Introduction
Breast cancer is a condition wherein the cells in
the mammary gland grow in a dysregulated manner, resulting in the
formation of a mass of cells called a tumor. The World Health
Organization statistics revealed that, in 2020, the worldwide scale
of breast cancer cases increased to 2.3 million, and, globally,
6,85,000 individuals lost their lives (1). Although breast cancer is generally
considered benign, a delayed diagnosis can cause the cancer to
spread to the surrounding lymph nodes, resulting in distant
metastasis and impacting vital organs such as the brain, liver and
lungs (2). Therefore, researchers
are striving hard to develop effective therapeutic approaches; in
this regard, the role of forkhead box proteins cannot be overlooked
due to their involvement in cancer development and progression.
Forkhead box proteins, commonly known as forkhead
box (FOX) proteins, belong to a family of transcription factors
that are evolutionarily conserved, distinguished by their DNA
binding domain, which is referred to as either ‘forkhead’ or
‘winged helix’ (3). These
transcription factors play pivotal roles as essential regulators of
gene expression in various pathological and physiological
processes, encompassing cell proliferation, differentiation and
survival (4). Interestingly,
numerous studies have attempted to establish the links between FOX
proteins and cellular events, including the cancer initiation and
progression, alongside the emergence of resistance against
anticancer drugs (5). These
discoveries highlight the diverse functions of FOX proteins in the
field of cancer biology.
The Forkhead box, or Fox, gene family is a group of
transcriptional regulators known for their ancient evolutionary
origins. They were named after the Drosophila melanogaster
fork head gene (fkh). It has been reported that mutations in fkh
can lead to abnormalities in head fold involution during embryonic
development, causing adult flies to exhibit a characteristic spiked
head phenotype (6). Fox genes are
found across a vast range of organisms, from yeast to humans. They
are further classified into subfamilies such as FOXA and FOXP. A
defining feature of Fox proteins is their DNA-binding domain, the
forkhead (FKH) region, which is remarkably similar across species
and consists of roughly 100 amino acids. The high degree of
conservation within the FKH domain extends across the entire Fox
family. This feature served as the basis for the initial
classification system, where Fox proteins were assigned to 15
classes, designated FOXA to FOXO, based on sequence similarity
within the FKH domain. More recent evolutionary analyses have
refined the Fox protein classification system, increasing the
number of classes to 19, ranging from FOXP to FOXS. This
categorization is based on sequence similarities within the FKH
domain, which typically consists of three alpha helices, three beta
sheets, and two unique ‘wing’ regions flanking the third beta
sheet. The ‘forkhead’ or ‘winged helix’ terminology originates from
the distinctive butterfly-like structure formed by these ‘wing’
regions in Fox proteins (7).
FOX transcription factors are crucial regulators in
embryonic development and maintaining cellular balance, driven by
evolutionary forces that shape their diverse functions (8). They act as master regulators of
fundamental cellular functions, including cell cycle progression,
proliferation, differentiation, DNA repair mechanisms, metabolism,
blood vessel formation (angiogenesis), and ultimately, a cell's
fate. Disruptions in Fox protein activity have been implicated in
the onset, spread, advancement and resistance to therapy of cancer.
They also influence pathways related to cancer, aiding cell
survival in adverse conditions. The involvement of FOX proteins in
cancer spans its entire spectrum, from initiation to metastasis,
orchestrated through intricate networks. While the importance of
Fox proteins is well-established, a comprehensive understanding of
how mutations within FOX-binding sites in the regulatory regions of
FOX-target genes remains incomplete. Further research is needed to
unravel these mechanisms in detail.
FOX proteins can also be regulated by microRNAs
(miRNAs), which are special non-coding RNAs whose role as a tumor
suppressor and oncogene is gradually coming into stronger light
(9,10). The present review has tried to
provide a brief overview about miRNAs and FOX proteins as other
multiple roles and association of FOX proteins have to be covered
in more detail. However, a thorough analysis of the crosslinks
between miRNAs and FOX proteins would help unlock the therapeutic
hurdles around breast cancer treatment.
Currently, the understanding of the FOX family is at
its nascent stage; therefore, a comprehensive study that unveils
the complexity revolving around the FOX transcription factor and
the detailed understanding of its association with breast cancer
would aid in the discovery of cancer biomarkers and improve the
existing treatment strategies. Keeping this in view, the article
has tried to document the majority of important associations
between multiple FOX proteins and breast cancer.
Role of various FOX proteins in breast
cancer
The discovery of significant findings often sparks
interest, and a similar scenario unfolded with FOX proteins, now
recognized for their role in positive and negative regulation of
breast carcinogenesis. Initially, the focus of the present review
centered on the FOXA protein family, which is extensively engaged
in hormonal signaling (10).
FOXA family and breast cancer
Considering the diverse functions and phenotypes of
normal breast epithelial cells, gene expression profiling has
facilitated the categorization of breast cancer into the following
five types: Luminal type A, luminal type B, normal-like,
ERα-negative, HER-2-positive and basal type (8). The luminal types A and B are
characterized by ERα-positive due to the elevated expression of
ERα, demonstrating an improved prognosis. FOXA1, a member of the
forkhead family, is garnering attention for its significant
involvement in the hormonal signaling network, which plays a
crucial role in regulating the growth and differentiation of
epithelial cells in breast cancer (10).
Hepatocyte nuclear factor 3α (HNF3α), also known as
FOXA1, is identified as the first transcription factor of the FOXA
family. It is enriched with hepatocytes and significantly regulates
α1-antitrypsin gene and transthyretin expressions (11). Apart from FOXA1, the FOXA family
comprises other members, namely FOXA2 (HNF3β) and FOXA3 (HNF3γ)
(12). These transcriptional
factors bind to ‘TGTTTAC’ or ‘TGTTTGC’ motifs present in the
promoter/enhancer regions of the target genes (13). Each factor binds to the target gene
with varying affinities, thereby modulating transcription. The FOXA
factors are referred to as the ‘pioneer factors’ as they exhibit a
unique capability to engage with target sites within silent
chromatin. This distinctive ability enables them to initiate
regulatory events, setting them apart from other transcription
factors.
FOXA1 comprises a helix-turn-helix motif of 110
amino acids as well as three α-helices, two β-strands, and two
polypeptide loops on either side, which are referred to as Wing1
and Wing2 subdomains (13,14). The DNA binding domain is equipped
with a polypeptide chain on either side, giving it a wing-like
appearance that is responsible for identifying the stability
associated with DNA binding (12).
The binding domain, along with the polypeptide chain, binds to the
consensus sequence ‘A(A/T)TRTT(G/T)RYTY’ wherein the binding domain
of the DNA interacts with the major grooves and the polypeptide
chains interact with the minor grooves of DNA.
FOXA1 resembles linker histones in DNA binding and
stabilizes the major groove by projecting into it, utilizing
polypeptide loops similar to histones 1 and 5. Unlike the linker
proteins, FOXA1 does not compact DNA around peptide sequences but
enhances the nucleosome accessibility to facilitate interactions
with transcriptional regulators (15,16).
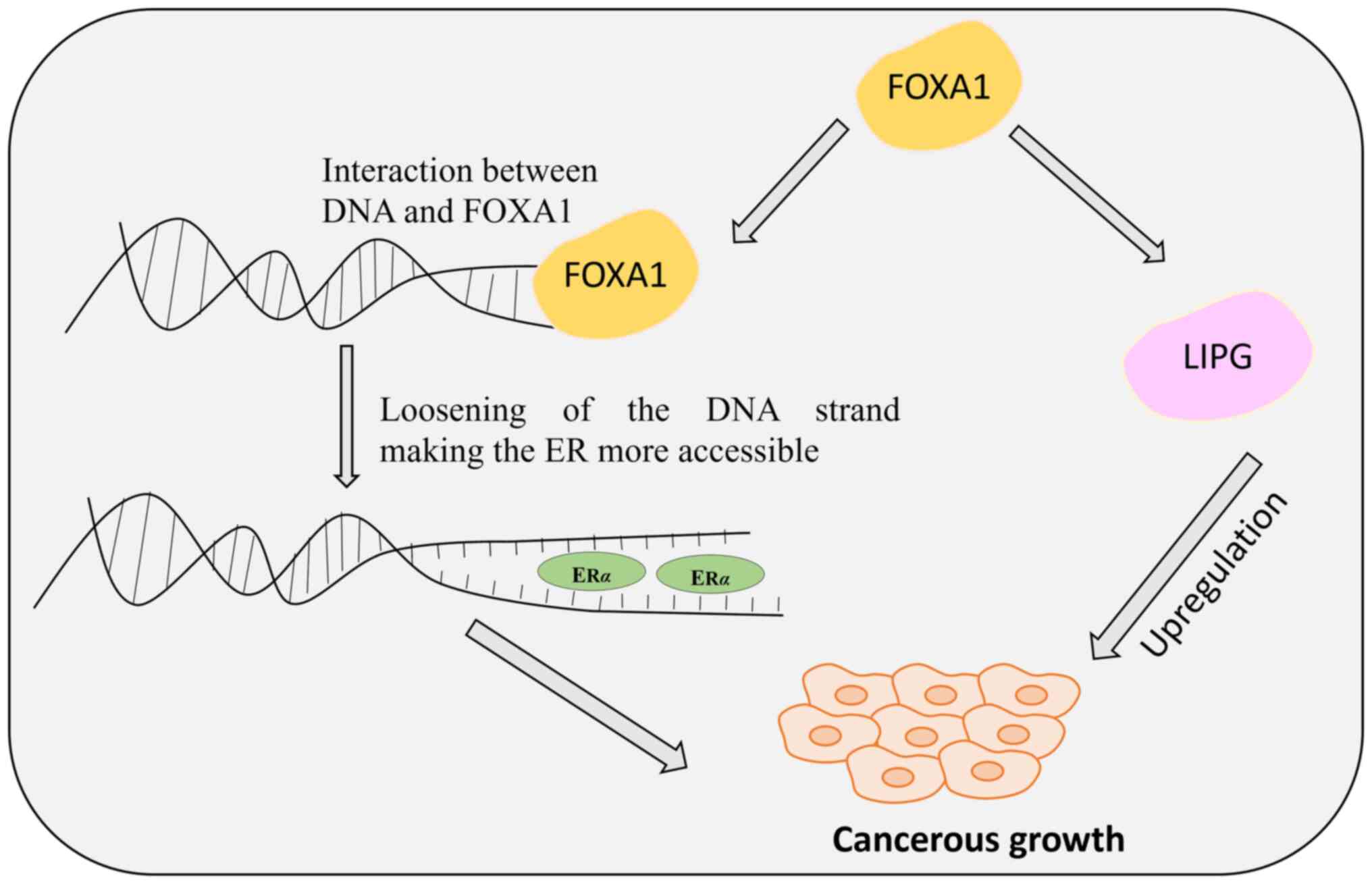
FOXA1 is vital for mammary morphogenesis and estrogen receptor (ER)
activity and is pivotal in normal breast tissue development
(17). In breast tissues, FOXA1
localizes within cell nuclei where it co-localizes with ER. FOXA1
interacts with specific DNA sequences termed as cis-regulatory
elements, located within heterochromatin, thereby aiding in the
binding of hormone receptors to chromatin. This ‘pioneering’
function opens up the chromatin structure, allowing other
transcription factors, such as nuclear receptors, to bind and
regulate gene expression (18).
