Cerebrovascular diseases occur following acute
cerebrovascular events whereby the arteries of the brain are
blocked or a brain blood vessel ruptures. Poor blood flow to the
brain subsequently results in cell death. There are three primary
types of cerebrovascular diseases: Ischemic stroke, hemorrhagic
stroke and transient ischemic attack (TIA). The high incidence of
cerebrovascular diseases worldwide is largely due to failed
management and prevention of modifiable risk factors, particularly
in ischemic stroke, which accounts for >85% of total
cerebrovascular diseases. Cerebrovascular diseases more commonly
affect people who are overweight, aged ≥55, have a unhealthy
lifestyle (limited exercise, heavy drinking, use of illicit drugs,
smoking or poor work/life balance), and who have a family history
of stroke, hypertension, moyamoya, vasculitis, arterial dissection
or venous occlusive disease (1–6).
Cerebrovascular disease is the leading cause of mortality and
chronic disability in China, and the third leading cause of
mortality and the leading cause of chronic disability in the USA
(7,8).
Notch signaling is a major intercellular
communication pathway, which is highly conserved in the majority of
multicellular organisms. Notch signaling is a crucial regulator of
numerous fundamental cellular processes, including proliferation,
stem cell maintenance and differentiation, during embryonic
development in vertebrate and invertebrate organisms (9–11).
In addition, Notch signaling is involved in cell differentiation,
proliferation, inflammation (12),
oxidative stress and apoptosis in a variety of cell types in adults
(10,13). The primary mechanisms underlying
the Notch signaling pathway in cerebrovascular disease have been
well-established by extensive investigation (10,14,15),
and include enhancing inflammation (16–18),
increasing oxidative stress (19),
promoting apoptosis (20) and
mediating adult subventricular zone neural progenitor cell
proliferation and differentiation following stroke (21). It has been demonstrated that
activation of the Notch signaling pathway exacerbates ischemic
brain damage, whereas inhibiting the Notch signaling pathway
decreases the infarct size and improves the functional outcome in a
mouse model of stroke (18,22).
The present review discusses the role of the Notch
signaling pathway in the pathogenesis of cerebrovascular diseases.
It primarily focuses on the association between Notch signaling and
neuroinflammation, oxidative stress and apoptosis in
cerebrovascular diseases. An overview is provided for the proposed
pathogenic mechanism underlying Notch signaling in stroke via
regulation of angiogenesis and the function of the blood–brain
barrier (BBB). Finally, the efficacy of regulating Notch signaling
as a novel therapeutic intervention for cerebrovascular diseases is
considered.
Extensive evidence has revealed that the Notch
signaling pathway is closely associated with the function and
structure of the nervous system. In the central nervous system
(CNS), the Notch signaling pathway regulates the normal development
of neural progenitor cells, neurons, oligodendrocytes and
astrocytes (35,36). Numerous diseases of the nervous
system are associated with Notch mutations, including sporadic
Alzheimer's disease (37,38), Down syndrome (39,40),
Pick's disease (38) and cerebral
autosomal dominant arteriopathy with subcortical infarcts and
leukoencephalopathy (CADASIL) (41–44).
The molecular and cellular mechanisms underlying the degeneration
of brain cells affected by cerebrovascular disease are complex,
involving bioenergetic failure, acidosis, excitotoxicity, oxidative
stress and inflammation, and resulting in necrotic or apoptotic
cell death (45,46). Various signaling pathways are
involved, including Notch. For example, in cerebral ischemia, the
activation of Notch regulates nerve damage repair, inflammation and
angiogenesis in the vascular ischemic area via regulating
proliferation and development of neuronal precursor cells,
mediating the release of inflammatory factors and promoting
angiogenesis (47–50). Studies in vitro and in
vivo have demonstrated that blood vessel angiogenesis,
endothelial cell proliferation, and artery and vein differentiation
are regulated by the Notch signaling pathway (51–53).
Enhancing Notch signaling activity promotes arteriogenesis via
vascular smooth muscle cell (VSMC) proliferation in the ischemic
brain following stroke (51,54,55).
Inflammation is a complex cascade that protects the
body from infection and injury. Similarly, neuroinflammation is a
response to neurological damage and may be divided into acute and
chronic process. A variety of inflammatory cytokines take part in
the neuroinflammation. Evidence indicates that acute
neuroinflammation is benefcial to damage repair in the nervous
system, whereas chronic neuroinflammation aggravates the
pathological events occurring in the brain (56–59).
In addition, neuroinflammation has been demonstrated to be crucial
for the pathogenesis of cerebrovascular diseases (56). Various studies have revealed that
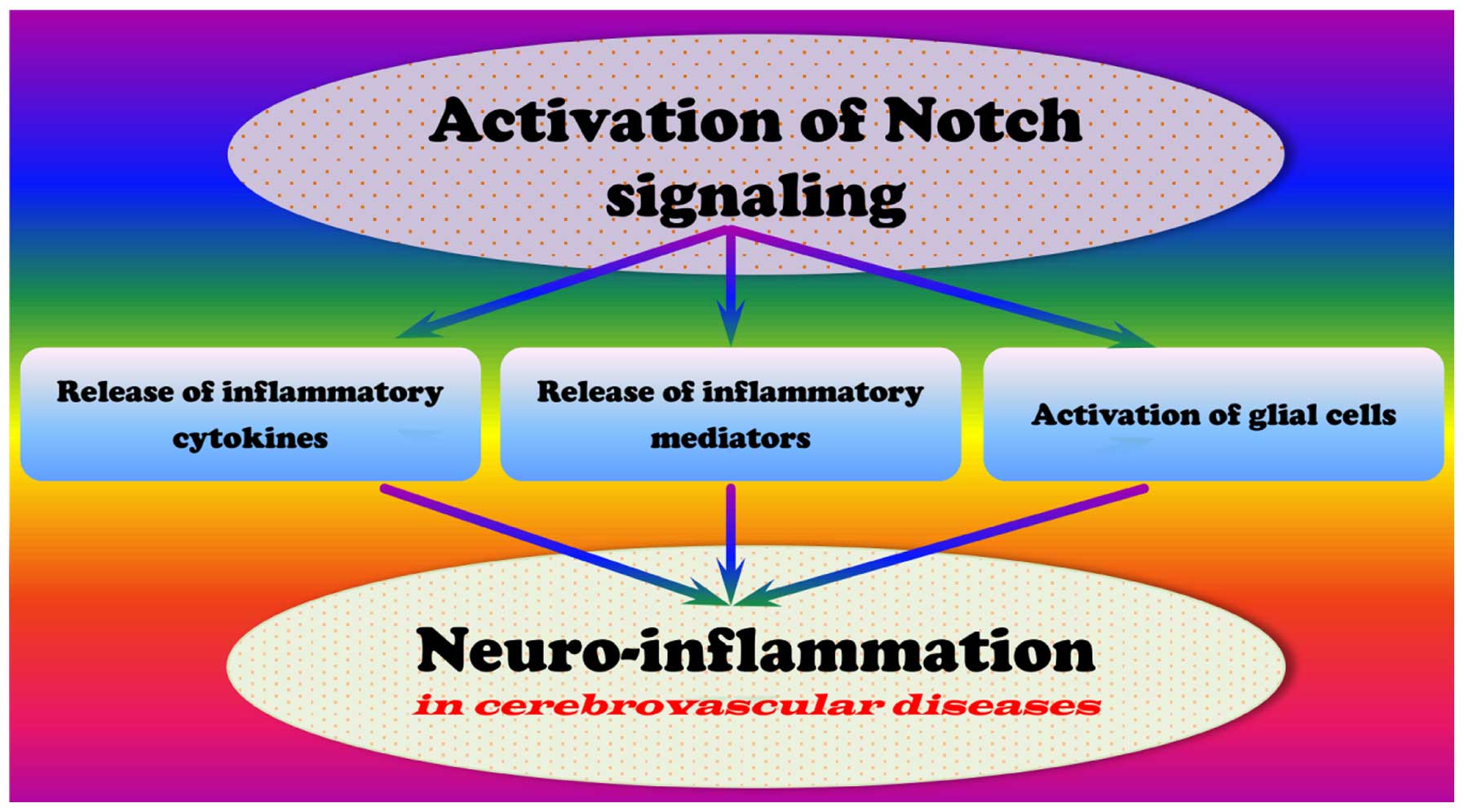
the activation of Notch signaling promotes the neuroinflammatory
response associated with cerebrovascular diseases (Fig. 1) (18,22,60).
Previous studies have demonstrated that cerebral
ischemia initiates an inflammatory response in the brain associated
with the release of a variety of inflammatory cytokines, including
tumor necrosis factor-α (TNF-α), interleukin (IL)-1β, and IL-6
(55,61,62).
Macrophages treated with Toll-like receptor (TLR) 3 or −4 agonists
increase their production of interferon (IFN)-β, TNF-α, IL-12 and
IL-23. Activation of glial cells and their release of neurotoxic
factors enhance inflammation in cerebrovascular disease. In
addition, activated glial cells increase the expression of
inflammatory cytokines in cerebral ischemia, including TNF-α, IL
-1β, IL-6, transforming growth factor β (TGF-β) and IL-8.
Notch signaling is evolutionarily conserved and
critical for the development and homeostasis of various tissues.
Activation of Notch signaling promotes macrophage polarization to
the IFN-γ-producing M1 (inflammatory) subtype (63). Inhibition of Notch signaling by
γ-secretase inhibitors (GSI) reduces nuclear factor-κB (NF-κB)
activity and suppresses inflammatory responses. Previous studies
have demonstrated that GSI significantly decreases peptidoglycan
and poly (I:C)-induced secretion of M1 (TNF-α, IL-6, IFN-γ and
IL-1α) and the anti-inflammatory subtype M2 (IL-10) cytokines
(63,64). Notch signaling is activated in
response to TLR ligands, thus amplifying the inflammatory response
by enhancing NF-κB signaling. Activation of Notch signaling has
been revealed to be involved in the sustained activation of NF-κB
and the resulting enhancement of inflammatory responses (65). It is becoming apparent that Notch
signaling is central to chronic inflammatory events involved in the
pathogenesis of cerebrovascular diseases, and Notch may therefore
provide a novel target for therapeutic strategies (15,16,18–20,22,63,65).
An ischemic stroke rat model induced by a 90-min occlusion of the
right middle cerebral artery demonstrated that inhibiting Notch
activation with
N-[N-(3,5-Difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl
ester (DAPT) limited NICD release, and production of IL-6 and IL-1β
in the ischemic penumbral cortex (18). Notch mutations may result in a
predisposition to stroke and cerebrovascular atherosclerosis, and
Notch mutations may also be involved in inflammation process, as
genes encoded by Notch mutations include the IL-1 receptor and
paraoxonase-1 (66).
Inflammatory mediators from plasma or cells, exert
their effects via binding to specific receptors on target cells.
Mediators may have one or numerous target cell types, and may even
have varying effects in distinct cell and tissue types. It has been
demonstrated that Notch signaling may reprogram mitochondrial
metabolism for proinflammatory macrophage activation, inducing the
release of inflammatory mediators (67). Nitric oxide (NO), which is produced
by cells that express NO synthase (NOS), is a prevalent
inflammatory mediator that may inhibit the activity of Notch1
signaling (68,69). A previous study indicates that
inducible NOS (iNOS) is directly involved in the generation of NO
and the inhibition of Notch1 signaling, and that NO inhibits the
binding of Notch1-IC and CSL protein transcriptional complexes to a
specific target sequence (69).
The dysfunction of Notch signaling pathway increases the
vulnerability of neurons and interacts with NF-κB to enhance the
inflammatory response following cerebral ischemia (70,71).
Numerous signaling pathways involved in neurodegenerative disorders
are activated in response to reactive oxygen species (ROS), which
induce apoptosis and increase NICD release and the expression of
hairy and enhancer of split-1 (HES-1) in cerebral ischemia
(71–73). The potential role of Notch
signaling in stroke via inflammatory mediators is summarized in
Table I.
Microglia are mononuclear phagocytes with various
functions in the CNS, with the stage and function of microglia
indicated by morphological characteristics. The phagocytic function
of microglia is critical for the removal of hematoma and other
debris; however, they additionally produce inflammatory mediators
(93). Microglia are typically
classified into three forms: Ameboid, ramified and activated.
Microglia, as the resident immune cells of the CNS, continually
sample the environment. Under normal conditions, they exist in a
ramified form and phagocytose debris (94). Previous studies indicate that Notch
signaling may regulate the different forms of microglia under
different conditions (71,95–97).
Notch signaling damages neurons by activating microglial cells and
stimulating the infiltration of proinflammatory leukocytes
(98). Following stroke, microglia
are activated, become amoeboid and release inflammatory cytokines
(M1 subtype). However, microglia may be differentially activated,
subsequently limiting inflammation and destroy tissue debris
through phagocytosis (M2 subtype) (63,99).
Microglia secrete various inflammatory molecules, including IL-1,
IL-6, IFN-γ and TNF-α (22).
Furthermore, Notch signaling may be involved in regulating
microglia activation following hypoxia, partially via the
TLR4/Myeloid differentiation primary response gene 88/TNF receptor
associated factor 6/NF-κB signaling pathway (71,100). A model of focal ischemic stroke
using mice transgenic for antisense Notch or wild-type mice treated
with GSI demonstrated that inhibiting Notch activation reduced
brain cell damage and improved functional outcome. This suggests
that Notch activation exacerbates brain damage and functional
outcome in ischemic stroke (98).
