Introduction
After water, tea is the most commonly consumed
beverage worldwide (1). Even in
Western countries, such as Germany, tea consumption is increasing.
In 2015, an average consumption of 28 liters per capita was
registered, corresponding to approximately 20,000 tons of total
domestic tea consumption. Since 2005, the share of green tea (2005:
20%) against black tea (2005: 80%) has increased to 30% (2). Similar to black tea, green tea is made
from the leaves of the tea plant Camellia sinensis (1).
Recently, matcha tea (MT) has been gaining in
popularity. The substrate concentrations are higher than that in
other green teas because the leaves are ground with a ceramic mill
to a fine powder that is later dissolved in hot water and
completely consumed. Fujioka et al published a study in 2016
in which they observed that the protective effect of MT against
oxygen radicals is significantly higher than the effect of normal
tea leaves due to increased catechin levels (3).
Research on polyphenols in green tea (GT) has
revealed considerable health benefits (4). The polyphenol family includes the
flavonoids which, in turn, incorporate flavanols and catechins.
Approximately 30% by mass of the dry substance of fresh tea leaves
is made up of phenolic compounds. In GT, 90% of these polyphenolic
compounds are catechins. The most abundant catechin in GT is
epigallocatechin gallate (EGCG) (5). Epigallocatechin (EGC), epicatechin
gallate (ECG) and epicatechin (EC) are also catechins found in GT.
However, these only make up 3–6% (EGC, ECG) and 1–3% (EC) of the
dry mass. Other components of the tea leaves by percentage of dry
mass are proteins (15%), amino acids (4%), caffeine (4%), raw
fibers (26%), lipids (7%), other carbohydrates (7%), pigments, such
as chlorophyll and carotenoids (2%) and minerals (5%) (6). A cup of GT (100 ml) contains
approximately 20–100 mg of EGCG. The US Department of Agriculture
states that 200 mg of EGCG are contained in 100 g of boiled GT
(7,8).
It was demonstrated in various studies that EGCG and
quercetin exhibit anticancer activity. Besides steroid receptor
(9) and PPARγ receptor (10) interaction, other assumed mechanisms
of action include interaction with the PI3K/Akt/mTOR signaling
pathway (11), VEGF (12), the the 67-kDa laminin receptor
(67-LR) (13), p53, Bax protein
(14) and Bcl-2 (15).
In the present study, we analyzed the effects of GT
and MT, as well as their components EGCG and quercetin, on MCF-7
and MDA-MB-231 breast carcinoma cells.
Materials and methods
Breast cancer cell lines and
preparation of the test substances
MCF-7 (HTB-22™; ATCC; American Type Culture
Collection, Manassas, VA, USA) (ER+, PR+) and
MDA-MB-231 (HTB-26™; ATCC) (ER−, PR−) cell
lines were purchased commercially. Green tea (GT) was dissolved in
water or 70% ethanol and was added to the cell culture at two
different concentrations. One gram of the MT powder or coarsely
ground Chinese tea leaves were weighed and were later dissolved in
10 ml of distilled water or 70% ethanol. The solutions were heated
in a microwave (Discover SP; CEM Corp., Matthews, NC, USA), with
the water extracts at 70°C and 80 W for 20 min and the ethanol
extracts at 60°C and 80 W for 20 min. The extracts were
subsequently centrifuged (1250 × g, 10 min), and the supernatant
was later filtered and concentrated by evaporation to a third of
the starting solution using a vacuum concentrator (Eppendorf
Concentrator S301; Eppendorf, Hamburg, Germany) at 45°C. The
extracts were frozen at −80°C for storage. Before application, the
tea extracts were centrifuged (10.410 × g for 10 min) again after
thawing.
EGCG was dissolved in distilled water and tested at
seven different concentrations in the cell cultures. Quercetin was
dissolved in 50% DMSO and tested at three different concentrations
in the cell cultures. Tamoxifen and estradiol solutions were
dissolved in 70% ethanol and examined at a single
concentration.
High-performance liquid
chromatography
To evaluate the concentration of EGCG and quercetin
in GT and MT, high-performance liquid chromatography (HPLC) was
performed. After starting the ClarityChrom® (Knauer
GmbH, Berlin, Germany), the sample syringe was filled with 20 µl of
the extract to be tested (GT, MT, quercetin and EGCG) and analyzed
for 40 min. The results were analyzed using ClarityChrom software
(version 1670-2). Since the concentrations of the active substances
were known, the quantity of the pure substance contained in the tea
extract could later be calculated by cross-multiplication.
ATP luminescence test
The ATP luminescence test assessed the viability of
the cells at different substrate concentrations. Next, 100 µl/well
cell suspensions was transferred to a 96-well plate for cell
incubation for 24 h at 37°C in 5% CO2. After the
addition of 1 µl/well of the extract, further cell incubation was
performed for 48 h at 37°C and 5% CO2. The CellTiter-Glo
(Promega, Mannheim, Germany) substrate was mixed with the
CellTiter-Glo (Promega) buffer and 100 µl/well was added. After 10
min of incubation at room temperature, the cell culture-reagent
mixtures were added to a non-transparent 96-well plate (to avoid
light spill to adjacent wells) and were measured using a
luminescence reader in the CellTiter-Glo program (Promega).
Proliferation measurement using the
MTT assay
Cell proliferation was analyzed using MTT (Roche,
Mannheim, Germany). After incubation of cells (500,000/ml) for 24 h
in the absence or presence of different substrate concentrations,
the MTT-labeled reagent was added at a final concentration of 0.5
mg/ml and was later incubated again for 4 h. During this time, the
metabolically active cells transformed the yellow tetrazolium salt
MTT to purple-colored formazan crystals. After adding the
solubilization solution, the plates were incubated overnight in a
humidified atmosphere at 37°C. With a microplate reader (Model 680;
Bio-Rad Laboratories, Hercules, CA, USA), the color intensity was
measured at 570 mm using a reference wavelength of 650 nm.
BrdU proliferation assay
Cell proliferation was analyzed using a
5-bromo-2-deoxyuridine (BrdU) labeling and detection kit (Roche).
MCF7 and MDA-MB-23 cells (500,000/ml) were grown in 96-well tissue
culture plates for 24 h in the absence or presence of different
substrate concentrations. After labeling with BrdU for 3 h, the
cells were fixed, and BrdU incorporation into DNA was measured by
ELISA. Cellular proliferation inhibition was expressed relative to
the controls (100%) ± SD.
Neutral Red uptake assay
Cellular viability was analyzed using the Neutral
Red uptake assay (Sigma-Aldrich, Steinheim, Germany). Next, 100
µl/well cell suspension was transferred to a 96-well plate for cell
incubation for 24 h at 37°C in 5% CO2. After the
addition of 1 µl/well extract, further cell incubation was
performed for 24 h at 37°C in 5% CO2. The medium was
replaced with Dulbeccos modified Eagles medium (DMEM) without
additives (pH adjusted to 7.2) and 3% Neutral Red test reagent was
added. After 60 min of incubation at 37°C in 5% CO2, the
medium was removed, and the wells were rinsed with 200 µl/well of
phosphate-buffered saline (PBS). Next, 100 µl/well of Neutral Red
solution was added, and the wells were placed on a plate vibrator.
Finally, the wells were measured using an ELISA reader at 570 nm
using a reference wavelength of 655 nm.
pH measurement
The pH was measured using a glass electrode. The
electrode was calibrated before each measurement series.