FOXA1 is crucial for various aspects of normal breast development,
such as mammary gland morphogenesis (13), and is essential for optimal ER
activity (19) (Fig. 1). Moreover, FOXA1 directly interacts
with GATA3 (20), emphasizing its
regulatory role in mammary gland development. Ghosh et al
(21) proposed that
hypermethylation in healthy breast tissue might reduce FOXA1
expression, potentially leading to impaired ER function and
influencing breast tumor development. However, the precise role of
FOXA1 in tumor initiation and progression remains a topic of
ongoing investigation. It is widely accepted that FOXA1 depletion
inhibits cancer cell proliferation (22), as both ER and androgen receptor
transcriptional activities rely on FOXA1 in breast and prostate
cancer cells. Additionally, during carcinogenesis, FOXA1 directly
enhances the transcription of several downstream genes that support
the luminal phenotype, such as the E-cadherin gene (CDH1) (23) and GATA3 (24), while simultaneously suppressing
basal differentiation (25).
The lipid metabolism pathway is the primary pathway
used by cancer cells to meet their energy requirements to
proliferate (26). Apart from the
ER levels, FOXA1 is known to directly regulate the endothelial
lipase (LIPG) enzyme expression levels. LIPG, belonging to the
lipoprotein lipase family, plays a substantial role in facilitating
tumor growth by promoting breast cancer cell-lipid addiction, and
its reduced expression causes tumor growth impairment. In breast
cancer cells, the increased expression of FOXA1 elevates the levels
of LIPG, which in turn stimulates tumor growth by enhancing cell
proliferation (Fig. 1). LIPG works
by generating lipid precursors extracellularly, which are
transported to manage the intracellular lipid species (22). The downregulation of LIPG leads to
impaired breast cancer cell growth, inhibition of proliferation,
and tumor development.
FOXA1 serves dual role as a therapeutic target and a
prognostic marker; however, further investigation is necessary to
understand the relationship between FOXA1 levels in normal luminal
breast tissue and the complex interactions between FOXA1, ER alpha
(ERα), and LIPG expression. This knowledge gap hinders the ability
to fully elucidate the mechanisms underlying breast cancer
initiation and progression.
FOXO family and breast cancer
FOXOs, constitute another group within the family of
forkhead proteins classified by a forkhead domain and a
winged-helix DNA binding motif (27), have been identified as tumor
suppressors, a key regulator in animal physiology, and control
various cellular processes, such as cellular growth,
differentiation, proliferation, apoptosis, DNA repair and stem cell
maintenance.
FOXOs negatively control cancer progression by
modulating angiogenesis and metastasis. At present, four members of
FOXO family have been identified: FOXO1, FOXO3A, FOXO4 and FOXO6;
each characterized by unique core functional domains (28). With the exception of FOXO6, the
other three members have similar structures, whereas FOXO6 differs
in structure as compared with the rest. However, despite the
structural differences, all four members have the same DNA binding
specificity and recognize the regulatory elements that contain the
sequence ‘TTGTTTAC’. Additionally, all members are known to work
with coactivators and corepressors to modulate their
functioning.
Previous studies have demonstrated that, in cancer,
FOXOs exist as chimeric products due to chromosomal translocation,
and the presence of these hybrids has directed the attention of
researchers toward the FOXOs' role in various cancers. FOXO1 is a
fusion protein, with the partner being either PAX3 or PAX7 DBD in
pediatric alveolar rhabdomyosarcoma (29). FOXO3A and FOXO4 were identified as
fusion proteins with the mixed-lineage leukemia gene in acute
myeloid leukemias (30). The
existence of FOXOs as fusion proteins indicates their role in human
cancers.
Reduced FOXO protein expression significantly
influences breast cancer cell proliferation (31), subject to intricate regulation
through processes such as phosphorylation, acetylation and
ubiquitination (32). The
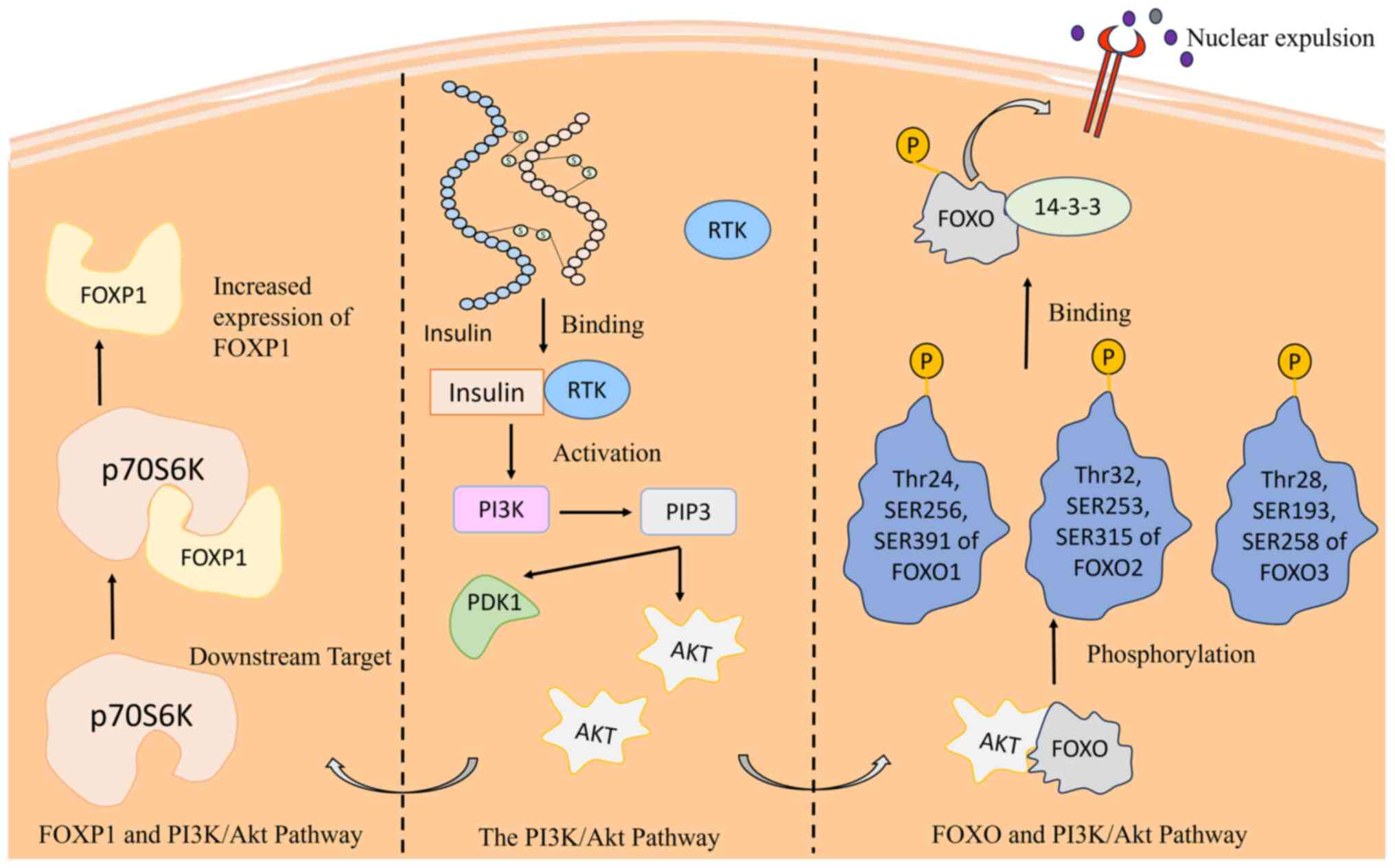
connection between FOXO and the PI3K/Akt pathway, a critical cell
cycle regulator, highlights its function in regulating cellular
proliferation and growth (33). The
activation of insulin triggers a cascade that involves the
production of phosphatidylinositol-3,4,5-triphosphate (PIP3) via
PI3K, ultimately leading to phosphorylation of FOXO by Akt
(34). This inhibits their
DNA-binding domains, thereby reducing the transcriptional activity
(31,35). Phosphorylated FOXOs are associated
with 14-3-3 proteins, causing nuclear exclusion. Insulin absence
restricts FOXO access to the nucleus, impairing its
tumor-suppressive activity. The intricate interplay underscores the
significance of FOXO proteins in breast cancer regulation,
proposing potential therapeutic targets within the PI3K/Akt pathway
(36). Various kinases, such as
PAK1 and the TAK1-Nemo-like kinase pathway, further modulate FOXO,
either inhibiting or promoting its activity (37,38).
Understanding these molecular mechanisms provides insights for
developing strategies to harness FOXO proteins for controlling
breast cancer progression (Fig. 2)
(39).
Moreover, FOXO activity is negatively regulated by
silent information regulators (SIRTs), which are members of the
class III histone deacetylases. SIRTs are acknowledged for their
crucial role in cell survival. SIRTs function by downregulating the
FOXO levels, thereby controlling the process of apoptosis (40). Oxidative stress results in the
nuclear localization of FOXOs. A complex formation occurs in the
nucleus by SIRT1, which then interacts with FOXO and deacetylates
it, thereby downregulating the tumor-suppressive role FOXO.
The expression of FOXO3a is modulated by the
methylation status of its promoter region (41). Liu et al (42) demonstrated that hypermethylation of
the FOXO3a promoter by DNMT1 is associated with reduced FOXO3a
expression in breast cancer (42).
It was further illustrated that FOXO3a suppresses breast cancer
stem cell (BCSC) properties and tumorigenicity by inhibiting the
FOXM1/SOX2 signaling pathway. Additionally, their findings suggest
that SOX2 negatively regulates FOXO3a expression through direct
activation of DNMT1, creating a feedback loop. Moreover, blocking
DNMT activity suppresses tumor growth by altering the signaling
between FOXO3a, FOXM1, and SOX2 proteins in breast cancer. This
finding underscores the critical role of the
DNMT1/FOXO3a/FOXM1/SOX2 signaling pathway in regulating BCSC
properties. It also suggests a promising therapeutic strategy for
targeting BCSCs and overcoming drug resistance.
FOXO has emerged as a promising therapeutic target
for breast cancer, with recent years witnessing substantial
advancements in understanding its role in the disease. Clearer
understanding of the processes related to FOXOs would help
researchers manipulate and develop more targeted therapies.
FOXM family and breast cancer
FOX proteins exhibit similarity in their DNA-binding
domain, characterized by three sheets, three helices, two wings or
loops that form a helix-turn-helix motif structure. The second and
third helices display structural variations, while the helices and
sheets contain conserved regions. The third helix primarily handles
DNA binding, with the second wing attaching to the major and minor
grooves of DNA (43). Another
crucial member of the FOX family, FOXM1, is situated on the
12p13.33 chromosomal band and comprises 10 exons (44). FOXM1 consists of a forkhead domain,
an activation domain associated with the C terminus, and a
repressor domain linked to the N terminus. The differential
splicing of FOXM1 exons gives rise to various variants, including
FOXM1A, FOXM1B, FOXM1C and FOXM1D (45). FOXM1 binds to core consensus
sequences ‘(A/C) AAACAAC’, and in certain instances, it
necessitates interaction with DNA binding factors for
transcriptional activity (46). The
FOXM1 protein plays a role in diverse processes, encompassing cell
proliferation, DNA damage repair and apoptosis. FOXM1 is
upregulated in various cancers, highlighting its role as the
principal regulator of cell cycle progression, governing various
transition points within the cell cycle (47). Additionally, FOXM1 plays a role in
regulating gene transcription associated with the cell cycle and
regulates various mitotic regulators, including CDC25B and
CENPF.
