Introduction
Multidrug resistant (MDR) cancer is a major problem
in cancer therapy and is often the result of overexpression of the
drug efflux protein, P-glycoprotein (P-gp). P-gp is a 170-kDa
protein that belongs to the ATP-binding cassette superfamily of
membrane transporter proteins (1,2). P-gp
is an energy-dependent drug efflux pump that maintains
intracellular drug concentrations below cytotoxic levels, thereby
decreasing the cytotoxic effects of a variety of chemotherapeutic
agents, including anthracyclines, vinca alkaloids and
epipodophyllotoxins (1–4). P-gp also plays a role in the
inhibition of drug accumulation and caspase activation in MDR
tumors (5,6). Importantly, recent lines of evidence
have shown that NF-κB- or SIRT1-mediated regulation of P-gp plays a
critical role in anticancer drug resistance (7,8).
Medicinal plants are a rich source of therapeutic
agents and have provided precursors for several synthetic drugs.
Despite the great development of organic synthesis technology,
currently 75% of prescribed drugs worldwide are derived from plant
sources (9). Some species of
Bryophyllum, which belongs to the plant genus
Kalanchoe of the Crassulaceae family, are perennial herbs
that grow in the wild and are used as traditional medicinal plants
in tropical Africa, China, Australia and tropical America (10). In traditional medicine, some species
of Bryophyllum have been used to treat ailments such as
infections, rheumatism and inflammation (11). A recent study also reported that
extracts from Brophyllum pinnata exhibit anticancer activity
against human cervical cancer cells (10). However, the molecular mechanisms by
which extracts of the plant show anticancer activity have not been
elucidated.
Since our previous research found that human A549
lung cancer cells resistant to etoposide (A549RT-eto) displayed
upregulation of P-gp, we screened natural compounds to reverse this
resistance to etoposide in these cells. In the present study, we
investigated whether an extract of B. laetivirens
efficiently induces cytotoxicity in A549RT-eto cells and further
examined possible molecular mechanisms by which an extract of B.
laetivirens can induce the reversal of MDR, leading to
apoptosis. We found that an extract of B. laetivirens
enhanced the apoptosis of A549RT-eto cells at least in part through
downregulation of P-gp, which was mediated by suppression of NF-κB
expression and activity.
Materials and methods
Reagents and antibodies
For immunoblotting, an antibody against cleaved PARP
(Asp214) was acquired from Cell Signaling Biotechnology (Beverly,
MA, USA). Anti-P-gp (Calbiochem, San Diego, CA, USA), NF-κB p65
(F-6), SIRT1 (H-300), Sp1 (1C6), caspase-9 p35 (H-170), cytochrome
c (A-8) and actin (C4) antibodies (Santa Cruz Biotechnology,
Santa Cruz, CA, USA) were used. Nicotinamide and BAY11-7082 were
purchased from Sigma-Aldrich (St. Louis, MO, USA).
Cell cultures
A549 or A549RT-eto cells, which were developed and
kindly provided by the Laboratory of Biochemistry, Chulabhorn
Research Institute, Thailand as described elsewhere (12), were cultured in RPMI-1640 medium
(Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine
serum (FBS) and 1% penicillin and streptomycin (Gibco) at 37ºC in a
humidified atmosphere of 5% CO2 in air.
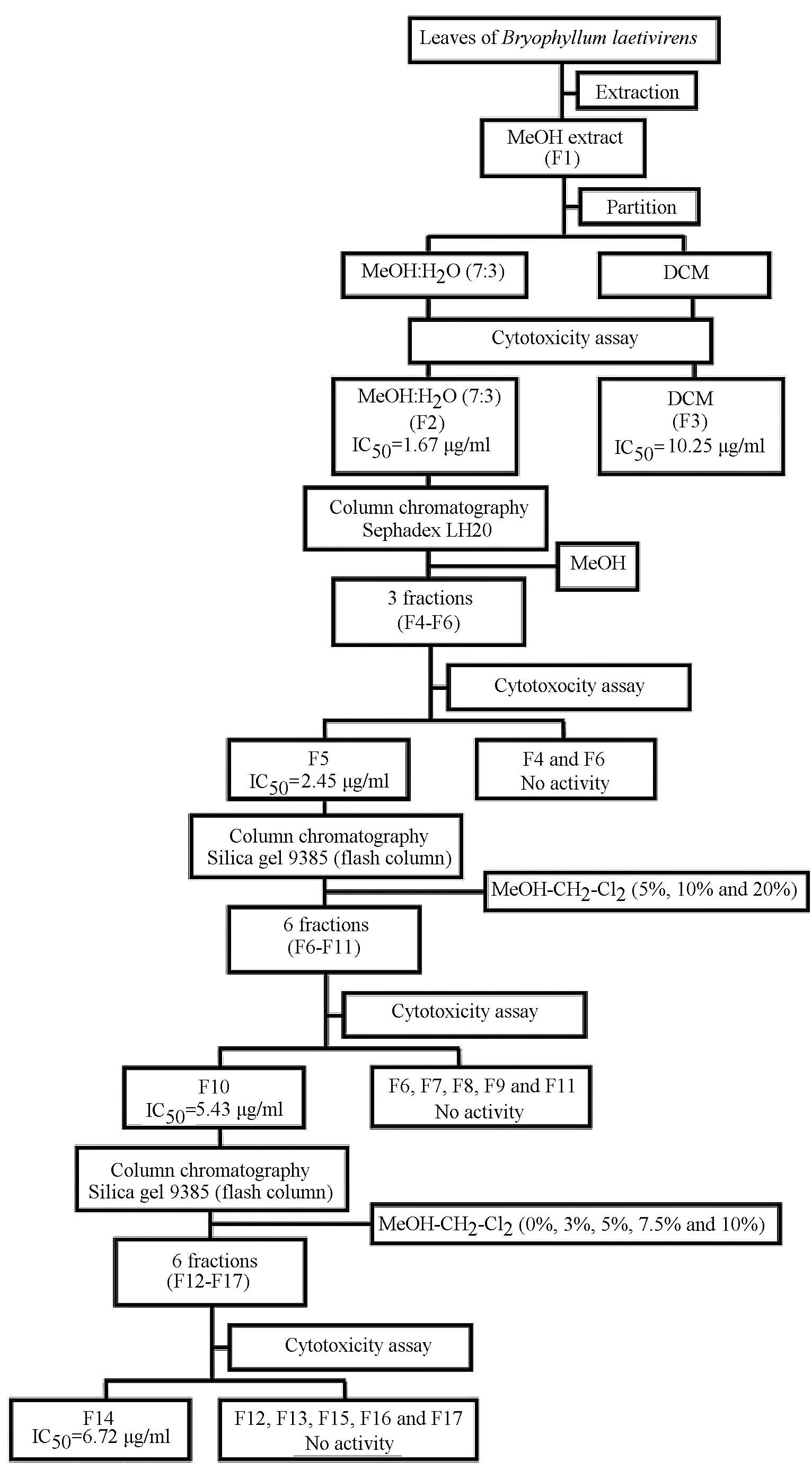
Preparation of extracts from B.