Therefore, Notch signaling may be a potential target for inhibition
of microglia activation implicated in brain damage (101).
Various studies have indicated that Notch activation
induces NF-κB-mediated expression of proinflammatory genes in
hypoxic astrocytes (102). Notch
signaling regulates the activation state of microglia, thus
contributing to the control of inflammatory reactions in the CNS
(18,96). Notch-1 signaling is activated in
hypoxic astrocytes, verified by increased NICD and HES-1,
regulating astrocytic proliferation and activation via the
suppression of the vascular endothelial growth factor (VEGF) or
NF-κB signaling pathways. Dysregulation of Notch may exert effects
following stroke via the activation of microglia and astrocytes
(63,72,87,103). NF-κB is crucial in promoting
ischemic brain damage following stroke. Activation of NF-κB induces
the expression of proinflammatory cytokines, the adhesion and
migration of leukocytes, thus increasing the inflammatory response
(102). The Notch1 signaling
pathway regulates the NF-κB signaling pathway via Jagged1 and
inhibitor of κB α (IκBα). The dysfunction of the Notch signaling
pathway occurs with NF-κB following cerebral ischemia via
activating microglia to produce inflammatory mediators (71,101,104). In addition, Notch activation
enhances postischemic inflammation by directly modulating the
microglial innate response (22,104). In rats with cerebral ischemia and
in activated BV-2 microglia, Notch signaling induces the migration
and morphological transformation of activated microglia (16). An ischemic rat model using
middle-cerebral-artery occlusion demonstrated that Notch-Jagged
signaling is involved in dysfunction of astrocyte-associated
capillary network (103).
Oxidative stress is broadly defined as a disturbance
in the balance between ROS production and antioxidant defenses
(105–107). In this state, abnormal levels of
ROS, including free radicals (hydroxyl, nitric acid and superoxide)
and non-radicals (hydrogen peroxide and lipid peroxide) result in
oxidative damage to cells or tissue (105,108–111). The oxidation state is the sum of
all redox processes producing ROS, reactive nitrogen species and
other reactive intermediates (106,108,112–114). ROS are crucial for physiological
processes, including apoptosis, regulation of neurotransmitters and
chemotaxis (114–116). ROS may destroy cell function and
promote injury to cellular lipids, nucleic acids and proteins, thus
inducing apoptosis. Oxidative stress is associated with the
pathological process of atherosclerosis, diabetes,
neurodegenerative disorders including Alzheimer's disease and
Parkinson's disease (117,118),
hypertension (119,120), cardiovascular diseases (121) and cerebrovascular diseases
(122,123). These diseases may promote the
production of ROS (105,107).
Oxidative stress is involved in the pathogenesis of
ischemic and hemorrhagic stroke (124–130) and appears to be a typical feature
in diverse models of cerebrovascular disease. Additionally,
oxidative stress may be involved in the pathogenesis of acute
ischemic stroke (131–136). Oxidative stress regulates
cerebral blood flow and controls permeability of the BBB (115,137). A high quantities of superoxide,
NO and peroxides are generated during cerebral
ischemia/reperfusion, and cellular macromolecules are destroyed by
oxygen radicals, resulting in apoptosis (138–142). Oxygen radicals activate matrix
metalloproteinases, resulting in the degradation of collagen and
laminin proteins in the basilar membrane, and destroying the
integrity of the vessel wall (143). In addition, ROS may induce cell
death through oxidative modification and fragmentation of DNA
mediated by nucleate endonuclease (144–146). Furthermore, oxidative stress
promotes transmigration of neutrophilic granulocytes from
peripheral blood to the CNS and the release of enzymes that degrade
the blood vessel basement membrane, resulting in increased
permeability of blood vessels (147–149). Oxidative stress may result in the
dysregulation of endothelial cell function, caused by
hyperglycemia, dyslipidemia and hyperinsulinemia, leading to
impaired vasoregulation, inflammation and altered BBB function
(150–152). The described pathological
processes result in cerebral parenchymal hemorrhage, vasogenic
brain edema and neutrophil infiltration, thus, aggravating cerebral
ischemic injury (142,153,154).
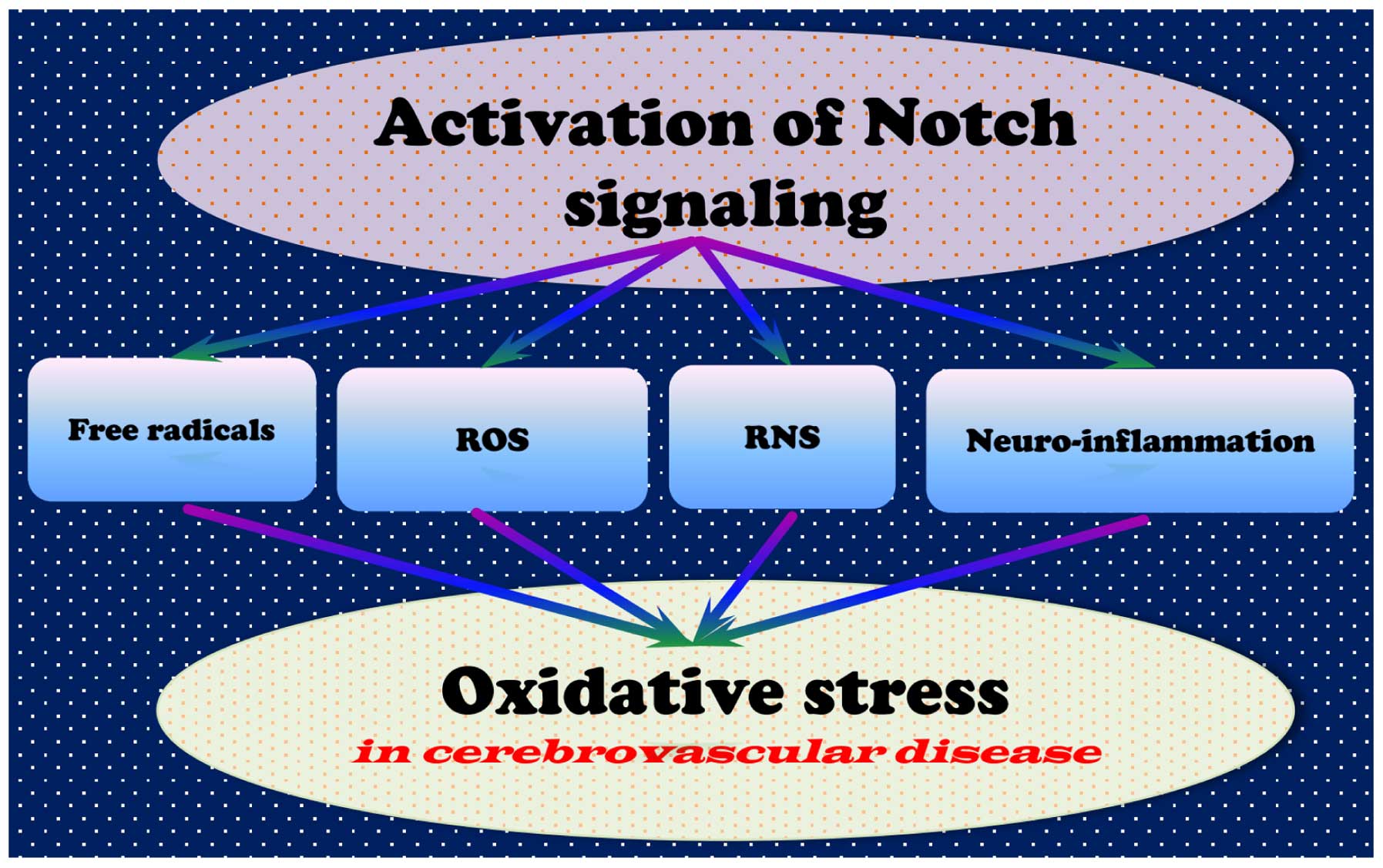
Studies have revealed that oxidative stress may
activate multiple signaling pathways associated with cell death;
the Notch signaling pathway is closely associated with oxidative
stress following cerebral ischemia, suggesting that dysregulation
of Notch signaling contributes to the occurrence of oxidative
stress (Fig. 2) (155–158). Notch activation results in cell
proliferation and metastasis, accompanied by a decrease in B-cell
lymphoma-2 (Bcl-2) associated protein X (Bax), Bcl-2
antagonist/killer, cytochrome c and caspase-3 and p53 expression
and an increase in Bcl-2 expression (159). It has been reported that
inhibiting Notch signaling abrogated cerebral ischemia/reperfusion
injury via inhibiting oxidative stress (68,160,161). Inhibiting the Notch signaling
pathway attenuates endothelial oxidative stress injury (158), suggesting that Notch inhibition
protects against cerebrovascular diseases via decreasing oxidative
stress-induced endothelial injury (158). A mutation in Notch3 has been
associated with mitochondrial disease, in which oxidative stress
caused by chronic hypoxia results in cerebral arteriopathy
(162).
Ischemia/reperfusion injury increases the oxidative
stress levels in tissue. The role of the Notch signaling pathway in
the oxidative stress-associated pathogenesis of cerebrovascular
diseases has been researched extensively (163). Further investigations to
elucidate the underlying molecular mechanisms of the Notch
signaling pathway in cerebrovascular disease may uncover potential
drug targets for the treatment of Notch-associated diseases.
However, decreasing the activity of Notch1 increases the production
of superoxide anion, iNOS, NO, nitrotyrosine and phosphatase and
tensin homolog deleted on chromosome 10 in mice subjected to
ischemia/reperfusion injury, whereas the phosphorylation levels of
NOS and protein kinase B (Akt) are decreased (68,163,164). As the inhibition or activation of
Notch signaling may be beneficial for the treatment of
cerebrovascular diseases, Notch signaling may exert distinct
functions under different conditions. Therefore, further studies
are required to elucidate the mechanisms underlying the role of
Notch signaling in cerebrovascular diseases.
Programmed cell death by apoptosis is crucial for
the development of multicellular organisms, and defects in
apop-tosis are associated with a wide variety of diseases (165). Inappropriate apoptosis results in
tissue atrophy, whereas a failure of apoptosis, as occurs in
cancer, leads to uncontrolled cell proliferation. Certain factors,
including Fas receptors and caspases, induce apoptosis, whereas
others, including certain Bcl-2 family members, suppress it
(166). Apoptosis is induced by
either the extrinsic or intrinsic pathways (167,168). Extrinsic stimuli include the
binding of ligands to cell surface death receptors, hormones,
TNF-α, growth factors, NO and cytokines (169–171). Intrinsic signals result from
cellular stress, including heat, radiation, nutrient deprivation
and viral infection. The expression of pro- and anti-apoptotic
proteins, the strength of the stimulus and the cell cycle stage all
alter the response of the cell to the extrinsic or intrinsic
trigger (172,173).
Notch is involved in various physiological
processes, via NICD translocation into the nucleus and binding to
target genes (189–191), including apoptosis (172). During apoptosis of tumor cells,
microRNA (miR)-100 was demonstrated to mediate Notch signaling
(192). A previous study
demonstrated that a Notch cis-regulatory element is responsive to
loss and gain of Drosophila p53 (Dp53) function and that
overexpression of Dp53 upregulates Notch mRNA and protein
expression levels (165).
Dp53-induced Notch activation and proliferation was revealed to
occur even when apoptosis was inhibited, and Dp53 may have a dual
role in regulating cell death and proliferation gene networks, to
control the balance between apoptosis and proliferation (165). In addition, Notch may be
important in the apoptosis- and drug-resistance of chronic
lymphocytic leukemia cells. Notch signaling has a cardioprotective
effect by regulating apoptosis via inhibiting Bcl-2 and the
activation of caspase-3 and -9. Furthermore, the Notch signaling
pathway mediates high-glucose-induced podocyte apoptosis via the
Bcl-2 and p53 pathways (193–195). It has been reported that miR-34c
overexpression increases the expression of anti-apoptotic Bcl-2,
and decreases the expression of pro-apoptotic Bax and cleaved
caspase-3 via targeting of Notch1 and Jaggged1 (193).
The Notch signaling pathway leads to apoptosis of
nerve cells and glia. Cell death in the brain following stroke is
the result of an alteration in the balance between pro- and
anti-apoptotic factors (196).
