Introduction
Pulmonary hypertension (PH) is a progressive and
life-threatening disease characterized by increased pulmonary
artery pressure and pulmonary vascular resistance. PH can be
divided into five clinical types according to different etiologies
(1). However, different categories
of PH share a common pathogenesis of vascular remodeling involving
thickening of pulmonary vasculature and invasive proliferation of
smooth muscle cells (2). Pulmonary
smooth muscle cell (PASMC) proliferation and migration to the
intima of pulmonary arteries serve a critical role in the process
of pulmonary vascular remodeling in PH (3).
The underlying mechanism and genes associated with
PASMC migration in PH have not been fully elucidated. Enhancer of
zeste homolog 2 (EZH2), a histone methyltransferase highly
expressed in multiple types of cancer, such as thyroid and prostate
cancer (4,5), promotes tumor cell proliferation and
migration via epigenetic regulation of cancer-associated gene
expression levels (6). EZH2
expression levels are also associated with tumor invasiveness
(7). Considering that epigenetic
alterations have been reported in certain types of PH that can be
regarded as a cancer-like disease, EZH2 may be involved in the
pulmonary arterial remodeling process in PH (8–10).
EZH2 has been demonstrated to be associated with hypoxia-induced PH
in mice and participates in the proliferation and migration of
PASMCs (11). To the best of our
knowledge, however, there is little evidence to demonstrate how
EZH2 functions in the pathogenesis of PASMC migration in PH.
Hypoxic pulmonary arterial hypertension (HPAH), a
common type of PH, is caused by high altitude or persistent hypoxic
conditions induced by pulmonary disease, particularly chronic
obstructive pulmonary disease (1).
Previous studies on HPAH demonstrated that structural remodeling in
pulmonary arteries contributes to its development and occur over a
relatively short time period (12,13).
Chronic thromboembolic pulmonary hypertension (CTEPH) is another
important form of PH characterized by one or multiple episodes of
pulmonary embolism. Remodeling of distal pulmonary vessels
following initial pulmonary thromboembolism may be responsible for
the formation of CTEPH (14). The
present study analyzed hypoxia-induced PASMCs and those isolated
from the endarterectomized tissue of patients with CTEPH. In order
to determine the role of EZH2 in PH, the effect of EZH2 on PASMC
migration was investigated using human CTEPH cell models. Pathways
that contribute to this process following EZH2 overexpression were
also identified.
The present results revealed high expression levels
of EZH2 in hypoxia-induced and CTEPH PASMCs. Furthermore, EZH2
affected PASMC migration. The present study also reported
genome-wide characteristics of EZH2 overexpression. These results
may provide insight into the mechanism associated with PH artery
remodeling.
Materials and methods
Ethics approval
The present study was approved by the Research
Ethics Committee of the Beijing Chao-Yang Hospital of Capital
Medical University. Written informed consent was provided by all
patients before the procedure was initiated.
Cell preparation
Normal human PASMC line (N-PASMC; cat. no. 3110) was
purchased from ScienCell Research Laboratories, Inc. and used for
EZH2 transfection. Cells were cultured in smooth muscle cell growth
medium (SMCM) (ScienCell Research Laboratories, Inc.), including
smooth muscle basal medium, FBS (2%), smooth muscle cell growth
supplement (1%), penicillin (200 µg/ml) and streptomycin (200
IU/ml). All cells were incubated in a 95% humidified incubator at
37°C with 5% CO2 and 21% O2 and passaged
after reaching 80–90% confluence.
A total of three consecutive patients with CTEPH
diagnosed by pulmonary angiography and right heart catheterization
at Beijing Chao-Yang Hospital (Beijing, China) between January 2013
and December 2014 were enrolled in the study. The patients included
two male patients and one female patient, aged between 49 and 61
years. All patients diagnosed with CTEPH met the following criteria
after 3 months of effective anticoagulation treatment: Mean
pulmonary arterial pressure ≥25 mmHg with normal wedge pressure ≤15
mmHg and at least one pulmonary segmental perfusion defect revealed
by lung perfusion scanning or pulmonary artery occlusion detected
by multi-detector computed tomography angiography or pulmonary
angiography. The World Health Organization (WHO) classification was
used to classify patients (15).
Exclusion criteria included ventriculo-atrial shunt, malignancies,
and other lung diseases. Clinical data were recorded.
PASMCs from patients with CTEPH were isolated and
cultured as previously described (16,17).
Briefly, pulmonary endarterectomy resection tissues from patients
were cut into 1 mm3 pieces and then centrifuged at 300 ×
g for 5 min at room temperature. Following resuspension in SMCM,
the resultant suspension was seeded in dishes and incubated in a
95% humidified incubator at 37°C with 5% CO2 for 1 week
until PASMCs were observed from adherent tissue pieces. The PASMC
phenotypes of isolated cells were characterized by
immunofluorescence using monoclonal antibodies against human
α-smooth muscle actin (α-SMA; 1:200; cat. no. ab124964; Abcam) and
smooth muscle myosin heavy chain (1:100; cat. no. sc-6956; Santa
Cruz Biotechnology, Inc.) respectively, for 1 h at room
temperature. All cells were passaged after reaching 80–90%
confluence prior to measurement of EZH2 expression levels.
Transient transfection of PASMCs
For EZH2 overexpression, the EZH2 gene was
subcloned into the pEZ-M98 vector (GeneCopoeia, Inc.) to generate
pM-EZH2. Cultured N-PASMCs were then transfected with pM-EZH2
vector using the Neon Transfection System (MPK5000) from Invitrogen
(Thermo Fisher Scientific, Inc.), according to the supplier's
instructions. Briefly, cells were cultured in 60-mm dishes to 70%
confluency and then transfected with 2 µg plasmid DNA (1,375 V; 20
ms) at room temperature. Empty vector pEZ-M98 (pM) was used as a
transfection control.
For EZH2 knockdown, PASMCs from CTEPH patient 3 were
cultured in 60-mm dishes and transfected with 160 pmol small
interfering (si)RNA (Shanghai GenePharma Co., Ltd.) and treated
with Lipofectamine RNAiMAX reagent from Invitrogen (Thermo Fisher
Scientific, Inc.) at room temperature, according to the
manufacturer's instructions. The sequences were sense,
5′-CCUGACCUCUGUCUUACUUTT-3′ and antisense,
5′-AAGUAAGACAGAGGUCAGGTT-3′. RNAi negative control that had no
homology with mammalian genes was used as the transfection control.
The RNAi negative control sequences were sense,
5′-UUCUCCGAACGUGUCACGUTT-3′ and antisense,
5′-ACGUGACACGUUCGGAGAATT-3′. After 48 h, transfection efficiency
was determined by both PCR and western blot analysis for EZH2.
Wound healing assay
At 12 h after transfection, the transfection mixture
was replaced with 0.2% FBS, and a straight scratch was created
using a 200-µl pipette tip on a monolayer of confluent PASMCs.
Images were captured at 0 and 12 h after scratching to visualize
migrated cells and wound healing using phase microscopy
(magnification, ×40). The distance of cell movement from the wound
edge into the wound area indicated the extent of cell migration. A
total of 10 points, evenly spaced, were selected on each edge, and
the minimum distance to the scratch edge was measured. The cell
migration rate was calculated with the following formula: [Scratch
width (0 h)-scratch width (12 h)]/scratch width (0 h) ×100%. The
experiment was repeated three times, and the average value was
calculated.
Gene microarray analysis
In order to evaluate the effect of EZH2 on PASMCs,
total RNA was isolated using TRIzol (Invitrogen; Thermo Fisher
Scientific, Inc.) following EZH2 overexpression. mRNA expression
levels were profiled using mRNA + lncRNA Human Gene Expression
Microarray V4.0 (CapitalBio Corporation) according to the
manufacturer's instructions. For microarray analysis, Agilent
Feature Extraction (V10.7; Agilent Technologies, Inc.) was used for
data extraction and quantification. Then, raw data were summarized
and normalized at the transcript level using the GeneSpring GX
program (V12.0; Agilent Technologies, Inc.). The unpaired t-test
was applied to filter genes with differential expression in the
control vs. experimental groups. Fold-change in differentially
expressed mRNAs >2.0 and P<0.05 were considered to indicate a
statistically significant difference between experimental and
control groups. Certain differentially expressed genes were then
validated by reverse transcription-quantitative (RT-q)PCR.
