Introduction
Uveitis is a leading cause of visual morbidity and
causes approximately 30,000 new cases of legal blindness annually
in the United States alone (1).
Although uveitis has been previously investigated, the exact
pathogenesis remains to be determined (2). It has been proposed that uveitis
results mainly from autoimmune reaction defects (1). However, since the exact pathogenesis
of uveitis remains to be identified, there are no specific and few
effective drugs for treating uveitis. Experimental autoimmune
uveoretinitis (EAU) is an attractive animal model for the study of
autoimmune uveitis and even other relevant autoimmune diseases that
affect humans (3) and it has been
recognized as an ideal animal model for drug treatment and
assessment. This model has many advantages, including the fact that
it is highly reproducible, the clinical manifestations in the rats
are easy to detect, and many test results are amenable to
quantitative processing.
Certain immunodeficiency syndromes and allergic
reactions are caused by excessive immune responses stimulated by
endogenous or exogenous antigens that mimic self-antigens (4). The majority of currently available
anti-allergy drugs are chemically synthetic drugs characterized by
their strong efficacy but also serious side effects (5). Compared with these chemically
synthetic drugs, natural drugs are advantageous as they produce
fewer side effects (6). Results of
previous studies suggested that the mutual effects of quercetin and
isoquercetin can reduce the incidence of asthma to some extent;
kaempferol, a flavonoid, can inhibit the degranulation of mast
cells; and triterpenes can effectively treat pleural
hypersensitivity (6).
In the present study, the curative effects of
natural compounds on EAU were investigated in a relevant rat model.
The findings provide a theoretical and experimental basis for the
treatment of EAU.
Materials and methods
Animals
A total of 25 EAU rats that were housed and fed in
animal cages in West China Hospital were enrolled in our study. The
rats were male, ranging from 6 to 8 weeks, with an average weight
of 160±32.5 g. The rats were randomly divided as follows (n=5 per
group): Alkaloids (berberine hydrochloride), saponins (steroidal
saponins), flavonoids (baicalein), phenols (chlorogenic acid) and
physiological saline groups. The present study was approved by the
ethics commitee of the Animal Centre of West China Hospital.
Main reagents
The main reagents used were: Goat anti-rabbit IL-17
primary antibody, and biotinylated donkey anti-sheep ABC
immunohistochemistry kit (Roche Diagnostics, Indianapolis, IN,
USA); RT-polymerase chain reaction (PCR) reagent (Takara Bio, Inc.,
Otsu, Japan); and cell apoptosis detection kit (Roche
Diagnostics).
Animal grouping and EAU model
establishment
EAU models were established in accordance with the
methods employed by Oh-i et al (7). The 25 rats were randomly divided into
groups as follows (n=5 per group): alkaloids group (2 mg/kg
berberine hydrochloride), saponins group (2.5 mg/kg steroidal
saponins), flavonoids group (2.5 mg/kg baicalein), phenols group (2
mg/kg chlorogenic acid) and physiological saline group (2 mg/kg
normal saline). The amounts of the natural compounds injected
intraperitoneally were confirmed to be appropriate by preliminary
experiments.
Slit lamp microscope observation
The preocular reactions of the rats treated by one
of the different natural compounds were observed once a day under a
slit-lamp microscope (Hangzhou Medical Science and Technology
Company, Hangzhou, China) and their inflammatory reactions were
recorded and scored according to the criteria of Kohno et al
(8). The criteria were as follows:
No inflammation present (0 points), iris congestion and mild
retinal vasculitis (1 point), mild fibrous tissue exudation in
anterior chamber (2 points), moderate exudation and mild empyema in
anterior chamber (3 points), severe bleeding and empyema in
anterior chamber (4 points).
Pathological observation of retina
Following treatment the rats in each group were kept
for 2 weeks and were fed as normal during that period of time,
prior to injection with 10% chloral hydrate. The rats were
subsequently sacrificed using cervical dislocation. The eye balls
were extracted and fixed in a 4% formaldehyde solution, sliced by
routine paraffin sectioning, stained with hematoxylin and eosin,
and observed under a microscope (Olympus CX23, Tokyo, Japan). The
severity of pathology was evaluated according to the Koho H
evaluation system (8): Normal (0
points), retinal receptor lesion (1–2 points), external granular
layer lesions of the retina (3–4 points), retinal internal granular
layer lesions (5–6 points), and retinal cellular layer lesions (7
points).
RT-PCR detection and ELISA detection
Tissue sample RNA extraction
Frozen tissue samples (0.1 g) were taken from liquid
nitrogen and allowed to melt on the ice. RNA Plus (0.45 ml)
(Takara, Shanghai, China) was added to each sample, then a
pre-cooled mortar was used to pulverize the tissues. After
transferring to a 15 ml Eppendorf (EP) tube (Axygen, New York, NY,
USA), 0.45 ml RNA Plus was used to wash the mortar, which was added
to the pulverized tissue sample. Chloroform (200 µl) was
added into a centrifuge tube and the mix was agitated for 15 sec,
after which the phases were allowed to separate on ice for 15 min.
