Introduction
Carcinoma of the uterine cervix is one of the most
common malignancies among women worldwide. Human papillomaviruses
(HPVs) have been identified as the major etiological factor in
cervical carcinogenesis (1).
However, HPV infection alone is insufficient to trigger the
tumorigenic cascade in human cervical cancer cells. This indicates
that multiple steps, as well as multiple factors, may be necessary
for the development of cervical cancer. Leptin is a metabolic
hormone that is required for the maintenance of normal energy
balance and body weight (2,3). It is a multifunctional peptide hormone
with wide ranging biological activities including appetite
regulation, bone formation, reproductive function and angiogenesis
(4–6). These biological activities indicate
that the leptin system may be involved in the development and
progression of neoplasms (7).
Leptin exerts its actions through its specific receptors present in
a variety of tissues localized to the cell membrane (8). The leptin receptor belongs to a family
of class I cytokine receptors, which typically contain a cytokine
receptor homologous domain in the extracellular region (9). Stimulation of the leptin receptor has
been reported to activate multiple signaling pathways such as
JAK/STAT, ERK and PI3K/AKT (10,11).
Here, we hypothesized that leptin expression may correlate with the
progression of cervical tumors. Our previous investigation showed
that leptin significantly increased the growth of HeLa cervical
cancer cells in a dose-dependent and time-dependent manner, and
leptin significantly upregulated c-myc mRNA and protein expression
in HeLa cells (12). In the present
study, we analyzed the relationship between leptin expression and
the clinical pathology of cervical cancer and examined the
relationships between leptin and c-myc and bcl-2. Furthermore, we
treated HeLa cells with recombinant leptin or silencing of leptin
and explored the effect on the cellular proliferation and apoptosis
and analyzed the mechanisms of leptin involved in cervical
carcinoma progression in vitro.
Materials and methods
Patients and specimens
Tissue samples were obtained from 80 women who
underwent partial or radical hysterectomy due to cervical lesions
during the period 2008–2010. The age of the patients ranged from 28
to 72 years (mean 52.8 years). All samples were pathologically
confirmed cervical intraepithelial neoplasia (CIN) or squamous cell
carcinoma (SCC). No patients had received treatment before tumor
excision. Tumor samples had previously been fixed in 10% buffered
formaldehyde solution and embedded in paraffin blocks for routine
histological examination.
Immunohistochemistry
The immunohistochemical analysis of leptin, Ki-67,
c-myc and bcl-2 was carried out in representative tissue sections
using antibodies for leptin (rabbit polyclonal Ab, dilution 1:100);
for bcl-2 (mouse monoclonal antibody, dilution 1:100) (both from
Boster, China); for c-myc (goat polyclonal Ab, dilution 1:200)
(Santa Cruz Biotechnology, Inc, Santa Cruz, CA, USA). Other
antibodies were working solutions. Final stainings were performed
with DAB staining system (Maxin, China). Slides were counterstained
with hematoxylin. In the negative controls the primary antibody was
omitted. The expression of leptin, bcl-2, Ki-67 and c-myc was
analyzed in 10 different tumor fields, and the mean percentage of
tumor cells with positive staining was evaluated. The expression of
the studied proteins in the cancer samples was classified using a
four-point scale: 0, <10% positive cells; 1+, 10–50% positive
cells with weak staining; 2+, >50% positive cells with weak
staining; 3+, >50% positive cells with strong staining.
Cell culture and colorimetric MTT
assay
The HeLa cell line was grown in Dulbecco’s modified
Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum
(FBS). For treatment, cells were seeded at a density of
1×106/100-mm tissue culture dish. Cells were
serum-deprived overnight and transfected with the leptin siRNA for
24, 48 or 72 h. The cells were then incubated with
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT;
0.5 mg/ml) for 4 h at 37°C. After removal of MTT, 150 μl of
dimethyl sulphoxide (DMSO) was added to the cells, and the
absorbance was measured at 540 nm using a microplate reader.
Flow cytometry and TUNEL analysis
For flow cytometric analysis, cells were maintained
at a density of 3×105 cells in 60-mm plates. After
transfection with leptin siRNA, the cells were harvested, rinsed
with ice-cold phosphate-buffered saline (PBS), and fixed with
ice-cold 70% ethanol. The cells were centrifuged for 5 min at 1,000
× g and resuspended in PBS containing 5 mM of
ethylenediaminetetraacetic acid (EDTA) and RNase A (1 mg/ml). After
incubation for 1 h at 37°C, the cells were treated for 15 min with
fluorescein isothiocyanate (FITC)-labeled Annexin V and propidium
iodide (PI), according to the supplier’s protocol (Dako, Denmark),
and then were analyzed with a flow cytometer. TUNEL analysis was
employed using the apoptosis detection kit (Maixin, China)
according to the manufacturer’s protocol. The apoptotic cells in 10
different fields were counted, and the apoptotic index was
calculated as the ratio of apoptotic cells over total cells.
