Introduction
Head and neck squamous cell carcinoma (HNSCC) is the
sixth most common cancer type in the world, with 500,000 new cases
diagnosed on average on an annual basis. The low 5-year overall
survival rate of ~50% is associated with high rates of metastasis
and recurrence (1,2). At the time of diagnosis, >50% of
patients are in an advanced stage of the disease (3). The majority of patients are presented
with advanced HNSCC despite no clinical history of pre-malignancy
(4). Surgery is the most commonly
used therapeutic procedure, followed by adjuvant radiation or
chemotherapy according to the stage of the disease (5). However, advanced HNSCC is associated
with poor prognosis and treatment options are currently limited,
highlighting the urgent need for novel therapeutic strategies.
Resistance to chemotherapy is a major reason for treatment failure,
which is partly mediated by suppression of tumor cell apoptosis.
Therefore, elucidation of the mechanisms underlying cancer cell
death should provide valuable insights for strategies to develop
new therapeutic agents (6).
Ferroptosis, a novel type of programmed cell death
characterized by accumulation of free iron and lipid peroxidation,
was originally reported by Dixon et al in 2012 (7). This iron-dependent cell death process
is caused by the accumulation of reactive oxygen species in cells
and is distinct from apoptosis, pyroptosis, autophagy and
necroptosis (8). Iron metabolism
disorder is a risk factor for cancer demonstrated to facilitate
tumor growth. Compared with normal cells, cancer cells show iron
addiction, typically manifesting as an excessive reliance on iron
for proliferation. In fact, activation of the ferroptosis pathway
may underlie resistance to chemotherapy drugs, representing a
promising new treatment frontier for cancer (9). A number of ferroptosis-related mRNAs
have been identified as biomarkers for the diagnosis, prognosis and
treatment of several cancer types. A recent study reported that
ferritin heavy chain 1 suppressed proliferation of HNSCC cells by
influencing the expression of glutathione (GSH) and Fe2+
and promoting the process of ferroptosis, along with expression of
glutathione peroxidase 4 (GPX4) (10). Caveolin-1 is reported to stimulate
HNSCC progression by inhibiting ferroptosis (11). However, the potential roles of
ferroptosis-related mRNAs in the diagnosis, prognosis and therapy
of HNSCC are yet to be clarified.
Small ubiquitin-like modifiers (SUMOs) are highly
conserved molecules that couple to lysine residues of target
proteins as a post-translational modification process (12). SUMOylation regulates several
biological processes, including DNA damage repair, immune response,
cancer formation, cell cycle progression and apoptosis.
SUMO-specific peptidase 1 (SENP1) is a widely investigated protease
that functions in decoupling the SUMO modifier from SUMOylated
proteins (13). Various
malignancies of the stomach, liver, pancreas, cervix, kidney and
breast are associated with abnormal expression and activation of
SENP1 (14–17). While earlier findings indicated that
SENP1-mediated protein deSUMOylation exerts a protective effect on
cardiomyocyte ferroptosis (18),
expression of SENP1 and its relationship with ferroptosis in HNSCC
are yet to be established.
The acyl CoA synthetase long chain 4 (ACSL4) gene
located on human X chromosome encodes an enzyme that preferentially
binds 20-carbon polyunsaturated fatty acid (PUFA) substrates, such
as linoleic acid (19). ACSL4 plays
a key regulatory role in ferroptosis, defined as a process of cell
death associated with iron-dependent lipid peroxidation that mainly
involves phospholipids containing PUFAs. Silencing of ACSL4
inhibits generation of PUFAs, thus suppressing ferroptosis
(20,21). ACSL4 is considered a key disease
marker for HNSCC (22). To the best
of the authors' knowledge, no studies on the mechanism by which
SENP1 regulates ACSL4 and ferroptosis in HNSCC have been documented
to date.
In the present study, the prognostic model of HNSCC
was created based on expression of ferroptosis-related mRNAs. SENP1
was validated as an important biomarker that inhibits the
ferroptosis pathway in HNSCC via deSUMOylation-mediated regulation
of ACSL4, in turn, promoting proliferation and invasion of HNSCC
cells. The findings of the present study suggested a novel
ferroptosis pathway in the initiation and progression of HNSCC,
providing new functional targets to guide future therapy.
Materials and methods
Access to The Cancer Genome Atlas
(TCGA) public data
RNA-sequencing (RNA-seq) transcriptomic data and the
accompanying clinical information for HNSCC and normal control
samples were obtained from the Cancer Genome Atlas (TCGA)
(https://gdc.cancer.gov/) (23). RNA-seq data were normalized using
the Expectation-Maximization method. A total of 502 HNSCC and 44
healthy subjects were subjected to randomized analysis. TCGA
transcriptomic data were used to obtain accurate and effective gene
expression matrices using Perl language. An expression matrix of
mRNAs and lncRNAs was additionally obtained using Perl language.
The ‘limma’ package in ‘R’ was utilized to determine differential
expression of ferroptosis genes and mRNAs using the following
screening conditions: P<0.05, false discovery rate <0.05 and
log2 fold change >1. Volcano plots and heatmaps of
ferroptosis-related mRNAs were generated. ‘Corfile >0.4,
P<0.01’ was set as the screening standard and the relationship
between differentially expressed mRNAs and ferroptosis genes
determined using R 4.1.3 software (The R Foundation) (24).
Co-expression network
To determine the action sites of key mRNAs, a
co-expression network of ferroptosis genes and mRNAs was
established, with the aim of screening mRNAs significantly related
to ferroptosis-related genes. Screening conditions were ‘P<0.01,
coef >0.4’ and the network was established using R software.
Establishment and verification of risk
signatures
The differentially expressed mRNA matrix was merged
with clinical data downloaded from TCGA and a prognostic model
constructed based on differentially expressed mRNAs. Patients with
HNSCC were divided into training and test groups. The training
group, accounting for 50% of the patients was used to establish a
prognostic model, which was validated in the test group. Univariate
Cox proportional hazards regression analysis was performed and
survival-related mRNAs screened based on P-values <0.05. A least
absolute shrinkage and selection operator (LASSO) regression model
was generated using the screened mRNAs and 10-fold cross-validation
performed with 1,000 cycles to identify the point with the smallest
cross-validation error. Significantly expressed mRNAs were
identified via LASSO regression and used to generate a Cox
model.
Patients and organizations
A total of 15 pairs of tumor tissue (T) and adjacent
non-tumor tissue (N) samples were obtained from the Department of
Stomatology of the Affiliated Hospital of Xuzhou Medical University
from patients with HNSCC without preoperative treatment (Xuzhou,
China). Among these 15 patients, there were 9 female patients and 6
male patients, all aged between 40–70 years-old. The tissues were
cut and subsequently stored in liquid nitrogen. Samples were
independently identified by two pathologists and detailed
clinicopathological features were collected. All tissue samples
were obtained from July 2020 to June 2022. Informed consent was
obtained from each patient prior to any specimen-related studies
and the study was approved by the Ethics Committee of the
Affiliated Hospital of Xuzhou Medical University (approval no.
XYFY2020-KL216-01; Xuzhou, China). Among them, 7 pairs of cancer
tissues and 3 cases of adjacent tissues were used for
transcriptomics; 15 pairs of cancer tissues and adjacent tissues
were applied for reverse transcription-quantitative PCR and western
blot experiments.
Transcriptome sequencing
Total RNA was isolated using TRIzol®
reagent from cancer tissues of seven patients with HNSCC and three
adjacent tissue samples. Detection of the concentration and purity
of extracted RNA was performed via spectrophotometry on a Nanodrop
2000 instrument (Agilent Technologies, Inc.); only high-quality RNA
sample (OD 260/280=1.8~2.2, OD 260/230 ≥2.0, RIN ≥6.5, 28S:18S
≥1.0, >1 µg) was used to construct sequencing library. Agarose
gel electrophoresis was used to determine the integrity of RNA, and
RNA integrity number values were established with the Agilent 2100
Bioanalyzer system (Agilent Technologies, Inc.). RNA-seq
transcriptome library was prepared following TruSeqTM RNA sample
preparation Kit from Illumina, Inc. using 1 µg of total RNA.
Shortly, messenger RNA was isolated according to polyA selection
method by oligo(dT) beads and then fragmented by fragmentation
buffer firstly. Secondly double-stranded cDNA was synthesized using
a SuperScript double-stranded cDNA synthesis kit (Invitrogen;
Thermo Fisher scientific, Inc.) with random hexamer primers
(Illumina, Inc.). Then the synthesized cDNA was subjected to
end-repair, phosphorylation and ‘A’ base addition according to
Illumina's library construction protocol. Libraries were size
selected for cDNA target fragments of 300 bp on 2% Low Range Ultra
Agarose followed by PCR amplified using Phusion DNA polymerase
(NEB) for 15 PCR cycles. After quantified by TBS380, paired-end
RNA-seq sequencing library was sequenced with the Illumina HiSeq
xten/NovaSeq 6000 sequencer (2×150 bp read length). according to
the manufacturer's protocol. Transcriptomes from all samples were
merged to reconstruct a comprehensive transcriptome using perl
script. Normalization was performed using R software (v4.1.3) and
differentially expressed genes were identified by their P-value and
expression fold change.
Cell lines and cell culture
CAL-27 (RRID: CVCL_0030) and HOK (RRID: CVCL_YE19)
cells purchased from the Procell Life Science & Technology Co.,
Ltd. were cultured in DMEM/F12 (1:1) medium (Gibco; Thermo Fisher
Scientific, Inc.) containing 10% heat-inactivated FBS (Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin and streptomycin.
Cells were cultured in a constant temperature incubator under 5%
CO2 at 37°C. Erastin (cat. no. HY-15763) was obtained
from MedChemExpress.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
(RT-qPCR) was used to detect Senp1
transcription levels. Total RNA was extracted from cells and
tissues using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc). Extracted RNA was reverse-transcribed into cDNA
with HiScript® II QRT Super Mix for qPCR (Vazyme Biotech
Co., Ltd.) and RT-qPCR was performed using SYBR Green qPCR Master
Mix (MedChemExpress) on the Roche Light Cycler 480 II instrument
(Roche Diagnostics) using β-actin as an internal standard. Relative
gene expression was normalized to β-actin mRNA expression
and analyzed using the comparative Cq method (ΔΔCq) (25). The same thermocycling profile
conditions were used for all primers sets: 95°C for 10 min, 40
cycles at 95°C for 15 sec, and 60°C for 1 min. The primers used
were as follows: β-actin forward, 5′-CTCCATCCTGGCCTCGCTGT-3′ and
reverse, 5′-GCTGTCACCTTCACCGTTCC-3′; SENP1 forward,
5′-CCAGCATTTTAACTAACCAGGAAC-3′ and reverse,
5′-GCTAAGTTATCTGGCTGATGTGG-3′; and ACSL4 forward,
5′-CATCCCTGGAGCAGATACTCT-3′ and reverse,
5′-TCACTTAGGATTTCCCTGGTCC-3′.