Neurons undergo apoptosis and necrosis. The Notch signaling pathway
is activated by various brain insults, including cerebrovascular
diseases (20,47,197), and is associated with the
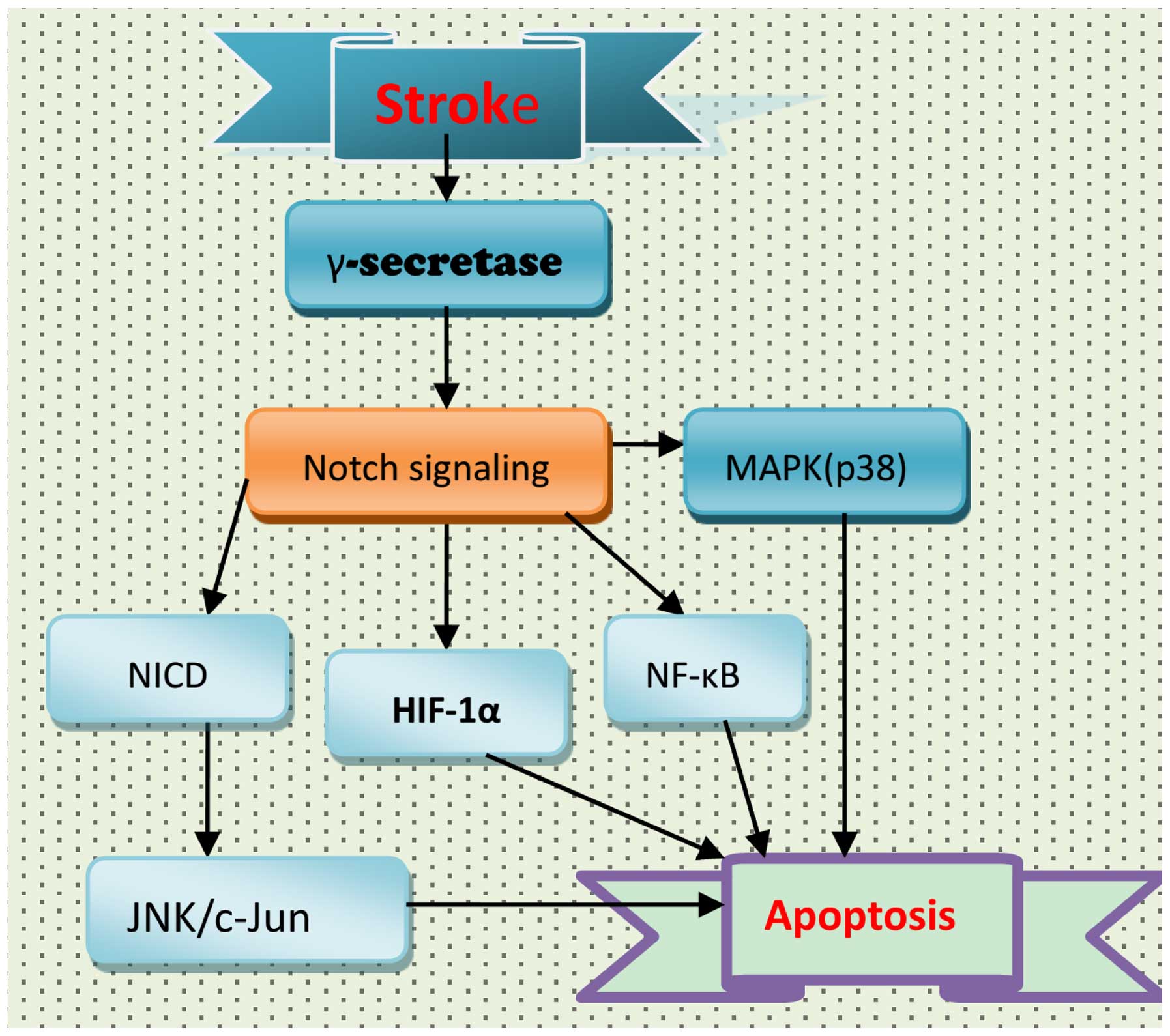
apoptosis involved in the pathogenesis of stroke (Fig. 3). Following stroke, activation of
the Notch signaling pathway may result in apoptosis of neurons via
NF-κB and hypoxia inducible factor-1α (HIF-1α) (20,197,198). In addition, Notch signaling may
affect mitogen-activated protein kinase (MAPK)-associated signaling
pathways. However, the role of Notch signaling in MAPK activation
following stroke remains to be fully elucidated. In wild-type and
NICD1-overexpressing HEK and SH-SY5Y cell lines, ischemic
conditions increased the expression levels of NICD1, JNK, p38-MAPK
and cleaved caspase-3; this increase in NICD1 and JNK was
attenuated by GSI (198). NICD
overexpression increased JNK expression levels, resulting in
enhanced cell death. Therefore, the Notch signaling pathway may
contribute to ischemic stroke via the JNK signaling pathway
(198), and the use of GSIs may
be a potential strategy for the treatment of ischemic stroke.
Neuronal cell apoptosis associated with Notch
signaling occurs in ischemic penumbra and ischemia/reperfusion
injury following ischemic cerebrovascular disease (85,199–201). Notch signaling may contribute to
apoptosis via the NF-κB, Bcl-like protein 11 and caspase pathways
(202). Calsenilin, the
expression of which is increased in the brain following
experimental ischemic stroke, was revealed to enhance the
γ-secretase-mediated cleavage of Notch and to contribute to
apoptosis (203). Peptidyl-prolyl
cis-trans isomerase NIMA-interacting 1 (Pin1) contributes to the
pathogenesis of ischemic stroke by promoting Notch signaling in
vitro and in a mouse stroke model, suggesting that Notch
signaling activation is involved in the pathogenesis of stroke, and
that inhibition of Pin1 may be a novel strategy for the treatment
of ischemic stroke (204).
However, Notch1 may inhibit neuronal apoptosis in cerebral
ischemia/reperfusion injury via increasing the phosphorylation of
Akt and promoting inactivation of Bcl-2-associated death promoter.
Notch1 may be neuroprotective in the immature brain against
ischemic injury, and future studies and clinical trials are
required to investigate the suitability of Notch1 inhibitors as a
treatment for perinatal ischemia. Inhibiting Notch2 was
demonstrated to alter the levels of apoptosis-regulating proteins
and slow the process of apoptosis in cerebral
ischemia/reperfusion-induced mice (199). Loss-of-function mutations in
Notch3 have been identified as the underlying cause of CADASIL
(205,206), in addition to complex regulation
of multiple pathways, including the Wnt/β-catenin signaling
pathway, TGF-β and Notch-induced apoptosis (207).
In summary, the role of Notch signaling in stroke
remains controversial. The majority of studies suggest that Notch
signaling activation is damaging following stroke, promoting
inflammation and apoptosis (20,83,98,202,206,208). However, certain studies have
indicated that enhancing Notch signaling may improve stroke
pathology (209–211). The effect of Notch on apoptosis
is summarized in Table II.
Therefore, further studies are required to fully elucidate the role
of Notch signaling in stroke.
Angiogenesis is a pathophysiological process of
vessel branching to form a new capillary network via vascular
endothelial cell proliferation and migration, and the sprouting and
division of blood vessels (233–236). The vasculature is primarily
comprised of vascular endothelial cells, VSMCs and extracellular
matrix, the structure and activity of which affect the morphology
and function of blood vessels. Angiogenesis is the result of the
interaction between endothelial cells, stromal cells and cytokines
mediated by a variety of positive and negative angiogenic
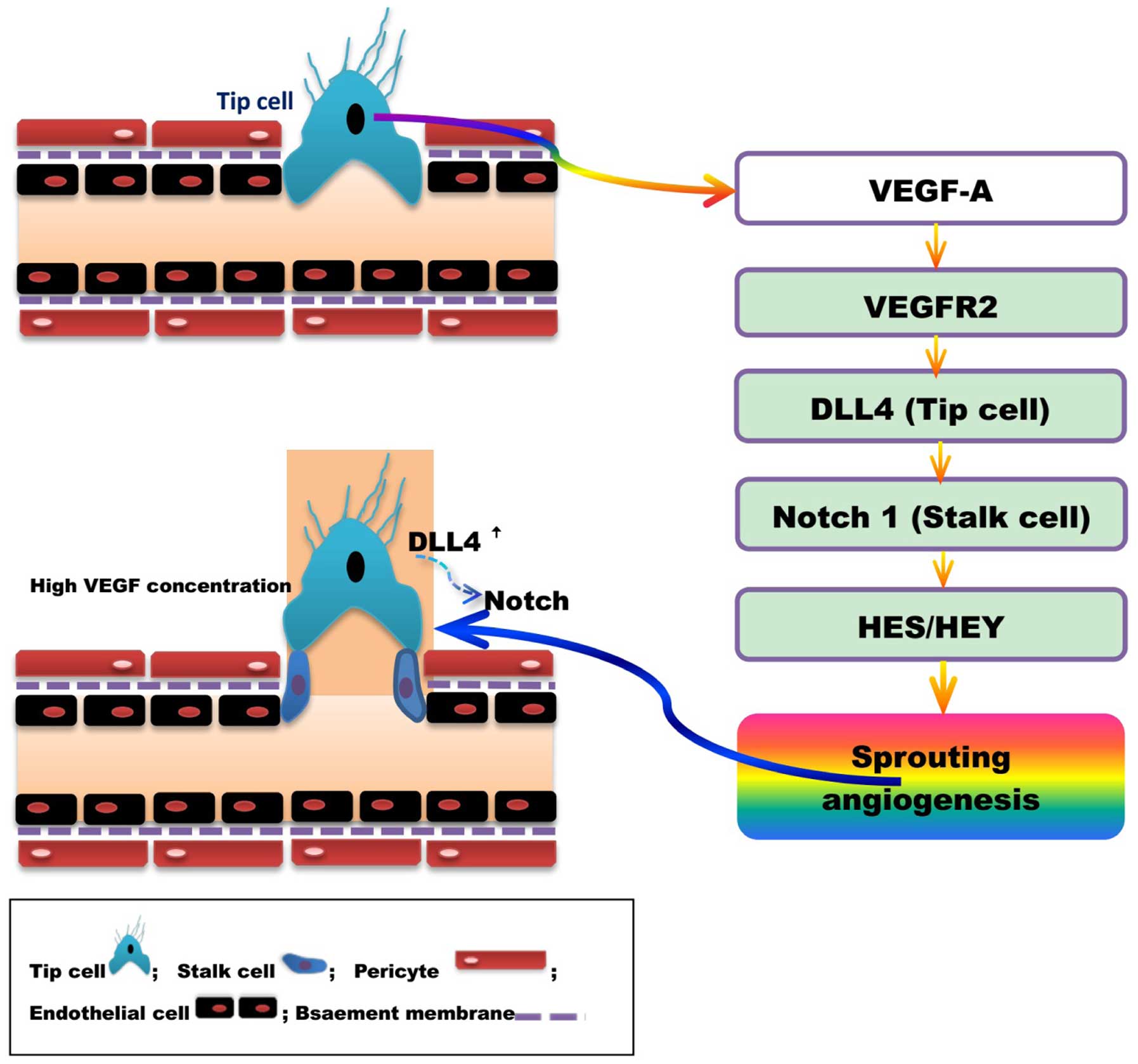
modulators. Studies have revealed that VEGF/VEGF receptor (VEGFR)
(237), Delta-like ligand 4
(DLL4)/Notch are the two primary pathways involved in the promotion
and coordination of angiogenesis (Fig.
4) (238,239).
Lumen formation is required to establish mature
blood vessels with complete structure and function. Vascular
endothelial cells are divided into acute (tip cell) and lotus cells
(trunk cell) depending on their location and characteristics, and
are involved in the formation of lumen. High concentrations of
VEGF-A induce endothelial cells to differentiate into tip cells.
Tip cells extend flopodia through the extracellular matrix, along
the VEGF-A gradient, providing direction to the new blood vessel
branch. The proliferation of trunk cells behind the tip cell
induces vascular sprouting, and the formation of the lumen and
extended vascular network. High levels of VEGF induce the synthesis
of DLL4 by tip cells, and thus increase Notchl expression in the
adjacent trunk cells. The activation of the DLL4/Notchl signaling
pathway promotes lumen formation (240,241). DLL4 expression in mouse tip cells
was reduced and angiogenesis attenuated following treatment with
VEGF antagonists or gene silencing (242,243). Studies have indicated that
DLL4/Notch regulate tip and trunk cell number and differentiation,
to control blood vessels sprouting and branching. Vascular
sprouting and branching proceeds following Notch inhibition,
however, these new blood vessels are dysfunctional (243,244).
Angiogenesis is a complex process regulated by
numerous factors. The most well-known of these regulators is VEGF,
which increases vascular permeability, promotes degradation of the
extracellular matrix and migration and proliferation of vascular
endothelial cells to induce angiogenesis. The expression of VEGF is
controlled by multiple factors, including fibroblast growth factor,
angiopoietins/Tie receptors, platelet-derived growth factor, TGF-β,
hepatocyte growth factor, HIF-1α, forkhead box (Fox) c1/Foxc2,
TNF-α, epidermal growth factor and matrix metalloproteinases
(Table III).
VEGF, a growth factor expressed in vascular
endothelial and other cells, acts directly on vascular endothelial
cells to promote mitosis, induce proliferation and migration,
maintain the integrity vessels and increase vascular permeability,
and is thus critical for angiogenesis. VEGF-A is the most
well-characterized of the VEGF family, and its receptor VEGFR2 is
the primary receptor involved in angiogenesis (237). The mammalian Notch signaling
pathway, comprised of four homologous Notch receptors (Notchl,
Notch2, Notch3 and Notch4) and five cognate ligands (DLL1, DLL3,
DLL4, Jaggedl and Jagged2) (254–256), is important for angiogenesis.
High concentrations of VEGF induce DLL4 expression, thus,
increasing Notchl expression on neighboring cells. The activation
of DLL4-Notchl signaling pathways promotes angiogenesis (47,257,258). Studies have revealed that
DLL4/Notch signaling mediates negative feedback; the expression of
DLL4 may suppress the proliferation and migration of endothelial
cells through the inhibition of VEGFR2 by HES-related protein 1
(259,260). VEGF, as a positive regulator of
angiogenesis, initiates and promotes angiogenesis, whereas Notch
signaling may negatively regulate the process to prevent
endothelial cell hyperplasia and, in conjunction with VEGF, promote
the formation of a well-differentiated vascular network (261–266).