Oxidative stress
The hydrogen peroxide colorimetric/fluorometric
assay kit (BioVision Inc., Milpitas, CA, USA) was used to determine
whether free oxygen radicals (ROS, reactive oxygen species) were
produced. The quantity of hydrogen peroxide
(H2O2) produced was determined
photometrically, with the concentration of
H2O2 being proportional to the measured
optical density. After incubation, OxiRed™ (LinRed IHC, Dossenheim,
Germany) was dissolved in 220 µl of DMSO (Sigma-Aldrich) and
horseradish peroxidase (LinRed IHC, HRP) in 220 µl of
H2O2-containing assay buffer. Next, 50 µl of
the cell culture supernatant was pipetted on 96-well plates, and a
H2O2 dilution series with concentrations of
0, 1, 2, 3, 4 and 5 nmol/well H2O2 was
prepared. Fifty microliters of reaction mixture (48 µl of
H2O2-containing assay buffer + 1 µl of HRP
solution + 1 µl of OxiRed™ solution, LinRed IHC) was added. The
samples were vortexed and incubated for 10 min at room temperature
before undergoing measurement using an ELISA reader at 570 nm.
Theory/calculation
The results were tested for normal distribution
using the Kolmogorov-Smirnov test. Subsequently, one-factor
variance analysis (ANOVA, analysis of variance) was carried out
using with Bonferronis post-hoc test. The statistics program IBM
SPSS Statistics version 22 (IBM Corp., Armonk, NY, USA) was used
for these analyses. A significance level of α = 0.05 and a
confidence interval of 95% were selected. Significant differences
were examined between the respective test results and negative
controls with the corresponding solvent, as well as between the two
cell lines. The calculation of the correlations took place for
interval-scaled features using the Pearsonian dimension correlation
coefficient with MS Excel 2010. A very high correlation was assumed
to be r>0.9.
Results
HPLC
For each tea extract and pure substance, three
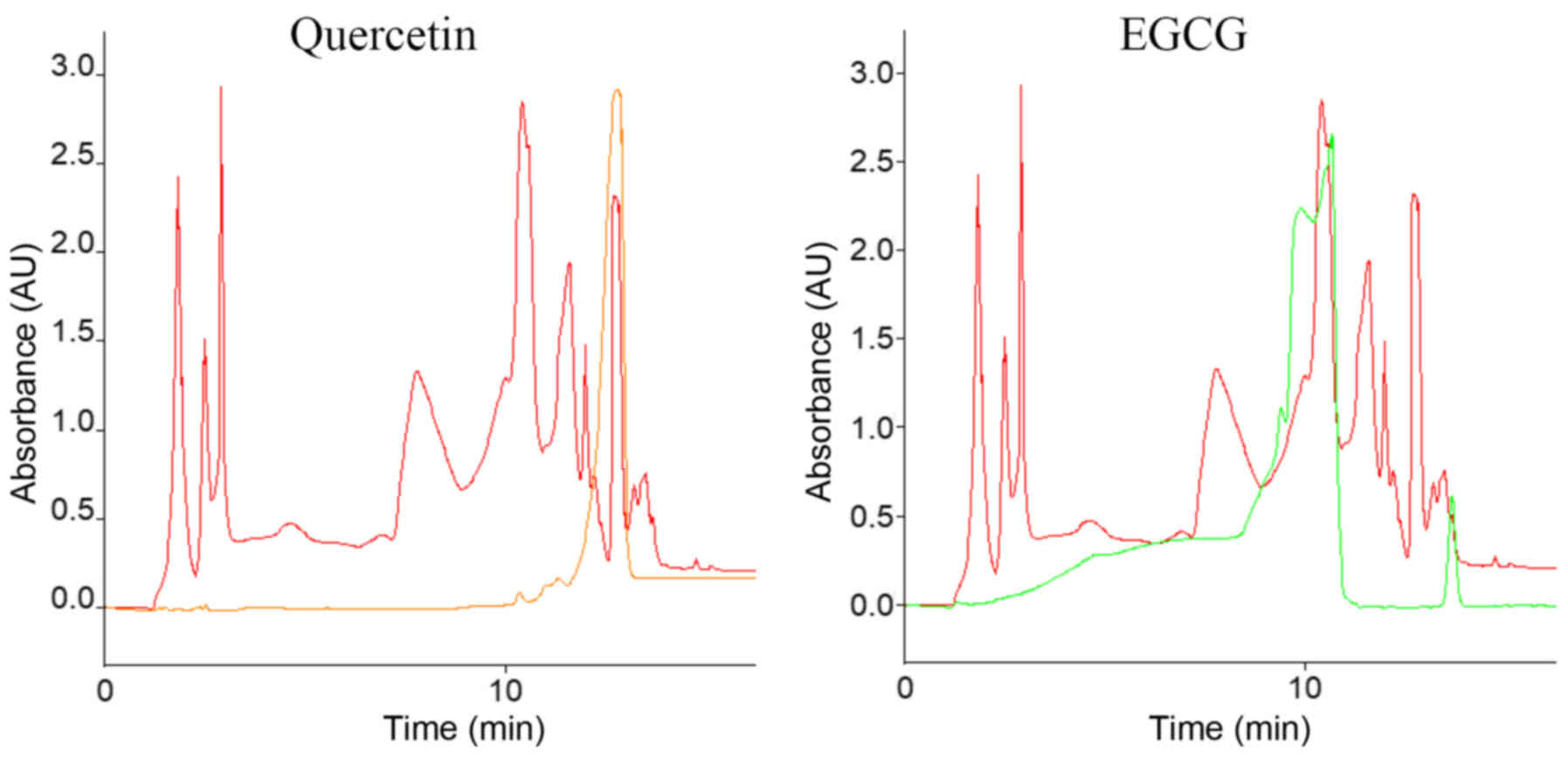
measurements were performed. Fig. 1
shows as an example the superimposed HPLC graph of one tea extract
+ one pure substance. Superimposed peaks represent similar
retention times, assuming that the two covering peaks are the same
substance.
Quercetin and EGCG could be detected in MT and in GT
in ethanol, as well as dissolved in H2O, using HPLC. The
retention times for quercetin were 12.9 min in each case and 10.7
min for EGCG. The concentrations of EGCG and quercetin in the
respective tea extracts were calculated based on the respective
peak areas. The results given are the mean values of two test
series. Table I demonstrates the
obtained concentrations.
 | Table I.Concentrations of EGCG and quercetin
in mg/ml in the tea extracts (0.3 g of tea/1 ml
H2O). |
Table I.