Reverse transcription-polymerase chain
reaction (RT-PCR)
Total RNAs were extracted using TRIzol reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA), and
single-stranded cDNA was synthesized from 1 μg of total RNA using
PrimeScript™ RT reagent kit following the protocol recommended by
the manufacturer (Takara, Japan). PCR primers were as follows:
c-myc, sense, GCCACGTCTCCACACATCAG and antisense,
TCTTGGCAGCAGGATAGTCCTT; Bcl-2, sense, TTTCTCATGGCTGTCCTTCAGGGT, and
antisense, AGGTCTGGCTTCATACCACAGGTT. In addition, specific primers
for the 18S RNA were used as control. The primers were sense,
5′-GAGGGAGCCTGAGAAACGG-3′, and antisense,
5′-GTCGGGAGTGGGTAATTTGC-3′. Finally, 30 cycles of PCR amplification
were carried out.
RNA interference
Using a fluorescein-labeled (FAM) siRNA (GenePharma
Co., Ltd., China), multiple targets were designed against leptin
mRNA (13), and specificity was
confirmed using the BLAST tool on the NCBI website. A non-specific
scramble control sequence was also generated. HeLa cells were
transfected with siRNAs at a final concentration of 100 nM
Lipofectamine™ 2000 according to the manufacturer’s
instructions.
Western blotting
Total extracts were obtained in the supernatant.
Approximately 20 μl of the samples was resolved on 10% SDS-PAGE,
and transferred to PVDF membranes. Western blot analyses were
conducted using the following antibodies: rabbit polyclonal
antibodies for leptin (1:200); a mouse polyclonal antibody for
c-myc (1:500) and a rabbit polyclonal antibody for β-actin (1:200)
to detect the corresponding proteins. The peroxidase-conjugated
goat anti-mouse/rabbit secondary antibody (Sigma, St. Louis, MO,
USA) was used. Immunodetection was carried out using enhanced
chemiluminescence (Pierce Biotechnology, Inc., Rockford, IL, USA)
