Introduction
Cervical cancer (CC) is the second most common
malignant tumor among women in Latin America (1,2). The
mortality of CC in developing countries is ~80%, with ~106,000 new
cases and ~48,000 deaths in China in 2018 (3). A large number of studies have
demonstrated that CC is induced by several factors, including
bacterial infections, sexual disorders, multiple pregnancies and
long-term oral contraceptives (4–6).
Harald zur Hausen demonstrated for the first time that human
papilloma virus (HPV) infection is a key cause of CC (7,8).
Although the development and application of HPV vaccines has
increased in popularity, the occurrence and development of tumors
is a complex regulation process involving multiple genes (9); therefore, activation of
proto-oncogenes and inactivation of tumor suppressor genes may
serve as a promising therapeutic strategy in CC.
Long non-coding RNAs (lncRNAs) are a type of RNA
that lack protein-encoding functions, and are widely involved in
cell proliferation, differentiation and apoptosis (10). lncRNAs are >200 nucleotides in
length (11) and the majority of
non-protein coding sequences are transcribed into lncRNAs (12). lncRNAs are tissue- and
cell-specific, and can regulate the growth and development of
single-cell eukaryotes, embryonic stem cells and adult stem cells
(13). Abnormal expression or
functions of lncRNAs are closely related to the occurrence and
development of cancer (14–16). An integrated analysis of
lncRNA-associated networks in CC indicated that several lncRNAs
might serve as novel molecular markers for the diagnosis and
prognosis of CC (17). Increasing
evidence demonstrates that lncRNAs, such as XLOC_006390 (18), small nucleolar RNA host gene 1
(19), are important regulators of
CC progression. However, the functions of lncRNAs in CC are not
completely understood.
ILF3 divergent transcript (ILF3-AS1) is an lncRNA
located on chromosome 19p 13.2 (20). lncRNA ILF3-AS1 is highly expressed
in melanoma cells, promoting cell proliferation, migration and
invasion via negative regulation of microRNA
(miR/miRNA)-200b/a/429, which suggests that ILF3-AS1 may serve as a
potential prognostic biomarker and therapeutic target for melanoma
(20,21). In addition, ILF3-AS1 expression is
significantly upregulated in osteosarcoma tissues and cells, which
promotes osteosarcoma development by regulating the miR-212/SOX5
axis (22). Moreover, ILF3-AS1 is a
potential biomarker for colon (23)
and prostate (24) cancer.
Similarly, previous studies reported that ILF3-AS1 might serve as a
prognostic biomarker for CC (25,26).
However, the specific regulatory mechanism underlying ILF3-AS1 in
CC is not completely understood, thus, the present study
investigated the effects and mechanisms underlying ILF3-AS1 in
CC.
Materials and methods
Ethics statement
All patients provided written informed consent. The
study was approved by the Ethics committee of Guangdong Clifford
Hospital (approval no. 2014007XHK).
CC specimen collection and survival
analysis
CC tissues (15 stage I/II cases and 17 stage III/IV
cases) and adjacent tissues (ANTs; distance from tumor margin, 2
cm; n=32) were collected during CC (age, 37–55 years; female
patients) resection at Guangdong Clifford Hospital (Guangzhou,
China) between January 2017 and December 2018. Patients with CC
were categorized into stages I–IV according to the criteria of the
International Society of Oncology and the International Association
of Obstetricians and Gynecologists (27). The exclusion criteria were as
follows: i) Combined with other malignancies; ii) receiving
radiotherapy and chemotherapy for CC; and iii) and acute
inflammation of the reproductive tract. Patients diagnosed with
cervical cancer via pathological examination were included in the
present study. Tissues were washed with sterile saline, snap-frozen
in liquid nitrogen for 15 min and stored at −80°C.
Database analysis
The expression and pathological stage plots of
ILF3-AS1 in cervical and endocervical cancer were obtained from the
Gene Expression Profiling Interactive Analysis (GEPIA) database
(gepia.cancer-pku.cn). Kaplan-Meier analysis was performed to
assess the survival of patients with CC based on data obtained from
the GEPIA database. Survival curves were compared using log-rank
tests.
Cell culture
Normal cervical epithelial cell line (NCE;
PCS-480-011) and CC cell lines, including C33A (HTB-31), ME-180
(HTB-33), SiHa (HTB-35), HeLa (CCL-2) and CaSki (CRM-CRL-1550),
were purchased from American Type Culture Collection. Cells were
cultured in DMEM (cat. no. 12800017; Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% FBS (cat. no. 10437085;
Gibco; Thermo Fisher Scientific, Inc.) at 37°C with 5%
CO2.
Transfection
The overexpression vector pcDNA 3.1 (cat. no.
V86020; Invitrogen; Thermo Fisher Scientific, Inc.) containing
ILF3-AS1 was constructed. SiHa and HeLa cells were transfected with
2 µg ILF3-AS1, miR-454-3p mimic, ILF3-AS1 + miR-454-3p mimic or the
corresponding controls [empty vector pcDNA 3.1 plasmid and mimic
control (scrambled), 5′-UAGGUCUAAAUGUCUAUUGAUGG-3′] using
Lipofectamine® 3000 (cat. no. L3000015; Thermo Fisher
Scientific, Inc.). Briefly, 1 µg ILF3-AS1 overexpression vector or
30 pmol miR-454-3p mimic (cat. no. 4464066; Thermo Fisher
Scientific, Inc.; 5′-UAGUGCAAUAUUGCUUAUAGGGU-3′) was added to
Opti-MEM® medium (cat. no. 31985062; Invitrogen; Thermo
Fisher Scientific, Inc.). Subsequently, 3 µl Lipofectamine 3000
reagent was added to Opti-MEM medium. Following gentle mixing of
the two mixtures at room temperature, CC cells (5×105
cells/well) were incubated with the combined mixture. Blank cells
were treated with transfection reagent alone. Cells in the negative
control (NC) group were transfected with empty pcDNA 3.1 plasmid.
Cells in the control group were co-transfected with empty pcDNA 3.1
plasmid and mimic control. The following mimic control was used:
5′-UAGCAAGGAGGUCUAUGUUAUUU-3′. Transfection was performed at room
temperature for 15 min. At 48 h post-transfection, subsequent
experiments were performed.
Target gene prediction
The target miRNA of ILF3-AS1 was predicted using
StarBase (starbase.sysu.edu.cn) (28). TargetScan (version 7.2; www.targetscan.org) was used to analyze miRNA-mRNA
interactions (29).
Luciferase assay
The dual-luciferase reporter assay was performed to
verify predictions. The following four reporter plasmids were
constructed using the pmirGLO plasmid (cat. no. CL414-01; Beijing
Biomed Gene Technology Co., Ltd.): i) Wild-type (WT) ILF3-AS1
(ILF3-AS1-WT); ii) mutant (MUT) ILF3-AS1 (ILF3-AS1-MUT); iii) WT
PTEN (PTEN-WT); and iv) mutant PTEN (PTEN-MUT). The four reporter
plasmids had the following sequences: i) ILF3-AS1-WT,
5′-AGCCGAGAUUGCUCCAUUGCACUC-3′; ii) ILF3-AS1-MUT,
5′-AGGGGUGUUUCGUCCUAACGUGAC-3′; iii) PTEN-WT,
5′-CAUUAUAAUGGGCUUUUGCACUG-3′; and iv) PTEN-MUT,
5′-CAUCGAUGUGGGCUUCCUUGACG-3′. Cells were seeded (5×105
cells/well) into 24-well plates and cultured overnight.
Subsequently, SiHa and HeLa cells were co-transfected with 50 ng
ILF3-AS1-WT, ILF3-AS1-MUT, PTEN-WT or PTEN-MUT reporter plasmid and
100 pmol miR-454-3p mimic or mimic control using
Lipofectamine® 3000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions for
48 h at 37°C. At 48 h post-transfection, firefly and Renilla
luciferase activities were detected using the Dual-luciferase
Reporter Assay System (cat. no. E1910; Promega Corporation).
