Introduction
Colorectal cancer is one of the common causes of
cancer-related deaths worldwide. Although recent advances in early
diagnosis and treatment have improved the clinical outcome in
patients with colorectal cancer, recurrence and metastasis are
still common (1–3). Therefore, a greater understanding of
the molecular mechanisms underlying tumor recurrence and metastasis
has proven useful in the management of colorectal cancer (4).
In patients with various types of cancers, cancer
cells in primary tumors disseminate to distant organs, through the
blood and lymphatic circulatory systems by metastasis (5,6).
Dysregulated angiogenesis and lymphangiogenesis occur in a variety
of pathological settings including growth and dissemination of
tumors (7–11). Several lines of evidence indicate
that progression of colorectal cancer depends on angiogenesis and
lymphangiogenesis (12–14).
The chemosensitivity of circulating cancer cells is
suggested to determine chemotherapeutic response in cancer
patients. Chemoresistance enables cancer cells to evade apoptotic
stimuli and leads to poor clinical prognosis (15–19).
Recent studies showed a correlation between poor treatment outcome
and development of chemoresistance in advanced colorectal cancer
patients (20–22).
Recepteur d'Origine Nantais (RON) is a member of the
c-MET protooncogene family of receptor tyrosine kinases (RTKs) that
has been implicated in regulating invasive growth phenotypes in a
variety of cancers (23–29). Specifically, activation of RON
mediates several biological events including cell growth, motility,
angiogenesis, epithelial-to-mesenchymal transition and drug
resistance in cancer cells (23–29).
Overexpression of RON has been reported in various cancer tissues
including that of the colon (30–37).
Furthermore, its overexpression correlates with aggressive disease
and poor prognosis in patients with colorectal, gastric or breast
cancers (30–33). Therefore, activation of RON may be
critical to the development and progression of cancers. However,
the impact of RON on chemosensitivity, angiogenesis and
lymphangiogenesis in colorectal cancer is yet to be elucidated.
The aims of the present study were to evaluate
whether RON affects chemosensitivity and oncogenic behavior of
colorectal cancer cells, and delineate the relationship between its
expression and various clinicopathological parameters in colorectal
cancer.
Materials and methods
Patients and tissue sample
collection
Colorectal cancer tissues were obtained from 88
patients who consecutively underwent surgery for colorectal cancer
at the Chonnam National University Hwasun Hospital between January
and December 2006. Pathological studies and clinical histories
recorded at the time of surgery were reviewed. Tissue blocks that
clearly showed the junction between normal colon epithelium and
tumoral region were selected by microscopic analyses of their
original pathologic slides. The tumors were staged at the time of
surgery using standard protocol for tumor-node-metastasis (TNM)
staging given by the American Joint Committee on Cancer. Survival
was tracked from the time of surgery until follow-up on December
31, 2014.
Cell culture and siRNA transfection
Human colorectal cancer cell line, DLD1 was obtained
from the American Type Culture Collection Line Inc., and were
maintained in Dulbecco's modified Eagle's medium (DMEM)
supplemented with 10% fetal bovine serum (both from HyClone, Logan,
UT, USA). Human umbilical vein endothelial cells (HUVECs; Lonza,
Walkersville, MD, USA) and human lymphatic endothelial cells
(HLECs; ScienCell, San Diego, CA, USA) were maintained in
EGM®-2 MV SingleQuots® (Lonza). The
RON-specific and scrambled small interfering RNA (siRNA) duplexes
were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA)
and Qiagen (Germantown, MD, USA), respectively. The siRNAs were
transfected using Lipofectamine™ RNAiMAX (Invitrogen, Carlsbad, CA,
USA) according to the manufacturer's instructions. To obtain the
conditioned medium (CM), transfected cells were incubated in
serum-free medium for 24 h.
5-Fluorouracil (FU) treatment
DLD1 cells were seeded onto 6-well plates at
densities of 3×105 cells/well, and transfected with
either RON-specific or scramble siRNA. Following incubation for 24
h, 5-FU (Julian®; Dongkook, Korea) was added at
concentrations ranging from 0 to 80 µg/ml and incubated for
24 h.
Cell proliferation assay
Cell proliferation was determined using the EZ-CyTox
(tetrazolium salts, WST-1) cell viability assay kit (Daeil Lab
Inc., Seoul, South Korea). Transfected DLD1 cells were treated with
5-FU at concentrations ranging from 0 to 80 µg/ml, incubated
for 24 h and treated with the WST-1 reagent. This was followed by
incubation for 1 h at 37°C. The absorbance was measured using a
microplate reader (Infinite M200) equipped with Magellan V6 data
analysis software (both from Tecan, Austria GmbH, Austria).
Apoptosis analysis
Transfected cells treated with 5-FU were collected
using trypsin and resuspended in binding buffer (BD Biosciences,
San Diego, CA, USA). The cell suspensions were then incubated with
APC Annexin V and 7-amino-actinomycin D (7-AAD; BD Biosciences).
The cells were gently vortexed and incubated in dark for 20 min at
room temperature. The population of Annexin V-positive cells were
analyzed using BD Cell Quest® version 3.3
(Becton-Dickinson, San Jose, CA, USA) and WinMDI version 2.9 (The
Scripps Research Institute, San Diego, CA, USA).
Cell cycle analysis
Transfected cells treated with 5-FU were fixed in
70% ethanol and washed with phosphate-buffered saline (PBS). Next,
the cells were incubated with 100 µg/ml each of ribonuclease
A and propidium iodide (both from Sigma-Aldrich, St. Louis, MO,
USA) at room temperature in the dark. The cell cycle profiles were
analyzed using BD CellQuest® version 3.3 and WinMDI
version 2.9.
Transwell invasion assay
Invasion assays were performed using Transwell
filter chambers (8.0-µm pore size; Costar, Cambridge, MA,
USA) with gelatin coating. The transfected, 5-FU-treated cells were
seeded at densities of 2×105, with 0.2% bovine serum
albumin (BSA) medium in the upper chambers. The lower chambers were
filled with 0.2% BSA medium containing fibronectin (10
µg/ml). After 24 h of incubation, invading cells on the
lower surface of the upper chamber were fixed with 70% ethanol and
stained with Diff-Quik solution (Sysmex, Kobe, Japan) following the
manufacturer's protocol. The stained cells were enumerated in five
randomly selected fields under a light microscope.
Cell migration assay
Transfected, 5-FU-treated cells were seeded at
densities of 1×105 culture inserts (2×0.22
cm2; Ibidi, Regensburg, Germany). Following incubation
for 24 h, the inserts were gently removed using sterile tweezers.
Cells migrated into the scratch were photographed at 0, 3, 6, 12
and 24 h time-points using an inverted microscope. Distance between
the gaps was normalized to 1 cm after capture of three random
sites.
Western blotting
Total proteins extracted with M-PER®
mammalian protein extraction reagent (Thermo, Rockford, IL, USA)
were resolved by sodium dodecyl sulfate-polyacrylamide gel
electrophoresis and transferred onto PVDF membranes (Millipore,
Billerica, MA, USA). Specific proteins were blotted with respective
primary antibodies, which are as follows: anti-RON,
anti-cyclooxygenase-2 (COX-2), anti-vascular endothelial growth
factor (VEGF)-A, -C and -D, anti-GAPDH, and anti-β-tubulin (Santa
Cruz Biotechnology). Antibodies against cleaved and intact forms of
caspase-3 and -9, and poly(ADP-ribose) polymerase (PARP), Bcl-xL,
Bax, Bim, cyclin B1, D1 and D3, cyclin-dependent kinase (CDK) 2, 4
and 6, p21, p27, hypoxia-inducible factor-1α (HIF-1α), and total
and phospho-β-catenin (Ser33/37/Thr41) were purchased from Cell
Signaling Technology (Danvers, MA, USA). Antibodies against
angiostatin and endostatin were purchased from Abcam (Cambridge,
UK). The target proteins were detected using the enhanced
chemiluminescence detection system horseradish peroxidase (HRP)
substrate (Millipore) and luminescent image analyzer LAS 4000
(Fujifilm, Tokyo, Japan).
In vitro tube formation assay
Matrigel basement membrane matrix (BD Biosciences)
was used to coat culture plates according to the manufacturer's
instructions. Approximately 50 µl of thawed Matrigel was
applied to each well of a 96-well plate and left to polymerize at
37°C for 1 h. Transfected HUVECs and HLECs (2×104
cells/well) in 100 µl CM were seeded on the substratum of
the Matrigel, and incubated for 24 h at 37°C and 5% CO2.
Next, the tube-like structures were photographed using an inverted
microscope. The tube lengths were measured using the WimTube image
analysis platform (Wimasis GmbH, Munich, Germany) on the Ibidi
website (www.ibidi.com).
Matrigel invasion assay
Transwell filters (8.0-µm pore size) were
coated with 1 mg/ml of Matrigel, and dried at room temperature.