Concentrations of EGCG and quercetin
in mg/ml in the tea extracts (0.3 g of tea/1 ml
H2O).
|
| EGCG | Quercetin |
|---|
| MT in
70%-ethanol | 13.5 | 1 |
| GT in
70%-ethanol | 15.6 | 1.1 |
| MT in
H2O | 6 | 1.2 |
| GT in
H2O | 11 | 1.1 |
| Mean | 11.5 | 1.1 |
ATP luminescence
test/CellTiter-Glo® test
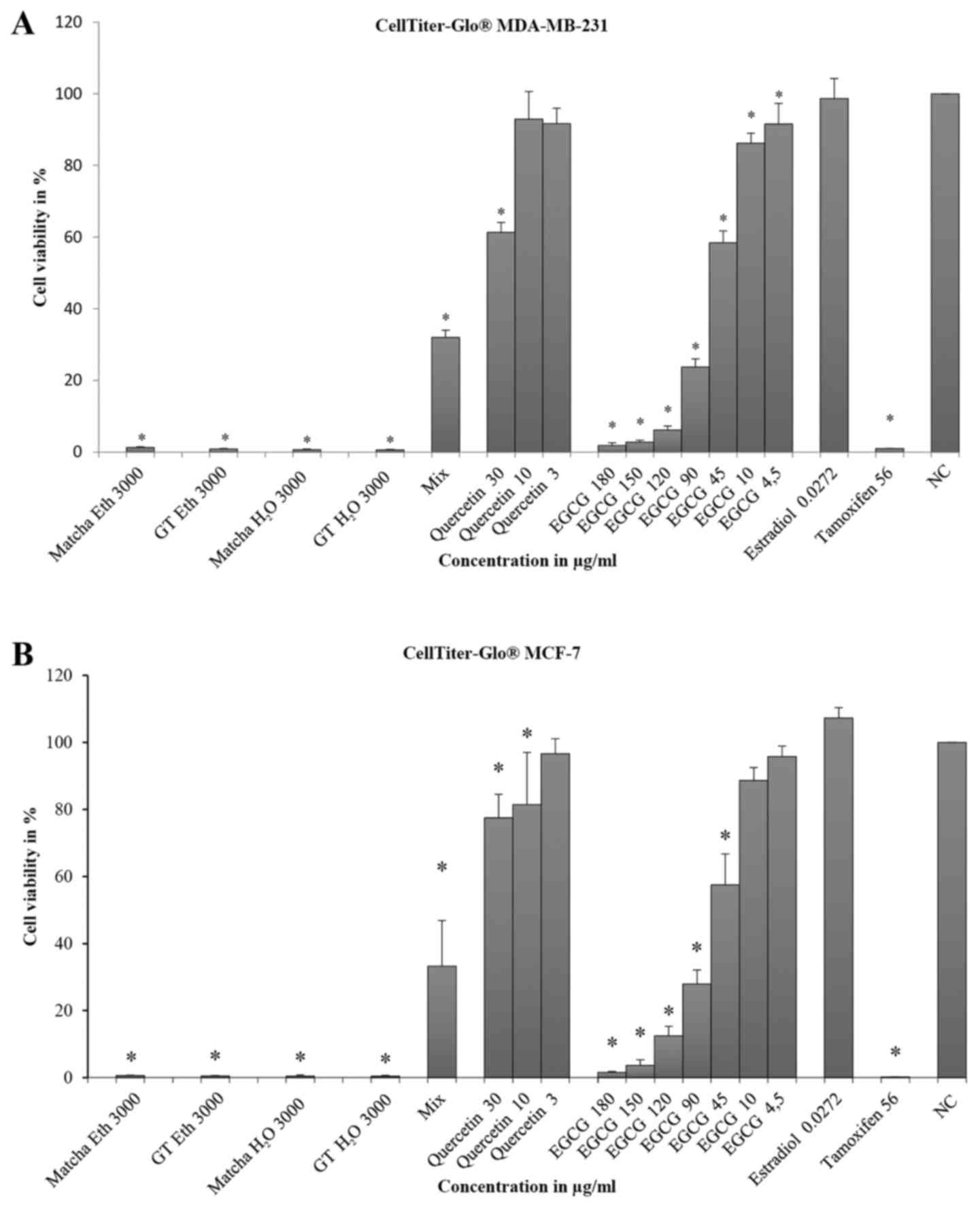
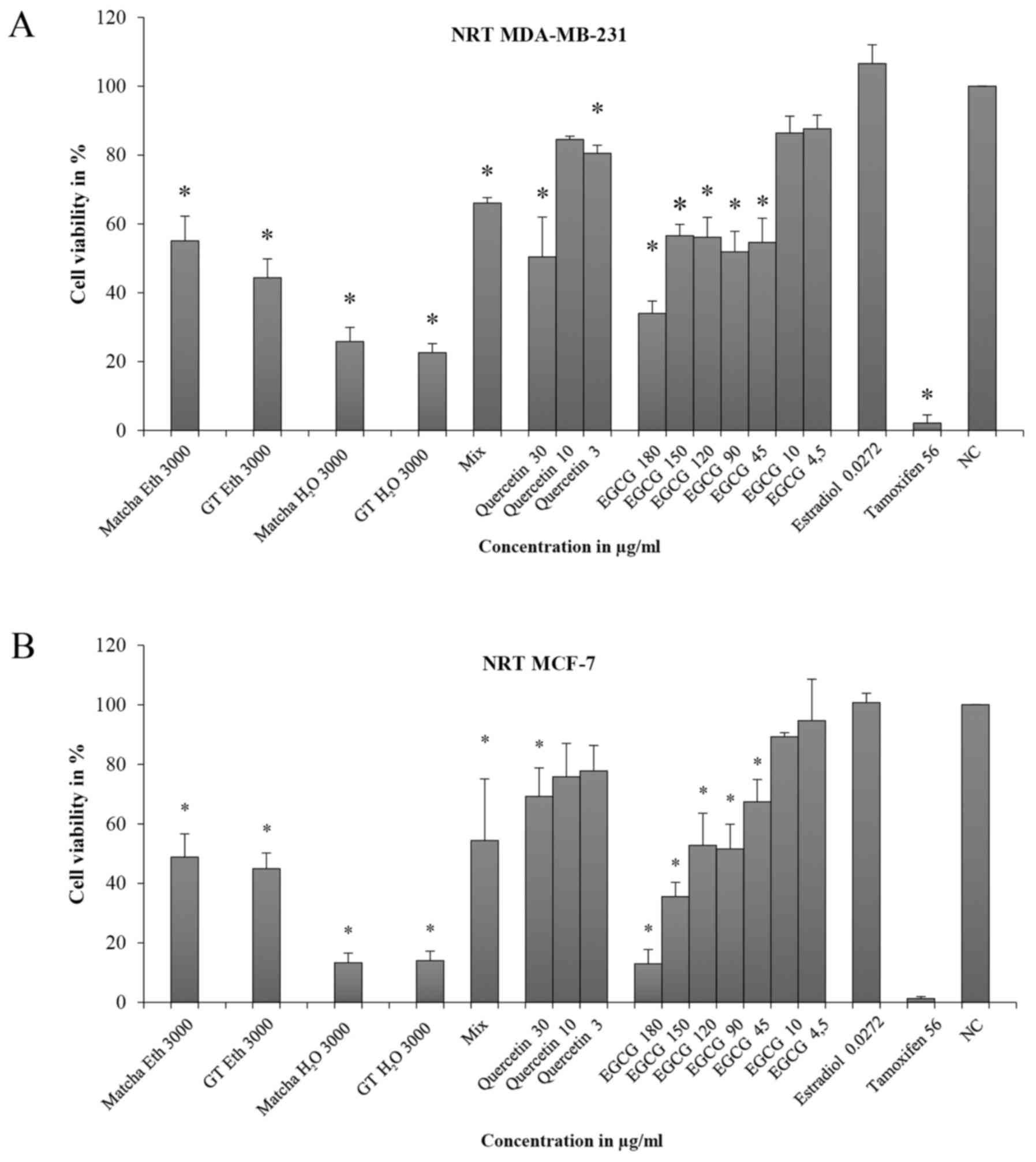
Tea extracts exhibited a significant inhibitory
effect on cell viability (CV) in MDA-MB-231 cells in ethanol and
water (Fig. 2A). Cell viability
(CV) was below 2% using tea extracts. Using the mixture of EGCG and
quercetin (Mix), significant inhibition of cell viability occurred
(CV=32%, SD=2.1). Quercetin alone did not reach the ED50
at all of the measured concentrations and inhibited cell viability
only at 30 µg/ml (CV=61.3%; SD=2.8). Different EGCG concentrations
revealed a dose-response relationship (correlation r=0.96, i.e.,
the amount at r>0.9). With increasing concentrations, the
inhibition of cell viability increased significantly for all
measurements. The concentrations of 180, 150, 120 and 90 µg/ml were
above ED50. Estradiol did not significantly alter cell
viability (98.7%, SD=5.6). Tamoxifen significantly inhibited the
cell viability of MDA-MB-231 cells above the ED50
(CV<1%).