Western blot analysis
Proteins were extracted from tissues and CAL-27
cells using RIPA lysis buffer (cat. no. 89900; Thermo Fisher
Scientific, Inc.) with Halt™ Protease Inhibitor Cocktail (cat. no.
78429; Thermo Fisher Scientific, Inc.). Subsequently, the BCA
method was used to quantify the tested protein. Premade adhesive
gels (4–20%) were prepared and 30 µg protein samples were loaded
per lane, marked on both sides to indicate protein molecular weight
and to distinguish left and right; then separated via sodium
dodecyl sulfate-polyacrylamide gel electrophoresis and transferred
to polyvinylidene fluoride membranes. Next, membranes were blocked
with 5% non-fat milk at room temperature for 2 h, followed by
overnight incubation at 4°C with anti-SENP1 (1:1,000; cat. no.
25349-1-AP), anti-GPX4 (1:1,000; cat. no. 67763-1-Ig), anti- solute
carrier family 7 member 11 (SLC7A11) (1:1,000; cat. no. 26864-1-AP)
and anti-β-actin (1:5,000; cat. no. 20536-1-AP; all from
Proteintech Group, Inc.) antibodies. After washing, membranes were
incubated with goat anti-mouse (1:5,000; cat. no. ab216776) or goat
anti-rabbit secondary antibody (1:5,000; cat. no. ab216773; both
from Abcam) for 2 h at room temperature. An enhanced
chemiluminescence reagent (Wuhan Servicebio Technology Co., Ltd.)
was used to visualize the protein bands. Protein bands were scanned
with an Odyssey infrared scanner (LI-COR Biosciences) and band
density was analyzed with ImageJ v1.8.0 software (National
Institutes of Health).
Cell Counting Kit-8 (CCK-8) assay
The CCK-8 assay (Beyotime Biotechnology Inc.) was
employed to detect cell activity. In brief, CAL-27 cells were
seeded into a 96-well plate at a density of 1×104
cells/well and cultured for 3 days. Next, cells were transfected
with Senp1 short hairpin RNA (shRNA) or overexpression
plasmid and survival rates were assessed via CCK-8 colorimetry at
72 h. In brief, CCK-8 was added to each well and was incubated for
1 h at 37°C. CCK-8 solution accounted for ~10% of the volume of the
culture medium. OD values were measured at 450 nm using a
microplate reader (BioTek Instruments, Inc.).
Wound healing assay
Transfected cells were cultured to ~90% confluence
in 12-well plates and a wound area introduced using a sterile
200-µl pipette tip. After washing twice with PBS, cells were
cultured in DMEM/F12 without FBS for 48 h. Cell images were
obtained using fluorescence microscopy (IX73; Olympus Corporation;
scale bars, 100 µm). Wound sizes were measured with ImageJ and
calculated as a percentage (from 0 h to 48 h).
Transwell assay
Cells were collected and resuspended in medium
(~1×105 cells/ml). An aliquot (200 µl) of the cell
suspension was placed in the upper chamber of a Transwell (8-µm
pore size; MilliporeSigma), which was inserted into a 24-well
plate. After incubation in RPMI-1640 containing 10% (v/v) FBS for
24 h at 37°C, cells on the upper membrane of the insert were
removed gently with a cotton swab while those migrating to the
bottom surface were fixed with 4% cold methanol for 30 min and
stained with 0.1% crystal violet for 1 h. The number of cells on
the bottom surface was counted and quantified using fluorescence
microscopy (IX73; Olympus Corporation; scale bars, 100 µm).
Lipid peroxidation assay
The TBA method kit (cat. no. A003; Nanjing Jiancheng
Bioengineering Institute) was used to measure the concentration of
MDA in cells. Cells were mixed well in RIPA lysis buffer. The
reagent was added to the centrifuge tube according to the
manufacturer's instructions and thoroughly mixed, followed by
boiling in a 95°C water bath for 40 min and cooling at room
temperature. After centrifugation at 1,600 × g for 10 min at 4°C,
absorbance of the supernatant was measured at 532 nm to calculate
malondialdehyde (MDA).
Iron assay
An iron assay kit (cat. no. ab83366; Abcam) was
employed to evaluate the relative intracellular iron levels.
According to the requirements of the reagent supplier, the prepared
iron probe, reducing agent, standard diluent (2–10 nmol/well) and
test sample (cell lysate) were mixed. Next, 5 µl iron reducing
agent was added to each standard well and incubated at 37°C for 30
min, followed by addition of 100 µl iron probe to each well at 37°C
and incubation in the dark for 60 min. The sample was immediately
evaluated with a colorimetric enzyme marker (593 nm). The total
iron content of test samples was determined from the standard
curve.
Immunohistochemistry (IHC)
Slices were immunohistochemically stained as
experimental procedure. Coverslips seeded with tumor cells or
tissue cryo-sections were fixed with 4% paraformaldehyde for 30
min, and then permeabilized with 0.5% Triton X-100 for 10 min.
After 2 h of incubation with goat serum (cat. no. 16210064; Thermo
Fisher Scientific, Inc.) at 37°C, Consecutively, 4-µm-thick
sections were analyzed using primary antibodies against SENP1
(1:1000; cat. no. 25349-1-AP; Proteintech Group, Inc.) at 4°C.
Then, sections were incubated with Alexa Fluor (cat. no. R37116;
Thermo Fisher Scientific, Inc.) secondary antibody for 1 h at 37°C.
Images were obtained using fluorescence microscopy (IX73; Olympus
Corporation; scale bars 200 µm).
GSH assay
A GSH colorimetric assay kit (cat. no. CS0260;
MilliporeSigma) was used to determine relative GSH levels. This
experiment used 5% of 5-thiosalicylic acid solution (SSA), GSH
standard solution, working mixture, NADPH solution and test sample
(cell extract). SSA (5%) was used as the reagent blank, a total of
10 µl GSH standard solution and 10 µl of test sample were added to
a separate well. An aliquot of 150 µl working mixture was added to
each well and was incubated at room temperature for 5 min. Finally,
a total of 50 µl NADPH solution was added to each well and mixed.
After absorbance of each well was determined at 412 nm, the GSH
concentration was calculated according to the standard curve.
Lentiviral transfection
To construct stable SENP1-knockdown CAL-27 cell
line, shRNA specifically targeting SENP1 was designed by the Thermo
Fisher Scientific, Inc. online tool (https://rnaidesigner.thermofisher.com/rnaiexpress/construct.do?pid=−431479708000205999)
and cloned into the lentiviral pLVX-sh1 vector. SENP1
overexpressing CAL27 cells were cloned into the lentivirus
pLVX–IRES-puro vector. Lentiviruses were synthesized by Genechem
Shanghai Co., Ltd. Cells were seeded in cell culture dishes and
used to transfect lentiviral with different MOI values (10, 20, 30)
at 30% density. On the second day of transfection, cells were
cultured in DMEM containing 10% FBS. After 3 days, the transfection
effect was observed under fluorescence microscopy (IX73; Olympus
Corporation). Then, transfected cells were passaged and cultured
with puromycin (5 ng/µl; Shanghai Yeasen Biotechnology Co., Ltd.)
used to selected cells for 1 week. Subsequently, stable cell lines
were cultured in DMEM containing 10% FBS and puromycin (2 ng/µl).
The sequences were as follows: sh-SENP1,
5′-CTCGATGTCTTAGTTCCAGTA-3′; and sh-NC,
5′-GGGACTCGATTCCCACGAATC-3′.
Co-IP
The cells were lysed with 250 µl NP-40 lysis buffer
(cat. no. P0013F; Beyotime Institute of Biotechnology). The cell
lysates were centrifuged at 4,800 × g for 5 min to remove the cell
fragments. The protein stock solution, with anti-SENP1 or ACSL4
antibody bound to 25 µl magnetic beads, was incubated overnight at
4°C. The small pallet was washed three times using the pyrolysis
method and sodium dodecyl sulfate samples were boiled for 5 min.
SENP1, ACSL4 and SUMO1 levels were analyzed via western blot
analysis using anti-SENP1 (1:1,000; cat. no. 25349-1-AP),
anti-ACSL4 (1:1,000; cat. no. 22401-1-AP) and anti-SUMO1 (1:1,000;
cat. no. 10329-1-AP; all from Proteintech Group, Inc.) antibodies,
respectively.
Statistical analysis
All data were analyzed with Graph Pad Prism 7.0
software (GraphPad Software; Dotmatics). Continuous data are
presented as the mean ± standard deviation and differences between
experimental groups were analyzed using one-way ANOVA or unpaired
two-tailed Student's t-test. In addition, to estimate the risk
differences between the main outcome groups, a post hoc analysis
was conducted using Bonferroni's test following ANOVA. Survival
analysis was performed using the Kaplan-Meier method(https://ualcan.path.uab.edu/). The screening of key
genes comes from the VENN diagram method (https://bioinfogp.cnb.csic.es/tools/venny/index.html).
Differences were considered statistically significant at P<0.05.
Experiments were repeated three times and representative
experiments are shown.