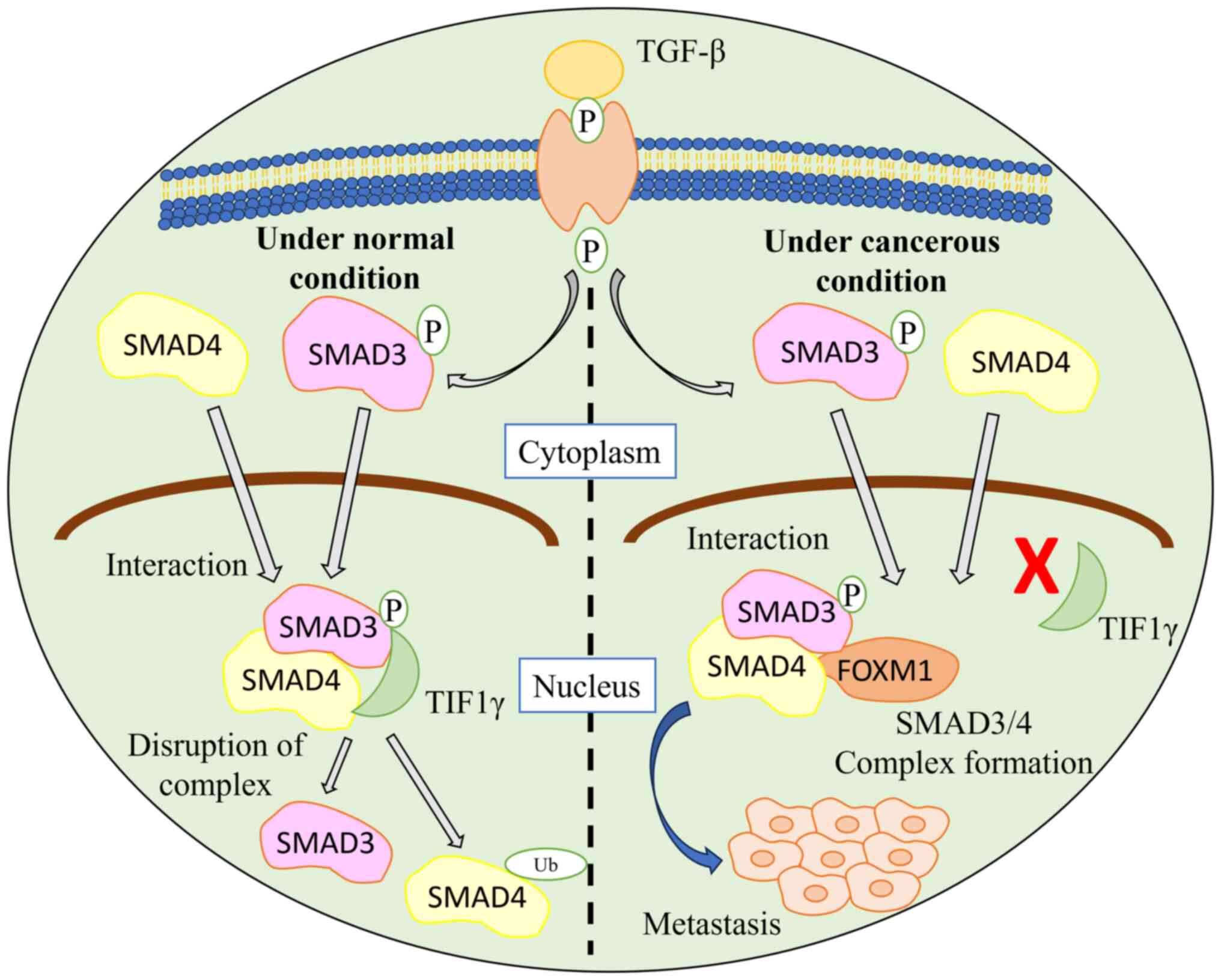
FOXM1 interacts with SMAD3, which activates the
TGF-β pathway, resulting in breast cancer cell metastasis. Under
normal conditions, the ubiquitin-protein ligase transcriptional
intermediary factor 1γ (TIF1γ) interacts with SMAD3 and SMAD4 and
causes ubiquitination. This interaction leads to a disruption of
the SMAD3/SMAD4 complex. When FOXM1 interacts with SMAD3, the TGF-β
pathway is activated as the interaction of SMAD3 and SMAD4 with
TIF1 is inhibited, thereby stabilizing the SMAD3/SMAD4 complex
(48). The FOXM1/SMAD3 interaction
results in breast cancer cell invasion induced by the TGF-β pathway
(Fig. 3).
In humans, two types of ER receptors, ERα and Erβ,
are responsible for mediating the effect of ER. Although ER binds
to both receptors with equal affinity, both exhibit different
activities; namely, ERα promote cell proliferation, whereas Erβ has
apoptotic and antiproliferative properties (49). FOXM1 has the ability to regulate the
levels of ER at both mRNA and protein levels, thereby positively
regulating the ER levels by interacting with forkhead-responsive
elements situated on the ERα promoter region, thereby influencing
its overall regulation.
Human epidermal growth receptor 2 (HER2), a member
of the epidermal growth factor receptor family, functions as a
tyrosine kinase receptor (50).
HER2 is directly correlated with FOXM1 activity. HER2
overexpression in breast cancer results in increased promoter
activity of FOXM1, whereas its downregulation upon administration
of the HER2 inhibitor (lapatinib) results in a reduced activity of
the promoter region of FOXM1 (51).
FOXM1 is an ideal therapeutic target because of its
significance in metastasis and disease progression. It is also
responsible for relaying resistance to anticancer drugs. However,
to date, FOXM1 therapy has not been implemented because of the
absence of a comprehensive analysis of the FOXM family. A thorough
understanding of FOXM1 mechanisms would help researchers develop
biomarkers and therapeutic strategies for early detection and
improved treatment strategies.
FOXC family and breast cancer
FOXC proteins are another category of forkhead
proteins with distinct DNA binding forkhead domains. Its two
members, FOXC1 and FOXC2, play roles in cardiac, embryonic and
ocular development (52). In
cancer, both members play a significant role in metastasis, cell
growth, invasion and angiogenesis (4). FOXC1 is a cell growth regulator in
breast cancer and its downregulation results in reduced cell
growth, whereas FOXC2 is also involved in carcinogenesis because
its increased expression is associated with breast cancer
development. FOXC1 is characterized as a single-exon gene situated
on the chromosomal band 6p25, encoding a 533 amino acid protein.
This encoded protein, localized in the nucleus, engages with DNA
and regulates targeted gene expression. Its DNA binding domain
contains a ‘winged-helix’ structure. The winged helix's third alpha
helix interacts with the major domain at 5′-GTAAATAAA-3′ (53).
Basal-like breast cancer (BLBC) falls under the
category of triple-negative breast cancer (TNBC), wherein all three
receptors, namely, HER2, progesterone receptor and ER, are not
expressed (54). The overexpression
of FOXC1 is observed in BLBC, leading to metastasis and invasion
(55). A previous study indicated
the function of FOXC1 in activating NF-κB signaling. In BLBC, FOXC1
upregulates peptidyl-prolyl cis-trans isomerase NIMA-interacting 1
expression, increasing p65/RelA protein stability (56). This activates the NF-κB signaling
pathway which mediates FOXC1 to promote proliferation, invasion and
metastasis (55).
In addition to activating the NF-κB signaling
pathway, FOXC1 is acknowledged for its role in inducing
epithelial-to-mesenchymal transition (EMT) in cancer cells. EMT is
a biological process characterized by the loss of cell polarity and
intercellular adhesion in embryonic cells, facilitating their
migration to various sites for tissue and organ development
(57). Overexpression of FOXC1 in
MCF12A mammary epithelial cells triggered EMT, as shown by reduced
E-cadherin levels. This EMT induction promoted tumor proliferation
and migration in BLBC (58,59). Furthermore, EMT is regulated by
various transcription factors, including Goosecoid, Snail and
Twist, which promote FOXC2 expression in cells undergoing EMT
(60). FOXC2 overexpression induces
mesenchymal differentiation along with MMP2 and MMP9 expressions.
Additionally, FOXC2 is responsible for E-cadherin downregulation,
whereas p120-catenin is directly downregulated by FOXC2. p120 is a
regulatory protein responsible for stabilizing E-cadherin (61).
The FOXC family is another important category known
to regulate tumor progression and metastasis. Relevant research is
being conducted, and there is markedly more to discover. Further
advances in the interlinkage between FOXC and breast cancer would
open the door to advanced therapeutics and treatment
strategies.
FOXP family and breast cancer
Similar to other FOX families, the FOXP family is
defined by a winged helix domain comprising α-helices, β-sheets and
loops or wings, forming a winged-helix forkhead DNA-binding domain
(FHD) and a helix-turn-helix motif (62). The FOXP family has four members:
FOXP1, FOXP2, FOXP3 and FOXP4, with FOXP1 and FOXP2 having a
C-terminal binding domain and FOXP3 and FOXP4 having an N-terminal
binding domain (63). Furthermore,
all four members exhibit a dimerized C-terminal winged helix FHD,
while the FOXP3 dimer is recognized for its stability compared with
the others (64).
Depending on the tumor type, FOXP1 acts as an
oncogene or tumor suppressor. Similar to FOXA1, the FOXP1 levels
are elevated, suggesting its association with ER (65). Elevated FOXP1 levels in MCF7
resulted in increased proliferation along with overexpression of
the ER-mediated gene, whereas cells in which the level of FOXP1 was
depleted presented reduced growth, with both situations presenting
a pro-tumorigenic role in breast cancer (65).
FOXP1, a crucial transcription factor, is
intricately linked to the PI3K/Akt signaling pathway which is a key
regulator of cellular functions including proliferation and
metastasis (66). Akt, a downstream
Ser/Thr kinase within this pathway, activates key targets,
including p70S6K, which subsequently coregulates diverse cellular
functions (67). Notably, FOXP1
levels are modulated by the PI3K/Akt pathway, as proposed by Banham
et al (67), who revealed
that the absence of Akt significantly reduces FOXP1 levels, whereas
the addition of Akt elevates them. Akt does not phosphorylate FOXP1
because of the absence of phosphorylation sites, emphasizing the
regulatory role of p70S6K (67)
(Fig. 2). A comprehensive
understanding of FOXP1 regulatory pathways holds promise for
developing innovative therapeutics and treatment strategies.
Liu et al (68) identified the interaction between
FOXP2 and FOXA2 and they confirmed that this interaction acts as an
EMT suppressor during metastasis in breast cancer cells,
highlighting its role as EMT regulator. To understand the in
vivo functioning of FOXP2 on the EMT of breast cancer cells,
they generated a metastasis model in nude mice and injected the
small hairpin (sh)FOXP2 and sh-control lentivirus-infected MCF-7
cells in the tail vein. The results revealed that FOXP2 knockdown
promoted metastasis, likely due to the downregulation of E-cadherin
and PHF2, both established EMT suppressors indicating that in
vivo FOXP2 inhibition could increase the metastasis of breast
cancer cells (68).
Conversely, FOXP3 functions as a tumor suppressor by
regulating the expression of numerous tumor genes (69). Originally recognized as a molecular
marker for regulatory T cells, the involvement of FOXP3 in breast
cancer is currently closely associated with metastasis-associated 1
(MTA1), a pivotal protein in downstream gene transcription
implicated in breast cancer (70).
Functioning as a negative regulator of MTA1 FOXP3 binds to its
promoter region, thereby inhibiting its expression and reducing the
metastatic capability of the cell (70,71). A
thorough investigation into the interplay between FOXP3 and MTA1
could help develop techniques to address metastatic breast cancer
and open new avenues for therapeutic advancements in oncology.
The effect of the dysregulated activity of the
various FOX proteins and the associated targeted gene are
summarized in Table I. While the
in vitro and ex vivo studies have helped researchers
to comprehend the dysregulated expression of FOX proteins in breast
cancer, the current understanding remains incomplete. Therefore,
further studies are required to understand the expression levels of
FOX proteins utilizing in vivo studies as this approach will
be crucial in developing effective therapeutic strategies.
 | Table I.The effect of the dysregulated
activity of the various FOX proteins and the associated targeted
gene. |
Table I.
