Introduction
Renal cell carcinoma (RCC) derived from the lining
of the proximal convoluted tubule is the most common tumor of the
kidney and is diagnosed with high incidence (1,2).
Indeed, RCC accounts for more than 90% of kidney neoplasms and
about 2% of malignant tumors (3,4).
Approximately 50% of patients with RCC have been reported to have
low survival rates because of metastasis (5). The most common metastatic sites are
the lung parenchyma (50–60%) and liver (30–40%) (6). Traditional cytotoxic chemotherapeutic
agents and immunotherapy generally have poor effects on RCC
(7). Surgery has therapeutic
effects for patients whose tumors are limited to the kidney, but 20
to 40% of patients develop metastasis after surgery (8,9).
Moreover, recurrence and metastasis have been reported in RCC
patients undergoing radical surgery (10). Therefore, research efforts are
needed to establish effective treatment strategies for the
treatment of malignant RCC.
Gene-directed enzyme prodrug therapy (GDEPT) is an
effective strategy for cancer therapy that can minimize adverse
drug reactions as it selectively inhibits cancer cells using the
bystander effect of suicide genes that convert pro-drugs to drugs
near the tumor sites (11,12). If there are vehicles by which these
genes can be effectively transferred toward cancer cells, these
genes are expressed near the neoplasm (13). For example, Escherichia coli
cytosine deaminase (CD), a suicide gene, alters 5-fluorocytosine
(5-FC) to its effective form, 5-fluorouracil (5-FU) (11,14).
5-FU specifically inhibits DNA synthesis and induces cell death in
human tumors (15,16). In addition, interferon-β (IFN-β), a
type I IFN known to block the synthesis phase of the cell cycle,
inhibits cancer proliferation at high concentrations, but is
problematic due to excessive toxicity (17,18).
Therefore, it is necessary to investigate promising treatments
using tools that can efficiently convey anticancer genes (e.g., CD
and IFN-β) while minimizing biological cytotoxicity in normal
tissues.
In the present study, we used HB1.F3 cells (human
neural stem cells) derived from a 15-week-old fetus as delivery
tools (19). hNSCs are suitable for
the treatment of metastatic tumors because they were effectively
recruited to a distant lesion in a previous study (20). We transduced HB1.F3 cells to express
CD and/or IFN-β genes. One of these was HB1.F3.CD cells expressing
only the CD gene (19), while the
other was HB1.F3.CD.IFN-β cells expressing CD and IFN-β genes. Many
researchers have demonstrated that these cells migrate to the tumor
and interfere with cancer cell proliferation (21–23).
Furthermore, we confirmed the anticancer effect of HB1.F3.CD and
HB1.F3.CD.IFN-β cells using RCC A498 cells, in a cellular and
metastasis model. Overall, the results of this study suggest that
hNSC therapy is a powerful approach for the treatment of patients
with metastasized nodules.
Materials and methods
Cell culture and media
All hNSCs (HB1.F3, HB1.F3.CD and HB1.F3.CD.IFN-β)
were kindly provided by Dr Seung U. Kim of the University of
British Columbia, Vancouver, BC, Canada. A498 cells were purchased
from the Korean Cell Line Bank. Hyclone™ Dulbecco's modified
Eagle's medium (DMEM, Hyclone Laboratories) supplemented with 10%
fetal bovine serum (GE Healthcare), 10 mM HEPES (Invitrogen Life
Technologies; Thermo Fisher Scientific, Inc.), 10 U/ml penicillin
and streptomycin of 100 µg/ml (Cellgro Mediatech) was used as the
culture medium for all cell lines.
Reverse transcriptase polymerase chain
reaction (PCR) analysis
The RNAs of HB1.F3.CD, HB1.F3.CD.IFN-β and A498
cells were isolated using Trizol (Invitrogen Life Technologies;
Thermo Fisher Scientific, Inc.), while M-MLV RT (iNtRON
Biotechnology) was used to obtain cDNAs. The expression of the
anticancer genes was confirmed using primers for human
glyceraldehydes-3-phosphate dehydrogenase (GAPDH, sense primer
5′-ATGTTCGTCATGGGTGTGAACCA-3′ and antisense
5′-TGGCAGGTTTTTCTAGACGGCAG-3′), CD (sense primer
5′-GCGCGAGTCACCGCCAGCCACACCACGGC-3′ and antisense,
5′-GTTTGTAATCGATGGCTTCTGGCTGC-3′) and IFN-β (sense primer,
5′-AAAGAAGCAGCAATTTTCAG-3′ and antisense,
5′-TTTCTCCAGTTTTTCTTCCA-3′) genes in HB1.F3.CD and HB1.F3.CD.IFN-β
cells. In addition, stem cell factor (SCF, sense primer,
5′-CCAGCTCCCTTAGGAATGACA-3′ and antisense,
5′-TAAATGAGACCCAAGTCCCGC-3′), vascular endothelial growth factor
(VEGF, sense primer, 5′-AAAACACAGACTCGCGTTGC-3′ and antisense,
5′-GGCCGCGGTGTGTCTA-3′) and C-X-C chemokine receptor type 4 (CXCR4,
sense primer, 5′-ATCCCTGCCCTCCTGCTGACTATTC-3′ and antisense,
5′-GAGGGCCTTGCGCTTCTGGTG-3′), which are chemoattractant factors,
were amplified from A498 cells by PCR. PCR amplification of cDNAs
was performed at 30 cycles in denaturation for 30 sec at 95°C,
annealing for 30 sec at 58°C, and extension for 30 sec at 72°C. All
PCR products were separated from 1.5% agarose gel containing NEO
Green (NEO Science) by electrophoresis. The results of
electrophoresis were confirmed using LuminoGraph II (ATTO
Corp.).
Cell viability assay
A498 cells (2,000 cells/well) were seeded in 96-well
plates on day 1. After 24 h, 5-FC and 5-FU of each concentration
diluted in phosphate buffered saline (PBS) were applied once a day
for three days. In another 96-well plate, A498 (1,000 cells/well)
and hNSCs (2,000 cells/well) were co-cultured to confirm the
effects of HB1.F3.CD and HB1.F3.CD.IFN-β cells expressing
anticancer genes in vitro. 5-FC of each concentration was
applied under the same conditions in wells containing A498 and
hNSCs, after which EZ-Cytox reagent (iTSBiO) was added to wells
according to the manufacturer's protocols. After 2 h, the optical
densities (ODs) (450 nm) were measured using a microplate reader
(BioTek Instruments, Inc.) to confirm the viability of the A498
cells.