Results
Expression of ferroptosis-related
mRNAs
Data from a total of 44 normal and 502 tumor samples
were obtained from TCGA database. Based on the co-expression
patterns of 40 ferroptosis genes (Table SI) and differentially expressed
mRNAs (|log2FC|>1 and P<0.01), a total of 457
ferroptosis-related mRNAs were obtained (correlation coefficients
>0.4 and P<0.001). All mRNAs and ferroptosis genes were
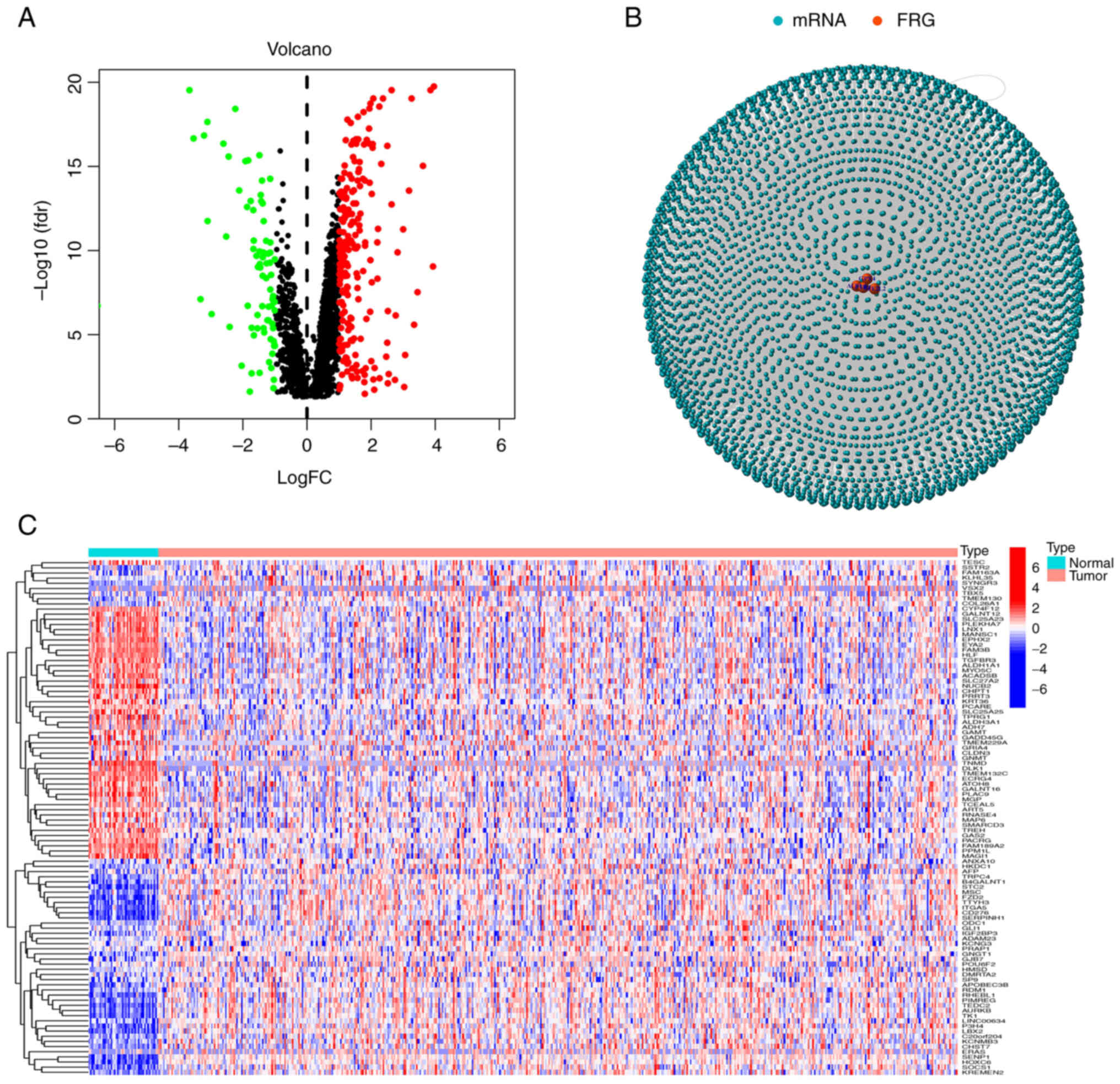
positively correlated. Volcano plots and heat maps were used to
examine expression patterns in normal and tumor groups (Fig. 1A and C) and the relationship between
mRNAs and ferroptosis genes was ultimately visualized via network
graphs (Fig. 1B).
Construction of an mRNA prognostic
model
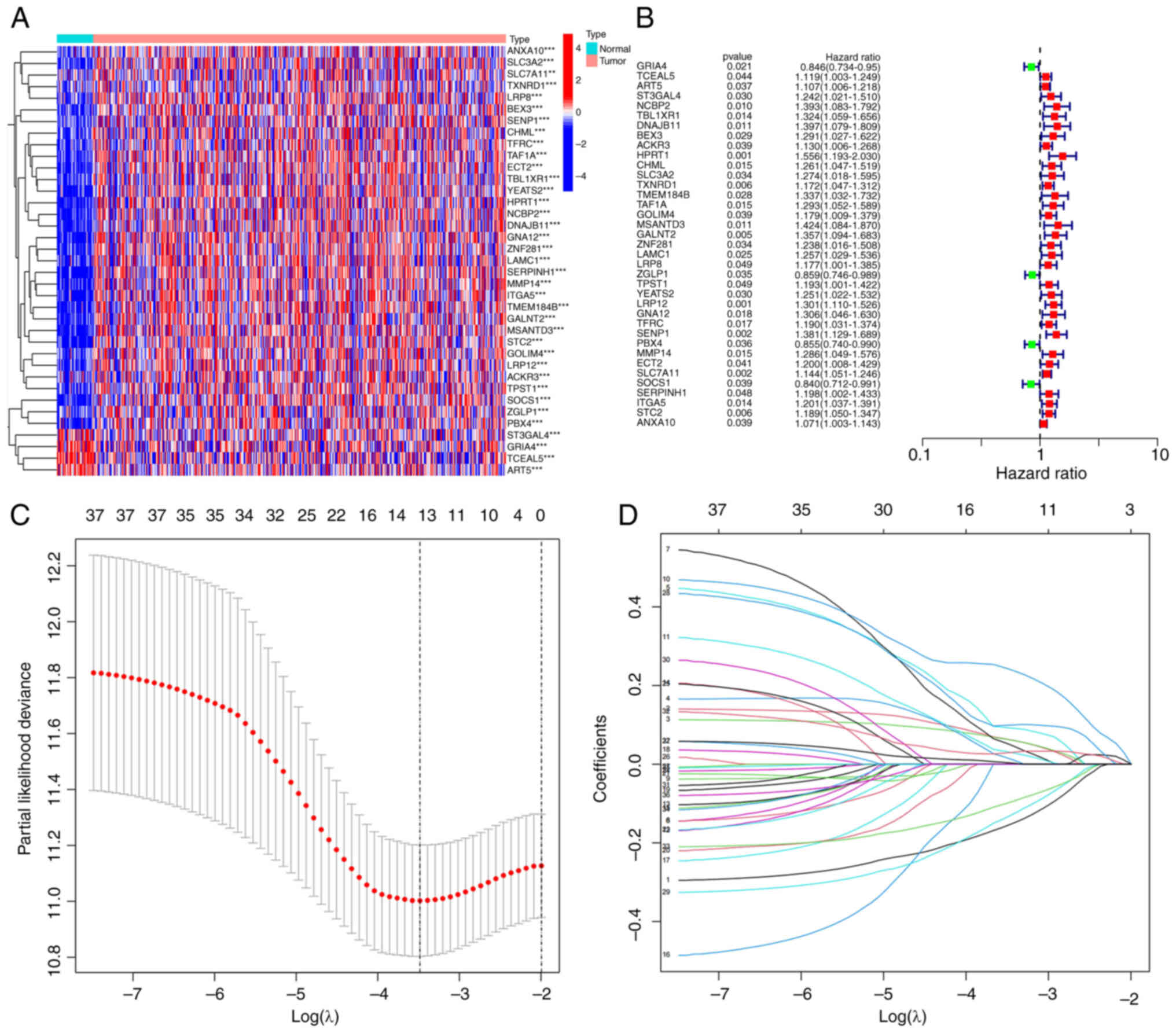
Using univariate Cox proportional hazards regression
analysis, 32 significant ferroptosis-related mRNAs among the 457
were identified and a heatmap was generated (Fig. 2A and B). The significant mRNAs
obtained via univariate Cox analysis were combined with clinical
data to construct LASSO regression curves (Fig. 2C). Cross-validation was performed
and the point of the smallest cross-validation error determined.
Ultimately, 13 ferroptosis-related mRNAs were screened (Fig. 2D). Following optimization of the Cox
model, seven mRNAs (SENP1, PBX4, SOCS1, DNAJB11, ART5,
TCEAL5 and GRIA4) were selected. Simultaneous
transcriptome analysis of HNSCC and corresponding paracancerous
tissues led to the identification of 427 differential genes (199
upregulated and 228 downregulated) (Fig. S1A). When these 427 genes were
analyzed with the VENN map with the aforementioned seven genes, a
key gene, SENP1, was obtained (Fig. S1B). Thus, SENP1 was selected
to start further verification.
