Introduction
Diabetes mellitus (DM) has become a global public
health crisis, affecting 537,000,000 individuals worldwide in 2021
(1) and has been identified as a
risk factor for dry eye (DE) disease (2,3).
Clinical studies have found that over half of patients with
diabetes have DE disease, which progresses as the duration of
diabetes increases (4,5). Diabetic DE disease is characterized
by decreased corneal sensitivity, reduced tear secretion, altered
tear proteins and corneal epithelial lesions (6–9).
Hyperglycemia impairs corneal epithelial basal cells and the
basement membrane (10), but the
specific mechanism by which corneal epithelial lesions form remains
unclear. Therefore, understanding the molecular mechanism of
corneal epithelial dysfunction in diabetic DE disease is
desirable.
Mucin1 (MUC1) is a member of the mucin family, which
contains O-glycosylated proteins that play key roles in the tear
film homeostasis and protect the ocular surface (11,12).
MUC1 is expressed across the cornea and conjunctival epitheliums in
humans but only in the corneal epithelium in mice (13,14).
The extracellular domain of the N-terminal alpha subunit of MUC1
(MUC1-ND) is responsible for cell adhesion and the cytoplasmic tail
of MUC1 (MUC1-CT) is involved in cell signaling (15). Previous studies have reported that
the MUC1-CT carries out extensive functions beyond its ability to
interact with several proteins, such as Src, catenin β1 (CTNNB1)
and ErbBs (16,17). For example, MUC1-CT promotes cell
adhesion by binding CTNNB1/β-catenin and junction
plakoglobin/γ-catenin. Additionally, MUC1-CT facilitates
RAB27A-mediated exosome secretion by interacting with the pocket of
RAB27A (18). Studies have also
found that MUC1 is extracellularly cleaved within the sperm
protein, enterokinase and agrin (SEA) module in MUC1-ND by ADAM17
or MT1MMP (19,20). Miyazaki et al (21) show that MUC1-ND functions as a
hydrophilic barrier that hinders the entry of lipophilic anticancer
drugs, leading to chemoresistance. Increasing evidence suggests
that MUC1 is also implicated in inflammatory diseases (22,23).
However, the function of MUC1 and the underlying molecular
mechanism involved in diabetic DE disease remain unclear.
Transient receptor potential cation channel
subfamily V member 1 (TRPV1), a nociceptive sensor, is associated
with ocular surface pain (24).
TRPV1 enhances the excitability of nociceptive sensors activated by
heat, protons and inflammatory mediators (25). TRPV1 channel dysfunction is shown
to induce decreased corneal sensitivity, which is correlated with
the pathogenesis of DE disease (6). Notably, TRPV1 is found to be
translocated to astrocyte membranes to promote migration and
inflammatory infiltration in models of neonatal hypoxic-ischemia
and hypoxic hypoglycemia (26,27),
but it is involved in the protection of diabetic rats after hypoxic
preconditioning (28), suggesting
that TRPV1 plays a complex role in the inflammatory process. The
TRPV1 channel activation is also shown to induce the proliferation
and migration of human corneal epithelial cells (29). MUC1 interacts with TRPV5 through
galectin 3, enhancing renal TRPV5 activity and preventing kidney
stone formation (30). In
addition, MUC1 decreases the internalization of Ca-selective TRP
channels TRPV5 and TRPV6 while promoting their presence on the cell
surface, aiding in maintaining Ca2+ balance (31). These findings highlight the close
relationship between MUC1 and TRP channels. However, whether MUC1
interacts with TRPV1 and the underlying signaling mechanism in
corneal epithelial cell proliferation remain to be elucidated.
The present study found decreased proliferation of
mouse corneal epithelial cells (MCECs), accompanied by decreased
levels of MUC1-ND and TRPV1, in diabetic DE mice. In addition,
capsazepine (CPZ; a TRPV1 antagonist) restricted the proliferation
of MCECs. There was an interaction between MUC1-ND and the TRPV1
protein in the control group but not in the diabetic DE group.
Furthermore, the activation of the AKT signaling pathway was
attenuated in diabetic DE mice and it was downstream of TRPV1. In
summary, the results illustrated that the MUC1-ND/TRPV1/AKT axis
plays an essential role in the proliferation of MCECs and provided
new insight into the pathogenesis of corneal epithelial dysfunction
in diabetic DE disease.
Materials and methods
Reagents
Primary antibodies against the following were
provided by Abcam: MUC1-ND (cat. no. ab45167), AKT1/2/3 (cat. no.
ab185633), p-AKT (Thr308; cat. no. ab38449), Bcl2 (cat. no.
ab182858), β-Actin (cat. no. ab8227) and β-tubulin (cat. no.
ab6046). Primary antibodies against the following were purchased
from Proteintech Group, Inc.: MUC1-CT (cat. no. 23614-1-AP), TRPV1
(cat. no. 66983-1-Ig) and phosphorylated (p-)AKT (Ser473; cat. no.
28731-1-AP). Primary antibodies against the following were
purchased from Cell Signaling Technology, Inc.: Caspase3 (cat. no.
9662) and IgG (cat. no. 2729). The primary antibodies of CK3+12
(cat. no. Bs-2369R) and TRPV1 (cat. no. Bs-23926R) were purchased
from BIOSS. The following secondary antibodies were obtained from
Invitrogen (Thermo Fisher Scientific, Inc.): Alexa Fluro 488 (goat
anti-mouse, cat. no. a11029; goat anti-rabbit, cat. no. a11008) and
Alexa Fluro 594 (goat anti-mouse, cat. no. a11032; goat
anti-rabbit, cat. no. a11012). The following secondary antibodies
were obtained from Abcam: goat anti-rabbit IgG/HRP (cat. no.
ab6721) and goat anti-mouse IgG/HRP (cat. no. ab6789). CPZ (cat.
no. HY15640) and MK2206 (cat. no. HY-10358) were purchased from
MedChemExpress.
Animals
Male C57BL/6 mice at aged of 6–8-weeks and weighing
18–22 g were purchased from the Experimental Animal Center of
Southern Medical University (Guangzhou, China). Mice were cared for
in accordance with the ARVO Statement for the Use of Animals in
Ophthalmic and Vision Research (32). All experimental procedures received
approval from the Institutional Animal Care and Use Committee of
Southern Medical University (Guangzhou, China; approval no.
LAEC-2021-032). The mice were housed in a specific pathogen-free
rated animal vivarium (temperature, 20–26°C; light/dark cycle,
12/12 h; humidity: 40–70%) and the research team monitored the
animals twice daily. Health assessments included monitoring weight
(twice weekly), food and water intake and a general evaluation of
activity levels, panting and fur condition. A total of 155 mice
were used, of which 149 were euthanized using carbon dioxide
(CO2) inhalation. The absence of a heartbeat and
dilation of pupils for a period of ≥5 min were verified in order to
confirm mortality; if these conditions were not met, cervical
dislocation was then performed. Sacrificed mice were used for
corneal and conjunctival histopathology, as well as for the primary
culture of mouse corneal epithelial cells. Of the diabetic group,
six succumbed unexpectedly, probably due to complications
associated with diabetes. The duration of the experiment ranged
from 1.5–2.5 months. The ocular surface parameter measurements were
conducted in a non-invasive and painless manner. Additionally, 3%
sodium pentobarbital was administered intraperitoneally at a dose
of 50 mg/kg to induce sedation in the mice.
Diabetic DE mouse model
A streptozotocin (STZ; MilliporeSigma)-induced
diabetic mouse model was established and Na-citrate buffer
administered to the control group. To induce DM, mice were fasted
for 4 h and injected with a low dose of STZ (50 mg/kg) for 5
consecutive days. Mice were fed a normal diet (control group) or a
high-fat/high-sugar diet (DM group). Corneal fluorescein staining
(CFS), tear ferning (TF) tests and phenol red thread tests were
performed before treatment and 7, 30 and 60 days after the final
injection. Corneal sections were conducted hematoxylin and eosin
(H&E) staining on days 30 and 60 after the final injection and
conjunctival sections were stained with periodic acid Schiff (PAS)
on days 30 and 60 after the final injection. After the
aforementioned evaluation, a diabetic DE mouse model was
established and the mice were divided into a control group and a
diabetic DE group.
CFS
To estimate the defects of corneal epithelium in the
mice, 1 µl of 1% liquid sodium fluorescein was dropped into the
lateral conjunctival sac. After 3 min, the eyes were imaged with a
slit-lamp microscope (SL-17; Kowa Optimed, Inc.) under cobalt blue
light. A total of four quadrants of cornea were scored by two
independent blinded individuals using a standardized four-scale
system: 0 points for no staining; 1 point for <30 stained dots;
2 points for >30 non-diffuse stained dots; 3 points for severe
diffuse staining but no positive plaques; and 4 points for positive
fluorescein plaques. The total score was the sum of four quadrants
scores (0–16).
TF tests
Tear samples (0.5 µl) collected from each eye of the
mice were dried on a microscopic glass slide at room temperature
and <40% humidity for 20 min. The TF patterns were imaged under
a Leica bright field microscope (Leica Microsystems GmbH;
magnification, ×10) and analyzed using a new 5-point grading scale
(33) from 0 to 4; a grade higher
than grade 2 indicated possible DE disease.
Tear production
The amount of tears was quantified by the phenol red
thread (Zone-Quick; Showa Yakuhin Kako Co., Ltd.). A smooth tweezer
placed the folded end of the thread into the temporal one-third of
the lateral conjunctival sac for 15 sec. After 15 sec, the wetting
length of thread (red portion) was measured in millimeters.
Histopathology
The eyeball and eyelid tissues of the mice were
excised and fixed in 4% paraformaldehyde (PFA) at 4°C overnight.