according to the manufacturer’s instructions.
Statistical analysis
Statistical analysis was carried out using SPSS 11.5
software. The relationships between the expression levels of leptin
and the clinicopathologic alterations of cervical cancer were
determined using Spearman correlation analysis. The results of
RT-PCR, western blotting, MTT assay and apoptosis were analyzed
using the paired or unpaired Student’s t-test, and a value of
p<0.05 was assumed to indicate a statistically significant
result. All experiments were independently carried out thrice in
triplicate.
Results
Leptin expression correlates with the
progression of cervical cancer
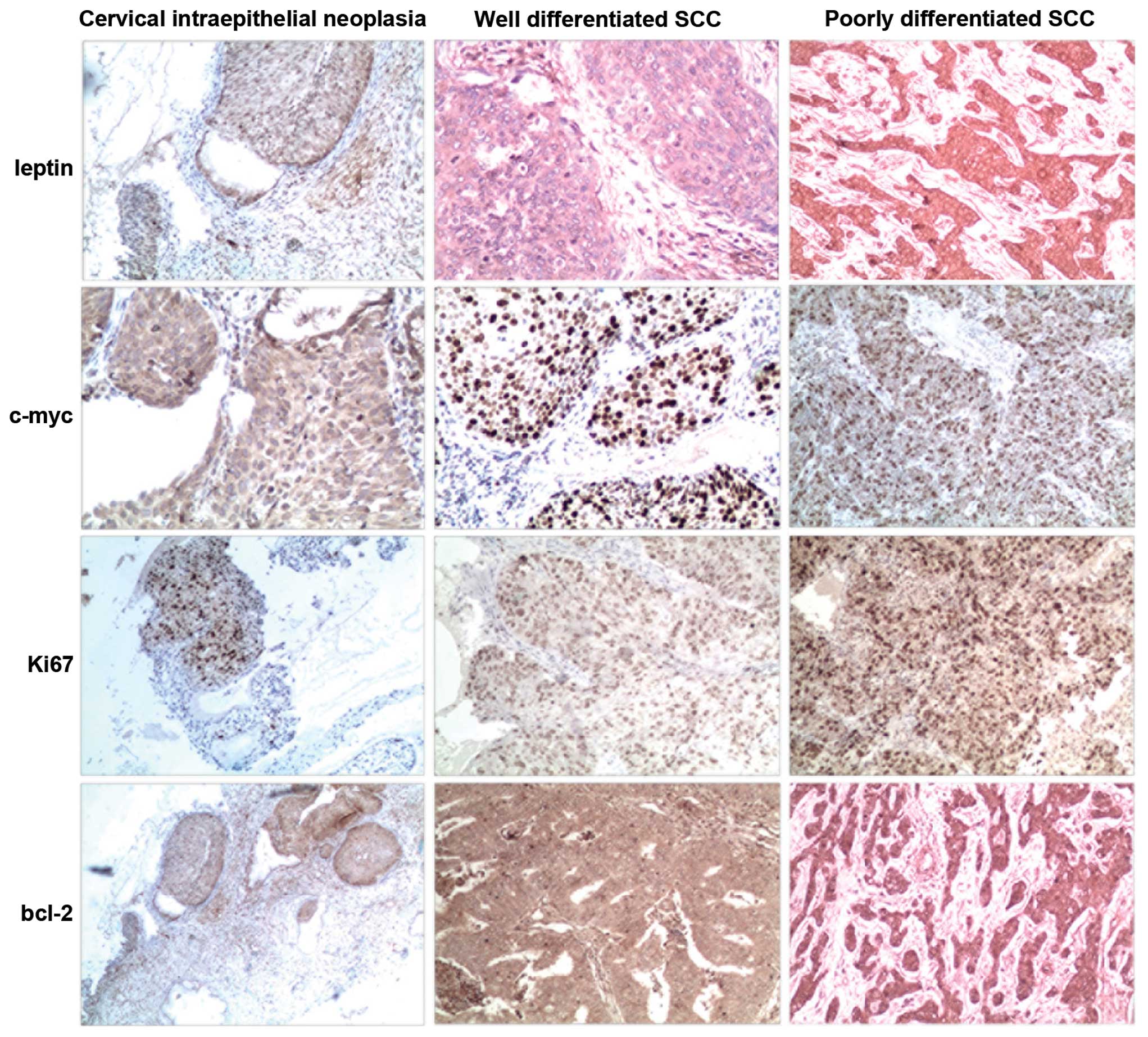
Immunohistochemistry was employed to investigate the
expression of leptin, bcl-2, c-myc and Ki-67 in the cervical cancer
samples (Fig. 1). As shown in
Table I, 21 cases in the 62 samples
of cervical cancer showed leptin-positive expression (33.9%). Ten
cases of the 23 squamous cell carcinoma grade III (43.5%), 8 cases
of the 26 squamous cell carcinoma grade II (30.8%), 3 cases of the
13 squamous cell carcinoma grade II (23.1%) and 3 cases of the 18
cervical intraepithelial neoplasia (CIN) samples (16.7%) showed
leptin-positive staining. Our data revealed that leptin expression
was significantly correlated with the grade of cervical neoplasia
(p<0.05). Analysis of the relationships between leptin and the
studied proteins showed a statistically significant positive
correlation between leptin and anti-apoptotic protein bcl-2
(p<0.05) as well as between leptin and oncogenic c-myc
(p<0.05). Leptin immunoactivity was correlated negatively with
the Ki-67 labeling index (p<0.05).
 | Table IExpression of the studied proteins in
cervical neoplasia. |
Table I
Expression of the studied proteins in
