: Frequent up-regulation of WNT2 in
primary gastric cancer and colorectal cancer. Int J Oncol.
19:1003–1007. 2001.
|
|
45
|
Nakashima N, Liu D, Huang CL, Ueno M,
Zhang X and Yokomise H: Wnt3 gene expression promotes tumor
progression in non-small cell lung cancer. Lung Cancer. 76:228–234.
2012. View Article : Google Scholar
|
|
46
|
Wang HS, Nie X, Wu RB, Yuan HW, Ma YH, Liu
XL, Zhang JY, Deng XL, Na Q, Jin HY, et al: Downregulation of human
Wnt3 in gastric cancer suppresses cell proliferation and induces
apoptosis. Onco Targets Ther. 9:3849–3860. 2016. View Article : Google Scholar
|
|
47
|
Nie XB, Xia FL, Liu Y, Zhou Y, Ye WL, Hean
PH, Meng JM, Liu HY, Liu L, Wen JX, et al: Downregulation of Wnt3
suppresses colorectal cancer development through inhibiting cell
proliferation and migration. Front Pharmacol. 10:11102019.
View Article : Google Scholar
|
|
48
|
Thiago L, Costa ES, Lopes DV, Otazu IB,
Nowill AE, Mendes FA, Portilho DM, Abreu JG, Mermelstein CS, Orfao
A, et al: The Wnt signaling pathway regulates Nalm-16 b-cell
precursor acute lymphoblastic leukemic cell line survival and
etoposide resistance. Biomed Pharmacother. 64:63–72. 2010.
View Article : Google Scholar
|
|
49
|
Zimmerman ZF, Kulikauskas RM, Bomsztyk K,
Moon RT and Chien AJ: Activation of Wnt/β-catenin signaling
increases apoptosis in melanoma cells treated with trail. PLoS One.
8:e695932013. View Article : Google Scholar
|
|
50
|
Annavarapu SR, Cialfi S, Dominici C, Kokai
GK, Uccini S, Ceccarelli S, McDowell HP and Helliwell TR:
Characterization of Wnt/β-catenin signaling in rhabdomyosarcoma.
Lab Invest. 93:1090–1099. 2013. View Article : Google Scholar
|
|
51
|
Fox SA, Richards AK, Kusumah I, Perumal V,
Bolitho EM, Mutsaers SE and Dharmarajan AM: Expression profile and
function of Wnt signaling mechanisms in malignant mesothelioma
cells. Biochem Biophys Res Commun. 440:82–87. 2013. View Article : Google Scholar
|
|
52
|
Wang SH, Li N, Wei Y, Li QR and Yu ZP:
β-catenin deacetylation is essential for WNT-induced proliferation
of breast cancer cells. Mol Med Rep. 9:973–978. 2014. View Article : Google Scholar
|
|
53
|
Akaboshi S, Watanabe S, Hino Y, Sekita Y,
Xi Y, Araki K, Yamamura K, Oshima M, Ito T, Baba H and Nakao M:
HMGA1 is induced by Wnt/beta-catenin pathway and maintains cell
proliferation in gastric cancer. Am J Pathol. 175:1675–1685. 2009.
View Article : Google Scholar
|
|
54
|
Zhao L, Wang LL, Zhang CL, Liu Z, Piao YJ,
Yan J, Xiang R, Yao YQ and Y S: E6-induced selective translation of
WNT4 and JIP2 promotes the progression of cervical cancer via a
noncanonical WNT signaling pathway. Signal Transduct Target Ther.
4:322019. View Article : Google Scholar
|
|
55
|
McDonald SL and Silver A: The opposing
roles of Wnt-5a in cancer. Br J Cancer. 101:209–214. 2009.
View Article : Google Scholar
|
|
56
|
Li J, Ying J, Fan Y, Wu L, Ying Y, Chan
AT, Srivastava G and Tao Q: WNT5A antagonizes WNT/β-catenin
signaling and is frequently silenced by promoter CpG methylation in
esophageal squamous cell carcinoma. Cancer Biol Ther. 10:617–624.
2010. View Article : Google Scholar
|
|
57
|
Ying J, Li H, Yu J, Ng KM, Poon FF, Wong
SC, Chan AT, Sung JJ and Tao Q: WNT5A exhibits tumor-suppressive
activity through antagonizing the Wnt/beta-catenin signaling, and
is frequently methylated in colorectal cancer. Clin Cancer Res.
14:55–61. 2008. View Article : Google Scholar
|
|
58
|
Kremenevskaja N, von Wasielewski R, Rao
AS, Schofl C, Andersson T and Brabant G: Wnt-5a has tumor
suppressor activity in thyroid carcinoma. Oncogene. 24:2144–2154.
2005. View Article : Google Scholar
|
|
59
|
Thiele S, Rachner TD, Rauner M and
Hofbauer LC: WNT5A and its receptors in the bone-cancer dialogue. J
Bone Miner Res. 31:1488–1496. 2016. View Article : Google Scholar
|
|
60
|
Kurayoshi M, Oue N, Yamamoto H, Kishida M,
Inoue A, Asahara T, Yasui W and Kikuchi A: Expression of Wnt-5a is
correlated with aggressiveness of gastric cancer by stimulating
cell migration and invasion. Cancer Res. 66:10439–10448. 2006.