laetivirens
Fresh leaves of B. laetivirens (3 kg) were
dried at 45ºC and ground into powder. The samples were extracted
with MeOH. The extracts were partitioned between H2O and
CH2Cl2 and then assessed for cytotoxicity. A
portion of the MeOH extract was chromatographed on Sephadex LH-20
gel (100 g; GE Healthcare, Piscataway, NJ, USA), and the fractions
showing cytotoxicity to A549RT-eto cells were collected. The active
fractions were then subjected to a silica gel (50 g; 60–230 mesh)
column in hexane and eluted with MeOH:CH2Cl2
(5, 10 and 20%). The active fractions were again subjected to this
column and eluted with MeOH:CH2Cl2 (0, 3, 5,
7.5 and 10%). The fractions showing a similar TLC profile were
combined and then evaluated for cytotoxic activity. Isolation of
the bioactive faction is diagrammed in Fig. 1, and the fraction, identified as
F14, showed one single spot on TLC and the highest cytotoxic effect
on A549RT-eto cells.
Immunoblotting
Cells were harvested and lysed with lysis buffer
[150 mM NaCl, 1% NP-40, 50 mM Tris-HCl (pH 7.5)]containing 0.1 mM
Na2VO3, 1 mM NaF and protease inhibitors
(Sigma). For immunoblotting, proteins from whole cell lysates were
resolved by 10 or 12% SDS-polyacrylamide gel electrophoresis (PAGE)
and then transferred to nitrocellulose membranes. Primary
antibodies were used at 1:1,000 or 1:2,000 dilutions, and secondary
antibodies conjugated with horseradish peroxidase were used at
1:2,000 dilutions in 5% non-fat dry milk. After the final washing,
nitrocellulose membranes were exposed for an enhanced
chemiluminescence assay using the LAS 4000 Mini (Fuji, Tokyo,
Japan).
Nuclear NF-κB pull-down assay
A549 or A549RT-eto cells (1×106 cells/ml)
were incubated with the F14 fraction or DMSO as control for 12 h,
and the nuclear extracts were prepared. Cells were pelleted and
resuspended in 0.4 ml hypotonic lysis buffer [20 mM HEPES (pH 7.9),
10 mM KCl, 1 mM EDTA, 0.2% Triton X-100 and 1 mM
Na2VO3 plus protease inhibitors] and kept on
ice for 20 min. After centrifugation at 14,000 × g for 5 min at
4ºC, the nuclear pellet was extracted with 0.1 ml hypertonic lysis
buffer on ice for a further 20 min. After centrifugation at 14,000
× g for 5 min at 4ºC, the supernatants were diluted with 100 mM
NaCl and incubated with 25 μl of agarose beads conjugated to a
consensus NF-κB binding oligonucleotide (Santa Cruz Biotechnology)
for 1 h at 4ºC. After 3 washes, sample buffer was added and boiled
for 5 min. The binding of NF-κB (p65) protein to the
oligonucleotide conjugated with agarose was detected by
immunoblotting using an anti-p65 NF-κB antibody (Santa Cruz
Biotechnology).
Short interference RNA transfection
Cells were trypsinized and incubated overnight to
achieve 60–70% confluency before siRNA transfection. SIRT1 siRNA
(commercially pre-made at Bioneer Corporation, Daejeon, Korea; 100
nM), sense, 5′-ACU UUG CUG UAA CCC UGU A(dTdT)-3′; antisense,
5′-UAC AGG GUU ACA GCA AAG U(dTdT)-3′ or negative control siRNA
(Bioneer) were mixed with Lipofectamine 2000 (Invitrogen, Carlsbad,
CA, USA). The cells were incubated with the transfection mixture
for 6 h and then rinsed with RPMI-1640 medium containing 10% FBS.
The cells were incubated for 48 h before harvesting.
Reverse transcriptase-polymerase chain
reaction (RT-PCR)
Total RNA was extracted from the cells using the
RNeasy Mini kit (Qiagen, Valencia, CA, USA) in accordance with the
manufacturer’s instructions. Three micrograms of total RNA was
converted to cDNA using SuperScript II reverse transcriptase
(Invitrogen), and PCR was performed using the following specific
primers: human MDR1, sense 5′-CCC ATC ATT GCA ATA GCA GG-3′
and antisense 5′-GTT CAA ACT TCT GCT CCT GA-3′; MRP2, sense
5′-ACA GAG GCT GGT GGC AAC C-3′ and antisense 5′-ACC ATT ACC TTG
TCA CTG TCC-3′; BCRP, sense 5′-GAT CAC AGT CTT CAA GGA GAT
C-3′ and antisense 5′-CAG TCC CAG TAC GAC TGT GAC A-3′. The cDNAs
of each sample were diluted, and PCR was run at the optimized cycle
number. β-actin mRNA was measured as an internal standard. After
amplification, the products were subjected to electrophoresis on
2.0% agarose gel and detected by ethidium bromide staining
Luciferase reporter assay
A549RT-eto cells were transfected with
hMDR1-luciferase or pGL3 empty vector as a control luciferase
vector. To normalize transfection efficiency, a pGK-β-gal vector
that expresses β-galactosidase from a phosphoglucokinase promoter
was included in the transfection mixture. F14 extract was added to
the transfected cells at 12 h before harvesting. At 48 h
post-transfection, cells were washed with cold PBS and lysed in
lysis solution [25 mM Tris (pH 7.8), 2 mM EDTA, 2 mM DTT, 10%
glycerol and 1% Triton X-100]. Luciferase activity was measured
with a luminometer by using a luciferase kit (Promega, Madison, WI,
USA).
SIRT1 activity assay
SIRT1 activity from A549, A549RT-eto or A549RT-eto
cells treated with F14 extract was measured using the SIRT1 direct
fluorescence kit provided by Cayman (Ann Arbor, MI, USA) according
to the manufacturer’s recommendations. Briefly, 25 μl of assay
buffer was added to the cell supernatant. Subsequently, 15 μl of
substrate solution (containing a final concentration of 125 μM
peptides and 3 mM NAD+) was added, and the sample
mixture was incubated for 45 min before addition of stop solution.
Fluorescence (an excitation wavelength 360 nm and an emission
wavelength 460 nm) was measured using the Victor3 microplate reader
(Perkin-Elmer, Waltham, MA, USA).
MTT assay
MTT assays were performed for the measurement of
cell survival as previously described (13). Dye solution containing tetrazolium
was added to the cells in a 96-well plate and incubated for 2 h.