The tubes were centrifuged at 8,000 × g for 15 min at 4°C. The
resultant supernatants were then transferred into de-RNase EP tubes
(Axygen), the same amount of isopropanol was added to each tube,
the tubes were inverted to mix the solutions, and then set aside on
ice for 10 min. The tubes were centrifuged at 8,000 × g for 10 min
at 4°C and the supernatants were discarded. Then, 750 µl 75%
ethanol was added to each tube, the tubes were gently mixed by
inversion, and then centrifuged at 8,000 × g for 10 min at 4°C. The
supernatants were discarded, and any residual ethanol was removed
by air drying. Subsequently, de-RNase H2O was added, and
after measuring the quality of the extracted RNA, the samples were
preserved for reverse transcription.
RT-PCR
PCR was used to detect the expression of
IL-17 gene, the EAU marker gene. The Oligon 7.0 software
(Molecular Biology Insights, Inc., Cascade, CO, USA) was used to
design the primers. The primer sequences used were: Upstream primer
5-′CCCATCATTGCAATAGCAGG-3′, and downstream primer
5′-GCTCAAACTYCTGCTCCTGA-3′. The length of the expected amplified
fragment was 170 bp. The parameters used for the fluorescent
quantitative PCR reaction system were: SYBR-Green reagent (5
µl), forward primer (0.5 µl), reverse primer (0.5
µl), and reverse transcriptase product (1 µl,
ddH2O: 3 µl). The PCR conditions were 95°C for 5
min and then 40 cycles of 95°C for 10 sec denaturation, followed by
60°C for 30 sec annealing/extension.
ELISA
ELISA (Roche Diagnostics) was applied to detect the
amount of IL-17 protein expression. Surgery was performed strictly
in accordance with the protocol of the Roche kit. The absorbance
value was measured at 450 nm after reaction completion and the
amount of protein expression was calculated according to a standard
protein curve (9).
Apoptosis detection using flow
cytometry
In the present study, apoptosis of the cells of
rat's retinas treated with different natural compounds were
observed using a flow cytometer and operations were performed
strictly in accordance with the protocol of the Roche kit.
Western blotting detection
A Roche animal cell protein extraction kit was used
to extract the samples of total protein according to the
manufacturer's instructions. The primary antibody was mouse
anti-human IL-17 monoclonal antibody (Santa Cruz Biotechnology, New
York, NY, USA, cat. no.: sc-374218. The secondary antibody was
HRP-goat anti-mouse antibody (Santa Cruz Biotechnology, cat. no.:
sc-395758). Western blotting was conducted as previously described
(7).
Statistical analysis
SPSS 20.0 software (IBM SPSS, Armonk, NY, USA) was
used for statistical analyses. Measurement data were shown as mean
± standard deviation and count data were analyzed by the
χ2 test. P<0.05 was considered to indicate
statistically significant differences.
Results
Ocular inflammation
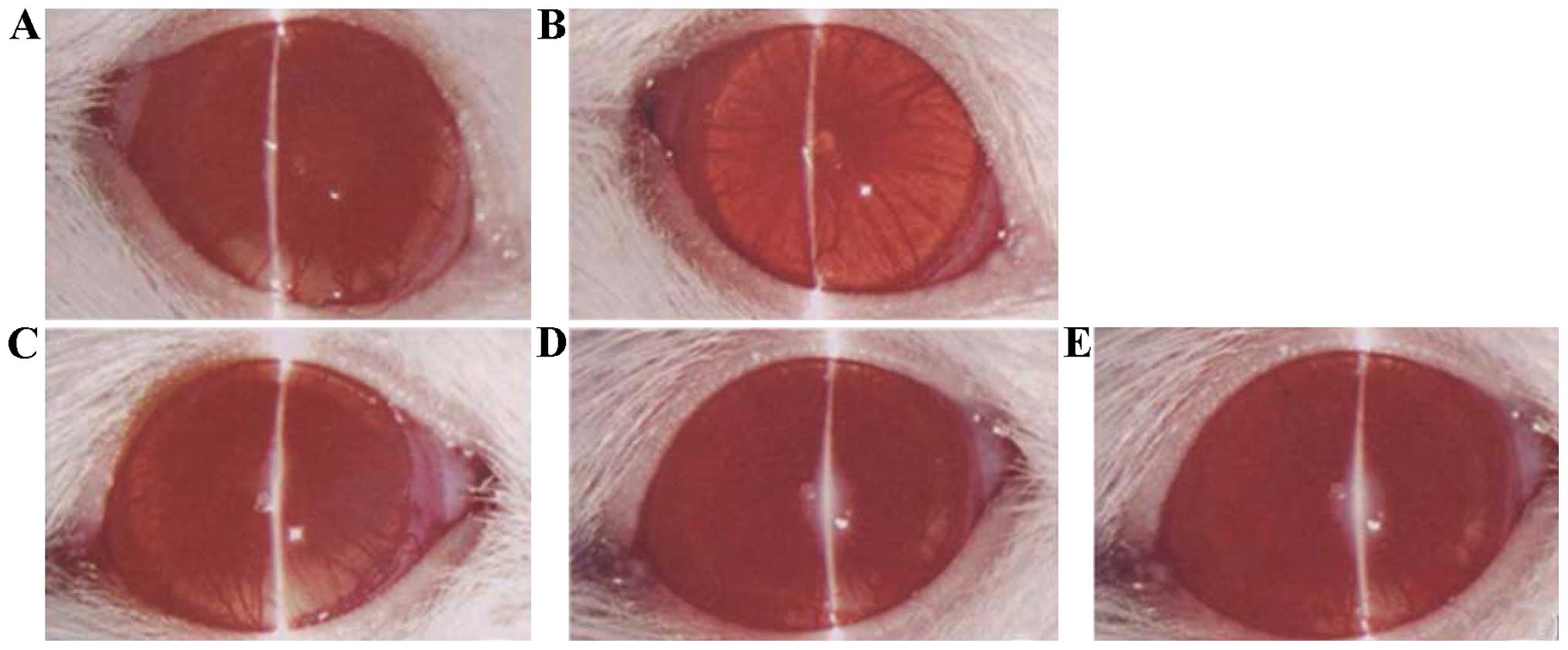
By observing the state of ocular inflammation of
rats treated with different natural compounds, it was found that
rats in the phenol (chlorogenic acid) and normal saline groups had
serious ocular vascular dilatation, iris hemorrhage and purulent
exudation; rats in the alkaloid (berberine hydrochloride) and
flavonoid (baicalein) groups had slight inflammation; and rats in
the saponin (steroidal saponins) group had the slightest
inflammation (Fig. 1).