View Article : Google Scholar
|
|
61
|
Huang CL, Liu D, Nakano J, Ishikawa S,
Kontani K, Yokomise H and Ueno M: Wnt5a expression is associated
with the tumor proliferation and the stromal vascular endothelial
growth factor-an expression in non-small-cell lung cancer. J Clin
Oncol. 23:8765–8773. 2005. View Article : Google Scholar
|
|
62
|
Bo H, Zhang S, Gao L, Chen Y, Zhang J,
Chang X and Zhu M: Upregulation of Wnt5a promotes
epithelial-tomesenchymal transition and metastasis of pancreatic
cancer cells. BMC Cancer. 13:4962013. View Article : Google Scholar
|
|
63
|
Navarrete-Meneses MDP and Perez-Vera P:
Epigenetic alterations in acute lymphoblastic leukemia. Bol Med
Hosp Infant Mex. 74:243–264. 2017.(In Spanish).
|
|
64
|
Stewart DJ: Wnt signaling pathway in
non-small cell lung cancer. J Natl Cancer Inst. 106:djt3562014.
View Article : Google Scholar
|
|
65
|
Kirikoshi H and Katoh M: Expression of
WNT7A in human normal tissues and cancer, and regulation of WNT7A
and WNT7B in human cancer. Int J Oncol. 21:895–900. 2002.
|
|
66
|
Vesel M, Rapp J, Feller D, Kiss E, Jaromi
L, Meggyes M, Miskei G, Duga B, Smuk G, Laszlo T, et al: ABCB1 and
ABCG2 drug transporters are differentially expressed in non-small
cell lung cancers (NSCLC) and expression is modified by cisplatin
treatment via altered Wnt signaling. Respir Res. 18:522017.
View Article : Google Scholar
|
|
67
|
Li J, Zhang Z, Wang L and Zhang Y: The
oncogenic role of Wnt10a in colorectal cancer through activation of
canonical Wnt/β-catenin signaling. Oncol Lett. 17:3657–3664.
2019.
|
|
68
|
Li P, Liu W, Xu Q and Wang C: Clinical
significance and biological role of Wnt10a in ovarian cancer. Oncol
Lett. 14:6611–6617. 2017.
|
|
69
|
Hsu RJ, Ho JY, Cha TL, Yu DS, Wu CL, Huang
WP, Chu P, Chen YH, Chen JT and Yu CP: WNT10A plays an oncogenic
role in renal cell carcinoma by activating WNT/beta-catenin
pathway. PLoS One. 7:e476492012. View Article : Google Scholar
|
|
70
|
Kirikoshi H, Inoue S, Sekihara H and Katoh
M: Expression of WNT10A in human cancer. Int J Oncol. 19:997–1001.
2001.
|
|
71
|
Dong T, Zhang Z, Zhou W, Zhou X, Geng C,
Chang LK, Tian X and Liu S: WNT10A/β-catenin pathway in
tumorigenesis of papillary thyroid carcinoma. Oncol Rep.
38:1287–1294. 2017. View Article : Google Scholar
|
|
72
|
Wend P, Runke S, Wend K, Anchondo B,
Yesayan M, Jardon M, Hardie N, Loddenkemper C, Ulasov I, Lesniak
MS, et al: WNT10B/beta-catenin signalling induces HMGA2 and
proliferation in metastatic triple-negative breast cancer. EMBO Mol
Med. 5:264–279. 2013. View Article : Google Scholar
|
|
73
|
Chen H, Wang Y and Xue F: Expression and
the clinical significance of Wnt10a and Wnt10b in endometrial
cancer are associated with the Wnt/β-catenin pathway. Oncol Rep.
29:507–514. 2013. View Article : Google Scholar
|
|
74
|
Saitoh T, Kirikoshi H, Mine T and Katoh M:
Proto-oncogene WNT10B is up-regulated by tumor necrosis factor
alpha in human gastric cancer cell line MKN45. Int J Oncol.
19:1187–1192. 2001.
|
|
75
|
Bartis D, Csongei V, Weich A, Kiss E,
Barko S, Kovacs T, Avdicevic M, D'Souza VK, Rapp J, Kvell K, et al:
Down-regulation of canonical and up-regulation of non-canonical Wnt
signalling in the carcinogenic process of squamous cell lung
carcinoma. PLoS One. 8:e573932013. View Article : Google Scholar
|
|
76
|
Tian S, Hu J, Tao K, Wang J, Chu Y, Li J,
Liu Z, Ding X, Xu L, Li Q, et al: Secreted AGR2 promotes invasion
of colorectal cancer cells via Wnt11-mediated non-canonical Wnt
signaling. Exp Cell Res. 364:198–207. 2018. View Article : Google Scholar
|
|
77
|
Toyama T, Lee HC, Koga H, Wands JR and Kim
M: Noncanonical Wnt11 inhibits hepatocellular carcinoma cell
proliferation and migration. Mol Cancer Res. 8:254–265. 2010.
View Article : Google Scholar
|
|
78
|
Yin P, Wang W, Zhang Z, Bai Y, Gao J and
Zhao C: Wnt signaling in human and mouse breast cancer: Focusing on
Wnt ligands, receptors and antagonists. Cancer Sci. 109:3368–3375.
2018. View Article : Google Scholar
|
|
79
|
Barker N, van Es JH, Kuipers J, Kujala P,
van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H,
Peters PJ and Clevers H: Identification of stem cells in small
intestine and colon by marker gene Lgr5. Nature. 449:1003–1007.