The results using MCF-7 cells were similar to those
using the MDA-MB-231 cell line (Fig.
2B). All tea extracts were above the ED50
(CV<2%), similar to the mixture of EGCG and quercetin (Mix)
(VC=33.2%; SD=13.6). In contrast to MDA-MB-231 cells, quercetin
demonstrated an inhibitory effect beginning at a concentration of
10 µg/ml (CV=81.5, SD=7.1). As in MDA-MB-231 cells, a dose-response
relationship was observed for different EGCG concentrations
(correlation r=0.97, i.e., the amount at r>0.97). At
concentrations of 90 to 180 µg/ml, the ED50 was reached.
At lower concentrations up to 45 µg/ml, significant results were
obtained. Estradiol led to slight cell proliferation (CV=107.3%,
SD=3.0), and tamoxifen caused strong significant inhibition
(CV=0.2%, SD=0.0). Comparing the results of MDA-MB-231 and MCF-7
cells, a significant difference between the two cell lines was
found only in the result for quercetin at 30 µg/ml.
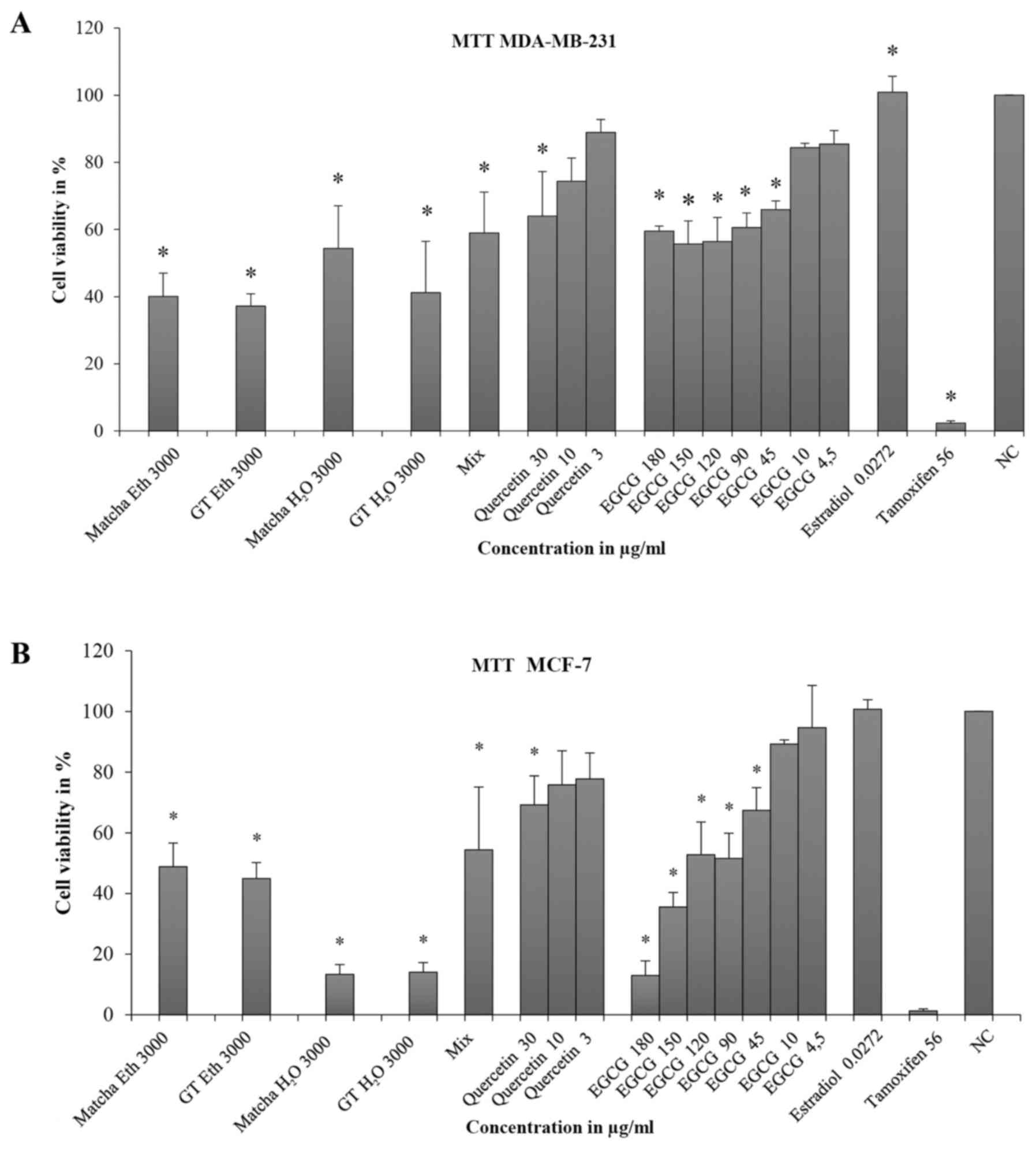
MTT assay
Tea extracts dissolved in ethanol had an inhibitory
effect on MDA-MB-231 cells in both solvents and all, besides those
in water, were above the ED50 (GT Eth: CV=40.1%, SD=6.9;
GT H2O: CV=41.2%, SD=15.3; Matcha H2O:
CV=54.4%, SD=12.7 (Fig. 3A). The
mixture (Mix) of quercetin and EGCG significantly inhibited cell
viability but did not reach the ED50 (CV=59.0%,
SD=12.2). In the case of quercetin, a dose-response relationship
was found, although statistical significance was only obtained at
30 µg/ml (CV=64.0%, SD=13.3). Although the cell viability was
significantly inhibited at 45–180 µg/ml of EGCG, the
ED50 could not be reached. Estradiol had no effect on
cell viability compared with the negative control (CV=100.8%,
SD=4.8) and tamoxifen was well above the ED50 (CV=2.3;
SD=0.6).
In MCF-7 cells, tea extracts achieved significant
inhibition of cell viability (>ED50), (Matcha
H2O: CV=32.4% and SD=4.1; GT H2O: CV=20.7%
and SD=2.1; Matcha Eth: CV=47.1% and SD=2.6; GT Eth: CV=30.8% and
SD=1.6) (Fig. 3B). The mixture
(EGCG + quercetin = Mix) had little effect on cell viability
(CV=91.4%, SD=2.0). Quercetin caused no significant inhibition of
cell viability; 3 and 10 µg/ml fell above the value of the negative
control (3 µg/ml: CV=116.6, SD=9.1, 10 µg/ml: CV=101.6, SD=5.3).
Only at 30 µg/ml was minimal inhibition noticed (CV=99%, SD=7.6).
EGCG only demonstrated significant inhibition of viability at 180
µg/ml (CV=66.0, SD=24.9). Concentrations between 45 and 4.5 µg/ml
had no effect with CV values above 100%.
