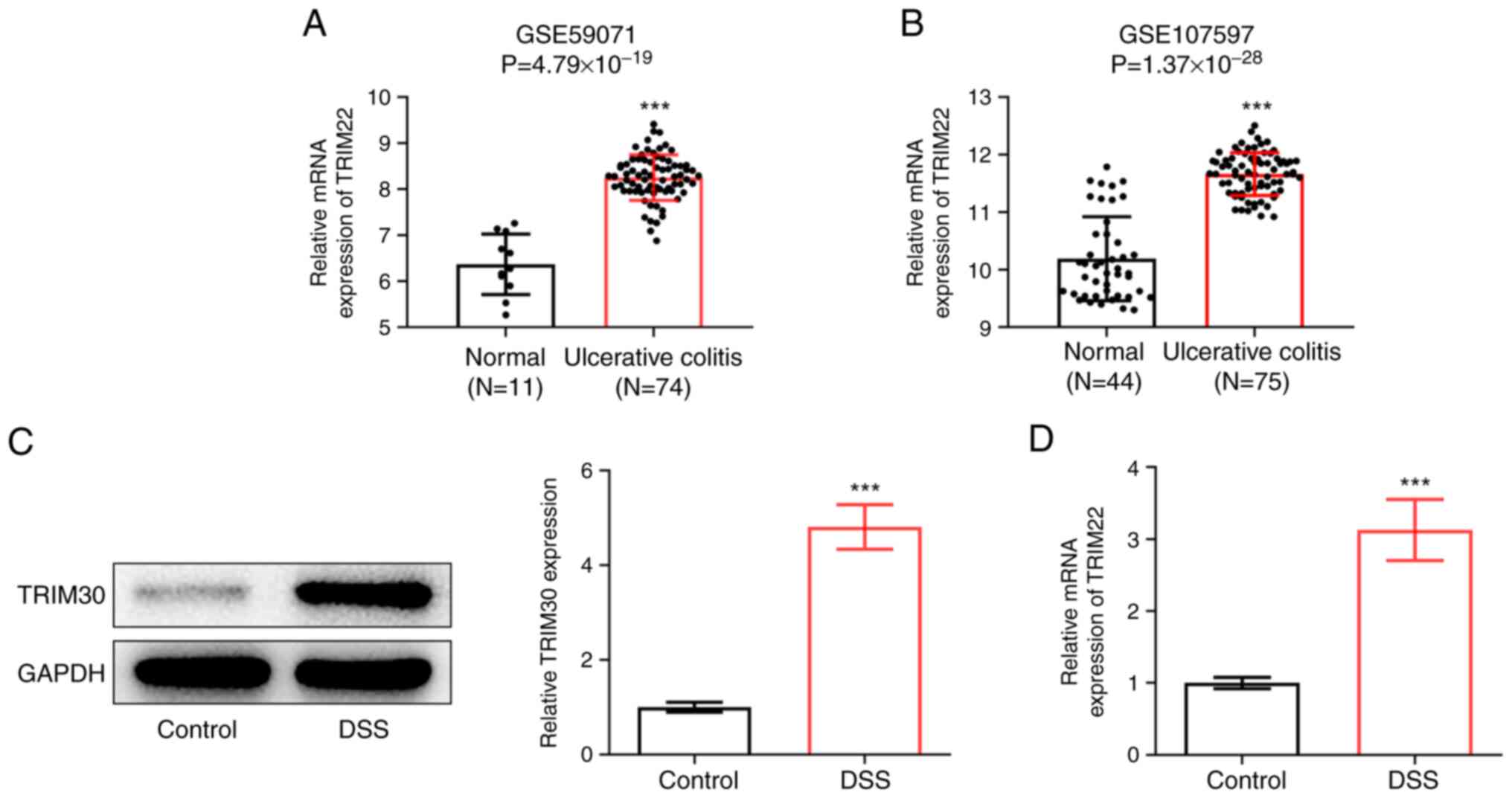
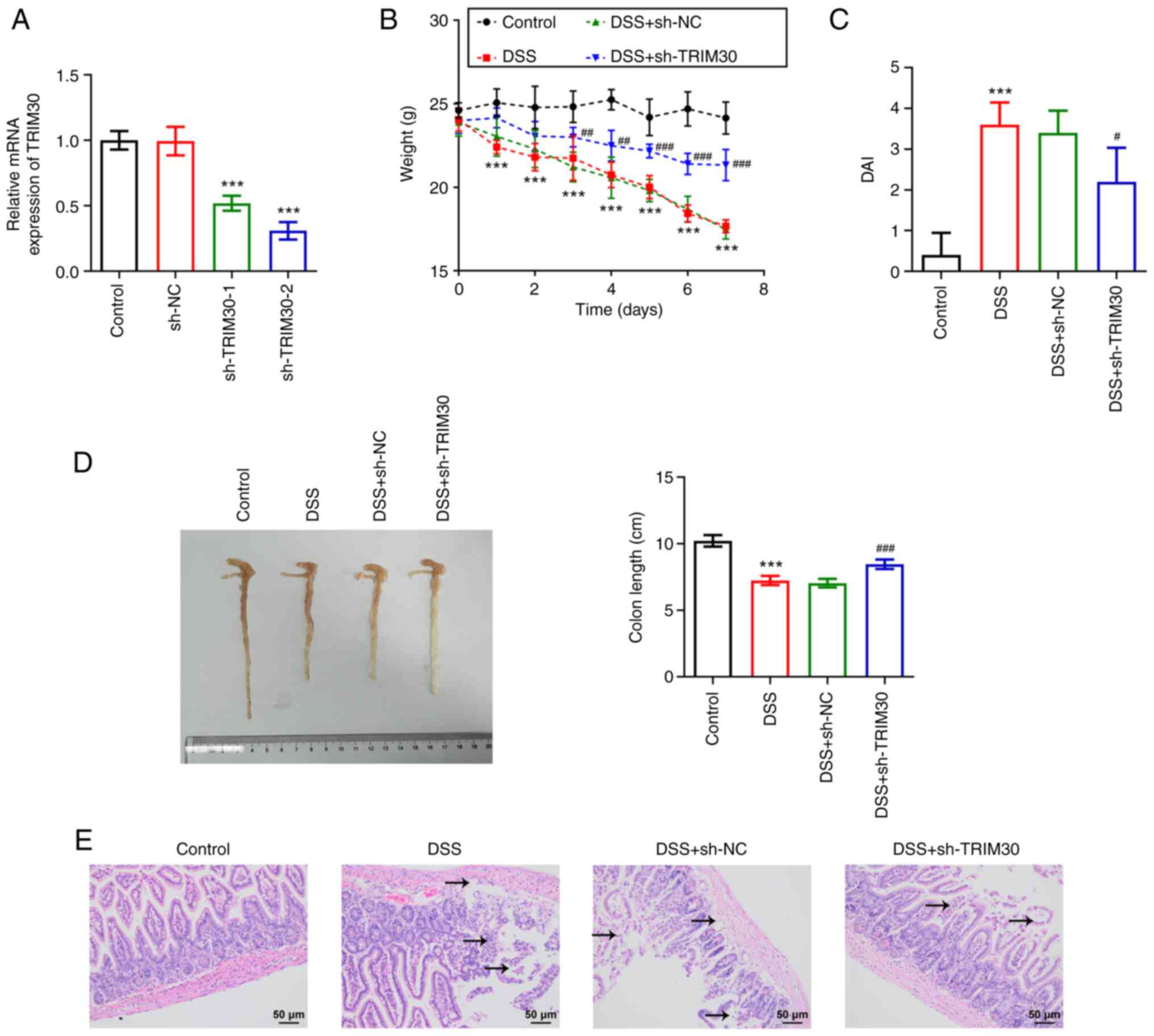