Survival analysis and construction of
risk curves
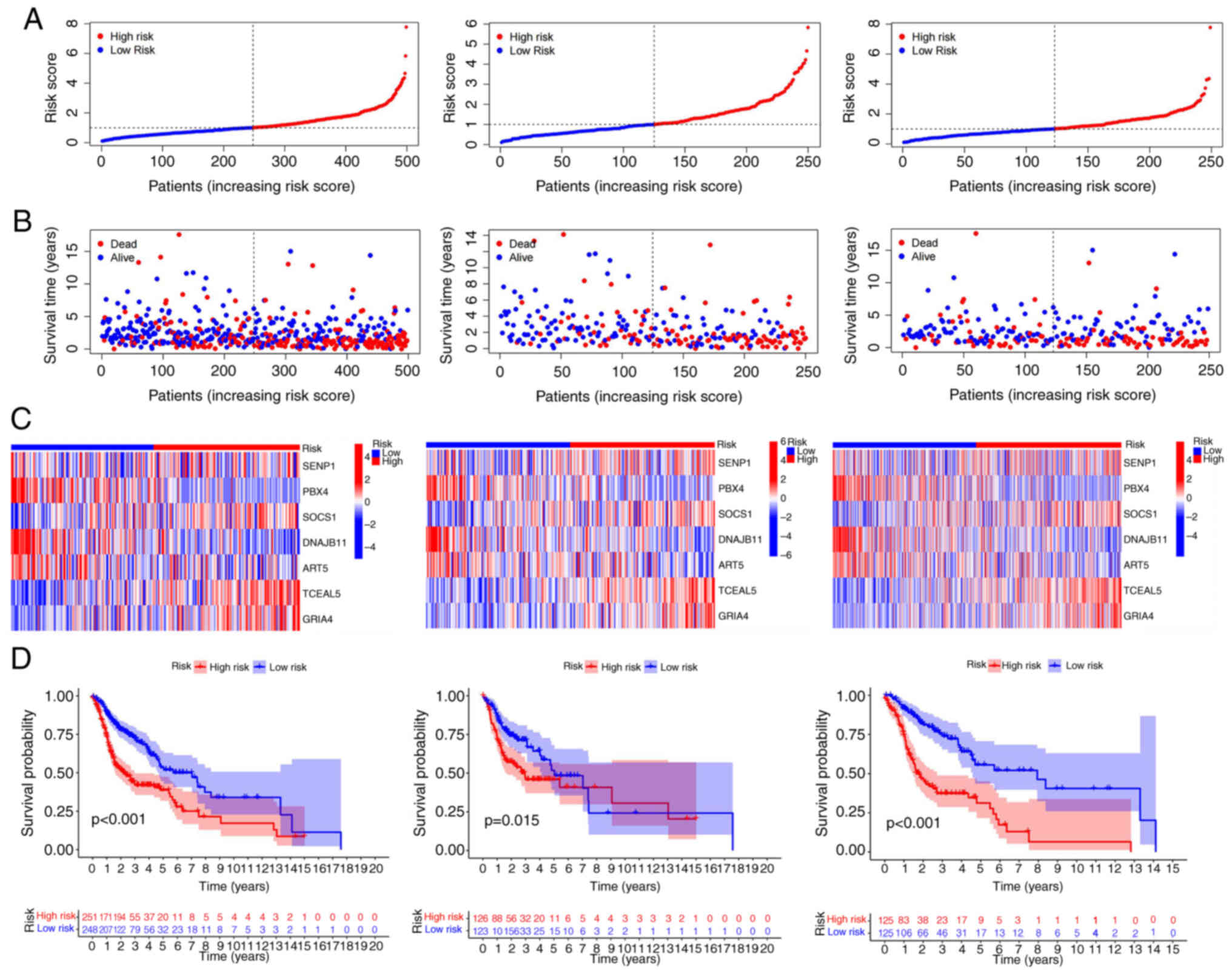
According to the risk score, a Kaplan-Meier curve of
overall survival (OS) probability of individuals with
ferroptosis-related genes was constructed for the training, test
and all groups. The survival rate was significantly lower in high
risk than low risk patients for all groups (Fig. 3D). Similar results were obtained for
age, sex, stage, type and tumor-node-metastasis (TNM) stage
(Fig. S2). Since HNSCC is prone to
lymph node metastasis, ‘N’ was specifically taken out as an
independent clinical prognostic factor. Effective independent
prognostic factors were screened via univariate Cox analysis
(screening condition, P<0.05). The risk score of the high-risk
group was obviously greater whereas survival time was markedly
lower than that of the low-risk group in the training, test and all
groups. The differences in expression levels of the seven
ferroptosis-related mRNAs in prognostic models of the high risk as
well as low risk groups were analyzed using heat maps (Fig. 3A-C).
Construction of a nomogram and
independent prognostic analysis
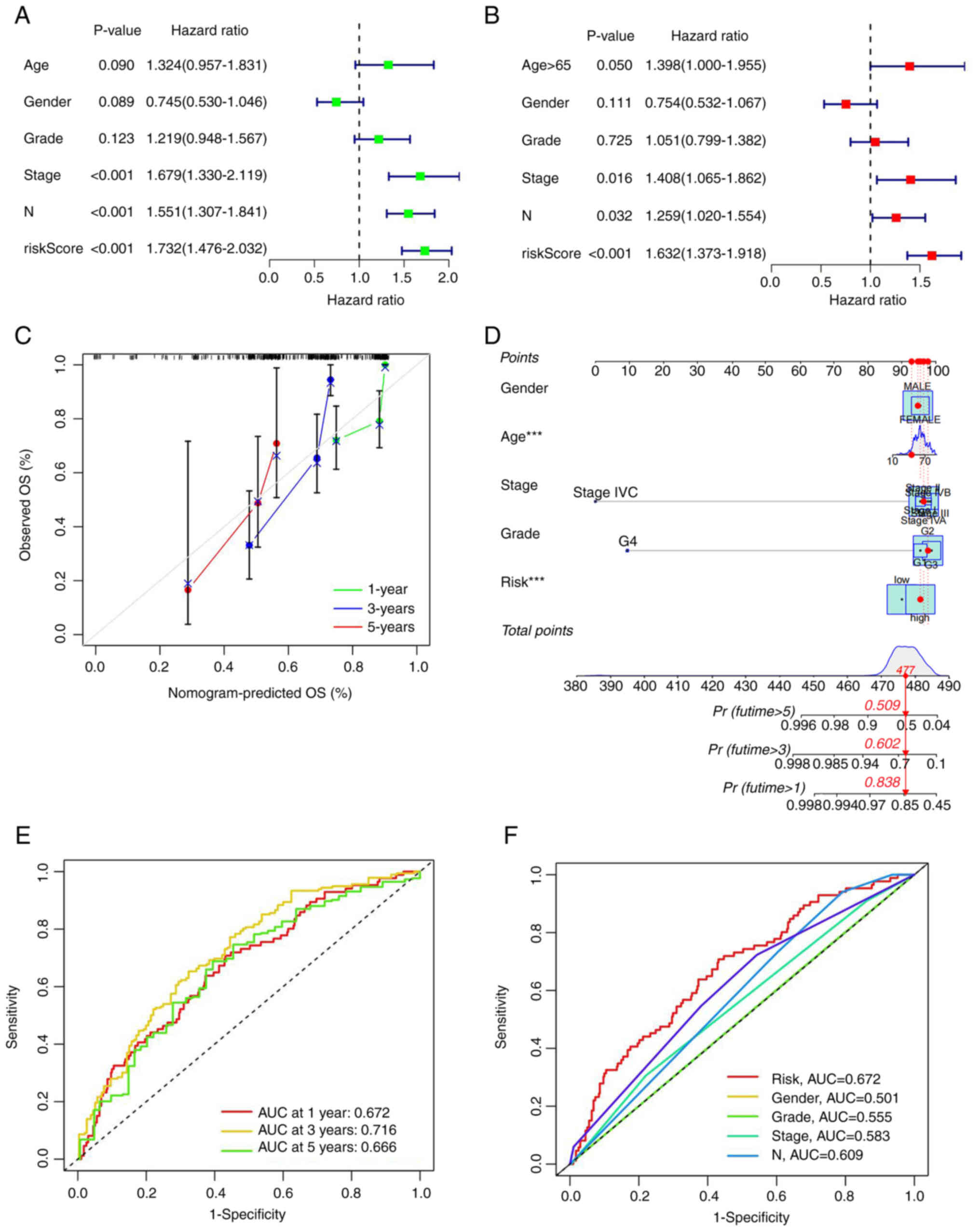
As aforementioned, effective independent prognostic
factors were screened via univariate Cox analysis (screening
condition, P<0.05). The hazard ratio (HR) for the risk score was
1.732 and 95% confidence interval (CI) was 1.476–2.032. ‘Stage’ and
‘N’ were identified as two additional prognostic parameters
(Fig. 4A). In multivariate Cox
analysis, HR of the risk score was 1.623 with 95% CI of
1.373–1.918. Age, stage and N could be used as additional
prognostic parameters (Fig. 4B). A
nomogram was generated according to the clinical risk factors
identified, specifically, age, sex, stage, grade, TNM stage and
comprehensive risk scores were obtained to assess 1-, 3- and 5-year
survival rates of patients with HNSCC (Fig. 4C). Calibration curves for 1-, 3- and
5-year survival rates were additionally generated to evaluate the
reliability of the nomogram (Fig.
4D). Receiver operation curve (ROC) analysis was utilized to
validate the preciseness of the model for predicting patient
survival. The predictive values of area under the curve (AUC) were
0.672, 0.716 and 0.666 for 5-, 3- and 1-year survival, respectively
(Fig. 4E). All AUC values were
>0.65, validating the accuracy of the prognostic model in
survival prediction. Moreover, AUCs were larger than those obtained
from other clinical traits, suggesting that accuracy of the
proposed model was greater than that of other clinical traits
(Fig. 4F). The data clearly
suggested that the seven mRNA ferroptosis-related models
constructed by the authors were closely associated with the
pathogenesis of HNSCC.
SENP1 is overexpressed in HNSCC
Kaplan-Meier method indicated that high SENP1
expression was generally associated with low survival rates
(Fig. S3). SENP1 mRNA that
displayed the highest association with ferroptosis was selected for
further verification (Fig. S4).