2007. View Article : Google Scholar
|
|
80
|
Bourroul GM, Fragoso HJ, Gomes JW,
Bourroul VS, Oshima CT, Gomes TS, Saba GT, Palma RT and Waisberg J:
The destruction complex of beta-catenin in colorectal carcinoma and
colonic adenoma. Einstein (Sao Paulo). 14:135–142. 2016. View Article : Google Scholar
|
|
81
|
Sawa M, Masuda M and Yamada T: Targeting
the Wnt signaling pathway in colorectal cancer. Expert Opin Ther
Targets. 20:419–429. 2016. View Article : Google Scholar
|
|
82
|
Amit S, Hatzubai A, Birman Y, Andersen JS,
Ben-Shushan E, Mann M, Ben-Neriah Y and Alkalay I: Axin-mediated
CKI phosphorylation of beta-catenin at Ser 45: A molecular switch
for the Wnt pathway. Genes Dev. 16:1066–1076. 2002. View Article : Google Scholar
|
|
83
|
He X, Semenov M, Tamai K and Zeng X: LDL
receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling:
Arrows point the way. Development. 131:1663–1677. 2004. View Article : Google Scholar
|
|
84
|
Bilic J, Huang YL, Davidson G, Zimmermann
T, Cruciat CM, Bienz M and Niehrs C: Wnt induces LRP6 signalosomes
and promotes dishevelled-dependent LRP6 phosphorylation. Science.
316:1619–1622. 2007. View Article : Google Scholar
|
|
85
|
Zarkou V, Galaras A, Giakountis A and
Hatzis P: Crosstalk mechanisms between the WNT signaling pathway
and long non-coding RNAs. Noncoding RNA Res. 3:42–53. 2018.
View Article : Google Scholar
|
|
86
|
Hammond WA, Swaika A and Mody K:
Pharmacologic resistance in colorectal cancer: A review. Ther Adv
Med Oncol. 8:57–84. 2016. View Article : Google Scholar
|
|
87
|
Gheidari F, Bakhshandeh B,
Teimoori-Toolabi L, Mehrtash A, Ghadir M and Zeinali S: TCF4
silencing sensitizes the colon cancer cell line to oxaliplatin as a
common chemotherapeutic drug. Anticancer Drugs. 25:908–916. 2014.
View Article : Google Scholar
|
|
88
|
Kosuri KV, Wu X, Wang L, Villalona-Calero
MA and Otterson GA: An epigenetic mechanism for capecitabine
resistance in mesothelioma. Biochem Biophys Res Commun.
391:1465–1470. 2010. View Article : Google Scholar
|
|
89
|
Rieth J and Subramanian S: Mechanisms of
intrinsic tumor resistance to immunotherapy. Int J Mol Sci.
19:13402018. View Article : Google Scholar
|
|
90
|
Ghadimi BM, Grade M, Difilippantonio MJ,
Varma S, Simon R, Montagna C, Fuzesi L, Langer C, Becker H, Liersch
T and Ried T: Effectiveness of gene expression profiling for
response prediction of rectal adenocarcinomas to preoperative
chemoradiotherapy. J Clin Oncol. 23:1826–1838. 2005. View Article : Google Scholar
|
|
91
|
Emons G, Spitzner M, Reineke S, Moller J,
Auslander N, Kramer F, Hu Y, Beissbarth T, Wolff HA, Rave-Frank M,
et al: Chemoradiotherapy resistance in colorectal cancer cells is
mediated by Wnt/beta-catenin signaling. Mol Cancer Res.
15:1481–1490. 2017. View Article : Google Scholar
|
|
92
|
Deng YH, Pu XX, Huang MJ, Xiao J, Zhou JM,
Lin TY and Lin EH: 5-Fluorouracil upregulates the activity of Wnt
signaling pathway in CD133-positive colon cancer stem-like cells.
Chin J Cancer. 29:810–815. 2010. View Article : Google Scholar
|
|
93
|
Vaish V, Kim J and Shim M: Jagged-2 (JAG2)
enhances tumorigenicity and chemoresistance of colorectal cancer
cells. Oncotarget. 8:53262–53275. 2017. View Article : Google Scholar
|
|
94
|
Kukcinaviciute E, Jonusiene V,
Sasnauskiene A, Dabkeviciene D, Eidenaite E and Laurinavicius A:
Significance of Notch and Wnt signaling for chemoresistance of
colorectal cancer cells HCT116. J Cell Biochem. 119:5913–5920.
2018. View Article : Google Scholar
|
|
95
|
Gonzalez-Exposito R, Semiannikova M,
Griffiths B, Khan K, Barber LJ, Woolston A, Spain G, von Loga K,
Challoner B, Patel R, et al: CEA expression heterogeneity and
plasticity confer resistance to the CEA-targeting bispecific
immunotherapy antibody cibisatamab (CEA-TCB) in patient-derived
colorectal cancer organoids. J Immunother Cancer. 7:1012019.
View Article : Google Scholar
|
|
96
|
Chang TC, Yeh CT, Adebayo BO, Lin YC, Deng
L, Rao YK, Huang CC, Lee WH, Wu AT, Hsiao M, et al:
4-Acetylantroquinonol B inhibits colorectal cancer tumorigenesis
and suppresses cancer stem-like phenotype. Toxicol Appl Pharmacol.