The absorbance of the formazan produced by living cells was
measured at 570 nm. The relative percentage of cell survival was
calculated by the following formula: % Cell survival =
(ODT/ODC), where ODT is the mean
absorbance of the treated cells and ODC is the mean
absorbance of the control cells.
Statistical analysis
Data are presented as means ± standard deviation
(SD). The Student’s t-test was used for statistical analysis, with
P-value <0.05 defined as indicative of statistical
significance.
Results
Extract of B. laetivirens induces
apoptosis of human A549 lung cells resistant to etoposide
We previously observed that human A549 lung cancer
cells resistant to etoposide (A549RT-eto) exhibit upregulation of
Stat1 and HDAC4, leading to the enhancement of P-gp protein levels
(unpublished data). Moreover, recent studies have shown that NF-κB
and SIRT1 are also involved in multidrug resistance (MDR). We thus
examined whether NF-κB and SIRT1 protein levels are upregulated in
A549RT-eto cells in addition to upregulation of Stat1 and HDAC4. We
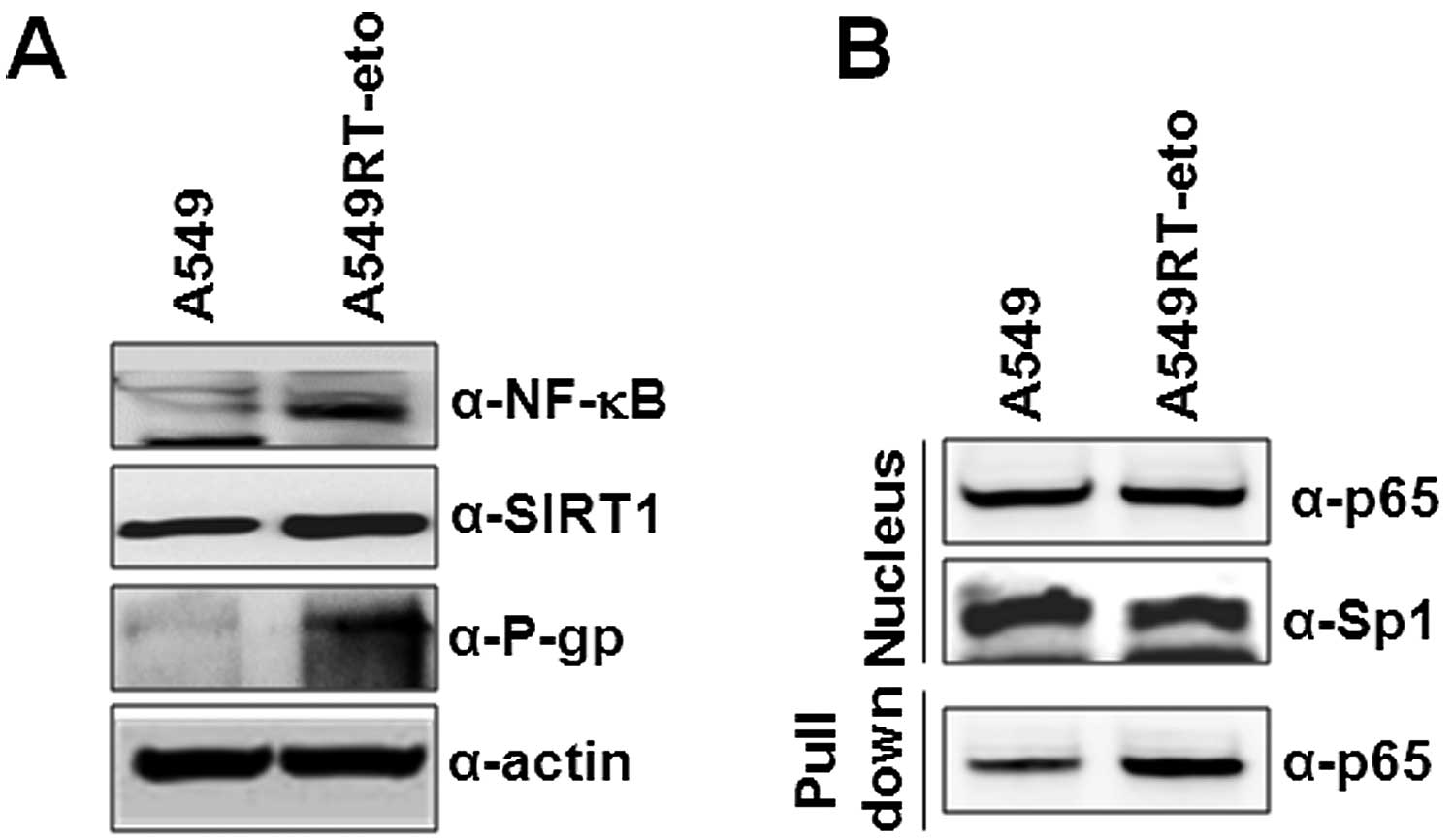
found that A549RT-eto cells showed higher levels of NF-κB and SIRT1
proteins when compared to these levels in the A549 parental cells
(Fig. 2A). We next wondered whether
the elevated protein levels of NF-κB in A549RT-eto cells indicate
higher activity of NF-κB compared to A549 cells. To address this
question, we prepared nuclear extracts from A549 and A549RT-eto
cells. Since the active NF-κB protein is translocated into the
nucleus and binds to its binding site, we examined whether p65
NF-κB protein found at higher levels in A659RT-eto cells can bind
to NF-κB oligonucleotides conjugated with agarose. We found that
more active p65 proteins were detected in the oligonucleotide
mixtures from A549RT-eto cells than those from A549 cells (Fig. 2B). With the same intent, we also
measured SIRT1 activity as we observed enhanced protein levels of
SIRT1 in A549RT-eto cells when compared to these levels in A549
cells. We found that A549RT-eto cells showed higher activity of
SIRT1 than that in A549 cells using a SIRT1 activity kit (Fig. 2C).
On the basis of our previous study to screen
extracts from medicinal plant sources exhibiting the the strongest
cytotoxic activity against A549RT-eto cells (13), we chose B. laetivirens. We,
thus, purified and collected an active pool from B.
laetivirens, and then named the active fraction as F14 extract
(Fig. 1). We first optimized the
concentration and treatment time of the F14 extract, and found that
20 μg/ml of F14 extract significantly induced the cell death of
A549RT-eto cells at 24 h post-treatment but not at 12 h
post-treatment (Fig. 3A). However,
when we examined the protein levels of NF-κB, SIRT1 and P-gp in the
A549RT-eto cells treated with F14 (20 μg/ml) at 12 h
post-treatment, we found that F14 treatment drastically reduced the
expression levels of NF-κB, SIRT1 and P-gp (Fig. 3B). In addition, we detected reduced
pro-caspase-9 levels, and enhanced cytochrome c levels in
the same lysates of A549RT-eto cells, indicating induction of
intrinsic apoptosis (Fig. 3A).