cervical neoplasia.
| Leptin
n (%) | bcl-2
n (%) | Ki-67
n (%) | c-myc
n (%) | Cases
N |
|---|
| CIN | 3 (16.7) | 4 (22.2) | 7 (38.9) | 4 (22.2) | 18 |
| SCC |
| Grade I | 3 (23.1) | 5 (38.5) | 6 (46.2) | 6 (26.1) | 13 |
| Grade II | 8 (30.8) | 16 (61.5) | 17 (65.4) | 11 (42.3) | 26 |
| Grade III | 10 (43.5) | 16 (69.6) | 22 (95.7) | 12 (52.2) | 23 |
Leptin affects apoptosis and
proliferation activity of cervical cancer cells
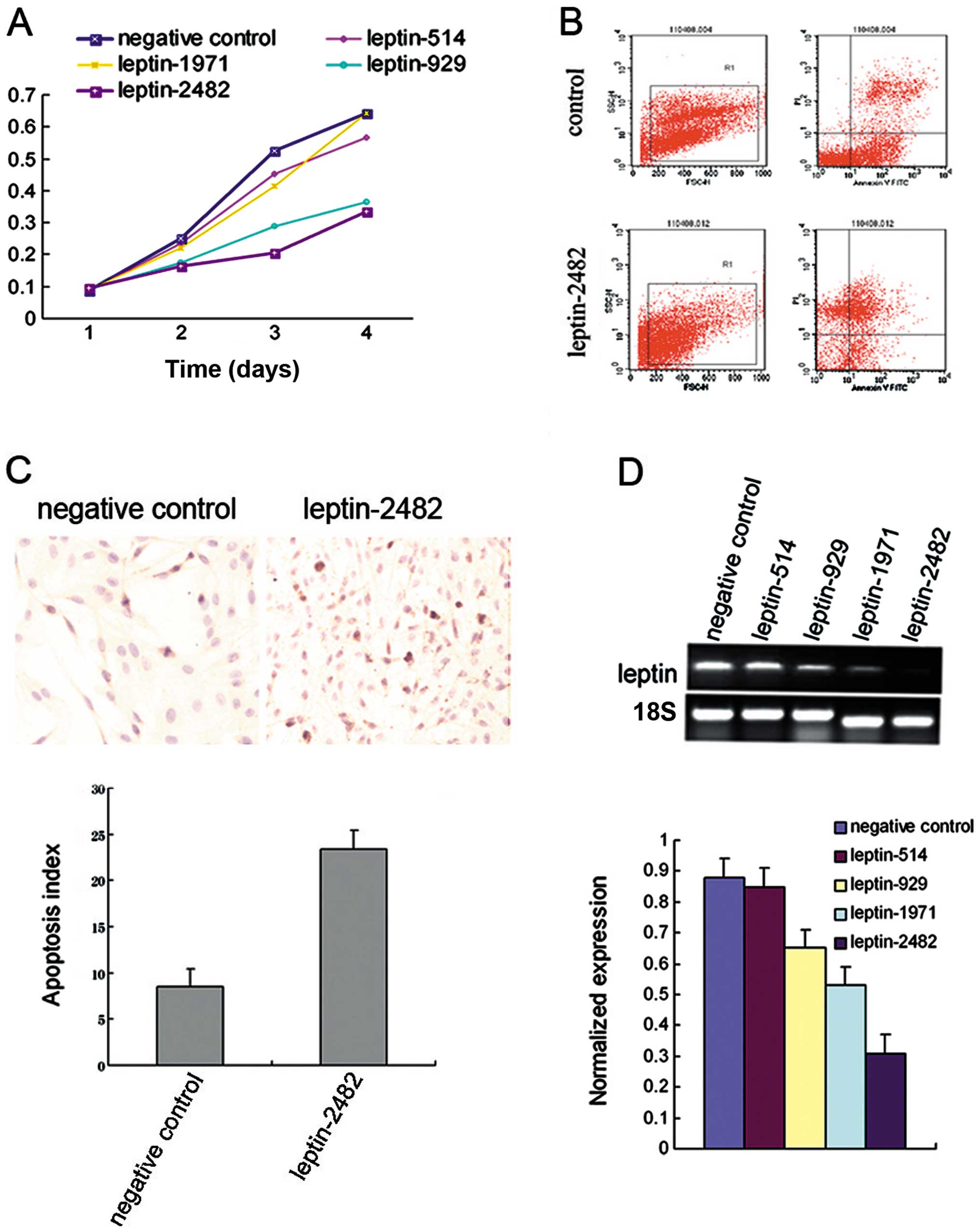
Our previous study revealed that recombinant leptin
dose-dependently (0, 50 and 100 nM) stimulated cell proliferation
and reduced the apoptosis in cervical cancer HeLa cells (12). To further study the functions of
leptin in HeLa cell growth, we knocked down the expression of
leptin in HeLa cells by leptin siRNA as described in Materials and
methods. Our results showed that leptin was successfully silenced
by leptin siRNA at both the mRNA level (Fig. 2D) and the protein level (Fig. 4B). Upon transfection, an apparent
suppression in cell growth was observed in the leptin-silenced HeLa
cells (Fig. 2). At 72 h after
transfection, the viability of the leptin siRNA-transfected HeLa
cells decreased by ~80% compared with the controls according to the
MTT assay (p<0.05) (Fig. 2A).
Apoptosis was detected by flow cytometric analysis and TUNEL
staining. As shown in Fig. 2C the
apoptotic index of the cells with knockdown of leptin significantly
increased when compared to the apoptotic index in the negative
control cells (p<0.05). Flow cytometric analysis revealed that
leptin RNAi increased the apoptosis of HeLa cells from 15.4±2.4 to
29.5±3.2% (p<0.05) (Fig. 2B).
These results indicate that knockdown of leptin inhibits the
proliferative activity and stimulates the apoptosis in cervical
cancer cells.