Comparing the MTT test results of MDA-MB-231 and
MCF-7 cells, a significant difference between the two cell lines
was found for quercetin at concentrations of 3 and 30 µg/ml and for
EGCG at concentrations of 45 and 90 µg/ml.
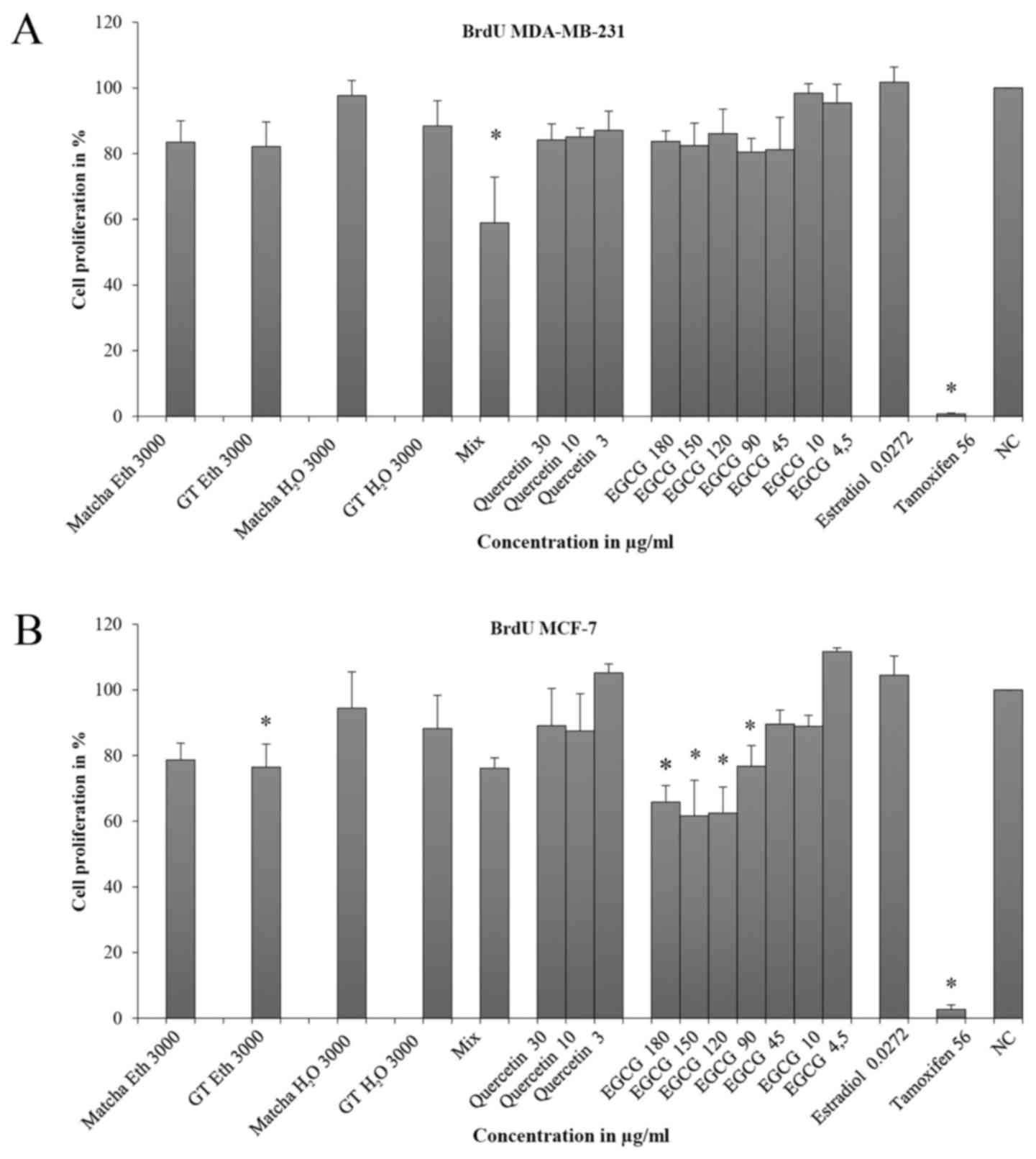
BrdU assay
In MDA-MB-231 cells, significant inhibitory effects
were only demonstrated using the mixture (EGCG + quercetin = Mix)
(cell proliferation (CP)=51.9%, SD=16.8) and tamoxifen (CP=1.0%;
SD=0.2) (Fig. 4A). The
ED50 was only reached using tamoxifen. The results for
quercetin showed a dose-response relationship (30 µg/ml: CP=84.2%,
SD=5.0; 10 µg/ml: CP=85.1%, SD=2.7; 3 µg/ml: CP=87.1%, SD=5.9).
Estradiol showed a strong positive effect on cell proliferation
(CP=0.71%, SD=0.23) compared with the negative control (NC)
(CP=101.7%, SD=4.7).
In the BrdU test for MCF-7, the ED50 was
not reached by any of the extracts except tamoxifen (CP=2.7%,
SD=1.3) (Fig. 4B). At the lowest
concentrations of quercetin (3 µg/ml: CP=105.2%, SD=2.7) and EGCG
(4.5 µg/ml: CP=111.7%, SD=1.1) a proliferation-promoting effect was
observed.
Neutral Red test
In MDA-MB-231 cells, a clear difference in the
strength of inhibition between the tea extracts dissolved in
ethanol or water was demonstrated (Fig.
5A). While the extracts dissolved in water showed strong
inhibition of cell viability (CV) of 22.6% (SD=2.6) in GT and 25.8%
(SD=4.1) in MT, ethanol extracts (Eth) only demonstrated a slight
inhibition of 44.3% (SD=5.5) in GT and 55.1% (SD=7.2) in MTE.
Statistical significance was achieved with all tea extracts. EGCG
showed significant inhibition of cell viability at concentrations
of 45–180 µg/ml, but the ED50 was reached only at 180
µg/ml.
In MCF-7 cells, as in the case of MDA-MB-231 cells,
it was demonstrated that tea extracts dissolved in water (Matcha
H2O: CV=13.3%; SD=3.3; GT H2O: CV=14.0%,
SD=3.2) have a significantly more potent inhibitory effect on cell
viability than when dissolved in ethanol (Matcha Eth: CV=45.0%,
SD=5.2; GT Eth: CV=48.8%, SD=7.8) (Fig.
5B).
For EGCG, the ED50 was reached at 180
µg/ml (CV=13.0%, SD=4.8), 150 µg/ml (CV=35.5%, SD=4.8) and 90 µg/ml
(CV=%, SD=7.1). Quercetin led to a significant inhibition of
viability only at the highest concentration used (CV=69.2%,
SD=9.5). None of the extracts led to an increase in viability
(>100%); only the result for estradiol was similar to that for
the negative control (NC) (CV=100.7%, SD=3.2). Tamoxifen treatment
resulted in only 1.3% cell viability.
Oxidative stress
The oxidative stress test was used to evaluate the
metabolism of the cells. As an indicator of oxidative stress, an
increase in the H2O2 concentration was
measured in the cell culture.
It was found that in all extract suspensions, the
concentration of H2O2 increased compared with
that in the negative control. GT extracts dissolved in ethanol
resulted in the strongest increase in H2O2
(up to 1.7 times the values observed in the control). EGCG also
showed a factor of 1.5 increase in the H2O2
concentration (data not shown).
Similar to MDA-MB-231 cells, the GT extracts
dissolved in ethanol and EGCG extract showed the highest
H2O2 concentrations in MCF-7 cells compared
with those in the negative control with a maximum 1.5-fold
increase.