Gene Ontology (GO) and pathway
functional enrichment analysis
Following microarray screening for mRNAs from PASMCs
transfected with pM-EZH2 and controls, the differentially expressed
genes were grouped into functional categories by performing GO
enrichment analysis according to the GO database (geneontology.org/), which categorizes genes into
regulatory networks on the basis of ‘Biological Process’,
‘Molecular Function’ and ‘Cellular Function’. The P-value of the
significance level for each gene with differential expression was
estimated with Fisher's exact test. P<0.05 was considered to
indicate a statistically significant difference.
Pathway enrichment analysis was performed using the
Kyoto Encyclopedia of Genes and Genomes (KEGG) (18), BioCarta (19) and Reactome databases (20) with KOBAS software (version 3.0)
(21). Significant enrichments were
calculated and filtered by P<0.05.
RT-qPCR
Relative expression levels of EZH2, superoxide
dismutase 3 (SOD3) and NADPH oxidase 1 (NOX1) were determined by
RT-qPCR analysis. Total RNA was isolated from PASMCs with EZH2
transfection using TRIzol® (Invitrogen; Thermo Fisher
Scientific, Inc.). RT was conducted using a ReverTra Ace qPCR kit
(Toyobo Life Science). Briefly, 0.5 µg total RNA was first reverse
transcribed using reverse transcriptase Mix at the following
conditions: 37°C for 15 min and 98°C for 5 min; 4°C. qPCR
quantification was performed as previously described (15). All experiments were performed in
triplicate. The primer sequences were: EZH2 forward,
5′-AATCATGGGCCAGACTGGGAAGAA-3′ and reverse,
5′-TCTTGAGCTGTCTCAGTCGCATGT-3′; SOD3 forward,
5′-GGCCTCCATTTGTACCGAAA-3′ and reverse, 5′-AGGGTCTGGGTGGAAAGGT-3′;
NOX1 forward, 5′-CCCCAAGTCTGTAGTGGGAGTT-3′ and reverse,
5′-CGCAGGCTCTTTGCCAAA-3′; and GAPDH forward,
5′-TGACTTCAACAGCGACACCCA-3′ and reverse,
5′-CACCCTGTTGCTGTAGCCAAA-3′. Human GAPDH was used as an endogenous
control to normalize gene expression levels.
Statistical analysis
All experiments were performed in triplicate. Data
are presented as the mean ± SD. Data were analyzed using SPSS
software (version 13.0; SPSS, Inc.) Statistical significance was
determined using an unpaired Student's t-test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Increased EZH2 expression levels in
PASMCs derived from patients with CTEPH
In order to analyze EZH2 expression levels, PASMCs
were isolated from tissue obtained during endarterectomy from 3