Transwell assay
Transwells (BD Biosciences) with an 8.0 µm pore size
were used to demonstrate the tumor-tropic ability of hNSCs toward
A498 cells. Briefly, A498 cells were separately seeded in a 24-well
plate (lower chambers) on day 1. Negative control (NC) groups
included only media without A498 cells. After incubation for 24 h,
three types of hNSCs were seeded into the Transwells (upper
chambers) pre-coated with fibronectin (Sigma-Aldrich; Merck KGaA).
There were 6 groups: HB1.F3 with or without A498 cells, HB1.F3.CD
with or without A498 cells and HB1.F3.CD.IFN-β with or without A498
cells. Within 24 h, the membranes of the Transwells were treated
with 3.7% formaldehyde (Sigma-Aldrich; Merck KGaA), and then
permeabilized with 100% methanol (Sigma-Aldrich; Merck KGaA). All
membranes of the Transwells were then stained with crystal violet
and the number of migrated stem cells was counted using an IX-73
inverted microscope (Olympus) and Cell Sense Dimension (Olympus
Corp.).
Metastasis model of RCC
Experiments were conducted with approval from the
Chungbuk National University Institutional Animal Care and Use
Committee (CBNUA-1089-17-01). We purchased 4 week-old female
athymic nude mice (KOATECH) and allowed them to acclimatize to the
experimental environment for one week. Thirty-fine mice were
randomly grouped by weight (initial weight 22–26 g). A498 cells
(2×106 cells) pre-stained with CMFDA Green Cell Tracker
(CMFDA) were mixed in PBS and then injected into the tail vein.
Treatment of genetically engineered
hNSCs in a mouse model
We divided the mice into five experimental groups
(seven mice per group). Group 1 (NC group) was a negative control
group without injections. Group 2 (A498 group) was injected with
A498 cells alone. One week after injection of the A498 cells, hNSCs
pre-stained with CM-DiI Red Cell Tracker (CM-DiI) were injected
into the tail vein (group 3 (F3 group) injected with HB1.F3; groups
4 (CD group) injected with HB1.F3.CD and groups 5 (CD-β group)
injected with HB1.F3.CD.IFN-β). Next, 5-FC (200 mg/kg/day) was
intravenously injected into mice in the F3, CD and CD-β groups
three times a week (24). The
injection cycle of hNSCs and 5-FC was repeated three times. At 30
days after the A498 injection, mice of all groups were euthanized
by inhalation with dimethyl ether, after which their organs were
extracted, weighed and stored in 10% formalin solution.
Fluorescence analysis
Tissue sections on slides were prepared as
previously described (22).
Briefly, slides were treated with DAPI solution (600 nM) for
fluorescence analysis, mounted with cover glasses and then analyzed
using an IX-73 inverted microscope (Olympus). Blue images indicate
the nuclei of all cells including hNSCs and A498 cells. Green
indicates CMFDA pre-stained A498 cells; red, CM-DiI pre-stained
hNSCs.
Data analysis
All experiments were repeated at least three times.
Statistical analyses were conducted using GraphPad Prism (v5.0;
GraphPad Software). Groups were compared by two-way analysis of
variance (ANOVA) followed by post hoc Dunnett's or Tukey's multiple
comparison tests or Student's t-tests. Data are presented as the
means ± standard error of the means. P-values <0.05 were
considered statistically significant.
Results
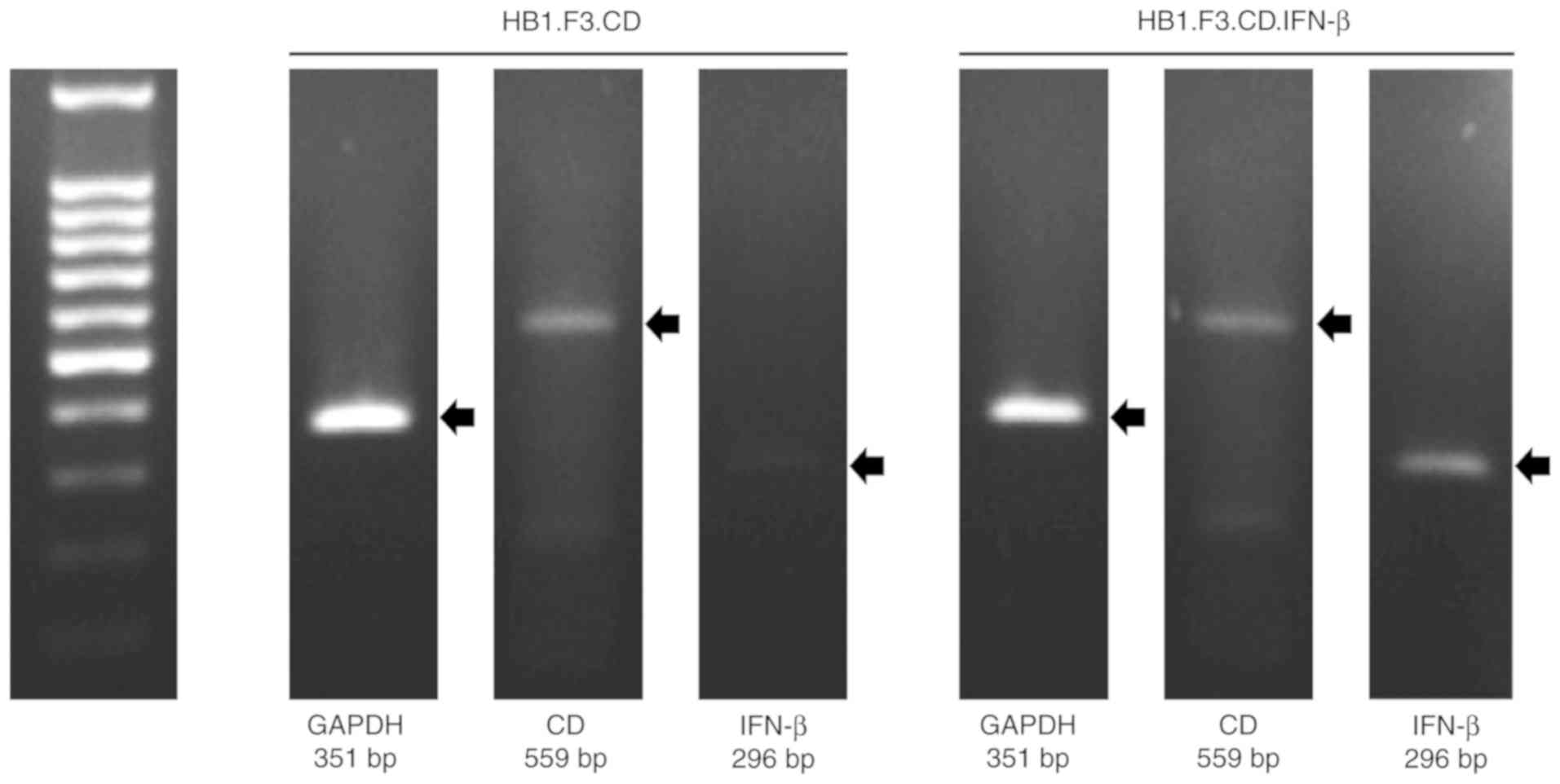
Expression of CD and/or IFN-β genes in
HB1.F3.CD and HB1.F3.CD.IFN-β cells
We conducted PCR to confirm the expression of
anticancer genes in the genetically engineered hNSCs using GAPDH,
CD and IFN-β primers. The GAPDH gene was used as an internal
control of each cell and the PCR products were electrophoresed on
the same gel. As a result, the CD gene was expressed in both
HB1.F3.CD and HB1.F3.CD.IFN-β cells and the IFN-β gene was
expressed only in HB1.F3.CD.IFN-β cells (Fig. 1).