Firefly luciferase activities were normalized to Renilla
luciferase activities.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from tissues and cells using
TRIzol® reagent (cat. no. 15596026; Invitrogen; Thermo
Fisher Scientific, Inc.). Total RNA concentrations were detected
using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific,
Inc.). Total RNAs were reverse transcribed into cDNA using the
Revert Aid First Strand cDNA Synthesis Kit (cat. no. k1622; Thermo
Fisher Scientific, Inc.). For detection of miR-454-3p expression,
reverse transcription was performed using the All-in-One™ miRNA
First-Reverse Transcription, Strand cDNA Synthesis Kit (cat. no.
QP056; GeneCopoeia, Inc.). Both RT kits were used according to the
manufacturer's protocol. Subsequently, qPCR was performed using a
Veriti™ 96-Well Fast Thermal Cycler (cat. no. 4375305; Thermo
Fisher Scientific, Inc.) and PowerUp™ SYBR™ Green Master Mix (cat.
no. A25742; Applied Biosystems; Thermo Fisher Scientific, Inc.).
The following thermocycling conditions were used for qPCR: Initial
denaturation at 95°C for 10 min; 40 cycles of denaturation at 95°C
for 15 sec, annealing at 60°C for 1 min and elongation at 95°C for
10 sec; and extension at 65°C for 60 sec. The following primers
were used for qPCR: lncRNA ILF3-AS1 forward,
5′-TAAACCCCACTGTCTTCC-3′ and reverse, 5′-TTCCTTGCTCTTCTTGCTC-3′;
PTEN forward, 5′-TTTGAAGACCATAACCCACCAC-3′ and reverse,
5′-ATTACACCAGTTCGTCCCTTTC-3′; GAPDH forward,
5′-AATGGACAACTGGTCGTGGAC-3′ and reverse,
5′-CCCTCCAGGGGATCTGTTTG-3′; miR-454-3p forward,
5′-GGGTGTCGTATCCAGTGCAA-3′ and reverse, 5′-GTCGTATCCAGTGCGTGTCG-3′;
and U6 forward, 5′-AGTAAGCCCTTGCTGTCAGTG-3′ and reverse,
5′-CCTGGGTCTGATAATGCTGGG-3′. miRNA and mRNA expression levels were
quantified using the 2−∆∆Cq method (30) and normalized to the internal
reference genes U6 and GAPDH, respectively.
Cell Counting Kit-8 (CCK-8) assay
SiHa and HeLa cell viability was assessed by
performing CCK-8 assays. Cells were seeded (5×103
cells/ml; 100 µl) into 96-well plates, with six replicate wells for
each experimental group. Following incubation for 24, 48 and 72 h
at 37°C, 10 µl CCK-8 reagent (cat. no. PA584814; Thermo Fisher
Scientific, Inc.) was added to each well and incubated for 4 h. The
absorbance was measured at a wavelength of 450 nm using an iMARK
microplate absorbance reader (Bio-Rad Laboratories, Inc.).
Colony formation assay
SiHa and HeLa cells were seeded (1×102
cells/well) into 6-well plates and incubated at 37°C with 5%
CO2. After 2 weeks, cells were thoroughly washed with
PBS, fixed with 5 ml 100% methanol (cat. no. M116119-4L; Shanghai
Aladdin Biochemical Technology Co., Ltd.) for 15 min at room
temperature and stained with Giemsa stain (cat. no. 32884-250ML;
Sigma-Aldrich; Merck KGaA) for 20 min at room temperature.
Following drying at room temperature, stained cells were counted
manually. The colony formation rate (%) was calculated according to
the following formula: (Number of clones/number of cells
inoculated) ×100.
Flow cytometry
The Annexin V-FITC Apoptosis Detection Kit (cat. no.
C1062M; Beyotime Institute of Biotechnology) was used to assess
cell apoptosis. SiHa or HeLa cell (3×105 cells)
suspensions were prepared using PBS. Subsequently, cells were
incubated with 5 µl Annexin V-FITC and 10 µl PI (cat. no. C1062M;
Beyotime Institute of Biotechnology) for 20 min in the dark at room
temperature. Apoptotic cells (early and late apoptotic cells) were
analyzed using a CytoFLEX flow cytometer (Beckman Coulter, Inc.)
and analyzed using FlowJo (version 10.0; FlowJo LLC).
Wound healing assay
SiHa and HeLa cell migration was detected by
performing wound healing assays. Cells were seeded
(5×105 cells/well) into a 6-well plate and incubated
overnight at 37°C. At 80% confluence, a pipette was used to create
a single wound in the cell monolayer. Following washing using PBS,
cells were incubated with serum-free DMEM medium at 37°C with 5%
CO2. SiHa and HeLa cell migration was observed at 0 and
24 h using a TS100 light microscope (Nikon Corporation;
magnification, ×100). The relative migration distance was
calculated according to the following formula: (wound width at 0
h-wound width at 24 h)/wound width at 0 h. Relative migration rates
were calculated as the ratio of the relative migration distance of
cells in the ILF3-AS1 group to the relative migration distance of
cells in the blank group.
Transwell invasion assay
Transwell invasion assays were performed to assess
SiHa and HeLa cell invasion. The upper chambers of Transwell
inserts (pore size, 8 µm; BD Biosciences) were coated with Matrigel
for 30 min at 37°C. Subsequently, cells were seeded
(1×104 cells/well) into the upper chamber with
serum-free DMEM. DMEM supplemented with 10% FBS was added into the
lower chamber. Following incubation for 48 h at 37°C, invading
cells were fixed with 4% paraformaldehyde (cat. no. C104188-500g;
Shanghai Aladdin Biochemical Technology Co., Ltd.) at room
temperature for 5 min and stained with 0.1% crystal violet (cat.
no. RBG1019-1; Guangzhou Rolls Biotechnology Co., Ltd.) for 15 min
at room temperature. Invading cells were observed using a light
microscope (magnification, ×100).
Western blotting
Following washing with PBS, total protein was
isolated from SiHa and HeLa cells using 100 µl cell lysis buffer
(RIPA buffer; cat. no. 9806; Cell Signaling Technology, Inc.).
Proteins were obtained from the cell lysate by centrifugation at
4°C for 10 min at 1,600 × g. Protein concentrations were determined
using the BCA method. Proteins (100 µg) were separated via 10%
SDS-PAGE and transferred to PVDF membranes. Following washing three
times with 1X TBST (0.5% Tween-20) for 10 min, the membranes were
blocked with 5% skimmed milk for 1.5 h at room temperature.
Subsequently, the membranes were incubated overnight at 4°C with
primary antibodies targeted against the following: E-Cadherin
(E-Cad; 1:1,000; cat. no. 14472; Cell Signaling Technology, Inc.),
N-Cadherin (N-Cad; 1:1,000; cat. no. 14215; Cell Signaling
Technology, Inc.), snail family transcriptional repressor 1 (Snail;
1:10,000; cat. no. ab53519; Abcam) and GAPDH (1:10,000; cat. no.
ab8245; Abcam). Following primary incubation, the membranes were
incubated with anti-goat (HRP-conjugated; 1:5,000; cat. no. ab6885;
Abcam) and anti-mouse (HRP-conjugated; 1:10,000; cat. no. ab6728;
Abcam) secondary antibodies for 1 h at 37°C. The membranes were
washed three times. Protein bands were visualized using ECL
luminescent liquid (cat. no. RBG2019-1; Guangzhou Rolls
Biotechnology Co., Ltd.) and observed using a GelDoc XR gel imager
(Bio-Rad Laboratories, Inc.). Protein expression levels were
semi-quantified using ImageJ software (version 1.47; National
Institutes of Health) with GAPDH as the loading control.
Statistical analysis
Statistical analyses were performed using SPSS
software (version 23.0; IBM Corp.). Data are presented as the mean
± SD. All experiments were repeated three times. For normally
distributed data, comparisons between two groups were analyzed
using an unpaired Student's t test, whereas comparisons among
multiple groups were analyzed using one-way ANOVA followed by
Bonferroni's post hoc test. Pearson's correlation coefficient was
used to analyze correlations. P<0.05 was considered to indicate
a statistically significant difference.