Injection or nasal feeding of rats with human
recombinant VEGF following focal cerebral ischemia in the middle
cerebral artery promoted neovascularization of the ischemic area
and the recovery of neurological function (267,268). In addition, delayed treatment
with VEGF alleviates brain injury, enhances endothelial cell
proliferation and augments total vascular volume following neonatal
stroke (269). Furthermore, the
overexpression of VEGF in close proximity to intracere-bral
hemorrhage lesions in mice undergoing transplantation of F3 human
neural stem cells (NSCs) facilitated differentiation and survival
of the grafted human NSCs, and resulted in renewed angiogenesis in
the host brain and functional recovery of mice (270). Studies have revealed that
strategies to enhance angiogenesis following focal cerebral
ischemia may improve recovery from stroke (271–274). The VEGF/Notch signaling pathway
is the primary signaling pathway regulating angiogenesis following
cerebral ischemia (47,275,276). VEGF and Notch are upregulated in
brain tissue following cerebral ischemia, which may significantly
promote angiogenesis in the ischemic region (277–280). Therefore, regulating the Notch
signaling pathway may provide a potential strategy for the
treatment of cerebrovascular diseases (281).
The BBB is a highly selective permeable barrier
separating circulating blood from the brain extracellular fluid, to
regulate the CNS microenvironment. The BBB is formed of a complex
network of endothelial cells, astroglia, pericytes, perivascular
macrophages and a basal membrane. Under physiological conditions,
BBB integrity is primarily maintained by endothelial cells, through
tight junctions, and the basal lamina; however, the structural and
functional integrity of the BBB is markedly altered during CNS
disorders, including neoplasia, ischemia, trauma, inflammation and
bacterial and viral infections.
Cerebrovascular BBB dysfunction is closely
associated with stroke, including intracranial hemorrhage and brain
ischemia disorders. Endothelial cells are critical for numerous
neurovascular functions, including angiogenesis, BBB formation and
maintenance, vascular stability and removal of cellular toxins.
Cerebrovascular endothelial cells interact with pericytes to
maintain a stable cerebral circulation in the CNS. A number of
studies have revealed that endothelial cell dysfunction in the CNS
results in breakdown of the BBB and brain hypoperfusion, leading to
neurodegeneration. It has been reported that disruption of Smad4
signaling, the central intracellular mediator of TGF-β signaling
(14), in endothelial cells leads
to the pathogenesis of intracranial hemorrhage and BBB breakdown
(14,282), indicating that Smad4 maintains
cerebrovascular integrity and that TGF-β/Smad signaling is involved
in the pathogenesis of cerebrovascular dysfunction. Notch signaling
is also critical in controlling BBB integrity via regulating the
normal function of endothelial cells and pericytes. However, the
underlying mechanisms regulating cerebral endothelial cell
functions remain to be elucidated.
The Notch signaling pathway is involved in blood
vessel integrity and BBB stability and function in the mammalian
vasculature (75,283–285). In vitro studies have
correlated BBB endothelial dysfunction with decreased Notch4
expression (286). Upon
activation, the constitutively expressed endothelial cell membrane
protein Notch4 appears to become primarily involved in the
stability and growth of mature endothelium (287). Permanent ischemia leads to the
redistribution of claudin decomposition fragments, zona occludens 1
and occludin protein from the membrane to the cytoplasm in BBB.
Additionally, the GSI, DAPT protects against permanent
ischemia-induced BBB damage, potentially via the modulation of
Notch/NICD/calpastatin homeostasis pathway in vascular endothelial
cells.
Increasing evidence indicates that Notch signaling
is critical in the pathogenesis of stroke, exerting effects via the
following underlying mechanisms: Neuroinfammation, oxidative
stress, apoptosis, angiogenesis and BBB function. Thus, regulating
Notch signaling may be an effective strategy for the prevention and
treatment of cerebrovascular diseases.
Studies have demonstrated that the activation of
Notch signaling is harmful and contributes to the pathogenesis of
cerebrovascular diseases including stroke (20,98,202,204,288–290). Acute inhibition of Notch
signaling has been revealed to rescue cerebral hypoperfusion,
reduce apoptosis in penumbra, decrease brain infarct size, elicit
certain morphologic features, including neurogenesis and
angiogenesis, associated with brain repair and functional recovery,
and enhance vascular densities in penumbra in the neonatal rat
brain following stroke (288).
However, activation of the Notch signaling pathway
may have a neuroprotective role via enhancing endogenous
neuroregeneration and brain arteriogenesis following stroke
(51,291). In a murine transient global
cerebral ischemia/reperfusion model, the neuroprotective effects of
preconditioning were mediated via the Notch signaling pathway, and
the expression of Notch1, NICD and HES-1 was upregulated (209). Notch signaling is widely accepted
to be a fundamental pathway controlling cell fate acquisition
through the regulation of adult neurogenesis. Studies have
demonstrated that Notch signaling is crucial for the maintenance,
proliferation and differentiation of NSCs in the developing brain
(292,293). Notch signaling induces the
neuronal expansion and differentiation following stroke (21). Increasing the expression level of
Notch signaling components may facilitate intrastriatal
transplantation therapy for ischemic stroke by promoting endogenous
regeneration in the hippocampus (294). Promoting Notch signaling activity
may facilitate increased arteriogenesis in a middle cerebral artery
occlusion stroke rat model (54).
In addition, Notch-induced rat and human bone marrow stromal cell
grafts inhibited ischemic cell loss and abrogated behavioral
deficits in chronic middle cerebral artery occlusion stroke rats
(295).
Therefore, the results on the effect of Notch
signaling on the pathogenesis of cerebrovascular diseases are
contradictory. Notch signaling may be damaging, as it promotes
inflammation, oxidative stress and apoptosis. However, the
activation of the Notch signaling pathway may exert neuroprotective
effects via enhancing endogenous neuroregeneration and brain
arteriogenesis following stroke. What is the exact role of Notch
signaling? Clarifying this question has potentially important
implications for the treatment of cerebrovascular disease, and will
provide novel strategies for future studies.
The present study was supported by grants from the
Natural Science Foundation of Hubei Province (grant no.
2015CFB260), the Hubei Province Health and Family Planning
Scientific Research Project (grant no. WJ2015MB219) the Shiyan
Natural Science Foundation (grant no. 15K70) and the Renmin
Hospital Natural Science Foundation (grant no. 2015CZY), to Dr
Zhiyou Cai.
|
1
|
Lama S, Dolati P and Sutherland GR:
Controversy in the management of lenticulostriate artery dissecting
aneurysm: A case report and review of the literature. World
Neurosurg. 81:441.e1–e7. 2014. View Article : Google Scholar
|
|
2
|
Dezmalj-Grbelja L, Bosnjak J,
Lovrencić-Huzjan A, Ivica M and Demarin V: Moyamoya disease in a
patient with brain tumor: Case report. Acta Clin Croat. 49:459–463.
2010.
|
|
3
|
Sharfstein SR, Ahmed S, Islam MQ, Najjar
MI and Ratushny V: Case of moyamoya disease in a patient with
advanced acquired immunodeficiency syndrome. J Stroke Cerebrovasc
Dis. 16:268–272. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Squizzato A, Gerdes VE, Brandjes DP,
Büller HR and Stam J: Thyroid diseases and cerebrovascular disease.
Stroke. 36:2302–2310. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vetrano DL, Landi F, De Buyser SL, Carfi
A, Zuccalà G, Petrovic M, Volpato S, Cherubini A, Corsonello A,
Bernabei R and Onder G: Predictors of length of hospital stay among
older adults admitted to acute care wards: A multicentre
observational study. Eur J Intern Med. 25:56–62. 2014. View Article : Google Scholar
|
|
6
|
Cicconetti P, Riolo N, Priami C, Tafaro L
and Ettore E: Risk factors for cognitive impairment. Recenti Prog
Med. 95:535–545. 2004.In Italian. PubMed/NCBI
|
|
7
|
Elkind MS: Epidemiology and risk factors.
Continuum (Minneap Minn). 17:1213–1232. 2011.
|
|
8
|
Jia Q, Liu LP and Wang YJ: Stroke in
China. Clin Exp Pharmacol Physiol. 37:259–264. 2010. View Article : Google Scholar
|
|
9
|
Bhoopathi P, Chetty C, Dontula R, Gujrati
M, Dinh DH, Rao JS and Lakka SS: SPARC stimulates neuronal
differentiation of medulloblastoma cells via the Notch1/STAT3
pathway. Cancer Res. 71:4908–4919. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yuan TM and Yu HM: Notch signaling: Key
role in intrauterine infection/inflammation, embryonic development,
and white matter damage? J Neurosci Res. 88:461–468. 2010.
|
|
11
|
Veenendaal LM, Kranenburg O, Smakman N,
Klomp A, Borel Rinkes IH and van Diest PJ: Differential Notch and
TGFbeta signaling in primary colorectal tumors and their
corresponding metastases. Cell Oncol. 30:1–11. 2008.PubMed/NCBI
|
|
12
|
Givogri MI, de Planell M, Galbiati F,
Superchi D, Gritti A, Vescovi A, de Vellis J and Bongarzone ER:
Notch signaling in astrocytes and neuroblasts of the adult
subventricular zone in health and after cortical injury. Dev
Neurosci. 28:81–91. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Quillard T and Charreau B: Impact of notch
signaling on inflammatory responses in cardiovascular disorders.
Int J Mol Sci. 14:6863–6888. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li F, Lan Y, Wang Y, Wang J, Yang G, Meng
F, Han H, Meng A and Yang X: Endothelial Smad4 maintains
cerebrovascular integrity by activating N-cadherin through
cooperation with Notch. Dev Cell. 20:291–302. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Dichgans M: Genetics of ischaemic stroke.
Lancet Neurol. 6:149–161. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yuan Y, Rangarajan P, Kan EM, Wu Y, Wu C
and Ling EA: Scutellarin regulates the Notch pathway and affects
the migration and morphological transformation of activated
microglia in experimentally induced cerebral ischemia in rats and
in activated BV-2 microglia. J Neuroinflammation. 12:112015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cheng YL, Choi Y, Sobey CG, Arumugam TV
and Jo DG: Emerging roles of the γ-secretase-notch axis in
inflammation. Pharmacol Ther. 147:80–90. 2015. View Article : Google Scholar
|
|
18
|
Wang Z, Huang W and Zuo Z: Perioperative
aspirin improves neurological outcome after focal brain ischemia
possibly via inhibition of Notch 1 in rat. J Neuroinflammation.
11:562014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li S, Zyang X, Wang Y, Ji H, Du Y and Liu
H: DAPT protects brain against cerebral ischemia by down-regulating
the expression of Notch 1 and nuclear factor κB in rats. Neurol
Sci. 33:1257–1264. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheng YL, Park JS, Manzanero S, Choi Y,
Baik SH, Okun E, Gelderblom M, Fann DY, Magnus T, Launikonis BS, et
al: Evidence that collaboration between HIF-1α and Notch-1 promotes
neuronal cell death in ischemic stroke. Neurobiol Dis. 62:286–295.
2014. View Article : Google Scholar
|
|
21
|
Wang L, Chopp M, Zhang RL, Zhang L,
Letourneau Y, Feng YF, Jiang A, Morris DC and Zhang ZG: The Notch
pathway mediates expansion of a progenitor pool and neuronal
differentiation in adult neural progenitor cells after stroke.
Neuroscience. 158:1356–1363. 2009. View Article : Google Scholar :
|
|
22
|
Wei Z, Chigurupati S, Arumugam TV, Jo DG,
Li H and Chan SL: Notch activation enhances the microglia-mediated
inflammatory response associated with focal cerebral ischemia.
Stroke. 42:2589–2594. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Morgan TH: The theory of the gene. Am
Naturalist. 51:513–544. 1917. View
Article : Google Scholar
|
|
24
|
Becker S, Oelschlaeger TA, Wullaert A,
Vlantis K, Pasparakis M, Wehkamp J, Stange EF and Gersemann M:
Bacteria regulate intestinal epithelial cell differentiation
factors both in vitro and in vivo. PLoS One. 8:e556202013.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maier D, Kurth P, Schulz A, Russell A,
Yuan Z, Gruber K, Kovall RA and Preiss A: Structural and functional
analysis of the repressor complex in the Notch signaling pathway of
Drosophila melanogaster. Mol Biol Cell. 22:3242–3252. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Braune EB and Lendahl U: Notch-a
goldilocks signaling pathway in disease and cancer therapy. Discov
Med. 21:189–196. 2016.PubMed/NCBI
|
|
27
|
Del Bianco C, Vedenko A, Choi SH, Berger
MF, Shokri L, Bulyk ML and Blacklow SC: Notch and MAML-1
complexation do not detectably alter the DNA binding specificity of
the transcription factor CSL. PLoS One. 5:e150342010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Faux CH, Turnley AM, Epa R, Cappai R and
Bartlett PF: Interactions between fbroblast growth factors and
Notch regulate neuronal differentiation. J Neurosci. 21:5587–5596.