The tissues were dehydrated using varying concentrations of
ethanol, followed by transparency treatment with xylene, immersion
in wax, and embedding in paraffin. Paraffin sections were cut into
4 µm sagittal sections with a microtome (RM2245; Leica Microsystems
GmbH). Frozen sections were sliced into sagittal sections (6–10 µm
thick) with a microtome (CM1950; Leica Microsystems GmbH). H&E
or PAS were performed on the paraffin sections, while the frozen
sections underwent immunofluorescence (IF) staining.
To evaluate defects in the corneal epithelium, eye
sections were stained with hematoxylin for 5 min and eosin for 1
min at room temperature. To evaluate the goblet cell density of the
conjunctiva, PAS staining of the conjunctiva sections was performed
using a PAS kit (Beijing Solarbio Science & Technology Co.,
Ltd.). The eye sections were stained with periodic acid for 5 min
and Schiff for 10 min at room temperature. Images of the staining
sections were captured under a microscope (DM2500; Leica
Microsystems GmbH). The thickness of the corneal epithelium in
captured images was measured with ImageJ 1.8 (National Institutes
of Health). The goblet cells in the superior and inferior
conjunctiva were counted by two independent blinded individuals in
three sections of each eye. The average number of the three
sections was recorded as the goblet cell density of the eye.
Isolation and culture of primary mouse
corneal epithelial cells (MCECs)
MCECs were isolated by enzymatic digestion. The mice
from the control group and diabetic DE group were sacrificed by
CO2. Animals were placed into CO2 anesthesia
box with ≥7.5% CO2 and after the animals gradually lost
consciousness, the CO2 concentration was raised to 100%
until respiratory cardiac arrest occurred. Their eyes were
surgically extracted, placed into a 10 cm dish (Corning Life
Sciences) and washed with PBS three times. Under a Leica S9
stereomicroscope (Leica Microsystems GmbH), the lens and posterior
eyecups were discarded to detach the corneas, which were dissected
into four equal pieces. Afterward, the corneas were transferred
into a new 10 cm dish containing 0.25% EDTA-trypsin (Gibco; Thermo
Fisher Scientific, Inc.) and digested at 37°C for 10 min. The
control group cells were maintained in DMEM/F12 (Gibco; Thermo
Fisher Scientific, Inc.) containing 10% FBS, 1% nonessential amino
acids (Gibco; Thermo Fisher Scientific, Inc.), insulin (5 µg/ml;
MilliporeSigma) and 100 U/ml penicillin/streptomycin (Gibco; Thermo
Fisher Scientific, Inc.). The diabetic DE group cells were cultured
in DMEM with high glucose and the same supplements used to maintain
the control group.
CCK8 assay
Cell Counting Kit-8 (CCK8; Gibco; Thermo Fisher
Scientific, Inc.) solution was used to detect the cell
proliferative capacity of each group or treated cells. Primary
MCECs from each group were cultured at 2,000 cells per well in
96-well plates (Corning Life Sciences) for 3 days. CCK8 solution
(10 µl) was added to each well at 4, 24, 48 and 72 h and then
incubated at 37°C under 5% CO2 for 2 h. The optical
density (OD) was measured at 450 nm wavelength via a microplate
reader (BioTek; Agilent Technologies, Inc.) and the cell growth
curves were plotted.
To evaluate the effect of CPZ or MK2206 on cell
viability, primary MCECs from each group were treated with
DMSO-soluble CPZ (0, 10, 30 µM) or water-soluble MK2206 (1 µM) for
24 h. After the addition of CCK8 solution and incubation for 1.5 h,
the OD was measured and the cell viability was calculated.
Cell IF staining
Primary MCECs were stained with cytokeratin 3+12
(CK3+12) for cell identification. The primary MCECs were cultured
on 6-well plates (Corning Life Sciences) with round cover slips (12
mm) for 24 h. Then, the cells were first fixed in 4% PFA for 15 min
at room temperature and blocked with 5% BSA (Thermo Fisher
Scientific, Inc.) in PBS. Following incubation with the primary
antibodies anti-CK3+12 (1:200), anti-MUC1-CT (1:200) and anti-TRPV1
(1:200) and fluorescent secondary antibodies (1:500), the cells
were counterstained with DAPI for 5 min at room temperature (cat.
no. ab285390; Abcam). Images were captured with an inverted
confocal microscope (Nikon Corporation).
IF colocalization analysis
Frozen corneal sections were fixed in cold acetone
for 10 min at 4°C. After blocking with 5% BSA in PBS for 1 h at
room temperature, they were incubated with anti-MUC1-ND (1:200) and
anti-TRPV1 (1:300) overnight at 4°C and stained with secondary
antibodies (1:500) for 1 h at room temperature, then counterstained
with DAPI for 5 min at room temperature. Images were captured with
a Nikon Ti-E microscope (Nikon Corporation) equipped with an A1R
confocal system. IF colocalization analysis was conducted with
ImageJ 1.8 (National Institutes of Health).
Western blotting
Primary MCECs from each group or treated cells were
lysed in RIPA (Thermo Fisher Scientific, Inc.) lysis buffer mixed
with 1% phosphate inhibitor cocktail 100X (Gibco; Thermo Fisher
Scientific, Inc.) and 1% protease inhibitor cocktail 100X (Gibco;
Thermo Fisher Scientific, Inc.). A BCA protein assay kit (Nanjing
KeyGen Biotech Co., Ltd.) was used to examine the protein
concentration. Equal amounts of protein samples were loaded to
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
(SDS-PAGE) and moved to polyvinylidene fluoride membranes
(MilliporeSigma). A total of 30 µg protein/lane was loaded, and 5%
stacking gel and 8, 10 and 12% separating gel was used. The
membranes were blocked with 5% BSA for 2 h at room temperature and
incubated with primary antibodies against the following overnight
at 4°C: p-AKT (Ser473) (1:1,000), MUC1-CT (1:800), MUC1-ND
(1:1,000), TRPV1 (1:1,000), AKT1/2/3 (1:1,500), p-AKT (Thr308)
(1:800), Bcl2 (1:2,000), caspase3 (1:1,000) and β-Actin (1:1,000).
After incubation with secondary antibodies (1:2,000) for 1 h, the
bands were detected by an enhanced chemiluminescence (ECL) system
(Bio-Rad ChemiDoc XRS+; Bio-Rad Laboratories, Inc.). Quantification
of the band intensity was conducted with ImageJ 1.8 (National
Institutes of Health).
Co-immunoprecipitation (Co-IP)
assays
Co-IP assays were performed by using a Pierce
cross-linked bead immunoprecipitation/coprecipitation kit (cat. no.
88805; Thermo Fisher Scientific, Inc.). First, primary MCECs were
lysed with IP lysis buffer and centrifuged at 10,000 × g for 10 min
at 4°C. Then, the lysates with 500 µg proteins were
immunoprecipitated with 5 µg anti-TRPV1 (1:20), 5 µg anti-MUC1-ND
(1:20) or 1 µg IgG (1:50) coupled to protein A/G magnetic beads at
room temperature for 1.5 h. The beads were washed in IP buffer and
eluted in IP elution buffer for 10 min. All incubations were taken
on a rotator or mixer. The beads were washed to remove unbound
proteins, and a low pH elution buffer was used to separate bound
antigen from the antibody cross-linked beads. Neutralization buffer
was included to prevent precipitation of the isolated antigen and
to ensure protein activity in downstream applications. Western
blotting was conducted to detect the bound proteins.
Statistical analysis
All data are shown as the mean ± standard deviation.
Statistical tests were performed by SPSS 20.0 (IBM Corp.) software
and graphs were prepared by Prism 8.0 (GraphPad; Dotmatics)
software. Student's t-test was used to compare differences of two
groups. Comparisons between more than two groups were made by
one-way ANOVA followed by the LSD test or Welch test with Dunn's
multiple-comparisons test. P<0.05 was considered to indicate a
statistically significant difference.
Results
Establishment of a diabetic DE mouse
model
To establish a diabetic DE mouse model, STZ-induced
diabetic mice were created on a high-fat and high-sugar diet and
phenol red thread tests, CFS, TF tests and histological
examinations were conducted to evaluate DE disease.
First, STZ was used to induce diabetes in mice and
the mice were then followed for 60 days following the final STZ
injection to determine changes in the ocular surface as the
duration of diabetes increased (Fig.
1A). Mice in the DM group were confirmed to have higher blood
glucose levels, lower body weights and increased food intake
compared with those of mice in the control group (Table I), indicating that the diabetic
mice had been successfully created. In addition, the mice were
observed at baseline and 7, 30 and 60 days following treatment by
phenol red thread, CFS and TF tests (Fig. 1B). As illustrated in Fig. 1C, the tear production was similar
level between the control and DM groups at baseline and on days 7
after treatment. However, the diabetic mice exhibited decreased
tear production on days 30 and 60 after STZ injection. These
results demonstrated that the lacrimal gland function in secreting
tear fluids was impaired in the DM group on days 30 and 60 after
STZ injection.