The effect of the dysregulated
activity of the various FOX proteins and the associated targeted
gene.
| Fox protein | Activity | Cell line | Cellosaurus ID | Sample | Estimation
technique | (Refs.) |
|---|
| FOXA1 | Upregulated | MDA-MB-231 | CVCL_0062 | - | Western
blotting | (26) |
| FOXA1 | Upregulated | MCF-10A | CVCL_0598 | - | Western
blotting | (26) |
| FOXA1 | Upregulated | MCF-7 | CVCL_0031 | - | Western
blotting | (26) |
| FOXA1 | Upregulated | MCF7 | CVCL_0031 | - | RT-qPCR and western
blotting | (22) |
| FOXA1 | Upregulated | MDA-MB-231 | CVCL_0062 | - | RT-qPCR and western
blotting | (22) |
| FoxA1 | Downregulated | - | - | Tissue | IHC staining | (72) |
| FoxA2 | Downregulated | - | - | Tissue | IHC staining | (73) |
| FOXO3A | Downregulated | MCF-7 | CVCL_0031 | - | RT-qPCR and western
blotting | (39,40) |
| FOXO3A | Downregulated | MDA-MB-231 | CVCL_0062 | - | RT-qPCR and western
blotting | (39,40) |
| FOXO3A | Upregulated | - | - | Tissue | IHC staining | (74) |
| FOXO1 | Downregulated | - | - | Tissue | IHC staining | (39) |
| FOXM1 | Upregulated | MDA-MB-231 | CVCL_0062 | - | Western
blotting | (40,51) |
| FOXM1 | Upregulated | MCF-7 | CVCL_0031 | - | Western
blotting | (40,51) |
| FOXM1 | Upregulated | MDA-MB-453 | CVCL_0418 | - | Western
blotting | (40,51) |
| FOXM1 | Upregulated | MDA-MB-468 | CVCL_0419 | - | Western
blotting | (40,51) |
| FOXM1 | Downregulated | MCF-10A | CVCL_0598 | - | Western
blotting | (51) |
| FOXM1 | Downregulated | MDA-MB-231 | CVCL_0062 | - | Western
blotting | (51) |
| FOXM1 | Upregulated | - | - | Tissue | IHC staining | (75) |
| FOXC1 | Upregulated | MDA-MB-231 | CVCL_0062 | - | Western
blotting | (55) |
| FOXC2 | Upregulated | 4T1 | CVCL_0125 | - | Western
blotting | (61) |
| FOXP3 | Downregulated | MDA-MB-231 | CVCL_0062 | - | RT-qPCR and western
blotting | (69) |
Overview of miRNA
MiRNAs are small, non-coding RNAs that exert
regulatory control over various biological processes. They have a
length of 22 nucleotides and function by negatively regulating the
mRNA transcription. miRNAs regulate mRNAs by interacting with the
3′ untranslated region (UTR) of the targeted mRNA (76). This interaction suppresses mRNA
expression. Apart from the 3′ UTR, miRNA also interacts with other
regions such as 5′ UTR, gene promoters and coding sequences
(77).
The biogenesis of miRNA involves a two-step process,
wherein DNA sequences are transcribed into primary miRNA
(pri-miRNA), subsequently processed into precursor and mature
miRNAs. The initial step entails transcription of the DNA sequence
into pri-miRNA within the nucleus (78). A microprocessor complex, comprising
DiGeorge syndrome Critical Region 8 (DGCR8) functioning as an RNA
binding domain and Drosha, a ribonuclease III enzyme, is
responsible for converting pri-miRNA into pre-miRNA. DGCR8 protein
specifically binds to the N6-methylated GGAC sequence in pri-miRNA.
This complex then recruits Drosha nuclease, which cleaves the
pri-miRNA duplex, resulting in the generation of a pre-miRNA with a
characteristic hairpin structure (79,80).
Following formation, the pre-miRNA is transported to the cytoplasm
via the Exportin 5 (XPO5)/RanGTP complex (81). In the cytoplasm, Dicer, an RNase III
enzyme, matures the pre-miRNA by cleaving off the terminal loop,
resulting in the formation of mature miRNA (82). The mature miRNA is loaded into the
Argonaute protein, which subsequently regulates the targeted
mRNA.
miRNAs play regulatory roles in various processes,
including cell proliferation and apoptosis. The involvement of
miRNAs in breast cancer, either as tumor suppressor miRNAs (tsmiRs)
or oncogenic miRNAs (oncomiRs), has been underscored in numerous
studies. OncomiRs are miRNAs capable of downregulating the
expression of tumor suppressors and are therefore elevated in
breast cancer (83). tsmiRs are
miRNAs capable of downregulating the expression of oncogenes and
are thereby downregulated in breast cancer (84). miRNAs are known for their regulatory
roles in cellular mechanisms, including cell proliferation,
metastasis, invasion and apoptosis; therefore, the altered miRNAs
levels can induce cancer-like conditions. For instance, miR-16 and
miR-497 are known to inhibit cell proliferation, thereby acting as
tsmiRs (85). Contrarily,
miR-200c/141 works as an oncogene, thereby promoting the metastasis
of breast cancer cells (86).
Interlinkage of miRNA and FOX proteins in
breast cancer
FOX proteins are a group of transcription factors
that play a regulatory role in controlling gene expression. FOX
proteins function as oncogenes and tumor suppressors; therefore,
any dysregulation in FOX protein levels results in cancer-like
conditions.
On the other hand, miRNA is a small non-coding RNA
that works by regulating target mRNA expression. In breast cancer,
miRNAs are known to function as either oncomiRs or tsmiRs, thereby
promoting the pathological condition. miRNA overexpression or
downregulation results in increased cell proliferation and
metastasis, resulting in tumorigenesis.
Research is being conducted to understand the
interlinkage between miRNA and various classes of FOX proteins as
miRNAs regulate the expression of FOX proteins, which leads to
breast cancer initiation and progression. Therefore, detailed
insights into the crosslinks between miRNA and FOX proteins and the
role of miRNA in modulating the activity of FOX proteins would help
in advancing the effective treatment for breast cancer.
miR-132, a tumor suppressor, is downregulated in
breast cancer. It regulates cell growth, proliferation and
apoptosis, and is known to downregulate the expression of FOXA1
(87). The overexpression of
miR-132 downregulates FOXA1, thereby affecting the expression of
LIPG (subsequent target of FOXA1). miR-132 downregulation results
in increased cell proliferation along with elevated levels of FOXA1
and LIPG, thereby resulting in tumorigenesis (88). In a study conducted by Wang et
al (88), it was observed that
introducing miR-132 significantly hindered colony formation in
MDA-MB-468 cells compared with the control group whereas
suppressing miR-132 enhanced colony formation efficiency in these
cells. Reducing FOXA1 expression led to a notable decrease in
colony formation efficiency in breast cancer cells compared with
the anti-miR-132 group. The aforementioned study revealed an
inverse correlation between miR-132 and FOXA1 expression in breast
cancer tissues. Furthermore, miR-132 was shown to suppress FOXA1
expression in breast cancer cells. These findings suggested that
miR-132 downregulation might contribute to FOXA1 upregulation in
breast cancer. Therefore, targeting miR-132 and FOXA1 together
could hold promise as therapeutic strategy for breast cancer
treatment. miR-802 is a tumor suppressor miRNA, responsible for
regulating cell proliferation (89). FOXM1, an oncogene, is negatively
regulated by miR-802 because it contains miR-802 binding sites.
miR-802 interacts with the 3′ UTR of FOXM1, downregulating its
expression. The upregulation of miR-802 results in the reduced
expression of FOXM1, thereby controlling cell proliferation,
whereas the downregulated expression of miR-802 elevates FOXM1
expression, contributing to cancer-like conditions. Yuan and Wang
(90) delved into the expression
pattern of miR-802 in cancerous vs. adjacent non-cancerous tissues,
revealing a significant decrease in miR-802 levels in cancer
tissues. It was demonstrated that increasing miR-802 levels
significantly reduced the ability of MCF-7 cells to proliferate
after transfection, compared with control cells. To confirm that
miR-802 directly targets FoxM1, they created a luciferase reporter
vector containing potential miR-802 binding sites within the 3′ UTR
of the FoxM1 gene. Their outcomes showed that increasing miR-802
levels significantly reduced luciferase activity in MCF-7 cells
when the reporter contained the FoxM1 3′ UTR. These findings
provide initial evidence that boosting miR-802 expression hinders
breast cancer cell proliferation both in cell culture (in
vitro) and in animal models (in vivo), likely by
regulating FoxM1 expression. This highlights the significance of
miR-802 in breast cancer development and suggests its potential as
a target for breast cancer therapy. miR-199a, identified as an
oncogenic miRNA, exhibits elevated levels in breast cancer cells.
The tumor suppressor FOXP2 is negatively regulated by miR-199a
(91). Overexpression of miR-199a
causes a decrease in FOXP2 protein expression in breast cancer
cells, consequently promoting increased proliferation, survival and
metastasis of breast cancer cells. Cuiffo et al (92) demonstrated that manipulating
specific miRNAs or silencing FOXP2 resulted in increased BCSC
properties, leading to enhanced tumor initiation and metastasis.
They validated their initial findings on FOXP2 expression using
independent reverse transcription-quantitative PCR (ΔΔCq) analysis,
revealing a significant decrease in FOXP2 protein levels in
BCC199a, BCC199a/214 and BCCMSC cell lines. Since the decrease in
FOXP2 was associated with maintaining progenitor cell identity and
inhibiting differentiation in these cellular contexts, it was
suggested that downregulation of FOXP2 might contribute to the
emergence of CSC-like phenotypes in BCC199a, BCC199a/214 and
BCCMSC. These findings contribute to a holistic comprehension of
miRNA-mediated regulation in breast cancer, as outlined in Table II.
 | Table II.List of miRNAs regulating various FOX
proteins in breast cancer. |
Table II.
List of miRNAs regulating various FOX
proteins in breast cancer.
| miRNA | Nature of
miRNA | Targeted FOX
protein |
Upregulation/Downregulation | (Refs.) |
|---|
| miR-132 | tsmiRs | FOXA1 | Downregulation | (88) |
| miR-96 | oncomiRs | FOXO1 and
FOXO3 | Downregulation | (93) |
| miR-182 | oncomiRs | FOXO1 and
FOXO3 | Downregulation | (93) |
| miR-96-5p | tsmiRs | FOXO1 | Downregulation | (94) |
| miR-802 | tsmiRs | FOXM1 | Downregulation | (90) |
| miR-671-5p | tsmiRs | FOXM1 | Downregulation | (95) |
| miR-495 | tsmiRs | FOXC1 | Downregulation | (96) |
| miR-199a | oncomiRs | FOXP2 | Downregulation | (92) |
FOX proteins and drug resistance
Despite the condition's heterogeneity, various
therapeutics are employed to treat breast cancer. Although
beneficial, the major difficulty faced by researchers is drug
resistance. After completing a few drug cycles, patients start to
develop resistance against the administered drug, thereby reducing
its efficiency. The development of resistance involves various
signaling pathways; recently, FOX proteins are also observed in
this light. Alterations in the expression levels of various FOX
proteins contribute to the development of resistance against
anticancer drugs. Endocrine therapeutics are administered to target
and regulate the estrogen response in breast cancer cells. Although
favorable, the major setback of endocrine therapies revolves around
the emergence of resistance against anticancer drugs. Tumors that
develop resistance can regulate the elevated ER expression by
employing various processes, including drug efflux, modulation of
ER cofactor expression, and activation of other pathways, including
growth factor signaling (97).
FOXA1 exhibits a positive association with all ER+
luminal breast cancer cells and works by interacting with DNA to
open up chromatin, which then elevates the ER interaction to make
its response element more accessible. The overall increased
interaction is responsible for mediating resistance to endocrine
therapeutics, including tamoxifen (98).
Tamoxifen is an FDA-approved ER modulator used as an
endocrine therapy for breast cancer treatment. Tamoxifen does not
entirely inhibit the function of ER; instead, it binds to ER, and
this complex is recruited to chromatin, subsequently leading to the
suppression of ER target gene transcription. Similar to normal
estrogen-bound ER, tamoxifen-bound ER also requires FOXM1 to access
the chromatin and perform its function (99).