Inhibition effect of CD/5-FC and IFN-β
gene in vitro
To confirm the inhibitory effect of 5-FU against
A498 cells, wells seeded with only A498 cells were treated with
5-FC and 5-FU, and the cell viability was evaluated. As shown in
Fig. 2B, A498 cells were
significantly influenced by treatment with 5-FU (500 µg/ml)
compared to the 5-FC (500 µg/ml) group, and a significant reduction
in cell viability was observed in response to all concentrations of
5-FU (Fig. 2A). Moreover, 5-FC was
applied to wells that contained co-cultured A498 cells and hNSCs to
evaluate the anti-proliferative effects of CD gene-expressing hNSCs
that convert 5-FC into 5-FU. The cell viability of A498 cells
decreased significantly in the HB1.F3.CD group compared to the
HB1.F3 group at all concentrations, and the synergism of the CD and
IFN-β genes was confirmed in the HB1.F3.CD.IFN-β group (Fig. 2C and D).
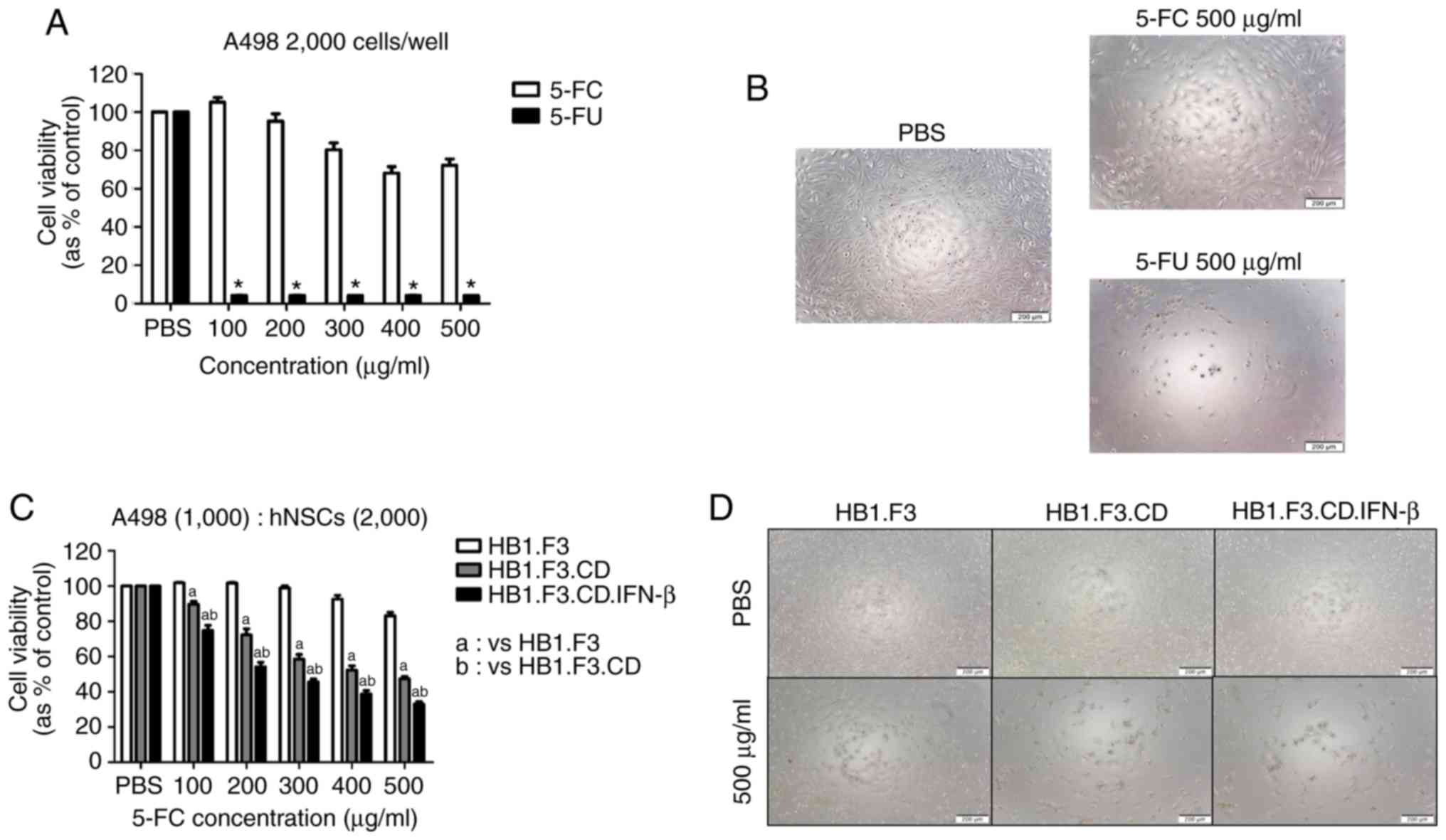 | Figure 2.A498-inhibitory effects of hNSCs
expressing CD and/or IFN-β genes against A498 cells in a cell
viability assay. (A and B) A498 cells (2×103 cells/well)
seeded on 96-well plates were treated with pro-drug (5-FC) or drug
(5-FU) of each concentration (0–500 µg/ml) once a day for 3 days.