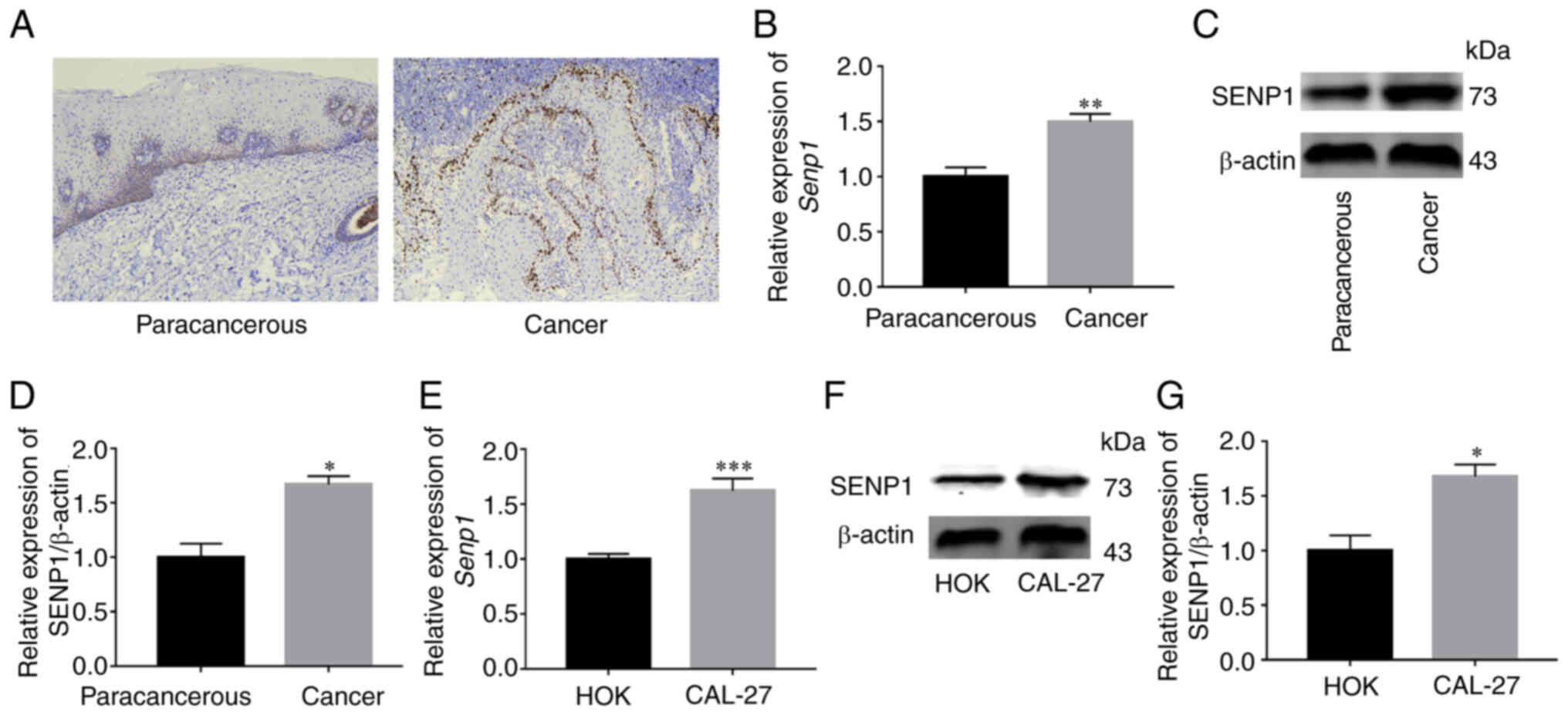
Subsequently, the expression of SENP1 in HNSCC tissue was examined
via IHC, western blotting and RT-qPCR and was revealed to be
significantly higher than that in paracancerous tissue (Fig. 5A-D). Simultaneously, the expression
of SENP1 in CAL-27 and HOK was examined via RT-qPCR and western
blot analyses. Both sets of experiments revealed higher expression
of SENP1 in the CAL-27 cell line (Fig.
5E-G). The results clearly indicated that SENP1 was
overexpressed in HNSCC.
SENP1 accelerates HNSCC cell
proliferation and migration
Stable knockout or overexpression cell lines using
shRNA and SENP1 overexpression lentivirus, respectively, were first
established with a view to investigate the biological role of SENP1
in HNSCC (Fig. 6A). Overexpression
and knockdown efficiency of SENP1 was validated at both protein and
transcriptional levels (Fig. 6C-E).
Data from the CCK-8 assay revealed increased CAL-27 cell
proliferation under conditions of overexpression of SENP1 (Fig. 6B). Conversely, knockdown of SENP1
significantly inhibited the viability of CAL-27 cells, suggesting
that SENP1 was an important regulator of HNSCC cell proliferation.
Transwell assays revealed that compared with the control group, the
number of migratory HNSCC cells transfected with sh-SENP1 decreased
while upon overexpression of SENP1, the number of migratory HNSCC
cells was significantly increased (Fig.
6F and G). The results of the wound healing assay consistently
demonstrated that SENP1 promoted cell migration. Compared with the
negative control group, the migration ability of oe-SENP1 CAL-27
cells was significantly increased while sh-SENP1 induced a decrease
in cell migration ability (Fig. 6H and
I). The collective findings indicated the pivotal role of SENP1
in the progression of HNSCC.
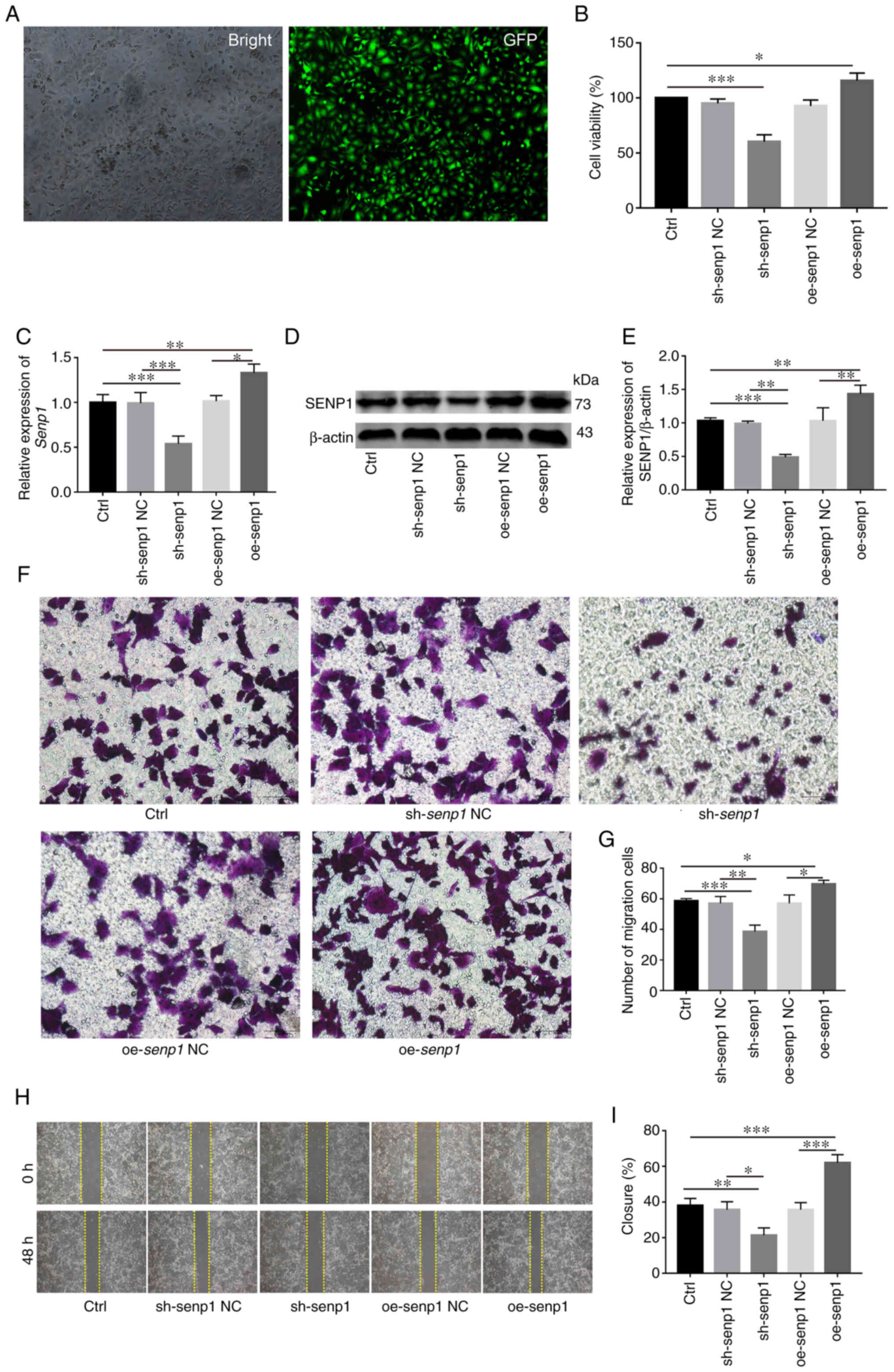 | Figure 6.SENP1 promotes the proliferation,
migration and invasion of HNSCC in vitro. (A and C) reverse
transcription-quantitative PCR revealed transfection efficiency of
sh-SENP1 and oe-SENP1. (B) Analysis and evaluation of the impact of
knocking down or overexpressing SENP1 on the proliferation ability
of HNSCC cells through Cell Counting Kit-8. (D and E) Western
blotting verified sh-SENP1l and oe-SENP1 transfection efficiency.
(F and G) Transwell assays were conducted to examine the cell
migration and invasion ability in HNSCC cells. (H and I) Wound
healing assay was employed to detect the cell migration ability of
HNSCC cells. Unpaired student's t-test for two-group comparison:
*P<0.05, **P<0.01 and ***P<0.001. One-way ANOVA for
multi-group comparisons: *P<0.05, **P<0.01 and ***P<0.001.