288:258–268. 2015. View Article : Google Scholar
|
|
97
|
Vermeulen L, Sprick MR, Kemper K, Stassi G
and Medema JP: Cancer stem cells-old concepts, new insights. Cell
Death Differ. 15:947–958. 2008. View Article : Google Scholar
|
|
98
|
Munro MJ, Wickremesekera SK, Peng L, Tan
ST and Itinteang T: Cancer stem cells in colorectal cancer: A
review. J Clin Pathol. 71:110–116. 2018. View Article : Google Scholar
|
|
99
|
Li N, Babaei-Jadidi R, Lorenzi F,
Spencer-Dene B, Clarke P, Domingo E, Tulchinsky E, Vries RGJ, Kerr
D, Pan Y, et al: An FBXW7-ZEB2 axis links EMT and tumour
microenvironment to promote colorectal cancer stem cells and
chemoresistance. Oncogenesis. 8:132019. View Article : Google Scholar
|
|
100
|
Prieti-Vila M, Takahashi R, Usuba W,
Kohama I and Ochiya T: Drug resistance driven by cancer stem cells
and their niche. Int J Mol Sci. 18:25742017. View Article : Google Scholar
|
|
101
|
Li L and Xie T: Stem cell niche: Structure
and function. Annu Rev Cell Dev Biol. 21:605–631. 2005. View Article : Google Scholar
|
|
102
|
Liu H, Zhang W, Jia Y, Yu Q, Grau GE, Peng
L, Ran Y, Yang Z, Deng H and Lou J: Single-cell clones of liver
cancer stem cells have the potential of differentiating into
different types of tumor cells. Cell Death Dis. 4:e8572013.
View Article : Google Scholar
|
|
103
|
Daverey A, Drain AP and Kidambi S:
Physical intimacy of breast cancer cells with mesenchymal stem
cells elicits trastuzumab resistance through src activation. Sci
Rep. 5:137442015. View Article : Google Scholar
|
|
104
|
Kim JY, Lee HY, Park KK, Choi YK, Nam JS
and Hong IS: CWP232228 targets liver cancer stem cells through
Wnt/β-catenin signaling: A novel therapeutic approach for liver
cancer treatment. Oncotarget. 7:20395–20409. 2016. View Article : Google Scholar
|
|
105
|
Fevr T, Robine S, Louvard D and Huelsken
J: Wnt/beta-catenin is essential for intestinal homeostasis and
maintenance of intestinal stem cells. Mol Cell Biol. 27:7551–7559.
2007. View Article : Google Scholar
|
|
106
|
Dahal Lamichane B, Jung SY, Yun J, Kang S,
Kim DY, Lamichane S, Kim YJ, Park JH, Jang WB, Ji ST, et al: AGR2
is a target of canonical Wnt/β-catenin signaling and is important
for stemness maintenance in colorectal cancer stem cells. Biochem
Biophys Res Commun. 515:600–606. 2019. View Article : Google Scholar
|
|
107
|
Liu YS, Hsu HC, Tseng KC, Chen HC and Chen
SJ: Lgr5 promotes cancer stemness and confers chemoresistance
through ABCB1 in colorectal cancer. Biomed Pharmacother.
67:791–799. 2013. View Article : Google Scholar
|
|
108
|
Zhan T, Ambrosi G, Wandmacher AM, Rauscher
B, Betge J, Rindtorff N, Haussler RS, Hinsenkamp I, Bamberg L,
Hessling B, et al: MEK inhibitors activate Wnt signalling and
induce stem cell plasticity in colorectal cancer. Nat Commun.
10:21972019. View Article : Google Scholar
|
|
109
|
Wu W, Cao J, Ji Z, Wang J, Jiang T and
Ding H: Co-expression of Lgr5 and CXCR4 characterizes cancer
stem-like cells of colorectal cancer. Oncotarget. 7:81144–81155.
2016. View Article : Google Scholar
|
|
110
|
Kobayashi S, Yamada-Okabe H, Suzuki M,
Natori O, Kato A, Matsubara K, Jau Chen Y, Yamazaki M, Funahashi S,
Yoshida K, et al: LGR5-positive colon cancer stem cells
interconvert with drug-resistant LGR5-negative cells and are
capable of tumor reconstitution. Stem Cells. 30:2631–2644. 2012.
View Article : Google Scholar
|
|
111
|
Villanueva-Toledo J, Ponciano-Gomez A,
Ortiz-Sanchez E and Garrido E: Side populations from
cervical-cancer-derived cell lines have stem-cell-like properties.
Mol Biol Rep. 41:1993–2004. 2014. View Article : Google Scholar
|
|
112
|
Steinbichler TB, Dudas J, Skvortsov S,
Ganswindt U, Riechelmann H and Skvortsova II: Therapy resistance
mediated by cancer stem cells. Semin Cancer Biol. 53:156–167. 2018.
View Article : Google Scholar
|
|
113
|
Singh A and Settleman J: EMT, cancer stem
cells and drug resistance: An emerging axis of evil in the war on
cancer. Oncogene. 29:4741–4751. 2010. View Article : Google Scholar
|
|
114
|
Martin-Orozco E, Sanchez-Fernandez A,
Ortiz-Parra I and Ayala-San Nicolas M: WNT signaling in tumors: The
way to evade drugs and immunity. Front Immunol. 10:28542019.