patients with CTEPH. The clinical information for patients is
summarized in Table SI. The mRNA
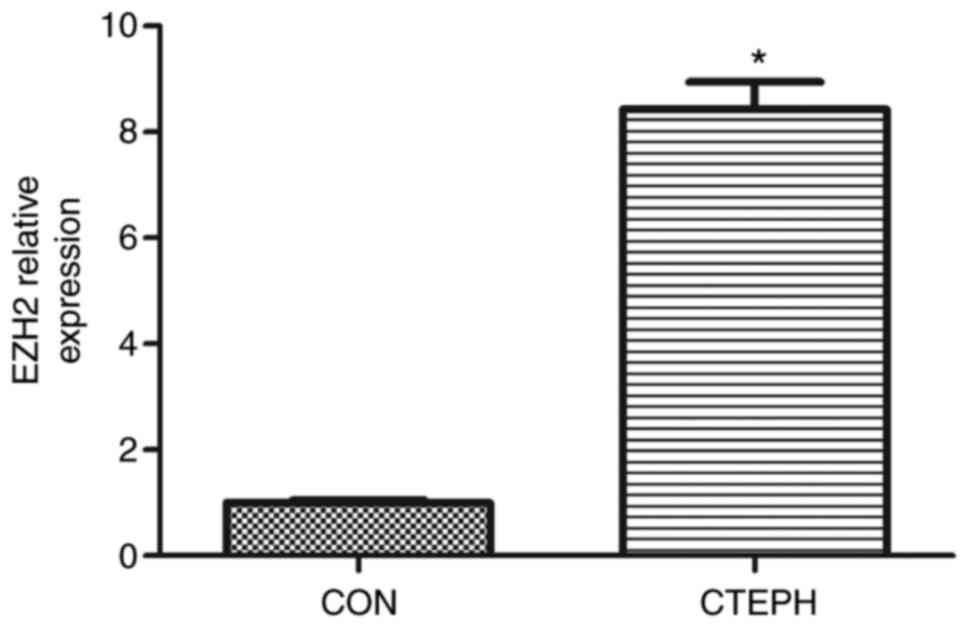
expression levels of EZH2 were assessed in PASMCs from CTEPH
patients. EZH2 mRNA expression levels were significantly increased
in PASMCs from CTEPH patients, compared with control PASMCs
(Fig. S1). PASMCs from CTEPH
patient 3 exhibited the strongest EZH2 expression levels among the
patients (Fig. 1 and Table SII).
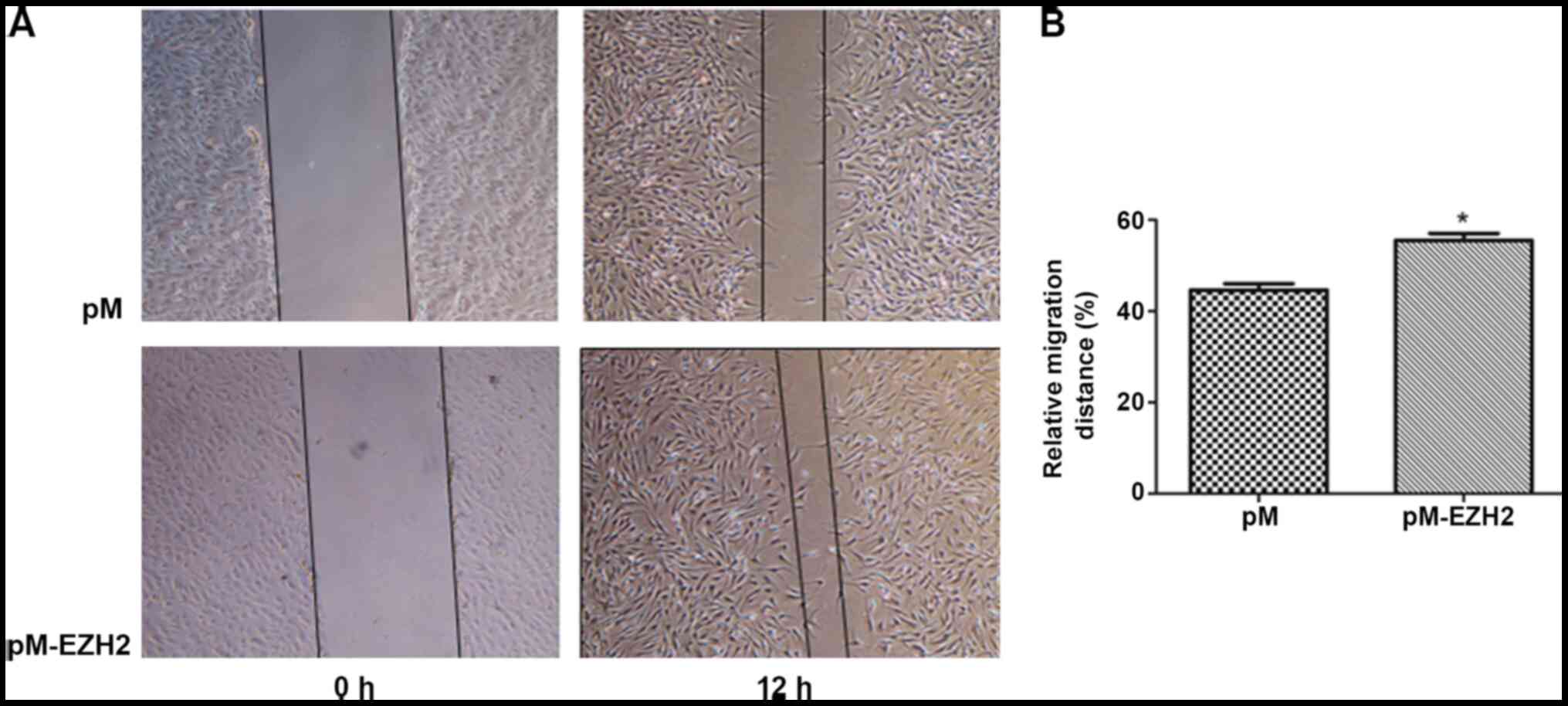
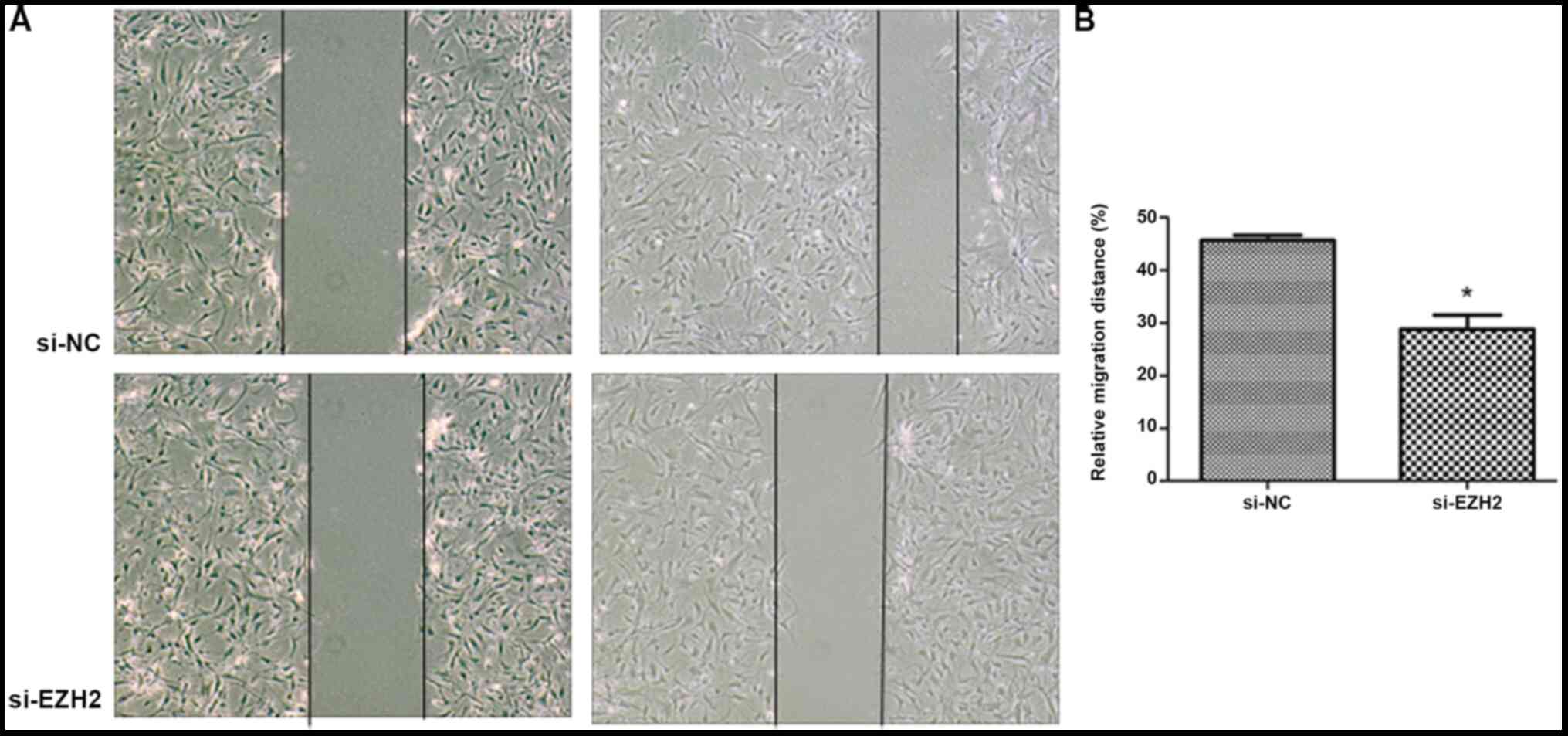
EZH2 affects PASMC migration
EZH2 expression levels were determined by western
blot analysis following overexpression by pM-EZH2 vector and
knockdown by siRNA (Fig. S2). The
role of the EZH2 gene in affecting PASMC migration was then
investigated by performing a wound healing assay. Following 12-h
wounding, the gap scratched by a pipette tip was filled with more
cells in the EZH2-overexpression group compared with the control.
However, the distance of the scratch between the migration edges of
the cells was significantly wider in the EZH2 interference group,
compared with the control. The migration distance of N-PASMCs
following EZH2 transfection was increased compared with that
observed for the control. Silencing of EZH2 by siRNA significantly
inhibited the migration of PASMCs derived from patients with CTEPH
(Figs. 2 and 3). This indicated that EZH2 increased the
migration of PASMCs and participated in the pathogenesis of
PAH.
EZH2-associated differences in gene
expression levels
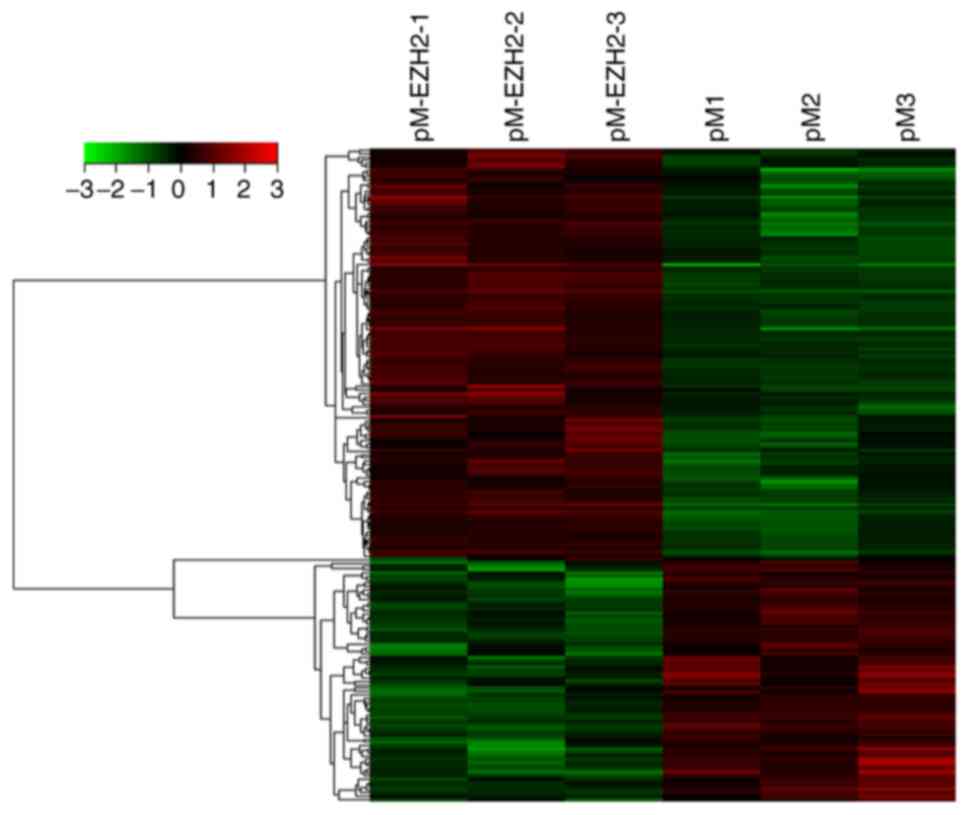
N-PASMCs transfected with EZH2 and control were
collected and microarray analysis was used to investigate changes
in gene expression levels. A data set from the microarray was
subjected to supervised hierarchical clustering analysis. Samples
were divided into two groups based on differential expression
levels of genes (Fig. 4). A total
of 192 genes with statistically significant changes were detected.
Among these, 122 genes were upregulated and 70 were downregulated
following EZH2 transfection. The top 10 significantly up- or
downregulated genes, including oncostatin M (OSM), Nod-like
receptor (NLR)P12, IL-31, serum amyloid A2 (SAA2) and arachidonate
5-lipoxygenase activating protein (ALOX5AP), according to P-value
are summarized in Table I.
 | Table I.Top 10 significantly altered
genes. |
Table I.