Either HUVECs or HLECs were resuspended in 120 µl
EGM®-2 MV Single Quotes® medium, and
inoculated into the upper chambers of the Matrigel-coated
Transwells. The lower chambers were filled with CM from transfected
cells. After incubation for 3 h, cells on the Transwell were
stained with Diff-Quik and non-invading cells on the upper surface
of the Transwell were wiped off with a cotton swab. The numbers of
invaded cells were enumerated in five randomly selected fields
under a light microscope. Data from three independent experiments
are expressed as the mean ± standard deviation (SD) of the number
of cells/field.
Immunohistochemistry
Paraffin-embedded tissue sections were
deparaffinized, rehydrated and retrieved. To block the endogenous
peroxidase activity, tissues were treated with peroxidase-blocking
solution (Dako, Carpinteria, CA, USA); and incubated overnight with
anti-RON, anti-D2–40 (Dako), and anti-CD34 (Abcam) antibodies in
primary diluent solution (Invitrogen) at 4°C. After washing in
Tris-buffered saline Tween-20, the tissues were stained using the
Dako Real™ EnVision HRP/DAB detection system and counterstained
with hematoxylin. The stained tissues were scored and photographed
using a light microscope.
Determination of RON expression
The immunostained specimens were examined by two
dependent observers, unaware of the clinicopathological data. The
staining intensities in cancer cells were graded as follows: 0, no
staining; 1, weak; 2, moderate; and 3, strong. The percentages of
stained cancer cells were also graded as follows: 0, none; 1,
<10%; 2, 10–50%; and 3, >50%. The intensity score was
multiplied by that of the percent staining to obtain an overall
score. The mean overall score for the 88 tumors analyzed was 6.0,
which was selected as the cut-off for discriminating between RON
expression levels. Specimens with scores >6 and ≤6 were
considered RON-positive and -negative, respectively.
Assessment of apoptosis in tissue
samples
In order to detect and quantify apoptosis, the
DeadEnd™ Colorimetric terminal deoxynucleotidyl transferase UTP
nick-end labeling (TUNEL) system (Promega, Madison, WI, USA) was
used according to the manufacturer's instructions. The number of
positively stained cells with apoptotic morphology was enumerated
in five random fields/sample. The apoptotic index (AI) was
expressed as the number of positively stained nuclei, including
apoptotic bodies among 1,000 tumor cell nuclei.
Determination of microvessel (MVD) and
lymphatic vessel (LVD) densities
Microvessels and lymphatic vessels were detected
using anti-CD34 and anti-D2–40 antibodies. MVD and LVD were
quantified based on an international consensus. The stained
sections were visualized at low magnification (×40) to identify the
areas with high vascular density (hot spots) within the peritumoral
and intratumoral regions. These high neovascular areas could occur
anywhere within the tumor, but were most frequent at the margins of
the carcinoma. Vessels in five areas with hot spots were enumerated
at high magnification (×200). MVD and LVD were expressed as the
mean number of vessels in these areas.
Statistical analyses
For intergroup comparisons, the Student's t-test was
used to determine statistical significance, where data are
presented as the mean ± SD. Correlations between various
clinicopathological parameters and RON expression were performed
using the χ2 and Fisher exact tests. The relationship of
RON expression with AI, MVD or LVD was also evaluated by the
Student's t-test. Actuarial survival rates of patients with either
positive or negative RON expression were evaluated according to the
Kaplan-Meier method, and the differences were tested with a
log-rank test. The Cox regression model was used to determine the
prognostic significance of each parameter by a multivariate
analysis. The statistical analysis was performed with Statistical
Package for the Social Sciences (SPSS/PC+ 15.0; SPSS, Inc.,
Chicago, IL, USA). P-values <0.05 were considered statistically
significant.
Results
Effect of RON on chemosensitivity of
colorectal cancer cell lines
In order to investigate the function of RON in the
oncogenic behavior of colorectal cancer cells, we used RON-specific
siRNAs to silence its endogenous gene expression in DLD1 cells.
5-FU is known to induce apoptosis and affect the cell cycle of
these colorectal cancer cells (20–22).
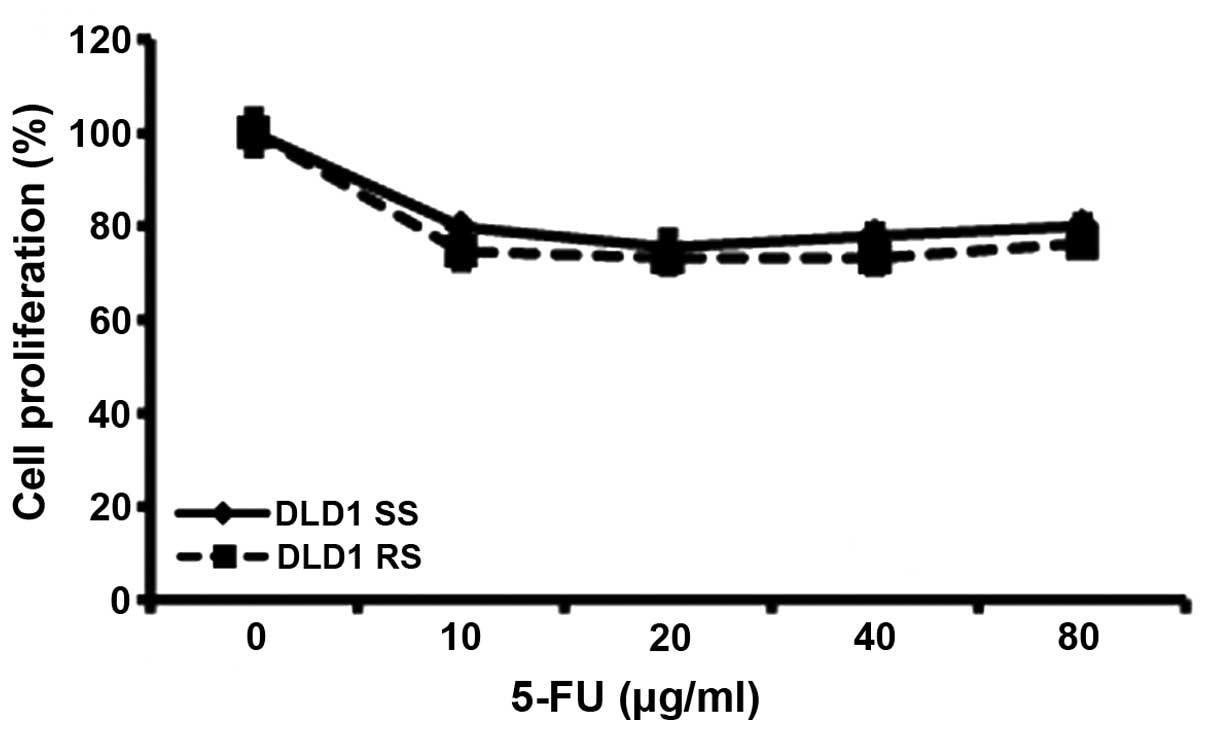
Cell viability assays revealed that RON knockdown did not enhance
5-FU-induced death in DLD1 cells (Fig.