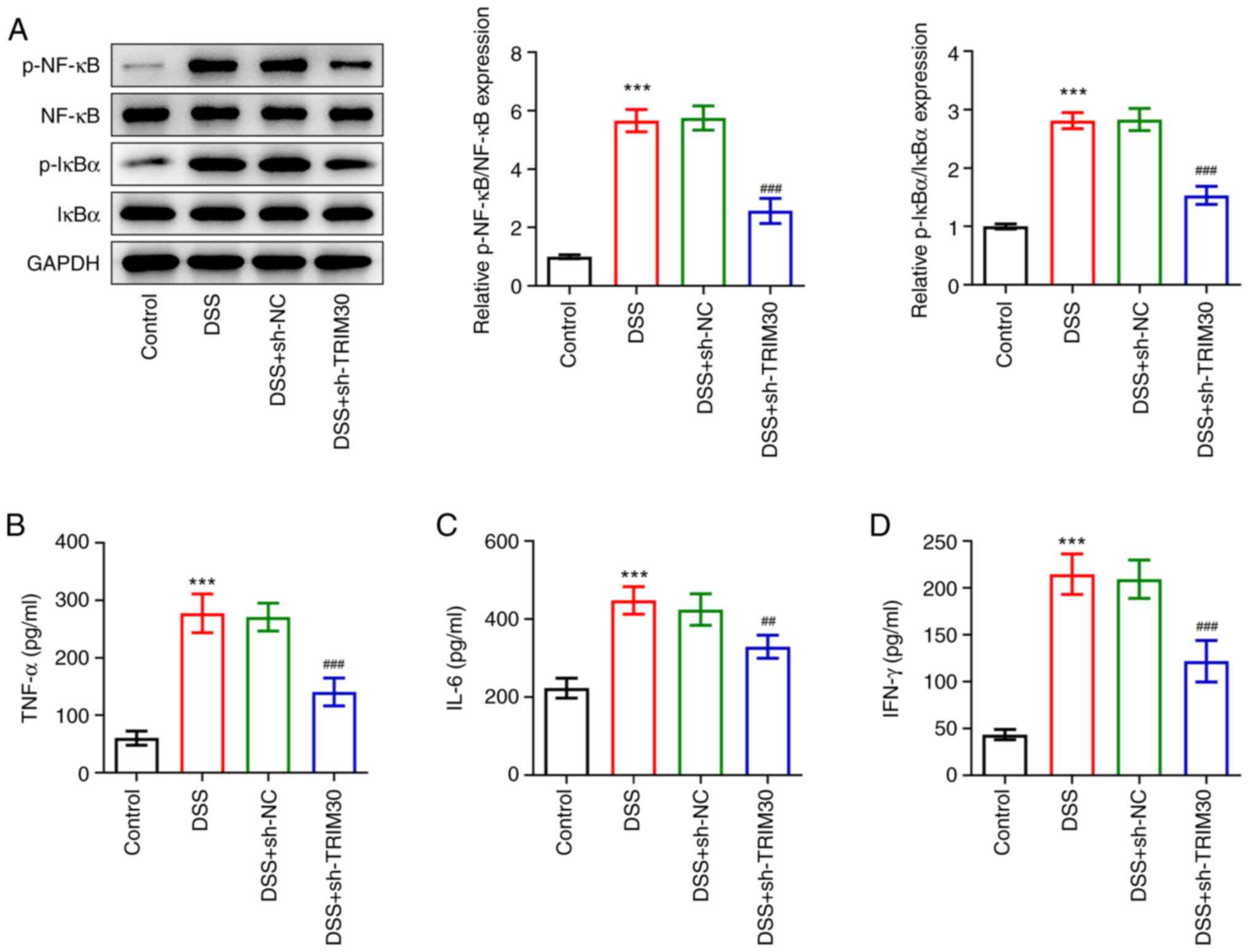
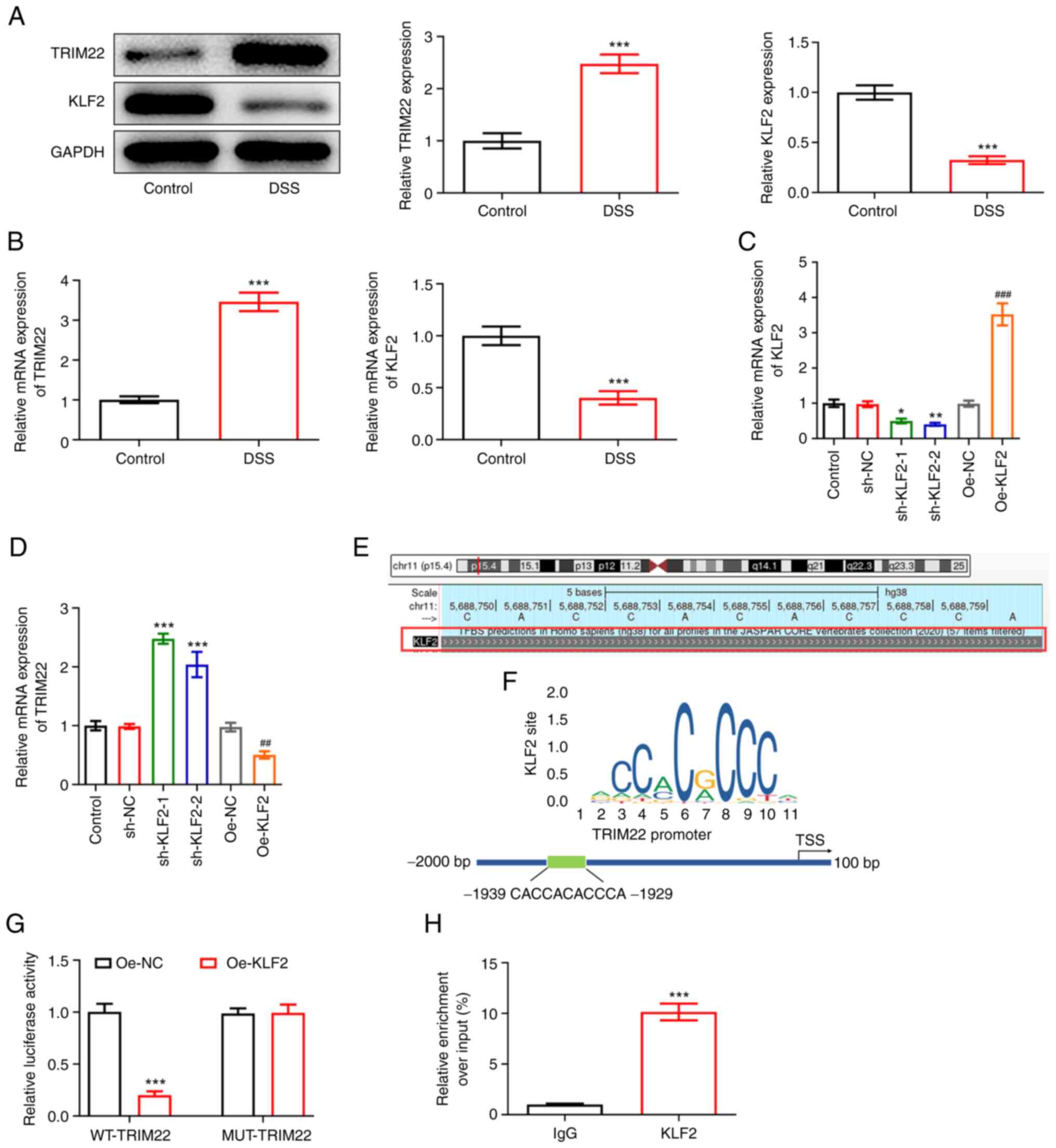