Results
ILF3-AS1 expression is downregulated
in CC tissues
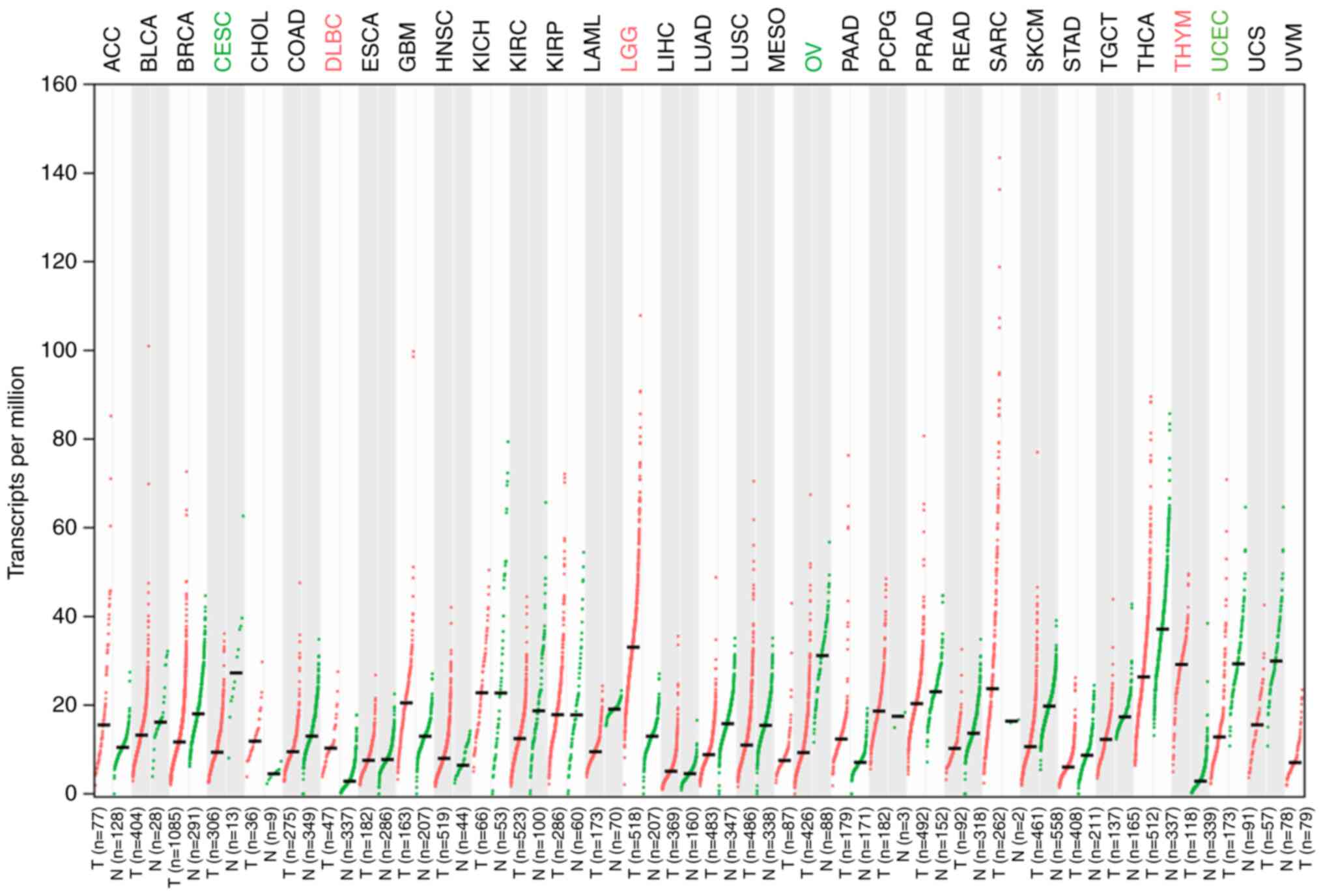
ILF3-AS1 expression in tumor tissues was detected
using the GEPIA database (Fig. 1).
The results demonstrated that ILF3-AS1 was expressed at lower
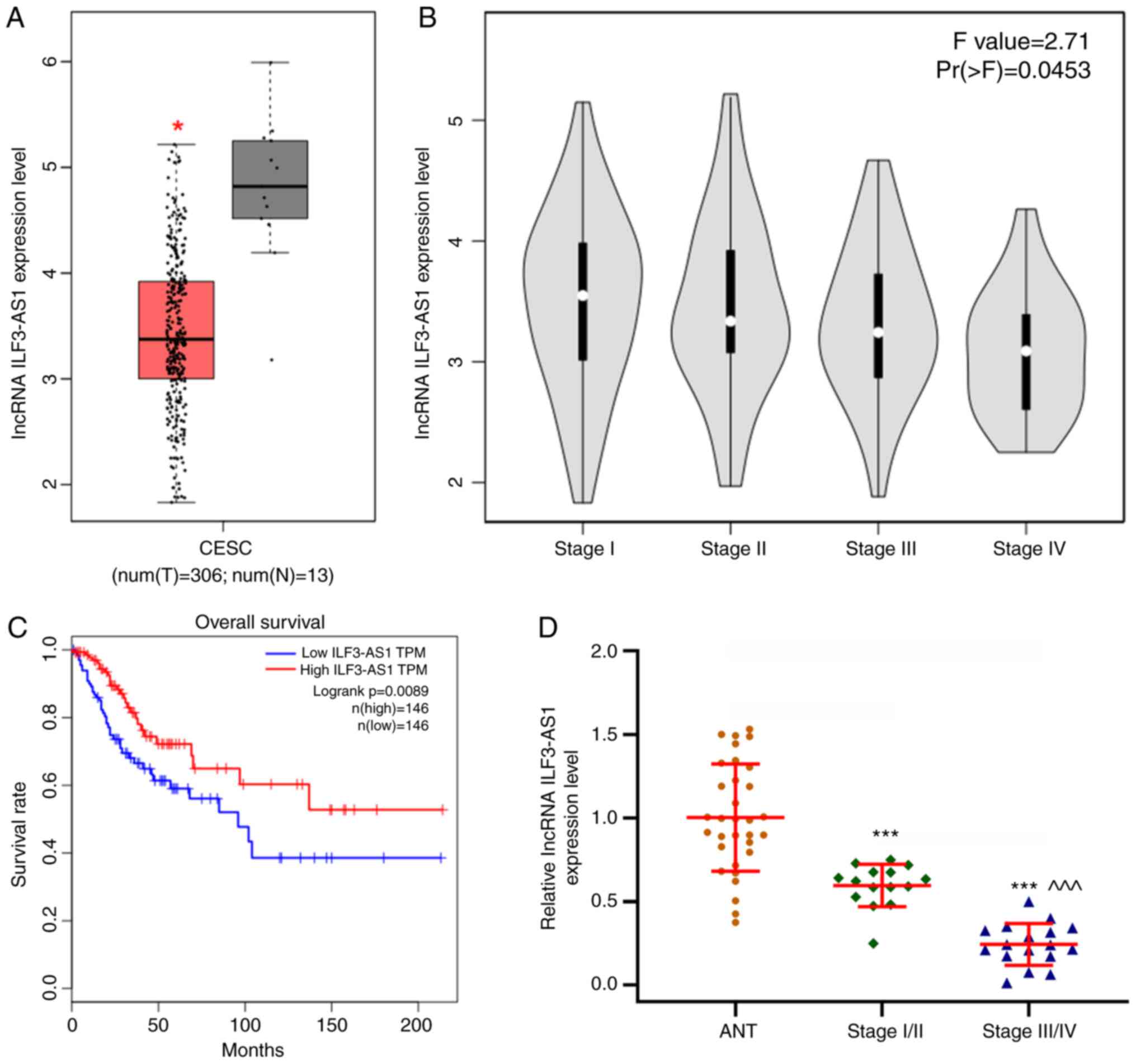
levels in tumor tissues compared with healthy tissues. In addition,
the GEPIA database predicted that ILF3-AS1 expression levels were
significantly lower in CC tissues compared with healthy tissues
(P<0.05; Fig. 2A). Moreover, low
ILF3-AS1 expression levels were significantly associated with more
advanced stages of CC (F=2.71; Pr(>F)=0.0453; Fig. 2B). The overall survival of patients
with low ILF3-AS1 expression was significantly worse compared with
patients with high ILF3-AS1 expression (P=0.0089; |log2FC|
cutoff=1; Fig. 2C). In addition,
ILF3-AS1 expression levels in CC tissues of different stages and
ANTs were detected via RT-qPCR (P<0.001; Fig. 2D). ILF3-AS1 expression was
significantly downregulated in CC tissues compared with ANTs, and
was significantly lower in stage III/IV CC tissues compared with
stage I/II CC tissues (P<0.001). The results demonstrated that
lncRNA ILF3-AS1 was expressed at lower levels in CC tissues
compared with ANTs, and that ILF3-AS1 expression in stage III/IV CC
tissues was decreased compared with stage I/II CC tissues.