After 3 days, cell viability assay and microscopic analysis were
performed. (C and D) A498 cells (1×103 cells/well) were
co-cultured with three types of hNSCs in 96-well plates. These 3
groups were treated with the six concentrations of 5-FC once a day
for 3 days. After microscopic analysis, cell viability was analyzed
using an EZ-Cytox kit. A PBS group was used as a negative control,
meaning that the concentration of treated 5-FC or 5-FU was 0 µg/ml.
Scale bars, 200 µm. *P<0.05 vs. 5-FC, aP<0.05 vs.
HB1.F3, bP<0.05 vs. HB1.F3.CD. CD, cytosine
deaminase; IFN-β, interferon-β; hNSCs, genetically engineered human
neural stem cells; 5-FC. 5-fluorocytosine; 5-FU,
5-fluorouracil. |
A498-specific migration ability of
hNSCs
hNSCs are known to have cancer-specific migration
capabilities induced by chemoattractant factors expressed in cancer
cells. Therefore, PCR was conducted to confirm the expression of
these factors in A498 cells. The chemoattractant factors SCF, CXCR4
and VEGF were expressed in A498 cells (Fig. 3A). The GAPDH gene was used as an
internal control of A498 cells.
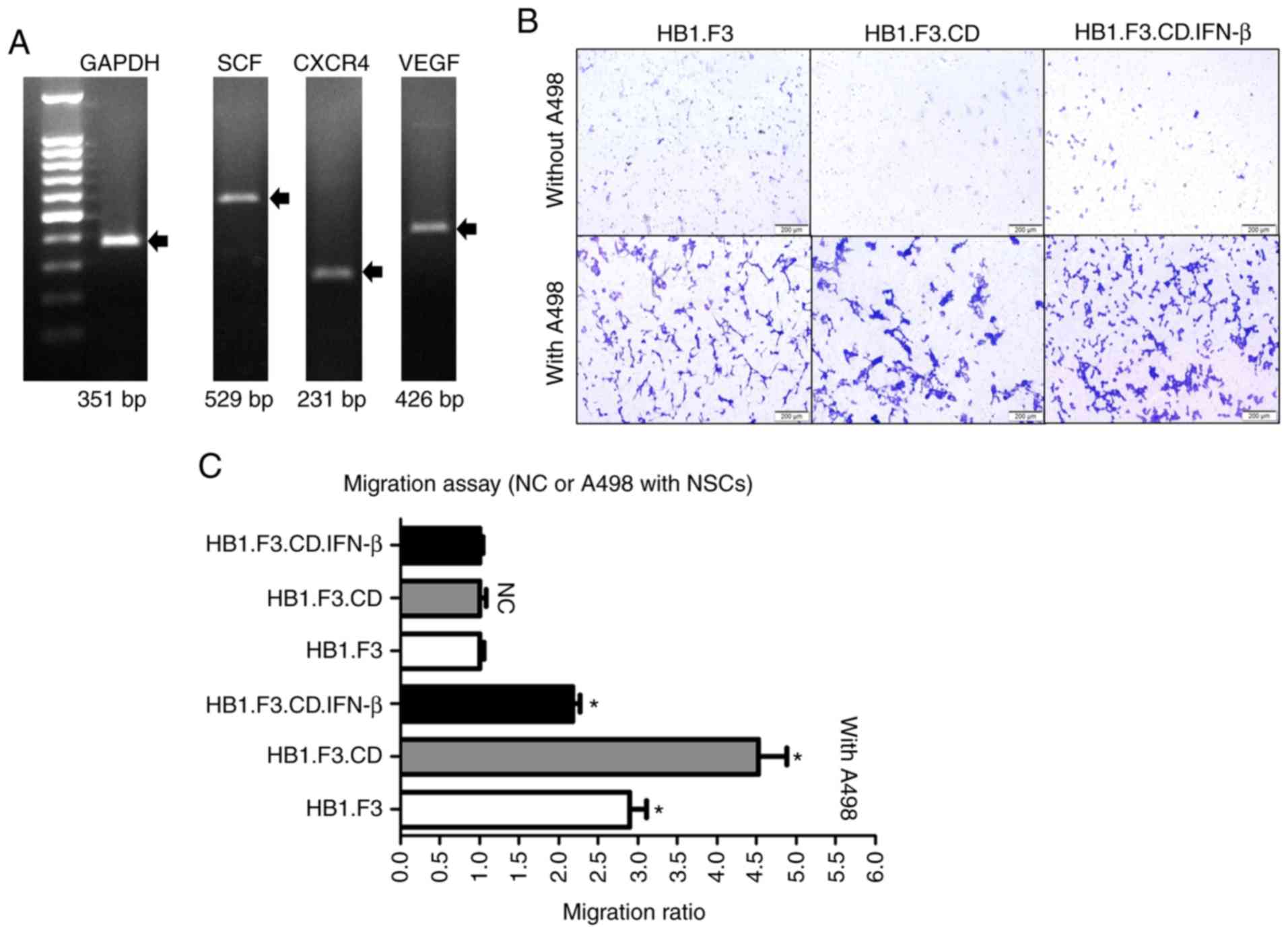 | Figure 3.Identification of A498-specific
migration ability of hNSCs by PCR and Transwell assay. (A) The
chemoattractant factors SCF (529 bp), CXCR4 (231 bp), and VEGF (426
bp) are expressed in A498 cells as determined by PCR. The results
of PCR were identified by LuminoGraph II. (B and C) A498 cells
(1×105 cells/well) were seeded into the lower chambers
of 24-well plates, while three types of hNSCs (1×105
cells/well) were seeded into the upper chambers of the Transwells
that had been pre-coated using fibronectin. After incubation for 24
h, membranes of the Transwells were stained with crystal violet.
hNSCs that migrated were enumerated using the cellSense Dimension
software (Olympus Life Science Solutions). NC groups did not
contain the A498 cells in the lower chambers. Scale bars, 200 µm.
hNSCs, genetically engineered human neural stem cells; NC, negative
control; SCF, stem cell factor; VEGF, vascular endothelial growth
factor; CXCR4, C-X-C chemokine receptor type 4. |
Transwells were used to identify the migration
capability of hNSCs toward A498 cells in vitro. The
membranes of the Transwells were stained with crystal violet
solution and the number of hNSCs that migrated in the direction of
the lower chambers, where the A498 cells were located, was
determined by counting using an IX-73 inverted microscope (Fig. 3B). As shown in Fig. 3C, a higher number of hNSCs migrated
in the A498 groups than that noted in the NC groups.