Bonferroni's test for multi-group comparisons: *P<0.05,
**P<0.01 and ***P<0.001. SENP1, SUMO-specific peptidase 1;
HNSCC, head and neck squamous cell carcinoma; Ctrl, control; shRNA,
short hairpin RNA; NC, negative control; oe, overexpressing. Scale
bars, 100 µm. |
SENP1 is an important regulator of
ferroptosis pathway in HNSCC
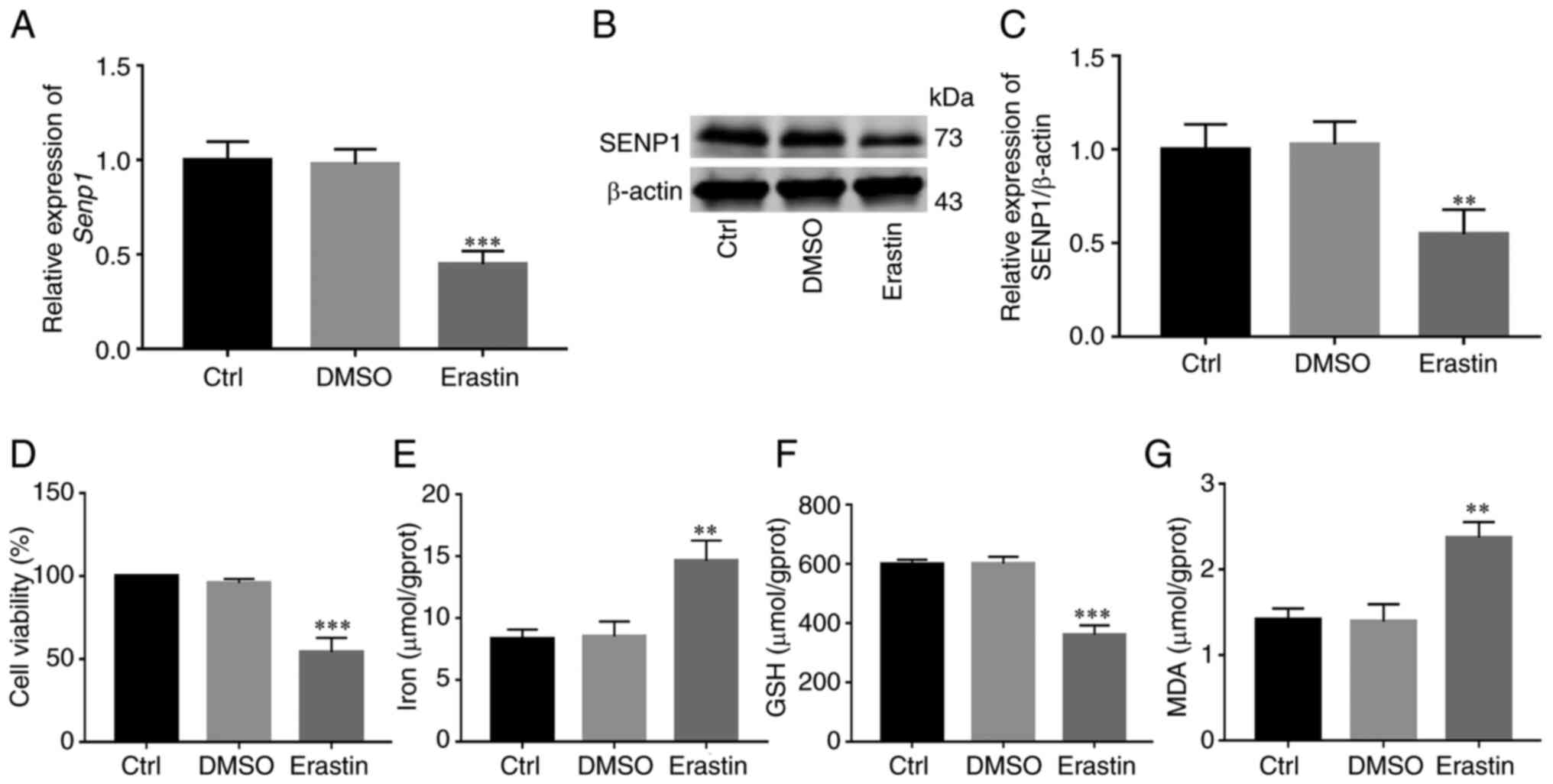
Erastin is a common inducer of ferroptosis.
Following activation of ferroptosis of HNSCC cells with erastin,
changes in SENP1 expression and its inhibitory mechanisms of action
on growth of HNSCC cells were examined. Initial RT-qPCR and western
blotting experiments revealed significantly reduced SENP1
expression in cells treated with erastin compared with those
treated with DMSO (Fig. 7A-C). To
further determine whether inhibition of cell growth was caused by
ferroptosis, under conditions of erastin-induced ferroptosis (10
µM, 24 h), the levels of iron death-related indicators (cellular
iron, GSH and MDA) in HNSCC cells were detected. Notably, viability
of HNSCC cells and levels of GSH were reduced (Fig. 7D and F), whereas levels of iron and
MDA, the end product of lipid peroxidation, were significantly
increased in cells (Fig. 7E and G).
Simultaneously, overexpression and knockdown efficiency of SENP1 at
the protein and transcriptional levels were validated under
conditions of ferroptosis induction (Fig. 8A-C). Following knockdown of SENP1,
compared with the NC group, cell proliferation ability reduced, the
end products of cellular iron and lipid peroxidation, MDA, were
significantly increased while that of endogenous GSH was
significantly decreased (Fig.
8D-G). In addition, the corresponding level of ferroptosis
under overexpression of SENP1 was reduced (Fig. 8F). The findings of the
aforementioned experiments clearly indicated that SENP1 served as
an important regulatory factor of ferroptosis in HSNCC cells.
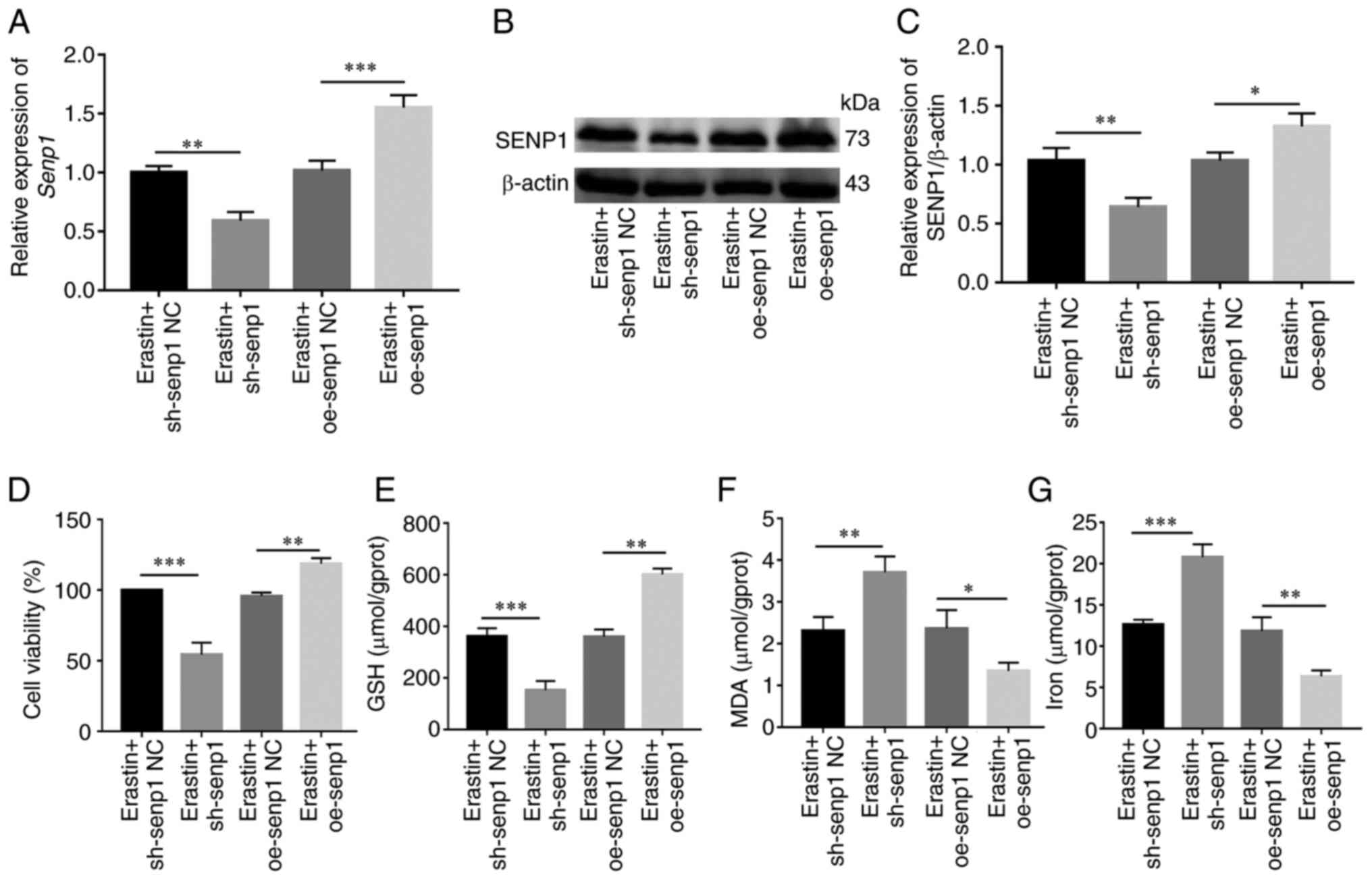 | Figure 8.SENP1 regulates ferroptosis in HNSCC
cells. (A) Reverse transcription-quantitative PCR detected SENP1
expression levels after erastin (10 µm, 24 h) treatment in sh-SENP1
and oe-SENP1. (B and C) Western blotting detected SENP1 expression
levels after erastin (10 µm, 24 h) treatment in sh-SENP1 and
oe-SENP1. (D-G) After erastin (10 µm, 24 h) treatment in HNSCC
cells, SENP1 regulated the (D) cell viability, (E) GSH, (F) MDA and
(G) levels of iron. *P<0.05, **P<0.01 and ***P<0.001.