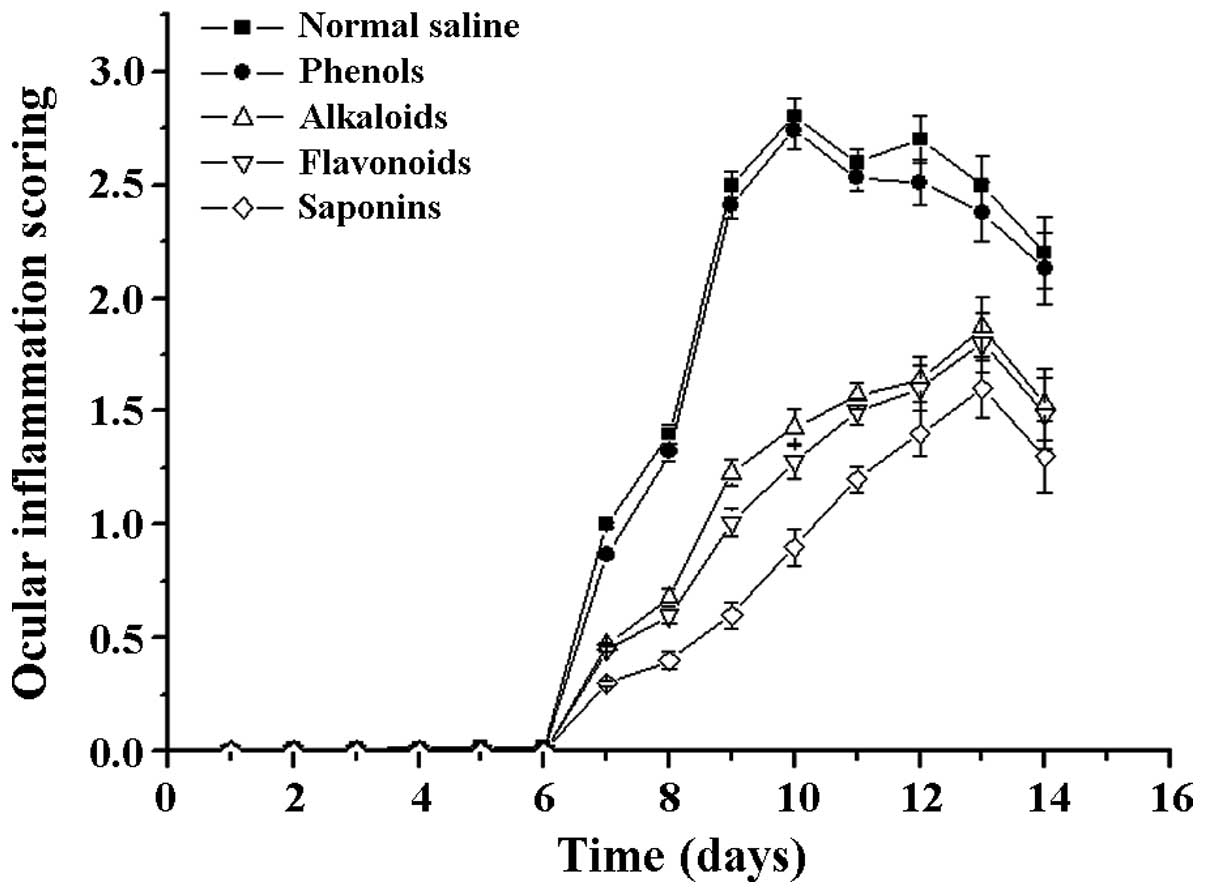
Following a comparison of the clinical scores of the
treatment groups, the differences were found to be statistically
significant (F=6.72 P=0.003). Compared with the scores of the
normal saline group, the clinical scores of the saponins (steroidal
saponins) group were significantly reduced, and the difference was
statistically significant (t=−0.634 P=0.002). By contrast, the
difference between the phenols (chlorogenic acid) and physiological
saline groups was not statistically significant (t=−0.334 P=0.538;
Fig. 2).