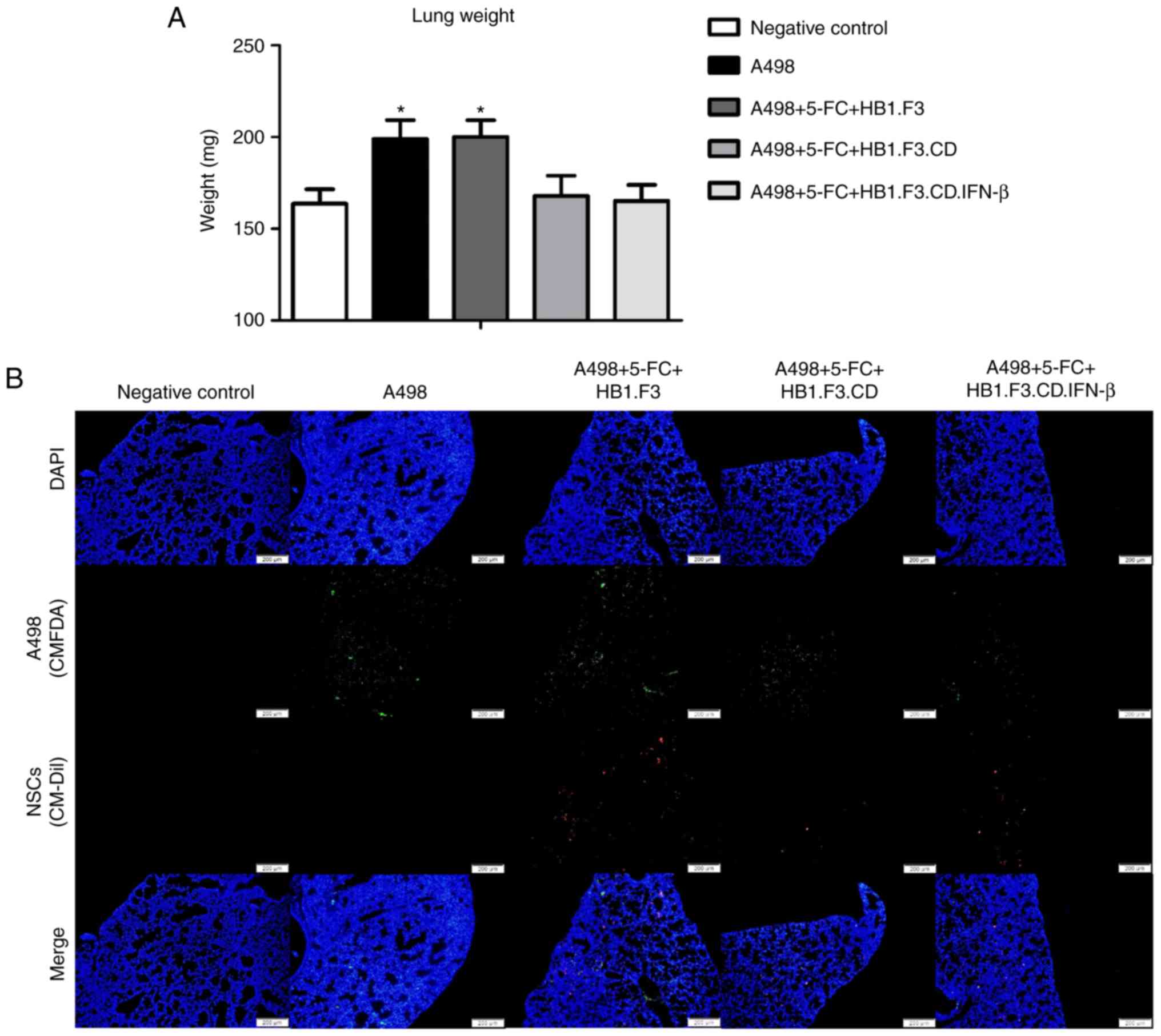
Changes in lungs in the metastasis
model
To confirm the metastasis-suppressive effects of
hNSCs in vivo, a metastasis model was established as
described in Materials and methods. After the experiment was
terminated, the weight of the lungs from all mice was determined
(Fig. 4A). Lung weight was
significantly increased in the A498 and F3 (A498+5-FC+HB1.F3)
groups compared to the NC (negative control) group. However, the
lung weights of the CD (A498+5-FC+HB1.F3.CD) and CD-β
(A498+5-FC+HB1.F3.CD.IFN-β) groups were unchanged compared to the
NC group. Fluorescence analysis revealed green fluorescence (A498
cells) in all groups except the NC group, while red fluorescence
(hNSCs) was confirmed in the F3, CD and CD-β groups (Fig. 4B).