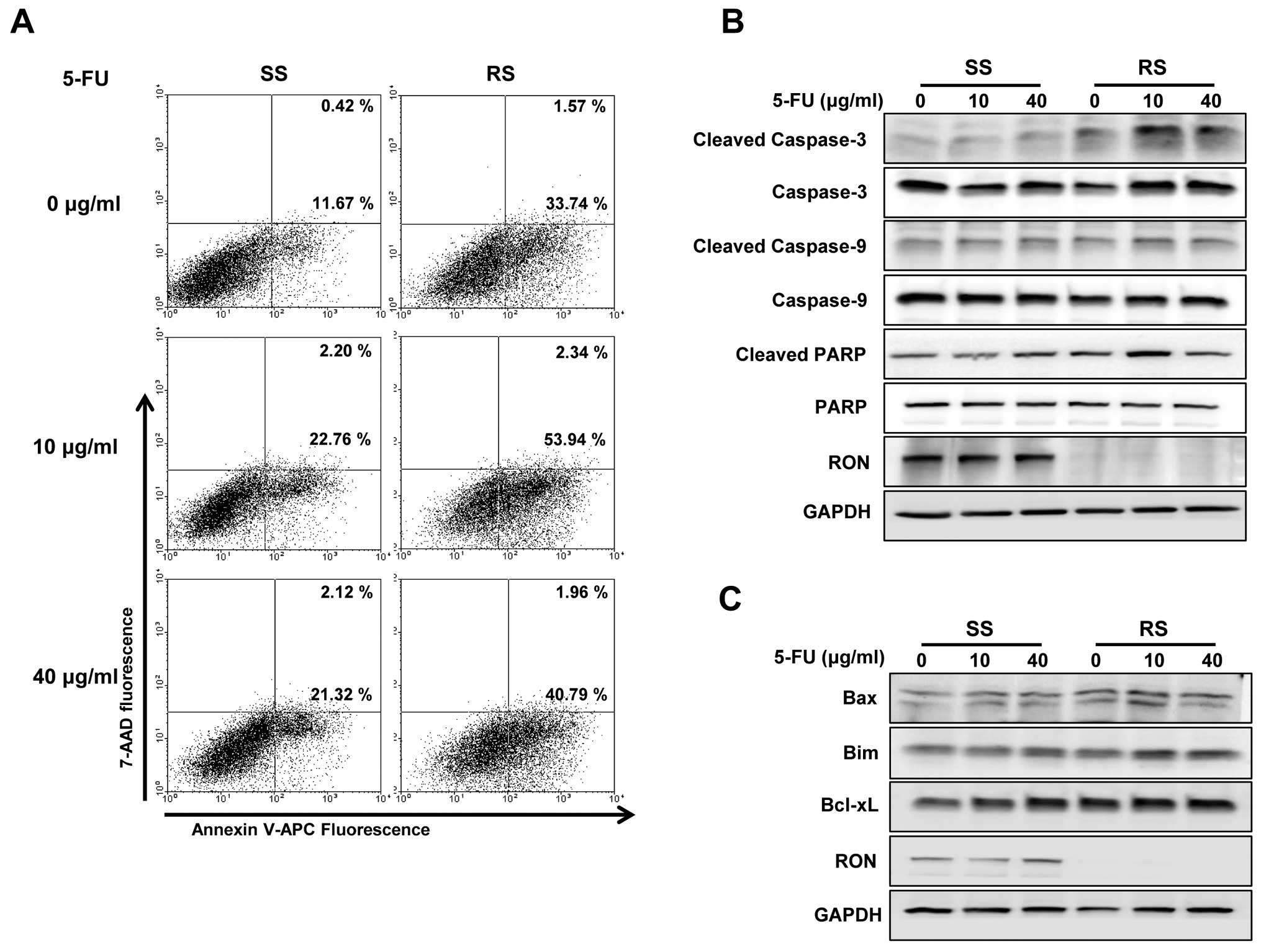
1). We next performed flow cytometric analyses to evaluate the
impact of RON on apoptosis and cell cycle distribution. The
apoptotic rate of DLD1 cells upon RON knockdown significantly
increased, when compared with that in presence of scramble siRNAs
(11.67 vs. 33.74%). It also augmented the 5-FU-induced apoptosis
compared with that upon scramble siRNA treatment in DLD1 cells
(22.76 and 21.32 vs. 53.94 and 40.79%, respectively) (Fig. 2A). We further investigated caspase
activities upon RON knockdown in these cell lines. Cleaved
caspase-3 and PARP levels were upregulated in DLD1 cells following
RON knockdown and 5-FU treatment (Fig.
2B). Fig. 2C illustrates that
Bax and Bim protein levels increased in RON knocked down DLD1
cells. However, Bcl-xL level remained unaltered upon RON knockdown
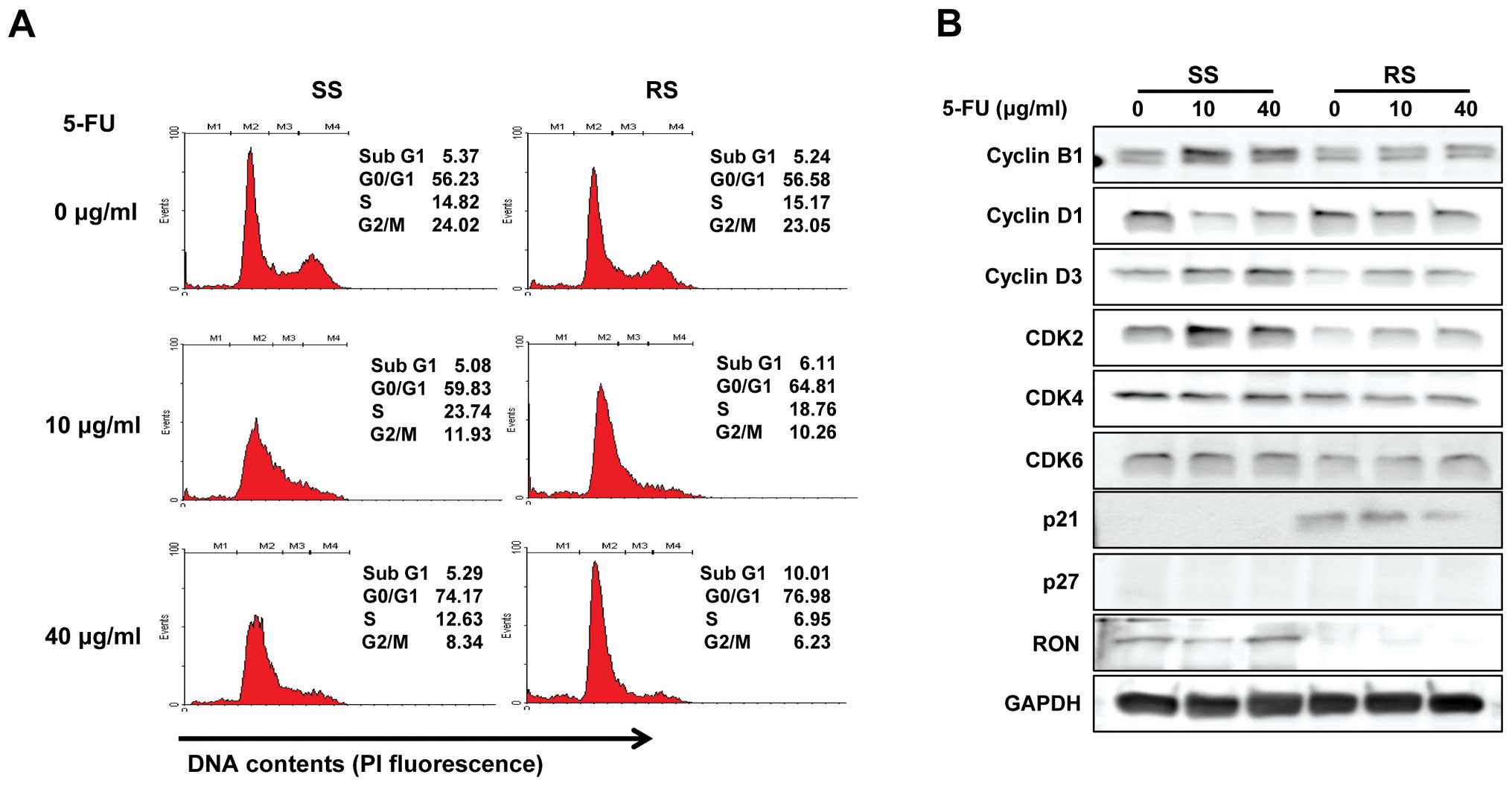
along with 5-FU treatment. However, this treatment arrested DLD1
cells in the subG1 and G0/G1 phases (Fig. 3A). Next, we evaluated the effect of
RON knockdown on various cell cycle regulatory proteins. As
Fig. 3B shows, cyclin B1 and D3,
and CDK2 and 6 were significantly downregulated at the protein
level. Moreover, the CDK inhibitor p21 was upregulated in the
presence of RON-specific siRNAs and 5-FU in DLD1 cells. To evaluate
whether RON affects the oncogenic behavior of colorectal cancer
cells, migration and invasion assays were performed. The number of
invading cells decreased in DLD1 cells treated with RON-specific
siRNAs, with 10 µg/ml 5-FU compared with scramble
siRNA-treated controls (P=0.263, 0.033 and 0.068, respectively;
Fig. 4A). The artificial wound gap
was also significantly broader on plates containing RON knocked
down DLD1 cells, with 10 µg/ml 5-FU than that on control
plates (P=0.218, 0.020 and 0.137, respectively; Fig. 4B).