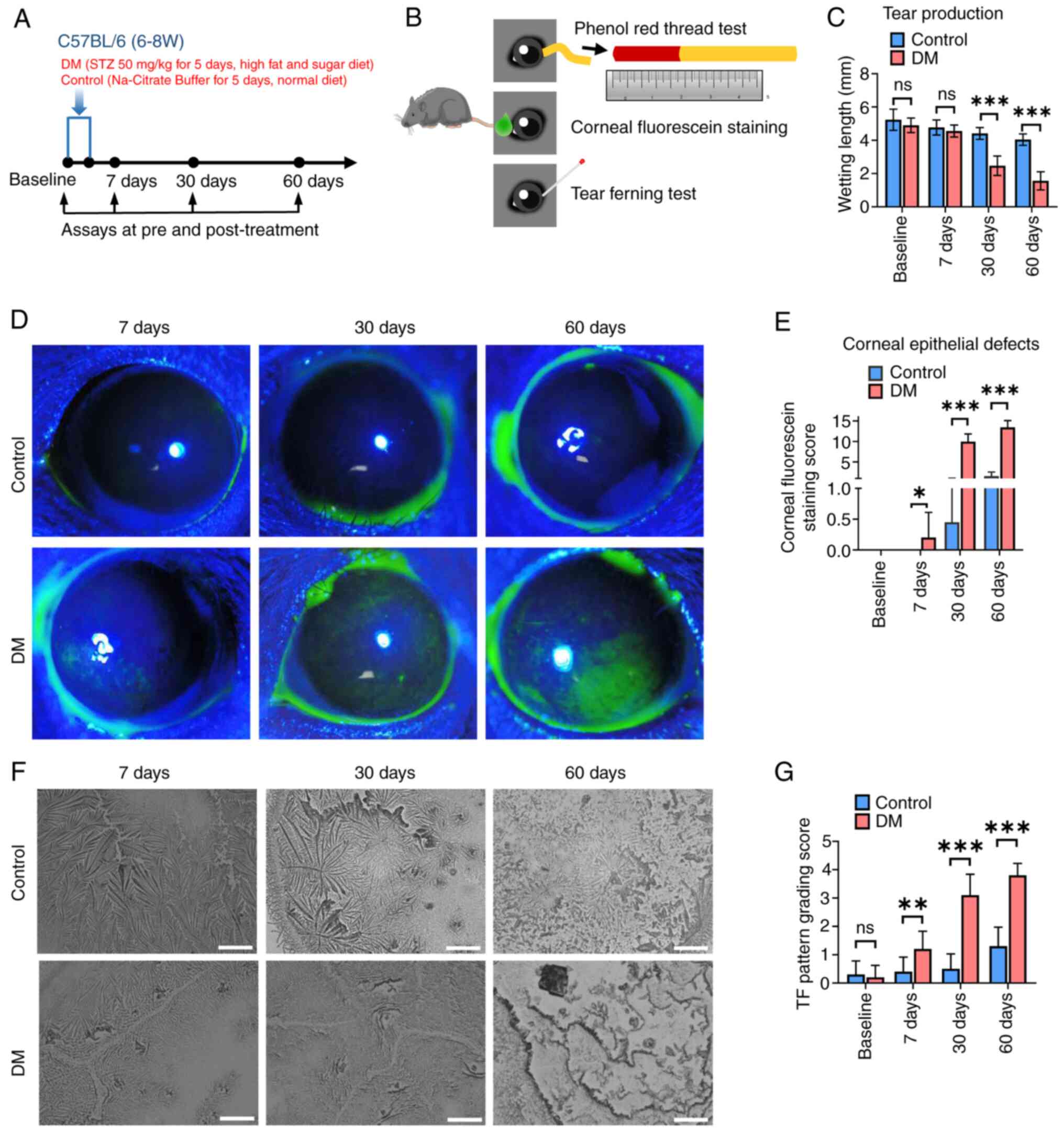 | Figure 1.STZ-induced diabetic mice and
evaluation of DE disease. (A and B) Experimental protocol for
diabetic mice preparation and assays to evaluate DE disease at
various time points. (C) Quantitative analysis of tear production
using the phenol red thread test. (D) CFS was imaged by a slit-lamp
microscope under cobalt blue light on days 7, 30 and 60 after final
injection. Green-stained areas represent corneal epithelial
defects. (E) Quantitative analysis of corneal epithelial damage.
(F) Representative images captured during the TF test at baseline
and days 7, 30 and 60 after final injection. Scale bar, 100 µm. (G)
Quantitative analysis of TF pattern grades. In C, E and G, the mean
values ± standard deviation are shown (n=20 eyes, 10 animals per
group). A two-sided Student's t test was used to calculate the
statistical significance. ns, not significant, *P<0.05,
**P<0.01, ***P<0.001. STZ, streptozotocin; DE, dry eye; CFS,
corneal fluorescein staining; TF, tear ferning. |
 | Table I.Blood glucose, weight and food intake
of mice before and after treatment. |
Table I.
Blood glucose, weight and food intake
of mice before and after treatment.
| Group |
| Control | Diabetes
mellitus |
|---|
| Blood glucose
(mmol/l) | Baseline | 6.24±0.78 | 5.18±0.57 |
|
| 7 days | 6.80±0.84 |
24.83±1.94a |
|
| 30 days | 7.72±0.62 |
25.49±1.83a |
| Body weight
(g) | Baseline | 20.18±0.64 | 20.35±0.71 |
|
| 7 days | 21.28±0.64 | 21.81±0.78 |
|
| 30 days | 26.96±0.81 |
23.61±1.08a |
| Food intake
(g/day) | Baseline | 4.70±0.43 | 5.05±0.50 |
|
| 7 days | 5.55±0.71 |
8.75±0.58a |
|
| 30 days | 6.16±0.58 |
8.83±0.46a |
CFS (Fig. 1D) of
the DM group after STZ injection showed scattered punctate staining
on days 7, severe diffuse staining on days 30 and positive
fluorescein plaque staining on days 60. However, no staining of the
corneas of the control group was observed. The DM group mice had a
higher CFS score than the control group mice (Fig. 1E). After treatment, corneal
epithelial defects in the diabetic mice increased on days 30 and
days 60 following STZ injection.
The diabetic mice exhibited decreased TF and poor TF
pattern grades on days 7, 30 and 60 after treatment (Fig. 1F and G). At baseline, there was no
significant difference between the two groups. On 7, 30 and 60 days
after treatment, the TF grade of the DM group was significantly
higher than that of the control group. These findings suggested
that in the diabetic mice, the composition and proportions of tear
proteins, especially mucins, were already changed.
Furthermore, histopathologic examination of the
cornea and conjunctiva were performed. During the 60-day follow-up,
it was found that compared with the control mice, the diabetic mice
exhibited a thinner corneal epithelium with reduced cell layers and
the disturbance of basal cells (Fig.
2A). The thickness of the corneal epithelium in H&E-stained
sections (Fig. 2B) was
significantly smaller in the DM group compared with the control
group on days 30 and 60 (P<0.01, P<0.01), suggesting that DM
had injured the corneal epithelium. As shown in Fig. 2C and D, the diabetic mice had a
lower goblet cell density than the control mice on days 30 and days
60 after treatment, illustrating that DM had impaired the number
and function of conjunctival goblet cells.
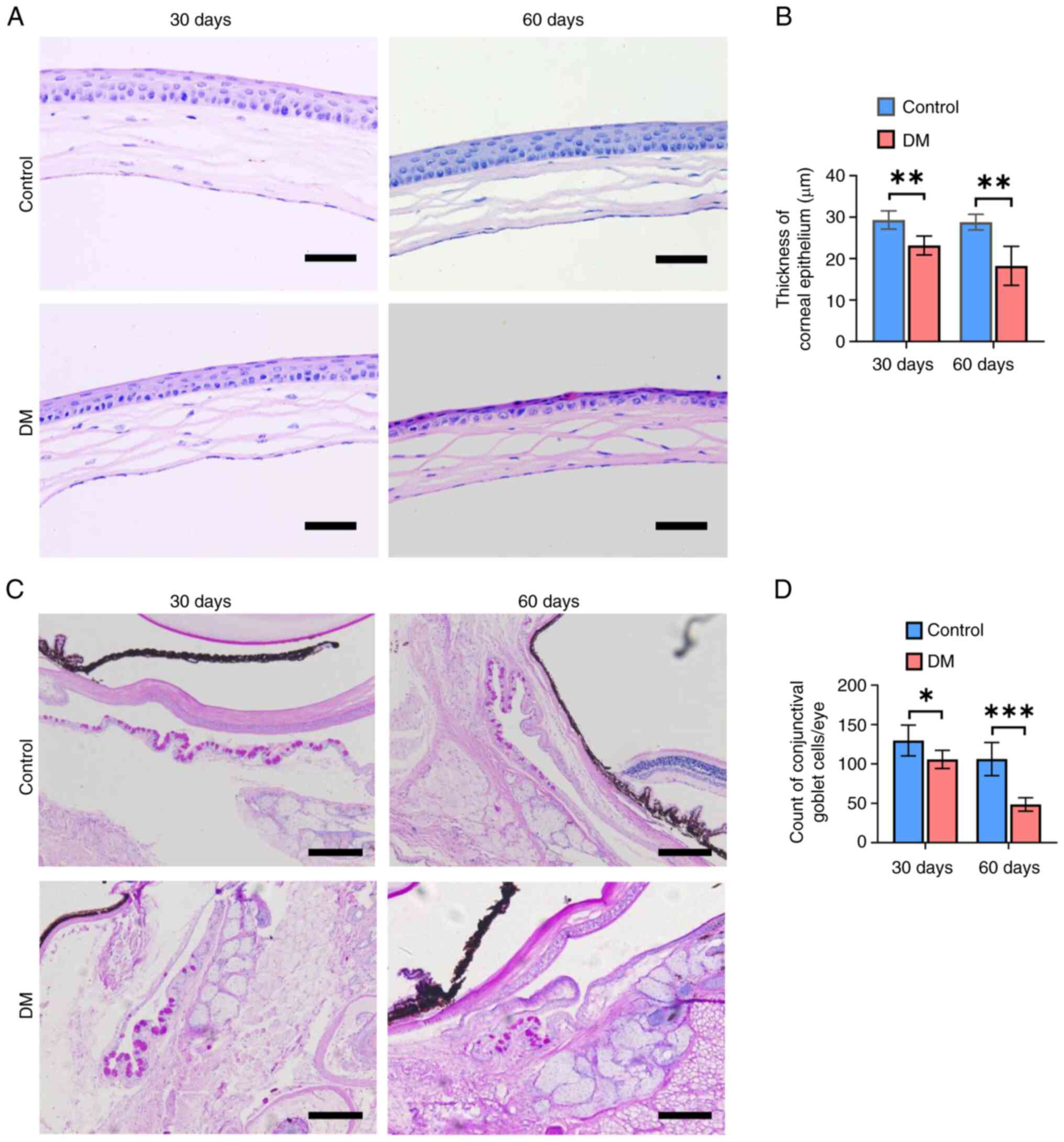 | Figure 2.Histopathology of the ocular surface
tissues. (A) Representative images of corneal sections following
hematoxylin and eosin staining. Scale bar, 50 µm. (B) Statistical
analysis of the thickness of the corneal epithelium, as measured in
images of (A). (C) Representative images of conjunctival sections
stained with PAS. Scale bar, 200 µm. (D) The goblet cell density in
PAS-stained sections. In (B) and (D), data are show as mean ±
standard deviation (n=5 animals per group). A two-tailed Student's
t-test was used to calculate the statistical significance. ns, not
significant, *P<0.05, **P<0.01, ***P<0.001. PAS, periodic
acid Schiff; DM, diabetes mellitus. |
After the aforementioned evaluation, a diabetic DE
mouse model was established. Taken together, the results indicated
that the diabetic mice exhibited decreased tear secretion,
increased corneal epithelial damage and poorer TF grades,
accompanied by corneal epithelium and conjunctiva injury, proving
that the diabetic DE mouse model had been created.