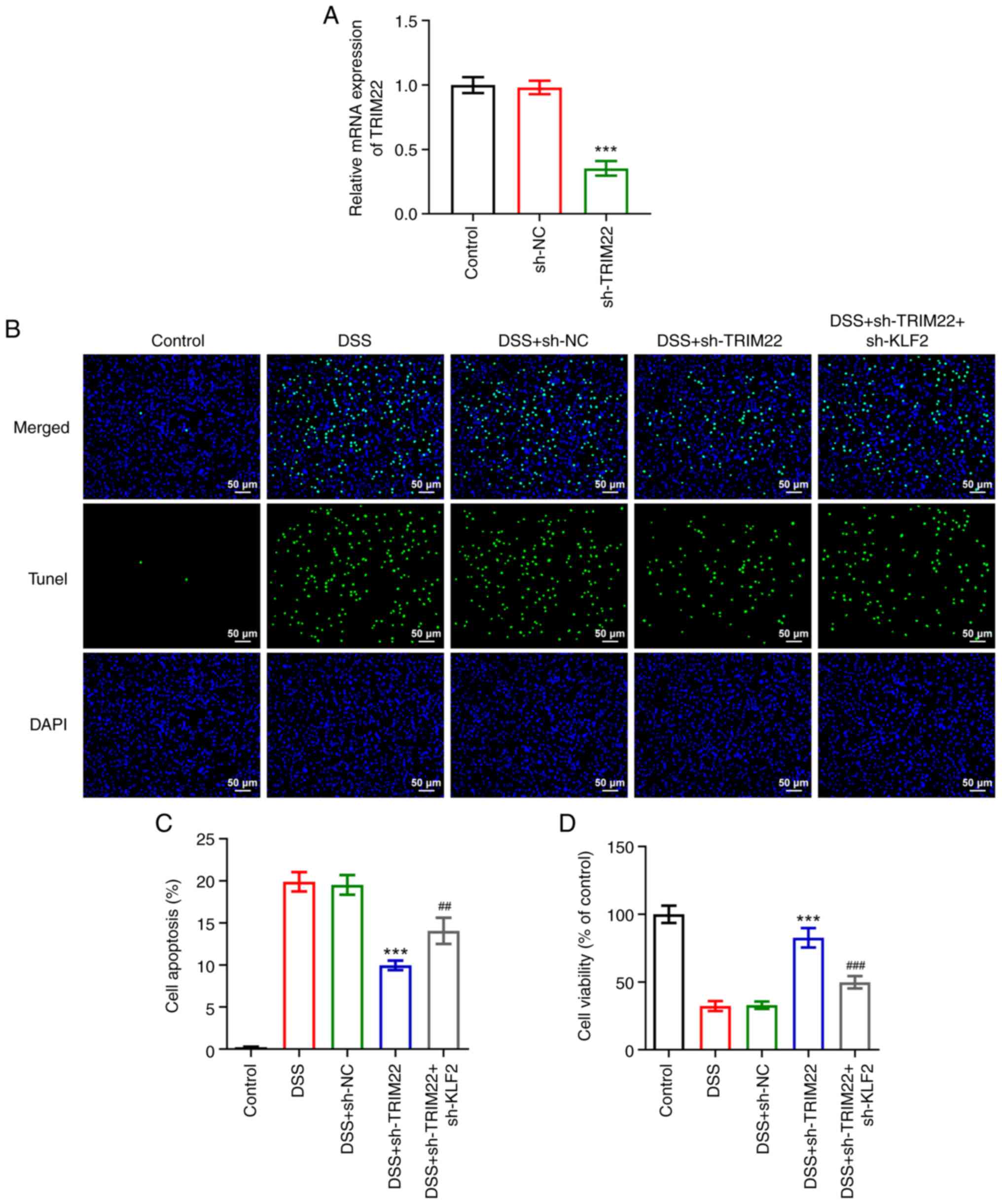
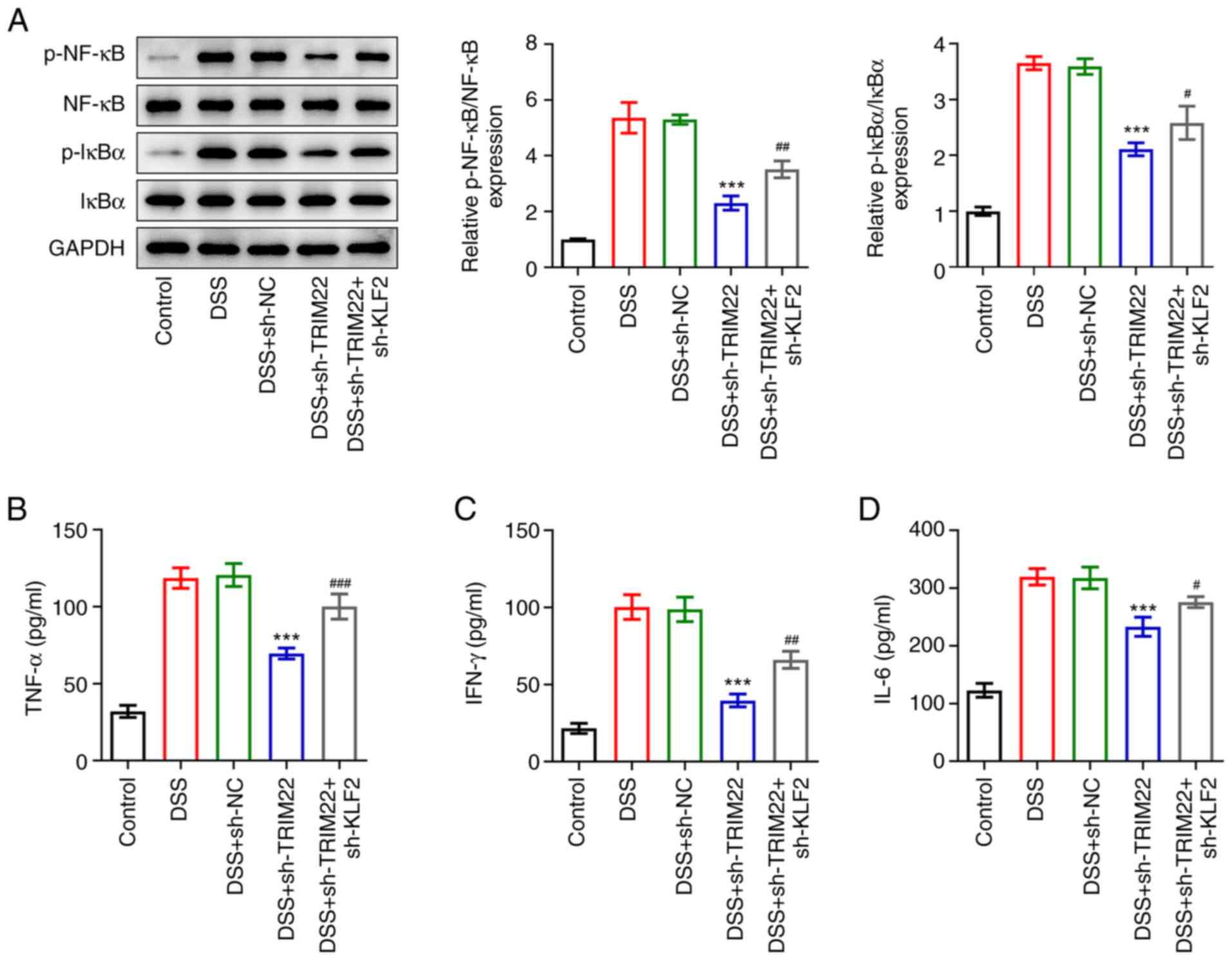
|
1
|
Adams SM and Bornemann PH: Ulcerative
colitis. Am Fam Physician. 87:699–705. 2013.PubMed/NCBI
|
|
2
|
Keshteli AH, Madsen KL and Dieleman LA:
Diet in the pathogenesis and management of ulcerative colitis; a
review of randomized controlled dietary interventions. Nutrients.