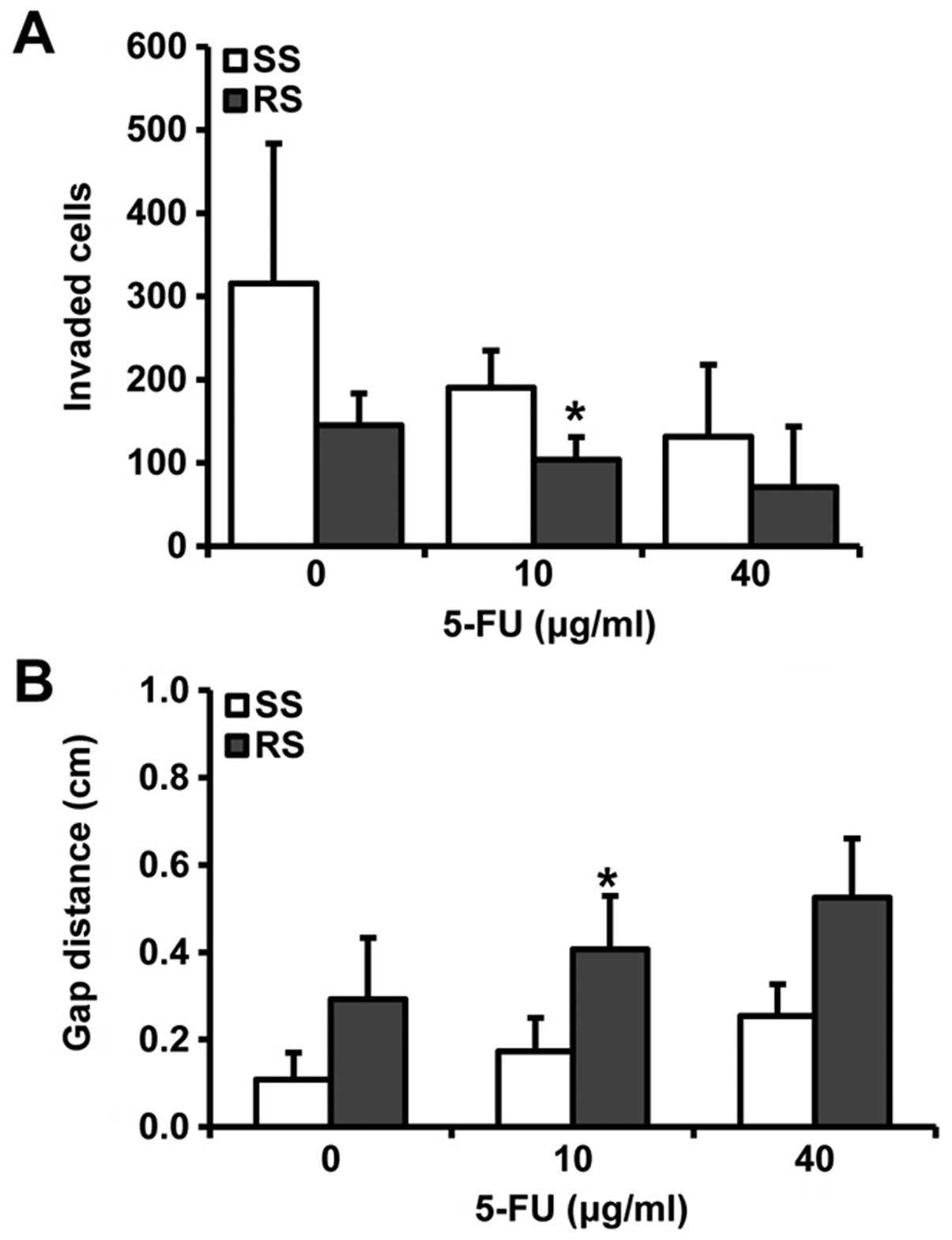 | Figure 4Effect of RON knockdown on invasion
and migration of colorectal cancer cells treated with
5-fluorouracil (FU) (0, 10 or 40 µg/ml). (A) The number of
invading cells decreased in DLD1 cells treated with RON-specific
siRNAs, with 10 µg/ml 5-FU compared with scramble
siRNA-treated controls (P=0.263, 0.033 and 0.068, respectively).
(B) The artificial wound gap was also significantly broader on
plates containing RON knocked down DLD1 cells, with 10 µg/ml
5-FU than that on control plates (P=0.218, 0.020 and 0.137,
respectively). Graphs for cell migration are represented as
relative healing distances (*P<0.05). RON, Recepteur
d'Origine Nantais; SS, scramble siRNA; RS, RON-specific siRNA. |
Effect of RON on the oncogenic signaling
pathway in colorectal cancer cells
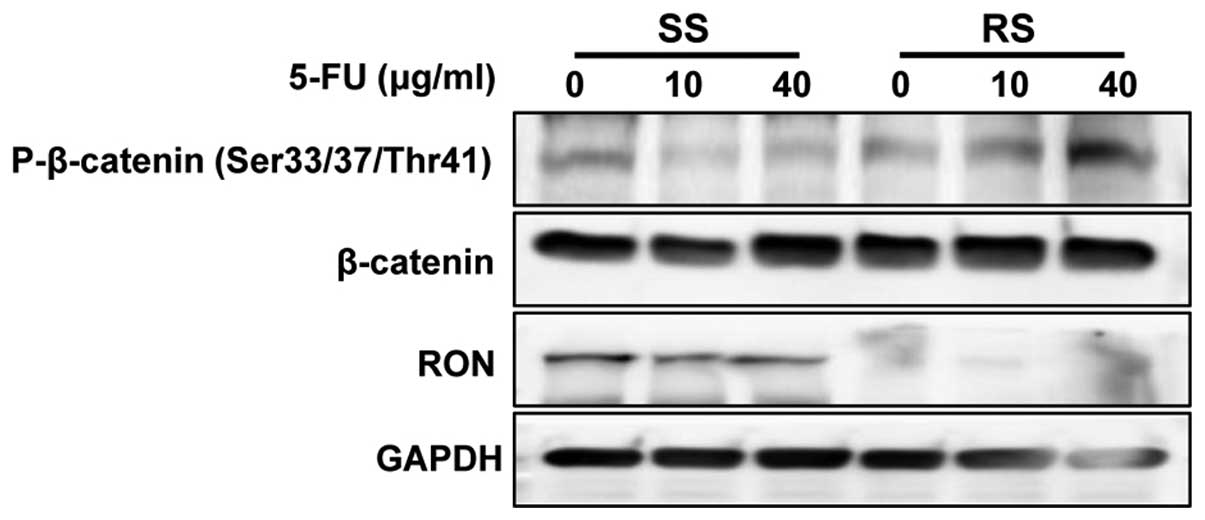
To examine whether RON activates intracellular
signaling pathways in colorectal cancer cells, we measured the
levels of phosphorylated β-catenin signaling proteins by western
blotting. The phosphorylation level of β-catenin was increased by
RON knockdown along with 5-FU treatment in DLD1 cells (Fig. 5).
Effect of RON on angiogenesis in
colorectal cancer cells
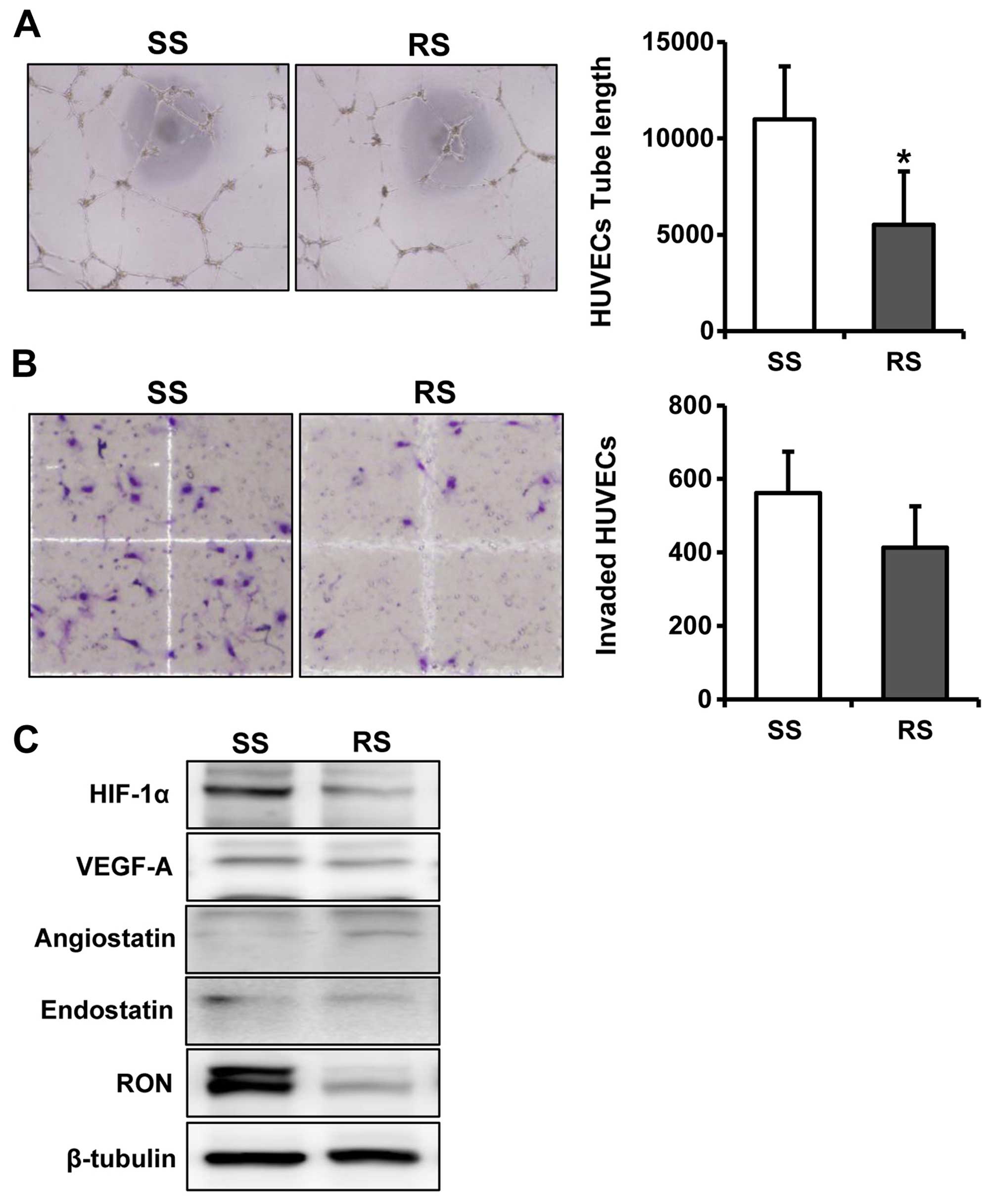
We determined whether conditioned media (CM) from
RON and scramble siRNA-transfected colorectal cancer cells affect
angiogenesis in HUVECs by performing Matrigel invasion and
endothelial tube formation assays. CM from RON knocked down DLD1
cells significantly inhibited endothelial tube formation compared
with that from the respective mock-treated control cells (P=0.045;
Fig. 6A). However, CM from RON
knocked down DLD1 cells did not inhibit invasion of HUVECs as
compared to the controls (P=0.091; Fig.