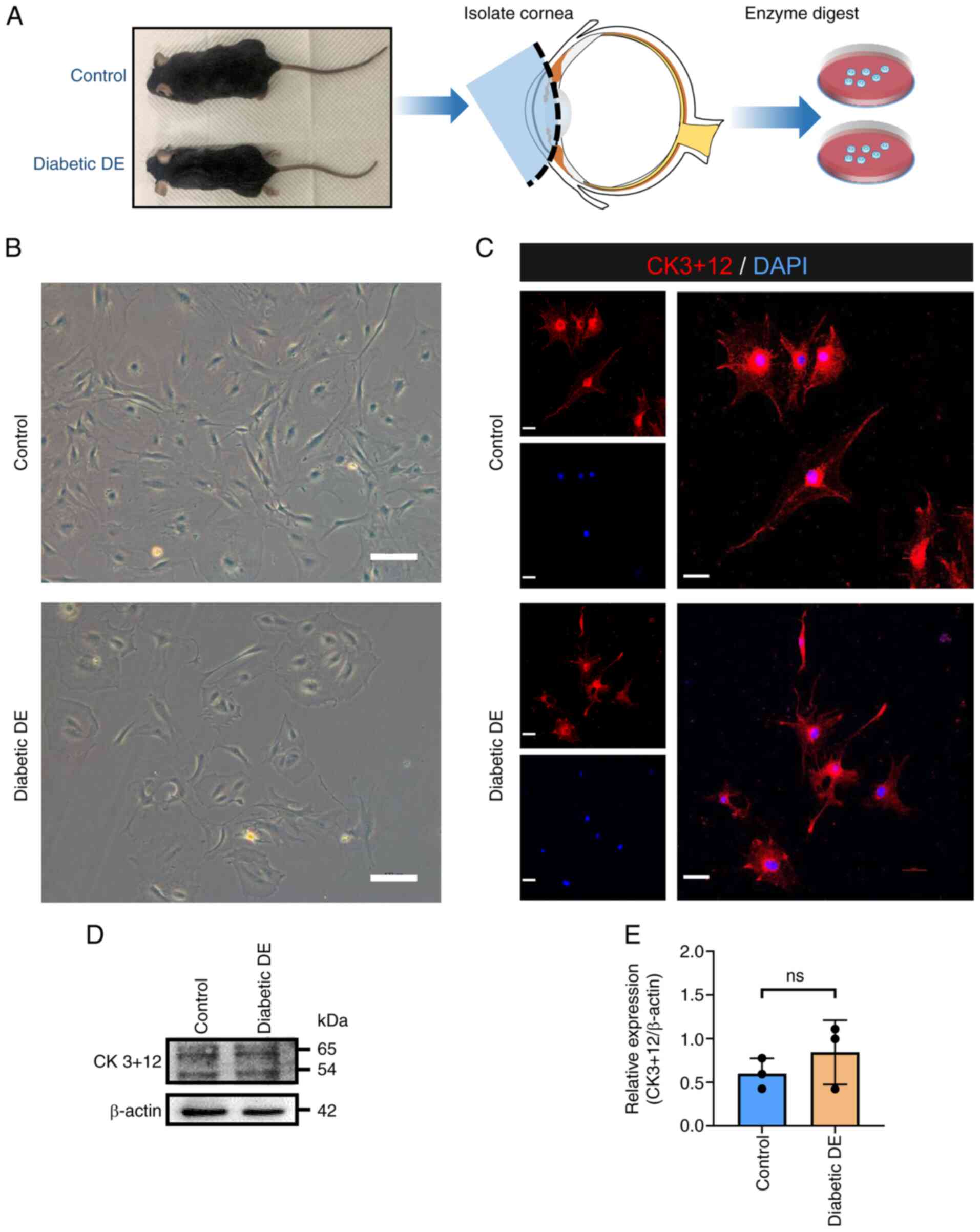
Isolation, culture and identification
of primary MCECs
To explore the mechanism of corneal epithelial
lesions in the diabetic DE mice in vitro, primary corneal
epithelial cells were cultured. The corneas of mice in the control
and diabetic DE groups were isolated and primary MCECs from the two
groups by cultured enzymatic digestion (Fig. 3A). The morphology of primary MCECs
in the two groups cultured on day 10 is shown in Fig. 3B. The MCECs grew by static
adherence and the cells in the two groups had similar shapes and
resembled spindles, ovals and polygons. The cells in the control
group grew rapidly, but those in the diabetic DE group grew slowly
and were sparse. This result indicated the poor proliferation of
cells in the diabetic DE group.
The cells were subjected to IF staining for CK3+12,
a marker of corneal epithelial cells. The CK3+12 protein
distributions in the two groups were similar, as illustrated in
Fig. 3C. The CK3+12 protein levels
in the cells from the two groups were did not significantly differ
(Fig. 3D and E). These results
identified the cultured cells as corneal epithelial cells.
Hyperglycemia impairs cell
proliferation, accompanied by decreased MUC1-ND and TRPV1
To further elucidate the functional changes in
primary MCECs, CCK8 assays were conducted, which demonstrated
attenuated cell proliferation in the diabetic DE group (Fig. 4A). The expression of MUC1-CT and
TRPV1 in primary MCECs of the control and diabetic DE groups was
detected and the protein localization was assessed by IF staining
(Fig. 4B). The difference in TRPV1
expression between cells in the control and diabetic DE groups was
visible, but a difference in MUC1-CT expression was not. The
colocalization of MUC1-CT and TRPV1 was also observed in the
membrane and cytoplasm. In addition, it was observed that TRPV1 and
MUC1-ND were significantly downregulated at the protein level in
cells of the diabetic DE group by western blotting and densitometry
analysis (Fig. 4C and D). However,
the MUC1-CT protein levels of the two groups of cells did not
significantly differ.
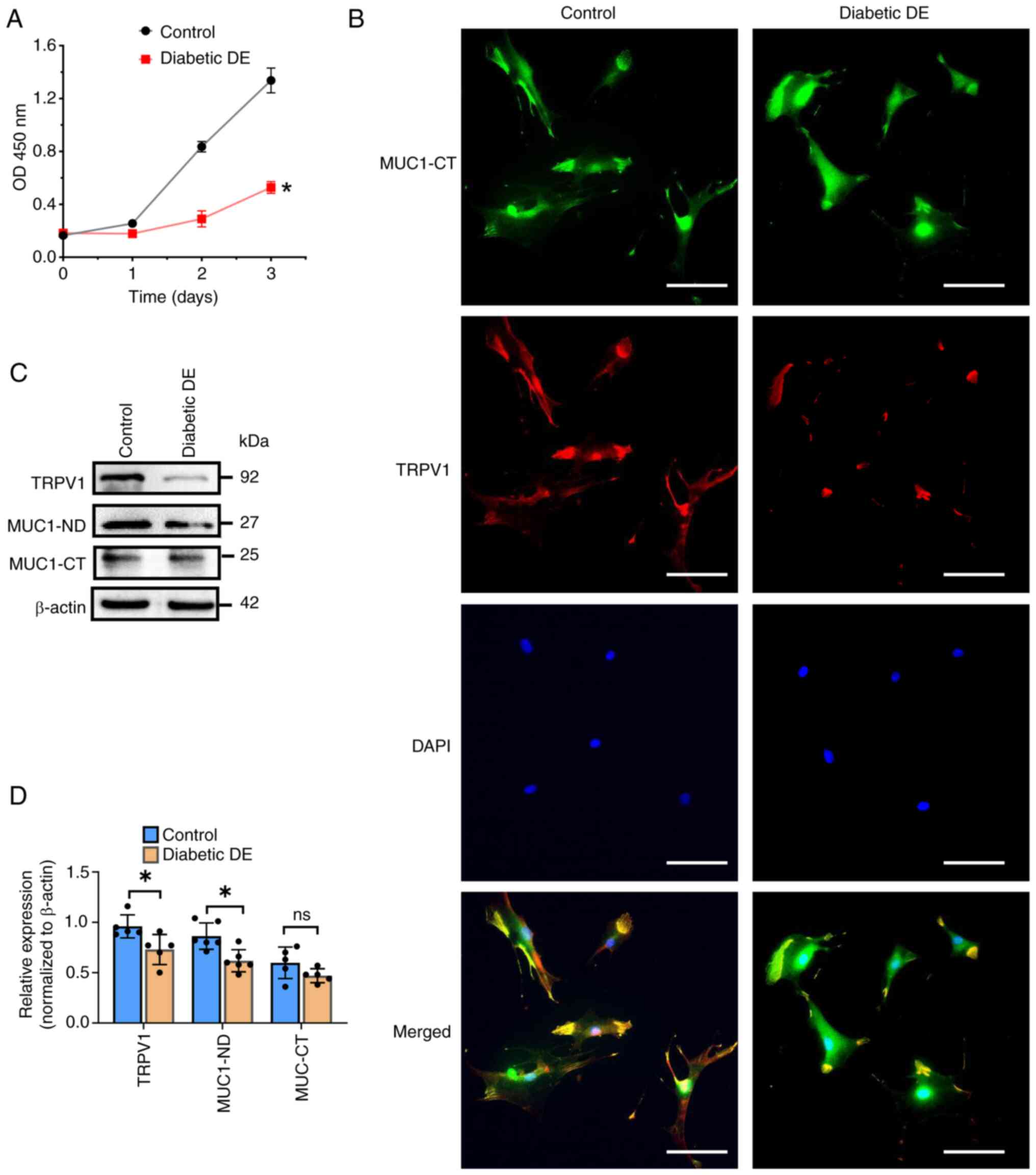 | Figure 4.Hyperglycemia impairs cell
proliferation, accompanied by decreased MUC1-ND and TRPV1 levels.