Histopathological examination
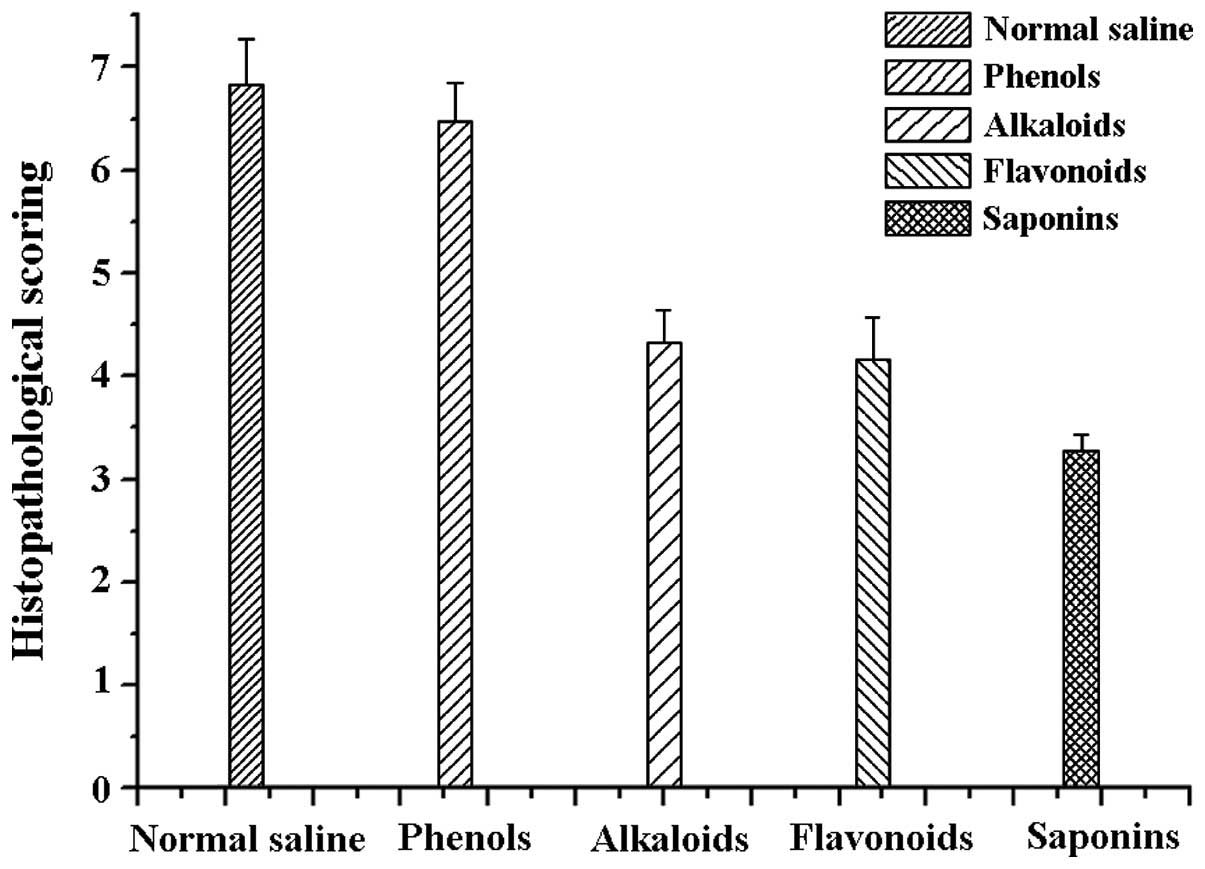
The ocular pathology scoring of rats differed
between groups. The rats on saponins had the smallest score
(3.28±0.159), which was in agreement with the above results. Rats
administered alkaloids (4.32±0.328), and flavonoids (4.17±0.401)
yielded similar results. Rats administered phenols (6.47±0.369) and
normal saline (6.83±0.437) had the most retinal damage. There was
no statistical difference between the normal saline and phenols
group (F=12.76 P=0.000). The differences between the normal saline
group and the remaining three groups were statistically significant
(t=−1.48, P=0.04). Specifically, the difference between the normal
saline and saponins group was statistically significant (t=−1.27,
P=0.01; Fig. 3).