6B). The RON knockdown also decreased the expression of
angiogenic inducers VEGF-A and HIF-1α, and increased that of the
angiogenic inhibitor angiostatin in DLD1 cells (Fig. 6C). Collectively, these results
suggest that RON is associated with angiogenesis and
neovascularization in colorectal cancer cells.
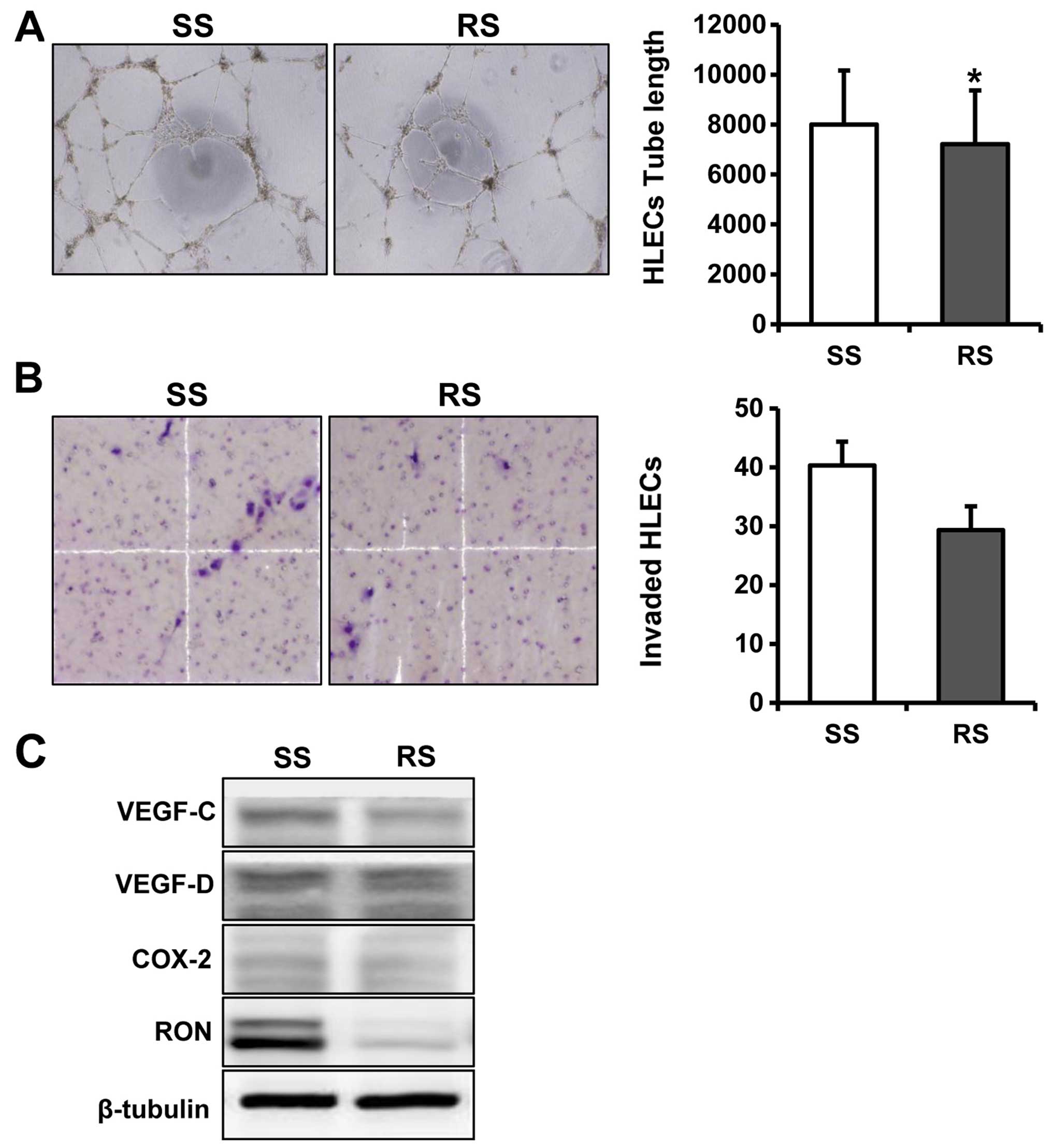
Effect of RON on lymphangiogenesis in
colorectal cancer cells
To examine the effects of RON on lymphangiogenesis
in HLECs, we performed Matrigel invasion and tube formation assays
using CM from RON and scramble siRNA-treated DLD1 cells. CM from
RON knocked down DLD1 cells significantly inhibited lymphatic
endothelial tube formation compared with that from the control
cells (P=0.031; Fig. 7A). However,
CM from RON knocked down DLD1 cells failed to inhibit invasion of
HLECs when compared with that from the respective controls
(P=0.325; Fig. 7B). RON knockdown
also suppressed expression of the lymphangiogenic inducers VEGF-C
and COX-2 in DLD1 cells (Fig.
7C).
Correlation between RON and
clinicopathological features of colorectal cancer
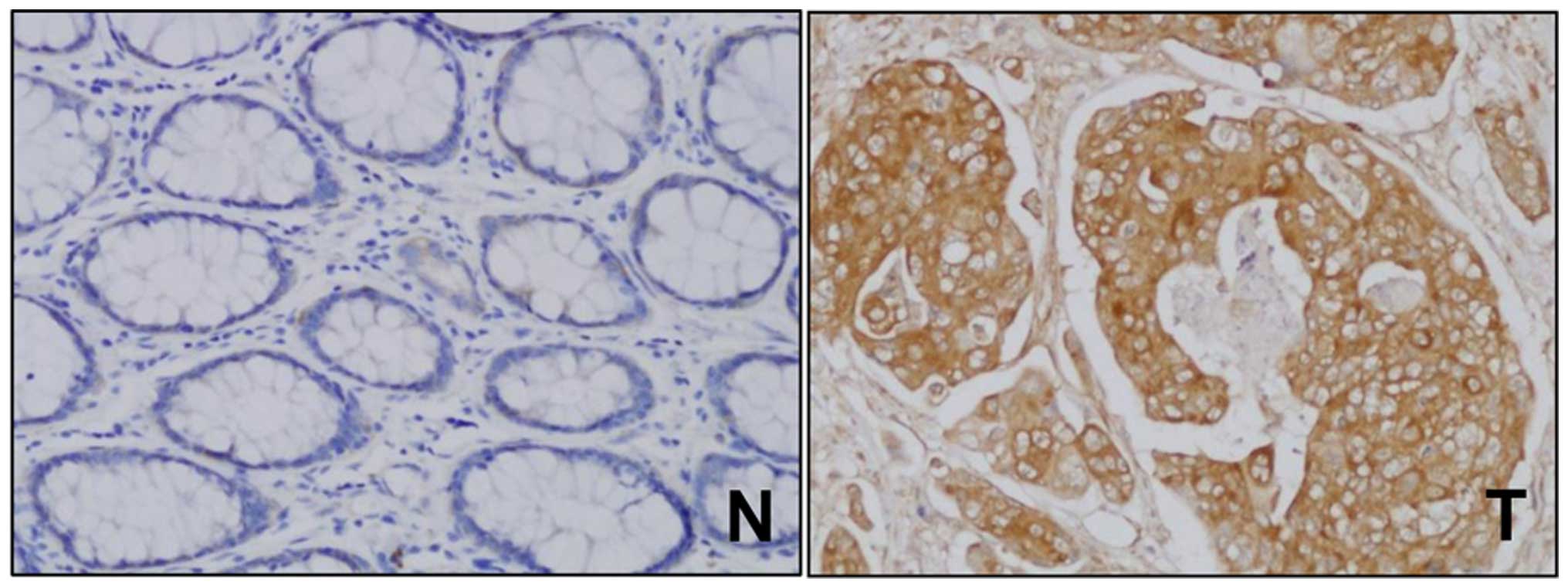
To study the role of RON in colorectal cancer
progression, we measured the protein levels of RON in
formalin-fixed, paraffin-embedded tissue blocks. These tissues were
obtained from 88 colorectal cancer patients whose
clinicopathological data were available. We subsequently analyzed
their survival rates, as well as the correlation between RON
expression (determined by immunohistochemistry) and
clinicopathological parameters. RON immunostaining was either
absent or weak in normal colorectal mucosa, but predominant in the
cytoplasm of cancer cells (Fig. 8).