ILF3-AS1 inhibits CC cell
proliferation and promotes CC cell apoptosis
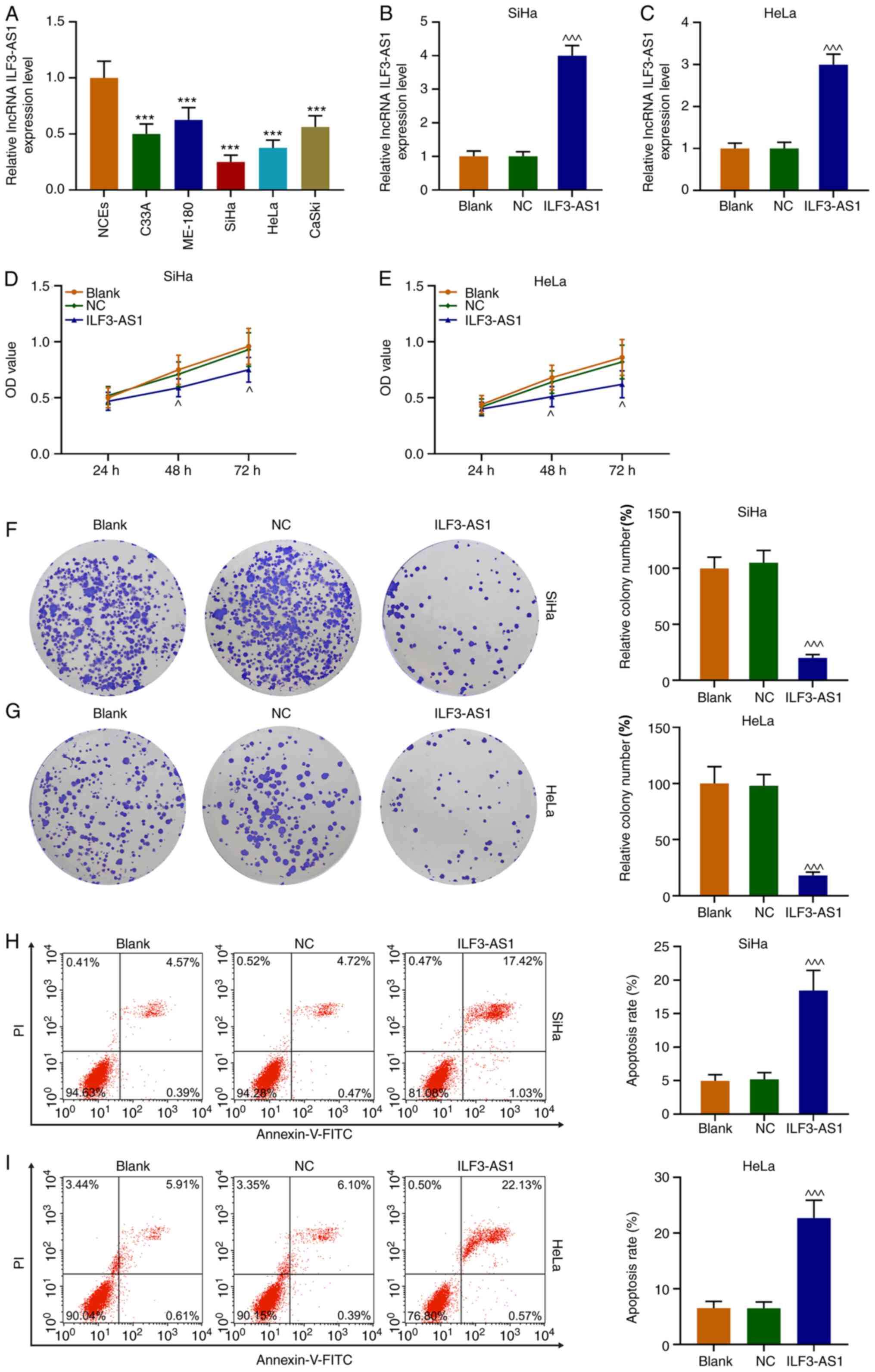
ILF3-AS1 expression levels in CC cells were detected
via RT-qPCR. The results demonstrated that ILF3-AS1 expression was
significantly downregulated in CC cells compared with NCEs
(P<0.001; Fig. 3A). Compared
with NCEs, SiHa and HeLa cells displayed the lowest expression
levels of ILF3-AS1 among the CC cell lines, thus were used for
subsequent experiments. Following transfection with ILF3-AS1,
ILF3-AS1 expression levels were significantly increased in SiHa and
HeLa cells compared with the NC group (P<0.001; Fig. 3B and C). Compared with the NC group,
ILF3-AS1 overexpression significantly inhibited SiHa and HeLa cell
viability at 48 and 72 h (P<0.05, Fig. 3D and E). Similarly, ILF3-AS1
overexpression significantly decreased SiHa and HeLa cell
proliferation compared with the NC group (P<0.001; Fig. 3F and G). The flow cytometry results
indicated that ILF3-AS1 overexpression significantly increased cell
apoptosis compared with the NC group (P<0.001; Fig. 3H and I). Thus, the results
demonstrated that ILF3-AS1 was expressed at lower levels in CC
cells compared with NCEs, and ILF3-AS1 overexpression inhibited
cell viability and promoted cell apoptosis compared with the NC
group.