SENP1, SUMO-specific peptidase 1; HNSCC, head and neck squamous
cell carcinoma; Ctrl, control; shRNA, short hairpin RNA; NC,
negative control; oe, overexpressing; GSH, glutathione; MDA,
malondialdehyde. |
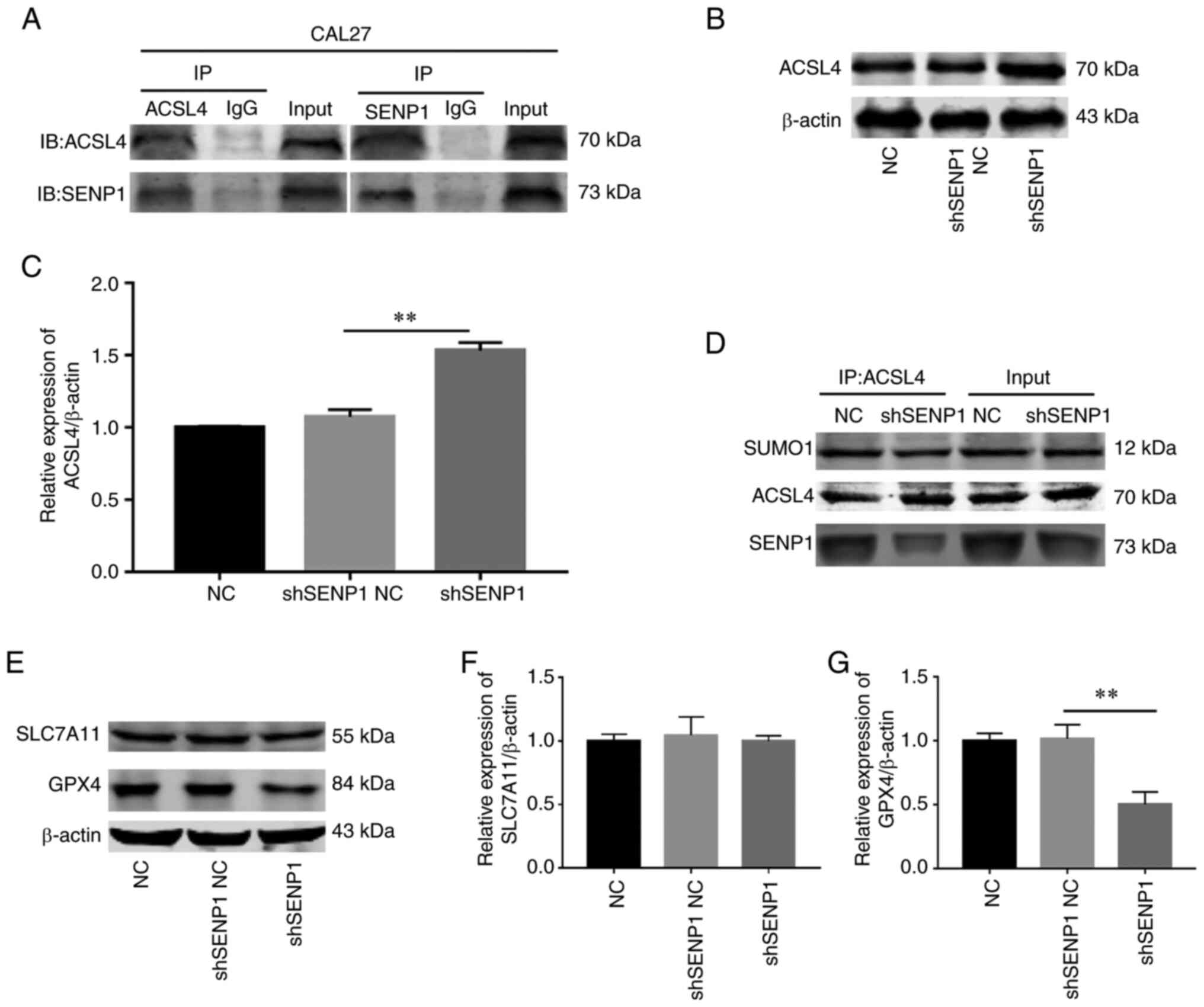
Silencing SENP1 propels ferroptosis by
suppressing deSUMOylation of ACSL4 to enhance its protein
stability
ACSL4 is a key enzyme in the lipid peroxidation
procedure that is highly expressed to promote ferroptosis. While
previous research has revealed that SENP1 regulates the process of
ferroptosis, the association between SENP1 and ACSL4 has not been
fully elucidated. It was proposed by the authors that SENP1 reduces
the stability of ACSL4 protein via deSUMOylation in HNSCC. Co-IP
experiments in CAL-27 cells revealed a significant direct
association between SENP1 and ACSL4 (Fig. 9A). Further TCGA and western blot
analysis disclosed a negative association between the two molecules
(Fig. 9B and C). To further
validate this hypothesis, IP experiments on SUMO1 conjugated to
ACSL4 in the control and gene knockout groups were conducted.
Notably, the SUMO1 level of ACSL4 was decreased in the gene
knockout group (Fig. 9D).
Furthermore, the impact of SENP1 knockdown on the iron death
pathway was evaluated. Protein immunoblotting was employed to
determine the levels of key proteins, GPX4 and SLC7A11, of the iron
death pathway in samples from each group following SENP1 knockdown,
which revealed that the GPX4 level was decreased while that of
SLC7A11 remained unchanged (Fig.
9E-G). These data supported a direct correlation between SENP1
and ACSL4. Knockdown of SENP1 may promote SUMO1 conjugation and
stability of ACSL4, further consuming GPX4 levels and ultimately,
facilitating ferroptosis.
Discussion
Ferroptosis-related mRNAs are associated with
progression of HNSCC (26). In the
present study, the TCGA database was utilized to explore
ferroptosis-related mRNAs in HNSCC. LASSO regression was merged
with clinical prognosis data to establish a panel containing seven
ferroptosis-related signature mRNAs, which were verified as
prognostic markers of HNSCC (Fig.
S4). The preciseness of the model was verified according to the
AUC values of ROC curves of the risk group. Transcriptome analysis
was employed for screening of the key ferroptosis-related SENP1
gene. High expression of SENP1 was associated with poor prognosis
and severe malignant progression of HNSCC. Compared with
non-neoplastic tissues, expression of SENP1 was elevated in HNSCC,
indicating its involvement in tumorigenesis. Notably, silencing of
SENP1 had a number of critical effects, including reduced
proliferation and migration of HNSCC cells, increased absorption of
iron ions, enhanced SUMOylation and stability of ACSL4, suppression
of GPX4 activity, and promotion of GSH metabolism, resulting in
accumulating cellular lipid peroxidation, and ultimately,
ferroptosis. The collective results suggested that SENP1 deficiency
promoted ferroptosis and inhibited tumor progression by reducing
SUMOylation of ACSL4 in HNSCC.
In recent years, research on ferroptosis was mainly
focused on the underlying molecular mechanisms and drug resistance
while related protein markers have been relatively overlooked
(27,28). To address this gap in knowledge, the
present study investigated the relationship between protein
biomarkers and ferroptosis in relation to the potential mechanisms
underlying HNSCC occurrence. SENP1 monitors cytoactivity by
eliminating the conjugation of SUMO proteins from substrate
proteins (13). SENP1 targets
several SUMO1-modified proteins with effects on cell proliferation,
migration and induction of macrophage inflammation and is closely
associated with the development of several cancer types (14,29).
Previous findings clearly suggested that SENP1 promotes cell
migration, invasion and proliferation by regulating DNA methylation
of E-cadherin in osteosarcoma (30). Similarly, SENP1 increases androgen
sensitivity to promote prostate cancer growth (31) and enhances invasion and metastasis
activities of triple-negative breast cancer through elevating CSN5
transcription mediated by GATA1 deSUMOylation (15). Overexpression of SENP1 in HNSCC was
originally uncovered via cDNA microarray analysis (32). The results of the present study
indicated that SENP1 was an important oncogene significantly
upregulated in HNSCC. High expression of SENP1 was positively
related to poor survival outcomes and effectively facilitated
proliferation and invasion of HNSCC cells.
ACSL4, a lipid droplet-related protein that converts
free long-chain fatty acids into fatty acid acyl coenzyme A esters,
is involved in both anabolic (fatty acid synthesis and lipogenesis)
and catabolic pathways (lipolysis and fatty acid metabolism) of
β-oxidation. ACSL4-encoded enzymes have a preferential affinity for
20-carbon PUFA substrates, such as adrenaline (ADA), catalyzing
their conversion to AACoA and ADA CoA while inhibiting the process
of reducing GSH inhibiting phospholipid synthesis and facilitating
lipid peroxidation to promote the ferroptosis process (20). A number of studies suggested that
SENP1 is a novel regulator of ferroptosis in cancer cells.
Silencing of SENP1 is reported to reduce the stability of A20 to
promote ferroptosis and apoptosis in lung cancer cells (33). Upregulation of SENP1 in myocardial
cells has also been demonstrated to inhibit erastin-induced
ferroptosis (18). However, the
relationship between SENP1 and ferroptosis in HNSCC is currently
unclear. In the present study, the ferroptosis inducer, erastin,
significantly promoted SENP1 expression in HNSCC cells. Low
expression of SENP1 in cells promoted ACSL4, thereby leading to
GSH-mediated growth of oxidized phospholipids; while promoting the
iron death pathway, lipid peroxidation (MDA) and the expression of
iron and suppressing the expression of GSH, led to promotion of
ferroptosis. However, the scope of the present study was limited to
the effect of SENP1 on HNSCC cells and its role in other cancer
types requires further exploration.
SENP1 is an important enzyme that induces
deSUMOylation (12).
Ubiquitination-mediated modification involves degradation of
proteasomes, primarily through degrading one or more proteins, to
achieve the desired effect. SUMOylation mainly affects the process
of protein stability (34,35), with SUMO modification playing a
critical role in complex protein regulatory networks. Disruption of
SUMOylation can promote the progression of a number of diseases and
tumors, highlighting the utility of SUMO as a potential therapeutic
target for cancer (36). SENP1
reduces the stability of PTEN proteins through regulating their
SUMOylation and ubiquitination. A different study demonstrated that
SENP-1 regulated glycolysis in prostate cancer cells by stabilizing
HIF-a (37). Moreover, SENP1 can
actively regulate the stability and activity of c-Myc in breast
cancer through deSUMOylation (38)
and reduce the stability of ACSL4 protein by deSUMOylation in
hypoxic environments, inhibiting ferroptosis of myocardial cells
(33). However, the expression
patterns and related mechanisms of associations between SENP1 and
ACSL4 in HNSCC have been largely unexplored to date. Data from the
present study revealed that SENP1 played an important role in HNSCC
progression by modifying ACSL4 through deSUMOylation.
SENP1-mediated ASCL4 phosphorylation affected activity, thereby
promoting GPX4 expression to inhibit the process of ferroptosis in
HNSCC.
In the present study, SENP1 was identified as a
novel oncogene with a key role in progression of HNSCC and
ferroptosis. SENP1 decreased the stability of ACSL4 protein through
deSUMOylation and indirectly inhibited its influence on
phospholipid metabolic pathways downstream of ferroptosis.
Additionally, SENP1 could serve as an important biomarker for
prognosis of patients. However, it must be noted that the present
study has a number of limitations that need to be addressed.
Additional experiments are necessary to verify SENP1-mediated
regulation of SUMOylation of ACSL4. Establishment and
characterization of mutant colonies based on predicted SUMO sites
of ACSL4 should be performed to confirm the finding that SENP1
regulates the SUMOylation domain of ACSL4 to enhance its stability.
Further research is required to facilitate the development of SENP1
as a clinical biomarker of HSNCC and potentially other cancer
types.
In conclusion, SENP1 deficiency promoted ferroptosis
and inhibited tumor progression through reducing SUMOylation of
ACSL4. The collective results of the present study supported the
utility of SENP1 as an effective predictive biomarker for targeted
treatment of HNSCC.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by Jiangsu College Natural
Science Foundation of China (grant no. 12KJB320015), Xuzhou Science
and Technology Planning Project (grant no. KC15SHO15) and Six
Talent Peaks Project in Jiangsu (grant no. 2014-wsw-068)
Availability of data and materials
The datasets generated and/or analyzed during the
current study are available in the Meiji Biotechnology Service
Network (https://report.majorbio.com/medical/species_general/task_id/rf7i_7cbvmpp12cp9c3a5fbn4u5).