Top 10 significantly altered
genes.
| A, Upregulated
genes |
|---|
|
|---|
| Gene symbol | P-value | Fold-change |
|---|
| OSM | 0.001 | 6.015 |
| FSHR | 0.009 | 4.793 |
| NLRP12 | 0.008 | 4.294 |
| LOC100996286 | <0.001 | 4.010 |
| LOC100131907 | <0.001 | 3.969 |
| CFAP61 | 0.001 | 3.876 |
| IL31 | 0.016 | 2.953 |
| SLC40A1 | 0.042 | 2.819 |
| CRX | 0.013 | 2.809 |
| lnc-DFNA5-3 | 0.006 | 2.675 |
|
| B, Downregulated
genes |
|
| Gene
symbol | P-value |
Fold-change |
|
| SAA2 | 0.013 | 4.615 |
| C3orf67 | 0.015 | 4.593 |
| ABCC6 | 0.013 | 3.744 |
| SLC38A4 | 0.040 | 3.404 |
| TTN | 0.009 | 3.349 |
| ALOX5AP | 0.047 | 3.161 |
| LOC101929689 | 0.001 | 3.132 |
| C1orf229 | 0.011 | 3.132 |
| AP1S2 | 0.001 | 3.025 |
| lnc-CBWD5-1 | 0.010 | 3.022 |
GO and KEGG functional enrichment
analysis reveals different genes are involved in inflammatory and
immune processes
GO analysis of Cell Component revealed that
differentially expressed genes were primarily enriched in
‘membranous components’ (data not shown). The GO analysis of
Molecular Function revealed differentially expressed genes
associated with ‘hydrogen ion channel activity’ and ‘oxidoreductase
activity’ (data not shown). GO analysis of ‘Biological Process’, as
shown in Table II, revealed a
strong signature for ‘cellular response to pH’, ‘inflammation and
the immune response’ related functions. Of the top 20 significant
GO terms, 7 (35%) were concerned with ‘cellular response to pH’ or
‘response to metal ion’; 8 (40%) were involved in ‘regulation of
interleukin-6 biosynthetic process’, ‘interleukin-6 biosynthetic
process’, ‘negative regulation of interleukin-6 biosynthetic
process’, ‘immune system process’ and ‘humoral immune response
mediated by circulating immunoglobulin’ (Table II). NLRP12, toll-like receptor 9,
complement C1q (C1Q)B and C chain were markedly enriched in these
GO terms.
 | Table II.Top 20 significant GO terms of
differential genes. |
Table II.
Top 20 significant GO terms of
differential genes.
| GO term | Genes | P-value |
|---|
| Cellular response
to pH | HVCN1, KD1L3,
NOX1 | 0.0002 |
| Response to metal
ion | TTN, SLC18A2, SOD3,
HVCN1, CYP11B1, SLC40A1, ZACN, ALOX5AP | 0.0003 |
| Negative regulation
of interleukin-6 biosynthetic process | PRG4, NLRP12 | 0.0008 |
| Regulation of
interleukin-18 production | TLR9, NLRP12 | 0.0008 |
| Response to
transition metal nanoparticle | HVCN1, ZACN,
SLC18A2, SOD3, SLC40A1 | 0.0008 |
| Negative regulation
of interleukin-6 production | PRG4, NLRP12,
TLR9 | 0.0011 |
| Interleukin-18
production | TLR9, NLRP12 | 0.0011 |
| Response to pH | HVCN1, PKD1L3,
NOX1 | 0.0012 |
| Cellular response
to acidic pH | PKD1L3, NOX1 | 0.0017 |
| Positive regulation
of response to wounding | TLR9, OSM, NLRP12,
ANO6, CPB2 | 0.0021 |
| Response to zinc
ion | HVCN1, ZACN,
SLC18A2 | 0.0025 |
| Humoral immune
response mediated by circulating immunoglobulin | C1QC, C1QB,
HLA | 0.0032 |
| Response to acidic
pH | PKD1L3, NOX1 | 0.0039 |
| Mitotic chromosome
condensation | TTN, CDCA5 | 0.0039 |
| Response to
inorganic substance | TTN, SLC18A2, SOD3,
HVCN1, CYP11B1, SLC40A1, ZACN, ALOX5AP | 0.0039 |
| Immune system
process | PSMB11, RAB17,
SLC7A10, PRG4, ANO6, PRKCG, CYP11B1, SLC40A1, INPP4B, IGSF6, C1QC,
OSM, C1QB, TLR9, CD160, PYDC1, DPP4, HLA, MLF1, GCSAM, CCL3L3,
IL31 |
|
| Regulation of
interleukin-6 biosynthetic process | PRG4, NLRP12 | 0.0051 |
| Cellular response
to metal ion | HVCN1, ALOX5AP,
CYP11B1, SLC40A1 | 0.0051 |
| Photoreceptor cell
differentiation | CEP290, CRX,
MYO7A | 0.0053 |
| Interleukin-6
biosynthetic process | PRG4, NLRP12 | 0.0062 |
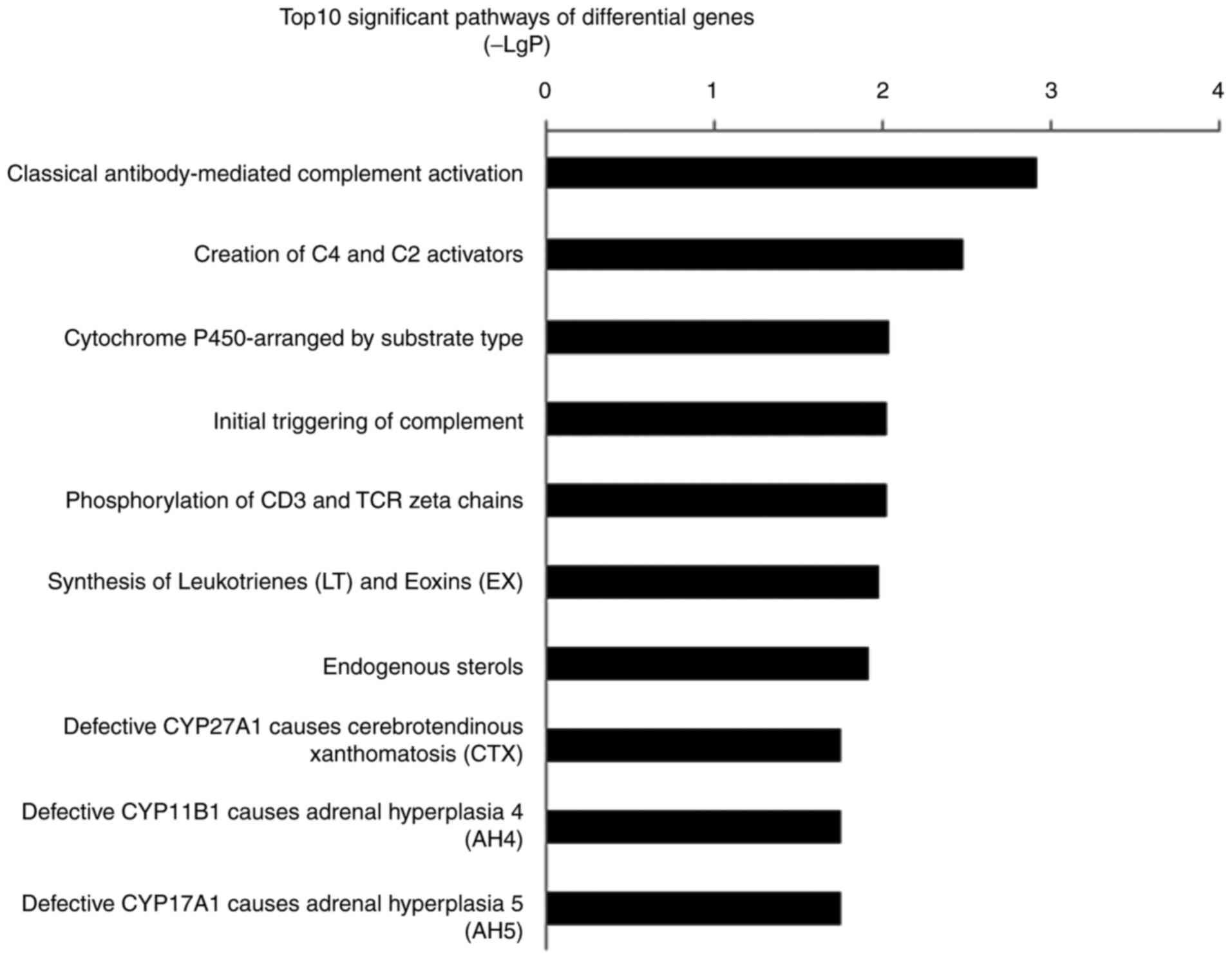
Pathway analysis of differentially expressed genes
also implicated inflammation and the immune response-related
pathways were involved in PASMCs migration mediated by EZH2. The
most significant pathway was ‘classical antibody-mediated
complement activation’ (P=0.001). The second most significant
pathway, ‘Creation of C4 and C2 activators’, and fourth most
significant pathway, ‘Initial triggering of complement’, were also
associated with complement-associated pathways (P=0.03 and 0.09,
respectively). The top 10 significant pathways included
inflammatory and immune response mechanisms such as
‘phosphorylation of CD3 and TCR ζ chains’, ‘synthesis of
leukotrienes (LT) and eoxins (EX)’ and the ‘cytochrome
P450-arranged by substrate type’ (Fig.