ILF3-AS1 inhibits CC metastasis by
inhibiting epithelial-mesenchymal transition (EMT)
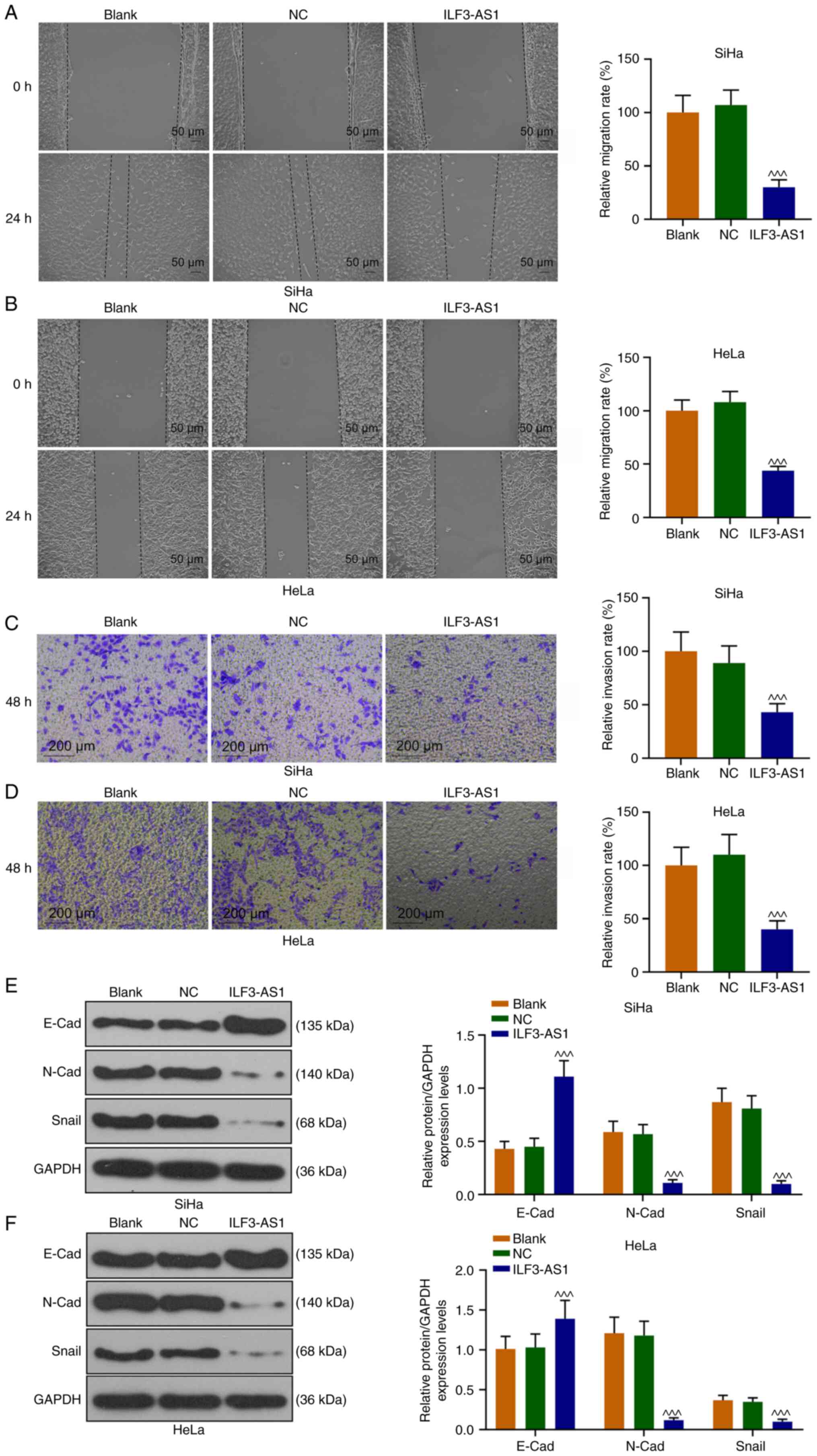
SiHa and HeLa cell migration and invasion were also
assessed (Fig. 4A-D). In
ILF3-AS1-overexpression cells, cell migration and invasion were
significantly inhibited compared with the NC group (P<0.001;
Fig. 4A-D). To further explore the
mechanism underlying ILF3-AS1-mediated alterations, the expression
levels of EMT-associated proteins were detected. ILF3-AS1
overexpression significantly upregulated E-Cad expression levels,
and significantly downregulated N-Cad and Snail expression levels
in SiHa and HeLa cells compared with the NC group (P<0.001;
Fig. 4E and F). The aforementioned
results indicated that ILF3-AS1 overexpression inhibited EMT in CC
cells.
miR-454-3p is highly expressed in CC
cells and is negatively correlated with ILF3-AS1
StarBase predicted that miR-454-3p was a target
miRNA of ILF3-AS1, which was verified by performing dual-luciferase
reporter assays (P<0.001; Fig.
5A-C). miR-454-3p expression was significantly upregulated in
CC tissues and cells compared with ANTs and NCEs, respectively
(P<0.001; Fig. 5D and H).
miR-454-3p expression levels were significantly higher in stage
III/IV CC tissues compared with stage I/II CC tissues (P<0.001;
Fig. 5D). Moreover, the results
indicated that ILF3-AS1 and miR-454-3p expression levels were
negatively correlated (P<0.05; r<0; Fig. 5E-G). miR-454-3p mimic significantly
upregulated miR-454-3p expression levels in SiHa and HeLa cells
compared with the mimic control group (P<0.001; Fig. 5I and J).
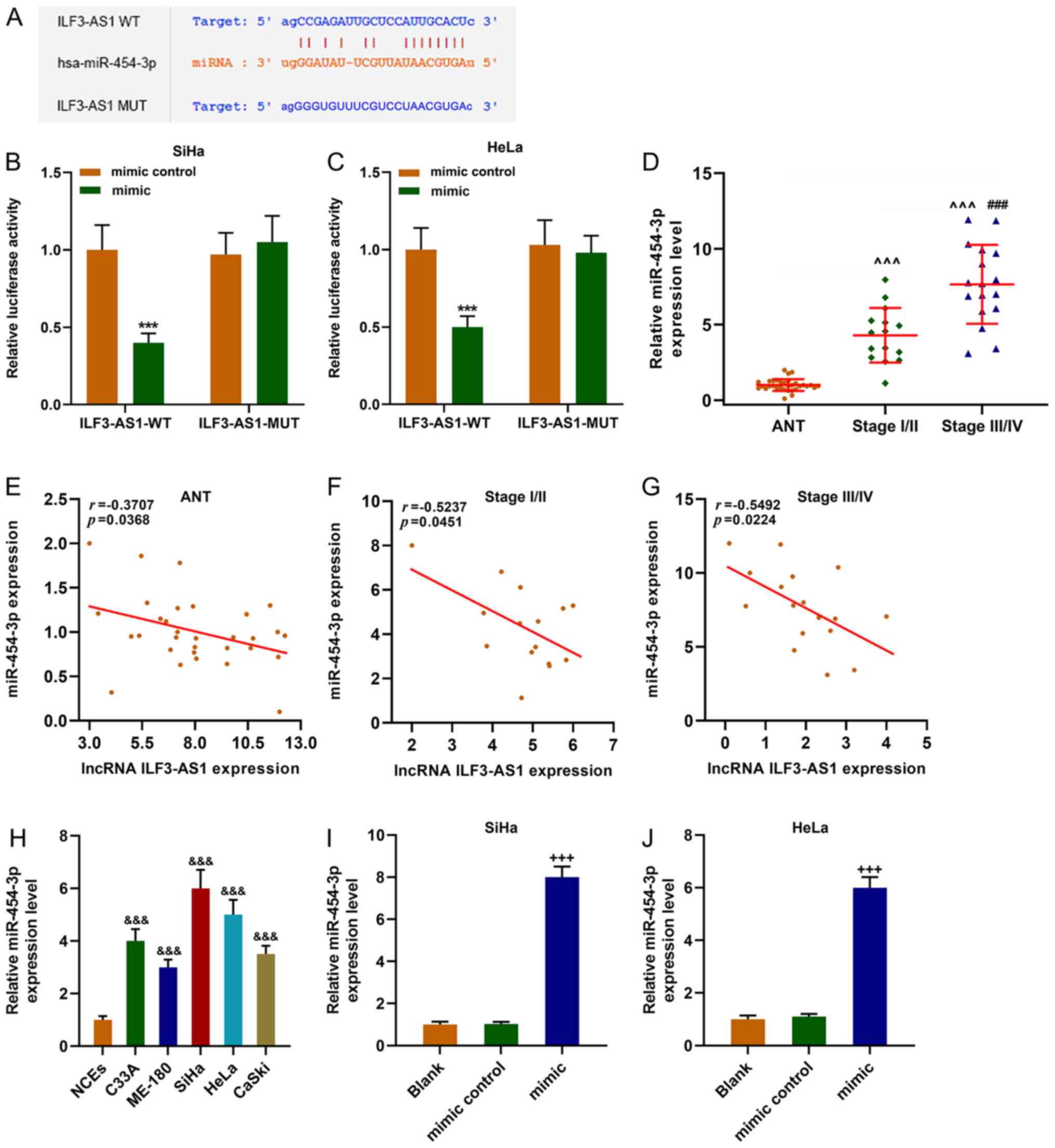 | Figure 5.miR-454-3p expression is upregulated
in CC cells and is negatively correlated with ILF3-AS1. (A)
StarBase predicted that miR-454-3p was the targeted miRNA of
ILF3-AS1. The sequences of ILF3-AS1 WT, ILF3-AS1 MUT and
miR-454-3p. Dual-luciferase reporter assays were performed to
verify the relationship between ILF3-AS1 and miR-454-3p in (B) SiHa
and (C) HeLa cells. (D) RT-qPCR was performed to measure miR-454-3p
expression levels in ANTs and CC tissues. Pearson's correlation
coefficient was used to analyze the correlation between miR-454-3p
and ILF3-AS1 expression in (E) ANTs, (F) stage I/II CC tissues and
(G) stage III/IV CC tissues. (H) RT-qPCR was performed to measure
miR-454-3p expression levels in NCEs and CC cells. Transfection
efficiency of miR-454-3p mimic in (I) SiHa and (J) HeLa cells. All
experiments were repeated three times. ***P<0.001 vs. mimic
control; ^^^P<0.001 vs. ANT; ###P<0.001
vs. stage I/II; &&&P<0.001 vs. NCEs;
+++P<0.001 vs. blank. miR/miRNA, microRNA; CC,
cervical cancer; ILF3-AS1, ILF3 divergent transcript; WT,
wild-type; MUT, mutant; RT-qPCR, reverse transcription-quantitative
PCR; ANT, adjacent tissue; NCE, normal cervical epithelial cell
line. |
PTEN is expressed at low levels in CC
cells, and is negatively correlated with miR-454-3p and positively
correlated with ILF3-AS1
TargetScan predicted that the target gene for
miR-454-3p was PTEN, which was further verified by performing
dual-luciferase reporter assays (P<0.001; Fig. 6A-C). The RT-qPCR results
demonstrated that PTEN expression levels were significantly lower
in CC tissues and cells compared with ANTs and NECs, respectively
(P<0.001; Fig. 6D and L). In
addition, PTEN expression levels in stage III/IV CC tissues were
significantly lower compared with stage I/II CC tissues
(P<0.001; Fig. 6D). Correlation
analysis demonstrated that PTEN expression was negatively
correlated with miR-454-3p expression (P<0.05; r=−0.5022;
Fig. 6E-G), but positively
correlated with ILF3-AS1 expression (P<0.05; r=0.4600; Fig. 6H-J). In addition, CC cells were
transfected with ILF3-AS1, miR-454-3p mimic or ILF3-AS1 +
miR-454-3p mimic. miR-454-3p expression levels in SiHa cells were
significantly decreased by ILF3-AS1 overexpression compared with
the control group, which was significantly reversed by
co-transfection miR-454-3p mimic (P<0.01; Fig. 6K). Compared with the control group,
miR-454-3p mimic significantly downregulated PTEN expression
levels, but ILF3-AS1 overexpression significantly upregulated PTEN
expression levels, which was significantly reversed by
co-transfection with miR-454-3p mimic (P<0.001; Fig. 6M and N). The results indicated that
ILF3-AS1 upregulated PTEN gene expression by negatively regulating
miR-454-3p.