(A) CCK8 assays showed the cell proliferative ability of the two
groups. The OD value at 450 nm was recorded. (B) IF staining was
used to locate and detect the expression of MUC1-CT and TRPV1 in
primary MCECs from the control and diabetic DE groups. Scale bar,
100 µm. (C) Western blot analysis of TRPV1, MUC1-ND and MUC1-CT in
primary MCECs. β-Actin was used as a loading control. (D)
Densitometry analysis of the data in (C). In (A) and (D), data are
shown as mean ± standard deviation from three or more independent
experiments. ns, not significant, *P<0.05 vs. the corresponding
control, as analyzed by two-tailed Student's t test. MUC1-ND, MUC1
extracellular domain; TRPV1, transient receptor potential cation
channel subfamily V member 1; IF, immunofluorescence; MCECs, mouse
corneal epithelial cells; DE, dry eye; MUC1-CT, cytoplasmic tail of
MUC1; MUC1, mucin1. |
These results indicated that hyperglycemia impaired
cell proliferation in the diabetic DE mice, accompanied by reduced
MUC1-ND and TRPV1 levels and that TRPV1 and MUC1 may interact in
primary MCECs.
MUC1-ND binds TRPV1 to positively
regulate the proliferation of MCECs MUC1-ND interacts with
TRPV1
To further validate the interaction between MUC1-ND
and TRPV1, IF colocalization analysis of frozen corneal sections
from the two groups and Co-IP analysis of the two groups of cells
were conducted. The distribution and expression of TRPV1 and
MUC1-ND are shown in Fig. 5A. IF
staining confirmed that TRPV1 and MUC1-ND colocalized in the cell
membrane and cytoplasm of the corneal epithelium. The present study
also observed reduced TRPV1 and MUC1-ND expression in corneal
tissues from the diabetic DE mice in comparison with the control
mice. IF colocalization analysis of IF staining images were
conducted by ImageJ software to obtain Pearson's correlation
coefficients, which were used for statistical analysis. As
illustrated in Fig. 5B and C, the
Pearson r between MUC1-ND and TRPV1 in the diabetic DE group was
0.60±0.12, which was significantly lower than that in the control
group (0.82±0.13). This result indicated that the colocalization of
MUC1-ND and TRPV1 was reduced in the diabetic DE mice.
Additionally, Co-IP analysis of the two groups of cells was carried
out and the association of TRPV1 and MUC1-ND was observed (Fig. 5D). The present study found evidence
that MUC1-ND interacted with TRPV1 in the control cells. By
contrast, TRPV1 did not coimmunoprecipitate with MUC1-ND in the
diabetic DE cells. The present study also tried IP:TRPV1 to
immunoblot mucin1-CT and mucin1-ND to immunoblot TRPV1 (Fig. S1), it found that TRPV1 did not
interact with mucin1-CT in the control or diabetic DE cells, which
may be related to the binding site between the two proteins. These
results illustrated that the interaction between MUC1-ND and TRPV1
in the corneal epithelium was decreased in the diabetic DE mice,
which may be related to the downregulation of MUC1-ND and TRPV1 in
the diabetic DE mice.
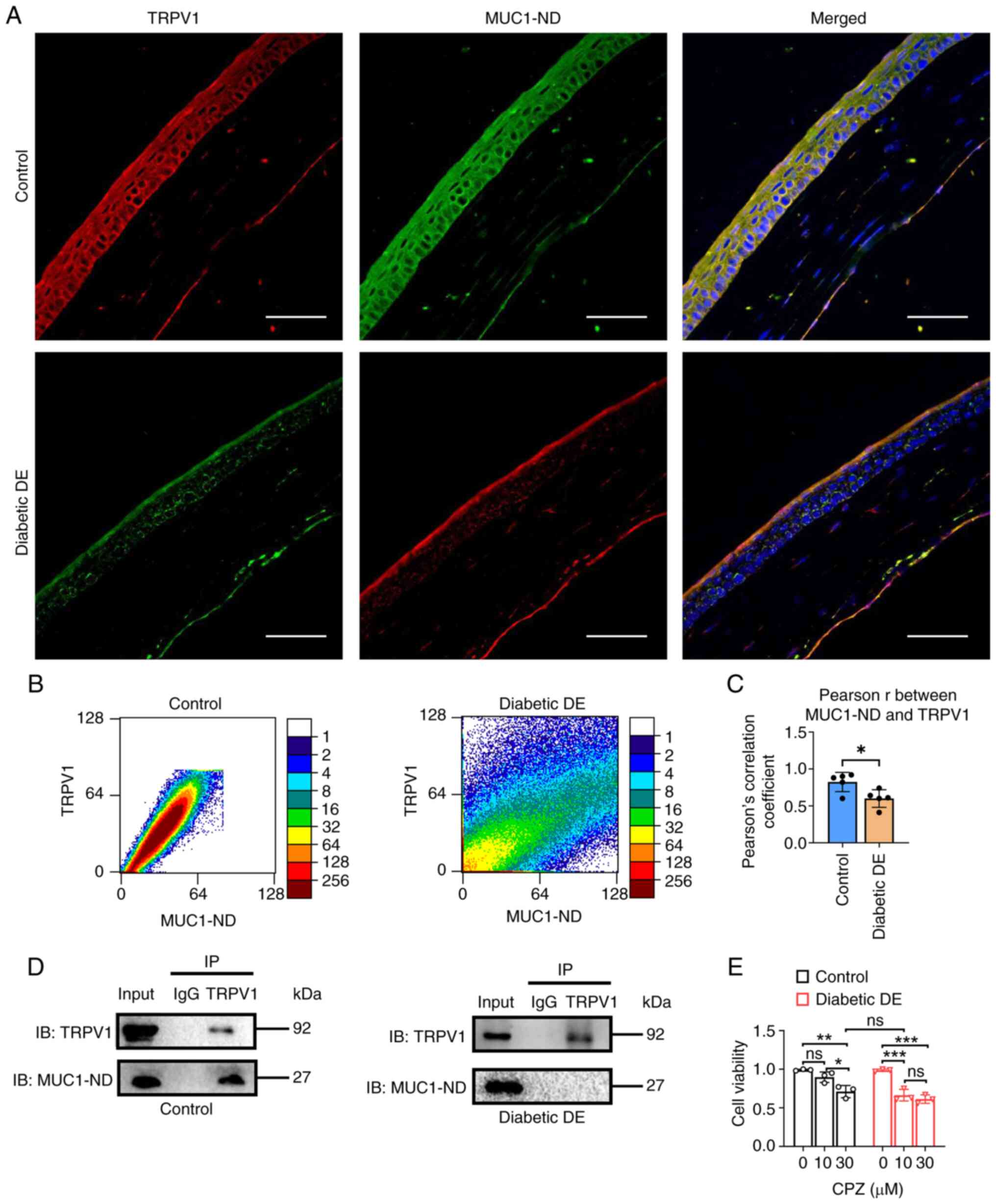 | Figure 5.TRPV1 interacts with MUC1-ND to
promote cell viability. (A) Frozen corneal sections were stained
for TRPV1 and MUC1-ND in the control and diabetic DE groups. Three
channels (red, blue and green, right panel) were used for IF
analysis. n=5 animals per group. Scale bar, 50 µm. (B) Scatter
diagram from IF colocalization analysis of the data in (A) by
ImageJ software. (C) Statistical analysis of the Pearson r between
TRPV1 and MUC1-ND in the corneal epithelium in the two groups. n=5
animals per group. (D) Coimmunoprecipitation and western blotting
showed that TRPV1 bound MUC1-ND in primary MCECs from the two
groups. (E) CCK8 assays detected the cell viability of the two
groups after treatment with CPZ (0, 10 and 30 µM), a TRPV1
antagonist. In (C) and (E), data are shown as mean ± standard
deviation from at least three independent experiments. ns: not
significant, *P<0.05, **P<0.01, ***P<0.001 vs. the
corresponding control, as analyzed by either two-tailed Student's t
test for two groups or by one-way ANOVA with LSD's
multiple-comparison test for more than two groups. TRPV1, transient
receptor potential cation channel subfamily V member 1; MUC1-ND,
MUC1 extracellular domain; MUC1, mucin1; DE, dry eye; IF,
immunofluorescence; MCECs, mouse corneal epithelial cells; CPZ,
capsazepine. |
TRPV1 positively regulates cell
proliferation
To verify the regulatory role of TRPV1 in cell
proliferation, primary MCECs of the control and diabetic DE groups
were treated with CPZ (0, 10 and 30 µM), a TRPV1 antagonist. Cell
viability was detected by CCK8 assays, as shown in Fig. 5E. The viability in control group
cells treated with CPZ at 30 µM was significantly lower than that
of cells treated without CPZ treatment (P<0.01) and cells
treated with CPZ at 10 µM (P<0.05). However, there was no
significant difference in cell viability between the untreated
cells and cells treated with 10 µM CPZ. In the diabetic DE group,
the lowest cell viability was found in cells treated with CPZ at 30
µM, followed by those treated with CPZ at 10 µM and those without
CPZ treatment. The viability of cells treated with 10 µM CPZ and 30
µM CPZ was significantly lower than that of cells without CPZ
treatment (P<0.001, P<0.001). However, the cell viability of
cells treated with 10 µM CPZ was similar to that of cell treated
with 30 µM CPZ. Collectively, these results demonstrated that CPZ,
a TRPV1 antagonist, restricted cell proliferation as its
concentration increased.