Furthermore, when we treated A549RT-eto cells with the F14 extract
at 0.5 and 5 μg/ml for 12 h, we found that the F14 extract at 5
μg/ml was sufficient to reduce not only expression levels of NF-κB,
SIRT1 and P-gp but also activities of NF-κB and SIRT1 (Fig. 3C and D).
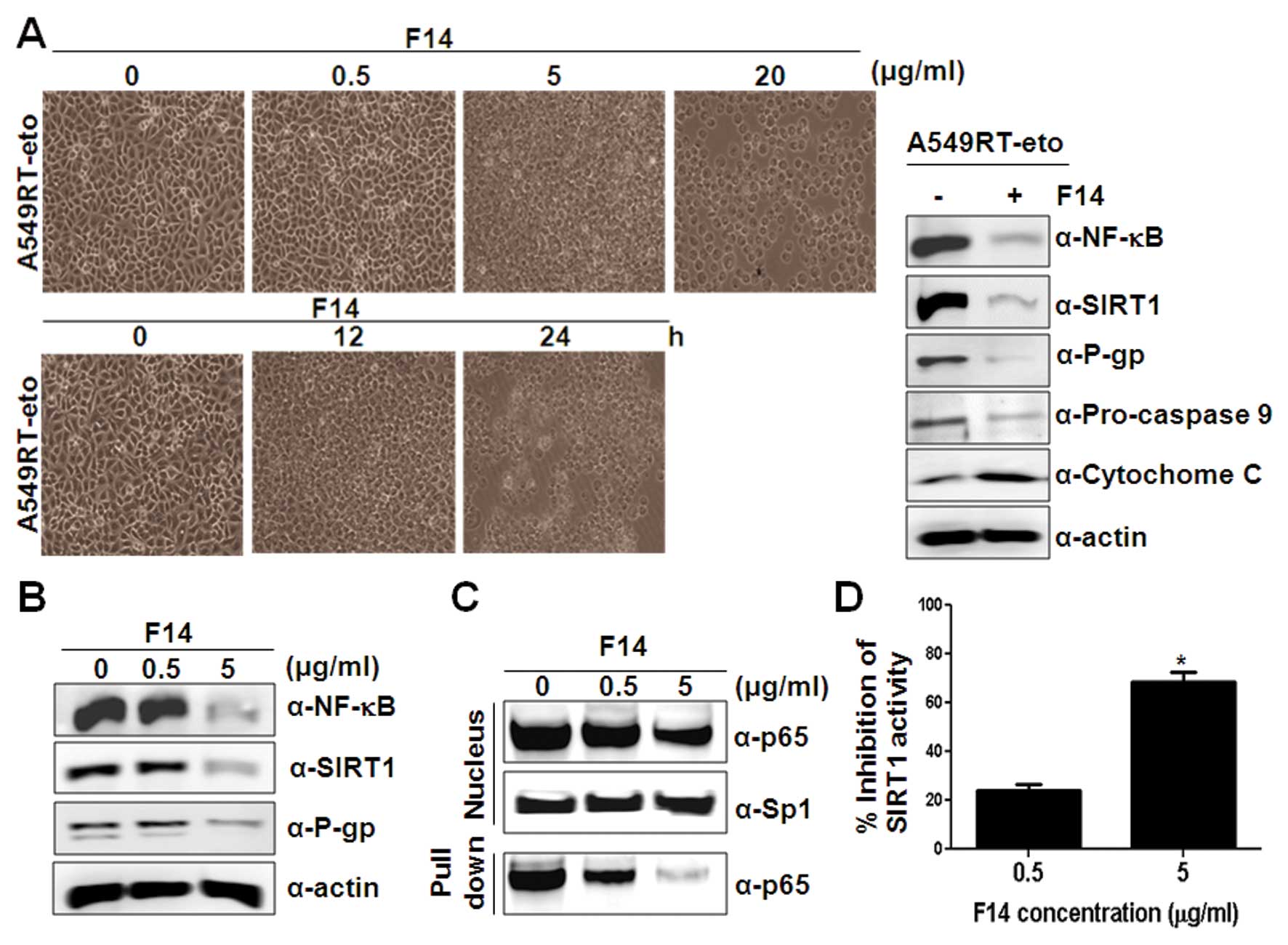 | Figure 3F14 extract suppresses levels of
NF-κB, SIRT1 and P-gp proteins, leading to F14-induced apoptosis.
(A) A549RT-eto cells were treated with F14 (0, 0.5, 5 and 20 μg/ml)
for 12 h or were treated with F14 (20 μg/ml) at 0, 12 and 24 h.
After treatment, the cells were observed under a light microscope.
Cell lysates from A549RT-eto cells in the presence of F14 (20
μg/ml) or absence of F14 at 12 h were prepared and separated on a
12% SDS-PAGE gel. The expression of pro-caspase-9 and cytochrome
c protein was measured as indicators of apoptosis by
immunoblotting, and protein levels of NF-κB, SIRT1 and P-gp were
compared by immunoblotting with the corresponding antibodies. (B)
Cell lysates from A549RT-eto cells in the presence (0, 0.5 and 5
μg/ml) or absence of F14 at 12 h were prepared for the detection of
protein levels of NF-κB, SIRT1 and P-gp with immunoblotting using
the corresponding antibodies. (C) NF-κB oligonucleotides conjugated
with agarose were added to nuclear extracts from the A549RT-eto
cells treated with F14 (0, 0.5 and 5 μg/ml) and the precipitation
mixtures were then isolated after centrifugation. The binding of
NF-κB (p65) to the oligonucleotide was detected by immunoblotting
using anti-NF-κB (p65) Ab. (D) Samples were prepared from
A549RT-eto cells treated with F14 (0, 0.5 and 5 μg/ml) at 12 h
post-treatment. SIRT1 activity was measured using a SIRT1
fluorometric kit according to the manufacturer’s protocol.