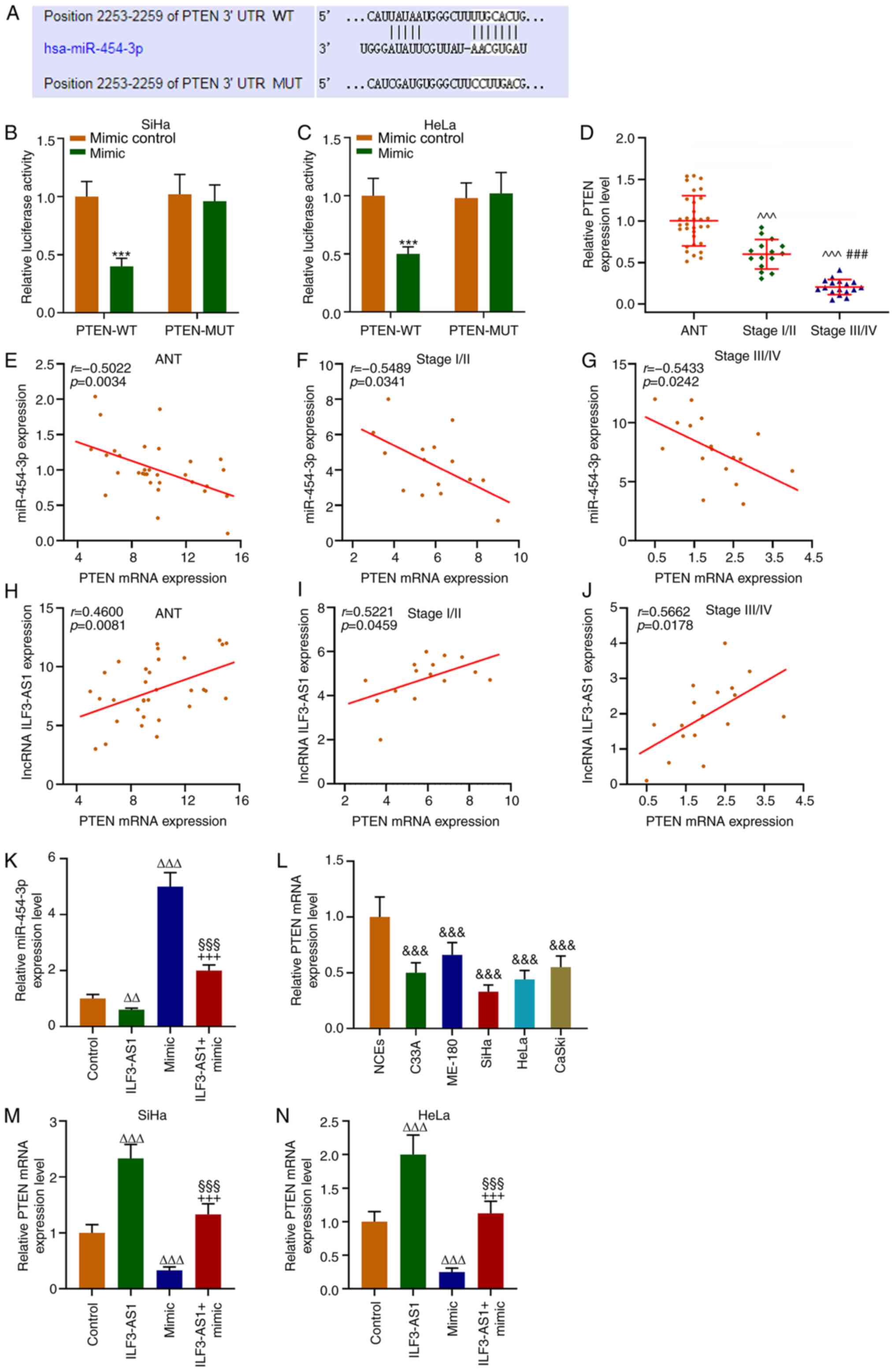 | Figure 6.PTEN is a target gene for miR-454-3p
and is correlated with miR-454-3p and ILF3-AS1 expression. (A)
TargetScan was used to predict the target gene of miR-454-3p.
Dual-luciferase reporter assays were performed to verify the
relationship between PTEN and miR-454-3p in (B) SiHa and (C) HeLa
cells. (D) RT-qPCR was performed to measure miR-454-3p expression
levels in ANTs and CC tissues. Pearson's correlation coefficient
was used to analyze the correlation between miR-454-3p and PTEN
expression in (E) ANT, (F) stage I/II CC tissues and (G) stage
III/IV CC tissues. Pearson's correlation coefficient was used to
analyze the correlation between PTEN and ILF3-AS1 in (H) ANTs, (I)
stage I/II CC tissues and (J) stage III/IV CC tissues. RT-qPCR was
performed to measure (K) miR-454-3p expression levels in SiHa cells
following transfection, (L) PTEN expression levels in NCEs and CC
cells, and PTEN expression levels in (M) SiHa and (N) HeLa cells
following transfection. All experiments were repeated three times.
***P<0.001 vs. mimic control; ^^^P<0.001 vs. ANT;
###P<0.001 vs. stage I/II;
&&&P<0.001 vs. NCEs;
∆∆P<0.01; ∆∆∆P<0.001 vs. control;
§§§P<0.001 vs. ILF3-AS1; +++P<0.001 vs.
miR-454-3p mimic. miR, microRNA; ILF3-AS1, ILF3 divergent
transcript; RT-qPCR, reverse transcription-quantitative PCR; ANT,
adjacent tissue; CC, cervical cancer; NCE, normal cervical
epithelial cell line; WT, wild-type; MUT, mutant; UTR, untranslated
region. |
Discussion
It has been reported that ILF3-AS1 can be used to
predict survival in patients with CC (25,26).