5). The signature genes that regulated inflammatory and immune
response processes included C1QC, CYP4F8 and ALOX5AP.
Validation of gene expression
levels
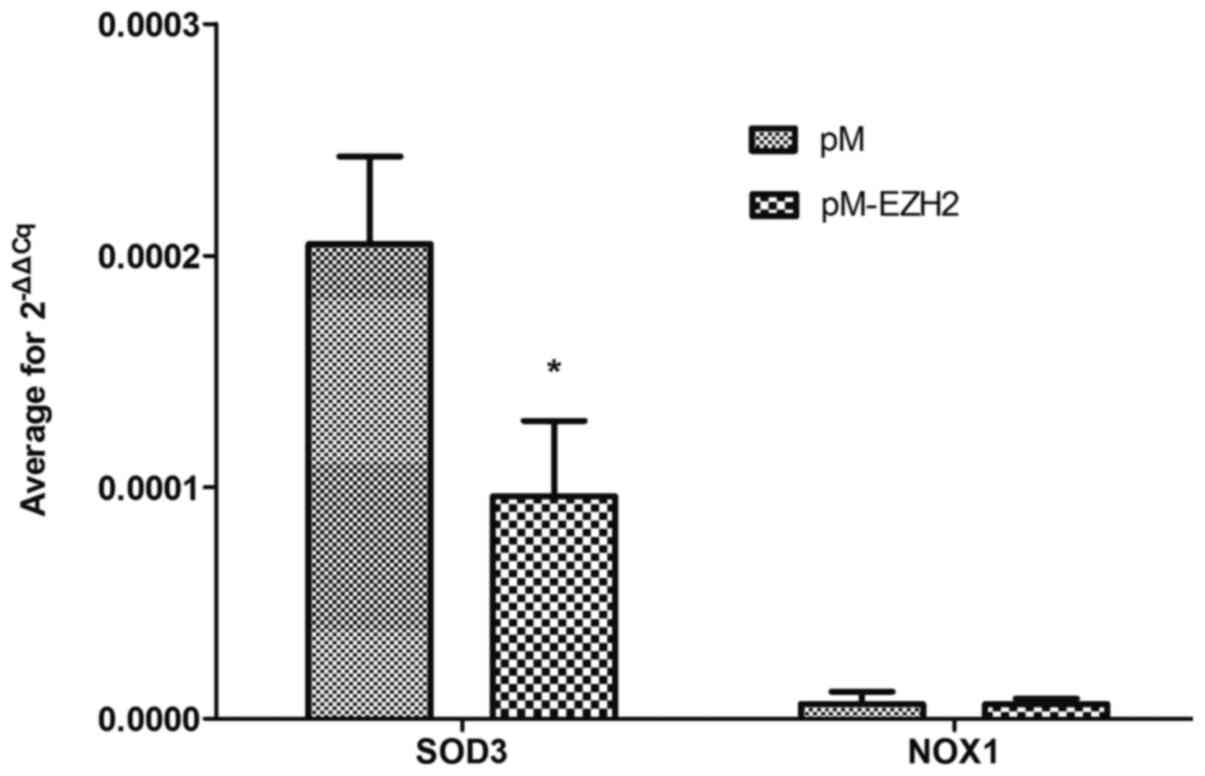
SOD3 and NOX1 were selected for validation of mRNA
expression levels via qPCR because functional analysis revealed
that both genes were significantly enriched following EZH2
transfection. qPCR demonstrated that the mRNA expression level of
SOD3 was decreased compared with the control (Fig. 6 and Table SIII), confirming the results of the
microarray detection. However, NOX1 expression levels were not
significantly altered.
Discussion
PASMCs are primary mediators of small pulmonary
arterial remodeling, which is an important pathological process in
the progression of PH. The proliferation and migration of PASMCs
serve key roles in this process (22). Epigenetic changes are involved in
the development of certain types of PH (10). Furthermore, our laboratory
previously revealed that DNA methylation changes in PASMCs are
associated with the pathogenesis of CTEPH (10,15).
In the present study, upregulation of EZH2 was observed in PASMCs
from patients with CTEPH and those induced by hypoxic conditions.
Moreover, EZH2 affected the migration ability of N-PASMCs. Gene
chips were used to identify the mechanism following EZH2
overexpression in N-PASMCs. Functional enrichment analysis revealed
that pathophysiological pathways that participated in inflammatory
and immune processes were controlled by EZH2-associated genes. The
present study provides further understanding into vascular
remodeling mechanisms underlying PH.
EZH2 is a conserved methyltransferase of histone
H3K27, which promotes cell proliferation, migration and
invasiveness in tumorigenesis via epigenetic modifications
(23,24). HPAH and CTEPH are two common types
of PH (1). PASMCs in HPAH and CTEPH
exhibit proliferation and migration phenotypes similar to those of
cancer cells (25,26). In the present study, EZH2 expression
levels were increased in both hypoxia-induced and patient-derived
PASMCs, which is consistent with a previous study in a
hypoxia-induced PAH mouse model (11). However, unlike CTEPH, HPAH is rarely
treated with surgery, thus tissue samples from patients with HPAH
are difficult to obtain. Hypoxia-induced human PASMCs were
therefore used instead of HPAH tissue samples for the measurement
of EZH2 mRNA expression levels. The present study did not
investigate EZH2 mRNA expression levels in HPAH and only focused on
CTEPH because EZH2 mRNA levels could not be measured in cells from
HPAH tissue. Increased migration ability of N-PASMCs overexpressing
EZH2 and decreased migration ability of patient-derived PASMCs
following EZH2 knockdown were confirmed, which was in line with
previously reported changes in hypoxia-induced HPASMCs and cancer
cells (11,27). Collectively, the present findings
indicated that EZH2 may serve a role in the migration of PASMCs in
PH vascular remodeling. However, no changes in the proliferation or
apoptosis of PASMCs following EZH2 transfection (data not shown)
were detected in the present study, which was inconsistent with
findings previously reported by Aljubran et al (11). It was speculated that different
PASMC phenotypes may partly explain this difference: Previous
studies have demonstrated that there may be multiple phenotypically
distinct SMC populations in pulmonary arteries, and these distinct
populations may serve different functions (28,29).
EZH2-associated changes in gene expression levels
and pathophysiological signaling pathways were investigated in the
present study. The results identified a number of candidate genes,
as well as pathophysiological pathways targeted by oxidoreductase
activity, inflammation and immune processes, that provide a basis
for further investigation of the mechanism of EZH2 in PH vascular
remodeling. The present results revealed that three of the top 10
significantly upregulated genes (OSM, NLRP12 and IL-31) were
closely associated with inflammatory and immune responses. As the
most differentially upregulated gene, OSM is a secreted member of
the IL-6 family and serves an important role in maintaining
homeostasis of the internal environment under chronic inflammation
(30). NLRP12 belongs to
non-classical NLRP molecules and has been reported to regulate
inflammation and tumorigenesis (31,32).