11:14982019. View Article : Google Scholar
|
|
3
|
Du L and Ha C: Epidemiology and
pathogenesis of ulcerative colitis. Gastroenterol Clin North Am.
49:643–654. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kaenkumchorn T and Wahbeh G: Ulcerative
colitis: Making the diagnosis. Gastroenterol Clin North Am.
49:655–669. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
da Silva BC, Lyra AC, Rocha R and Santana
GO: Epidemiology, demographic characteristics and prognostic
predictors of ulcerative colitis. World J Gastroenterol.
20:9458–9467. 2014. View Article : Google Scholar
|
|
6
|
Huang Y, Chen J, Wong T and Liow JL:
Experimental and theoretical investigations of non-Newtonian
electro-osmotic driven flow in rectangular microchannels. Soft
Matter. 12:6206–6213. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nowarski R, Jackson R, Gagliani N, de
Zoete MR, Palm NW, Bailis W, Low JS, Harman CC, Graham M, Elinav E
and Flavell RA: Epithelial IL-18 equilibrium controls barrier
function in colitis. Cell. 163:1444–1456. 2015. View Article : Google Scholar
|
|
8
|
Pugliese D, Felice C, Papa A, Gasbarrini
A, Rapaccini GL, Guidi L and Armuzzi A: Anti TNF-α therapy for
ulcerative colitis: Current status and prospects for the future.
Expert Rev Clin Immunol. 13:223–233. 2017. View Article : Google Scholar
|
|
9
|
Li L, Qi Y, Ma X, Xiong G, Wang L and Bao
C: TRIM22 knockdown suppresses chronic myeloid leukemia via
inhibiting PI3K/Akt/mTOR signaling pathway. Cell Biol Int.
42:1192–1199. 2018. View Article : Google Scholar
|
|
10
|
Lou J, Wang Y, Zheng X and Qiu W: TRIM22
regulates macrophage autophagy and enhances Mycobacterium
tuberculosis clearance by targeting the nuclear factor-multiplicity
κB/beclin 1 pathway. J Cell Biochem. 119:8971–8980. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kang C, Lu Z, Zhu G, Chen Y and Wu Y:
Knockdown of TRIM22 relieves oxygen-glucose
deprivation/reoxygenation-induced apoptosis and inflammation
through inhibition of NF-κB/NLRP3 axis. Cell Mol Neurobiol.
41:341–351. 2021. View Article : Google Scholar
|
|
12
|
Li Q, Lee CH, Peters LA, Mastropaolo LA,
Thoeni C, Elkadri A, Schwerd T, Zhu J, Zhang B, Zhao Y, et al:
Variants in TRIM22 that affect NOD2 signaling are associated with
very-early-onset inflammatory bowel disease. Gastroenterology.
150:1196–1207. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu R, Zhao W, Wang H and Wang J: Long
noncoding RNA LINC01207 promotes colon cancer cell proliferation
and invasion by regulating miR-3125/TRIM22 axis. Biomed Res Int.
2020:12163252020. View Article : Google Scholar
|
|
14
|
Chen C, Zhao D, Fang S, Chen Q, Cheng B,
Fang X and Shu Q: TRIM22-mediated apoptosis is associated with bak
oligomerization in monocytes. Sci Rep. 7:399612017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ji F and Sadreyev RI: RNA-seq: Basic
bioinformatics analysis. Curr Protoc Mol Biol. 124:e682018.
View Article : Google Scholar
|
|
16
|
Canzoneri R, Lacunza E and Abba MC:
Genomics and bioinformatics as pillars of precision medicine in
oncology. Medicina (B Aires). 79:(Spec 6/1). 587–592.