Leptin regulates the expression of c-myc
and bcl-2 in HeLa cervical cancer cells
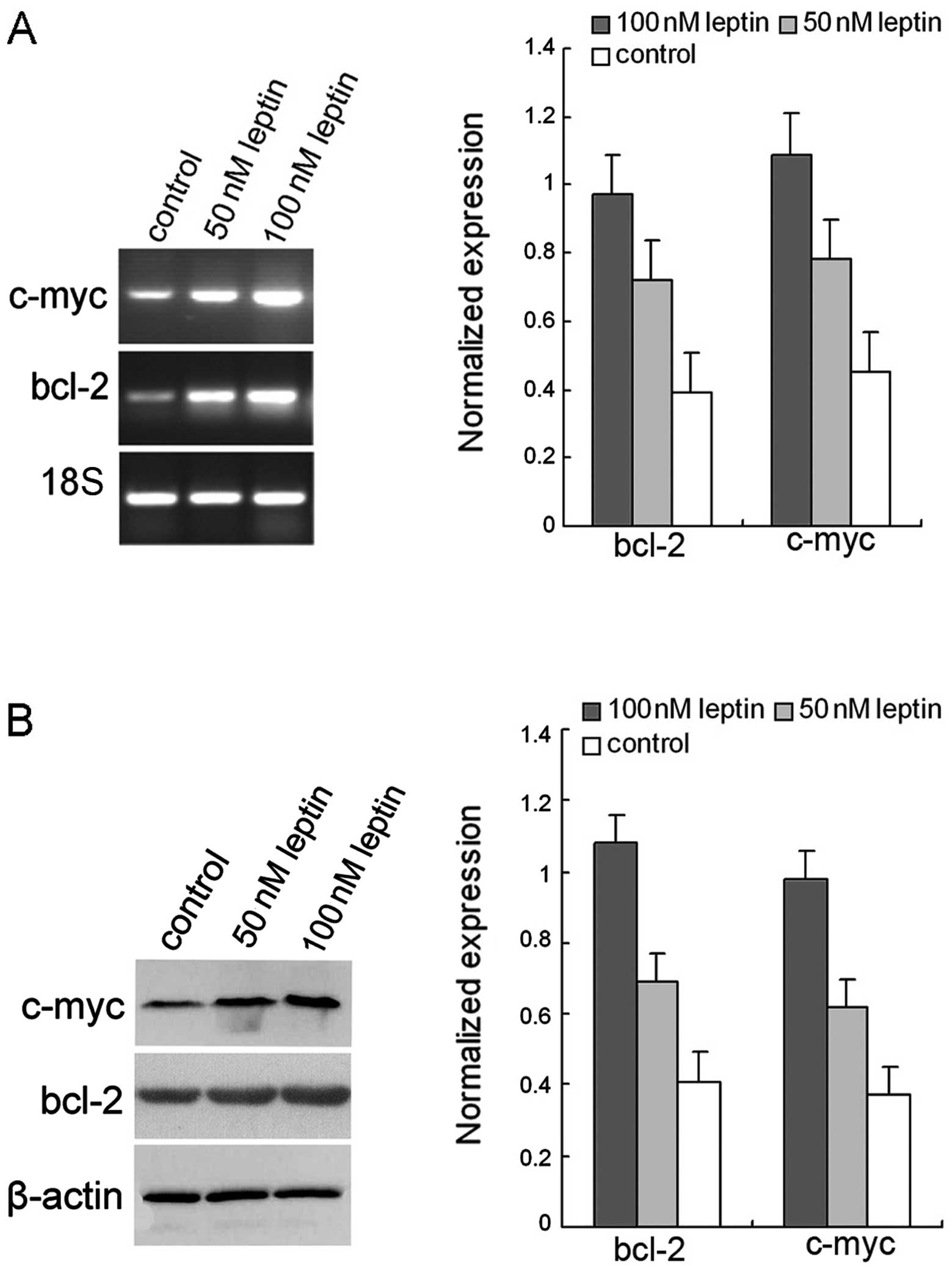
To investigate the mechanism(s) involved in
leptin-mediated cell proliferation in HeLa cells, the expression of
bcl-2 and c-myc was initially measured by RT-PCR following
treatment with recombinant leptin in HeLa cells. As shown in
Fig. 3A, the mRNA level of c-myc in
HeLa cells following leptin treatment markedly increased when
compared with the blank control (p<0.05). Moreover, the mRNA
level of bcl-2 was significantly increased compared with the
control groups (p<0.05). Similarly, the immunoblot analysis
(Fig. 3B) revealed that leptin
induced the expression of c-myc and bcl-2 in HeLa cells. These
results suggest that leptin leads to the activation of c-myc and
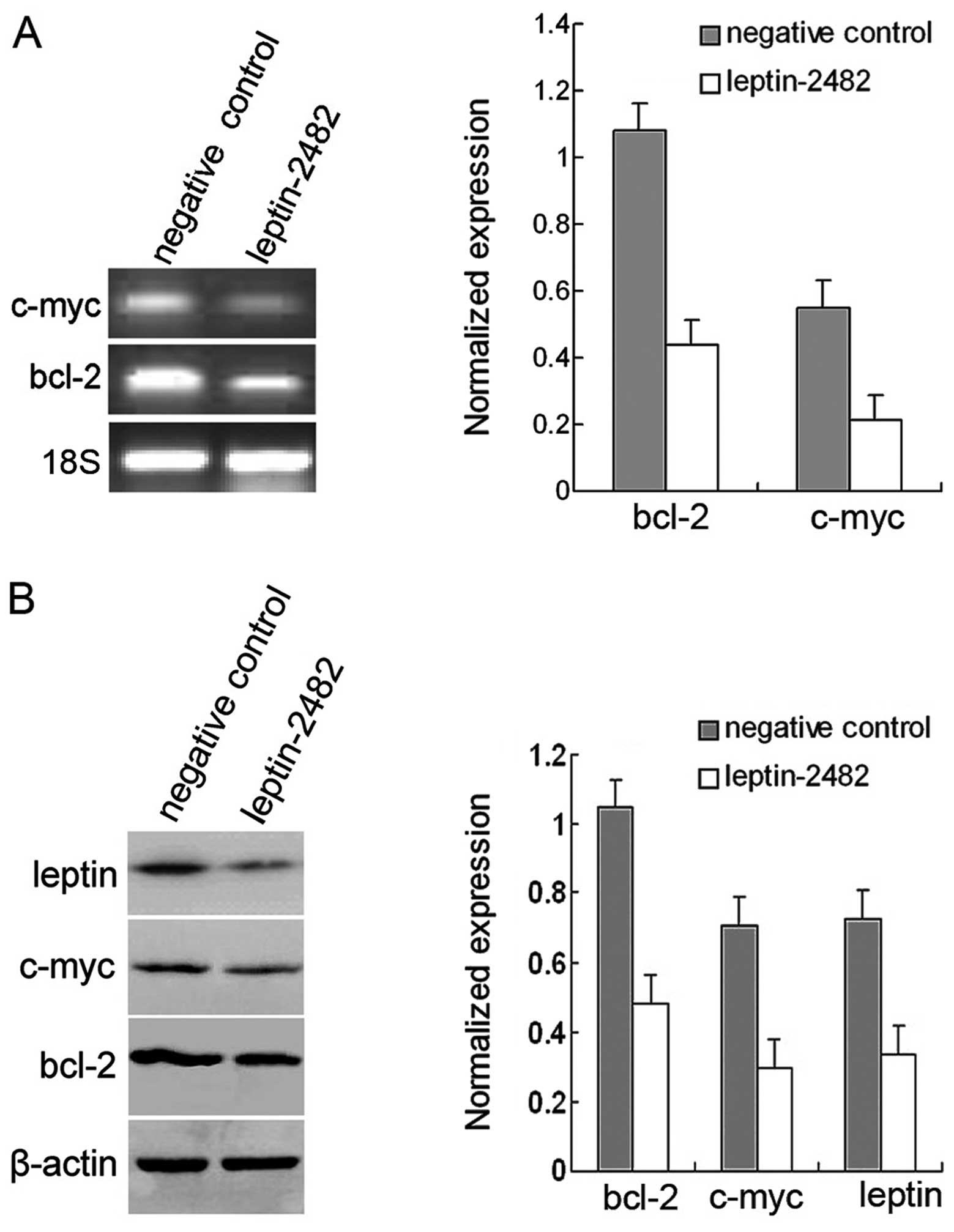
bcl-2, which are known to regulate apoptosis. Then RNA interference
of leptin was employed to gain further insight into the role of
leptin in HeLa cells. Using western blot analysis (Fig. 4B), a significant reduction in leptin
protein levels was noted in cells treated with siRNA, relative to
the negative control (p<0.05; 48 h). The activity of c-myc and
bcl-2 (Fig. 4) as detected by
RT-PCR and western blotting revealed that silencing of leptin
inhibited the expression of c-myc (p<0.05), as well as bcl-2
(p<0.05). This reinforced the finding that leptin is an
efficient regulator of c-myc signaling.
Discussion
The development and progression of cancer involve a
series of molecular changes affected by the interaction of a cell
and its environment. Such processes as angiogenesis,
lymphangiogenesis, cell motility, proliferation, and cell apoptosis
are also important. Leptin, a product of the obese (ob) gene, is a
neuroendocrine hormone that has attracted attention since its
identification in 1995 (2,3). Previous reports suggest that leptin is
overexpressed in various types of cancer cells and plays a role in