2001.PubMed/NCBI
|
|
29
|
Shimizu K, Chiba S, Kumano K, Hosoya N,
Takahashi T, Kanda Y, Hamada Y, Yazaki Y and Hirai H: Mouse jagged1
physically interacts with notch2 and other notch receptors.
Assessment by quantitative methods. J Biol Chem. 274:32961–32969.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang S, Chung WC, Wu G, Egan SE and Xu K:
Tumor-suppressive activity of Lunatic Fringe in prostate through
differential modulation of Notch receptor activation. Neoplasia.
16:158–167. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bresnick EH, Chu J, Christensen HM, Lin B
and Norton J: Linking Notch signaling, chromatin remodeling, and
T-cell leukemogenesis. J Cell Biochem Suppl. 35(Suppl): S46–S53.
2000. View Article : Google Scholar
|
|
32
|
Nam Y, Weng AP, Aster JC and Blacklow SC:
Structural requirements for assembly of the CSL. Intracellular
Notch1. Mastermind-like 1 transcriptional activation complex. J
Biol Chem. 278:21232–21239. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Portin P: General outlines of the
molecular genetics of the Notch signalling pathway in Drosophila
melanogaster. A review Hereditas. 136:89–96. 2002. View Article : Google Scholar
|
|
34
|
Li Y and Baker NE: Proneural enhancement
by Notch overcomes Suppressor-of-Hairless repressor function in the
developing Drosophila eye. Curr Biol. 11:330–338. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang J, Ye Z, Zheng S, Chen L, Wan Y, Deng
Y and Yang R: Lingo-1 shRNA and Notch signaling inhibitor DAPT
promote differentiation of neural stem/progenitor cells into.
neurons Brain Res. 1634:34–44. 2016. View Article : Google Scholar
|
|
36
|
Cardano M, Diaferia GR, Cattaneo M, Dessí
SS, Long Q, Conti L, Deblasio P, Cattaneo E and Biunno I: mSEL-1L
(Suppressor/enhancer Lin12-like) protein levels influence murine
neural stem cell self-renewal and lineage commitment. J Biol Chem.
286:18708–18719. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Berezovska O, Xia MQ and Hyman BT: Notch
is expressed in adult brain, is coexpressed with presenilin-1, and
is altered in Alzheimer disease. J Neuropathol Exp Neurol.
57:738–745. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nagarsheth MH, Viehman A, Lippa SM and
Lippa CF: Notch-1 immunoexpression is increased in Alzheimer's and
Pick's disease. J Neurol Sci. 244:111–116. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Cairney CJ, Sanguinetti G, Ranghini E,
Chantry AD, Nostro MC, Bhattacharyya A, Svendsen CN, Keith WN and
Bellantuono I: A systems biology approach to Down syndrome:
Identification of Notch/Wnt dysregulation in a model of stem cells
aging. Biochim Biophys Acta. 1792:353–363. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fernandez-Martinez J, Vela EM,
Tora-Ponsioen M, Ocaña OH, Nieto MA and Galceran J: Attenuation of
Notch signalling by the Down-syndrome-associated kinase DYRK1A. J
Cell Sci. 122:1574–1583. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
García-Estévez DA, Barros-Angueira F and
Navarro C: CADASIL: Brief report on a family with a new p.G296C
mutation in exon 6 of the Notch-3 gene. Rev Neurol. 51:729–732.
2010.In Spanish.
|
|
42
|
Tang SC, Jeng JS, Lee MJ and Yip PK: Notch
signaling and CADASIL. Acta Neurol Taiwan. 18:81–90.
2009.PubMed/NCBI
|
|
43
|
Louvi A, Arboleda-Velasquez JF and
Artavanis-Tsakonas S: CADASIL: A critical look at a Notch disease.
Dev Neurosci. 28:5–12. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tan ZX, Li FF, Qu YY, Liu J, Liu GR, Zhou
J, Zhu YL and Liu SL: Identification of a known mutation in Notch 3
in familiar CADASIL in China. PLoS One. 7:e365902012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Posada-Duque RA, Barreto GE and
Cardona-Gomez GP: Protection after stroke: Cellular effectors of
neurovascular unit integrity. Front Cell Neurosci. 8:2312014.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cotena S, Piazza O and Tufano R: The use
of erythtropoietin in cerebral diseases. Panminerva Med.
50:185–192. 2008.PubMed/NCBI
|
|
47
|
Lou YL, Guo F, Liu F, Gao FL, Zhang PQ,
Niu X, Guo SC, Yin JH, Wang Y and Deng ZF: miR-210 activates notch
signaling pathway in angiogenesis induced by cerebral ischemia. Mol
Cell Biochem. 370:45–51. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Corada M, Morini MF and Dejana E:
Signaling pathways in the specifcation of arteries and veins.
Arterioscler Thromb Vasc Biol. 34:2372–2377. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Grieskamp T, Rudat C, Lüdtke TH, Norden J
and Kispert A: Notch signaling regulates smooth muscle
differentiation of epicardium-derived cells. Circ Res. 108:813–823.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
del Monte G, Casanova JC, Guadix JA,
MacGrogan D, Burch JB, Pérez-Pomares JM and de la Pompa JL:
Differential Notch signaling in the epicardium is required for
cardiac inflow development and coronary vessel morphogenesis. Circ
Res. 108:824–836. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Proweller A, Wright AC, Horng D, Cheng L,
Lu MM, Lepore JJ, Pear WS and Parmacek MS: Notch signaling in
vascular smooth muscle cells is required to pattern the cerebral
vasculature. Proc Natl Acad Sci USA. 104:16275–16280. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Koga J, Nakano T, Dahlman JE, Figueiredo
JL, Zhang H, Decano J, Khan OF, Niida T, Iwata H, Aster JC, et al:
Macrophage Notch Ligand Delta-Like 4 Promotes Vein Graft Lesion
Development: Implications for the Treatment of Vein Graft Failure.
Arterioscler Thromb Vasc Biol. 35:2343–2353. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Quillien A, Moore JC, Shin M, Siekmann AF,
Smith T, Pan L, Moens CB, Parsons MJ and Lawson ND: Distinct Notch
signaling outputs pattern the developing arterial system.
Development. 141:1544–1552. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zacharek A, Chen J, Cui X, Yang Y and
Chopp M: Simvastatin increases notch signaling activity and
promotes arteriogenesis after stroke. Stroke. 40:254–260. 2009.
View Article : Google Scholar
|
|
55
|
Chen J, Cui X, Zacharek A, Ding GL,
Shehadah A, Jiang Q, Lu M and Chopp M: Niaspan treatment increases
tumor necrosis factor-alpha-converting enzyme and promotes
arteriogenesis after stroke. J Cereb Blood Flow Metab. 29:911–920.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Di Napoli M and Shah IM: Neuroinflammation
and cerebrovascular disease in old age: A translational medicine
perspective. J Aging Res. 2011:8574842011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Felsky D, De Jager PL, Schneider JA,
Arfanakis K, Fleischman DA, Arvanitakis Z, Honer WG, Pouget JG,
Mizrahi R, Pollock BG, et al: Cerebrovascular and microglial states
are not altered by functional neuroinflammatory gene variant. J
Cereb Blood Flow Metab. 36:819–830. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Cacabelos R, Torrellas C, Fernández-Novoa
L and Aliev G: Neuroimmune Crosstalk in CNS Disorders: The
Histamine Connection. Curr Pharm Des. 22:819–848. 2016. View Article : Google Scholar
|
|
59
|
Silva J, Polesskaya O, Knight W, Zheng JT,
Granger M, Lopez T, Ontiveros F, Feng C, Yan C, Kasischke KA and
Dewhurst S: Transient hypercapnia reveals an underlying
cerebrovascular pathology in a murine model for HIV-1 associated
neuroinflammation: Role of NO-cGMP signaling and normalization by
inhibition of cyclic nucleotide phosphodiesterase-5. J
Neuroinflammation. 9:2532012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Meschia JF and Worrall BB: New advances in
identifying genetic anomalies in stroke-prone probands. Curr
Atheroscler Rep. 5:317–323. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Heo R, Park JS, Jang HJ, Kim SH, Shin JM,
Suh YD, Jeong JH, Jo DG and Park JH: Hyaluronan nanoparticles
bearing γ-secretase inhibitor: In vivo therapeutic effects on
rheumatoid arthritis. J Control Release. 192:295–300. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Lucitti JL, Mackey JK, Morrison JC, Haigh
JJ, Adams RH and Faber JE: Formation of the collateral circulation
is regulated by vascular endothelial growth factor-A and a
disintegrin and metal-loprotease family members 10 and 17. Circ
Res. 111:1539–1550. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Brifault C, Gras M, Liot D, May V, Vaudry
D and Wurtz O: Delayed pituitary adenylate cyclase-activating
polypeptide delivery after brain stroke improves functional
recovery by inducing m2 microglia/macrophage polarization. Stroke.
46:520–528. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Holden JA, Attard TJ, Laughton KM, Mansell
A, O'Brien-Simpson NM and Reynolds EC: Porphyromonas gingivalis
lipopolysaccharide weakly activates M1 and M2 polarized mouse
macrophages but induces inflammatory cytokines. Infect Immun.
82:4190–4203. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Zhang Y, He K, Wang F, Li X and Liu D:
Notch-1 signaling regulates astrocytic proliferation and activation
after hypoxia exposure. Neurosci Lett. 603:12–18. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Meschia JF and Worrall BB: New advances in
identifying genetic anomalies in stroke-prone probands. Curr Neurol
Neurosci Rep. 4:420–426. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Xu J, Chi F, Guo T, Punj V, Lee WN, French
SW and Tsukamoto H: NOTCH reprograms mitochondrial metabolism for
proinflammatory macrophage activation. J Clin Invest.
125:1579–1590. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Pei H, Song X, Peng C, Tan Y, Li Y, Li X,
Ma S, Wang Q, Huang R, Yang D, et al: TNF-α inhibitor protects
against myocardial ischemia/reperfusion injury via Notch1-mediated
suppression of oxidative/nitrative stress. Free Radic Biol Med.
82:114–121. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Qin WD, Zhang F, Qin XJ, Wang J, Meng X,
Wang H, Guo HP, Wu QZ, Wu DW and Zhang MX: Notch1 inhibition
reduces low shear stress-induced plaque formation. Int J Biochem
Cell Biol. 72:63–72. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Palaga T, Buranaruk C, Rengpipat S, Fauq
AH, Golde TE, Kaufmann SH and Osborne BA: Notch signaling is
activated by TLR stimulation and regulates macrophage functions.
Eur J Immunol. 38:174–183. 2008. View Article : Google Scholar
|
|
71
|
Cao Q, Kaur C, Wu CY, Lu J and Ling EA:
Nuclear factor-kappa β regulates Notch signaling in production of
proinflammatory cytokines and nitric oxide in murine BV-2
microglial cells. Neuroscience. 192:140–154. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Fang M, Yuan Y, Rangarajan P, Lu J, Wu Y,
Wang H, Wu C and Ling EA: Scutellarin regulates microglia-mediated
TNC1 astrocytic reaction and astrogliosis in cerebral ischemia in
the adult rats. BMC Neurosci. 16:842015. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhou D, Huang C, Lin Z, Zhan S, Kong L,
Fang C and Li J: Macrophage polarization and function with emphasis
on the evolving roles of coordinated regulation of cellular
signaling pathways. Cell Signal. 26:192–197. 2014. View Article : Google Scholar
|
|
74
|
Qiu Y, Du B, Xie F, Cai W, Liu Y, Li Y,
Feng L and Qiu L: Vaccarin attenuates high glucose-induced human
EA•hy926 endothelial cell injury through inhibition of Notch
signaling. Mol Med Rep. 13:2143–2150. 2016.PubMed/NCBI
|
|
75
|
Henshall TL, Keller A, He L, Johansson BR,
Wallgard E, Raschperger E, Mäe MA, Jin S, Betsholtz C and Lendahl
U: Notch3 is necessary for blood vessel integrity in the central
nervous system. Arterioscler Thromb Vasc Biol. 35:409–420. 2015.
View Article : Google Scholar
|
|
76
|
Yu LM, Chen DX, Zhou QX, Fang N and Liu
ZL: Effects of histamine on immunophenotype and notch signaling in
human HL-60 leukemia cells. Exp Biol Med (Maywood). 231:1633–1637.
2006.
|
|
77
|
Boulos N, Helle F, Dussaule JC, Placier S,
Milliez P, Djudjaj S, Guerrot D, Joutel A, Ronco P, Boffa JJ and
Chatziantoniou C: Notch3 is essential for regulation of the renal
vascular tone. Hypertension. 57:1176–1182. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Fischer AJ, Zelinka C, Gallina D, Scott MA
and Todd L: Reactive microglia and macrophage facilitate the
formation of Müller glia-derived retinal progenitors. Glia.