These data suggested that MUC1-ND interacts with
TRPV1, which can positively regulate cell proliferation.
TRPV1 promotes cell proliferation by
partly activating the AKT signaling pathway
To clarify the mechanism by which the binding of
MUC1-ND and TRTV1 regulates cell proliferation, western blotting
and CCK8 assays were conducted. Primary MCECs in the control and
diabetic DE groups were treated with CPZ (0, 10 and 30 µM) for 24 h
and then harvested for western blotting. CPZ was formulated with
DMSO as a solvent, so CPZ 0 µM meant cells treated with DMSO in the
control or diabetic DE groups. As shown in Fig. 6A, there was decreased expression of
TRPV1 in control cells treated with CPZ at 30 µM and diabetic DE
cells treated with CPZ at 0, 10 and 30 µM, indicating that TRPV1
expression was inhibited by CPZ. As shown in Fig. 6A and B, similar levels of AKT1/2/3
in the control groups were observed but in diabetic DE groups, it
was significantly increased in 30 µM CPZ group compared with 0 µM
CPZ group. The level in the diabetic DE + 10 µM CPZ group was
similar to the other two diabetic DE groups. The expression of
p-AKT(Ser473) decreased significantly with CPZ treatment in a
dose-dependent manner in the control groups and the lowest level
was observed in the diabetic DE + 30 µM CPZ group (Fig. 6A and C). The expression in diabetic
DE + 10 µM CPZ group was not significantly different from the other
two diabetic DE groups. The expression of Bcl2 decreased with CPZ
treatment in a dose-dependent manner in the control and diabetic DE
groups. The protein level of caspase3 increased in diabetic DE
groups compared with the control groups with different CPZ
treatment. It was more abundant in the control + 30 µM CPZ group
than in control groups treated with 0 and 10 µM CPZ. Similar levels
was found in the three diabetic DE groups. These results suggested
that CPZ, a TRPV1 antagonist, attenuated the AKT signaling
pathway.
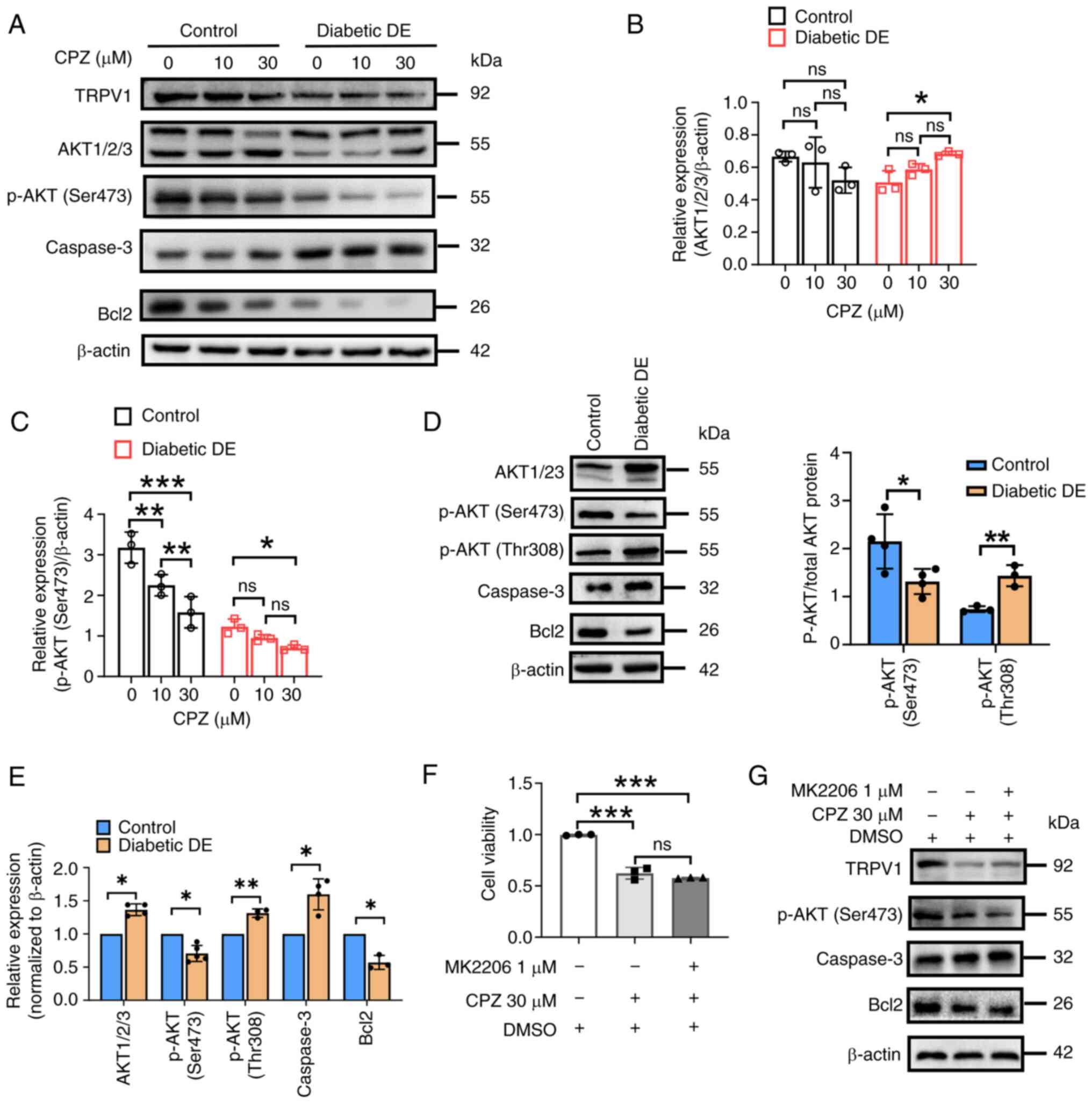 | Figure 6.TRPV1 positively regulates cell
proliferation by partly activating the AKT pathway. (A) Western
blotting showing the protein expression of TRPV1, AKT1/2/3, p-AKT
(Ser473), p-AKT (Thr308), caspase3 and Bcl2 in the two groups of
cells upon treatment with the TRPV1 antagonist CPZ (0, 10 and 30
µM) for 24 h. (B and C) Densitometry analysis of AKT1/2/3, p-AKT
(Ser473) in (A). (D) Western blotting and (E) densitometry analysis
of the protein expression of AKT1/2/3, p-AKT (Ser473), p-AKT
(Thr308), caspase3 and Bcl2 in primary MCECs of the control and
diabetic DE groups. (F) CCK8 assays showed the cell viability in
the control group without CPZ treatment and upon treatment with CPZ
at 30 µM or with CPZ at 30 µM + MK2206 (an AKT inhibitor) at 1 µM
for 24 h. Western blotting showing the protein expression of TRPV1,
p-AKT (Ser473), caspase3 and Bcl2 in primary MCECs of the diabetic
DE group treated as described in (G). In (B, C and F), quantified
data are shown as mean ± standard deviation from three or more
independent experiments. ns, not significant, *P<0.05,
**P<0.01, ***P<0.001 vs. the corresponding control, by either
two-tailed Student's t-test for two groups or by one-way ANOVA
followed by LSD multiple-comparison test for more than two groups.
TRPV1, transient receptor potential cation channel subfamily V
member 1; p-, phosphorylated; CPZ, capsazepine; DE, dry eye; STZ,
streptozotocin. |
In addition, western blotting was performed to
detect the protein expression of AKT1/2/3, p-AKT (Ser473), p-AKT
(Thr308), caspase3 and Bcl2. As illustrated in Fig. 6D and E, the protein levels of p-AKT
(Ser473) and Bcl2 were significantly reduced in the diabetic DE
group, but the protein levels of AKT1/2/3, p-AKT (Thr308) and
caspase3 was significantly increased in the diabetic DE group when
compared with the control group. The results indicated that
activation of the AKT signaling pathway was attenuated in the
diabetic DE mice.
Furthermore, cells in the control group were treated
with DMSO, 30 µM CPZ or treated with 30 µM CPZ + 1 µM MK2206 (an
AKT inhibitor) for 24 h. Then, CCK8 assays were conducted to detect
cell viability and western blotting. As shown in Fig. 6F, the cell viability of the 30 µM
CPZ-treated group was significantly lower than that of the
untreated group but was not significantly different from that of
the CPZ + MK2206-treated group. The cell viability of the CPZ +
MK2206-treated group was significantly lower than that of the DMSO
group (P<0.001). These results demonstrated that AKT signaling
pathway inhibition was partly responsible for the effect of CPZ (a
TRPV1 antagonist) on cell proliferation. As shown in Fig. 6G, the protein expressions of TRPV1,
p-AKT (Ser473) and Bcl2 were reduced in other two groups compared
with that of the DMSO group. The caspase3 protein level was
increased in the 30 µM CPZ-treated group and the CPZ +
MK2206-treated group.
The aforementioned results suggested that TRPV1
positively regulated cell proliferation by partly activating the
AKT signaling pathway.