View Article : Google Scholar
|
|
115
|
Islam MO, Kanemura Y, Tajria J, Mori H,
Kobayashi S, Shofuda T, Miyake J, Hara M, Yamasaki M and Okano H:
Characterization of ABC transporter ABCB1 expressed in human neural
stem/progenitor cells. FEBS Lett. 579:3473–3480. 2005. View Article : Google Scholar
|
|
116
|
Falasca M and Linton KJ: Investigational
ABC transporter inhibitors. Expert Opin Investig Drugs. 21:657–666.
2012. View Article : Google Scholar
|
|
117
|
Duan Z, Li X, Huang H, Yuan W, Zheng SL,
Liu X, Zhang Z, Choy E, Harmon D, Mankin H and Hornicek F:
Synthesis and evaluation of
(2-(4-methoxyphenyl)-4-quinolinyl)(2-piperidinyl)methanol
(NSC23925) isomers to reverse multidrug resistance in cancer. J Med
Chem. 55:3113–3121. 2012. View Article : Google Scholar
|
|
118
|
Huang XC, Sun YL, Salim AA, Chen ZS and
Capon RJ: Parguerenes: Marine red alga bromoditerpenes as
inhibitors of P-glycoprotein (ABCB1) in multidrug resistant human
cancer cells. Biochem Pharmacol. 85:1257–1268. 2013. View Article : Google Scholar
|
|
119
|
Zinzi L, Contino M, Cantore M, Capparelli
E, Leopoldo M and Colabufo NA: ABC transporters in CSCs membranes
as a novel target for treating tumor relapse. Front Pharmacol.
5:1632014. View Article : Google Scholar
|
|
120
|
Liu YY, Gupta V, Patwardhan GA, Bhinge K,
Zhao Y, Bao J, Mehendale H, Cabot MC, Li YT and Jazwinski SM:
Glucosylceramide synthase upregulates MDR1 expression in the
regulation of cancer drug resistance through cSrc and beta-catenin
signaling. Mol Cancer. 9:1452010. View Article : Google Scholar
|
|
121
|
Kugimiya N, Nishimoto A, Hosoyama T, Ueno
K, Enoki T, Li TS and Hamano K: The c-MYC-ABCB5 axis plays a
pivotal role in 5-fluorouracil resistance in human colon cancer
cells. J Cell Mol Med. 19:1569–1581. 2015. View Article : Google Scholar
|
|
122
|
Wang T, Chen Z, Zhu Y, Pan Q, Liu Y, Qi X,
Jin L, Jin J, Ma X and Hua D: Inhibition of transient receptor
potential channel 5 reverses 5-Fluorouracil resistance in human
colorectal cancer cells. J Biol Chem. 290:448–456. 2015. View Article : Google Scholar
|
|
123
|
Plaks V, Kong N and Werb Z: The cancer
stem cell niche: How essential is the niche in regulating stemness
of tumor cells? Cell Stem Cell. 16:225–238. 2015. View Article : Google Scholar
|
|
124
|
Osthus RC, Shim H, Kim S, Li Q, Reddy R,
Mukherjee M, Xu Y, Wonsey D, Lee LA and Dang CV: Deregulation of
glucose transporter 1 and glycolytic gene expression by c-Myc. J
Biol Chem. 275:21797–21800. 2000. View Article : Google Scholar
|
|
125
|
Wang T, Ning K, Lu TX and Hua D: Elevated
expression of TrpC5 and GLUT1 is associated with chemoresistance in
colorectal cancer. Oncol Rep. 37:1059–1065. 2017. View Article : Google Scholar
|
|
126
|
Matsui M and Corey DR: Non-coding RNAs as
drug targets. Nat Rev Drug Discov. 16:167–179. 2017. View Article : Google Scholar
|
|
127
|
Ling H, Fabbri M and Calin GA: MicroRNAs
and other non-coding RNAs as targets for anticancer drug
development. Nat Rev Drug Discov. 12:847–865. 2013. View Article : Google Scholar
|
|
128
|
Laurent GS, Wahlestedt C and Kapranov P:
The landscape of long noncoding RNA classification. Trends Genet.
31:239–251. 2015. View Article : Google Scholar
|
|
129
|
Sato-Kuwabara Y, Melo SA, Soares FA and
Calin GA: The fusion of two worlds: Non-coding RNAs and
extracellular vesicles-diagnostic and therapeutic implications
(Review). Int J Oncol. 46:17–27. 2015. View Article : Google Scholar
|
|
130
|
Ebbesen KK, Kjems J and Hansen TB:
Circular RNAs: Identification, biogenesis and function. Biochim
Biophys Acta. 1859:163–168. 2016. View Article : Google Scholar
|
|
131
|
Chen HY, Lang YD, Lin HN, Liu YR, Liao CC,
Nana AW, Yen Y and Chen RH: miR-103/107 prolong Wnt/β-catenin
signaling and colorectal cancer stemness by targeting Axin2. Sci
Rep. 9:96872019. View Article : Google Scholar
|
|
132
|
Zhou H, Lin C, Zhang Y, Zhang X, Zhang C,
Zhang P, Xie X and Ren Z: miR-506 enhances the sensitivity of human
colorectal cancer cells to oxaliplatin by suppressing MDR1/P-gp
expression. Cell Prolif. 50:e123412017. View Article : Google Scholar
|
|
133
|
Lu ML, Zhang Y, Li J, Fu Y, Li WH, Zhao
GF, Li XH, Wei L, Liu GB and Huang H: MicroRNA-124 inhibits
colorectal cancer cell proliferation and suppresses tumor growth by
interacting with PLCB1 and regulating Wnt/β-catenin signaling
pathway. Eur Rev Med Pharmacol Sci. 23:121–136. 2019.
|
|
134
|
Liang CQ, Fu YM, Liu ZY, Xing BR, Jin Y
and Huang JL: The effect of miR-224 down-regulation on SW80 cell
proliferation and apoptosis and weakening of ADM drug resistance.