IL-31 is a cytokine belonging to the IL-6 family and shares a
common signaling receptor subunit with OSM (33). IL-6 has been reported to be involved
in airway inflammation and remodeling in vascular remodeling
(34,35). As is true for a number of proteins
in the IL-6 family, the roles of IL-31 and OSM in vascular
remodeling remain unclear. The microarray analysis identified SAA2
and ALOX5AP among the 10 most downregulated genes. This result
appeared to contradict the proinflammatory roles of SAA2 and
ALOX5AP reported in previous studies, which have established that
SAA is expressed at increased levels in numerous inflammatory
conditions, including trauma, infection and tumor growth, and
secreted by the liver as an acute-phase protein (36–38).
However, the potential roles of local SAA variants remain to be
elucidated. De Buck et al (39) described the extrahepatic production
of SAA variants by a number of types of tissues, such as brain and
colon tissues, with differential expression levels of SAA1 and
SAA2. Jumeau et al (40)
also identified SAA1 as an important SAA acute-phase gene induced
in human monocytes and derived macrophages in the presence of
stimulants. The ALOX5AP gene is required for LT synthesis, which is
involved in numerous types of inflammatory response (37). The present microarray analysis
revealed downregulated expression levels of ALOX5AP (P=0.047). As
demonstrated by Wang et al (41), microarrays may not provide accurate
results when used to measure differential gene expression at levels
near the threshold for detection. No significant change in the
expression levels of ALOX5AP was detected (data not shown).
Therefore, results reported in relation to ALOX5AP expression
levels require further study.
The present results demonstrated significant
downregulation of SOD3 levels. In 2010, Archer et al
(9) confirmed that deficiency of
SOD2 causes PAH by impairing redox signaling and inducing PASMC
phenotype transition, which is highly associated with epigenetic
regulation by EZH2. Soon et al (42) also demonstrated that BMPR-II
deficiency instigates the development of PAH by decreasing SOD3
expression levels and exaggerating the inflammatory response both
in vitro and in vivo. Therefore, the role of EZH2 in
PASMC migration may be associated with redox and inflammation
reactions.
GO term enrichment analysis confirmed the
aforementioned observations. GO analysis revealed immune system
processes that involve IL-6, −18 and −1 and NF-κB regulation and
complement activation predominated. Increasing evidence has
confirmed that these inflammatory factors promote vascular
remodeling in PH (43–45). Significantly enriched GO terms also
included cellular response to hydrogen potential, pH and
oxidoreductase activity, which are associated with abnormal energy
metabolism in mitochondria, reactive oxygen species (ROS) and an
acidic microenvironment. A potential mechanism of cancer-like
abnormalities such as a PASMC phenotype shift in PH may be
mitochondrial disorder and glycolysis, which is known as the
Warburg phenotype in tumors (46–48).
In PAH and cancer, abnormal mitochondrial metabolism and redox
signaling lead to decreased ROS levels and a shift from oxidative
to glycolytic metabolism (49).
There is a close association between ROS and the occurrence and
development of inflammation (50).
Sutendra et al (51)
reported that abnormalities of mitochondrial metabolism and
decreased ROS levels are linked to the chronic inflammatory process
in PAH and progressive expansion of SMC-like cells that obliterate
the vascular lumen. SOD has been recognized as a redox-signaling
molecule that regulates pulmonary vascular tone and structure and
may have therapeutic potential in inflammation (52). Soon et al (42) reported that SOD3 loss is necessary
for the proinflammatory phenotype in PAH. The aforementioned
studies suggest that there may be an important interplay between
ROS and inflammation in SMC. Therefore, we hypothesize that EZH2
may lead to epigenetic repression of SOD3, thus promoting PASMC
migration in PAH via inflammatory and immune processes. This chain
of events is consistent with the pathogenic mechanism of PAH
(53).
Further pathway analysis supported these
inflammatory changes, including effects on CD3 and TCR ζ chains and
LT synthesis. Pathway analysis also identified a role for
complement-associated pathways in the EZH2-mediated migration of
PASMCs. A previous study revealed that C3 complement contributes to
the development of hypoxia-induced PH in mice (54), and this process is associated with
pulmonary vascular remodeling. C3 complement levels are
significantly altered in patients with idiopathic PAH (55). In CTEPH, the inflammatory response
has also been revealed to play a critical role (56–58).
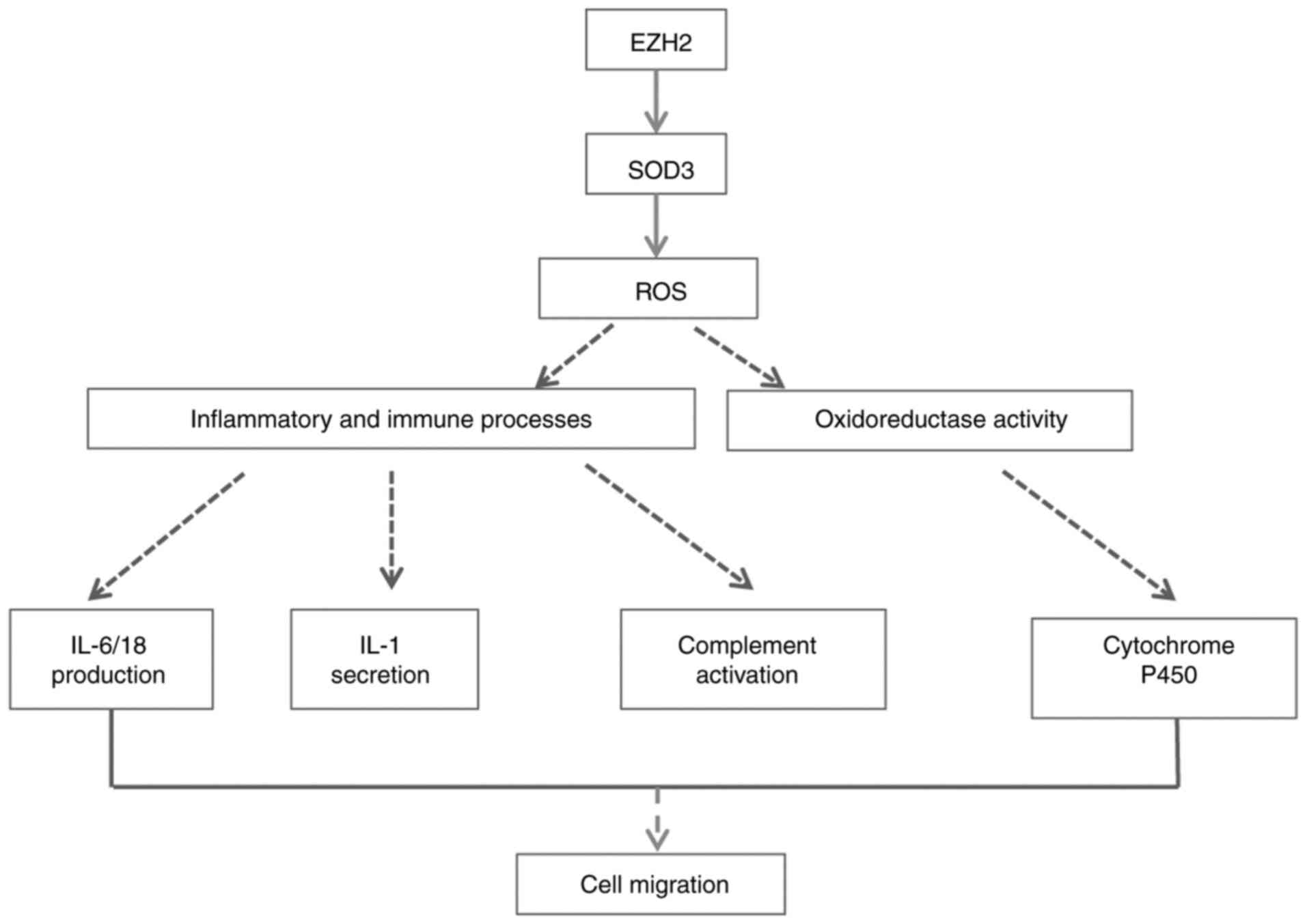
A total of two genes significantly enriched in GO
terms and pathways were selected to verify mRNA expression levels.
The decreased expression levels of SOD3 were consistent with the
microarray results. Potential mechanism pathways are presented in
Fig. 7. The mechanisms of the
aforementioned genes and clear functions modulated by EZH2 in PH
should be investigated in further studies.