Discussion
The present study investigated the biological role
of the MUC1-ND/TRPV1/AKT axis in the moderation of corneal
epithelial cells in diabetic mice with DE disease. First, diabetic
DE model mice were established with significantly increased corneal
epithelial defects, reduced tear production, poor TF pattern grades
and an impaired ocular surface structure. Second, the expression
levels of MUC1-ND and TRPV1 were found to be significantly reduced
in the corneal epithelium of diabetic DE mice. Third, TRPV1 was
shown to positively regulate the proliferation of MCECs. Notably,
MUC1-ND was shown to interact with the TRPV1 protein in the control
group but not in the diabetic DE group. Furthermore, it was found
that TRPV1 positively regulated cell proliferation by partly
activating the AKT signaling pathway. In general, the present study
revealed that the interaction between MUC1-ND and TRPV1 partly
activated the AKT signaling pathway to promote MCEC
proliferation.
DE disease is a multifactorial disorder and its most
distinguished feature is the loss of homeostasis of the tear film
(2). The establishment of DE
animal models is necessary to study the fundamental mechanisms of
DE disease, which is nearly identical in animals and in humans
(34). Previous studies have
confirmed that DE animal models, including chemical- or
surgery-induced DE models, environmental factor-induced DE models,
genetically engineered DE models and models prepared with combined
methods, are reliable and effective in imitating ocular surface
microenvironments (34,35). Common parameters used to evaluate
DE disease in mouse models are CFS to assess surface structure, the
TF test to assess tear proteins, the phenol red thread test to
assess tear secretion and histopathological examination (34,35).
In the present study, the diabetic mice were followed for 60 days
and it was confirmed that they exhibited significantly increased
corneal epithelial defects, decreased tear production, poor TF
pattern grades, impaired corneal epithelial structure and a lower
goblet cell density on day 30 following the final STZ injection.
The method used to establish the diabetic DE model was similar to
that used in another previous study (36). The diabetic DE model mice were
identified to be reliable tools in the study of pathophysiological
mechanisms and the underlying molecular cascade in diabetic DE
disease. Notably, it was also observed that TF was lessened and
even disappeared in diabetic mice with DE, indicating that the tear
proteins differed in diabetic mice with DE disease. Then, primary
MCECs from mice in the control and diabetic DE groups were cultured
and identified, consistent with a previous study (37).
MUC1 is a key member of the membrane-associated
mucin (MAM) family with an important barrier function (15). MUC1 accumulates with other MAMs on
the corneal epithelia and it is cross-linked by galectin-3 to form
an interlocking lattice for pathogen exclusion (14). Damage to the ocular surface
epithelium and barrier disruption causes ocular surface diseases,
including DE disease. One clinical study reported that MUC1
expression levels were decreased in the conjunctival epithelium of
patients with DE disease (38).
MUC1 is also confirmed to be cleaved at its extracellular domain
(MUC1-ND) (19,20). Part of the soluble extracellular
subunit of MUC1 can be detected in human tears (39). In the present study, MUC1-CT
expression was found decreased in diabetic DE mice, but the
difference was not significant compared with that of the control
mice, whereas MUC1-ND levels were significantly decreased in the
corneal epithelial cells of diabetic DE mice compared with control
mice. This suggested that MUC1 was cleaved and that its soluble
extracellular large subunit (MUC1-ND) was shed from the corneal
epithelial cell surface in diabetic mice with DE disease. The
present study revealed that MUC1 abnormalities may be closely
linked with corneal epithelial homeostasis in diabetic DE
disease.
Growing evidence shows the downregulation of TRPV1
in colorectal, endometrial, renal and skin cancers but the
upregulation of TRPV1 in the U373 glioblastoma line, high-grade
astrocytes and the RT4 renal cell carcinoma line, indicating that
TRPV1 mediates the balance between cell proliferation and apoptosis
(40). TRPV1 activation acts as a
double-edged sword in that it induces both improvements in cell
proliferation and migration in corneal injury (41–43)
and the release of proinflammatory cytokines in human corneal
epithelial cells stimulated by hyperosmolarity (25,44,45).
Previous studies (30,31,46)
have also shown that MUC1 regulates inflammation via interactions
with several different signaling pathways and correlates with the
stabilization of ion channels, which are essential to corneal
epithelial homeostasis (15). MUC1
forms a lattice with TRPV5 via galectin-3 to increase renal TRPV5
activity to protect against nephrolithiasis (30). The present study first confirmed
the downregulation of TRPV1 in the corneal epithelia of diabetic DE
mice compared with control mice. It next analyzed the relationship
between TRPV1 levels and corneal cell proliferation. It was found
that CPZ, a TRPV1 antagonist, restricted proliferation in primary
MCECs, indicating that TRPV1 played a key role in promoting MCEC
proliferation in diabetic DE disease. The present study further
investigated the interaction between MUC1 and TRPV1 and the results
demonstrated that MUC1-ND interacted with the TRPV1 protein in the
control group but not in the diabetic DE group. These results
demonstrated that the interaction between MUC1-ND and TRPV1
positively promoted corneal epithelial cell proliferation. This
interaction was attenuated in the diabetic mice and may contribute
to corneal epithelial dysfunction in DE disease.
The Ser and Thr kinase AKT, also known as protein
kinase B (PKB), has become the focus in biological and medicinal
studies, together with its function and regulation in diabetes
(47). AKT activation occurs
downstream of inositol phosphate 3-kinase (PI3K), a lipid kinase
associated with cell transformation and the insulin response. The
activation of PI3K leads to the phosphorylation of two key residues
on AKT1, catalyzing the activation of Thr308 in the T loop and
Ser473 in the C-terminal hydrophobic motif. AKT activation is
greatly decreased when Ser473 is unphosphorylated, which stabilizes
Thr308 phosphorylation and AKT activation. Attenuated activation of
the AKT signaling pathway inhibits corneal epithelial wound healing
in a model of corneal epithelial trauma in diabetic mice (48). Some studies have found that TRPV1
channel activation can result in the phosphorylation of AKT for
signal transduction (29,49). The present study found that the AKT
signaling pathway was attenuated in diabetic mice with DE disease,
accompanied by the decreased phosphorylation of AKT (Ser473).
Additionally, it was found that CPZ, a TRPV1 antagonist, reduced
the activation of the AKT signaling pathway. However, this
inhibition was partly responsible for the effect of CPZ (a TRPV1
antagonist) on cell proliferation. Collectively, these findings
revealed that TRPV1 positively regulated cell proliferation by
partly activating the AKT signaling pathway. Notably, the present
study revealed the interaction between MUC1-ND and TRPV1 and its
regulation in the corneal epithelial cells of diabetic DE mice. In
the present study it was found that MUC1-ND acts as a protein
partner that interacts with TRPV1, which positively regulates MCEC
proliferation by partly activating the AKT signaling pathway. The
decrease in this interaction in the diabetic mice may have induced
corneal epithelial dysfunction in the progression of diabetic DE
disease.
There was limitation to the present study. MUC1-ND
in the tears of diabetic mice failed to be detected via western
blotting, due to very little tear secretion from the mice. It may
be an improvement to collect the tear fluid of diabetic patients
with dry eye disease to detect the mucin1-ND level. Relevant
clinical trials are required to assess the role of MUC1-ND and
TRPV1 in diabetic DE disease.
In summary, the present study illustrated that the
interaction of MUC1-ND with TRPV1 promoted MCEC proliferation by
partly activating the AKT signaling pathway. The findings provided
new insight into the pathogenesis of corneal epithelial dysfunction
in diabetic DE disease.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by Natural Science Foundation of
Guangdong Province, China (project no. 2022A1515012346) and the
Science and Technology Program of Guangzhou, China (project no.
202002020046).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HQL, XHL, YZ and YYZ contributed to the study
design. HQL, YZ, YTC, RZ, XHL, YYZ, WKZ, HC, JH and SFF conducted
experiments. HQL and YZ performed statistical analysis and drafted
the manuscript. HQL and YZ confirm the authenticity of all the raw
data. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All experimental procedures were approved by the
Institutional Animal Care and Use Committee of Southern Medical
University (Guangzhou, China; approval no. LAEC-2021-032) and
conducted in compliance with the ARVO statement for the Use of
Animals in Ophthalmic and Vision Research (32).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ogurtsova K, Guariguata L, Barengo NC,
Ruiz PL, Sacre JW, Karuranga S, Sun H, Boyko EJ and Magliano DJ:
IDF diabetes Atlas: Global estimates of undiagnosed diabetes in
adults for 2021. Diabetes Res Clin Pract. 183:1091182022.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Craig JP, Nichols KK, Akpek EK, Caffery B,
Dua HS, Joo CK, Liu Z, Nelson JD, Nichols JJ, Tsubota K and
Stapleton F: TFOS DEWS II definition and classification report.
Ocul Surf. 15:276–283. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Stapleton F, Alves M, Bunya VY, Jalbert I,
Lekhanont K, Malet F, Na KS, Schaumberg D, Uchino M, Vehof J, et
al: TFOS DEWS II epidemiology report. Ocul Surf. 15:334–365. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Manaviat MR, Rashidi M, Afkhami-Ardekani M
and Shoja MR: Prevalence of dry eye syndrome and diabetic
retinopathy in type 2 diabetic patients. BMC Ophthalmol. 8:102008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang X, Zhao L, Deng S, Sun X and Wang N:
Dry eye syndrome in patients with diabetes mellitus: Prevalence,
etiology, and clinical characteristics. J Ophthalmol.
2016:82010532016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Belmonte C, Nichols JJ, Cox SM, Brock JA,
Begley CG, Bereiter DA, Dartt DA, Galor A, Hamrah P, Ivanusic JJ,
et al: TFOS DEWS II pain and sensation report. Ocul Surf.
15:404–437. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shih KC, Lam KS and Tong L: A systematic
review on the impact of diabetes mellitus on the ocular surface.
Nutr Diabetes. 7:e2512017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Han SB, Yang HK and Hyon JY: Influence of
diabetes mellitus on anterior segment of the eye. Clin Interv
Aging. 14:53–63. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kaiserman I, Kaiserman N, Nakar S and
Vinker S: Dry eye in diabetic patients. Am J Ophthalmol.