Eur Rev Med Pharmacol Sci. 21:5008–5016. 2017.
|
|
135
|
Lucero OM, Dawson DW, Moon RT and Chien
AJ: A re-evaluation of the ‘oncogenic’ nature of Wnt/beta-catenin
signaling in melanoma and other cancers. Curr Oncol Rep.
12:314–318. 2010. View Article : Google Scholar
|
|
136
|
Song C, Lu P, Sun G, Yang L and Wang Z and
Wang Z: miR-34a sensitizes lung cancer cells to cisplatin via
p53/miR-34a/MYCN axis. Biochem Biophys Res Commun. 482:22–27. 2017.
View Article : Google Scholar
|
|
137
|
Schulz-Heddergott R, Stark N, Edmunds SJ,
Li J, Conradi LC, Bohnenberger H, Ceteci F, Greten FR, Dobbelstein
M and Moll UM: Therapeutic ablation of gain-of-function mutant p53
in colorectal cancer inhibits stat3-mediated tumor growth and
invasion. Cancer Cell. 34:298–314 e297. 2018. View Article : Google Scholar
|
|
138
|
Nakayama M, Sakai E, Echizen K, Yamada Y,
Oshima H, Han TS, Ohki R, Fujii S, Ochiai A, Robine S, et al:
Intestinal cancer progression by mutant p53 through the acquisition
of invasiveness associated with complex glandular formation.
Oncogene. 36:5885–5896. 2017. View Article : Google Scholar
|
|
139
|
Lane DP, Cheok CF and Lain S: p53-based
cancer therapy. Cold Spring Harb Perspect Biol. 2:a0012222010.
View Article : Google Scholar
|
|
140
|
Tsou SH, Hou MH, Hsu LC, Chen TM and Chen
YH: Gain-of-function p53 mutant with 21-bp deletion confers
susceptibility to multidrug resistance in MCF-7 cells. Int J Mol
Med. 37:233–242. 2016. View Article : Google Scholar
|
|
141
|
Li XL, Zhou J, Chen ZR and Chng WJ: P53
mutations in colorectal cancer-molecular pathogenesis and
pharmacological reactivation. World J Gastroenterol. 21:84–93.
2015. View Article : Google Scholar
|
|
142
|
Kwak B, Kim DU, Kim TO, Kim HS and Kim SW:
MicroRNA-552 links Wnt signaling to p53 tumor suppressor in
colorectal cancer. Int J Oncol. 53:1800–1808. 2018.
|
|
143
|
Zhou AD, Diao LT, Xu H, Xiao ZD, Li JH,
Zhou H and Qu LH: β-Catenin/LEF1 transactivates the
microRNA-371-373 cluster that modulates the Wnt/β-catenin-signaling
pathway. Oncogene. 31:2968–2978. 2012. View Article : Google Scholar
|
|
144
|
Wang LQ, Yu P, Li B, Guo YH, Liang ZR,
Zheng LL, Yang JH, Xu H, Liu S, Zheng LS, et al: miR-372 and
miR-373 enhance the stemness of colorectal cancer cells by
repressing differentiation signaling pathways. Mol Oncol.
12:1949–1964. 2018. View Article : Google Scholar
|
|
145
|
Han P, Li JW, Zhang BM, Lv JC, Li YM, Gu
XY, Yu ZW, Jia YH, Bai XF, Li L, et al: The lncRNA CRNDE promotes
colorectal cancer cell proliferation and chemoresistance via
miR-181a-5p-mediated regulation of Wnt/β-catenin signaling. Mol
Cancer. 16:92017. View Article : Google Scholar
|
|
146
|
Xiao Z, Qu Z, Chen Z, Fang Z, Zhou K,
Huang Z, Guo X and Zhang Y: LncRNA HOTAIR is a prognostic biomarker
for the proliferation and chemoresistance of colorectal cancer via
MiR-203a-3p-mediated Wnt/ß-catenin signaling pathway. Cell Physiol
Biochem. 46:1275–1285. 2018. View Article : Google Scholar
|
|
147
|
Wu KF, Liang WC, Feng L, Pang JX, Waye MM,
Zhang JF and Fu WM: H19 mediates methotrexate resistance in
colorectal cancer through activating Wnt/β-catenin pathway. Exp
Cell Res. 350:312–317. 2017. View Article : Google Scholar
|
|
148
|
Deng X, Ruan H, Zhang X, Xu X, Zhu Y, Peng
H, Zhang X, Kong F and Guan M: Long noncoding RNA CCAL transferred
from fibroblasts by exosomes promotes chemoresistance of colorectal
cancer cells. Int J Cancer. 146:1700–1716. 2020. View Article : Google Scholar
|
|
149
|
Ma Y, Yang Y, Wang F, Moyer MP, Wei Q,
Zhang P, Yang Z, Liu W, Zhang H, Chen N, et al: Long non-coding RNA
CCAL regulates colorectal cancer progression by activating
Wnt/β-catenin signalling pathway via suppression of activator
protein 2α. Gut. 65:1494–1504. 2016. View Article : Google Scholar
|
|
150
|
Hanahan D and Coussens LM: Accessories to
the crime: Functions of cells recruited to the tumor
microenvironment. Cancer Cell. 21:309–322. 2012. View Article : Google Scholar
|
|
151
|
Sun Y: Tumor microenvironment and cancer
therapy resistance. Cancer Lett. 380:205–215. 2016. View Article : Google Scholar
|
|
152
|
Castellone MD, Teramoto H, Williams BO,
Druey KM and Gutkind JS: Prostaglandin E2 promotes colon cancer
cell growth through a Gs-axin-beta-catenin signaling axis. Science.