62:1608–1628. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Shipp LE, Hill RZ, Moy GW, Gokirmak T and
Hamdoun A: ABCC5 is required for cAMP-mediated hindgut invagination
in sea urchin embryos. Development. 142:3537–3548. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Bartosh TJ, Ylostalo JH, Bazhanov N,
Kuhlman J and Prockop DJ: Dynamic compaction of human mesenchymal
stem/precursor cells into spheres self-activates caspase-dependent
IL1 signaling to enhance secretion of modulators of inflammation
and immunity (PGE2, TSG6, and STC1). Stem Cells. 31:2443–2456.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Clement N, Gueguen M, Glorian M, Blaise R,
Andréani M, Brou C, Bausero P and Limon I: Notch3 and IL-1beta
exert opposing effects on a vascular smooth muscle cell
inflammatory pathway in which NF-kappaB drives crosstalk. J Cell
Sci. 120:3352–3361. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Ali M, Heyob K and Rogers LK: DHA
suppresses primary macrophage inflammatory responses via Notch
1/Jagged 1 signaling. Sci Rep. 6:222762016. View Article : Google Scholar
|
|
83
|
Yin J, Li H, Feng C and Zuo Z: Inhibition
of brain ischemia-caused notch activation in microglia may
contribute to isoflurane postconditioning-induced neuroprotection
in male rats. CNS Neurol Disord Drug Targets. 13:718–732. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Liu Q, Fan X, Zhu J, Xu G, Li Y and Liu X:
Co-culturing improves the OGD-injured neuron repairing and NSCs
differentiation via Notch pathway activation. Neurosci Lett.
559:1–6. 2014. View Article : Google Scholar
|
|
85
|
Albéri L, Chi Z, Kadam SD, Mulholland JD,
Dawson VL, Gaiano N and Comi AM: Neonatal stroke in mice causes
long-term changes in neuronal Notch-2 expression that may
contribute to prolonged injury. Stroke. 41(Suppl 10): S64–S71.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Lipsey CC, Harbuzariu A, Daley-Brown D and
Gonzalez-Perez RR: Oncogenic role of leptin and Notch interleukin-1
leptin crosstalk outcome in cancer. World J Methodol. 6:43–55.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Grill M, Syme TE, Nocon AL, Lu AZ, Hancock
D, Rose-John S and Campbell IL: Strawberry notch homolog 2 is a
novel inflammatory response factor predominantly but not
exclusively expressed by astrocytes in the central nervous system.
Glia. 63:1738–1752. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Wang H, Tian Y, Wang J, Phillips KL, Binch
AL, Dunn S, Cross A, Chiverton N, Zheng Z, Shapiro IM, et al:
Inflammatory cytokines induce NOTCH signaling in nucleus pulposus
cells: Implications in intervertebral disc degeneration. J Biol
Chem. 288:16761–16774. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Keuylian Z, de Baaij JH, Gueguen M,
Glorian M, Rouxel C, Merlet E, Lipskaia L, Blaise R, Mateo V and
Limon I: The Notch pathway attenuates interleukin 1β
(IL1β)-mediated induction of adenylyl cyclase 8 (AC8) expression
during vascular smooth muscle cell (VSMC) trans-differentiation. J
Biol Chem. 287:24978–24989. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Mirandola L, Apicella L, Colombo M, Yu Y,
Berta DG, Platonova N, Lazzari E, Lancellotti M, Bulfamante G,
Cobos E, et al: Anti-Notch treatment prevents multiple myeloma
cells localization to the bone marrow via the chemokine system
CXCR4/SDF-1. Leukemia. 27:1558–1566. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Fukuda D, Aikawa E, Swirski FK,
Novobrantseva TI, Kotelianski V, Gorgun CZ, Chudnovskiy A, Yamazaki
H, Croce K, Weissleder R, et al: Notch ligand delta-like 4 blockade
attenuates atherosclerosis and metabolic disorders. Proc Natl Acad
Sci USA. 109:E1868–E1877. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Al Haj Zen A, Oikawa A, Bazan-Peregrino M,
Meloni M, Emanueli C and Madeddu P: Inhibition of
delta-like-4-mediated signaling impairs reparative angiogenesis
after ischemia. Circ Res. 107:283–293. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Kumari B, Jain P, Das S, Ghosal S, Hazra
B, Trivedi AC, Basu A, Chakrabarti J, Vrati S and Banerjee A:
Dynamic changes in global microRNAome and transcriptome reveal
complex miRNA-mRNA regulated host response to Japanese Encephalitis
Virus in microglial cells. Sci Rep. 6:202632016. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Yao L, Cao Q, Wu C, Kaur C, Hao A and Ling
EA: Notch signaling in the central nervous system with special
reference to its expression in microglia. CNS Neurol Disord Drug
Targets. 12:807–814. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Salta E, Lau P, Sala Frigerio C, Coolen M,
Bally-Cuif L and De Strooper B: A self-organizing miR-132/Ctbp2
circuit regulates bimodal notch signals and glial progenitor fate
choice during spinal cord maturation. Dev Cell. 30:423–436. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Grandbarbe L, Michelucci A, Heurtaux T,
Hemmer K, Morga E and Heuschling P: Notch signaling modulates the
activation of microglial cells. Glia. 55:1519–1530. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Morgan SC, Taylor DL and Pocock JM:
Microglia release activators of neuronal proliferation mediated by
activation of mitogen-activated protein kinase,
phosphatidylinositol-3-kinase/Akt and delta-Notch signalling
cascades. J Neurochem. 90:89–101. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Arumugam TV, Chan SL, Jo DG, Yilmaz G,
Tang SC, Cheng A, Gleichmann M, Okun E, Dixit VD, Chigurupati S, et
al: Gamma secretase-mediated Notch signaling worsens brain damage
and functional outcome in ischemic stroke. Nat Med. 12:621–623.
2006. View
Article : Google Scholar : PubMed/NCBI
|
|
99
|
Liu HC, Zheng MH, Du YL, Wang L, Kuang F,
Qin HY, Zhang BF and Han H: N9 microglial cells polarized by LPS
and IL4 show differential responses to secondary environmental
stimuli. Cell Immunol. 278:84–90. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Yao L, Kan EM, Kaur C, Dheen ST, Hao A, Lu
J and Ling EA: Notch-1 signaling regulates microglia activation via
NF-κB pathway after hypoxic exposure in vivo and in vitro. PLoS
One. 8:e784392013. View Article : Google Scholar
|
|
101
|
Cao Q, Lu J, Kaur C, Sivakumar V, Li F,
Cheah PS, Dheen ST and Ling EA: Expression of Notch-1 receptor and
its ligands Jagged-1 and Delta-1 in amoeboid microglia in postnatal
rat brain and murine BV-2 cells. Glia. 56:1224–1237. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Morga E, Mouad-Amazzal L, Felten P,
Heurtaux T, Moro M, Michelucci A, Gabel S, Grandbarbe L and
Heuschling P: Jagged1 regulates the activation of astrocytes via
modulation of NFkappaB and JAK/STAT/SOCS pathways. Glia.
57:1741–1753. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Nardai S, Dobolyi A, Pál G, Skopál J,
Pintér N, Lakatos K, Merkely B and Nagy Z: Selegiline promotes
NOTCH-JAGGED signaling in astrocytes of the peri-infarct region and
improves the functional integrity of the neurovascular unit in a
rat model of focal ischemia. Restor Neurol Neurosci. 33:1–14.
2015.
|
|
104
|
Monsalve E, Ruiz-García A, Baladrón V,
Ruiz-Hidalgo MJ, Sánchez-Solana B, Rivero S, García-Ramírez JJ,
Rubio A, Laborda J and Díaz-Guerra MJ: Notch1 upregulates
LPS-induced macrophage activation by increasing NF-kappaB activity.
Eur J Immunol. 39:2556–2570. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Jones DP: Extracellular redox state:
Refining the definition of oxidative stress in aging. Rejuvenation
Res. 9:169–181. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Darley-Usmar V and Halliwell B: Blood
radicals: Reactive nitrogen species, reactive oxygen species,
transition metal ions, and the vascular system. Pharm Res.
13:649–662. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Wu JQ, Kosten TR and Zhang XY: Free
radicals, antioxidant defense systems, and schizophrenia. Prog
Neuropsychopharmacol Biol Psychiatry. 46:200–206. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Catarino MD, Alves-Silva JM, Pereira OR
and Cardoso SM: Antioxidant capacities of favones and benefts in
oxidative-stress related diseases. Curr Top Med Chem. 15:105–119.
2015. View Article : Google Scholar
|
|
109
|
Lee JC and Won MH: Neuroprotection of
antioxidant enzymes against transient global cerebral ischemia in
gerbils. Anat Cell Biol. 47:149–156. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Valko M, Morris H and Cronin MT: Metals,
toxicity and oxidative stress. Curr Med Chem. 12:1161–1208. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wu D and Yotnda P: Production and
detection of reactive oxygen species (ROS) in cancers. J Vis Exp.
pii: 3357. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
112
|
Reiter RJ, Tan DX, Manchester LC and Qi W:
Biochemical reactivity of melatonin with reactive oxygen and
nitrogen species: A review of the evidence. Cell Biochem Biophys.
34:237–256. 2001. View Article : Google Scholar
|
|
113
|
Reiter RJ, Acuña-Castroviejo D, Tan DX and
Burkhardt S: Free radical-mediated molecular damage. Mechanisms for
the protective actions of melatonin in the central nervous system.
Ann N Y Acad Sci. 939:200–215. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Hemnani T and Parihar MS: Reactive oxygen
species and oxidative DNA damage. Indian J Physiol Pharmacol.
42:440–452. 1998.
|
|
115
|
Rodrigo R, Fernández-Gajardo R, Gutiérrez
R, Matamala JM, Carrasco R, Miranda-Merchak A and Feuerhake W:
Oxidative stress and pathophysiology of ischemic stroke: Novel
therapeutic opportunities. CNS Neurol Disord Drug Targets.
12:698–714. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Oprea E, Berteanu M, Cintezã D and
Manolescu BN: The effect of the ALAnerv nutritional supplement on
some oxidative stress markers in postacute stroke patients
undergoing rehabilitation. Appl Physiol Nutr Metab. 38:613–620.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Liang H, Zhang Y, Shi X, Wei T and Lou J:
Role of Notch-1 signaling pathway in PC12 cell apoptosis induced by
amyloid beta-peptide (25–35). Neural Regen Res. 9:1297–1302. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Braidy N, Jayasena T, Poljak A and Sachdev
PS: Sirtuins in cognitive ageing and Alzheimer's disease. Curr Opin
Psychiatry. 25:226–230. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Nakane H, Kamouchi M, Hata J, Ibayashi S,
Kusuda K, Omae T, Nagao T, Ago T and Kitazono T; EMINENT Study
Investigators: Effects of hydrochlorothiazide on oxidative stress
and pulse pressure in hypertensive patients with chronic stroke:
The EMINENT study. Intern Med. 54:573–577. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Nakagawa T, Hasegawa Y, Uekawa K, Ma M,
Katayama T, Sueta D, Toyama K, Kataoka K, Koibuchi N, Maeda M, et
al: Renal denervation prevents stroke and brain injury via
attenuation of oxidative stress in hypertensive rats. J Am Heart
Assoc. 2:e0003752013. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Das UN: Can free radicals induce coronary
vasospasm and acute myocardial infarction? Med Hypotheses.
39:90–94. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Manzanero S, Santro T and Arumugam TV:
Neuronal oxidative stress in acute ischemic stroke: Sources and
contribution to cell injury. Neurochem Int. 62:712–718. 2013.
View Article : Google Scholar
|
|
123
|
Cojocaru IM, Cojocaru M, Sapira V and
Ionescu A: Evaluation of oxidative stress in patients with acute
ischemic stroke. Rom J Intern Med. 51:97–106. 2013.PubMed/NCBI
|
|
124
|
Icme F, Erel Ö, Avci A, Satar S, Gülen M
and Acehan S: The relation between oxidative stress parameters,
ischemic stroke, and hemorrhagic stroke. Turk J Med Sci.
45:947–953. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Simão AN, Lehmann MF, Alferi DF, Meloni
MZ, Flauzino T, Scavuzzi BM, de Oliveira SR, Lozovoy MA, Dichi I
and Reiche EM: Metabolic syndrome increases oxidative stress but
does not influence disability and short-time outcome in acute
ischemic stroke patients. Metab Brain Dis. 30:1409–1416. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Tsai NW, Chang YT, Huang CR, Lin YJ, Lin
WC, Cheng BC, Su CM, Chiang YF, Chen SF, Huang CC, et al:
Association between oxidative stress and outcome in different
subtypes of acute ischemic stroke. Biomed Res Int. 2014:2568792014.
View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Pantcheva P, Elias M, Duncan K, Borlongan
CV, Tajiri N and Kaneko Y: The role of DJ-1 in the oxidative stress
cell death cascade after stroke. Neural Regen Res. 9:1430–1433.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Nabavi SF, Dean OM, Turner A, Sureda A,
Daglia M and Nabavi SM: Oxidative stress and post-stroke
depression: Possible therapeutic role of polyphenols? Curr Med
Chem. 22:343–351. 2015. View Article : Google Scholar
|
|
129
|
Gonullu H, Aslan M, Karadas S, Kati C,
Duran L, Milanlioglu A, Aydin MN and Demir H: Serum prolidase
enzyme activity and oxidative stress levels in patients with acute
hemorrhagic stroke. Scand J Clin Lab Invest. 74:199–205. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
El Kossi MM and Zakhary MM: Oxidative
stress in the context of acute cerebrovascular stroke. Stroke.
31:1889–1892. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Milanlioglu A, Aslan M, Ozkol H, Çilingir
V, Nuri Aydin M and Karadas S: Serum antioxidant enzymes activities
and oxidative stress levels in patients with acute ischemic stroke:
Influence on neurological status and outcome. Wien Klin Wochenschr.