Moreover, it was undetected in the tumor stroma. The percentage of
positively stained tumor cells and staining intensity for each
sample were recorded. Of the 88 samples, RON-positivity was
observed in 45 (51.1%) tissues (Table
I). Additionally, immunostaining of RON was significantly
associated with age, tumor size, lymphovascular and perineural
invasion, tumor stage and lymph node and distant metastasis
(P=0.049, 0.003, 0.003, 0.001, <0.001, <0.001 and 0.001,
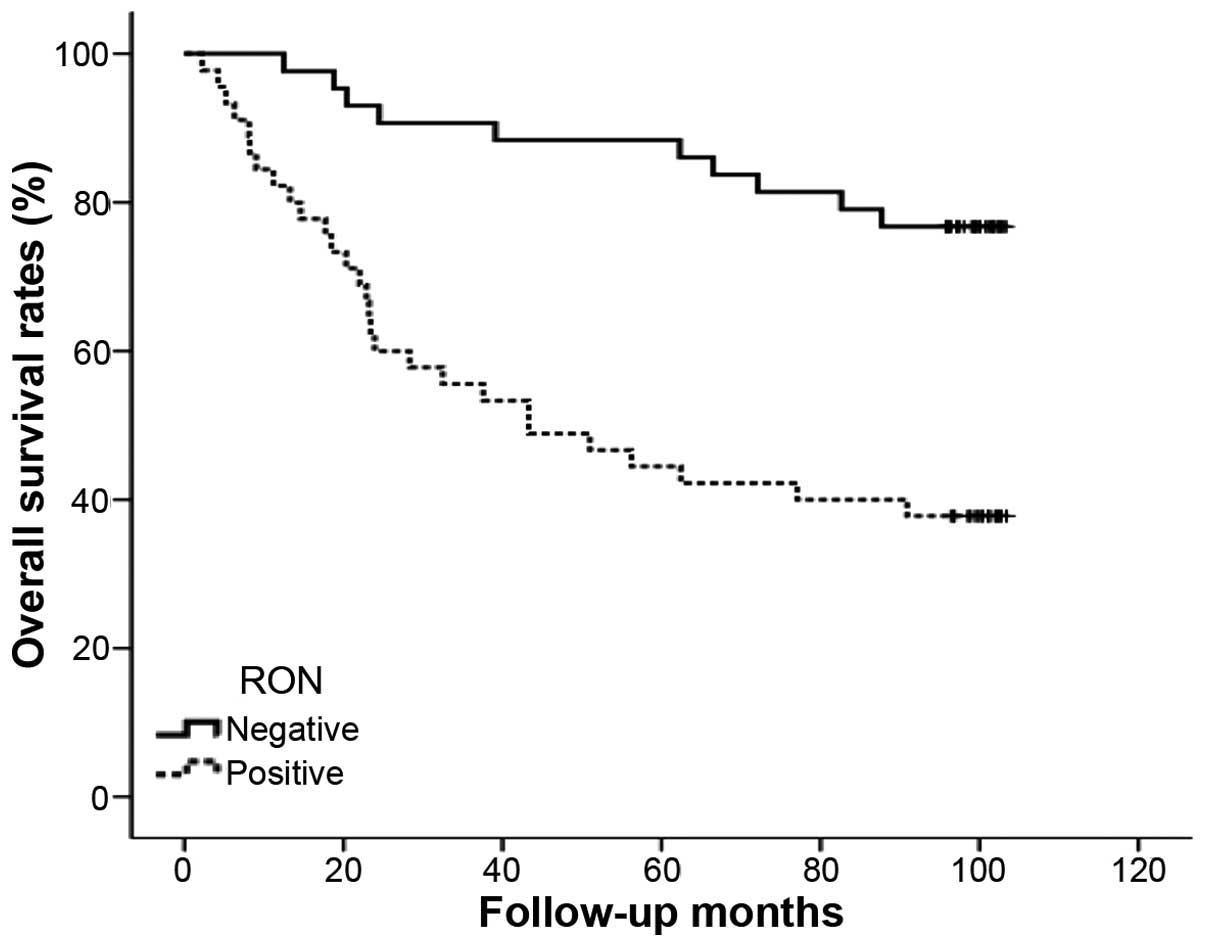
respectively; Table I). Moreover,
the overall survival rate for patients positive for RON
immunostaining was significantly lower than that for patients with
negative immunostaining (P<0.001; Fig. 9).
 | Table ICorrelation between RON expression
and the clinicopathological parameters of patients with colorectal
cancer. |
Table I
Correlation between RON expression
and the clinicopathological parameters of patients with colorectal
cancer.
| Total
(n=88) | RON
| P-value |
|---|
Negative
(n=43) | Positive
(n=45) |
|---|
| Age (years) | | | | 0.049 |
| <65.8 | 38 | 14 | 24 | |
| ≥65.8 | 50 | 29 | 21 | |
| Gender | | | | 0.859 |
| Male | 52 | 25 | 27 | |
| Female | 36 | 18 | 18 | |
| Tumor size
(cm) | | | | 0.003 |
| <4.8 | 45 | 29 | 16 | |
| ≥4.8 | 43 | 14 | 29 | |
| Histologic
type | | | | 0.687 |
|
Differentiated | 77 | 37 | 40 | |
|
Undifferentiated | 11 | 6 | 5 | |
| Lymphovascular
invasion | | | | 0.003 |
| Negative | 58 | 35 | 23 | |
| Positive | 30 | 8 | 22 | |
| Perineural
invasion | | | | 0.001 |
| Negative | 63 | 38 | 25 | |
| Positive | 25 | 5 | 20 | |
| Stage | | | | <0.001 |
| I/II | 42 | 32 | 10 | |
| III/IV | 46 | 11 | 35 | |
| Depth of invasion
(T) | | | | 0.068 |
| T1/T2 | 23 | 15 | 8 | |
| T3/T4 | 65 | 28 | 37 | |
| Lymph node
metastasis (N) | | | | <0.001 |
| N0 | 45 | 33 | 12 | |
| N1–3 | 43 | 10 | 33 | |
| Distant metastasis
(M) | | | | 0.001 |
| M0 | 78 | 43 | 35 | |
| M1 | 10 | 0 | 10 | |
Correlation of RON expression with
apoptosis, angiogenesis, and lymphangiogenesis in colorectal
cancers
All tumor samples were assessed by TUNEL assays and
immunostaining for CD34 and D2–40 in order to determine apoptosis,
angiogenesis and lymphangiogenesis. AI of the 88 tumor samples
ranged from 0.6 to 30, with a mean of 8.6 6.0. There was no
significant difference observed between RON expression and AI
(P=0.752). Moreover, MVD values for the samples ranged from 23.0 to
429.0, with a mean of 116.0 76.4. The mean MVD value for
RON-positive tumors was 133.9 82.4, significantly higher than that
for RON-negative ones (P=0.040; Table
II). Furthermore, LVD values for the tumor samples ranged from
4.0 to 31.3 with a mean of 13.5 5.7. There was no significant
difference between RON expression and LVD values (P=0.170; Table II).
 | Table IICorrelation of RON expression with
tumor cell apoptosis, angiogenesis and lymphangiogenesis in
colorectal cancer. |
Table II
Correlation of RON expression with
tumor cell apoptosis, angiogenesis and lymphangiogenesis in
colorectal cancer.
| Indices | Total | RON expression
| P-value |
|---|
| Negative | Positive |
|---|
| AI | 8.6±6.0 | 9.1±7.2 | 8.2±4.7 | 0.752 |
| MVD | 116.0±76.4 | 97.3±65.5 | 133.9±82.4 | 0.040 |
| LVD | 13.5±5.7 | 12.2±4.8 | 15.3±6.4 | 0.170 |
Correlation of AI, MVD, and LVD with
clinicopathological features of colorectal cancer
The correlation of AI, MVD and LVD with
clinicopathological parameters is summarized in Table III. Although LVD was associated
with lymph node metastasis (P=0.041), no significant correlation
was observed between either AI or MVD and various
clinicopathological parameters (Table
III).