the development and/or progression of a variety of malignancies
including colon (14,15), gastric (16), endometrial (17), ovarian (18) and breast cancers (19,20).
In the present study, we firstly examined the prevalence of the
expression of leptin and several genes which regulate apoptosis and
proliferation activity in 80 samples of cervical neoplasia. Leptin
protein was detected in 33.9% of the examined cases. The samples
with high leptin expression showed significantly poorer
differentiation. In contrast, reduced leptin expression was noted
in cervical intraepithelial neoplasia and well differentiated
cancer. We also observed a significant correlation between leptin
expression and the Ki-67 labeling index in the cervical cancers.
This suggests that leptin, through an autocrine mechanism,
activates cervical cancer to increased progression of the disease,
and the expression level of leptin may provide useful information
for estimating the malignancy of cervical cancer.
Leptin was previously reported to stimulate the
proliferation of various types of tumor cells. Mauro et
al(21) observed that leptin
stimulated the proliferation of MCF-7 breast cancer cells in
vivo and this action was correlated with increased expression
of E-cadherin and cyclin D1. Rouet-Benzineb et al(22) reported that leptin enhanced the
apoptosis of human colon cancer HT-29 cells probably through
activation of the NF-κB pathways. Escobar-Morreale and San Millán
(23) and Sharma et
al(24) observed increased
proliferation by leptin in endometrial and ovarian cancer cells.
Moreover, Saxena et al(10)
found the leptin promoted invasion and migration of hepatocellular
carcinoma cells. Our previous experiment revealed that recombinant
leptin significantly induced proliferation and reduced apoptosis in
HeLa cells (12). Here, we found
that leptin siRNA efficiently reversed the growth of HeLa cells.