Dysregulated FOXA1 expression promotes resistance to
tamoxifen, thereby limiting its effectiveness. The overexpression
of FOXA1 leads to the upregulation of IL-8, a cytokine responsible
for metastases and survival of cancer cells and contributes to
resistance to ER inhibitors. Fu et al (98) investigated the contribution of FOXA1
to endocrine therapy resistance in ER-positive (ER+) breast cancer.
They employed various endocrine-resistant (Endo-R) breast cancer
cell lines derived from parental ER+ lines. Notably, they observed
elevated FOXA1 expression in these Endo-R cell lines compared with
their ER+ counterparts. Additionally, elevated levels of FOXA1 mRNA
were predictive of unfavorable outcomes in patients with ER+ tumor
receiving tamoxifen. Importantly, IL-8 emerged as a critical
mediator of the FOXA1/ER transcriptional reprogramming process,
promoting the growth and invasion of Endo-R cells. The study
suggests that targeting the IL-8 signaling pathway might be a
promising therapeutic approach for treating ER+ breast cancers with
elevated levels of FOXA1.
The downregulation of FOXA1 results in upregulation
of IL-6, thus promoting cancer-like properties in
tamoxifen-resistant breast cancer cells (100). In a study by Yamaguchi et
al (100), tamoxifen-resistant
(TAM-R) breast cancer cells were developed by prolonged exposure of
ER+ MCF7 cells to tamoxifen. Interestingly, TAM-R cells displayed
lower levels of both ERα, a major ER form in breast cancer, and its
co-regulator FOXA1. By contrast, these cells displayed increased
activation of the transcription factor NF-κB and its target gene,
IL6. Through experimental interventions, stable expression of
FOXA1, but not ERα, led to reduced IL6 levels in both MDA-MB-231
cells (lacking FOXA1 and ERα) and TAM-R cells, without
significantly affecting NF-κB activity. Conversely, depletion of
FOXA1 increased IL6 expression in MCF7 cells.
FOXM1, a key regulator in therapeutic resistance
among breast cancer cells, is associated with various cancer
conditions due to its influence on cell cycle regulation, autophagy
and senescence. Elevated FOXM1 levels contribute to drug
resistance, notably against the anthracycline drug epirubicin
(101). OTU domain-containing
ubiquitin aldehyde-binding protein 1 (OTUB1) acts as a regulator,
with upregulated FOXM1 resulting in increased cell proliferation
and resistance development against epirubicin. Lys48-linked
polyubiquitin chains, pivotal in proteasomal regulation, interact
with FOXM1 to modulate its levels. However, FOXM1-OTUB1 interaction
prevails, enhancing FOXM1 expression and conferring resistance
(102). Elevated FOXM1 levels also
facilitate improved DNA damage repair induced by epirubicin.
Additionally, FOXM1 regulates antiapoptotic genes XIAP and
survivin, fostering resistance against doxorubicin,
epirubicin, docetaxel and paclitaxel (103). Understanding the intricate role of
FOXM1 provides insights for targeted therapeutic interventions.
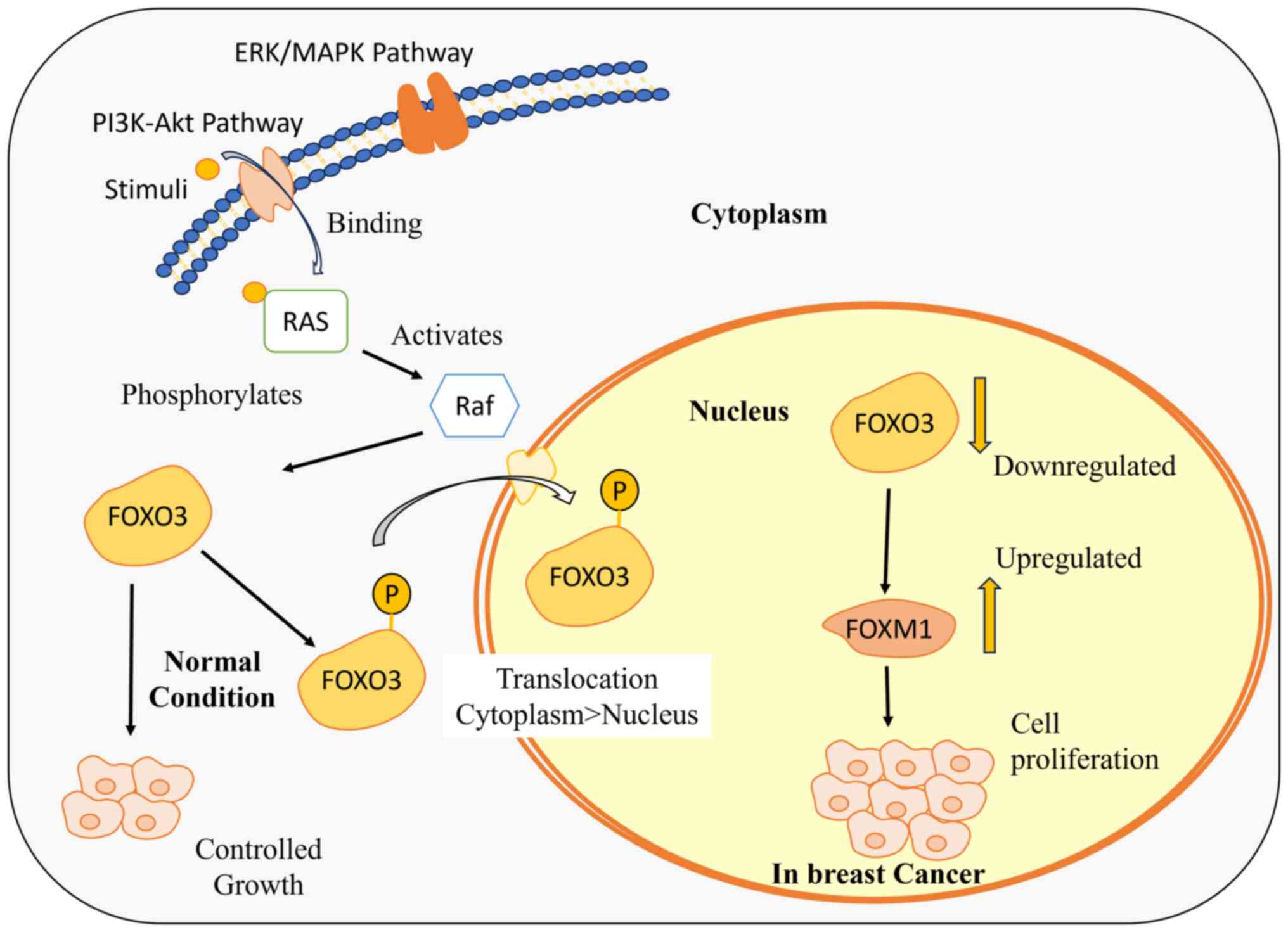
The FOXO3-FOXM1 axis serves as an informative marker
to comprehend the resistance of drug, as its dysregulation impacts
the cellular fate. FOXO3 is recognized as a tumor suppressor that
regulates cell growth, proliferation and apoptosis (30). The activity of FOXO3 is
downregulated by Ras-Raf-MEK-extracellular signal-regulated kinase
(ERK) and phosphatidylinositol 3-kinase (PI3K)-Akt/Protein Kinase B
(PKB) signaling pathways in response to various external stimuli
and growth factors. Apart from all the targets, FOXO3 downregulates
the FOXM1 expression levels and competes with FOXM1 to interact
with the same binding sites at the gene promoters (102). FOXM1 is an oncogene responsible
for cell growth and proliferation, thereby promoting tumorigenesis
(104). The downregulation of
FOXO3 induces an upregulation of FOXM1, promoting drug efflux and
cell survival. This upregulation contributes to an increase in
reactive oxygen species levels, ultimately leading to the
development of resistance against the administered drug (105) (Fig.
4).
FOX therapeutics: Current trends and future
perspectives
Chemotherapy, a widely used treatment for breast
cancer, targets and destroys cancer cells, with ongoing research
aimed at enhancing its effectiveness against the disease. Distinct
subclasses within the FOX family are identified as either oncogenes
or tumor suppressors, exerting regulatory control over cellular
fate through diverse mechanisms. This underscores their pivotal
role in the initiation and progression of tumors (106). Targeting FOX proteins as a
therapeutic intervention holds promise in the development of drugs
responsible for regulation of FOX protein levels, thereby
effectively inhibiting the growth of breast cancer cells which may
potentially improve the efficacy of existing treatment
strategies.
FOXM1 emerges as a crucial therapeutic target for
breast cancer, particularly in HER2-positive cases. Lapatinib, an
FDA-approved tyrosine kinase inhibitor, effectively downregulates
elevated HER2 levels, concurrently decreasing FOXM1 expression.
Francis et al (51)
elucidated the connection between HER2 and FOXM1, establishing the
ability of lapatinib to regulate FOXM1 at both mRNA and protein
levels. Alternatively, honokiol, a plant-derived compound,
demonstrates anti-inflammatory, antioxidative and anticancer
properties, including FOXM1 inhibition. Honokiol disrupts the
positive feedback loop regulating FOXM1 transcription, leading to
reduced mRNA and protein levels. This dual approach, targeting
FOXM1 with lapatinib and honokiol, offers a promising avenue for
developing comprehensive breast cancer therapeutics, complementing
existing strategies by Halasi et al (107).
Imipramine blue (IB), an anticancer derivative of
the FDA-approved antidepressant imipramine, demonstrates efficacy
in downregulating elevated FOXM1 expression in breast cancer, as
demonstrated by Rajamanickam et al (108). The ability of IB to target FOXM1
and inhibit homologous recombination-mediated DNA repair positions
it as a potential therapeutic agent for regulating tumor growth and
metastasis in breast cancer. Integrating IB into treatment
protocols could enable effective targeting of FOXM1 and improve the
outcomes of breast cancer therapies.
Combination therapy has become pivotal in cancer
treatment due to the limited efficacy of single-agent targeted
drugs in clinical trials despite the expanding array of options
available (109). Combining
targeted therapies with hormone therapies (endocrine treatments)
has been successful in improving the outlook for patients with
breast cancer. However, some combinations of these drugs may have a
higher chance of causing adverse effects. Therefore, identifying
precise combinations and dosing regimens to optimize therapeutic
efficacy while minimizing side effects is crucial in both
preclinical and clinical investigations (110). A notable success in breast cancer
combination therapy involves incorporating the anti-HER2 monoclonal
antibody trastuzumab into first-line chemotherapy, significantly
enhancing treatment benefits in patients with HER2-overexpressing
metastatic breast cancer (111).
Currently, the trastuzumab and paclitaxel combination is a potent
regimen for treating low-risk HER2-positive tumors, yielding
sustained positive outcomes (112). In a recent study, researchers
investigated the synergistic potential of FOXM1 inhibitors
alongside proteasome inhibitors such as Bortezomib, revealing
significant inhibition of proliferation in both ER+ and TNBC
(113). Similarly, the combined
efficacy of FOXM1 inhibitors with CDK4/6 inhibitors including
palbociclib, ribociclib and abemaciclib was demonstrated in ER+
breast cancer cells (113). These
findings offer promise for advancing translational efforts and
initiating clinical trials investigating agents targeting FOXM1,
potentially improving outcomes for patients with breast cancer.
Targeting Fox proteins, transcription factors linked
to breast cancer, is a challenge due to selectivity issues
(4). Ideally, therapies should
target only the cancer-promoting functions of Fox proteins in
tumors, leaving out the healthy tissues. However, this objective
presents several key challenges. Inhibitor off-target effects can
disrupt similar proteins in healthy cells, causing side effects.