310:1504–1510. 2005. View Article : Google Scholar
|
|
153
|
Yang L, Lin C and Liu ZR: P68 RNA helicase
mediates PDGF-induced epithelial mesenchymal transition by
displacing Axin from beta-catenin. Cell. 127:139–155. 2006.
View Article : Google Scholar
|
|
154
|
Gupta GP and Massague J: Cancer
metastasis: Building a framework. Cell. 127:679–695. 2006.
View Article : Google Scholar
|
|
155
|
Gross JC, Chaudhary V, Bartscherer K and
Boutros M: Active Wnt proteins are secreted on exosomes. Nat Cell
Biol. 14:1036–1045. 2012. View Article : Google Scholar
|
|
156
|
Hu JL, Wang W, Lan XL, Zeng ZC, Liang YS,
Yan YR, Song FY, Wang FF, Zhu XH, Liao WJ, et al: CAFs secreted
exosomes promote metastasis and chemotherapy resistance by
enhancing cell stemness and epithelial-mesenchymal transition in
colorectal cancer. Mol Cancer. 18:912019. View Article : Google Scholar
|
|
157
|
Xu X, Chang W, Yuan J, Han X, Tan X, Ding
Y, Luo Y, Cai H, Liu Y, Gao X, et al: Periostin expression in
intra-tumoral stromal cells is prognostic and predictive for
colorectal carcinoma via creating a cancer-supportive niche.
Oncotarget. 7:798–813. 2016. View Article : Google Scholar
|
|
158
|
Sun Y, Campisi J, Higano C, Beer TM,
Porter P, Coleman I, True L and Nelson PS: Treatment-induced damage
to the tumor microenvironment promotes prostate cancer therapy
resistance through WNT16B. Nat Med. 18:1359–1368. 2012. View Article : Google Scholar
|
|
159
|
Sun Y, Zhu D, Chen F, Qian M, Wei H, Chen
W and Xu J: SFRP2 augments WNT16B signaling to promote therapeutic
resistance in the damaged tumor microenvironment. Oncogene.
35:4321–4334. 2016. View Article : Google Scholar
|
|
160
|
Izumi D, Toden S, Ureta E, Ishimoto T,
Baba H and Goel A: TIAM1 promotes chemoresistance and tumor
invasiveness in colorectal cancer. Cell Death Dis. 10:2672019.
View Article : Google Scholar
|
|
161
|
Takada K, Zhu D, Bird GH, Sukhdeo K, Zhao
JJ, Mani M, Lemieux M, Carrasco DE, Ryan J, Horst D, et al:
Targeted disruption of the BCL9/β-catenin complex inhibits
oncogenic Wnt signaling. Sci Transl Med. 4:148ra1172012. View Article : Google Scholar
|
|
162
|
Tan Z, Huang Q, Zang J, Teng SF, Chen TR,
Wei HF, Song DW, Liu TL, Yang XH, Fu CG, et al: HIF-1α activates
hypoxia-induced BCL-9 expression in human colorectal cancer cells.
Oncotarget. 8:25885–25896. 2017. View Article : Google Scholar
|
|
163
|
Wu X, Gu Z, Chen Y, Chen B, Chen W, Weng L
and Liu X: Application of PD-1 blockade in cancer immunotherapy.
Comput Struct Biotechnol J. 17:661–674. 2019. View Article : Google Scholar
|
|
164
|
Yaghoubi N, Soltani A, Ghazvini K,
Hassanian SM and Hashemy SI: PD-1/PD-L1 blockade as a novel
treatment for colorectal cancer. Biomed Pharmacother. 110:312–318.
2019. View Article : Google Scholar
|
|
165
|
Payandeh Z, Khalili S, Somi MH,
Mard-Soltani M, Baghbanzadeh A, Hajiasgharzadeh K, Samadi N and
Baradaran B: PD-1/PD-L1-dependent immune response in colorectal
cancer. J Cell Physiol. 235:5461–5475. 2020. View Article : Google Scholar
|
|
166
|
Topalian SL, Taube JM, Anders RA and
Pardoll DM: Mechanism-driven biomarkers to guide immune checkpoint
blockade in cancer therapy. Nat Rev Cancer. 16:275–287. 2016.
View Article : Google Scholar
|
|
167
|
Galon J, Costes A, Sanchez-Cabo F,
Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M,
Berger A, Wind P, et al: Type, density, and location of immune
cells within human colorectal tumors predict clinical outcome.