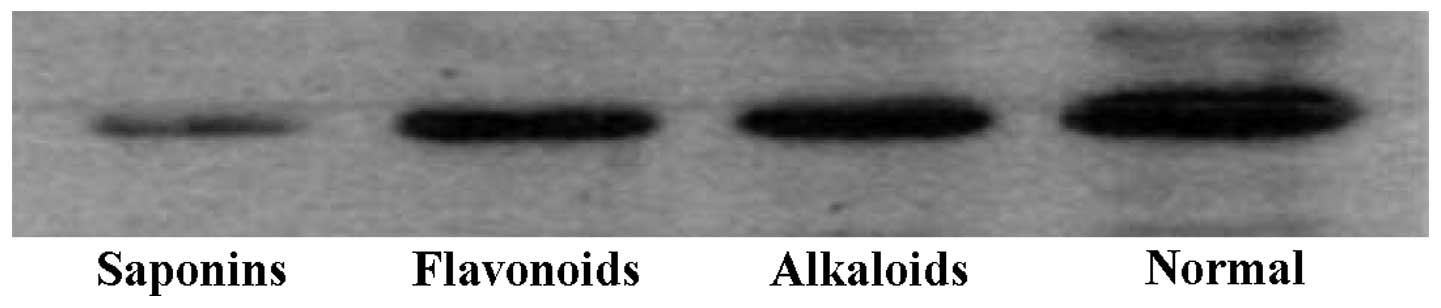
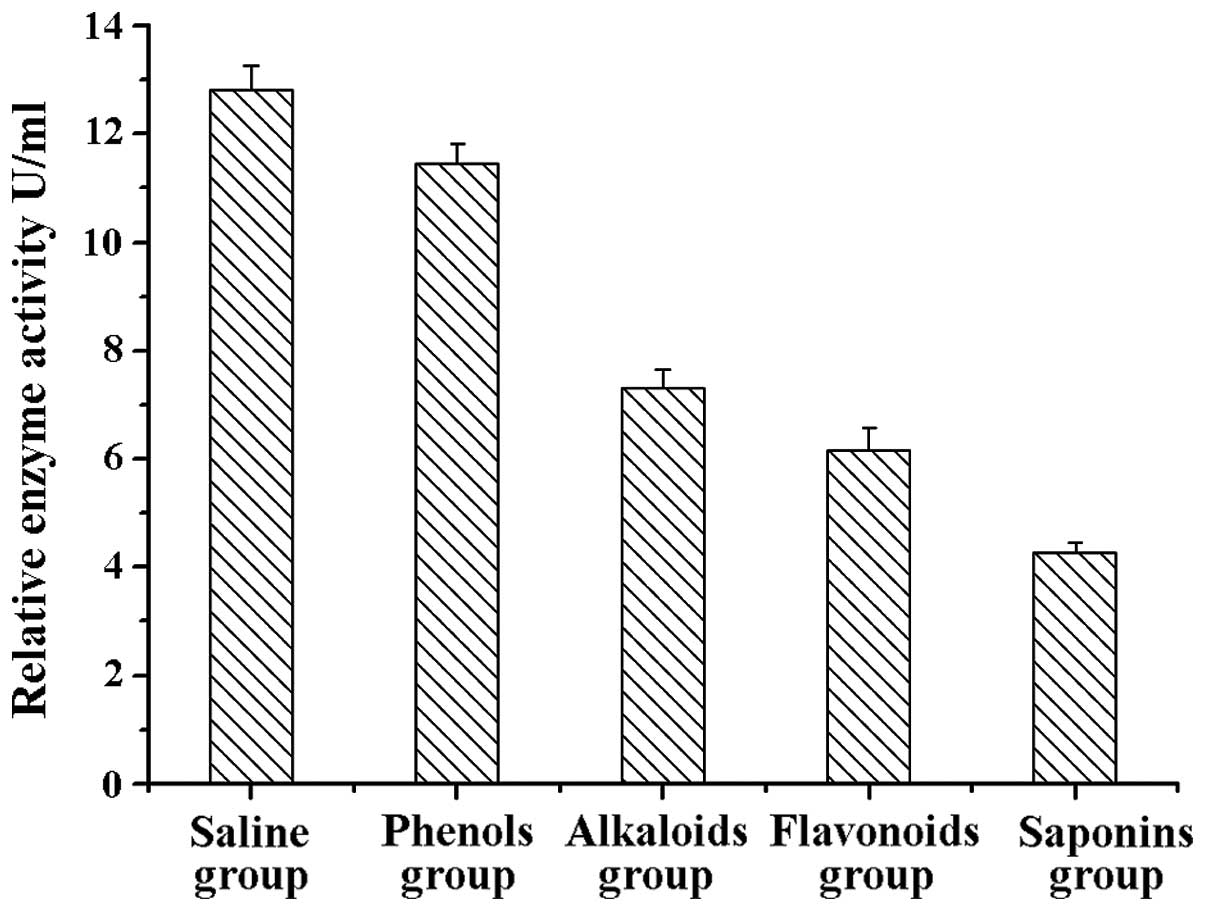
RT-PCR, ELISA, and western blot
analysis
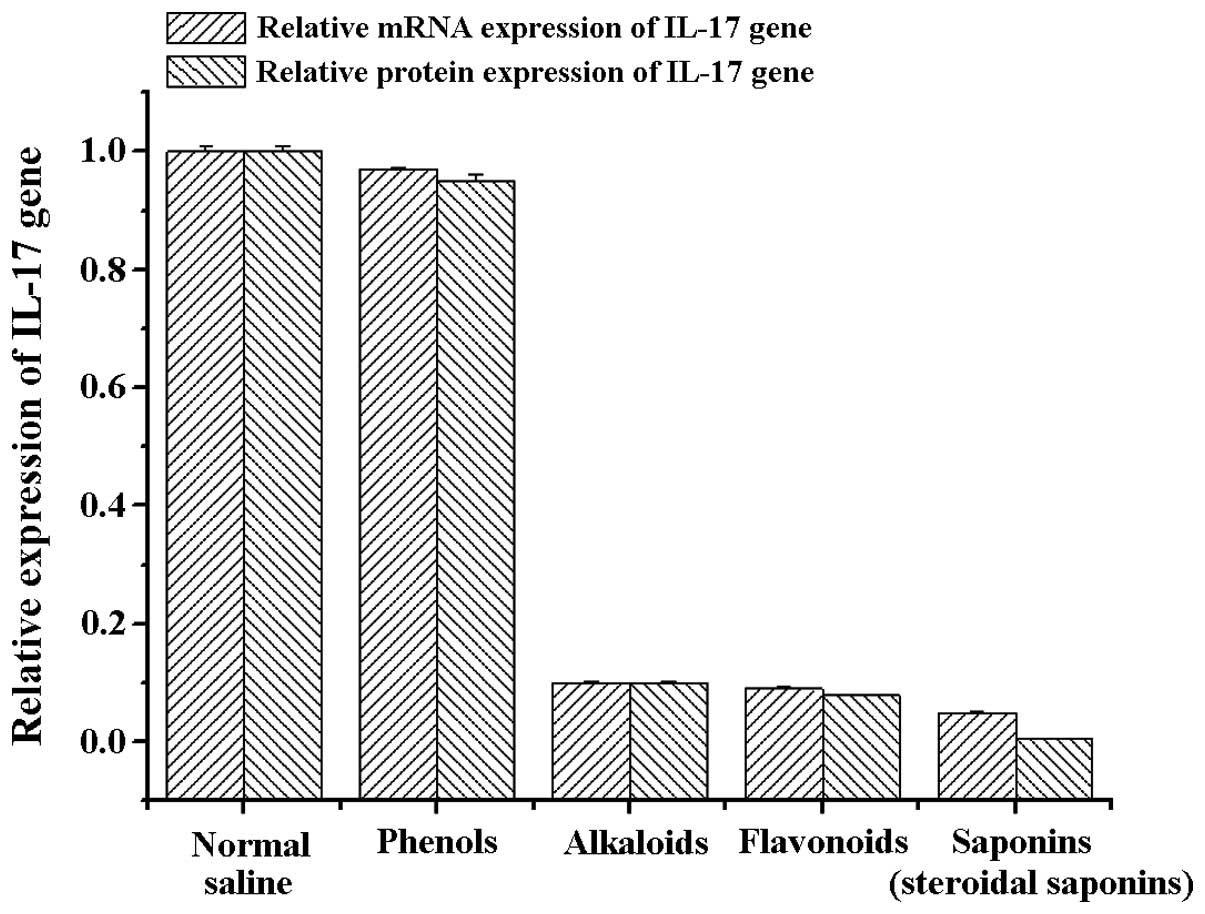
Previous studies have indicated that IL-17
expression is closely associated with the pathogenesis and
deterioration of EAU, and IL-17 can be used as a marker to identify
the severity of uveitis, rheumatoid arthritis, multiple sclerosis,
systemic lupus erythematosus and asthma (10). Therefore, in the present study, we
extracted RNA from the retinal tissues of rats in the different
treatment groups and detected the mRNA and protein expression of
IL-17. Detection results showed that the amount of mRNA and protein
expression of IL-17 genes in the alkaloids (berberine
hydrochloride) group, saponins (steroidal saponins) group and
flavonoids (baicalein) group was significantly lower than the
normal saline and phenols (chlorogenic acid) groups (Fig. 4). These results were further
supported by western blot analysis and ELISA with anti-IL-17
antibodies, indicating that the the three natural compounds may
effectively reduce the amount of IL-17 protein expression (Figs. 5 and 6).
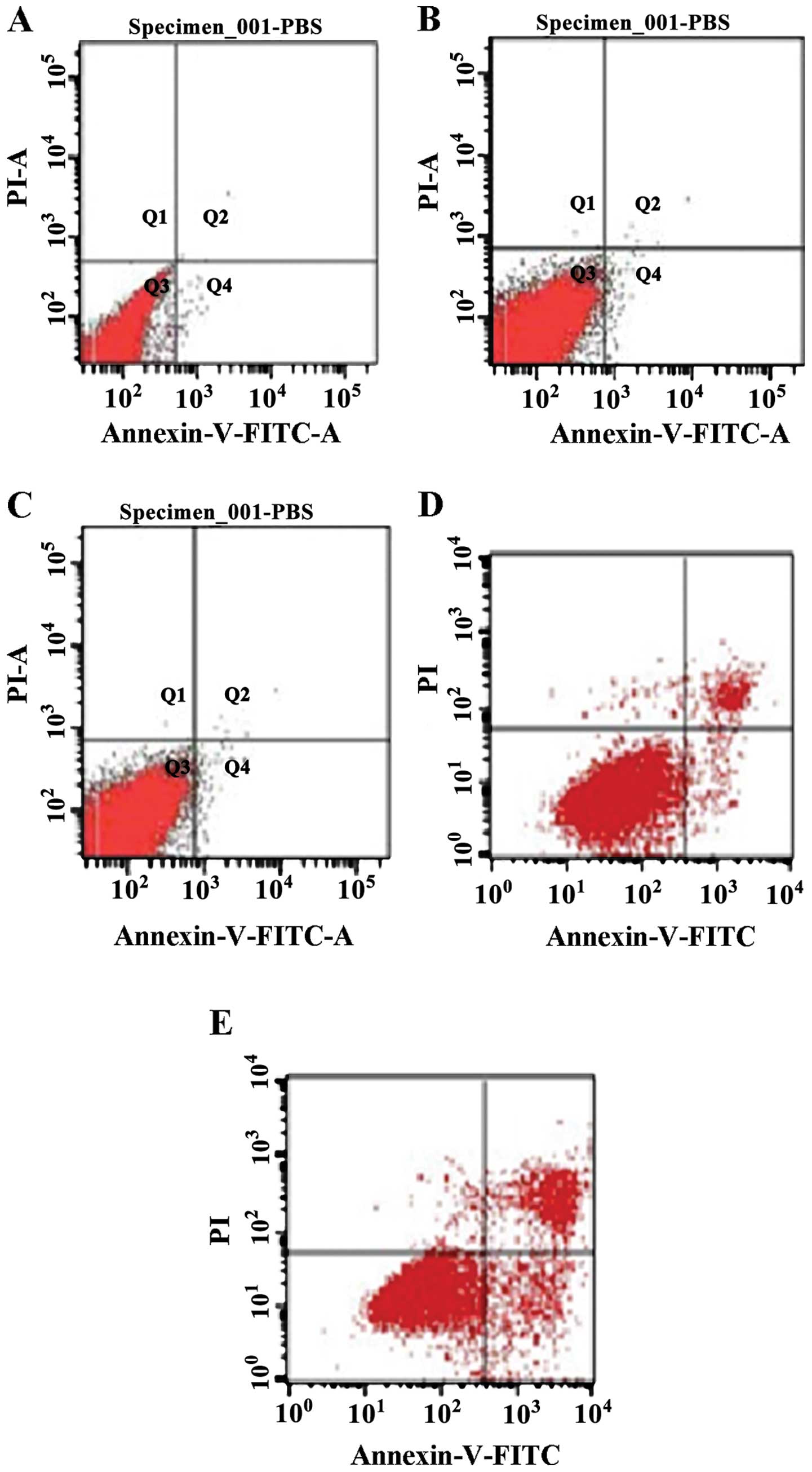
Detection of cell apoptosis
A search was conducted for apoptosis markers on the
ocular cells of rats in the different treatment groups using flow
cytometry. The results showed that cell apoptosis in the alkaloids,
saponins, and flavonoids groups was significantly higher than that
in the control group (normal saline group) (Table I), indicating that these natural
compounds can promote apoptosis of relevant inflammatory cells,