Our experiments clearly showed that leptin has an oncogenic effect
on HeLa cells and this oncogenic effect is due to a combination of
cell proliferation and inhibition of apoptosis by leptin.
The c-myc gene was found to be the cellular homolog
of retroviral v-myc oncogene about 30 years ago (25). It is located on chromosomal region
8q24.1 and encodes a nuclear phosphoprotein (26) and is generally recognized as an
important regulator of proliferation, growth, differentiation and
apoptosis (27). c-myc regulates
transcription from its targets through several mechanisms,
including recruitment of histone acetylases, chromatin modulating
proteins, basal transcription factors and DNA methyltransferase
(28). Oncogenic alterations of myc
are commonly induced by events such as point mutations, gene
amplification, chromosomal translocation, viral insertion at the
myc locus, and resistance of myc protein to ubiquitin-mediated
proteolysis and enhanced transcription or translation by other
oncogenic signaling pathways. Expression of the c-myc oncogene or
its protein product is elevated in virtually all types of malignant
diseases (29). Therefore, it is
also accepted that the deregulation of myc expression is a major
event in cancer pathogenesis or progression (30). Groups of genes involved in cell
cycle regulation, metabolism, ribosome biogenesis, protein
synthesis and mitochondrial function are overrepresented in the myc
target gene network. Bcl-2, which is regarded as a significant
target gene, has been investigated for prognostic significance in
various malignancies, including carcinoma of the cervix (31,32).
Bcl-2 has been shown in some studies as an independent predictor of
poor prognosis in carcinoma of the cervix (33). To elucidate the target regulators
involved in leptin-mediated induction of the cancerous properties
of HeLa cells, we examined the effect of leptin on the activation
of c-myc and bcl-2. Our data showed that leptin rapidly stimulated
c-myc and bcl-2 thus activating these key signaling regulators
associated with cell growth. In addition, gene silencing of leptin
in HeLa cell lines abrogated c-myc activation as well as the
expression of anti-apoptotic gene bcl-2, suggesting that
leptin-mediated HeLa cell growth is due to the modulation of
oncogene c-myc and anti-apoptotic gene bcl-2 expression.
In conclusion, leptin expression was significantly
correlated with the grade of cervical carcinoma, as well as a high
Ki-67 labeling index. Furthermore, the levels of leptin were
significantly positively correlated with oncogene c-myc and its
target gene, bcl-2. This suggests an important role for leptin in
cervical carcinogenesis. Moreover, our data revealed that leptin
regulates the expression of c-myc and bcl-2 in HeLa cells, and the
molecular mechanism responsible for leptin-mediated cell
proliferation in cervical cancer was further elucidated, thus
establishing a direct association between leptin and cervical
carcinogenesis. Our findings have potential clinical implication in
the prevention of development and progression of cervical
cancer.
Acknowledgements
The present study was supported by a grant from the
Science and Technology Department of Shaanxi, China
(2010k14-03-11). We are grateful to Dr Zhizhong Wang for help with
the statistical analysis and Dr Weidong Lü for his critical
comments concerning the manuscript.
References
|
1
|
Jemal A, Bray F, Center MM, et al: Global
cancer statistics. CA Cancer J Clin. 61:69–90. 2011. View Article : Google Scholar
|
|
2
|
MacDougald OA, Hwang CS, Fan H, Lane MD,
et al: Regulated expression of the obese gene product (leptin) in
white adipose tissue and 3T3-L1 adipocytes. Proc Natl Acad Sci USA.
92:9034–9037. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Halaas JL, Gajiwala KS, Maffei M, et al:
Weight-reducing effects of the plasma protein encoded by the obese
gene. Science. 269:543–546. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Huang L and Li C: Leptin: a
multifunctional hormone. Cell Res. 10:81–92. 2000. View Article : Google Scholar
|
|
5
|
Bouloumié A, Drexler HC, Lafontan M and
Busse R: Leptin, the product of Ob gene, promotes angiogenesis.
Circ Res. 83:1059–1066. 1998.PubMed/NCBI
|
|
6
|
Sierra-Honigmann MR, Nath AK, Murakami C,
et al: Biological action of leptin as an angiogenic factor.
Science. 281:1683–1686. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Somasunder P, McFadden DW, Hileman SM and
Vona-Davis L: Leptin is a growth factor in cancer. J Surg Res.
116:337–349. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bjorbaek C, Uotani S, da Silva B and Flier
JS: Divergent signaling capacities of the long and short isoforms
of the leptin receptor. J Biol Chem. 272:32686–32695. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tartaglia LA: The leptin receptor. J Biol
Chem. 272:6093–6096. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Saxena NK, Sharma D, Ding X, et al:
Concomitant activation of the JAK/STAT, PI3K/AKT, and ERK signaling
is involved in leptin-mediated promotion of invasion and migration
of hepatocellular carcinoma cells. Cancer Res. 67:2497–2507. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yin N, Wang D, Zhang H, et al: Molecular
mechanisms involved in the growth stimulation of breast cancer
cells by leptin. Cancer Res. 64:5870–5875. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yuan Y, Wang X, Lu J, et al: Effects of
leptin on biological behavior of cervical cancer Hela cells and its
mechanism. Chin J Clin Exp Pathol. 28:162–165. 2012.(In
Chinese).
|
|
13
|
Elbashir SM, Harborth J, Weber K and
Tuschl T: Analysis of gene function in somatic mammalian cells
using small interfering RNAs. Methods. 26:199–213. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hoda MR, Keely SJ, Bertelsen LS, Junger
WG, Dharmasena D and Barrett KE: Leptin acts as a mitogenic and
antiapoptotic factor for colonic cancer cells. Br J Surg.