Fluorescence was measured using a fluorescence microplate at an
excitation wavelength of 360 nm and an emission wavelength of 460
nm. The results shown are the average of triplicates; bar indicates
standard deviation (*P<0.01). |
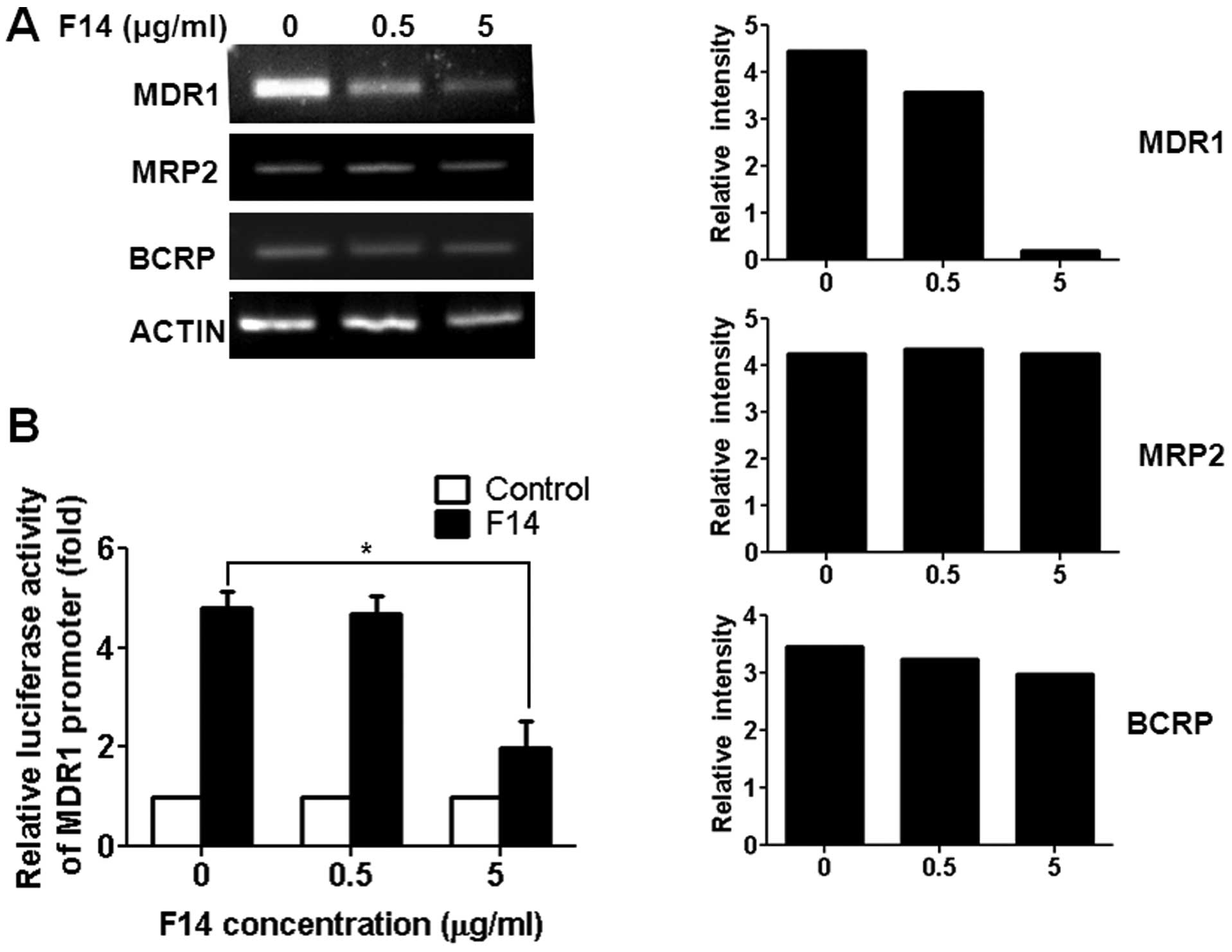
F14 extract reduces MDR1 transcript
levels and its transcriptional activity in A549RT-eto cells
Since not only the MDR1 gene but also other
genes such as MRP2 and BCRP are known to be involved
in drug resistance (14,15), we examined levels of transcripts
related to MDR1 such as the MDR1, MRP2 and
BCRP genes in A549RT-eto cells during F14 treatment. In
addition, we aimed to ascertain whether the decrease in P-gp
protein levels is regulated at the transcriptional level by F14
treatment. Cells were thus treated with the F14 extract (5 μg/ml)
for 12 h, and total RNAs were isolated. After cDNA synthesis,
MDR1, MRP2 and BCRP genes were amplified in
A549RT-eto cells treated with F14. We found that A549RT-eto
drastically reduced levels of MDR1 transcripts and slightly reduced
levels of BCRP in a dose-dependent manner, but not MRP2 (Fig. 4A). Furthermore, we examined MDR1
transcriptional activity in the cells using a MDR1-promoter
luciferase reporter vector (7). As
shown in Fig. 4B, F14 treatment
induced a drastic decrease in MDR1-mediated luciferase activity in
A549RT-eto cells (Fig. 4B),
indicating that the F14 extract reduced MDR1 transcriptional
activity. On the basis of these results, we suggest that the F14
extract inhibited MDR1 transcription, resulting in a decrease in
P-gp protein levels, which eventually sensitized A549RT-eto cells
to apoptosis.
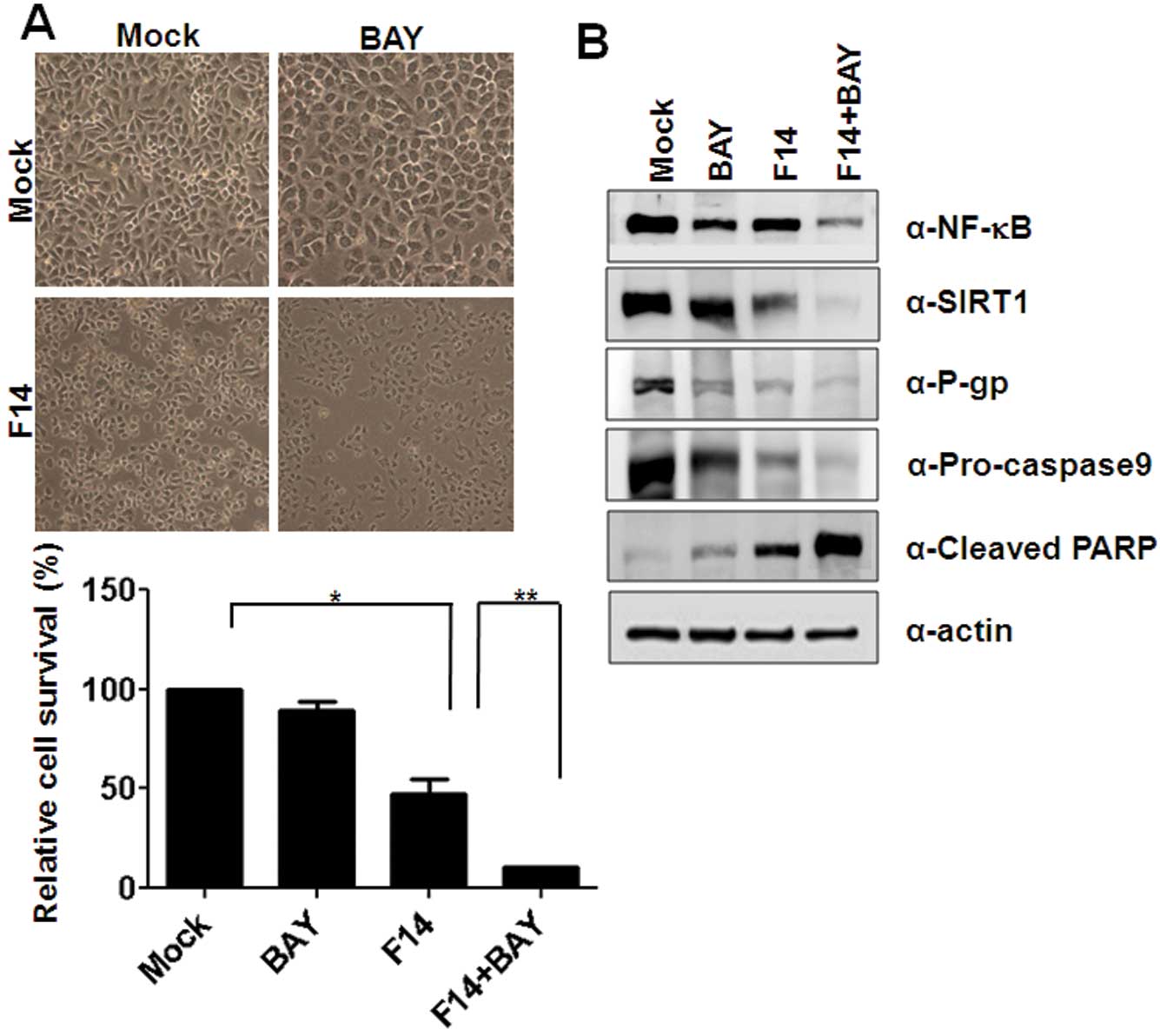
Inhibition of NF-κB sensitizes
F14-induced apoptosis of A549RT-eto cells through downregulation of
P-gp
Since we observed enhanced NF-κB protein levels and
activity in A549RT-eto cells, we explored whether NF-κB is involved
in resistance to etoposide in A549 cells through upregulation of
P-gp. We treated A549RT-eto cells with BAY11-7082 (BAY; 10 μM), an
inhibitor of NF-κB, and examined cell viability with MTT assays.
BAY treatment alone did not cause inhibition of cell growth in
A549RT-eto cells while the F14 extract (1 μg/ml) inhibited ~50%
cell growth at 24 h post-treatment (Fig. 5A). Furthermore, we found that the