thereby exerting a beneficial effect for the rehabilitation of
inflammation (Fig. 7).
 | Table ICell apoptosis of different treatment
groups (mean ± standard deviation, n=12). |
Table I
Cell apoptosis of different treatment
groups (mean ± standard deviation, n=12).
| Group | Cell apoptotic rate
(%) |
|---|
| Normal saline | 2.13±0.017 |
| Phenols (chlorogenic
acid) | 54.8±0.042a |
| Alkaloids (berberine
hydrochloride) | 65.7±0.012a |
| Flavonoids
(baicalein) | 74.2±0.068a |
| Saponins (steroidal
saponins) | 83.4±0.037b |
Discussion
Uveitis is an important eye disease that may cause
blindness. It has been extensively investigated, however, its exact
pathogenesis remains to be determined (10), resulting in a lack of specifically
efficient drugs for its treatment (11,12).
Arsenic trioxide has been shown to be effective in the treatment of
acute promyelocytic leukemia and other tumors, and more recently
investigators tested it against EAU, via intraperitoneal injection,
with promising results, albeit a high incidence of adverse effects
(13). Other investigators have
employed cyclophosphamide because of its inhibitory effects on CD
lymphocytes (supressing humoral immunity and IL-17 expression)
(14). However, the results have
shown that although cyclophosphamide suppresses humoral immunity
and reduces the gene expression of peripheral IL-17, its effects on
EAU are only mildy beneficial (15). At least one group of investigators
has used traditional drugs that seem to act more directly on the
eye, improving the clinical syndromes associated with the ocular
inflammation caused by uvetis, albeit those treatments do not