128:169–174. 2016. View Article : Google Scholar
|
|
132
|
Newton DF, Naiberg MR and Goldstein BI:
Oxidative stress and cognition amongst adults without dementia or
stroke: Implications for mechanistic and therapeutic research in
psychiatric disorders. Psychiatry Res. 227:127–134. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Nakajima H, Kubo T, Ihara H, Hikida T,
Danjo T, Nakatsuji M, Shahani N, Itakura M, Ono Y, Azuma YT, et al:
Nuclear-translocated Glyceraldehyde-3-phosphate dehydrogenase
promotes poly(ADP-ribose) polymerase-1 activation during
Oxidative/Nitrosative stress in stroke. J Biol Chem.
290:14493–14503. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Kotur-Stevuljevic J, Bogavac-Stanojevic N,
Jelic-Ivanovic Z, Stefanovic A, Gojkovic T, Joksic J, Sopic M,
Gulan B, Janac J and Milosevic S: Oxidative stress and paraoxonase
1 status in acute ischemic stroke patients. Atherosclerosis.
241:192–198. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Han Z, Shen F, He Y, Degos V, Camus M,
Maze M, Young WL and Su H: Activation of α-7 nicotinic
acetylcholine receptor reduces ischemic stroke injury through
reduction of pro-inflammatory macrophages and oxidative stress.
PLoS One. 9:e1057112014. View Article : Google Scholar
|
|
136
|
Lagowska-Lenard M, Bielewicz J, Raszewski
G, Stelmasiak Z and Bartosik-Psujek H: Oxidative stress in cerebral
stroke. Pol Merkur Lekarski. 25:205–208. 2008.In Polish.
|
|
137
|
Takemori K, Murakami T, Kometani T and Ito
H: Possible involvement of oxidative stress as a causative factor
in blood-brain barrier dysfunction in stroke-prone spontaneously
hypertensive rats. Microvasc Res. 90:169–172. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Hung LM, Huang JP, Liao JM, Yang MH, Li
DE, Day YJ and Huang SS: Insulin renders diabetic rats resistant to
acute ischemic stroke by arresting nitric oxide reaction with
superoxide to form peroxynitrite. J Biomed Sci. 21:922014.
View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Fabian RH and Kent TA: Hyperglycemia
accentuates persistent 'functional uncoupling' of cerebral
microvascular nitric oxide and superoxide following focal
ischemia/reperfusion in rats. Transl Stroke Res. 3:482–490. 2012.
View Article : Google Scholar
|
|
140
|
Fabian RH, Perez-Polo JR and Kent TA:
Perivascular nitric oxide and superoxide in neonatal cerebral
hypoxia-ischemia. Am J Physiol Heart Circ Physiol. 295:H1809–H1814.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Gümüştaş K, Meta Güzeyli FM, Atükeren P,
Sanus GZ, Kemerdere R, Tanriverdi T and Kaynar MY: The effects of
vitamin E on lipid peroxidation, nitric oxide production and
superoxide dismutase expression in hyperglycemic rats with cerebral
ischemia-reperfusion injury. Turk Neurosurg. 17:78–82. 2007.
|
|
142
|
Forman LJ, Liu P, Nagele RG, Yin K and
Wong PY: Augmentation of nitric oxide, superoxide, and
peroxynitrite production during cerebral ischemia and reperfusion
in the rat. Neurochem Res. 23:141–148. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Brosnan MJ, Hamilton CA, Graham D, Lygate
CA, Jardine E and Dominiczak AF: Irbesartan lowers superoxide
levels and increases nitric oxide bioavailability in blood vessels
from spontaneously hypertensive stroke-prone rats. J Hypertens.
20:281–286. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Baumeister P, Huebner T, Reiter M,
Schwenk-Zieger S and Harréus U: Reduction of oxidative DNA
fragmentation by ascorbic acid, zinc and N-acetylcysteine in nasal
mucosa tissue cultures. Anticancer Res. 29:4571–4574.
2009.PubMed/NCBI
|
|
145
|
Mikhaĭlov VF, Mazurik VK and Burlakova EB:
Signal function of the reactive oxygen species in regulatory
networks of the cell reaction to damaging effects: Contribution of
radiosensitivity and genome instability. Radiats Biol Radioecol.
43:5–18. 2003.In Russian.
|
|
146
|
Fischer-Nielsen A, Corcoran GB, Poulsen
HE, Kamendulis LM and Loft S: Menadione-induced DNA fragmentation
without 8-oxo-2′-deoxyguanosine formation in isolated rat
hepatocytes. Biochem Pharmacol. 49:1469–1474. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Zhou T, He Q, Tong Y, Zhan R, Xu F, Fan D,
Guo X, Han H, Qin S and Chui D: Phospholipid transfer protein
(PLTP) deficiency impaired blood-brain barrier integrity by
increasing cerebrovascular oxidative stress. Biochem Biophys Res
Commun. 445:352–356. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Tóth AE, Walter FR, Bocsik A, Sántha P,
Veszelka S, Nagy L, Puskás LG, Couraud PO, Takata F, Dohgu S, et
al: Edaravone protects against methylglyoxal-induced barrier damage
in human brain endothelial cells. PLoS One. 9:e1001522014.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Elmorsy E, Elzalabany LM, Elsheikha HM and
Smith PA: Adverse effects of antipsychotics on micro-vascular
endothelial cells of the human blood-brain barrier. Brain Res.
1583:255–268. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Sathanoori R, Swärd K, Olde B and Erlinge
D: The ATP Receptors P2X7 and P2X4 modulate high glucose and
Palmitate-Induced inflammatory responses in endothelial cells. PLoS
One. 10:e01251112015. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Okada R, Wu Z, Zhu A, Ni J, Zhang J,
Yoshimine Y, Peters C, Saftig P and Nakanishi H: Cathepsin D
deficiency induces oxidative damage in brain pericytes and impairs
the blood-brain barrier. Mol Cell Neurosci. 64:51–60. 2015.
View Article : Google Scholar
|
|
152
|
Abdul-Muneer PM, Chandra N and Haorah J:
Interactions of oxidative stress and neurovascular infammation in
the pathogenesis of traumatic brain injury. Mol Neurobiol.
51:966–979. 2015. View Article : Google Scholar
|
|
153
|
Ste-Marie L, Hazell AS, Bémeur C,
Butterworth R and Montgomery J: Immunohistochemical detection of
inducible nitric oxide synthase, nitrotyrosine and manganese
superoxide dismutase following hyperglycemic focal cerebral
ischemia. Brain Res. 918:10–19. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Kumura E, Yoshimine T, Kubo S, Tanaka S,
Hayakawa T, Shiga T and Kosaka H: Effects of superoxide dismutase
on nitric oxide production during reperfusion after focal cerebral
ischemia is rats. Neurosci Lett. 200:137–140. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Al-Maghrebi M and Renno WM: Genistein
alleviates testicular ischemia and reperfusion injury-induced
sper-matogenic damage and oxidative stress by suppressing abnormal
testicular matrix metalloproteinase system via the Notch 2/Jagged
1/Hes-1 and caspase-8 pathways. J Physiol Pharmacol. 67:129–137.
2016.PubMed/NCBI
|
|
156
|
Xie H, Sun J, Chen Y, Zong M, Li S and
Wang Y: EGCG attenuates uric Acid-Induced inflammatory and
oxidative stress responses by medicating the NOTCH pathway. Oxid
Med Cell Longev. 2015:2148362015. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Xie F, Cai W, Liu Y, Li Y, Du B, Feng L
and Qiu L: Vaccarin attenuates the human EA.hy926 endothelial cell
oxidative stress injury through inhibition of Notch signaling. Int
J Mol Med. 35:135–142. 2015.
|
|
158
|
Yang Y, Duan W, Liang Z, Yi W, Yan J, Wang
N, Li Y, Chen W, Yu S, Jin Z and Yi D: Curcumin attenuates
endothelial cell oxidative stress injury through Notch signaling
inhibition. Cell Signal. 25:615–629. 2013. View Article : Google Scholar
|
|
159
|
Li M, Chen F, Clifton N, Sullivan DM,
Dalton WS, Gabrilovich DI and Nefedova Y: Combined inhibition of
Notch signaling and Bcl-2/Bcl-xL results in synergistic antimyeloma
effect. Mol Cancer Ther. 9:3200–3209. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Chen C, Cui H, Li Z, Wang R and Zhou C:
Normobaric oxygen for cerebral ischemic injury. Neural Regen Res.
8:2885–2894. 2013.
|
|
161
|
Zhu B, Yang P, Mammat N, Ding H, He J,
Qian Y, Fei J and Abdukerim K: Aiweixin, a traditional Uyghur
medicinal formula, protects against chromium toxicity in
Caenorhabditis elegans. BMC Complement Altern Med. 15:2852015.
View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Finsterer J: Neuromuscular implications in
CADASIL. Cerebrovasc Dis. 24:401–404. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Santoni M, Pantano F, Amantini C, Nabissi
M, Conti A, Burattini L, Zoccoli A, Berardi R, Santoni G, Tonini G,
et al: Emerging strategies to overcome the resistance to current
mTOR inhibitors in renal cell carcinoma. Biochim Biophys Acta.
1845:221–231. 2014.PubMed/NCBI
|
|
164
|
Pei H, Yu Q, Xue Q, Guo Y, Sun L, Hong Z,
Han H, Gao E, Qu Y and Tao L: Notch1 cardioprotection in myocardial
ischemia/reperfusion involves reduction of oxidative/nitrative
stress. Basic Res Cardiol. 108:3732013. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Simón R, Aparicio R, Housden BE, Bray S
and Busturia A: Drosophila p53 controls Notch expression and
balances apoptosis and proliferation. Apoptosis. 19:1430–1443.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Zheng WX, Cao XL, Wang F, Wang J, Ying TZ,
Xiao W, Zhang Y, Xing H, Dong W, Xu SQ, et al: Baicalin inhibiting
cerebral ischemia/hypoxia-induced neuronal apoptosis via
MRTF-A-mediated transactivity. Eur J Pharmacol. 767:201–210. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Baroja-Mazo A, Martín-Sánchez F, Gomez AI,
Martínez CM, Amores-Iniesta J, Compan V, Barberà-Cremades M, Yagüe
J, Ruiz-Ortiz E, Antón J, et al: The NLRP3 inflammasome is released
as a particulate danger signal that amplifes the inflammatory
response. Nat Immunol. 15:738–748. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Yang RY and Liu FT: Galectins in cell
growth and apoptosis. Cell Mol Life Sci. 60:267–276. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Bao JX, Su YT, Cheng YP, Zhang HJ, Xie XP
and Chang YM: Vascular sphingolipids in physiological and
pathological adaptation. Front Biosci (Landmark Ed). 21:1168–1186.
2016. View Article : Google Scholar
|
|
170
|
Kagiya G, Ogawa R, Tabuchi Y, Feril LB Jr,
Nozaki T, Fukuda S, Yamamoto K and Kondo T: Expression of heme
oxygenase-1 due to intracellular reactive oxygen species induced by
ultrasound. Ultrason Sonochem. 13:388–396. 2006. View Article : Google Scholar
|
|
171
|
Santiago B, Galindo M, Palao G and Pablos
JL: Intracellular regulation of Fas-induced apoptosis in human
fibroblasts by extracellular factors and cycloheximide. J Immunol.
172:560–566. 2004. View Article : Google Scholar
|
|
172
|
Wang L, Song G, Liu M, Chen B, Chen Y,
Shen Y, Zhu J and Zhou X: MicroRNA-375 overexpression influences
P19 cell proliferation, apoptosis and differentiation through the
Notch signaling pathway. Int J Mol Med. 37:47–55. 2016.
|
|
173
|
Aboutaleb N, Shamsaei N, Khaksari M,
Erfani S, Rajabi H and Nikbakht F: Pre-ischemic exercise reduces
apoptosis in hippocampal CA3 cells after cerebral ischemia by
modulation of the Bax/Bcl-2 proteins ratio and prevention of
caspase-3 activation. J Physiol Sci. 65:435–443. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Zhang JF, Shi LL, Zhang L, Zhao ZH, Liang
F, Xu X, Zhao LY, Yang PB, Zhang JS and Tian YF: MicroRNA-25
negatively regulates cerebral Ischemia/Reperfusion Injury-Induced
cell apoptosis through Fas/FasL pathway. J Mol Neurosci.
58:507–516. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Xue R, Wu G, Wei X, Lv J, Fu R, Lei X,
Zhang Z, Li W, He J, Zhao H, et al: Tea polyphenols may attenuate
the neurocognitive impairment caused by global cerebral
ischemia/reperfusion injury via anti-apoptosis. Nutr Neurosci.
19:63–69. 2016. View Article : Google Scholar
|
|
176
|
Yang Y, Gao K, Hu Z, Li W, Davies H, Ling
S, Rudd JA and Fang M: Autophagy upregulation and apoptosis
downregulation in DAHP and triptolide treated cerebral ischemia.
Mediators Inflamm. 2015:1201982015. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Wu L, Zhao QS, Li TW, Li HY, Wang QB, Bi
XY, Cai XK and Tang N: Yifei Xuanfei Jiangzhuo formula, a Chinese
herbal decoction, improves memory impairment through inhibiting
apoptosis and enhancing PKA/CREB signal transduction in rats with
cerebral ischemia/reperfusion. Mol Med Rep. 12:4273–4283.