2019.PubMed/NCBI
|
|
17
|
Wang ZL, Wang YD, Wang K, Li JA and Li L:
KFL2 participates in the development of ulcerative colitis through
inhibiting inflammation via regulating cytokines. Eur Rev Med
Pharmacol Sci. 22:4941–4948. 2018.
|
|
18
|
Liang Y, Luo J, Yang N, Wang S, Ye M and
Pan G: Activation of the IL-1β/KLF2/HSPH1 pathway promotes STAT3
phosphorylation in alveolar macrophages during LPS-induced acute
lung injury. Biosci Rep. 40:BSR201935722020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Vanhove W, Peeters PM, Staelens D, Anica
S, der Goten JV, Cleynen I, De Schepper S, Van Lommel L, Reynaert
NL, Schuit F, et al: Strong upregulation of AIM2 and IFI16
inflammasomes in the mucosa of patients with active inflammatory
bowel disease. Inflamm Bowel Dis. 21:2673–2682. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ouahed J, Gordon W, Canavan JB, Zhou HY,
Du S, von Schack D, Phillips K, Wang L, Dunn WA III, Field M, et
al: Mucosal gene expression in pediatric and adult patients with
ulcerative colitis permits modeling of ideal biopsy collection
strategy for transcriptomic analysis. Inflamm Bowel Dis.
24:2565–2578. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cheng F, Li Q, Wang J, Zeng F, Wang K and
Zhang Y: Identification of differential intestinal mucosa
transcriptomic biomarkers for ulcerative colitis by bioinformatics
analysis. Dis Markers. 2020:88765652020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cardon AD, Bailey MR and Bennett BT: The
animal welfare act: From enactment to enforcement. J Am Assoc Lab
Anim Sci. 51:301–305. 2012.
|
|
23
|
Kihara N, de la Fuente SG, Fujino K,
Takahashi T, Pappas TN and Mantyh CR: Vanilloid receptor-1
containing primary sensory neurones mediate dextran sulphate sodium
induced colitis in rats. Gut. 52:713–719. 2003. View Article : Google Scholar
|
|
24
|
Chen Q, Fang X, Yao N, Wu F, Xu B and Chen
Z: Suppression of miR-330-3p alleviates DSS-induced ulcerative
colitis and apoptosis by upregulating the endoplasmic reticulum
stress components XBP1. Hereditas. 157:182020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Mehods. 25:402–408. 2001.
|
|
26
|
Yamamoto-Furusho JK, Gutiérrez-Grobe Y,
López-Gómez JG, Bosques-Padilla F and Rocha-Ramírez JL; Grupo del
Consenso Mexicano de Colitis Ulcerosa Crónica Idiopática, : The
Mexican consensus on the diagnosis and treatment of ulcerative
colitis. Rev Gastroenterol Mex (Engl Ed). 83:144–167. 2018.(In
English, Spanish). PubMed/NCBI
|
|
27
|
Ashton JJ, Mossotto E, Stafford IS,
Haggarty R, Coelho TAF, Batra A, Afzal NA, Mort M, Bunyan D,
Beattie RM and Ennis S: Genetic sequencing of pediatric patients
identifies mutations in monogenic inflammatory bowel disease genes
that translate to distinct clinical phenotypes. Clin Transl
Gastroenterol. 11:e001292020. View Article : Google Scholar
|
|
28
|
Lawrence T: The nuclear factor NF-kappaB
pathway in inflammation. Cold Spring Harb Perspect Biol.
1:a0016512009. View Article : Google Scholar
|
|
29
|
Ji J, Ding K, Luo T, Zhang X, Chen A,
Zhang D, Li G, Thorsen F, Huang B, Li X and Wang J: TRIM22
activates NF-κB signaling in glioblastoma by accelerating the
degradation of IκBα. Cell Death Differ. 28:367–381. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jha P and Das H: KLF2 in regulation of
NF-κB-mediated immune cell function and inflammation. Int J Mol
Sci. 18:23832017. View Article : Google Scholar
|
|
31
|
Zhuang T, Liu J, Chen X, Zhang L, Pi J,
Sun H, Li L, Bauer R, Wang H, Yu Z, et al: Endothelial Foxp1
suppresses atherosclerosis via modulation of Nlrp3 inflammasome
activation. Circ Res. 125:590–605. 2019. View Article : Google Scholar
|