Discussion
This study confirmed that EGCG and quercetin are
contained in GT and that the single substances, as well as GT
itself, have an anticarcinogenic effect on both ER/PR
receptor-positive and -negative breast cancer cells, suggesting
estrogen receptor-independent pathways.
EGCG is the most abundant anticarcinogenic catechin
in GT (4). In the German breast
cancer guideline of 2012, EGCG has been previously described as a
complementary drug used by patients that should not be applied
outside of clinical trials (16).
In the ATP luminescence test, a correlative (|r|=0.97)
concentration-dependent inhibition of cell viability was observed
in both cell lines. At the highest concentration tested, the effect
of EGCG corresponded with that of tamoxifen. Wang et al
performed the same ATP assay with MCF-7 cells and EGCG, testing
EGCG concentrations between 4.5 and 54 µg/ml. These researchers
results showed an inhibition of cell viability even at low
concentrations after 24 h. In the MTT assay, Wang et al
reported viability values after 48 h that are comparable to those
of our study (18). In MDA-MB-231
cells, Thangapazham et al (17) described lower viability values in
the MTT assay with EGCG at different concentrations after 48 h. The
ED50 was reached at EGCG 50 µg/ml; however, in this
study, the ED50 was not achieved by any of the tested
concentrations. In the studies of both Wang et al and
Thangapazham et al, the measurements were also carried out
after 24, 48 and 72 h, and even 96 h, as reported by Thangapazham
et al. All of the results showed that the cell viability was
decreased further over time. An explanation for the difference in
viability values, despite identical test methods and identical EGCG
concentrations, could be the pH value. As early as 1997, Zhu et
al reported that catechins are considerably more stable at
acidic pH values than at alkaline pH values (19). Our pH measurements always showed
alkaline pH values, suggesting instability of the EGCG molecules.
In the investigations of Wang et al and Thangapazham et
al, no information was available on the pH values of the cell
cultures. Therefore, it is possible that they worked at lower pH
values, leading to higher stability of the catechins and increased
inhibition of cellular viability.
Not every EGCG concentration in the BrdU test in
MCF-7 cells reached the ED50. However, in MDA-MB-231
cells, the mixture of EGCG and quercetin exhibited a stronger
effect than the two single substances alone. An explanation could
be the influence of the two substances on the PI3K/Akt/mTOR
signaling pathway and thus on endothelial growth factor (VEGF). Van
Aller et al (20)
demonstrated that EGCG has an inhibitory effect on this signaling
pathway in MDA-MB-231 cells. Bruning confirmed the inhibitory
effect of quercetin on mTOR (21).
Gu et al observed that the oral administration of EGCG in
mice resulted in a VEGF-mediated reduction of tumor volume compared
with the control group (22). Wang
et al demonstrated in 2014, using a xenograft prostate tumor
mouse model, that the combination of EGCG and quercetin resulted in
greater bioavailability and lower methylation of the catechins.
Tumor size could be reduced compared with that in the control
groups. They also found that the quercetin and EGCG combination led
to a significant reduction in phosphorylated AKT (pAKT) (23). Scandlyn et al combined EGCG
with tamoxifen, provoking a reduction of mTOR by 78% in
receptor-negative tumors in vivo (24).
Our results showed no significant differences
between the effects of EGCG on estrogen receptor-negative
MDA-MB-231 and ER-positive MCF-7 cells; therefore, it can be
assumed that the effect is not, or at least not exclusively,
generated via the estrogen receptor. Tachibana et al
(26) and Umeda et al
(25) found that EGCG exerts its
anticarcinogenic effect on its binding behavior with the surface
the 67-kDa laminin receptor (67-LR). The 67-LR is important in the
metastasis of tumor cells (27).
Mittal et al showed that EGCG inhibits telomerase in MCF-7
cells, which is increased in more than 90% of all cases in tumors
(28). Roy et al (29) described increased expression of p53
and the proapoptotic protein Bax, as well as reduced expression of
the anti-apoptotic protein Bcl-2 in estrogen receptor-negative
breast cancer cells under EGCG influence. Moreover, although
MDA-MB-231 cells are triple negative, these cells express ERβ.
Therefore, tamoxifen treatment is possible (30).
In the tests performed using GT and MT extracts, a
reduction in cellular viability, vitality, and proliferation was
observed (Fig. 2A and B). It is
assumed that EGCG is responsible for most of the anticancer effects
of GT (4,31). As demonstrated in our results, Wang
et al observed significantly higher viability values in the
MTT assay than the ATP measurements (18). An explanation could be offered by
the studies of Bruggisser et al (32) and Peng et al (48), who observed that antioxidants and
flavonoids result in a reduced reaction of MTT to the dye formazan,
even without the presence of living cells. The amount of formazan
formed is measured photometrically, and false-high results are
measured.
The flavonoid quercetin was detected by HPLC in GT.
In almost all tests performed, inhibition of cell viability was
demonstrated, except for the MCF-7 cells in the MTT assay, in which
all results were above 100% cell viability but without a
statistical significance. In a study by Scambia et al,
quercetin inhibited the growth of MCF-7, as well as that of
MDA-MB-231 cells, in a dose-dependent manner by binding to the
estrogen-binding domain type II (EBS II) (33). Lee et al described that the
induction of apoptosis of MCF-7 cells is caused by the formation of
free oxygen radicals that activate the pro-apoptotic
AMPKα1/ASK1/p38 signaling pathways (34). Duo et al recognized the
apoptotic effect of quercetin in the upregulation of Bax protein
and reduction of Bcl-2 protein (35). Similar to EGCG, quercetin also has
an inhibitory effect on the mTOR signal cascade (36).
As already mentioned, EGCG is a very sensitive
molecule. It is assumed that EGCG changes its structure under cell
culture conditions by auto-oxidation and dimerization. Sang et
al observed increased instability of the EGCG molecule in
normal tap water, attributable to its iron constituents (37). Other factors that influence the
molecular stability are pH, temperature and the concentration of
EGCG (20,37). Sang et al demonstrated that
20 µmol/l of EGCG in RPMI-1640 medium (named after the Roswell Park
Memorial Institute), similar to the DMEM used in our work, lost
integrity after 1 h at 37°C. At high concentrations, EGCG tends to
change the stereoconformation and become GCG (gallocatechingallate)
and the molecules half-life is prolonged. At lower concentrations,
dimer formation occurs, resulting in molecular instability.
Inconsistent amounts of time passed between the preparation and
measurement may contribute to the heterogeneous results in the
literature on the amount of EGCG in a cup of tea, presuming that
some of the EGCG may have already decayed after the preparation of
the extracts at the time of the determination of the quantity
(37).
Importantly, this study was performed in
vitro; therefore, the bioavailability of EGCG in humans was not
considered. Yang et al found in a patient study (n=18) that
the consumption of 3 g of decaffeinated GT (EGCG content: 73 mg/g)
resulted in a plasma concentration of no more than 326 ng/ml.
However, an increase from 1.5 to 3 g resulted in a triple EGCG
plasma concentration; a further increase at a dose of 4.5 g could
not be observed. Thus, it appears that a saturation of EGCG uptake
is achieved around 3 g (38). Chow
et al tested the bioavailability of EGCG by administering
EGCG capsules. They measured the plasma levels of 439 ng/ml after
administering 800 mg of EGCG or 388 ng/ml after the administering
800 mg of uncontaminated EGCG (polyphenol E) (39). These results show that the
bioavailability of EGCG in plasma is very low.