There are limitations of the present study. First,
increased PASMC migration was detected following EZH2
overexpression. Since increased SMC migration is a feature of the
synthetic phenotype, the expression levels of synthetic phenotype
markers should be assessed to determine the effects of EZH2
overexpression on the PASMC phenotype. Second, these findings
indicated that EZH2 is increased in PASMC models of PH and provide
a potential preliminary mechanism for the effects of EZH2 on PASMC
migration in PH. However, the exact molecular mechanism requires
further investigation. For example, T-cell coculture experiments
should be performed to validate our finding that EZH2 may regulate
inflammatory and immune processes. Third, the present findings are
also limited by cell type, since there are distinct SMC populations
in pulmonary arteries that may serve different functions. Further
validation should be performed using different cells and
patient-derived cell lines.
In conclusion, the role of histone methyltransferase
EZH2 in PH was investigated. The results indicated that EZH2
promoted human PASMC migration and was increased in CTEPH PASMCs.
The enrichment functions affected by EZH2 focused on inflammatory
and immune processes. qPCR confirmed significantly altered gene
expression levels of SOD3, which suggested an association between
EZH2, SOD3, PASMC migration and inflammatory and immune processes
in PAH. The present findings may aid the search for therapeutic
targets for PH and further investigation on EZH2 in PH is
warranted.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by Capital's Funds
for Health Improvement and Research (grant no. CFH
2018-2-2042).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YW, XXH and DL performed the experiments. YW drafted
the manuscript. JFL analyzed the data. YL analyzed and interpreted
the data. TJ directed and designed the study. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Research
Ethics Committee of the Beijing Chao-Yang Hospital of Capital
Medical University. Written informed consent was provided by all
patients before the procedure was initiated.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Simonneau G, Gatzoulis MA, Adatia I,
Celermajer D, Denton C, Ghofrani A, Gomez Sanchez MA, Krishna Kumar
R, Landzberg M, Machado RF, et al: Updated clinical classification
of pulmonary hypertension. J Am Coll Cardiol. 62 (25
Suppl):D34–D41. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xiong PY, Potus F, Chan W and Archer SL:
Models and molecular mechanisms of world health organization group
2 to 4 pulmonary hypertension. Hypertension. 71:34–55. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Shi N and Chen SY: Mechanisms
simultaneously regulate smooth muscle proliferation and
differentiation. J Biomed Res. 28:40–46. 2014.PubMed/NCBI
|
|
4
|
Masudo K, Suganuma N, Nakayama H, Oshima
T, Rino Y, Iwasaki H, Matsuzu K, Sugino K, Ito K, Kondo T, et al:
EZH2 overexpression as a useful prognostic marker for aggressive
behaviour in thyroid cancer. In Vivo. 32:25–31. 2018.PubMed/NCBI
|
|
5
|
Simon JA and Lange CA: Roles of the EZH2
histone methyltransferase in cancer epigenetics. Mutat Res.
647:21–29. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chang CJ and Hung MC: The role of EZH2 in
tumour progression. Br J Cancer. 106:243–247. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Rao ZY, Cai MY, Yang GF, He LR, Mai SJ,
Hua WF, Liao YJ, Deng HX, Chen YC, Guan XY, et al: EZH2 supports
ovarian carcinoma cell invasion and/or metastasis via regulation of
TGF-beta1 and is a predictor of outcome in ovarian carcinoma
patients. Carcinogenesis. 31:1576–1583. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nephew KP and Huang TH: Epigenetic gene
silencing in cancer initiation and progression. Cancer Lett.
190:125–133. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Archer SL, Marsboom G, Kim GH, Zhang HJ,
Toth PT, Svensson EC, Dyck JR, Gomberg-Maitland M, Thébaud B,
Husain AN, et al: Epigenetic attenuation of mitochondrial
superoxide dismutase 2 in pulmonary arterial hypertension: A basis
for excessive cell proliferation and a new therapeutic target.
Circulation. 121:2661–2671. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Philibert RA, Sears RA, Powers LS, Nash E,
Bair T, Gerke AK, Hassan I, Thomas CP, Gross TJ and Monick MM:
Coordinated DNA methylation and gene expression changes in smoker
alveolar macrophages: Specific effects on VEGF receptor 1
expression. J Leukoc Biol. 92:621–631. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Aljubran SA, Cox R Jr, Tamarapu
Parthasarathy P, Kollongod Ramanathan G, Rajanbabu V, Bao H,
Mohapatra SS, Lockey R and Kolliputi N: Enhancer of zeste homolog 2
induces pulmonary artery smooth muscle cell proliferation. PLoS
One. 7:e377122012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Stenmark KR, Fagan KA and Frid MG:
Hypoxia-induced pulmonary vascular remodeling: Cellular and
molecular mechanisms. Circ Res. 99:675–691. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Groves BM, Reeves JT, Sutton JR, Wagner
PD, Cymerman A, Malconian MK, Rock PB, Young PM and Houston CS:
Operation everest II: Elevated high-altitude pulmonary resistance
unresponsive to oxygen. J Appl Physiol (1985). 63:521–530. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Galiè N and Kim NH: Pulmonary
microvascular disease in chronic thromboembolic pulmonary
hypertension. Proc Am Thorac Soc. 3:571–576. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Barst RJ, McGoon M, Torbicki A, Sitbon O,
Krowka MJ, Olschewski H and Gaine S: Diagnosis and differential
assessment of pulmonary arterial hypertension. J Am Coll Cardiol.
43 (12 Suppl S):40S–47S. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang Y, Huang X, Leng D, Li J, Wang L,
Liang Y, Wang J, Miao R and Jiang T: DNA methylation signatures of
pulmonary arterial smooth muscle cells in chronic thromboembolic
pulmonary hypertension. Physiol Genomics. 50:313–322. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ogawa A, Firth AL, Yao W, Madani MM, Kerr
KM, Auger WR, Jamieson SW, Thistlethwaite PA and Yuan JX:
Inhibition of mTOR attenuates store-operated Ca2+ entry
in cells from endarterectomized tissues of patients with chronic
thromboembolic pulmonary hypertension. Am J Physiol Lung Cell Mol
Physiol. 297:L666–L676. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kanehisa M and Goto S: KEGG: Kyoto
encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rouillard AD, Gundersen GW, Fernandez NF,
Wang Z, Monteiro CD, McDermott MG and Ma'ayan A: The harmonizome: A
collection of processed datasets gathered to serve and mine
knowledge about genes and proteins. Database (Oxford).
2016:baw1002016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jassal B, Matthews L, Viteri G, Gong C,
Lorente P, Fabregat A, Sidiropoulos K, Cook J, Gillespie M, Haw R,
et al: The reactome pathway knowledgebase. Nucleic Acids Res.
48(D1): D498–D503. 2020.PubMed/NCBI
|
|
21
|
Xie C, Mao X, Huang J, Ding Y, Wu J, Dong
S, Kong L, Gao G, Li CY and Wei L: KOBAS 2.0: A web server for
annotation and identification of enriched pathways and diseases.
Nucleic Acids Res. 39((Web Server Issue)): W316–W322. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Humbert M: Pulmonary arterial hypertension
and chronic thromboembolic pulmonary hypertension: Pathophysiology.
Eur Respir Rev. 19:59–63. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Czermin B, Melfi R, McCabe D, Seitz V,
Imhof A and Pirrotta V: Drosophila enhancer of Zeste/ESC complexes
have a histone H3 methyltransferase activity that marks chromosomal
Polycomb sites. Cell. 111:185–196. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bracken AP, Pasini D, Capra M, Prosperini
E, Colli E and Helin K: EZH2 is downstream of the pRB-E2F pathway,
essential for proliferation and amplified in cancer. EMBO J.