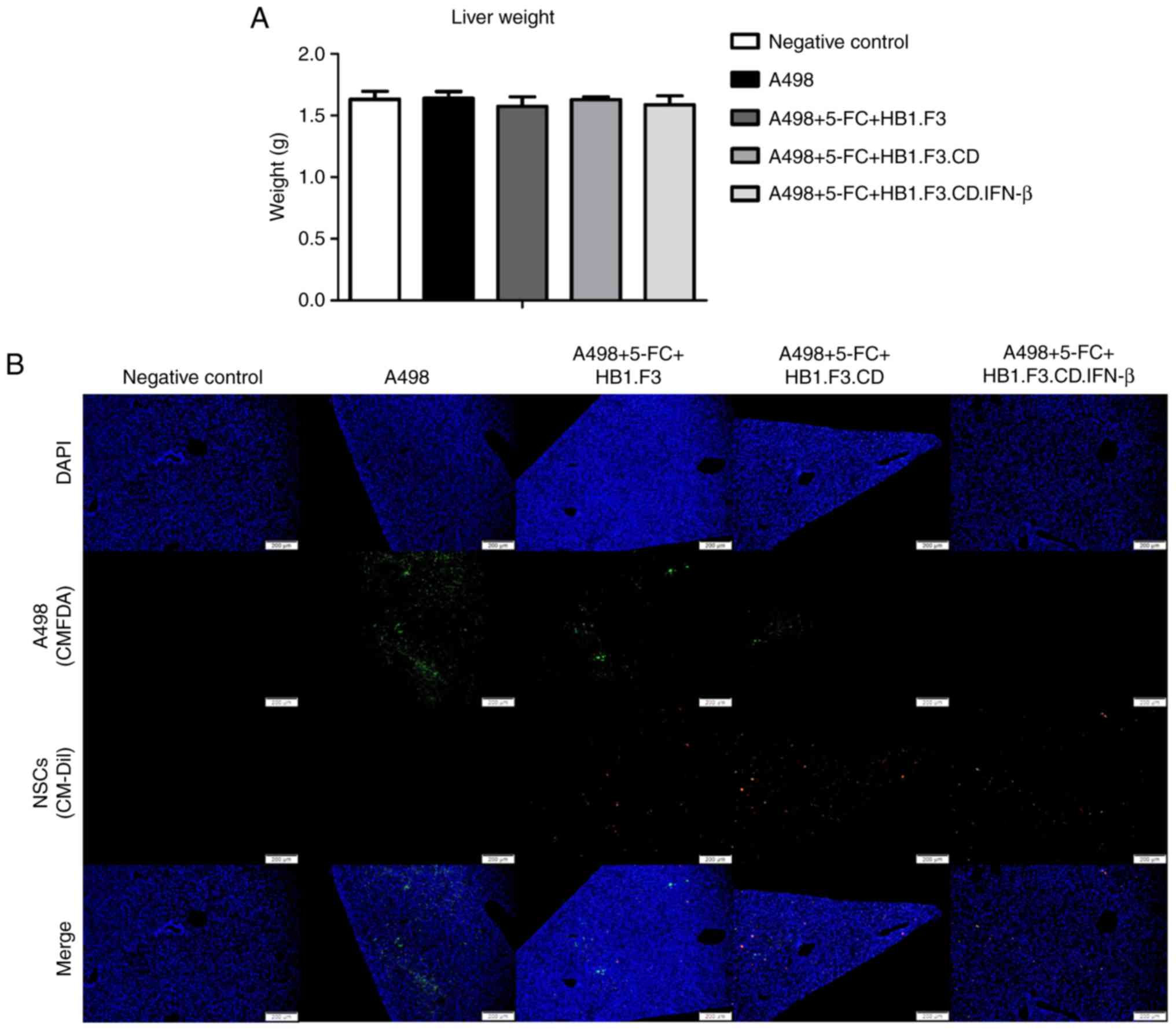
Metastasis inhibition effect of
HB1.F3.CD and HB1.F3.CD.IFN-β cells in the liver
The weights of the extracted livers were measured
using the same method as for the lungs. Unlike the results for the
lungs, there were no significant differences in the weights of the
livers between any of the groups and the NC (negative control)
group (Fig. 5A). However, as shown
in Table I, there was a significant
difference in the incidence of metastases. In particular, the CD-β
(A498+5-FC+HB1.F3.CD.IFN-β) group showed significantly fewer
incidences of metastases than the other groups in the liver.
Fluorescence analysis of livers from the F3 (A498+5-FC+HB1.F3), CD
(A498+5-FC+HB1.F3.CD) and CD-β (A498+5-FC+HB1.F3.CD.IFN-β) groups
revealed a difference in the number of mice in which green
fluorescence was confirmed, but red fluorescence was present in all
mice (Fig. 5B). Taken together,
these results indicate that the incidence of metastasis differs
between CD/5-FC and IFN-β treated and non-treated groups.
 | Table I.Reduction in the incidence of
metastasis by hNSCs expressing CD and/or IFN-β genes. |
Table I.
Reduction in the incidence of
metastasis by hNSCs expressing CD and/or IFN-β genes.
|
| Incidence of
metastasis |
|---|
|
|
|
|---|
| Groups | Lung | Liver |
|---|
| Negative control | 0/7 | 0/7 |
| A498 | 7/7 | 5/7 |
| A498+5-FC+HB1.F3 | 7/7 | 5/7 |
|
A498+5-FC+HB1.F3.CD | 7/7 | 4/7 |
|
A498+5-FC+HB1.F3.CD.IFN-β | 7/7 | 1/7 |
Discussion
Renal cell carcinoma (RCC), which is the most common
renal cancer, spreads to the lymph nodes, lungs and liver when
metastasized (25). If it spreads
to organs outside the kidney, multiple therapies such as
chemotherapy and surgery are attempted, but most cases of RCC are
resistant to traditional treatment modalities (26). A previous study revealed that 40% of
patients diagnosed with RCC die of cancer progression, 25–30% of
whom are identified with metastatic RCC (27). Therefore, it is necessary to
discover promising treatments that can serve as an alternative to
conventional therapies.
In the present study, we used genetically engineered
human neural stem cells (hNSCs) as a vehicle that can specifically
deliver anticancer genes to tumor sites. It is well known that
hNSCs have the ability to target multiple tumor types by the
interaction with chemoattractant factors (27–30).
According to a previous study, hNSCs preferentially migrated to
hypoxic areas in glioma xenografts. Hypoxia is a critical
microenvironment of cancer and hypoxia-induced migration of hNSCs
to glioma region was found to be mediated by the interactions
between chemoattractant factors such as SDF-1/CXCR4, uPA/uPAR,
VEGF/VEGFR2 (31). We confirmed the
tumor-specific tropism of hNSCs targeting A498 RCC cells in a PCR
and Transwell assay. As a result, three chemoattractant factors
such SCF, CXCR4 and VEGF were found to be expressed in the A498
cells, and the number of migrated hNSCs was 2–5 times higher in the
A498 cell-seeded groups than that in the negative control (NC)
groups. Accordingly, HB1.F3.CD and HB1.F3.CD.IFN-β cells have the
potential to minimize adverse effects due to A498-specific
targeting ability.
CD/5-FC therapy, a type of gene-directed enzyme
prodrug therapy (GDEPT), has considerable advantages compared to
conventional chemotherapy as the CD gene can convert the inactive
drug 5-FC to an effective drug near cancer cells. Moreover, we
confirmed the synergism of CD and IFN-β genes in a cell viability
assay. The cell viability of A498 cells was reduced by
approximately 10–50% by the CD gene alone according to the
concentration of 5-FC, and decreased by about 70% in
HB1.F3.CD.IFN-β cells expressing both genes.