However, the functions and mechanism underlying ILF3-AS1 in CC are
not completely understood. The present study demonstrated that
ILF3-AS1 overexpression significantly inhibited CC cell migration
and invasion compared with the NC group. Therefore, the expression
levels of EMT-related factors were measured. E-Cad, N-Cad and Snail
are important regulators of EMT (31). Downregulation or deletion of E-Cad
protein can cause tumor cell release contact inhibition, and the
conversion of E-Cad to N-Cad can enhance tumor cell migration and
invasion, which promotes EMT (32–34).
Snail downregulates the expression levels of epithelial phenotype
genes by binding to the E-box sequence of the target gene (35). The present study demonstrated that
compared with the NC group, ILF3-AS1 overexpression significantly
upregulated E-Cad protein expression levels, and significantly
downregulated N-Cad and Snail expression levels, which indicated
that ILF3-AS1 overexpression inhibited CC cell EMT. EMT, a
biological process by which epithelial cells are transformed into
mesenchymal cells (36), serves an
important role in epithelial-derived cancer cell migration and
invasion (37). Therefore,
investigating the molecular mechanism underlying EMT in the
occurrence, development and metastasis of tumors, and developing
diagnostic and molecular therapeutic strategies based on EMT may
improve CC treatment.
lncRNAs affect tumor cell proliferation by
inhibiting miRNA expression (38).
lncRNAs can compete with miRNAs for binding to genes, and can also
cleave precursors of miRNAs in cells or directly inhibit miRNA
expression (39). In the present
study, starBase predicted that miR-454-3p was a target miRNA of
ILF3-AS1. miR-454-3p serves a regulatory role in a variety of
neoplastic diseases, including breast and bladder cancer (40,41).
Wang et al (42) reported
that miR-454-3p is expressed at low levels in bladder cancer cells,
which negatively regulated cell invasion and migration by targeting
ZEB2 antisense RNA 1. Shao et al (43) demonstrated that miR-454-3p is an
exosome biomarker, and miR-454-3p overexpression inhibits glioma
cell proliferation, migration, invasion and autophagy. However, Ren
et al (40) reported that
inhibition of regulation of nuclear pre-mRNA domain containing 1A
by miR-454-3p activates the Wnt/β-catenin signaling pathway,
thereby promoting breast cancer metastasis and growth (40). In addition, miR-454-3p expression is
upregulated in colorectal cancer tissues and participates in the
development of colorectal cancer by promoting liver cancer cell
proliferation, invasion and migration (44). The aforementioned studies suggested
that miR-454-3p might display different effects in different types
of cancer. To explore the role of miR-454-3p in CC, miR-454-3p
expression levels in CC tissues and cells were measured. The
results demonstrated that miR-454-3p expression levels were
significantly increased in CC tissues and cells compared with ANTs
and NCEs, respectively. Moreover, miR-454-3p was identified as a
target miRNA of ILF3-AS1, and its expression was negatively
regulated by ILF3-AS1.
In the present study, TargetScan also identified an
interaction between miR-454-3p and PTEN. PTEN, which is 200 kb in
length and is located on the human chromosome 10q23.3 (45), displays a variety of biological
activities and encodes a dual phosphatase with lipid and protein
phosphatase activities (46).
Previous studies demonstrated that PTEN inhibits cell
proliferation, migration and adhesion, induces apoptosis, and
participates in embryonic development and other physiological
functions via multiple signaling pathways, including the Akt
signaling pathway (47,48). PTEN is a tumor suppressor gene and
one of the most susceptible genes in tumors (46). By inhibiting FAK expression
activity, PTEN reduces integrin-mediated cell proliferation and
local adhesion, thereby inhibiting tumor cell migration and
invasion (49). In addition, PTEN
can suppress the phosphorylation of ERK, RAS and SHC adaptor
protein 1, which are upstream of the MAPK/ERK signaling pathway,
thereby inhibiting tumor cell proliferation (50). Therefore, the aforementioned studies
indicated that increased PTEN expression levels display a
significant inhibitory effect on tumor cell migration and invasion.
The results of the present study demonstrated that PTEN expression
was significantly decreased in CC cells compared with NCEs, which
was partially reversed by ILF3-AS1 overexpression. Moreover,
miR-454-3p negatively regulated ILF3-AS1.
In conclusion, the present study demonstrated that
ILF3-AS1 was expressed at significantly lower levels in CC tissues
and cells compared with ANTs and NCEs, respectively. Compared with
the NC group, ILF3-AS1 overexpression significantly inhibited CC
cell viability, reduced CC cell migration and invasion, and
promoted CC cell apoptosis by inhibiting EMT. Moreover, compared
with the control group, ILF3-AS1 overexpression significantly
upregulated the expression levels of the tumor suppressor gene PTEN
by negatively regulating miR-454-3p. Therefore, the results of the
present study may aid with improving the treatment of CC.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Medical
Scientific Research Foundation of Guangdong Province (grant no.
B2020211), the Scientific Research Project of Guangdong Provincial
Bureau of Traditional Chinese Medicine (grant no. 20201284) and the
Guangzhou Health Science and Technology Project (grant no.
20202A010024).
Availability of data and materials
The datasets analyzed during the current study are
available from the corresponding author on reasonable request.
Authors' contributions
LZ and RC substantially contributed to the
conception and design of the present study. CJ, QX, WZ, XG and HH
acquired, analyzed and interpreted the data. LZ and RC drafted the
manuscript and critically revised it for important intellectual
content. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All patients provided written informed consent. The
study was approved by Guangdong Clifford Hospital (approval no.
2014007XHK).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CC
|
cervical cancer
|
|
lncRNA
|
long non-coding RNA
|
|
RT-qPCR
|
reverse transcription-quantitative
PCR
|
|
CCK-8
|
Cell Counting Kit-8
|
References
|
1
|
Burki TK: Novel mutations in cervical
cancer. Lancet Oncol. 18:e1372017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bychkovsky BL, Ferreyra ME,
Strasser-Weippl K, Herold CI, de Lima Lopes G Jr, Dizon DS,
Schmeler KM, Del Carmen M, Randall TC, Nogueira-Rodrigues A, et al:
Cervical cancer control in Latin America: A call to action. Cancer.
122:502–514. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Arbyn M, Weiderpass E, Bruni L, de Sanjosé
S, Saraiya M, Ferlay J and Bray F: Estimates of incidence and
mortality of cervical cancer in 2018: a worldwide analysis. Lancet
Glob Health. 8:e191–e203. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fang J, Zhang H and Jin S: Epigenetics and
cervical cancer: From pathogenesis to therapy. Tumour Biol.
35:5083–5093. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kanyina EW, Kamau L and Muturi M: Cervical
precancerous changes and selected cervical microbial infections,
Kiambu County, Kenya, 2014: A cross sectional study. BMC Infect
Dis. 17:6472017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tavakoli F, Khatami SS, Momeni F,
Azadbakht J and Ghasemi F: Cervical cancer diagnosis: Insights into
biochemical biomarkers and Imaging techniques. Comb Chem High
Throughput Screen. Aug 31–2020.(Epub ahead of print).
|
|
7
|
zur Hausen H: Papillomaviruses and cancer:
From basic studies to clinical application. Nat Rev Cancer.
2:342–350. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
zur Hausen H: Papillomaviruses in the
causation of human cancers-a brief historical account. Virology.
384:260–265. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gillison ML: Human papillomavirus-related
diseases: Oropharynx cancers and potential implications for
adolescent HPV vaccination. J Adolesc Health. 43 (Suppl 4):S52–S60.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mattick JS and Makunin IV: Non-coding RNA.
Hum Mol Genet. 15:R17–R29. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hombach S and Kretz M: Non-coding RNAs:
Classification, biology and functioning. Adv Exp Med Biol.
937:3–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cabianca DS, Casa V and Gabellini D: A
novel molecular mechanism in human genetic disease: A DNA
repeat-derived lncRNA. RNA Biol. 9:1211–1217. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jarroux J, Morillon A and Pinskaya M:
History, discovery, and classification of lncRNAs. Adv Exp Med
Biol. 1008:1–46. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim J, Piao HL, Kim BJ, Yao F, Han Z, Wang
Y, Xiao Z, Siverly AN, Lawhon SE, Ton BN, et al: Long noncoding RNA
MALAT1 suppresses breast cancer metastasis. Nat Genet.