 | Table IIICorrelation of AI, MVD and LVD with
clinicopathological parameters of patients with colorectal
cancer. |
Table III
Correlation of AI, MVD and LVD with
clinicopathological parameters of patients with colorectal
cancer.
| Parameters | AI (mean ± SD) | P-value | MVD (mean ±
SD) | P-value | LVD (mean ±
SD) | P-value |
|---|
| Stage | | 0.179 | | 0.098 | | 0.062 |
| I/II | 10.4±7.6 | | 105.6±70.0 | | 12.2±4.4 | |
| III/IV | 7.3±4.1 | | 125.6±81.5 | | 14.7±6.5 | |
| Depth of invasion
(T) | | 0.549 | | 0.297 | | 0.222 |
| T1/T2 | 11.5±10.1 | | 130.8±84.8 | | 11.4±3.9 | |
| T3/T4 | 7.8±4.2 | | 110.8±73.3 | | 14.2±6.1 | |
| LN metastasis
(N) | | 0.274 | | 0.232 | | 0.041 |
| N0 | 10.4±7.6 | | 108.3±71.7 | | 12.2±4.4 | |
| N1–3 | 7.3±4.1 | | 124.1±81.2 | | 14.7±6.5 | |
| Distant metastasis
(M) | | 0.389 | | 0.795 | | 0.102 |
| M0 | 8.7±6.4 | | 113.2±71.6 | | 13.4±5.9 | |
| M1 | 8.4±2.9 | | 138.4±109.4 | | 15.1±3.5 | |
Discussion
Overexpression and/or activation of RON has been
implicated in the progression and metastasis of diverse epithelial
cancers, where it plays a causal role in tumor progression by
promoting growth, survival and motility of tumor cells (23–29).
Cancer cells are typically characterized by
increased resistance to apoptosis and cell cycle control (5,6). In
this context, 5-FU is known to induce apoptosis and affects the
cell cycle of cancer cells (19).
It is a widely used first-line drug for colorectal cancer
treatment, but limited by drug resistance and severe toxicity
(38,39).
In the present study, we evaluated whether RON
knockdown affects chemosensitivity of colorectal cancer cells.
Apoptosis is a highly regulated process, and various proapoptotic
and anti-apoptotic genes are involved in this process (40,41).
Our study showed that RON knockdown enhances 5-FU-induced apoptosis
by upregulating the activities of caspase-3 and PARP; as well as
the expression of proapoptotic genes encoding Bax and Bim. In
contrast, cell cycle progression is regulated by positive (cyclins
and CDKs) and negative (CDKIs) effectors (42–44).
Moreover, we showed that RON knockdown augments 5-FU-induced cell
cycle arrest by decreasing the expression of cyclin B1 and D3, CDK2
and CDK6 and by inducing that of p21. Furthermore, regulation of
cell migration, invasion and proliferation is crucial for
maintaining cellular homeostasis, and its loss is a hallmark of
cancer cells (5,6). In our study, RON knockdown enhanced
the 5-FU-induced inhibitory effect on the invasion and migration of
colorectal cancer cells. These results suggest that RON functions
to resist 5-FU sensitivity by inhibiting apoptosis, cell cycle
arrest, invasion and migration in colorectal cancer cells.
To elucidate the mechanisms underlying the effects
of RON knockdown observed in the present study, we investigated the
intracellular signaling pathway that leads to inhibition of
apoptosis, cell cycle arrest, migration and invasion. We found that
the β-catenin signaling cascade is blocked upon RON knockdown along
with 5-FU treatment. Previous studies have presented this cascade
as one of the oncogenic signaling pathways activated by RON
(29,45,46).
Therefore, our results suggest that RON may regulate 5-FU
sensitivity of colorectal cancer cells via the β-catenin signaling
cascade.
Angiogenesis and lymphangiogenesis are critical
features of tumor growth and metastasis (7–11).
Increasing evidence from in vitro and in vivo studies
has shown that angiogenesis and lymphangiogenesis are associated
with tumor growth, progression and poor clinical outcome in
colorectal cancer (12–14). In the present study, we examined the
impact of RON expression on angiogenic and lymphangiogenic
phenotypes using in vitro assays. RON knockdown decreased
umbilical vein endothelial tube formation and VEGF-A and HIF-1α
expression; and increased angiostatin expression in colorectal
cancer cells. Previous studies showed that RON modulates angiogenic
chemokine production and subsequent endothelial recruitment in
prostatic cancers (47). Together,
these results indicate that RON plays an important role in tumor
progression by stimulating angiogenesis, and angiogenic and
angiostatic factors in colorectal cancer.
In addition, RON knockdown decreased lymphatic
endothelial cell tube formation and the expression of VEGF-C and
COX-2 in colorectal cancer cells. Previous studies reported that
VEGF-C, -D and COX-2 are associated with tumor lymphangiogenesis
and lymph node metastasis (9–11,48).
Therefore, we suggest that RON contributes to tumor
lymphangiogenesis in colorectal cancer.
RON is overexpressed in several cancer types and is
associated with tumor progression (30–37).
We next examined RON expression in a well-defined series of
colorectal cancer tissue samples, with special reference to patient
prognosis. RON expression was significantly increased in the cancer
tissues compared with that in normal colorectal mucosa. It was also
associated with age, tumor size and stage, lymphovascular and
perineural invasion, lymph node and distant metastasis, and poor
survival. Previous studies have associated RON expression with
aggressive disease and poor clinical outcome in colorectal cancer
patients (30,31). RON expression has also been
associated with progression and poor prognosis of gastric, ovarian
and bladder cancers (32,33,49,50).
Our results, along with these studies, suggest that RON expression
may help predict poor clinical outcome of colorectal cancer.
Finally, we evaluated the correlation of RON
expression with tumor cell apoptosis, angiogenesis and
lymphangiogenesis in colorectal cancer tissues, in order to confirm
our in vitro observations. The mean MVD value for
RON-positive tumors was significantly higher than that of
RON-negative ones. However, there was no significant difference of
RON expression with either AI or LVD.
In conclusion, RON influences tumor progression by
inhibiting chemosensitivity and enhancing angiogenesis in
colorectal cancer.
References
|
1
|
Brenner H, Kloor M and Pox CP: Colorectal
cancer. Lancet. 383:1490–1502. 2014. View Article : Google Scholar
|
|
2
|
Park SH, Song CW, Kim YB, Kim YS, Chun HR,
Lee JH, Seol WJ, Yoon HS, Lee MK, Lee JH, et al:
Clinicopathological characteristics of colon cancer diagnosed at
primary health care institutions. Intest Res. 12:131–138. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lee CK: Clinicopathological
characteristics of newly diagnosed colorectal cancers in community
gastroenterology practice. Intest Res. 12:87–89. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kim ER and Kim YH: Clinical application of
genetics in management of colorectal cancer. Intest Res.
12:184–193. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Riethdorf S, Wikman H and Pantel K:
Review: Biological relevance of disseminated tumor cells in cancer
patients. Int J Cancer. 123:1991–2006. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chambers AF, Groom AC and MacDonald IC:
Dissemination and growth of cancer cells in metastatic sites. Nat
Rev Cancer. 2:563–572. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mittal K, Ebos J and Rini B: Angiogenesis
and the tumor microenvironment: Vascular endothelial growth factor
and beyond. Semin Oncol. 41:235–251. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhao Y and Adjei AA: Targeting
angiogenesis in cancer therapy: Moving beyond vascular endothelial
growth factor. Oncologist. 20:660–673. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gomes FG, Nedel F, Alves AM, Nör JE and
Tarquinio SB: Tumor angiogenesis and lymphangiogenesis:
Tumor/endothelial crosstalk and cellular/microenvironmental
signaling mechanisms. Life Sci. 92:101–107. 2013. View Article : Google Scholar :
|
|
10
|
Stacker SA, Williams SP, Karnezis T,
Shayan R, Fox SB and Achen MG: Lymphangiogenesis and lymphatic
vessel remodelling in cancer. Nat Rev Cancer. 14:159–172. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Duong T, Koopman P and Francois M: Tumor
lymphangiogenesis as a potential therapeutic target. J Oncol.
2012:2049462012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Marques I, Araújo A and de Mello RA:
Anti-angiogenic therapies for metastatic colorectal cancer: Current
and future perspectives. World J Gastroenterol. 19:7955–7971. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Royston D and Jackson DG: Mechanisms of
lymphatic metastasis in human colorectal adenocarcinoma. J Pathol.
217:608–619. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun XF and Zhang H: Clinicopathological
significance of stromal variables: Angiogenesis, lymphangiogenesis,
inflammatory infiltration, MMP and PINCH in colorectal carcinomas.