provide successful treatment for the disease (16).
Many natural compounds can be effective for the
treatment of certain anaphylactic diseases (17). Compared with conventional drugs,
the negative effects of natural compounds are less prominent. In a
previous study, volatile oil of Angelica, a natural compound,
effectively treated passive cutaneous anaphylaxis and parietal
periosteum hypertrophy in rats (18). A different study confirmed that the
volatile oil of schizonepeta indirectly inhibited inflammation by
suppressing the metabolic products of arachidonic acid epoxidase
and lipoxidase (19). Based on the
abovementioned results, the present study was carried out to
examine the curative effects of natural compounds on uveitis
(20,21). The results show that natural
compounds were more effective for EAU treatment than normal saline.
However, the curative effects of the different natural compounds
tested varied. Of the four types of natural compounds, the curative
effects of saponins were the most promising, with alkaloids and
flavonoids providing the second best results, while phenols were
basically ineffective. Such differences are probably associated
with the different compositions of the tested compounds. Terpenoids
are the major component of saponins. Recent findings have shown the
effectiveness of certain terpenoids for dealing with inflammation
(18). Additionally, the major
components of phenols are alkaline substances, which probably cause
many toxic effects on tissues. The results of the present study
require further investigation with regard to the use of saponins
and terpenoids for the treatment of uveitis.