One way to increase EGCG bioavailability is
described by Landis-Piwowar et al, who demonstrated in
vitro as well as in animal experiments in vivo that a
prodrug of EGCG, in which the hydroxyl groups were protected by
peracetic acid groups before methylation, leads to greater
bioavailability and results in higher concentrations in breast
cancer cells (40). Moreover, in
2007, they found that the methylation of EGCG by catechol
O-methyltransferase (COMT) leads to decreased
anticarcinogenic activity of the catechins (41). This observation was also consistent
with the work of Wu et al who demonstrated that
predominantly, carriers of the COMT-L allele, a low-activity form
of COMT, benefited from the anti-carcinogenic effects of the
catechins; however, in carriers of the highly active COMT-H allele,
there was no difference in breast cancer risk between tea consumers
and non-consumers (42).
Landis-Piwowars et al demonstrated that, in MDA-MB-231 cells
expressing the COMT-H form, the effect of catechins can be
increased using COMT inhibitors (43).
In conclusion, based on our results and the findings
reported in the literature, the anti-carcinogenic activity by GT
and its components EGCG and quercetin can be assumed.
A problem in the therapeutic setting of EGCG is the
low oral bioavailability. In animal studies (e.g., in athymic
mice), the possibility of intravenous EGCG administration to avoid
the first-pass effect could be tested (44). Another approach would be to
administer an EGCG prodrug, as described by Landis-Piwowar et
al, leading to better bioavailability (40).
However, more studies are crucial to determine the
potential toxicity of EGCG at higher doses, as hepatotoxic effects
at high concentrations are described. The drug interactions of EGCG
and associated risks, e.g., effects of the pharmacokinetics of
concomitant medication by influencing enzymes such as the
cytochrome P450 system, need further evaluation (45). It is also worth including Her-2
positive cell lines in further studies. But for this study, we
focused on ERα/ERβ-positive cells.
EGCG and tamoxifen could represent a promising
combination in breast cancer therapy as synergistic
anti-proliferative and cytotoxic effects in both MCF-7 and
MDA-MB-231 cells were observed (46,47).
Scandlyn et al observed an anti-proliferative effect of EGCG
in combination with low tamoxifen doses (75 µg/kg) in animals with
triple-negative breast cancer (24). To better understand this effect,
active compounds of the two substances and pathways associated
should be investigated more intensively. Therefore, studies
regarding the influence on apoptosis and neovascularization should
be considered. To understand the mode of action of EGCG, it would
be interesting to further investigate the role of the 67-kDa
laminin receptor (25,27).
Acknowledgements
Not applicable.
Funding
The presents tudy was funded by the Department of
Obstetrics and Gynecology of the LMU Munich and by the ‘Deutsche
Krebshilfe’ for D.U. Richter.
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors contributions
LS and PM performed the experiments. SH added
additional experiments. DUR conceived and designed the study. JGK,
HH, TV, TPB, SM and UJ were also involved in the conception of the
study and revised it critically. All authors read and approved the
manuscript and agree to be accountable for all aspects of the
research in ensuring that the accuracy or integrity of any part of
the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
References
|
1
|
Wolf A, Bray GA and Popkin BM: A short
history of beverages and how our body treats them. Obes Rev.
9:151–164. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
A B. Beling A Jahresbericht_2015 n.d.;
250516. Lay.
|
|
3
|
Fujioka K, Iwamoto T, Shima H, Tomaru K,
Saito H, Ohtsuka M, Yoshidome A, Kawamura Y and Manome Y: The
powdering process with a set of ceramic mills for green tea
promoted catechin extraction and the ROS inhibition effect.
Molecules. 21:4742016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Cabrera C, Artacho R and Giménez R:
Beneficial effects of green tea: a review. J Am Coll Nutr.
25:79–99. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tachibana H: Molecular basis for cancer
chemoprevention by green tea polyphenol EGCG. Forum Nutr.
61:156–169. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Belitz HD, Grosch W and Schieberle P: Food
Chemistry 4th edition. Springer-Verlag Berlin Heidelberg.
https://doi.org/10.1007/978-3-40-69934-7
|
|
7
|
Stangl V, Lorenz M and Stangl K: The role
of tea and tea flavonoids in cardiovascular health. Mol Nutr Food
Res. 50:218–228. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bhagwat S, Haytowitz DB and Holden JM:
USDA Database for the Flavonoid Content of Selected Foods.
2011.
|
|
9
|
Hallman K, Aleck K, Quigley M, Dwyer B,
Lloyd V, Szmyd M and Dinda S: The regulation of steroid receptors
by epigallocatechin-3-gallate in breast cancer cells. Breast Cancer
(Dove Med Press). 9:365–373. 2017.PubMed/NCBI
|
|
10
|
Wu M, Liu D, Zeng R, Xian T, Lu Y, Zeng G,
Sun Z, Huang B and Huang Q: Epigallocatechin-3-gallate inhibits
adipogenesis through down-regulation of PPARγ and FAS expression
mediated by PI3K-AKT signaling in 3T3-L1 cells. Eur J Pharmacol.
795:134–142. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ding ML, Ma H, Man YG and Lv HY:
Protective effects of a green tea polyphenol,
epigallocatechin-3-gallate, against sevoflurane-induced neuronal
apoptosis involve regulation of CREB/BDNF/TrkB and PI3K/Akt/mTOR
signalling pathways in neonatal mice. Can J Physiol Pharmacol.
95:1396–1405. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rashidi B, Malekzadeh M, Goodarzi M,
Masoudifar A and Mirzaei H: Green tea and its anti-angiogenesis
effects. Biomed Pharmacother. 89:949–956. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li YF, Wang H, Fan Y, Shi HJ, Wang QM,
Chen BR, Khurwolah MR, Long QQ, Wang SB, Wang ZM, et al:
Epigallocatechin-3-gallate inhibits matrix metalloproteinase-9 and
monocyte chemotactic protein-1 expression through the 67-kDa
laminin receptor and the TLR4/MAPK/NF-κB signalling pathway in
lipopolysaccharide-induced macrophages. Cell Physiol Biochem.
43:926–936. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Moradzadeh M, Hosseini A, Erfanian S and
Rezaei H: Epigallocatechin-3-gallate promotes apoptosis in human
breast cancer T47D cells through down-regulation of PI3K/AKT and
Telomerase. Pharmacol Rep. 69:924–928. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Huang CY, Han Z, Li X, Xie HH and Zhu SS:
Mechanism of EGCG promoting apoptosis of MCF-7 cell line in human
breast cancer. Oncol Lett. 14:3623–3627. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
S3 Leitlinie Mammakarzinom: n.d.
https://www.awmf.org/uploads/tx_szleitlinien/032-045OLl_S3_Mammakarzinom_2018-09.pdf
|
|
17
|
Thangapazham RL, Singh AK, Sharma A,
Warren J, Gaddipati JP and Maheshwari RK: Green tea polyphenols and
its constituent epigallocatechin gallate inhibits proliferation of
human breast cancer cells in vitro and in vivo. Cancer Lett.
245:232–241. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wang P, Henning SM and Heber D:
Limitations of MTT and MTS-based assays for measurement of
antiproliferative activity of green tea polyphenols. PLoS One.
5:e102022010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhu QY, Zhang A, Tsang D, Huang Y and Chen
Z-Y: Stability of Green Tea Catechins. J Agric Food Chem.