Science. 313:1960–1964. 2006. View Article : Google Scholar
|
|
168
|
Tumeh PC, Harview CL, Yearley JH, Shintaku
IP, Taylor EJ, Robert L, Chmielowski B, Spasic M, Henry G, Ciobanu
V, et al: PD-1 blockade induces responses by inhibiting adaptive
immune resistance. Nature. 515:568–571. 2014. View Article : Google Scholar
|
|
169
|
Spranger S, Koblish HK, Horton B, Scherle
PA, Newton R and Gajewski TF: Mechanism of tumor rejection with
doublets of CTLA-4, PD-1/PD-L1, or IDO blockade involves restored
IL-2 production and proliferation of CD8+ T cells
directly within the tumor microenvironment. J Immunother Cancer.
2:32014. View Article : Google Scholar
|
|
170
|
Spranger S, Dai D, Horton B and Gajewski
TF: Tumor-residing Batf3 dendritic cells are required for effector
t cell trafficking and adoptive t cell therapy. Cancer Cell.
31:711–723. 2017. View Article : Google Scholar
|
|
171
|
Feng M, Jin JQ, Xia L, Xiao T, Mei S, Wang
X, Huang X, Chen J, Liu M, Chen C, et al: Pharmacological
inhibition of β-catenin/BCL9 interaction overcomes resistance to
immune checkpoint blockades by modulating T reg. Sci
Adv. 5:eaau52402019. View Article : Google Scholar
|
|
172
|
Bottcher JP and Reis e Sousa C: The role
of type 1 conventional dendritic cells in cancer immunity. Trends
Cancer. 4:784–792. 2018. View Article : Google Scholar
|
|
173
|
Liu K and Li J, Wu X, Chen M, Luo F and Li
J: GSK-3β inhibitor 6-bromo-indirubin-3′-oxime promotes both
adhesive activity and drug resistance in colorectal cancer cells.
Int J Oncol. 51:1821–1830. 2017. View Article : Google Scholar
|
|
174
|
Yan L, Yu HH, Liu YS, Wang YS and Zhao WH:
Esculetin enhances the inhibitory effect of 5-Fluorouracil on the
proliferation, migration and epithelial-mesenchymal transition of
colorectal cancer. Cancer Biomark. 24:231–240. 2019. View Article : Google Scholar
|
|
175
|
Cai MH, Xu XG, Yan SL, Sun Z, Ying Y, Wang
BK and Tu YX: Regorafenib suppresses colon tumorigenesis and the
generation of drug resistant cancer stem-like cells via modulation
of miR-34a associated signaling. J Exp Clin Cancer Res. 37:1512018.
View Article : Google Scholar
|
|
176
|
Siraj AK, Kumar Parvathareddy S,
Pratheeshkumar P, Padmaja Divya S, Ahmed SO, Melosantos R, Begum R,
Concepcion R, Al-Sanea N, Ashari LH, et al: APC truncating
mutations in middle eastern population: Tankyrase inhibitor is an
effective strategy to sensitize APC mutant CRC To 5-FU
chemotherapy. Biomed Pharmacother. 121:1095722020. View Article : Google Scholar
|
|
177
|
Wang J, Min H, Hu B, Xue X and Liu Y:
Guanylate-binding protein-2 inhibits colorectal cancer cell growth
and increases the sensitivity to paclitaxel of paclitaxel-resistant
colorectal cancer cells by interfering Wnt signaling. J Cell
Biochem. 121:1250–1259. 2020. View Article : Google Scholar
|
|
178
|
Wu CE, Zhuang YW, Zhou JY, Liu SL, Wang RP
and Shu P: Cinnamaldehyde enhances apoptotic effect of oxaliplatin
and reverses epithelial-mesenchymal transition and stemnness in
hypoxic colorectal cancer cells. Exp Cell Res. 383:1115002019.
View Article : Google Scholar
|
|
179
|
Zhao B, Wang L, Qiu H, Zhang M, Sun L,
Peng P, Yu Q and Yuan X: Mechanisms of resistance to anti-EGFR
therapy in colorectal cancer. Oncotarget. 8:3980–4000. 2017.
View Article : Google Scholar
|
|
180
|
Sansom OJ, Meniel V, Wilkins JA, Cole AM,
Oien KA, Marsh V, Jamieson TJ, Guerra C, Ashton GH, Barbacid M and
Clarke AR: Loss of Apc allows phenotypic manifestation of the
transforming properties of an endogenous K-ras oncogene in vivo.
Proc Natl Acad Sci USA. 103:14122–14127. 2006. View Article : Google Scholar
|
|
181
|
He L, Zhu H, Zhou S, Wu T, Wu H, Yang H,
Mao H, SekharKathera C, Janardhan A, Edick AM, et al: Wnt pathway
is involved in 5-FU drug resistance of colorectal cancer cells. Exp
Mol Med. 50:1012018. View Article : Google Scholar
|
|
182
|
Zhao Q, Zhuang K, Han K, Tang H, Wang Y,
Si W and Yang Z: Silencing DVL3 defeats MTX resistance and
attenuates stemness via Notch signaling pathway in colorectal
cancer. Pathol Res Pract. 216:1529642020. View Article : Google Scholar
|
|
183
|
Zhang F, Sun H, Zhang S, Yang X, Zhang G
and Su T: Overexpression of PER3 inhibits self-renewal capability
and chemoresistance of colorectal cancer stem-like cells via
inhibition of notch and β-catenin signaling. Oncol Res. 25:709–719.
2017. View Article : Google Scholar
|