References
|
1
|
Witkowski L, Cywinska A, Paschalis-Trela
K, Crisman M and Kita J: Multiple etiologies of equine recurrent
uveitis - A natural model for human autoimmune uveitis: A brief
review. Comp Immunol Microbiol Infect Dis. 44:14–20. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Wakefield D and Abi-Hanna D: HLA antigens
and their significance in the pathogenesis of anterior uveitis: A
mini review. Curr Eye Res. 5:465–473. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Maca SM, Amirian A, Prause C, Gruber K,
Mejdoubi L and Barisani-Asenbauer T: Understanding the impact of
uveitis on health-related quality of life in adolescents. Acta
Ophthalmol. 91:e219–e224. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Berthet F, Le Deist F, Duliege AM,
Griscelli C and Fischer A: Clinical consequences and treatment of
primary immunodeficiency syndromes characterized by functional T
and B lymphocyte anomalies (combined immune deficiency).
Pediatrics. 93:265–270. 1994.PubMed/NCBI
|
|
5
|
Zhang Q, Lu J, Liang X and Zhi L: The
level and clinical significance of serum IL-17 in patients with
systemic lupus erythematosus. Xibao Yu Fenzi Mianyixue Zazhi.
28:301–303. 2012.In Chinese.
|
|
6
|
Cui Y, Wang G, Li Y, Wang Y, Wang X and Bi
H: Optical coherence tomography and histopathology of macular
uveitis. Optom Vis Sci. 91:1335–1342. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Oh-i K, Keino H, Goto H, Yamakawa N,
Murase K, Usui Y, Kezuka T, Sakai J, Takeuchi M and Usui M:
Intravitreal injection of Tacrolimus (FK506) suppresses ongoing
experimental autoimmune uveoretinitis in Rats. Br J Ophthalmol.
91:237–242. 2007. View Article : Google Scholar
|
|
8
|
Kohno H, Sakai T, Saito S, Okano K and
Kitahara K: Treatment of experimental autoimmune uveoretinitis with
atorvastatin and lovastatin. Exp Eye Res. 84:569–576. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kezic JM, Glant TT, Rosenbaum JT and
Rosenzweig HL: Neutralization of IL-17 ameliorates uveitis but
damages photo-receptors in a murine model of spondyloarthritis.
Arthritis Res Ther. 14:R182012. View
Article : Google Scholar
|
|
10
|
Chi W, Zhu X, Yang P, Liu X, Lin X, Zhou
H, Huang X and Kijlstra A: Upregulated IL-23 and IL-17 in Behçet
patients with active uveitis. Invest Ophthalmol Vis Sci.
49:3058–3064. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sng CC, Ang M and Barton K: Uveitis and
glaucoma: New insights in the pathogenesis and treatment. Prog
Brain Res. 221:243–269. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wells JM and Smith JR: Uveitis in juvenile
idiopathic arthritis: Recent therapeutic advances. Ophthalmic Res.
54:124–127. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zou ZP, Qiang FY, Zhang T, Sun L, Jia T,
Zhu XC and Xu H: Treatment of experimental autoimmune uveitis in
rats with arsenic trioxide. Chin Ophthalmic Res. 28:306–310.
2010.
|
|
14
|
Klímová A, Seidler Štanogová P,
Heissigerová J, Svozílková P and Kučera T: Mycophenolate mofetil
and cyclophosphamide treatments suppress inflammation intensity in
an experimental model of autoimmune uveitis. Folia Biol (Praha).
60:228–234. 2014.
|
|
15
|
Tang Y and Cai L: Research progress of
regulatory T cells and uveitis. Int J Ophthalmol. 11:450–452.
2011.
|
|
16
|
Li J, Chen X, Liu Z, Yue Q and Liu H:
Expression of Th17 cytokines in skin lesions of patients with
psoriasis. J Huazhong Univ Sci Technolog Med Sci. 27:330–332. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
de Smet MD, Taylor SRJ, Bodaghi B,
Miserocchi E, Murray PI, Pleyer U, Zierhut M, Barisani-Asenbauer T,
LeHoang P and Lightman S: Understanding uveitis: The impact of
research on visual outcomes. Prog Retin Eye Res. 30:452–470. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mira A, Tanaka A, Tateyama Y, Kondo R and
Shimizu K: Comparative biological study of roots, stems, leaves,
and seeds of Angelica shikokiana Makino. J Ethnopharmacol.
148:980–987. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Byun MW: Schizonepeta tenuifolia ethanol
extract exerts anti-inflammatory activity through the inhibition of
TLR4 signaling in lipopolysaccharide-stimulated macrophage cells. J
Med Food. 17:350–356. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jäger A and Kuchroo VK: Effector and
regulatory T-cell subsets in autoimmunity and tissue inflammation.
Scand J Immunol. 72:173–184. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tang K, Si J-K, Guo D-D, Cui Y, Du YX, Pan
XM and Bi HS: Ranibizumab alone or in combination with photodynamic
therapy vs photodynamic therapy for polypoidal choroidal
vasculopathy: A systematic review and Meta-analysis. Int J
Ophthalmol. 8:1056–1066. 2015.PubMed/NCBI
|