Lin et al (114)
demonstrated that FOXC1 promotes EMT and metastasis in TNBC cell
lines. Their study revealed that inhibiting FOXC1 with a specific
inhibitor significantly suppressed TNBC cell invasion and
metastasis in preclinical models. However, a critical challenge
identified in the aforementioned study is the potential for
off-target effects of the inhibitor. Further optimization is
necessary to minimize adverse consequences on healthy cells
(114). FOXA1 plays a crucial role
in ER signaling and tumor progression in hormone receptor (HR)+
breast cancer. Robinson et al (17) employed whole-genome analysis of HR+
breast cancer and identified FOXA1 as a key regulator of
ER-mediated transcription. While targeting FOXA1 holds promise for
disrupting ER signaling and inhibiting tumor growth, a major
challenge lies in developing selective inhibitors that specifically
target FOXA1 within cancer cells without compromising its normal
functions in ER+ healthy breast tissue (17). It is important to understand that
the functional similarity between FOX proteins in healthy and
cancerous cells possess a significant challenge. The development of
inhibitors that precisely target the tumorigenic functions of FOX
proteins is challenging. This underscores the challenge of
specifically targeting the cancer-promoting functions of FOX
proteins without affecting their essential physiological roles in
healthy tissues; therefore, a deeper understanding of Fox protein
activity unique to cancer cells is crucial to overcome this
(4).
Although therapeutics targeting specific miRNAs have
not yet been applied clinically in breast cancer, notable progress
is being made. For example, a miR-122 antagonist is currently
undergoing Phase II clinical trials for hepatitis C treatment,
suggesting the potential for miRNAs to serve as viable therapeutic
targets in breast cancer (115).
Efforts are also being directed towards developing therapeutics
targeting miR-21 and miR-221. These microRNAs have been linked to
drug resistance in breast cancer, and scientists are investigating
their potential as therapeutic targets for hepatocellular carcinoma
and other cancers (116). Clinical
trials testing miRNA inhibitors as a treatment for patients with
breast cancer with multidrug resistance could be initiated in the
near future. Addressing the epigenetic alterations associated with
drug resistance in breast cancer presents significant challenges.
However, histone deacetylation inhibitors are emerging as promising
candidates for all breast cancer subtypes (117,118). It is crucial to proceed
cautiously, as these inhibitors may inadvertently alter the
expression of key receptors such as ERα, HER2 and MDR1, potentially
exacerbating drug resistance. Ongoing clinical trials of various
histone deacetylation inhibitors will provide valuable insights
into their efficacy and safety profiles (118,119).
Understanding the factors involved in cancerous cell
initiation, invasion and metastasis is essential for developing new
treatment strategies and improving existing ones. Numerous factors
have been discovered so far that have been targeted to develop
various therapeutics. FOX proteins are an ideal biomarker and
therapeutic target that would aid in understanding and improving
the existing therapies for breast cancer. Clinical trials targeting
FOX proteins, therapeutics and breast cancer, that are currently
underway are summarized in Table
III.
 | Table III.Ongoing clinical trials targeting FOX
proteins in breast cancer. |
Table III.
Ongoing clinical trials targeting FOX
proteins in breast cancer.
| Targeted FOX | Clinical trial
number | Phase | Description | (Refs.) |
|---|
| FOXM1 | NCT02187991 | Phase-II | Targeting FOXM1 as
a potential biomarker in breast cancer | (120) |
| FOXP3 | NCT04504669 | Phase-I | Analyzing change in
the mRNA expression of FOXP3 after treatment | (121) |
| FOXP3 | NCT04443348 | Phase-II | Quantifying changes
in FOXP3 during pre and post treatment | (122) |
| FOXP3 | NCT02890069 | Phase-I | Characterization of
FOXP3 using immunohistochemistry | (123) |
| FOXO3 | NCT02260661 | Phase-I | Analyzing of FOXO3
localization in tumor samples | (124) |
Conclusions
In recent years, significant advancements have been
achieved in the realm of breast cancer research, owing to
researchers' adeptness in selectively addressing multiple signaling
pathways whose dysregulation contributes to the initiation and
progression of breast cancer. Apart from the pathways, diverse
transcription factors are involved in breast cancer tumorigenesis.
FOX proteins function as transcription factors and have been linked
to numerous processes, including cell growth and apoptosis. The
altered levels of distinct subclasses of FOX proteins can act as
either oncogenes or tumor suppressors, thus inducing abnormal
proliferation in breast cancer cells. miRNAs regulate FOX proteins,
thereby controlling their expression, and the dysregulation of FOX
protein levels caused by altered miRNA levels contributes to
tumorigenesis.
Research focusing on FOX factors has provided
valuable insights into the underlying mechanisms driving cancer
progression. FOX factors often play diverse roles in cancer
progression, influencing various aspects of tumor development and
metastasis. Understanding the specific functions of FOX factors in
cancer modulation and identifying potential therapeutic targets
capable of modulating their activities could expand treatment
options for cancer patients. Inhibiting FOX factors or the pathways
they regulate holds potential for reversing drug resistance,
overcoming immune evasion and halting metastatic spread.
Therapeutic strategies targeting FOX factors may involve inhibitors
of the factors themselves, the kinases involved in FOX-mediated
pathways, or agents utilizing RNA interference to silence FOX
factor expression. These approaches represent promising avenues for
treating various types of cancer and offer hope for improved
patient outcomes. Additionally, FOX proteins can potentially relay
drug resistance, making them an appropriate therapeutic target,
with only few FOX proteins being investigated as potential
therapeutic targets. Thus, a thorough understanding of the
mechanisms behind the various classes of FOX proteins would help to
develop drugs that target the FOX proteins, control their levels,
and thereby assist in the development of efficient treatment
approaches.
Acknowledgements
Not applicable.
Funding
This research has been funded by Scientific Research Deanship at
University of Ha'il-Saudi Arabia through project number (RG-23
131).
Availability of data and materials
Not applicable.
Authors' contributions
SA and MAK wrote the major parts of the manuscript
and prepared the figures and tables. MZ and MAH revised the
manuscript. NI, SA, AA and SK performed the bibliographic research
and prepared the table and figures. S, SK and MZN conceptualized
the study and oversaw the process. All authors contributed to
writing the manuscript. All authors read and approved the final
manuscript. Data authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Weiderpass E and Stewart BW: World cancer
report: Cancer research for cancer prevention. International Agency
for Research on Cancer; Lyon: 2020
|
|
2
|
Feng Y, Spezia M, Huang S, Yuan C, Zeng Z,
Zhang L, Ji X, Liu W, Huang B, Luo W, et al: Breast cancer
development and progression: risk factors, cancer stem cells,
signaling pathways, genomics, and molecular pathogenesis. Genes
Dis. 5:77–106. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Laissue P: The forkhead-box family of
transcription factors: Key molecular players in colorectal cancer
pathogenesis. Mol Cancer. 18:52019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bach DH, Long NP, Luu TT, Anh NH, Kwon SW
and Lee SK: The dominant role of forkhead box proteins in cancer.
Int J Mol Sci. 19:32792018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Myatt SS and Lam EW: The emerging roles of
forkhead box (Fox) proteins in cancer. Nat Rev Cancer. 7:847–859.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Weigel D, Jürgens G, Küttner F, Seifert E
and Jäckle H: The homeotic gene fork head encodes a nuclear protein
and is expressed in the terminal regions of the Drosophila embryo.
Cell. 57:645–658. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Vaidya HJ, Briones Leon A and Blackburn
CC: FOXN1 in thymus organogenesis and development. Eur J Immunol.
46:1826–1837. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lam EW and Gomes AR: Forkhead box
transcription factors in cancer initiation, progression and
chemotherapeutic drug response. Front Oncol. 4:3052014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li C, Zhang K, Chen J, Chen L, Wang R and
Chu X: MicroRNAs as regulators and mediators of forkhead box
transcription factors function in human cancers. Oncotarget.
8:12433–12450. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Seachrist DD, Anstine LJ and Keri RA:
FOXA1: A pioneer of nuclear receptor action in breast cancer.
Cancers (Basel). 13:52052021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Czerny CC, Borschel A, Cai M, Otto M and
Hoyer-Fender S: FOXA1 is a transcriptional activator of
Odf2/Cenexin and regulates primary ciliation. Sci Rep.
12:214682022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cirillo LA and Zaret KS: Specific
interactions of the wing domains of FOXA1 transcription factor with
DNA. J Mol Biol. 366:720–724. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bernardo GM and Keri RA: FOXA1: A
transcription factor with parallel functions in development and
cancer. Biosci Rep. 32:113–130. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Dai X, Cheng H, Bai Z and Li J: Breast
cancer cell line classification and its relevance with breast tumor
subtyping. J Cancer. 8:3131–3141. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu Y, Zhao Y, Skerry B, Wang X,
Colin-Cassin C, Radisky DC, Kaestner KH and Li Z: Foxa1 is
essential for mammary duct formation. Genesis. 54:277–285. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brisken C and O'Malley B: Hormone action
in the mammary gland. Cold Spring Harb Perspect Biol.
2:a0031782010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Robinson JL, Macarthur S, Ross-Innes CS,
Tilley WD, Neal DE, Mills IG and Carroll JS: Androgen receptor
driven transcription in molecular apocrine breast cancer is
mediated by FoxA1. EMBO J. 30:3019–3027. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang YA, Zhao JC, Fong KW, Kim J, Li S,
Song C, Song B, Zheng B, He C and Yu J: FOXA1 potentiates
lineage-specific enhancer activation through modulating TET1
expression and function. Nucleic Acids Res. 44:8153–8164. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bernardo GM, Lozada KL, Miedler JD,
Harburg G, Hewitt SC, Mosley JD, Godwin AK, Korach KS, Visvader JE,
Kaestner KH, et al: FOXA1 is an essential determinant of ERalpha
expression and mammary ductal morphogenesis. Development.
137:2045–2054. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Takaku M, Grimm SA, De Kumar B, Bennett BD
and Wade PA: Cancer-specific mutation of GATA3 disrupts the
transcriptional regulatory network governed by Estrogen Receptor
alpha, FOXA1 and GATA3. Nucleic Acids Res. 48:4756–4768. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ghosh S, Gu F, Wang CM, Lin CL, Liu J,
Wang H, Ravdin P, Hu Y, Huang TH and Li R: Genome-wide DNA
methylation profiling reveals parity-associated hypermethylation of
FOXA1. Breast Cancer Res Treat. 147:653–659. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Slebe F, Rojo F, Vinaixa M, García-Rocha
M, Testoni G, Guiu M, Planet E, Samino S, Arenas EJ, Beltran A, et
al: FoxA and LIPG endothelial lipase control the uptake of
extracellular lipids for breast cancer growth. Nat Commun.
7:111992016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Anzai E, Hirata K, Shibazaki M, Yamada C,
Morii M, Honda T and Yamaguchi N and Yamaguchi N: FOXA1 induces
E-cadherin expression at the protein level via suppression of slug
in epithelial breast cancer cells. Biol Pharm Bull. 40:1483–1489.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ambrosone CB and Higgins MJ: Relationships
between breast feeding and breast cancer subtypes: Lessons learned
from studies in humans and in mice. Cancer Res. 80:4871–4877. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xia K, Huang W, Zhao X, Huang X, Chen Y,
Yu L and Tan Y: Increased FOXA1 levels induce apoptosis and inhibit
proliferation in FOXA1-low expressing basal breast cancer cells. Am
J Cancer Res. 12:2641–2658. 2022.PubMed/NCBI
|
|
26
|
Cantor JR and Sabatini DM: Cancer cell
metabolism: One hallmark, many faces. Cancer Discov. 2:881–898.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Y, Zhou Y and Graves DT: FOXO
transcription factors: Their clinical significance and regulation.