45:4624–4628. 1997. View Article : Google Scholar
|
|
20
|
Van Aller GS, Carson JD, Tang W, Peng H,
Zhao L, Copeland RA, Tummino PJ and Luo L: Epigallocatechin gallate
(EGCG), a major component of green tea, is a dual
phosphoinositide-3-kinase/mTOR inhibitor. Biochem Biophys Res
Commun. 406:194–199. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bruning A: Inhibition of mTOR signaling by
quercetin in cancer treatment and prevention. Anticancer Agents Med
Chem. 13:1025–1031. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gu JW, Makey KL, Tucker KB, Chinchar E,
Mao X, Pei I, Thomas EY and Miele L: EGCG, a major green tea
catechin suppresses breast tumor angiogenesis and growth via
inhibiting the activation of HIF-1α and NF-κB, and VEGF expression.
Vasc Cell. 5:92013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang P, Vadgama JV, Said JW, Magyar CE,
Doan N, Heber D and Henning SM: Enhanced inhibition of prostate
cancer xenograft tumor growth by combining quercetin and green tea.
J Nutr Biochem. 25:73–80. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Scandlyn MJ, Stuart EC, Somers-Edgar TJ,
Menzies AR and Rosengren RJ: A new role for tamoxifen in oestrogen
receptor-negative breast cancer when it is combined with
epigallocatechin gallate. Br J Cancer. 99:1056–1063. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Umeda D, Yano S, Yamada K and Tachibana H:
Green tea polyphenol epigallocatechin-3-gallate signaling pathway
through 67-kDa laminin receptor. J Biol Chem. 283:3050–3058. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tachibana H, Koga K, Fujimura Y and Yamada
K: A receptor for green tea polyphenol EGCG. Nat Struct Mol Biol.
11:380–381. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nelson J, McFerran NV, Pivato G, Chambers
E, Doherty C, Steele D and Timson DJ: The 67 kDa laminin receptor:
Structure, function and role in disease. Biosci Rep. 28:33–48.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mittal A, Pate MS, Wylie RC, Tollefsbol TO
and Katiyar SK: EGCG down-regulates telomerase in human breast
carcinoma MCF-7 cells, leading to suppression of cell viability and
induction of apoptosis. Int J Oncol. 24:703–710. 2004.PubMed/NCBI
|
|
29
|
Roy AM, Baliga MS and Katiyar SK:
Epigallocatechin-3-gallate induces apoptosis in estrogen
receptor-negative human breast carcinoma cells via modulation in
protein expression of p53 and Bax and caspase-3 activation. Mol
Cancer Ther. 4:81–90. 2005.PubMed/NCBI
|
|
30
|
Madeira M, Mattar A, Logullo ÂF, Soares FA
and Gebrim LH: Estrogen receptor alpha/beta ratio and estrogen
receptor beta as predictors of endocrine therapy responsiveness-a
randomized neoadjuvant trial comparison between anastrozole and
tamoxifen for the treatment of postmenopausal breast cancer. BMC
Cancer. 13:4252013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Stuart EC, Scandlyn MJ and Rosengren RJ:
Role of epigallocatechin gallate (EGCG) in the treatment of breast
and prostate cancer. Life Sci. 79:2329–2336. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bruggisser R, von Daeniken K, Jundt G,
Schaffner W and Tullberg-Reinert H: Interference of plant extracts,
phytoestrogens and antioxidants with the MTT tetrazolium assay.
Planta Med. 68:445–448. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Scambia G, Ranelletti FO, Panici PB,
Piantelli M, De Vincenzo R, Ferrandina G, Bonanno G, Capelli A and
Mancuso S: Quercetin induces type-II estrogen-binding sites in
estrogen-receptor-negative (MDA-MB231) and
estrogen-receptor-positive (MCF-7) human breast-cancer cell lines.
Int J Cancer. 54:462–466. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lee YK, Hwang JT, Kwon DY, Surh YJ and
Park OJ: Induction of apoptosis by quercetin is mediated through
AMPKalpha1/ASK1/p38 pathway. Cancer Lett. 292:228–236. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Duo J, Ying GG, Wang GW and Zhang L:
Quercetin inhibits human breast cancer cell proliferation and
induces apoptosis via Bcl-2 and Bax regulation. Mol Med Rep.
5:1453–1456. 2012.PubMed/NCBI
|
|
36
|
Klappan AK, Hones S, Mylonas I and Brüning
A: Proteasome inhibition by quercetin triggers macroautophagy and
blocks mTOR activity. Histochem Cell Biol. 137:25–36. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sang S, Lee MJ, Hou Z, Ho CT and Yang CS:
Stability of tea polyphenol (−)-epigallocatechin-3-gallate and
formation of dimers and epimers under common experimental
conditions. J Agric Food Chem. 53:9478–9484. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yang CS, Chen L, Lee MJ, Balentine D, Kuo
MC and Schantz SP: Blood and urine levels of tea catechins after
ingestion of different amounts of green tea by human volunteers.
Cancer Epidemiol Biomarkers Prev. 7:351–354. 1998.PubMed/NCBI
|
|
39
|
Chow HHS, Cai Y, Alberts DS, Hakim I, Dorr
R, Shahi F, Crowell JA, Yang CS and Hara Y: Phase I pharmacokinetic
study of tea polyphenols following single-dose administration of
epigallocatechin gallate and polyphenon E. Cancer Epidemiol
Biomarkers Prev. 10:53–58. 2001.PubMed/NCBI
|
|
40
|
Landis-Piwowar KR, Huo C, Chen D, Milacic
V, Shi G, Chan TH and Dou QP: A novel prodrug of the green tea
polyphenol (−)-epigallocatechin-3-gallate as a potential anticancer
agent. Cancer Res. 67:4303–4310. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Landis-Piwowar KR, Wan SB, Wiegand RA,
Kuhn DJ, Chan TH and Dou QP: Methylation suppresses the
proteasome-inhibitory function of green tea polyphenols. J Cell
Physiol. 213:252–260. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wu AH, Tseng CC, Van Den Berg D and Yu MC:
Tea intake, COMT genotype, and breast cancer in Asian-American
women. Cancer Res. 63:7526–7529. 2003.PubMed/NCBI
|
|
43
|
Landis-Piwowar K, Chen D, Chan TH and Dou
QP: Inhibition of catechol-Omicron-methyltransferase activity in
human breast cancer cells enhances the biological effect of the
green tea polyphenol (−)-EGCG. Oncol Rep. 24:563–569.
2010.PubMed/NCBI
|
|
44
|
Miyazawa T: Absorption, metabolism and
antioxidative effects of tea catechin in humans. Biofactors.
13:55–59. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Schönthal AH: Adverse effects of
concentrated green tea extracts. Mol Nutr Food Res. 55:874–885.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chisholm K, Bray BJ and Rosengren RJ:
Tamoxifen and epigallocatechin gallate are synergistically
cytotoxic to MDA-MB-231 human breast cancer cells. Anticancer
Drugs. 15:889–897. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sakata M, Ikeda T, Imoto S, Jinno H and
Kitagawa Y: Prevention of mammary carcinogenesis in C3H/OuJ mice by
green tea and tamoxifen. Asian Pac J Cancer Prev. 12:567–571.
2011.PubMed/NCBI
|
|
48
|
Peng L, Wang B and Ren P: Reduction of MTT
by flavonoids in the absence of cells. Colloids Surf B
Biointerfaces. 45:108–111. 2005. View Article : Google Scholar : PubMed/NCBI
|