139:498–503. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhu L, Titone R and Robertson DM: The
impact of hyperglycemia on the corneal epithelium: Molecular
mechanisms and insight. Ocul Surf. 17:644–654. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Georgiev GA, Eftimov P and Yokoi N:
Contribution of mucins towards the physical properties of the tear
film: A modern update. Int J Mol Sci. 20:61322019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ablamowicz AF and Nichols JJ: Ocular
surface membrane-associated mucins. Ocul Surf. 14:331–341. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shirai K and Saika S: Ocular surface
mucins and local inflammation-studies in genetically modified mouse
lines. BMC Ophthalmol. 15 (Suppl 1):S1542015. View Article : Google Scholar
|
|
14
|
Martinez-Carrasco R, Argüeso P and Fini
ME: Membrane-associated mucins of the human ocular surface in
health and disease. Ocul Surf. 21:313–330. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fini ME, Jeong S, Gong H,
Martinez-Carrasco R, Laver NMV, Hijikata M, Keicho N and Argüeso P:
Membrane-associated mucins of the ocular surface: New genes, new
protein functions and new biological roles in human and mouse. Prog
Retin Eye Res. 75:1007772020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Schroeder JA, Thompson MC, Gardner MM and
Gendler SJ: Transgenic MUC1 interacts with epidermal growth factor
receptor and correlates with mitogen-activated protein kinase
activation in the mouse mammary gland. J Biol Chem.
276:13057–13064. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li Y, Ren J, Yu W, Li Q, Kuwahara H, Yin
L, Carraway KL III and Kufe D: The epidermal growth factor receptor
regulates interaction of the human DF3/MUC1 carcinoma antigen with
c-Src and beta-catenin. J Biol Chem. 276:35239–35242. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Morimoto Y, Yamashita N, Daimon T, Hirose
H, Yamano S, Haratake N, Ishikawa S, Bhattacharya A, Fushimi A,
Ahmad R, et al: MUC1-C is a master regulator of MICA/B NKG2D ligand
and exosome secretion in human cancer cells. J Immunother Cancer.
11:e0062382023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Thathiah A, Blobel CP and Carson DD: Tumor
Necrosis Factor-alpha Converting Enzyme/ADAM 17 Mediates MUC1
Shedding. J Biol Chem. 278:3386–3394. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Thathiah A and Carson DD: MT1-MMP mediates
MUC1 shedding independent of TACE/ADAM17. Biochem J. 382((Pt 1)):
363–373. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Miyazaki K, Kishimoto H, Kobayashi H,
Suzuki A, Higuchi K, Shirasaka Y and Inoue K: The glycosylated
N-terminal domain of MUC1 is involved in chemoresistance by
modulating drug permeation across the plasma membrane. Mol
Pharmacol. 103:166–175. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Liu R, Chen L, Zhao X, Bao L, Wei R and Wu
X: MUC1 promotes RIF by regulating macrophage ROS-SHP2 signaling
pathway to up-regulate inflammatory response and inhibit
angiogenesis. Aging (Albany NY). 16:3790–3802. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu L, Zhou L, Wang L, Mao Z, Zheng P,
Zhang F, Zhang H and Liu H: MUC1 attenuates neutrophilic airway
inflammation in asthma by reducing NLRP3 inflammasome-mediated
pyroptosis through the inhibition of the TLR4/MyD88/NF-κB pathway.
Respir Res. 24:2552023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Comes N, Gasull X and Callejo G: Proton
sensing on the ocular surface: Implications in eye pain. Front
Pharmacol. 12:7738712021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang Y, Yang H, Wang Z, Okada Y, Saika S
and Reinach PS: Wakayama symposium: Dependence of corneal
epithelial homeostasis on transient receptor potential function.
Ocul Surf. 11:8–11. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang XL, Wang X, Shao L, Jiang GT, Min JW,
Mei XY, He XH, Liu WH, Huang WX and Peng BW: TRPV1 mediates
astrocyte activation and interleukin-1β release induced by hypoxic
ischemia (HI). J Neuroinflammation. 16:1142019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang X, Yang XL, Kong WL, Zeng ML, Shao L,
Jiang GT, Cheng JJ, Kong S, He XH, Liu WH, et al: TRPV1
translocated to astrocytic membrane to promote migration and
inflammatory infiltration thus promotes epilepsy after hypoxic
ischemia in immature brain. J Neuroinflammation. 16:2142019.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lu MJ, Chen YS, Huang HS and Ma MC:
Hypoxic preconditioning protects rat hearts against
ischemia-reperfusion injury via the
arachidonate12-lipoxygenase/transient receptor potential vanilloid
1 pathway. Basic Res Cardiol. 109:4142014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang H, Wang Z, Capó-Aponte JE, Zhang F,
Pan Z and Reinach PS: Epidermal growth factor receptor
transactivation by the cannabinoid receptor (CB1) and transient
receptor potential vanilloid 1 (TRPV1) induces differential
responses in corneal epithelial cells. Exp Eye Res. 91:462–471.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nie M, Bal MS, Yang Z, Liu J, Rivera C,
Wenzel A, Beck BB, Sakhaee K, Marciano DK and Wolf MT: Mucin-1
Increases Renal TRPV5 activity in vitro, and urinary level
associates with calcium nephrolithiasis in patients. J Am Soc
Nephrol. 27:3447–3458. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Al-Bataineh MM, Kinlough CL, Marciszyn A,
Lam T, Ye L, Kidd K, Maggiore JC, Poland PA, Kmoch S, Bleyer A, et
al: Influence of glycoprotein MUC1 on trafficking of the
Ca2+-selective ion channels, TRPV5 and TRPV6, and on in vivo
calcium homeostasis. J Biol Chem. 299:1029252023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
The Association for Research in Vision and
Ophthalmology:ARVO Statement for the Use of Animals in Ophthalmic
and Vision Research. The Association for Research in Vision and
Ophthalmology; Rockville, MD: 2021
|
|
33
|
Masmali AM, Murphy PJ and Purslow C:
Development of a new grading scale for tear ferning. Cont Lens
Anterior Eye. 37:178–184. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rahman MM, Kim DH, Park CK and Kim YH:
Experimental models, induction protocols, and measured parameters
in dry eye disease: Focusing on practical implications for
experimental research. Int J Mol Sci. 22:121022021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhu J, Inomata T, Shih KC, Okumura Y,
Fujio K, Huang T, Nagino K, Akasaki Y, Fujimoto K, Yanagawa A, et
al: Application of animal models in interpreting dry eye disease.
Front Med (Lausanne). 9:8305922022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Qu M, Wan L, Dong M, Wang Y, Xie L and
Zhou Q: Hyperglycemia-induced severe mitochondrial bioenergetic
deficit of lacrimal gland contributes to the early onset of dry eye
in diabetic mice. Free Radic Biol Med. 166:313–323. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bai J, Fu H, Bazinet L, Birsner AE and
D'Amato RJ: A method for developing novel 3D Cornea-on-a-Chip using
primary murine corneal epithelial and endothelial cells. Front
Pharmacol. 11:4532020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Corrales RM, Narayanan S, Fernández I,
Mayo A, Galarreta DJ, Fuentes-Páez G, Chaves FJ, Herreras JM and
Calonge M: Ocular mucin gene expression levels as biomarkers for
the diagnosis of dry eye syndrome. Invest Ophthalmol Vis Sci.
52:8363–8369. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Spurr-Michaud S, Argüeso P and Gipson I:
Assay of mucins in human tear fluid. Exp Eye Res. 84:939–950. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhai K, Liskova A, Kubatka P and
Büsselberg D: Calcium Entry through TRPV1: A potential target for
the regulation of proliferation and apoptosis in cancerous and
healthy cells. Int J Mol Sci. 21:41772020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sumioka T, Okada Y, Reinach PS, Shirai K,
Miyajima M, Yamanaka O and Saika S: Impairment of corneal
epithelial wound healing in a TRPV1-deficient mouse. Invest
Ophthalmol Vis Sci. 55:3295–3302. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Okada Y, Reinach PS, Shirai K, Kitano A,
Kao WW, Flanders KC, Miyajima M, Liu H, Zhang J and Saika S: TRPV1
involvement in inflammatory tissue fibrosis in mice. Am J Pathol.
178:2654–2664. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Nidegawa-Saitoh Y, Sumioka T, Okada Y,
Reinach PS, Flanders KC, Liu CY, Yamanaka O, Kao WW and Saika S:
Impaired healing of cornea incision injury in a TRPV1-deficient
mouse. Cell Tissue Res. 374:329–338. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Pan Z, Wang Z, Yang H, Zhang F and Reinach
PS: TRPV1 activation is required for hypertonicity-stimulated
inflammatory cytokine release in human corneal epithelial cells.
Invest Ophthalmol Vis Sci. 52:485–493. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Reinach PS, Mergler S, Okada Y and Saika
S: Ocular transient receptor potential channel function in health
and disease. BMC Ophthalmol. 15 (Suppl 1):S1532015. View Article : Google Scholar
|
|
46
|
Jeon BH, Yoo YM, Jung EM and Jeung EB:
Dexamethasone treatment increases the intracellular calcium level
through TRPV6 in A549 cells. Int J Mol Sci. 21:10502020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Manning BD and Toker A: AKT/PKB Signaling:
Navigating the network. Cell. 169:381–405. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Li Y, Li J, Zhao C, Yang L, Qi X, Wang X,
Zhou Q and Shi W: Hyperglycemia-reduced NAD+ biosynthesis impairs
corneal epithelial wound healing in diabetic mice. Metabolism.
114:1544022021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Pham TH, Jin SW, Lee GH, Park JS, Kim JY,
Thai TN, Han EH and Jeong HG: Sesamin induces endothelial nitric
oxide synthase activation via transient receptor potential
vanilloid type 1. J Agric Food Chem. 68:3474–3484. 2020. View Article : Google Scholar : PubMed/NCBI
|