Biomed Res Int. 2014:9253502014.PubMed/NCBI
|
|
28
|
Jiramongkol Y and Lam EW: FOXO
transcription factor family in cancer and metastasis. Cancer
Metastasis Rev. 39:681–709. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Dumont SN, Lazar AJ, Bridge JA, Benjamin
RS and Trent JC: PAX3/7-FOXO1 fusion status in older
rhabdomyosarcoma patient population by fluorescent in situ
hybridization. J Cancer Res Clin Oncol. 138:213–220. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Parry P, Wei Y and Evans G: Cloning and
characterization of the t(X;11) breakpoint from a leukemic cell
line identify a new member of the forkhead gene family. Genes
Chromosomes Cancer. 11:79–84. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Guttilla IK and White BA: Coordinate
regulation of FOXO1 by miR-27a, miR-96, and miR-182 in breast
cancer cells. J Biol Chem. 284:23204–23216. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bullock M: FOXO factors and breast cancer:
Outfoxing endocrine resistance. Endocr Relat Cancer. 23:R113–R130.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Farhan M, Wang H, Gaur U, Little PJ, Xu J
and Zheng W: FOXO signaling pathways as therapeutic targets in
cancer. Int J Biol Sci. 13:815–827. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Di Blasio L, Gagliardi PA, Puliafito A and
Primo L: Serine/threonine kinase 3-phosphoinositide-dependent
protein Kinase-1 (PDK1) as a key regulator of cell migration and
cancer dissemination. Cancers (Basel). 9:252017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shaw RJ and Cantley LC: Ras, PI(3)K and
mTOR signalling controls tumour cell growth. Nature. 441:424–430.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tzivion G, Dobson M and Ramakrishnan G:
FoxO transcription factors; regulation by AKT and 14-3-3 proteins.
Biochim Biophys Acta. 1813:1938–1945. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kim S, Kim Y, Lee J and Chung J:
Regulation of FOXO1 by TAK1-Nemo-like kinase pathway. J Biol Chem.
285:8122–8129. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu H, Liu K and Dong Z: The role of
p21-activated kinases in cancer and beyond: Where are we heading?
Front Cell Dev Biol. 9:6413812021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Khan MA, Massey S, Ahmad I, Sada f, Akhter
N, Habib M, Mustafa S, Deo SVS and Husain SA: FOXO1 gene
downregulation and promoter methylation exhibits significant
correlation with clinical parameters in Indian breast cancer
patients. Front Genet. 13:8429432022. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Peck B, Chen CY, Ho KK, Di Fruscia P,
Myatt SS, Coombes RC, Fuchter MJ, Hsiao CD and Lam EW: SIRT
inhibitors induce cell death and p53 acetylation through targeting
both SIRT1 and SIRT2. Mol Cancer Ther. 9:844–855. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Gong C, Yao S, Gomes AR, Man EP, Lee HJ,
Gong G, Chang S, Kim SB, Fujino K, Kim SW, et al: BRCA1 positively
regulates FOXO3 expression by restricting FOXO3 gene methylation
and epigenetic silencing through targeting EZH2 in breast cancer.
Oncogenesis. 5:e2142016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu H, Song Y, Qiu H, Liu Y, Luo K, Yi Y,
Jiang G, Lu M, Zhang Z, Yin J, et al: Downregulation of FOXO3a by
DNMT1 promotes breast cancer stem cell properties and
tumorigenesis. Cell Death Differ. 27:966–983. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sanders DA, Gormally MV, Marsico G,
Beraldi D, Tannahill D and Balasubramanian S: FOXM1 binds directly
to non-consensus sequences in the human genome. Genome Biol.
16:1302015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Korver W, Roose J, Heinen K, Weghuis DO,
de Bruijn D, van Kessel AG and Clevers H: The human
TRIDENT/HFH-11/FKHL16 gene: Structure, localization, and promoter
characterization. Genomics. 46:435–442. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Kalathil D, John S and Nair AS: FOXM1 and
cancer: Faulty cellular signaling derails homeostasis. Front Oncol.
10:6268362021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ye H, Kelly TF, Samadani U, Lim L, Rubio
S, Overdier DG, Roebuck KA and Costa RH: Hepatocyte nuclear factor
3/fork head homolog 11 is expressed in proliferating epithelial and
mesenchymal cells of embryonic and adult tissues. Mol Cell Biol.
17:1626–1641. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Halasi M and Gartel AL: FOX(M1) news-it is
cancer. Mol Cancer Ther. 12:245–254. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Xue J, Lin X, Chiu WT, Chen YH, Yu G, Liu
M, Feng XH, Sawaya R, Medema RH, Hung MC and Huang S: Sustained
activation of SMAD3/SMAD4 by FOXM1 promotes TGF-β-dependent cancer
metastasis. J Clin Invest. 124:564–579. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Speirs V and Walker RA: New perspectives
into the biological and clinical relevance of oestrogen receptors
in the human breast. J Pathol. 211:499–506. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Iqbal N and Iqbal N: Human epidermal
growth factor Receptor 2 (HER2) in cancers: Overexpression and
therapeutic implications. Mol Biol Int. 2014:8527482014. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Francis RE, Myatt SS, Krol J, Hartman J,
Peck B, McGovern UB, Wang J, Guest SK, Filipovic A, Gojis O, et al:
FoxM1 is a downstream target and marker of HER2 overexpression in
breast cancer. Int J Oncol. 35:57–68. 2009.PubMed/NCBI
|
|
52
|
Chen X, Wei H, Li J, Liang X, Dai S, Jiang
L, Guo M, Qu L, Chen Z, Chen L and Chen Y: Structural basis for DNA
recognition by FOXC2. Nucleic Acids Res. 47:3752–3764. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Pierrou S, Enerbäck S and Carlsson P:
Selection of high-affinity binding sites for sequence-specific, DNA
binding proteins from random sequence oligonucleotides. Anal
Biochem. 229:99–105. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Yin L, Duan JJ, Bian XW and Yu SC:
Triple-negative breast cancer molecular subtyping and treatment
progress. Breast Cancer Res. 22:612020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Han B, Bhowmick N, Qu Y, Chung S, Giuliano
AE and Cui X: FOXC1: An emerging marker and therapeutic target for
cancer. Oncogene. 36:3957–3963. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wang J, Ray PS, Sim MS, Zhou XZ, Lu KP,
Lee AV, Lin X, Bagaria SP, Giuliano AE and Cui X: FOXC1 regulates
the functions of human basal-like breast cancer cells by activating
NF-kappaB signaling. Oncogene. 31:4798–4802. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Nieto MA: Epithelial plasticity: A common
theme in embryonic and cancer cells. Science. 342:12348502013.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Bloushtain-Qimron N, Yao J, Snyder EL,
Shipitsin M, Campbell LL, Mani SA, Hu M, Chen H, Ustyansky V,
Antosiewicz JE, et al: Cell type-specific DNA methylation patterns
in the human breast. Proc Natl Acad Sci USA. 105:14076–14081. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Powell AA, Talasaz AH, Zhang H, Coram MA,
Reddy A, Deng G, Telli ML, Advani RH, Carlson RW, Mollick JA, et
al: Single cell profiling of circulating tumor cells:
Transcriptional heterogeneity and diversity from breast cancer cell
lines. PLoS One. 7:e337882012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Lindley LE and Briegel KJ: Molecular
characterization of TGFbeta-induced epithelial-mesenchymal
transition in normal finite lifespan human mammary epithelial
cells. Biochem Biophys Res Commun. 399:659–664. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Clark KL, Halay ED, Lai E and Burley SK:
Co-crystal structure of the HNF-3/fork head DNA-recognition motif
resembles histone H5. Nature. 364:412–420. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Perumal K, Dirr HW and Fanucchi S: A
single amino acid in the hinge loop region of the FOXP forkhead
domain is significant for dimerisation. Protein J. 34:111–121.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Stroud JC, Wu Y, Bates DL, Han A, Nowick
K, Paabo S, Tong H and Chen L: Structure of the forkhead domain of
FOXP2 bound to DNA. Structure. 14:159–166. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Shigekawa T, Ijichi N, Ikeda K,
Horie-Inoue K, Shimizu C, Saji S, Aogi K, Tsuda H, Osaki A, Saeki T
and Inoue S: FOXP1, an estrogen-inducible transcription factor,
modulates cell proliferation in breast cancer cells and 5-year
recurrence-free survival of patients with tamoxifen-treated breast
cancer. Horm Cancer. 2:286–297. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Beelen K, Hoefnagel LD, Opdam M, Wesseling
J, Sanders J, Vincent AD, van Diest PJ and Linn SC: PI3K/AKT/mTOR
pathway activation in primary and corresponding metastatic breast
tumors after adjuvant endocrine therapy. Int J Cancer.
135:1257–1263. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Banham AH, Beasley N, Campo E, Fernandez
PL, Fidler C, Gatter K, Jones M, Mason DY, Prime JE, Trougouboff P,
et al: The FOXP1 winged helix transcription factor is a novel
candidate tumor suppressor gene on chromosome 3p. Cancer Res.
61:8820–8829. 2001.PubMed/NCBI
|
|
68
|
Liu Y, Chen T, Guo M, Li Y, Zhang Q, Tan
G, Yu L and Tan Y: FOXA2-interacting FOXP2 prevents
epithelial-mesenchymal transition of breast cancer cells by
stimulating E-cadherin and PHF2 transcription. Front Oncol.
11:6050252021. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Sada f, Akhter N, Alharbi RA, Sindi AAA,
Najm MZ, Alhumaydhi FA, Khan MA, Deo SVS and Husain SA: Epigenetic
alteration and its association with downregulated FOXP3 gene in
indian breast cancer patients. Front Genet. 12:7814002021.
View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Liu C, Han J, Li X, Huang T, Gao Y, Wang
B, Zhang K, Wang S, Zhang W, Li W, et al: FOXP3 inhibits the
metastasis of breast cancer by downregulating the expression of
MTA1. Front Oncol. 11:6561902021. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Ma B, Miao W, Xiao J, Chen X, Xu J and Li
Y: The role of FOXP3 on tumor metastasis and its interaction with
traditional Chinese medicine. Molecules. 27:67062022. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Dai X, Cheng H, Chen X, Li T, Zhang J, Jin
G, Cai D and Huang Z: FOXA1 is prognostic of triple negative breast
cancers by transcriptionally suppressing SOD2 and IL6. Int J Biol
Sci. 15:1030–1041. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Cao J, Wang X, Wang D, Ma R, Li X, Feng H,
Wang J, Liu S and Wang L: PGC-1β cooperating with FOXA2 inhibits
proliferation and migration of breast cancer cells. Cancer Cell
Int. 19:932019. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Song Y, Zeng S, Zheng G, Chen D, Li P,
Yang M, Luo K, Yin J, Gu Y, Zhang Z, et al: FOXO3a-driven miRNA
signatures suppresses VEGF-A/NRP1 signaling and breast cancer
metastasis. Oncogene. 40:777–790. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Wei G, Yang X, Lu H, Zhang L, Wei Y, Li H,
Zhu M and Zhou X: Prognostic value and immunological role of FOXM1
in human solid tumors. Aging (Albany NY). 14:9128–9148. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Ha M and Kim VN: Regulation of microRNA
biogenesis. Nat Rev Mol Cell Biol. 15:509–524. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Broughton JP, Lovci MT, Huang JL, Yeo GW
and Pasquinelli AE: Pairing beyond the seed supports microRNA
targeting specificity. Mol Cell. 64:320–333. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Arora T, Kausar MA, Aboelnaga SM, Anwar S,
Hussain MA, Sadaf S, Kaur S, Eisa AA, Shingatgeri VMM, Najm MZ and
Aloliqi AA: miRNAs and the Hippo pathway in cancer: Exploring the
therapeutic potential (Review). Oncol Rep. 48:1352022. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Denli AM, Tops BB, Plasterk RH, Ketting RF
and Hannon GJ: Processing of primary microRNAs by the
microprocessor complex. Nature. 432:231–235. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Alarcón CR, Lee H, Goodarzi H, Halberg N
and Tavazoie SF: N6-methyladenosine marks primary microRNAs for
processing. Nature. 519:482–485. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Okada C, Yamashita E, Lee SJ, Shibata S,
Katahira J, Nakagawa A, Yoneda Y and Tsukihara T: A high-resolution
structure of the pre-microRNA nuclear export machinery. Science.