Many previous studies have demonstrated the ability
of these stem cells to inhibit tumor growth in xenograft models of
various types of cancer (16,22).
However, few studies have investigated the inhibitory effects of
therapeutic hNSCs in metastasis models. Therefore, to verify the
metastasis-inhibitory effect of HB1.F3.CD and HB1.F3.CD.IFN-β cells
against RCC, A498 cells were injected into mice intravenously to
establish a metastasis model. After all injections were completed,
the lungs and livers, the most common metastatic sites of RCC, were
extracted. The weight of the extracted organs did not differ in
regards to the livers, but the weight of the lungs was altered by
hNSCs/5-FC. Lung weights were 120% higher in the A498 and F3 groups
than that noted in the NC group, but the CD (A498+5-FC+HB1.F3.CD)
and CD-β (A498+5-FC+HB1.F3.CD.IFN-β) groups injected with hNSCs
expressing anticancer genes showed similar results to that of the
NC group. Furthermore, as shown in Table I, the incidence of liver metastases
was significantly reduced in the CD-β group. Fluorescence analysis
revealed that there were no A498 cells in some mice, while stem
cells were present in the livers of all mice in the CD-β group.
These findings indicate that metastasis of A498 cells is inhibited
by hNSCs expressing anticancer genes in the presence of 5-FC. In
the present study, the metastasis of RCC in lung and liver by A498
RCC cells and anti-metastatic effect of hNSCs were confirmed by the
presence of A498 cells and hNSCs in lung and liver and their weight
changes. However, the identification of altered expression of
metastasis-specific markers such as cytokeratins and vimentin in
lung and liver by A498 cells and hNSCs via immunohistochemistry are
still required to be conducted in subsequent studies. In addition,
the issues concerned associated with stem cell delivery need to be
identified and solved in further studies. In the present study,
although hNSCs were found in both lung and liver where A498 cancer
cells resided in advance, the pulmonary first-pass effect occurring
during intravenous stem cell delivery (which means that most of
stem cells are trapped in the lungs before they can play a role) is
considered as a major obstacle (32). Achieving a superior stem cell effect
has been considered to involve placing the cells into the lesion to
date. Therefore, various attempts should be made to solve the
pulmonary first-pass effect and maximize the tumor-specific tropism
of hNSCs, its inherent property.
The results of this study demonstrated that
HB1.F3.CD and HB1.F3.CD.IFN-β cells with 5-FC diminished the cell
viability of RCC and retarded metastasis to other organs in a
cellular and metastasis model. In conclusion, hNSC therapy in
combination with current therapies is expected to exhibit superior
efficacy for patients suffering from metastatic RCC.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Basic Science
Research Program (2020R1A2C2006060) and the Global Research and
Development Center (GRDC) Program (2017K1A4A3014959)through the
National Research Foundation (NRF) of Korea funded by the Ministry
of Science and ICT.
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors' contributions
KCC and SUK conceived and designed the study. GSK
and SMK performed the experiments and analyzed the results. GSK
completed the draft of the stufdy and KCC revised the manuscript.
SUK and GL provided some materials and essential techniques for
this study. GL and KCC reviewed and edited the manuscript. All
authors read and approved the manuscript and agree to be
accountable for all aspects of the research in ensuring that the
accuracy or integrity of any part of the work are appropriately
investigated and resolved.
Ethics approval and consent to
participate
Experiments were conducted with approval from the
Chungbuk National University Institutional Animal Care and Use
Committee (CBNUA-1089-17-01).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests to report.
References
|
1
|
Williams RT, Yu AL, Diccianni MB,
Theodorakis EA and Batova A: Renal cancer-selective englerin A
induces multiple mechanisms of cell death and autophagy. J Exp Clin
Cancer Res. 32:572013. View Article : Google Scholar :
|
|
2
|
Curti BD: Renal cell carcinoma. JAMA.
292:97–100. 2004. View Article : Google Scholar
|
|
3
|
Gupta K, Miller JD, Li JZ, Russell MW and
Charbonneau C: Epidemiologic and socioeconomic burden of metastatic
renal cell carcinoma (mRCC): A literature review. Cancer Treat Rev.
34:193–205. 2008. View Article : Google Scholar
|
|
4
|
Basu B and Eisen T: Perspectives in drug
development for metastatic renal cell cancer. Target Oncol.
5:139–156. 2010. View Article : Google Scholar
|
|
5
|
Saini S, Liu J, Yamamura S, Majid S,
Kawakami K, Hirata H and Dahiya R: Functional significance of
secreted frizzled-related protein 1 in metastatic renal cell
carcinomas. Cancer Res. 69:6815–6822. 2009. View Article : Google Scholar
|
|
6
|
Matias M, Casa-Nova M, Borges-Costa J and
Ribeiro L: Unusual head metastasis of kidney cancer. BMJ Case Rep.
2013:bcr20132000042013. View Article : Google Scholar :
|
|
7
|
Zhang F, Shang D, Zhang Y and Tian Y:
Interleukin-22 suppresses the growth of A498 renal cell carcinoma
cells via regulation of STAT1 pathway. PLoS One. 6:e203822011.
View Article : Google Scholar :
|
|
8
|
Amato RJ: Renal cell carcinoma: Review of
novel single-agent therapeutics and combination regimens. Ann
Oncol. 16:7–15. 2005. View Article : Google Scholar
|
|
9
|
Lane BR, Rini BI, Novick AC and Campbell
SC: Targeted molecular therapy for renal cell carcinoma. Urology.
69:3–10. 2007. View Article : Google Scholar
|
|
10
|
Antonelli A, Cozzoli A, Zani D, Zanotelli
T, Nicolai M, Cunico SC and Simeone C: The follow-up management of
non-metastatic renal cell carcinoma: Definition of a surveillance
protocol. BJU Int. 99:296–300. 2007. View Article : Google Scholar
|
|
11
|
Huber BE, Austin EA, Richards CA, Davis ST
and Good SS: Metabolism of 5-fluorocytosine to 5-fluorouracil in
human colorectal tumor cells transduced with the cytosine deaminase
gene: Significant antitumor effects when only a small percentage of
tumor cells express cytosine deaminase. Proc Natl Acad Sci USA.