The datasets and The Cancer Genome Atlas (https://gdc.cancer.gov/) repositories. The datasets
used and/or analyzed during the current study are available from
the corresponding author on reasonable request.
Authors' contributions
XX and ZF conceived and designed the study and
participated in the processing of the experiments, drafting and
revising the first draft. YM conducted data analysis and
composition. FD conducted data collection. TG conducted a design
for bioinformatics analysis. JZ participated in design of all
experiments and wrote the final manuscript determination. All
authors read and approved the final manuscript. TG and JZ confirm
the authenticity of all the raw data.
Ethics approval and consent to
participate
Informed consent was obtained from all the patients
and the study was approved by the Ethics Committee of the
Affiliated Hospital of Xuzhou Medical University (approval no.
XYFY2020-KL216-01; Xuzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
HNSCC
|
head and neck squamous cell
carcinoma
|
|
GPX4
|
glutathione peroxidase 4
|
|
SUMO
|
small ubiquitin-like modifiers
|
|
SENP1
|
SUMO-specific peptidase 1
|
|
ACSL4
|
acyl CoA synthetase long chain 4
|
|
PUFA
|
polyunsaturated fatty acid
|
|
LASSO
|
least absolute shrinkage and selection
operator
|
|
cDNA
|
complementary DNA
|
|
CCK-8
|
Cell Counting Kit-8 assay
|
|
TCGA
|
The Cancer Genome Atlas
|
|
GSH
|
glutathione
|
|
TNM
|
tumor-node-metastasis
|
|
CI
|
confidence interval
|
|
HR
|
hazard ratio
|
|
ROC
|
receiver operation curve
|
|
AUC
|
area under the curve
|
|
AA
|
anthotetraenoic acid
|
|
ADA
|
adrenaline
|
References
|
1
|
Johnson DE, Burtness B, Leemans CR, Lui
VWY, Bauman JE and Grandis JR: Head and neck squamous cell
carcinoma. Nat Rev Dis Primers. 6:922020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Solomon B, Young RJ and Rischin D: Head
and neck squamous cell carcinoma: Genomics and emerging biomarkers
for immunomodulatory cancer treatments. Semin Cancer Biol.
52:228–240. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kitamura N, Sento S, Yoshizawa Y, Sasabe
E, Kudo Y and Yamamoto T: Current trends and future prospects of
molecular targeted therapy in head and neck squamous cell
carcinoma. Int J Mol Sci. 22:2402020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Panarese I, Aquino G, Ronchi A, Longo F,
Montella M, Cozzolino I, Roccuzzo G, Colella G, Caraglia M and
Franco R: Oral and oropharyngeal squamous cell carcinoma:
Prognostic and predictive parameters in the etiopathogenetic route.
Expert Rev Anticancer Ther. 19:105–119. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hanoteau A, Newton JM, Krupar R, Huang C,
Liu HC, Gaspero A, Gartrell RD, Saenger YM, Hart TD, Santegoets SJ,
et al: Tumor microenvironment modulation enhances immunologic
benefit of chemoradiotherapy. J Immunother Cancer. 7:102019.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Göttgens EL, Ostheimer C, Span PN, Bussink
J and Hammond EM: HPV, hypoxia and radiation response in head and
neck cancer. Br J Radiol. 92:201800472019.PubMed/NCBI
|
|
7
|
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta
R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS,
et al: Ferroptosis: An iron-dependent form of nonapoptotic cell
death. Cell. 149:1060–1072. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang C, Liu X, Jin S, Chen Y and Guo R:
Ferroptosis in cancer therapy: A novel approach to reversing drug
resistance. Mol Cancer. 21:472022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhao L, Zhou X, Xie F and Zhang L, Yan H,
Huang J, Zhang C, Zhou F, Chen J and Zhang L: Ferroptosis in cancer
and cancer immunotherapy. Cancer Commun (Lond). 42:88–116. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xu L, Li YY, Zhang YC, Wu YX, Guo DD, Long
D and Liu ZH: A novel ferroptosis-related gene signature to predict
prognosis in patients with head and neck squamous cell carcinoma.
Dis Markers. 2021:57599272021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lu T, Zhang Z, Pan X, Zhang J, Wang X,
Wang M, Li H, Yan M and Chen W: Caveolin-1 promotes cancer
progression via inhibiting ferroptosis in head and neck squamous
cell carcinoma. J Oral Pathol Med. 51:52–62. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yau TY, Sander W, Eidson C and Courey AJ:
SUMO interacting motifs: Structure and function. Cells.
10:28252021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhao X: SUMO-mediated regulation of
nuclear functions and signaling processes. Mol Cell. 71:409–418.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li H, Chen L, Li Y and Hou W:
SUMO-specific protease 1 inhibitors-A literature and patent
overview. Expert Opin Ther Pat. 32:1207–1216. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gao Y, Wang R, Liu J, Zhao K, Qian X, He X
and Hong L: SENP1 promotes triple-negative breast cancer invasion
and metastasis via enhancing CSN5 transcription mediated by GATA1
deSUMOylation. Int J Biol Sci. 18:2186–2201. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Dong B, Gao Y, Kang X, Gao H, Zhang J, Guo
H, You MJ, Xue W, Cheng J and Huang Y: SENP1 promotes proliferation
of clear cell renal cell carcinoma through activation of
glycolysis. Oncotarget. 7:80435–80449. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sun W, Liu X, Yang X, Jing X, Duan C, Yang
G, Wu C, Huang H, Luo Q, Xia S, et al: SENP1 regulates the
transformation of lung resident mesenchymal stem cells and is
associated with idiopathic pulmonary fibrosis progression. Cell
Commun Signal. 20:1042022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bai YT, Xiao FJ, Wang H, Ge RL and Wang
LS: Hypoxia protects H9c2 cells against ferroptosis through
SENP1-mediated protein DeSUMOylation. Int J Med Sci. 18:1618–1627.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kuwata H and Hara S: Role of acyl-CoA
synthetase ACSL4 in arachidonic acid metabolism. Prostaglandins
Other Lipid Mediat. 144:1063632019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Doll S, Proneth B, Tyurina YY, Panzilius
E, Kobayashi S, Ingold I, Irmler M, Beckers J, Aichler M, Walch A,
et al: ACSL4 dictates ferroptosis sensitivity by shaping cellular
lipid composition. Nat Chem Biol. 13:91–98. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen X, Li J, Kang R, Klionsky DJ and Tang
D: Ferroptosis: Machinery and regulation. Autophagy. 17:2054–2081.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Quan J, Bode AM and Luo X: ACSL family:
The regulatory mechanisms and therapeutic implications in cancer.
Eur J Pharmacol. 909:1743972021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Blum A, Wang P and Zenklusen JC: SnapShot:
TCGA-analyzed tumors. Cell. 173:5302018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Onofri A, Terzaroli N and Russi L: Linear
models for diallel crosses: A review with R functions. Theor Appl
Genet. 134:585–601. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
He F, Chen Z, Deng W, Zhan T, Huang X,
Zheng Y and Yang H: Development and validation of a novel
ferroptosis-related gene signature for predicting prognosis and
immune microenvironment in head and neck squamous cell carcinoma.
Int Immunopharmacol. 98:1077892021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang W, Zhang J, Wang Y, Xu Y and Zhang S:
Identifies microtubule-binding protein CSPP1 as a novel
cancer biomarker associated with ferroptosis and tumor
microenvironment. Comput Struct Biotechnol J. 20:3322–3335. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhu T, Shi L, Yu C, Dong Y, Qiu F, Shen L,
Qian Q, Zhou G and Zhu X: Ferroptosis promotes photodynamic
therapy: Supramolecular photosensitizer-inducer nanodrug for
enhanced cancer treatment. Theranostics. 9:3293–3307. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Han ZJ, Feng YH, Gu BH, Li YM and Chen H:
The post-translational modification, SUMOylation, and cancer
(Review). Int J Oncol. 52:1081–1094. 2018.PubMed/NCBI
|
|
30
|
Wang X, Liang X, Liang H and Wang B:
SENP1/HIF-1α feedback loop modulates hypoxia-induced cell
proliferation, invasion, and EMT in human osteosarcoma cells. J
Cell Biochem. 119:1819–1826. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hao L, Dong Y, Zhang JJ, He HG, Chen JG,
Zhang SQ, Zhang QJ, Wu W, Han CH and Shi ZD: Melatonin decreases
androgen-sensitive prostate cancer growth by suppressing SENP1
expression. Transl Androl Urol. 11:91–103. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shen X, Ni R, Qian X, Yu C, Wu H, Ni J,
Zhu W and Gao X: A verification study on the genes associated with
laryngeal squamous cell carcinoma by cDNA microarray. Lin Chuang Er
Bi Yan Hou Tou Jing Wai Ke Za Zhi. 24:411–413. 2010.(In Chinese).
PubMed/NCBI
|
|
33
|
Gao C, Xiao F, Zhang L, Sun Y and Wang L,
Liu X, Sun H, Xie Z, Liang Y, Xu Q and Wang L: SENP1
inhibition suppresses the growth of lung cancer cells through
activation of A20-mediated ferroptosis. Ann Transl Med.
10:2242022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vertegaal ACO: Signalling mechanisms and
cellular functions of SUMO. Nat Rev Mol Cell Biol. 23:715–731.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mevissen TET and Komander D: Mechanisms of
deubiquitinase specificity and regulation. Annu Rev Biochem.
86:159–192. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kukkula A, Ojala VK, Mendez LM, Sistonen
L, Elenius K and Sundvall M: Therapeutic potential of targeting the
SUMO pathway in cancer. Cancers (Basel). 13:44022021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bawa-Khalfe T, Yang FM, Ritho J, Lin HK,
Cheng J and Yeh ET: SENP1 regulates PTEN stability to dictate
prostate cancer development. Oncotarget. 8:17651–17664. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sun XX, Chen Y, Su Y, Wang X, Chauhan KM,
Liang J, Daniel CJ, Sears RC and Dai MS: SUMO protease SENP1
deSUMOylates and stabilizes c-Myc. Proc Natl Acad Sci USA.
115:10983–10988. 2018. View Article : Google Scholar : PubMed/NCBI
|