326:1275–1279. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Zhang H, Kolb FA, Jaskiewicz L, Westhof E
and Filipowicz W: Single processing center models for human Dicer
and bacterial RNase III. Cell. 118:57–68. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Wang W and Luo YP: MicroRNAs in breast
cancer: Oncogene and tumor suppressors with clinical potential. J
Zhejiang Univ Sci B. 16:18–31. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Corcoran C, Friel AM, Duffy MJ, Crown J
and O'Driscoll L: Intracellular and extracellular microRNAs in
breast cancer. Clin Chem. 57:18–32. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Guo X, Connick MC, Vanderhoof J, Ishak MA
and Hartley RS: MicroRNA-16 modulates HuR regulation of cyclin E1
in breast cancer cells. Int J Mol Sci. 16:7112–7132. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Jin T, Suk Kim H, Ki Choi S, Hye Hwang E,
Woo J, Suk Ryu H, Kim K, Moon A and Kyung Moon W: microRNA-200c/141
upregulates SerpinB2 to promote breast cancer cell metastasis and
reduce patient survival. Oncotarget. 8:32769–32782. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Chen S, Wang Y, Ni C, Meng G and Sheng X:
HLF/miR-132/TTK axis regulates cell proliferation, metastasis and
radiosensitivity of glioma cells. Biomed Pharmacother. 83:898–904.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Wang D, Ren J, Ren H, Fu JL and Yu D:
MicroRNA-132 suppresses cell proliferation in human breast cancer
by directly targeting FOXA1. Acta Pharmacol Sin. 39:124–131. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Gao T, Zou M, Shen T and Duan S:
Dysfunction of miR-802 in tumors. J Clin Lab Anal. 35:e239892021.
View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Yuan F and Wang W: MicroRNA-802 suppresses
breast cancer proliferation through downregulation of FoxM1. Mol
Med Rep. 12:4647–4651. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Wang Q, Ye B, Wang P, Yao F, Zhang C and
Yu G: Overview of microRNA-199a regulation in cancer. Cancer Manag
Res. 11:10327–10335. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Cuiffo BG, Campagne A, Bell GW, Lembo A,
Orso F, Lien EC, Bhasin MK, Raimo M, Hanson SE, Marusyk A, et al:
MSC-regulated microRNAs converge on the transcription factor FOXP2
and promote breast cancer metastasis. Cell Stem Cell. 15:762–774.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Lin H, Dai T, Xiong H, Zhao X, Chen X, Yu
C, Li J, Wang X and Song L: Unregulated miR-96 induces cell
proliferation in human breast cancer by downregulating
transcriptional factor FOXO3a. PLoS One. 5:e157972010. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Yin Z, Wang W, Qu G, Wang L, Wang X and
Pan Q: MiRNA-96-5p impacts the progression of breast cancer through
targeting FOXO3. Thorac Cancer. 11:956–963. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Tan X, Li Z, Ren S, Rezaei K, Pan Q,
Goldstein AT, Macri CJ, Cao D, Brem RF and Fu SW: Dynamically
decreased miR-671-5p expression is associated with oncogenic
transformation and radiochemoresistance in breast cancer. Breast
Cancer Res. 21:892019. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kumar U, Hu Y, Masrour N,
Castellanos-Uribe M, Harrod A, May ST, Ali S, Speirs V, Coombes RC
and Yagüe E: MicroRNA-495/TGF-β/FOXC1 axis regulates multidrug
resistance in metaplastic breast cancer cells. Biochem Pharmacol.
192:1146922021. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Badve S, Turbin D, Thorat MA, Morimiya A,
Nielsen TO, Perou CM, Dunn S, Huntsman DG and Nakshatri H: FOXA1
expression in breast cancer-correlation with luminal subtype A and
survival. Clin Cancer Res. 13:4415–4421. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Fu X, Jeselsohn R, Pereira R,
Hollingsworth EF, Creighton CJ, Li F, Shea M, Nardone A, De Angelis
C, Heiser LM, et al: FOXA1 overexpression mediates endocrine
resistance by altering the ER transcriptome and IL-8 expression in
ER-positive breast cancer. Proc Natl Acad Sci USA. 113:E6600–E6609.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Hurtado A, Holmes KA, Ross-Innes CS,
Schmidt D and Carroll JS: FOXA1 is a key determinant of estrogen
receptor function and endocrine response. Nat Genet. 43:27–33.
2011. View
Article : Google Scholar : PubMed/NCBI
|
|
100
|
Yamaguchi N, Nakayama Y and Yamaguchi N:
Down-regulation of Forkhead box protein A1 (FOXA1) leads to cancer
stem cell-like properties in tamoxifen-resistant breast cancer
cells through induction of interleukin-6. J Biol Chem.
292:8136–8148. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Karunarathna U, Kongsema M, Zona S, Gong
C, Cabrera E, Gomes AR, Man EP, Khongkow P, Tsang JW, Khoo US, et
al: OTUB1 inhibits the ubiquitination and degradation of FOXM1 in
breast cancer and epirubicin resistance. Oncogene. 35:1433–1444.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Nestal de Moraes G, Delbue D, Silva KL,
Robaina MC, Khongkow P, Gomes AR, Zona S, Crocamo S, Mencalha AL,
Magalhães LM, et al: FOXM1 targets XIAP and Survivin to modulate
breast cancer survival and chemoresistance. Cell Signal.
27:2496–2505. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Nestal de Moraes G, Bella L, Zona S,
Burton MJ and Lam EW: Insights into a critical role of the
FOXO3a-FOXM1 axis in DNA damage response and genotoxic drug
resistance. Curr Drug Targets. 17:164–177. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Di Fruscia P, Zacharioudakis E, Liu C,
Moniot S, Laohasinnarong S, Khongkow M, Harrison IF, Koltsida K,
Reynolds CR, Schmidtkunz K, et al: The discovery of a highly
selective
5,6,7,8-tetrahydrobenzo[4,5]thieno[2,3-d]pyrimidin-4(3H)-one SIRT2
inhibitor that is neuroprotective in an in vitro Parkinson's
disease model. ChemMedChem. 10:69–82. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Saba R, Alsayed A, Zacny JP and Dudek AZ:
The role of forkhead box protein M1 in breast cancer progression
and resistance to therapy. Int Breast Cancer.
2016:97681832016.PubMed/NCBI
|
|
106
|
Ji X, Tian X, Feng S, Zhang L, Wang J, Guo
R, Zhu Y, Yu X, Zhang Y, Du H, et al: Intermittent F-actin
perturbations by magnetic fields inhibit breast cancer metastasis.
Research (Wash DC). 6:00802023.PubMed/NCBI
|
|
107
|
Halasi M, Hitchinson B, Shah BN, Váraljai
R, Khan I, Benevolenskaya EV, Gaponenko V, Arbiser JL and Gartel
AL: Honokiol is a FOXM1 antagonist. Cell Death Dis. 9:842018.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Rajamanickam S, Panneerdoss S, Gorthi A,
Timilsina S, Onyeagucha B, Kovalskyy D, Ivanov D, Hanes MA,
Vadlamudi RK, Chen Y, et al: Inhibition of FoxM1-mediated DNA
repair by imipramine blue suppresses breast cancer growth and
metastasis. Clin Cancer Res. 22:3524–3536. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Lopez JS and Banerji U: Combine and
conquer: Challenges for targeted therapy combinations in early
phase trials. Nat Rev Clin Oncol. 14:57–66. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Martel S, Bruzzone M, Ceppi M, Maurer C,
Ponde NF, Ferreira AR, Viglietti G, Del Mastro L, Prady C, de
Azambuja E and Lambertini M: Risk of adverse events with the
addition of targeted agents to endocrine therapy in patients with
hormone receptor-positive metastatic breast cancer: A systematic
review and meta-analysis. Cancer Treat Rev. 62:123–132. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Pegram MD, Konecny GE, O'Callaghan C,
Beryt M, Pietras R and Slamon DJ: Rational combinations of
trastuzumab with chemotherapeutic drugs used in the treatment of
breast cancer. J Natl Cancer Inst. 96:739–749. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Waks AG and Winer EP: Breast cancer
treatment: A review. JAMA. 321:288–300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Guillen VS, Ziegler Y, Gopinath C, Kumar
S, Dey P, Plotner BN, Dawson NZ, Kim SH, Katzenellenbogen JA and
Katzenellenbogen BS: Effective combination treatments for breast
cancer inhibition by FOXM1 inhibitors with other targeted cancer
drugs. Breast Cancer Res Treat. 198:607–621. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Lin Z, Huang W, He Q, Li D, Wang Z, Feng
Y, Liu D, Zhang T, Wang Y, Xie M, et al: FOXC1 promotes HCC
proliferation and metastasis by Upregulating DNMT3B to induce DNA
Hypermethylation of CTH promoter. J Exp Clin Cancer Res. 40:502021.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Bader AG and Lammers P: The therapeutic
potential of microRNAs. Innov Pharm Technol. 52–55. 2011.
|
|
116
|
Broderick JA and Zamore PD: MicroRNA
therapeutics. Gene Ther. 18:1104–1110. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Tate CR, Rhodes LV, Segar HC, Driver JL,
Pounder FN, Burow ME and Collins-Burow BM: Targeting
triple-negative breast cancer cells with the histone deacetylase
inhibitor panobinostat. Breast Cancer Res. 14:R792012. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Linares A, Dalenc F, Balaguer P, Boulle N
and Cavailles V: Manipulating protein acetylation in breast cancer:
A promising approach in combination with hormonal therapies? J
Biomed Biotechnol. 2011:8569852011. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Khafaga AF, Shamma RN, Abdeen A, Barakat
AM, Noreldin AE, Elzoghby AO and Sallam MA: Celecoxib repurposing
in cancer therapy: Molecular mechanisms and nanomedicine-based
delivery technologies. Nanomedicine (Lond). 16:1691–1712. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
National Library of Medicine (NIH), .
Study to Compare Alisertib with Paclitaxel vs Paclitaxel Alone in
Metastatic or Locally Recurrent Breast Cancer. Clinical trial:
NCT02187991. NIH; Bethesda, MD: 2022, https://www.mycancergenome.org/content/clinical_trials/NCT02187991/December
1–2022
|
|
121
|
National Library of Medicine (NIH), .
First Time in Human Study of AZD8701 With or Without Durvalumab in
Participants with Advanced Solid Tumours. Clinical trial:
NCT04504669. NIH; Bethesda, MD: 2022, https://www.mycancergenome.org/content/clinical_trials/NCT04504669/December
1–2022
|
|
122
|
National Library of Medicine (NIH), .
Pre-op Pembro + Radiation Therapy in Breast Cancer (P-RAD).
Clinical trial: NCT04443348. NIH; Bethesda, MD: 2022, https://www.mycancergenome.org/content/clinical_trials/NCT04443348/December
1–2022
|
|
123
|
National Library of Medicine (NIH), . A
Study of PDR001 in Combination with LCL161, Everolimus or
Panobinostat. Clinical trial: NCT02890069. NIH; Bethesda, MD: 2022,
https://www.mycancergenome.org/content/clinical_trials/NCT02890069/December
1–2022
|
|
124
|
National Library of Medicine (NIH), .
Phase I, Dose Study to Look at the Safety and Pharmacokinetics of
AZD8835 in Patients with Advanced Solid Tumours. Clinical trial:
NCT02260661. NIH; Bethesda, MD: 2022, https://www.mycancergenome.org/content/clinical_trials/NCT02260661/December
1–2022
|