91:8302–8306. 1994. View Article : Google Scholar
|
|
12
|
Nawa A, Tanino T, Luo C, Iwaki M, Kajiyama
H, Shibata K, Yamamoto E, Ino K, Nishiyama Y and Kikkawa F: Gene
directed enzyme prodrug therapy for ovarian cancer: Could GDEPT
become a promising treatment against ovarian cancer? Anticancer
Agents Med Chem. 8:232–239. 2008. View Article : Google Scholar
|
|
13
|
Qiao J, Doubrovin M, Sauter BV, Huang Y,
Guo ZS, Balatoni J, Akhurst T, Blasberg RG, Tjuvajev JG, Chen SH
and Woo SL: Tumor-specific transcriptional targeting of suicide
gene therapy. Gene Ther. 9:168–175. 2002. View Article : Google Scholar
|
|
14
|
Ye L, Tao K, Yu Y and Wang G: Reduction of
G0 phase cells of colon cancer caco-2 cells may enhance
5-fluorouracil efficacy. J Biomed Res. 24:64–68. 2010. View Article : Google Scholar :
|
|
15
|
Kim KY, Kim SU, Leung PC, Jeung EB and
Choi KC: Influence of the prodrugs 5-fluorocytosine and CPT-11 on
ovarian cancer cells using genetically engineered stem cells:
Tumor-tropic potential and inhibition of ovarian cancer cell
growth. Cancer Sci. 101:955–962. 2010. View Article : Google Scholar
|
|
16
|
Yi BR, Kang NH, Hwang KA, Kim SU, Jeung EB
and Choi KC: Antitumor therapeutic effects of cytosine deaminase
and interferon-β against endometrial cancer cells using genetically
engineered stem cells in vitro. Anticancer Res. 31:2853–2861.
2011.
|
|
17
|
Garrison JI, Berens ME, Shapiro JR,
Treasurywala S and Floyd-Smith G: Interferon-beta inhibits
proliferation and progression through S phase of the cell cycle in
five glioma cell lines. J Neurooncol. 30:213–223. 1996. View Article : Google Scholar
|
|
18
|
Ren C, Kumar S, Chanda D, Kallman L, Chen
J, Mountz JD and Ponnazhagan S: Cancer gene therapy using
mesenchymal stem cells expressing interferon-beta in a mouse
prostate cancer lung metastasis model. Gene Ther. 15:1446–1453.
2008. View Article : Google Scholar :
|
|
19
|
Kim SU, Nakagawa E, Hatori K, Nagai A, Lee
MA and Bang JH: Production of immortalized human neural crest stem
cells. Methods Mol Biol. 198:55–65. 2002.
|
|
20
|
Jeong SW, Chu K, Jung KH, Kim SU, Kim M
and Roh JK: Human neural stem cell transplantation promotes
functional recovery in rats with experimental intracerebral
hemorrhage. Stroke. 34:2258–2263. 2003. View Article : Google Scholar
|
|
21
|
Kim GS, Heo JR, Kim SU and Choi KC:
Cancer-Specific inhibitory effects of genetically engineered stem
cells expressing cytosine deaminase and interferon-β against
choriocarcinoma in xenografted metastatic mouse models. Transl
Oncol. 11:74–85. 2017. View Article : Google Scholar :
|
|
22
|
Park GT, Kim SU and Choi KC:
Anti-proliferative effect of engineered neural stem cells
expressing cytosine deaminase and interferon-β against lymph
node-derived metastatic colorectal adenocarcinoma in cellular and
xenograft mouse models. Cancer Res Treat. 9:79–91. 2017. View Article : Google Scholar
|
|
23
|
Kim SU, Jeung EB, Kim YB, Cho MH and Choi
KC: Potential tumor-tropic effect of genetically engineered stem
cells expressing suicide enzymes to selectively target invasive
cancer in animal models. Anticancer Res. 31:1249–1258. 2011.
|
|
24
|
Li C, Penet MF, Winnard P Jr, Artemov D
and Bhujwalla ZM: Image-guided enzyme/prodrug cancer therapy. Clin
Cancer Res. 14:515–522. 2008. View Article : Google Scholar
|
|
25
|
Singer EA, Gupta GN, Marchalik D and
Srinivasan R: Evolving therapeutic targets in renal cell carcinoma.
Curr Opin Oncol. 25:273–280. 2013. View Article : Google Scholar
|
|
26
|
Schwartz MJ, Liu H, Hwang DH, Kawamoto H
and Scherr DS: Antitumor effects of an imidazoquinoline in renal
cell carcinoma. Urology. 73:1156–1162. 2009. View Article : Google Scholar
|
|
27
|
Lam JS, Leppert JT, Belldegrun AS and
Figlin RA: Novel approaches in the therapy of metastatic renal cell
carcinoma. World J Urol. 23:202–212. 2005. View Article : Google Scholar
|
|
28
|
Lash GE, Warren AY, Underwood S and Baker
PN: Vascular endothelial growth factor is a chemoattractant for
trophoblast cells. Placenta. 24:549–556. 2003. View Article : Google Scholar
|
|
29
|
Koshiba T, Hosotani R, Miyamoto Y, Ida J,
Tsuji S, Nakajima S, Kawaguchi M, Kobayashi H, Doi R, Hori T, et
al: Expression of stromal cell-derived factor 1 and CXCR4 ligand
receptor system in pancreatic cancer: A possible role for tumor
progression. Clin Cancer Res. 6:3530–3535. 2000.
|
|
30
|
Yi BR, O SN, Kang NH, Hwang KA, Kim SU,
Jeung EB, Kim YB, Heo GJ and Choi KC: Genetically engineered stem
cells expressing cytosine deaminase and interferon-β migrate to
human lung cancer cells and have potentially therapeutic anti-tumor
effects. Int J Oncol. 39:833–839. 2011.
|
|
31
|
Zhao D, Najbauer J, Garcia E, Metz MZ,
Gutova M, Glackin CA, Kim SU and Aboody KS: Neural stem cell
tropism to glioma: Critical role of tumor hypoxia. Mol Cancer Res.
6:1819–1829. 2008. View Article : Google Scholar
|
|
32
|
Fischer UM, Harting MT, Jimenez F,
Monzon-Posadas WO, Xue H, Savitz SI, Laine GA and Cox CS Jr:
Pulmonary passage is a major obstacle for intravenous stem cell
delivery: The pulmonary first-pass effect. Stem Cells Dev.
18:683–692. 2009. View Article : Google Scholar
|