94:346–354. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ogunwobi OO and Beales IL: The
anti-apoptotic and growth stimulatory actions of leptin in human
colon cancer cells involves activation of JNK mitogen activated
protein kinase, JAK2 and PI3 kinase/Akt. Int J Colorectal Dis.
22:401–409. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Pai R, Lin C, Tran T and Tarnawski A:
Leptin activates STAT and ERK2 pathways and induces gastric cancer
cell proliferation. Biochem Biophys Res Commun. 331:984–992. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Koda M, Sulkowska M, Wincewicz A,
Kanczuga-Koda L, Musiatowicz B, Szymanska M and Sulkowski S:
Expression of leptin, leptin receptor, and hypoxia-inducible factor
1 alpha in human endometrial cancer. Ann NY Acad Sci. 1095:90–98.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Choi JH, Park SH, Leung PC and Choi KC:
Expression of leptin receptors and potential effects of leptin on
the cell growth and activation of mitogen-activated protein kinases
in ovarian cancer cells. J Clin Endocrinol Metab. 90:207–210. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ishikawa M, Kitayama J and Nagawa H:
Enhanced expression of leptin and leptin receptor (OB-R) in human
breast cancer. Clin Cancer Res. 10:4325–4331. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hu X, Juneja SC, Maihle NJ and Cleary MP:
Leptin - a growth factor in normal and malignant breast cells and
for normal mammary gland development. J Natl Cancer Inst.
94:1704–1711. 2002. View Article : Google Scholar
|
|
21
|
Mauro L, Catalano S, Bossi G, et al:
Evidences that leptin up-regulates E-cadherin expression in breast
cancer: effects on tumor growth and progression. Cancer Res.
67:3412–3421. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rouet-Benzineb P, Aparicio T, Guilmeau S,
Pouzet C, Descatoire V, Buyse M and Bado A: Leptin counteracts
sodium butyrate-induced apoptosis in human colon cancer HT-29 cells
via NF-κB signaling. J Biol Chem. 279:16495–16502. 2004.PubMed/NCBI
|
|
23
|
Escobar-Morreale HF and San Millán JL:
Abdominal adiposity and the polycystic ovary syndrome. Trends
Endocrinol Metab. 18:266–272. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sharma D, Saxena NK, Vertino PM and Anania
FA: Leptin promotes the proliferative response and invasiveness in
human endometrial cancer cells by activating multiple
signal-transduction pathways. Endocr Relat Cancer. 13:629–640.
2006. View Article : Google Scholar
|
|
25
|
Sheiness D, Fanshier L and Bishop JM:
Identification of nucleotide sequences which may encode the
oncogenic capacity of avian retrovirus MC29. J Virol. 28:600–610.
1978.PubMed/NCBI
|
|
26
|
Persson H and Leder P: Nuclear
localization and DNA binding properties of a protein expressed by
human c-myc oncogene. Science. 225:718–721. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vita M and Henriksson M: The Myc
oncoprotein as a therapeutic target for human cancer. Semin Cancer
Biol. 16:318–330. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
O’Connell BC, Cheung AF, Simkevich CP, Tam
W, Ren X, Mateyak MK and Sedivy JM: A large scale genetic analysis
of c-Myc-regulated gene expression patterns. J Biol Chem.
278:12563–12573. 2003.PubMed/NCBI
|
|
29
|
Nesbit CE, Tersak JM and Prochownik EV:
MYC oncogenes and human neoplastic disease. Oncogene. 18:3004–3016.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chung HJ and Levens D: c-myc expression:
keep the noise down! Mol Cells. 20:157–166. 2005.PubMed/NCBI
|
|
31
|
Skirnisdóttir I, Seidal T, Gerdin E and
Sorbe B: The prognostic importance of p53, bcl-2, and bax in early
stage epithelial ovarian carcinoma treated with adjuvant
chemotherapy. Int J Gynecol Cancer. 12:265–276. 2002.PubMed/NCBI
|
|
32
|
Oltvai ZN, Milliman CL and Korsmeyer SJ:
Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that
accelerates programmed cell death. Cell. 74:609–619. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chung TK, Cheung TH, Lo WK, et al:
Expression of apoptotic regulators and their significance in
cervical cancer. Cancer Lett. 180:63–68. 2002. View Article : Google Scholar : PubMed/NCBI
|