combined treatment with the F14 extract and BAY accelerated F14
extract-mediated apoptosis in A549RT-eto cells (Fig. 5A), which was confirmed by
observation of cleaved PARP and pro-caspase-9 (Fig. 5B). Moreover, when we examined
protein levels of NF-κB, SIRT1 and P-gp after treatment with Bay
alone, F14 extract alone, or Bay plus F14 extract, we found that
BAY treatment alone decreased the expression levels of NF-κB and
P-gp but did not influence protein levels of SIRT1 in the cells
(Fig. 5B). F14 treatment alone
drastically diminished the protein levels of NF-κB, SIRT1 and P-gp
in the A549RT-eto cells (Figs. 3A
and 5B). We also observed that the
combined treatment more significantly reduced protein levels of
NF-κB, SIRT1 and P-gp (Fig. 5B).
These results suggest that NF-κB is involved in MDR in A549 cells
by upregulation of P-gp, resulting in resistance to etoposide.
Suppression of SIRT1 expression does not
enhance susceptibility to F14-induced apoptosis of A549RT-eto
cells
We observed the enhanced levels of SIRT1 in
A549RT-eto cells (Fig. 2A) while
F14 treatment reduced SIRT1 as well as P-gp expression levels
(Fig. 3B). We aimed to ascertain
whether downregulation of the SIRT1 protein level was attributed to
a decrease in P-gp expression, leading to F14 fraction-induced
apoptosis. Thus, we introduced SIRT1 siRNA to suppress SIRT1 levels
in A549RT-eto cells. We first optimized the SIRT1 siRNA
concentration (100 nM; data not shown). We then treated A549RT-eto
cells with F14 under suppression of SIRT1 using its siRNA and
examined cell viability with MTT assays. Importantly, we found that
suppression of SIRT1 did not accelerate F14-induced apoptosis of
A549RT-eto cells (Fig. 6A) as noted
in the combined treatment with Bay and F14 (Fig. 5). We also observed that inhibition
of SIRT1 activity with nicotinamide (NAM; 500 μM) did not
accelerate F14 extract-induced apoptosis of A549RT-eto cells
(Fig. 6C). Furthermore, neither
suppression of SIRT1 protein levels nor inhibition of SIRT1
activity significantly affected P-gp expression (Fig. 6B and D). However, F14 treatment (1
μg/ml) decreased expression of P-gp irrespective of SIRT1 protein
levels and activity (Fig. 6B and
D). These results suggest that the elevated levels of SIRT1
were not related to the upregulation of P-gp in A549RT-eto
cells.