Mol Cancer. 5:432006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhang W, Meng Y, Liu N, Wen XF and Yang T:
Insights into chemoresistance of prostate cancer. Int J Biol Sci.
11:1160–1170. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ojha R, Bhattacharyya S and Singh SK:
Autophagy in cancer stem cells: A potential link between
chemoresistance, recurrence, and metastasis. Biores Open Access.
4:97–108. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang X, Li X, Fu X, Bai M, Li X, Mei Q,
Nie J, Wu Z and Han W: Eliminating ovarian cancer stem cells: A
potential therapeutic target for ovarian cancer chemoresistance.
Curr Protein Pept Sci. 16:270–278. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Carta A, Chetcuti R and Ayers D: An
introspective update on the influence of miRNAs in breast carcinoma
and neuroblastoma chemoresistance. Genet Res Int.
2014:7430502014.PubMed/NCBI
|
|
19
|
Hoshiba T and Tanaka M: Optimization of
the tissue source, malignancy, and initial substrate of tumor
cell-derived matrices to increase cancer cell chemoresistance
against 5-fluorouracil. Biochem Biophys Res Commun. 457:353–357.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Garza-Treviño EN, Said-Fernández SL and
Martínez-Rodríguez HG: Understanding the colon cancer stem cells
and perspectives on treatment. Cancer Cell Int. 15:22015.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Paldino E, Tesori V, Casalbore P,
Gasbarrini A and Puglisi MA: Tumor initiating cells and
chemoresistance: Which is the best strategy to target colon cancer
stem cells? Biomed Res Int. 2014:8598712014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Grossi V, Peserico A, Tezil T and Simone
C: p38α MAPK pathway: A key factor in colorectal cancer therapy and
chemoresistance. World J Gastroenterol. 20:9744–9758. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lu Y, Yao HP and Wang MH: Multiple
variants of the RON receptor tyrosine kinase: Biochemical
properties, tumorigenic activities, and potential drug targets.
Cancer Lett. 257:157–164. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Leonis MA, Thobe MN and Waltz SE:
Ron-receptor tyrosine kinase in tumorigenesis and metastasis.
Future Oncol. 3:441–448. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Camp ER, Liu W, Fan F, Yang A, Somcio R
and Ellis LM: RON, a tyrosine kinase receptor involved in tumor
progression and metastasis. Ann Surg Oncol. 12:273–281. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang MH, Yao HP and Zhou YQ: Oncogenesis
of RON receptor tyrosine kinase: A molecular target for malignant
epithelial cancers. Acta Pharmacol Sin. 27:641–650. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang MH, Wang D and Chen YQ: Oncogenic and
invasive potentials of human macrophage-stimulating protein
receptor, the RON receptor tyrosine kinase. Carcinogenesis.
24:1291–1300. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wagh PK, Peace BE and Waltz SE:
Met-related receptor tyrosine kinase Ron in tumor growth and
metastasis. Adv Cancer Res. 100:1–33. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Danilkovitch-Miagkova A: Oncogenic
signaling pathways activated by RON receptor tyrosine kinase. Curr
Cancer Drug Targets. 3:31–40. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee CT, Chow NH, Su PF, Lin SC, Lin PC and
Lee JC: The prognostic significance of RON and MET receptor
coexpression in patients with colorectal cancer. Dis Colon Rectum.
51:1268–1274. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Park YL, Lee GH, Kim KY, Myung E, Kim JS,
Myung DS, Park KJ, Cho SB, Lee WS, Jung YD, et al: Expression of
RON in colorectal cancer and its relationships with tumor cell
behavior and prognosis. Tumori. 98:652–662. 2012.PubMed/NCBI
|
|
32
|
Song YA, Park YL, Kim KY, Myung E, Chung
CY, Cho SB, Lee WS, Jung YD, Kweon SS and Joo YE: RON is associated
with tumor progression via the inhibition of apoptosis and cell
cycle arrest in human gastric cancer. Pathol Int. 62:127–136. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Maggiora P, Marchio S, Stella MC, Giai M,
Belfiore A, De Bortoli M, Di Renzo MF, Costantino A, Sismondi P and
Comoglio PM: Overexpression of the RON gene in human breast
carcinoma. Oncogene. 16:2927–2933. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim SA, Yoon TM, Lee DH, Park YL, Lee KH,
Lim SC, Joo YE and Lee JK: RON (recepteur d'origine Nantais)
expression and its association with tumor progression in laryngeal
squamous cell carcinoma. Auris Nasus Larynx. 41:201–206. 2014.
View Article : Google Scholar
|
|
35
|
Yoon TM, Kim SA, Park YL, Lee KH, Sung MW,
Lee JK, Lim SC, Chung IJ and Joo YE: Expression of the receptor
tyrosine kinase recepteur d'origine Nantais and its association
with tumor progression in hypopharyngeal cancer. Head Neck.
35:1106–1113. 2013. View Article : Google Scholar
|
|
36
|
Chung CY, Park YL, Song YA, Myung E, Kim
KY, Lee GH, Ki HS, Park KJ, Cho SB, Lee WS, et al: Knockdown of RON
inhibits AP-1 activity and induces apoptosis and cell cycle arrest
through the modulation of Akt/FoxO signaling in human colorectal
cancer cells. Dig Dis Sci. 57:371–380. 2012. View Article : Google Scholar
|
|
37
|
Cho SB, Park YL, Song YA, Kim KY, Lee GH,
Cho DH, Myung DS, Park KJ, Lee WS, Chung IJ, et al: Small
interfering RNA-directed targeting of RON alters invasive and
oncogenic phenotypes of human hepatocellular carcinoma cells. Oncol
Rep. 26:1581–1586. 2011.PubMed/NCBI
|
|
38
|
American Joint Committee on Cancer
Classification (AJCC): Cancer Staging Manual. 6th edition. revised.
Lippincott-Raven; Philadelphia: pp. 113–123. 2002
|
|
39
|
Paschall AV and Liu K: Epigenetic
regulation of apoptosis and cell cycle regulatory genes in human
colon carcinoma cells. Genom Data. 5:189–191. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Mason EF and Rathmell JC: Cell metabolism:
An essential link between cell growth and apoptosis. Biochim
Biophys Acta. 1813:645–654. 2011. View Article : Google Scholar
|
|
41
|
Schultz DR and Harrington WJ Jr:
Apoptosis: Programmed cell death at a molecular level. Semin
Arthritis Rheum. 32:345–369. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Morgan DO: Principles of CDK regulation.
Nature. 374:131–134. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Graña X and Reddy EP: Cell cycle control
in mammalian cells: Role of cyclins, cyclin dependent kinases
(CDKs), growth suppressor genes and cyclin-dependent kinase
inhibitors (CKIs). Oncogene. 11:211–219. 1995.PubMed/NCBI
|
|
44
|
Johnson N and Shapiro GI: Cyclin-dependent
kinases (cdks) and the DNA damage response: Rationale for cdk
inhibitor-chemotherapy combinations as an anticancer strategy for
solid tumors. Expert Opin Ther Targets. 14:1199–1212. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wang D, Shen Q, Xu XM, Chen YQ and Wang
MH: Activation of the RON receptor tyrosine kinase attenuates
transforming growth factor-beta1-induced apoptotic death and
promotes phenotypic changes in mouse intestinal epithelial cells.
Carcinogenesis. 26:27–36. 2005. View Article : Google Scholar
|
|
46
|
Danilkovitch-Miagkova A, Miagkov A, Skeel
A, Nakaigawa N, Zbar B and Leonard EJ: Oncogenic mutants of RON and
MET receptor tyrosine kinases cause activation of the beta-catenin
pathway. Mol Cell Biol. 21:5857–5868. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Thobe MN, Gurusamy D, Pathrose P and Waltz
SE: The Ron receptor tyrosine kinase positively regulates
angiogenic chemokine production in prostate cancer cells. Oncogene.
29:214–226. 2010. View Article : Google Scholar :
|
|
48
|
Karnezis T, Shayan R, Fox S, Achen MG and
Stacker SA: The connection between lymphangiogenic signalling and
prostaglandin biology: A missing link in the metastatic pathway.
Oncotarget. 3:893–906. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Maggiora P, Lorenzato A, Fracchioli S,
Costa B, Castagnaro M, Arisio R, Katsaros D, Massobrio M, Comoglio
PM and Flavia Di Renzo M: The RON and MET oncogenes are
co-expressed in human ovarian carcinomas and cooperate in
activating invasiveness. Exp Cell Res. 288:382–389. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Cheng HL, Liu HS, Lin YJ, Chen HH, Hsu PY,
Chang TY, Ho CL, Tzai TS and Chow NH: Co-expression of RON and MET
is a prognostic indicator for patients with transitional-cell
carcinoma of the bladder. Br J Cancer. 92:1906–1914. 2005.
View Article : Google Scholar : PubMed/NCBI
|