50:1705–1715. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang Y, He L, Du Y, Zhu P, Huang G, Luo J,
Yan X, Ye B, Li C, Xia P, et al: The long noncoding RNA lncTCF7
promotes self-renewal of human liver cancer stem cells through
activation of Wnt signaling. Cell Stem Cell. 16:413–425. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hu X, Feng Y, Zhang D, Zhao SD, Hu Z,
Greshock J, Zhang Y, Yang L, Zhong X, Wang LP, et al: A functional
genomic approach identifies FAL1 as an oncogenic long noncoding RNA
that associates with BMI1 and represses p21 expression in cancer.
Cancer Cell. 26:344–357. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wu WJ, Shen Y, Sui J, Li CY, Yang S, Xu
SY, Zhang M, Yin LH, Pu YP and Liang GY: Integrated analysis of
long non-coding RNA competing interactions revealed potential
biomarkers in cervical cancer: Based on a public database. Mol Med
Rep. 17:7845–7858. 2018.PubMed/NCBI
|
|
18
|
Luan X and Wang Y: LncRNA XLOC_006390
facilitates cervical cancer tumorigenesis and metastasis as a ceRNA
against miR-331-3p and miR-338-3p. J Gynecol Oncol. 29:e952018.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Liu Y, Yang Y, Li L, Liu Y, Geng P, Li G
and Song H: LncRNA SNHG1 enhances cell proliferation, migration,
and invasion in cervical cancer. Biochem Cell Biol. 96:38–43. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen X, Liu S, Zhao X, Ma X, Gao G, Yu L,
Yan D, Dong H and Sun W: Long noncoding RNA ILF3-AS1 promotes cell
proliferation, migration, and invasion via negatively regulating
miR-200b/a/429 in melanoma. Biosci Rep. 37:BSR201710312017.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gao G, Li W, Liu S, Han D, Yao X, Jin J,
Han D, Sun W and Chen X: The positive feedback loop between ILF3
and lncRNA ILF3-AS1 promotes melanoma proliferation, migration, and
invasion. Cancer Manag Res. 10:6791–6802. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hu XH, Dai J, Shang HL, Zhao ZX and Hao
YD: SP1-mediated upregulation of lncRNA ILF3-AS1 functions a ceRNA
for miR-212 to contribute to osteosarcoma progression via
modulation of SOX5. Biochem Biophys Res Commun. 511:510–517. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou M, Hu L, Zhang Z, Wu N, Sun J and Su
J: Recurrence-associated long non-coding RNA signature for
determining the risk of recurrence in patients with colon cancer.
Mol Ther Nucleic Acids. 12:518–529. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ye G, Guo L, Xing Y, Sun W and Yuan M:
Identification of prognostic biomarkers of prostate cancer with
long non-coding RNA-mediated competitive endogenous RNA network.
Exp Ther Med. 17:3035–3040. 2019.PubMed/NCBI
|
|
25
|
Wu W, Sui J, Liu T, Yang S, Xu S, Zhang M,
Huang S, Yin L, Pu Y and Liang G: Integrated analysis of two-lncRNA
signature as a potential prognostic biomarker in cervical cancer: A
study based on public database. PeerJ. 7:e67612019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mao X, Qin X, Li L, Zhou J, Zhou M, Li X,
Xu Y, Yuan L, Liu QN and Xing H: A 15-long non-coding RNA signature
to improve prognosis prediction of cervical squamous cell
carcinoma. Gynecol Oncol. 149:181–187. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Tsikouras P, Zervoudis S, Manav B, Tomara
E, Iatrakis G, Romanidis C, Bothou A and Galazios G: Cervical
cancer: Screening, diagnosis and staging. J BUON. 21:320–325.
2016.PubMed/NCBI
|
|
28
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42((Database Issue)): D92–D97. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Agarwal V, Bell GW, Nam JW and Bartel DP:
Predicting effective microRNA target sites in mammalian mRNAs.
Elife. 4:e050052015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Qu BL, Yu W, Huang YR, Cai BN, Du LH and
Liu F: 6-OH-BDE-47 promotes human lung cancer cells epithelial
mesenchymal transition via the AKT/Snail signal pathway. Environ
Toxicol Pharmacol. 39:271–279. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wong SHM, Fang CM, Chuah LH, Leong CO and
Ngai SC: E-cadherin: Its dysregulation in carcinogenesis and
clinical implications. Crit Rev Oncol Hematol. 121:11–22. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
van Roy F: Beyond E-cadherin: Roles of
other cadherin superfamily members in cancer. Nat Rev Cancer.
14:121–134. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Angst BD, Marcozzi C and Magee AI: The
cadherin superfamily: Diversity in form and function. J Cell Sci.
114:629–641. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Osorio LA, Farfán NM, Castellón EA and
Contreras HR: SNAIL transcription factor increases the motility and
invasive capacity of prostate cancer cells. Mol Med Rep.
13:778–786. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Diepenbruck M and Christofori G:
Epithelial-mesenchymal transition (EMT) and metastasis: Yes, no,
maybe? Curr Opin Cell Biol. 43:7–13. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cheng N, Wu J, Yin M, Xu J, Wang Y, Chen
X, Nie Z and Yin J: LncRNA CASC11 promotes cancer cell
proliferation in hepatocellular carcinoma by inhibiting
miRNA-188-5p. Biosci Rep. 39:BSR201902512019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Slaby O, Laga R and Sedlacek O:
Therapeutic targeting of non-coding RNAs in cancer. Biochem J.
474:4219–4251. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ren L, Chen H, Song J, Chen X, Lin C,
Zhang X, Hou N, Pan J, Zhou Z, Wang L, et al: MiR-454-3p-mediated
Wnt/β-catenin signaling antagonists suppression promotes breast
cancer metastasis. Theranostics. 9:449–465. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Song Y, Guo Q, Gao S and Hua K: miR-454-3p
promotes proliferation and induces apoptosis in human cervical
cancer cells by targeting TRIM3. Biochem Biophys Res Commun.
516:872–879. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang S, Zhang G, Zheng W, Xue Q, Wei D,
Zheng Y and Yuan J: MiR-454-3p and miR-374b-5p suppress migration
and invasion of bladder cancer cells through targetting ZEB2.
Biosci Rep. 38:BSR201814362018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Shao N, Xue L, Wang R, Luo K, Zhi F and
Lan Q: miR-454-3p is an exosomal biomarker and functions as a tumor
suppressor in glioma. Mol Cancer Ther. 18:459–469. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li W, Feng Y, Ma Z and Lu L: Expression of
miR-454-3p and its effect on proliferation, invasion and metastasis
of colon cancer. Nan Fang Yi Ke Da Xue Xue Bao. 38:1421–1426.
2018.(In Chinese). PubMed/NCBI
|
|
45
|
Malaney P, Uversky VN and Davé V: PTEN
proteoforms in biology and disease. Cell Mol Life Sci.
74:2783–2794. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chen CY, Chen J, He L and Stiles BL: PTEN:
Tumor suppressor and metabolic regulator. Front Endocrinol
(Lausanne). 9:3382018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ortega-Molina A and Serrano M: PTEN in
cancer, metabolism, and aging. Trends Endocrinol Metab. 24:184–189.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Nitulescu GM, Van De Venter M, Nitulescu
G, Ungurianu A, Juzenas P, Peng Q, Olaru OT, Grădinaru D, Tsatsakis
A, Tsoukalas D, et al: The Akt pathway in oncology therapy and
beyond (Review). Int J Oncol. 53:2319–2331. 2018.PubMed/NCBI
|
|
49
|
Milella M, Falcone I, Conciatori F, Cesta
Incani U, Del Curatolo A, Inzerilli N, Nuzzo CM, Vaccaro V, Vari S,
Cognetti F and Ciuffreda L: PTEN: Multiple functions in human
malignant tumors. Front Oncol. 5:242015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ye Z, Li Q, Guo Q, Xiong Y, Guo D, Yang H
and Shu Y: Ketamine induces hippocampal apoptosis through a
mechanism associated with the caspase-1 dependent pyroptosis.
Neuropharmacology. 128:63–75. 2018. View Article : Google Scholar : PubMed/NCBI
|