22:5323–5335. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang YX, Li JF, Yang YH, Zhai ZG, Gu S,
Liu Y, Miao R, Zhong PP, Wang Y, Huang XX, et al: Renin-angiotensin
system regulates pulmonary arterial smooth muscle cell migration in
chronic thromboembolic pulmonary hypertension. Am J Physiol Lung
Cell Mol Physiol. 314:L276–L286. 2018.PubMed/NCBI
|
|
26
|
Wang L, Liu J, Wang W, Qi X, Wang Y, Tian
B, Dai H, Wang J, Ning W, Yang T and Wang C: Targeting IL-17
attenuates hypoxia-induced pulmonary hypertension through
downregulation of β-catenin. Thorax. 74:564–578. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Varambally S, Cao Q, Mani RS, Shankar S,
Wang X, Ateeq B, Laxman B, Cao X, Jing X, Ramnarayanan K, et al:
Genomic loss of microRNA-101 leads to overexpression of histone
methyltransferase EZH2 in cancer. Science. 322:1695–1699. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Frid MG, Moiseeva EP and Stenmark KR:
Multiple phenotypically distinct smooth muscle cell populations
exist in the adult and developing bovine pulmonary arterial media
in vivo. Circ Res. 75:669–681. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pugliese SC, Poth JM, Fini MA, Olschewski
A, El Kasmi KC and Stenmark KR: The role of inflammation in hypoxic
pulmonary hypertension: From cellular mechanisms to clinical
phenotypes. Am J Physiol Lung Cell Mol Physiol. 308:L229–L252.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rose-John S: Interleukin-6 family
cytokines. Cold Spring Harb Perspect Biol. 10:a0284152018.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Karan D, Tawfik O and Dubey S: Expression
analysis of inflammasome sensors and implication of NLRP12
inflammasome in prostate cancer. Sci Rep. 7:43782017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Allen IC: Non-inflammasome forming NLRs in
inflammation and tumorigenesis. Front Immunol. 5:1692014.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hermanns HM: Oncostatin M and
interleukin-31: Cytokines, receptors, signal transduction and
physiology. Cytokine Growth Factor Rev. 26:545–558. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Miyata M, Ito M, Sasajima T, Ohira H and
Kasukawa R: Effect of a serotonin receptor antagonist on
interleukin-6-induced pulmonary hypertension in rats. Chest.
119:554–561. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Unver N and McAllister F: IL-6 family
cytokines: Key inflammatory mediators as biomarkers and potential
therapeutic targets. Cytokine Growth Factor Rev. 41:10–17. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ye RD and Sun L: Emerging functions of
serum amyloid A in inflammation. J Leukoc Biol. 98:923–929. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Helgadottir A, Manolescu A, Thorleifsson
G, Gretarsdottir S, Jonsdottir H, Thorsteinsdottir U, Samani NJ,
Gudmundsson G, Grant SF, Thorgeirsson G, et al: The gene encoding
5-lipoxygenase activating protein confers risk of myocardial
infarction and stroke. Nat Genet. 36:233–239. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Malle E, Sodin-Semrl S and Kovacevic A:
Serum amyloid A: An acute-phase protein involved in tumour
pathogenesis. Cell Mol Life Sci. 66:9–26. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
De Buck M, Gouwy M, Wang JM, Van Snick J,
Opdenakker G, Struyf S and Van Damme J: Structure and expression of
different serum amyloid a (SAA) variants and their
concentration-dependent functions during host insults. Curr Med
Chem. 23:1725–1755. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jumeau C, Awad F, Assrawi E, Cobret L,
Duquesnoy P, Giurgea I, Valeyre D, Grateau G, Amselem S, Bernaudin
JF and Karabina SA: Expression of SAA1, SAA2 and SAA4 genes in
human primary monocytes and monocyte-derived macrophages. PLoS One.
14:e02170052019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wang Y, Barbacioru C, Hyland F, Xiao W,
Hunkapiller KL, Blake J, Chan F, Gonzalez C, Zhang L and Samaha RR:
Large scale real-time PCR validation on gene expression
measurements from two commercial long-oligonucleotide microarrays.
BMC Genomics. 7:592006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Soon E, Crosby A, Southwood M, Yang P,
Tajsic T, Toshner M, Appleby S, Shanahan CM, Bloch KD, Pepke-Zaba
J, et al: Bone morphogenetic protein receptor type II deficiency
and increased inflammatory cytokine production. A gateway to
pulmonary arterial hypertension. Am J Respir Crit Care Med.
192:859–872. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Humbert M, Monti G, Brenot F, Sitbon O,
Portier A, Grangeot-Keros L, Duroux P, Galanaud P, Simonneau G and
Emilie D: Increased interleukin-1 and interleukin-6 serum
concentrations in severe primary pulmonary hypertension. Am J
Respir Crit Care Med. 151:1628–1631. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Furuya Y, Satoh T and Kuwana M:
Interleukin-6 as a potential therapeutic target for pulmonary
arterial hypertension. Int J Rheumatol. 2010:7203052010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Yang X, Coriolan D, Murthy V, Schultz K,
Golenbock DT and Beasley D: Proinflammatory phenotype of vascular
smooth muscle cells: Role of efficient Toll-like receptor 4
signaling. Am J Physiol Heart Circ Physiol. 289:H1069–H1076. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Bonnet S, Michelakis ED, Porter CJ,
Andrade-Navarro MA, Thébaud B, Bonnet S, Haromy A, Harry G, Moudgil
R, McMurtry MS, et al: An abnormal mitochondrial-hypoxia inducible
factor-1alpha-Kv channel pathway disrupts oxygen sensing and
triggers pulmonary arterial hypertension in fawn hooded rats:
Similarities to human pulmonary arterial hypertension. Circulation.
113:2630–2641. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sutendra G and Michelakis ED: The
metabolic basis of pulmonary arterial hypertension. Cell Metab.
19:558–573. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Bonnet S, Archer SL, Allalunis-Turner J,
Haromy A, Beaulieu C, Thompson R, Lee CT, Lopaschuk GD, Puttagunta
L, Bonnet S, et al: A mitochondria-K+ channel axis is
suppressed in cancer and its normalization promotes apoptosis and
inhibits cancer growth. Cancer Cell. 11:37–51. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Archer SL, Gomberg-Maitland M, Maitland
ML, Rich S, Garcia JG and Weir EK: Mitochondrial metabolism, redox
signaling, and fusion: A mitochondria-ROS-HIF-1alpha-Kv1.5
O2-sensing pathway at the intersection of pulmonary hypertension
and cancer. Am J Physiol Heart Circ Physiol. 294:H570–H578. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cho KA, Suh JW, Lee KH, Kang JL and Woo
SY: IL-17 and IL-22 enhance skin inflammation by stimulating the
secretion of IL-1β by keratinocytes via the ROS-NLRP3-caspase-1
pathway. Int Immunol. 24:147–158. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sutendra G, Dromparis P, Bonnet S, Haromy
A, McMurtry MS, Bleackley RC and Michelakis ED: Pyruvate
dehydrogenase inhibition by the inflammatory cytokine TNFα
contributes to the pathogenesis of pulmonary arterial hypertension.
J Mol Med (Berl). 89:771–783. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yasui K and Baba A: Therapeutic potential
of superoxide dismutase (SOD) for resolution of inflammation.
Inflamm Res. 55:359–363. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Rabinovitch M, Guignabert C, Humbert M and
Nicolls MR: Inflammation and immunity in the pathogenesis of
pulmonary arterial hypertension. Circ Res. 115:165–175. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bauer EM, Zheng H, Comhair S, Erzurum S,
Billiar TR and Bauer PM: Complement C3 deficiency attenuates
chronic hypoxia-induced pulmonary hypertension in mice. PLoS One.
6:e285782011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhang J, Zhang Y, Li N, Liu Z, Xiong C, Ni
X, Pu Y, Hui R, He J and Pu J: Potential diagnostic biomarkers in
serum of idiopathic pulmonary arterial hypertension. Respir Med.
103:1801–1806. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Langer F, Schramm R, Bauer M, Tscholl D,
Kunihara T and Schäfers HJ: Cytokine response to pulmonary
thromboendarterectomy. Chest. 126:135–141. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
von Haehling S, von Bardeleben RS, Kramm
T, Thiermann Y, Niethammer M, Doehner W, Anker SD, Munzel T, Mayer
E and Genth-Zotz S: Inflammation in right ventricular dysfunction
due to thromboembolic pulmonary hypertension. Int J Cardiol.
144:206–211. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Gu S, Su P, Yan J, Zhang X, An X, Gao J,
Xin R and Liu Y: Comparison of gene expression profiles and related
pathways in chronic thromboembolic pulmonary hypertension. Int J
Mol Med. 33:277–300. 2014. View Article : Google Scholar : PubMed/NCBI
|