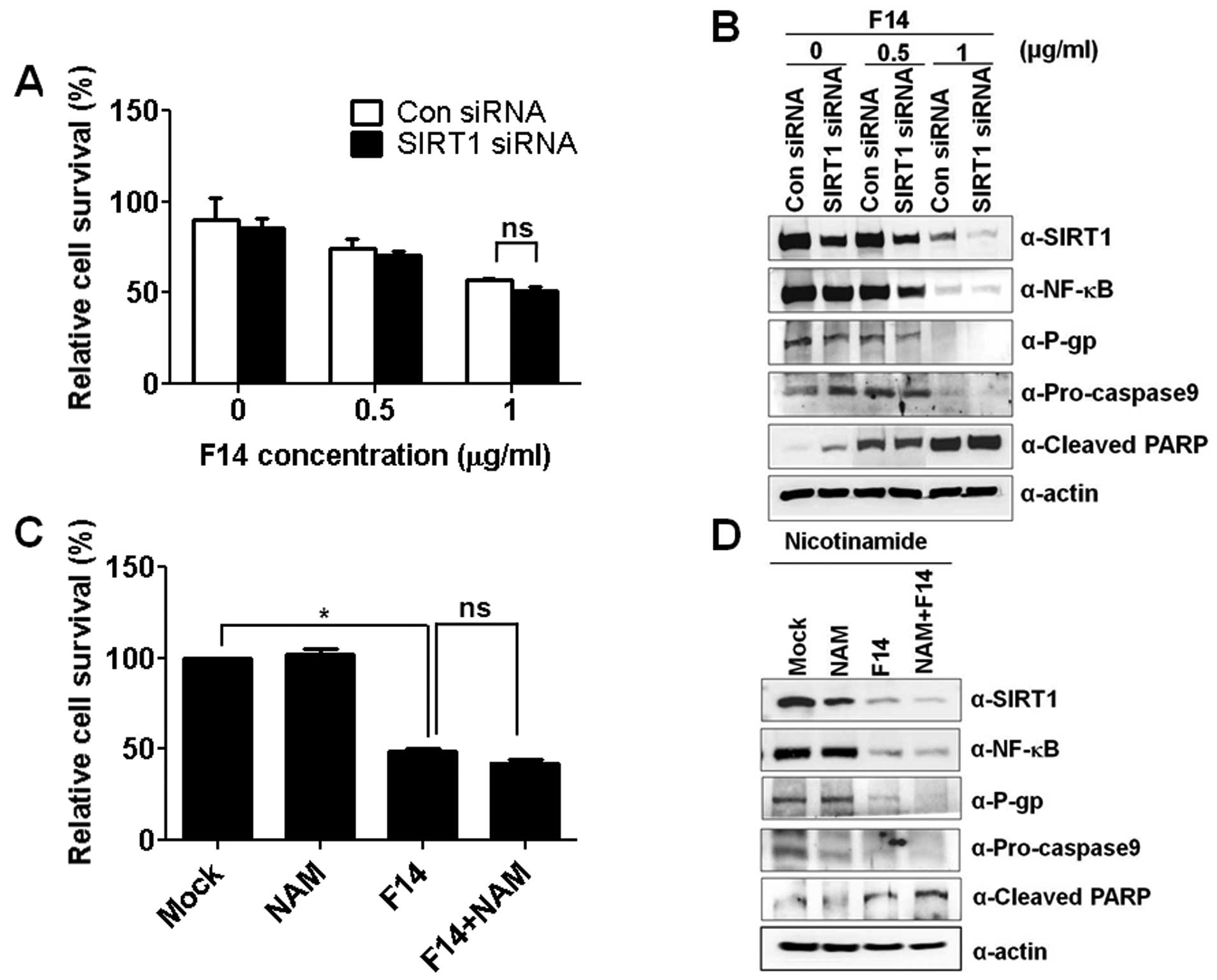 | Figure 6Neither SIRT1 suppression nor
inhibition decreases P-gp protein levels or accelerates F14-induced
apoptosis. (A) A549RT-eto cells were treated with F14 (0.5 and 1
μg/ml) alone, SIRT1 siRNA (100 nM) alone, and F14 plus SIRT siRNA
for 48 h, and cell growth was measured by MTT assay. The results
shown are the average of triplicates; bar indicates standard
deviation (ns, not significant). (C) A549RT-eto cells were treated
with F14 (1 μg/ml) alone, NAM (500 μM) alone, and F14 plus BAY for
24 h, and cell growth was measured by MTT assay. The results shown
are the average of triplicates; bar indicates standard deviation
(*P<0.05; ns, not significant). (B and D) Cell
lysates from the treated A549RT-eto cells (in A and C,
respectively) were prepared and separated on a 12% SDS-PAGE gel.
The expression of pro-caspase-9 and PARP protein were measured as
indicators of apoptosis by immunoblotting, and protein levels of
NF-κB, SIRT1 and P-gp were compared by immunoblotting with the
corresponding antibodies. |
Discussion
Medicinal plants are important sources for the
potential development of effective anticancer agents (16). In fact, more than half of the
current anticancer drugs were originally synthesized from natural
products and their derivatives. In the present study, we found that
a purified fraction (F14) from Bryophyllum laetivirens
exhibits significant antiproliferative effects against
multidrug-resistant A549 cancer cells. A majority of the effective
anticancer concentrations of plant extracts are >100 μg/ml
(16,17); however, in the present study, cell
proliferation was inhibited by treatment with 6.72 μg/ml and the
viability was <50% after a 12-h exposure (Fig. 1 and data not shown). Therefore, the
F14 fraction may include a highly effective candidate compound for
further study as a future anticancer drug. Another study revealed
that methanol extracts of Kalanchoe (Bryophyllum) hybrid
exhibited cytotoxicity toward MCF-7, NCI-H460 and SF-268 tumor cell
lines (18). Moreover, kalanchoside
compounds from Kalanchoe (Bryophyllum) gracilis also
exhibited significant cytotoxic activity against gastric and
nasopharyngeal carcinoma cell lines (19). However, these studies did not
provide detailed mechanisms by which the extracts or compounds from
Bryophyllum species induce cytotoxicity in various tumor
cell lines.
Here, we demonstrated the mechanism by which the F14
fraction from Bryophyllum laetivirens sensitized human lung
A549 cells that are resistant to etoposide-induced apoptosis, by
reversing their MDR phenotype. It is known that several proteins
including Ras, Sp1, p53, PKC and NF-κB are involved in the
regulation of P-gp expression at the transcriptional level
(20). We herein report that the
F14 extract not only diminishes NF-κB protein levels but also
blocks translocation of NF-κB into the nucleus and binding to its
binding sites, leading to a decrease in MDR1 transcription levels.
Furthermore, the combined treatment with the F14 fraction and
BAY-11-7082 (an irreversible NF-κB-specific inhibitor) accelerated
apoptosis, suggesting that NF-κB has a crucial role in MDR and
resistance to apoptosis. In addition, since our previous research
showed that Stat1 and HDAC4 also play a crucial role in P-gp
expression (unpublished data), we examined whether the F14 fraction
reduces expression levels of Stat1 and HDAC4 protein. We found that
the F14 fraction decreased protein levels of Stat1 and HDAC4
(unpublished data). Furthermore, we observed that the F14 fraction
sensitized etoposide-induced apoptosis in A549RT-eto cells
(unpublished data). Since we observed that the F14 extract targets
multiple proteins including NF-κB, Stat1 and HDAC4, we undertook a
search for the detailed mechanism of action of the F14
fraction.
SIRT1 is a NAD+-dependent deacetylase
which deacetylates histones and non-histone proteins and has been
involved in various biological responses including aging,
metabolism and cancer (21,22). Since previous studies have shown
that overexpression of SIRT1 induces the expression of P-gp and
leads to resistance to chemotherapy in tumor cells (8,23), we
speculated that the enhanced SIRT1 expression in the A549RT-eto
cells might also be involved in MDR in this case. We then found
that treatment with the F14 fraction reduced SIRT1 protein levels
and its activity. However, we could not determine from this result
whether SIRT1 itself modulates P-gp expression levels as F14 also
reduces NF-κB expression and its activity which is involved in
regulating transcription of P-gp. To resolve this issue, we
specifically suppressed SIRT1 protein levels by siRNA or inhibited
SIRT1 activity with NAM in the absence of F14 extracts. We found
that neither suppression of SIRT1 expression nor its activity
caused a decrease in P-gp protein levels, suggesting that SIRT1
itself is not directly involved in the regulation of P-gp
expression in A549RT-eto cells. Moreover, combined treatment with
the F14 fraction and SIRT1 siRNA, or NAM did not accelerate
apoptosis of A549RT-eto cells when compared to the F14 fraction
alone. When we consider the results of other studies, we suggest
that the observation of a possible SIRT1 role in MDR might be
attributed to dual functions of SIRT1 on oncogene and tumor
suppressor activity, depending on the cellular contexts and
subcellular localization of SIRT1 (21,22).
Thus, the possible role of enhanced SIRT1 protein levels in
A549RT-eto cells requires further investigation.
In summary, the present study showed that F14, a
methanol extract from B. laetivirens, reverses etoposide
resistance in A549 lung cancer cells though downregulation of
NF-κB, leading to decreased transcription and expression of P-gp,
which eventually promotes F14 extract-mediated apoptosis. Since we
herein provide important evidence for the development of a novel
anticancer therapeutic drug against MDR cancer, in subsequent
studies we will further purify B. laetivirens leaf extracts
in order to obtain a single compound for use as a new and
efficacious anticancer drug.
Acknowledgements
The present study was supported by a grant from the
World Class University Program (R31-2008-000-20004-0) through the
National Research Foundation funded by the Korean government. It
was also supported by the Office of the Higher Education
Commission, Thailand, under the Strategic Scholarships Fellowships
Frontier Research Networks (specific for the Southern region) for
the Join PhD Program Thai Doctoral degree program; a CHE-SSR-Ph.D.
SW Scholarship to C.K.
References
|
1
|
Biedler JL: Drug resistance: genotype
versus phenotype - thirty-second G. H. A. Clowes Memorial Award
Lecture. Cancer Res. 54:666–678. 1994.PubMed/NCBI
|
|
2
|
Bosch I and Croop J: P-glycoprotein
multidrug resistance and cancer. Biochim Biophys Acta. 9:37–54.
1996.
|
|
3
|
Goldstein LJ, Galski H, Fojo A, et al:
Expression of a multidrug resistance gene in human cancers. J Natl
Cancer Inst. 81:116–124. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gottesman MM, Fojo T and Bates SE:
Multidrug resistance in cancer: role of ATP-dependent transporters.
Nat Rev Cancer. 2:48–58. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Friedrich K, Wieder T, Von Haefen C, et
al: Overexpression of caspase-3 restores sensitivity for
drug-induced apoptosis in breast cancer cell lines with acquired
drug resistance. Oncogene. 20:2749–2760. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ruefli AA, Tainton KM, Darcy PK, Smyth MJ
and Johnstone RW: P-glycoprotein inhibits caspase-8 activation but
not formation of the death inducing signal complex (disc) following
Fas ligation. Cell Death Differ. 9:1266–1272. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bentires-Alj M, Barbu V, Fillet M, et al:
NF-κB transcription factor induces drug resistance through
MDR1 expression in cancer cells. Oncogene. 22:90–97.
2003.
|
|
8
|
Chu F, Chou PM, Zheng X, Mirkin BL and
Rebbaa A: Control of multidrug resistance gene mdr1 and
cancer resistance to chemotherapy by the longevity gene
sirt1. Cancer Res. 65:10183–10187. 2005.PubMed/NCBI
|
|
9
|
Tan G, Gyllenhaal C and Soejarto DD:
Biodiversity as a source of anticancer drugs. Curr Drug Targets.
7:265–277. 2006.PubMed/NCBI
|
|
10
|
Mahata S, Maru S, Shukla S, et al:
Anticancer property of Bryophyllum pinnata(Lam.) Oken. leaf
on human cervical cancer cells. BMC Complement Altern Med.
12:152012. View Article : Google Scholar
|
|
11
|
Jain Vineet C, Patel Natvarlal M, Shah
Dhiren P, Patel Paras K and Joshi Bhavesh H: Antioxidant and
antimicrobial activities of Bryophyllum Calycinum salisb
leaf. Pharmacologyonline. 1:393–405. 2010.
|
|
12
|
Kanintronkul Y, Worayuthakarn R, Thasana
N, et al: Overcoming multidrug resistance in human lung cancer with
novel benzo[a]quinolizin-4-ones. Anticancer Res. 31:921–928.
2011.PubMed/NCBI
|
|
13
|
Kaewpiboon C, Lirdprapamongkol K,
Srisomsap C, et al: Studies of the in vitro cytotoxic,
antioxidant, lipase inhibitory and antimicrobial activities of
selected Thai medicinal plants. BMC Complement Altern Med.
12:2172012.
|
|
14
|
Sugimoto Y, Tsukahara S, Ishikawa E and
Mitsuhashi J: Breast cancer resistance protein: molecular target
for anticancer drug resistance and
pharmacokinetics/pharmacodynamics. Cancer Sci. 96:457–465. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Young LC, Campling BG, Cole SPC, Deeley RG
and Gerlach JH: Multidrug resistance proteins MRP3, MRP1, and MRP2
in lung cancer: correlation of protein levels with drug response
and messenger RNA levels. Clin Cancer Res. 7:1798–1804.
2001.PubMed/NCBI
|
|
16
|
Tang Y-Q, Jaganath IB and Sekaran SD:
Phyllanthus spp. induces selective growth inhibition of PC-3
and MeWo human cancer cells through modulation of cell cycle and
induction of apoptosis. PLoS One. 5:e126442010. View Article : Google Scholar
|
|
17
|
Ganguly A, Yang H and Cabral F:
Paclitaxel-dependent cell lines reveal a novel drug activity. Mol
Cancer Ther. 9:2914–2923. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kuo PC, Kuo TH, Su CR, Liou MJ and Wu TS:
Cytotoxic principles and α-pyrone ring-opening derivatives of
bufadienolides from Kalanchoe hybrida. Tetrahedron.
64:3392–3396. 2008.
|
|
19
|
Wu PL, Hsu YL, Wu TS, Bastow KF and Lee
KH: Kalanchosides A-C, new cytotoxic bufadienolides from the aerial
parts of Kalanchoe gracilis. Org Lett. 8:5207–5210. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sun J, Yeung CA, Co NN, et al: Clitocine
reversal of P-glycoprotein associated multi-drug resistance through
down-regulation of transcription factor NF-κB in R-HepG2 cell line.
PLoS One. 7:222012.PubMed/NCBI
|
|
21
|
Song NY and Surh YJ: Janus-faced role of
SIRT1 in tumorigenesis. Ann NY Acad Sci. 1271:10–19. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yi J and Luo J: SIRT1 and p53, effect on
cancer, senescence and beyond. Biochim Biophys Acta.
1804:1684–1689. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang Z and Chen W: Emerging roles of SIRT1
in cancer drug resistance. Genes Cancer. 4:82–90. 2013. View Article : Google Scholar : PubMed/NCBI
|