2015.PubMed/NCBI
|
|
178
|
Saad MA, Abdelsalam RM, Kenawy SA and
Attia AS: Ischemic preconditioning and postconditioning alleviates
hippocampal tissue damage through abrogation of apoptosis modulated
by oxidative stress and inflammation during transient global
cerebral ischemia-reperfusion in rats. Chem Biol Interact.
232:21–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Fan M, Jin W, Zhao H, Xiao Y, Jia Y, Yin
Y, Jiang X, Xu J, Meng N and Lv P: Lithium chloride administration
prevents spatial learning and memory impairment in repeated
cerebral ischemia-reperfusion mice by depressing apoptosis and
increasing BDNF expression in hippocampus. Behav Brain Res.
291:399–406. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Garrigue P, Giacomino L, Bucci C, Muzio V,
Filannino MA, Sabatier F, Dignat-George F, Pisano P and Guillet B:
Single photon emission computed tomography imaging of cerebral
blood flow, blood-brain barrier disruption, and apoptosis time
course after focal cerebral ischemia in rats. Int J Stroke.
11:117–126. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Cao G, Zhou H, Jiang N, Han Y, Hu Y, Zhang
Y, Qi J, Kou J and Yu B: YiQiFuMai powder injection ameliorates
cerebral ischemia by inhibiting endoplasmic reticulum
Stress-Mediated neuronal apoptosis. Oxid Med Cell Longev.
2016:54932792016. View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Yan XG, Cheng BH, Wang X, Ding LC, Liu HQ,
Chen J and Bai B: Lateral intracerebroventricular injection of
Apelin-13 inhibits apoptosis after cerebral ischemia/reperfusion
injury. Neural Regen Res. 10:766–771. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Saad MA, Abdel Salam RM, Kenawy SA and
Attia AS: Pinocembrin attenuates hippocampal inflammation,
oxidative perturbations and apoptosis in a rat model of global
cerebral ischemia reperfusion. Pharmacol Rep. 67:115–122. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Jiang Y, Li L, Tan X, Liu B, Zhang Y and
Li C: miR-210 mediates vagus nerve stimulation-induced antioxidant
stress and anti-apoptosis reactions following cerebral
ischemia/reperfusion injury in rats. J Neurochem. 134:173–181.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Chopp M and Li Y: Apoptosis in focal
cerebral ischemia. Acta Neurochir Suppl. 66:21–26. 1996.PubMed/NCBI
|
|
186
|
Wu X, Li L, Zhang L, Wu J, Zhou Y, Zhou Y,
Zhao Y and Zhao J: Inhibition of thioredoxin-1 with siRNA
exacerbates apoptosis by activating the ASK1-JNK/p38 pathway in
brain of a stroke model rats. Brain Res. 1599:20–31. 2015.
View Article : Google Scholar
|
|
187
|
Baregamian N, Song J, Bailey CE,
Papaconstantinou J, Evers BM and Chung DH: Tumor necrosis
factor-alpha and apoptosis signal-regulating kinase 1 control
reactive oxygen species release, mitochondrial autophagy, and c-Jun
N-terminal kinase/p38 phosphorylation during necrotizing
enterocolitis. Oxid Med Cell Longev. 2:297–306. 2009. View Article : Google Scholar
|
|
188
|
Bedogni B, Warneke JA, Nickoloff BJ,
Giaccia AJ and Powell MB: Notch1 is an effector of Akt and hypoxia
in melanoma development. J Clin Invest. 118:3660–3670. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Petit A, Bihel F, Alvès da Costa C,
Pourquié O, Checler F and Kraus JL: New protease inhibitors prevent
gamma-secretase-mediated production of Abeta40/42 without affecting
Notch cleavage. Nat Cell Biol. 3:507–511. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Okochi M, Steiner H, Fukumori A, Tanii H,
Tomita T, Tanaka T, Iwatsubo T, Kudo T, Takeda M and Haass C:
Presenilins mediate a dual intramembranous gamma-secretase cleavage
of Notch-1. EMBO J. 21:5408–5416. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Ikeuchi T and Sisodia SS: The Notch
ligands, Delta1 and Jagged2, are substrates for
presenilin-dependent 'gamma-secretase' cleavage. J Biol Chem.
278:7751–7754. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Yang G, Gong Y, Wang Q, Wang Y and Zhang
X: The role of miR-100-mediated Notch pathway in apoptosis of
gastric tumor cells. Cell Signal. 27:1087–1101. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
193
|
Liu XD, Zhang LY, Zhu TC, Zhang RF, Wang
SL and Bao Y: Overexpression of miR-34c inhibits high
glucose-induced apoptosis in podocytes by targeting Notch signaling
pathways. Int J Clin Exp Pathol. 8:4525–4534. 2015.PubMed/NCBI
|
|
194
|
Wang XM, Yao M, Liu SX, Hao J, Liu QJ and
Gao F: Interplay between the Notch and PI3K/Akt pathways in high
glucose-induced podocyte apoptosis. Am J Physiol Renal Physiol.
306:F205–F213. 2014. View Article : Google Scholar
|
|
195
|
Gao F, Yao M, Shi Y, Hao J, Ren Y, Liu Q,
Wang X and Duan H: Notch pathway is involved in high
glucose-induced apoptosis in podocytes via Bcl-2 and p53 pathways.
J Cell Biochem. 114:1029–1038. 2013. View Article : Google Scholar
|
|
196
|
Yang Y, Li X, Zhang L, Liu L, Jing G and
Cai H: Ginsenoside Rg1 suppressed infammation and neuron apoptosis
by activating PPAR γ/HO-1 in hippocampus in rat model of cerebral
ischemia-reperfusion injury. Int J Clin Exp Pathol. 8:2484–2494.
2015.
|
|
197
|
Zhao Y, Deng B, Li Y, Zhou L, Yang L, Gou
X, Wang Q, Chen G, Xu H and Xu L: Electroacupuncture pretreatment
attenuates cerebral ischemic injury via Notch Pathway-Mediated
Up-Regulation of hypoxia inducible Factor-1α in Rats. Cell Mol
Neurobiol. 35:1093–1103. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Cheng YL, Choi Y, Seow WL, Manzanero S,
Sobey CG, Jo DG and Arumugam TV: Evidence that neuronal Notch-1
promotes JNK/c-Jun activation and cell death following ischemic
stress. Brain Res. 1586:193–202. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Meng S, Su Z, Liu Z, Wang N and Wang Z:
Rac1 contributes to cerebral ischemia reperfusion-induced injury in
mice by regulation of Notch2. Neuroscience. 306:100–114. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Ma M, Wang X, Ding X, Teng J, Shao F and
Zhang J: Numb/Notch signaling plays an important role in cerebral
ischemia-induced apoptosis. Neurochem Res. 38:254–261. 2013.
View Article : Google Scholar
|
|
201
|
Sun J, Ling Z, Wang F, Chen W, Li H, Jin
J, Zhang H, Pang M, Yu J and Liu J: Clostridium butyricum
pretreatment attenuates cerebral ischemia/reperfusion injury in
mice via anti-oxidation and anti-apoptosis. Neurosci Lett.
613:30–35. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Arumugam TV, Cheng YL, Choi Y, Choi YH,
Yang S, Yun YK, Park JS, Yang DK, Thundyil J, Gelderblom M, et al:
Evidence that gamma-secretase-mediated Notch signaling induces
neuronal cell death via the nuclear factor-kappaB-Bcl-2-interacting
mediator of cell death pathway in ischemic stroke. Mol Pharmacol.
80:23–31. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Park JS, Manzanero S, Chang JW, Choi Y,
Baik SH, Cheng YL, Li YI, Gwon AR, Woo HN, Jang J, et al:
Calsenilin contributes to neuronal cell death in ischemic stroke.
Brain Pathol. 23:402–412. 2013. View Article : Google Scholar
|
|
204
|
Baik SH, Fane M, Park JH, Cheng YL,
Yang-Wei Fann D, Yun UJ, Choi Y, Park JS, Chai BH, Back SH, et al:
Pin1 promotes neuronal death in stroke by stabilizing Notch
intracellular domain. Ann Neurol. 77:504–516. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Viswanathan A, Gray F, Bousser MG,
Baudrimont M and Chabriat H: Cortical neuronal apoptosis in
CADASIL. Stroke. 37:2690–2695. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Kalimo H, Ruchoux MM, Viitanen M and
Kalaria RN: CADASIL: A common form of hereditary arteriopathy
causing brain infarcts and dementia. Brain Pathol. 12:371–384.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Liu XY, Gonzalez-Toledo ME, Fagan A, Duan
WM, Liu Y, Zhang S, Li B, Piao CS, Nelson L and Zhao LR: Stem cell
factor and granulocyte colony-stimulating factor exhibit
therapeutic effects in a mouse model of CADASIL. Neurobiol Dis.
73:189–203. 2015. View Article : Google Scholar
|
|
208
|
Wang S, Yuan Y, Xia W, Li F, Huang Y, Zhou
Y and Guo Y: Neuronal apoptosis and synaptic density in the dentate
gyrus of ischemic rats' response to chronic mild stress and the
effects of Notch signaling. PLoS One. 7:e428282012. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Zhang HP, Sun YY, Chen XM, Yuan LB, Su BX,
Ma R, Zhao RN, Dong HL and Xiong L: The neuroprotective effects of
isofurane preconditioning in a murine transient global cerebral
ischemia-reperfusion model: The role of the Notch signaling
pathway. Neuromolecular Med. 16:191–204. 2014. View Article : Google Scholar
|
|
210
|
Yang Q, Yan W, Li X, Hou L, Dong H, Wang
Q, Wang S, Zhang X and Xiong L: Activation of canonical notch
signaling pathway is involved in the ischemic tolerance induced by
sevo-flurane preconditioning in mice. Anesthesiology. 117:996–1005.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Yao J and Qian C: Over-activated Notch-1
protects gastric carcinoma BGC-823 cells from TNFalpha-induced
apoptosis. Dig Liver Dis. 41:867–874. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Yang X, Klein R, Tian X, Cheng HT, Kopan R
and Shen J: Notch activation induces apoptosis in neural progenitor
cells through a p53-dependent pathway. Dev Biol. 269:81–94. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
de Antonellis P, Medaglia C, Cusanelli E,
Andolfo I, Liguori L, De Vita G, Carotenuto M, Bello A, Formiggini
F, Galeone A, et al: MiR-34a targeting of Notch ligand delta-like 1
impairs CD15+/CD133+ tumor-propagating cells and supports neural
differentiation in medulloblastoma. PLoS One. 6:e245842011.
View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Sionov RV, Kfr-Erenfeld S, Spokoini R and
Yefenof E: A role for bcl-2 in notch1-dependent transcription in
thymic lymphoma cells. Adv Hematol. 2012:4352412012. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Ye QF, Zhang YC, Peng XQ, Long Z, Ming YZ
and He LY: Silencing Notch-1 induces apoptosis and increases the
chemo-sensitivity of prostate cancer cells to docetaxel through
Bcl-2 and Bax. Oncol Lett. 3:879–884. 2012.PubMed/NCBI
|
|
216
|
Cao H, Hu Y, Wang P, Zhou J, Deng Z and
Wen J: Down-regulation of Notch receptor signaling pathway induces
caspase-dependent and caspase-independent apoptosis in lung
squamous cell carcinoma cells. APMIS. 120:441–450. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Brockhaus M, Grünberg J, Röhrig S,
Loetscher H, Wittenburg N, Baumeister R, Jacobsen H and Haass C:
Caspase-mediated cleavage is not required for the activity of
presenilins in amyloidogenesis and NOTCH signaling. Neuroreport.
9:1481–1486. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Wu K, Hu L and Hou J: Selective
suppression of Notch1 inhibits proliferation of renal cell
carcinoma cells through JNK/p38 pathway. Oncol Rep. 35:2795–2800.
2016.PubMed/NCBI
|
|
219
|
Smith KA, Voiriot G, Tang H, Fraidenburg
DR, Song S, Yamamura H, Yamamura A, Guo Q, Wan J, Pohl NM, et al:
Notch activation of Ca(2+) signaling in the development of hypoxic
pulmonary vasoconstriction and pulmonary hypertension. Am J Respir
Cell Mol Biol. 53:355–367. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Rothschild SC, Lahvic J, Francescatto L,
McLeod JJ, Burgess SM and Tombes RM: CaMK-II activation is
essential for zebrafsh inner ear development and acts through
Delta-Notch signaling. Dev Biol. 381:179–188. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Kim SK, Park HJ, Hong HS, Baik EJ, Jung MW
and Mook-Jung I: ERK1/2 is an endogenous negative regulator of the
gamma-secretase activity. FASEB J. 20:157–159. 2006.
|
|
222
|
Servín-González LS, Granados-López AJ and
López JA: Families of microRNAs expressed in clusters regulate cell
signaling in cervical cancer. Int J Mol Sci. 16:12773–12790. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Aguirre A, Rubio ME and Gallo V: Notch and
EGFR pathway interaction regulates neural stem cell number and
self-renewal. Nature. 467:323–327. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Nagaraj R and Banerjee U: Regulation of
Notch and Wingless signalling by phyllopod, a transcriptional
target of the EGFR pathway. EMBO J. 28:337–346. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Elmadhun NY, Sabe AA, Lassaletta AD, Chu
LM, Kondra K, Sturek M and Sellke FW: Metabolic syndrome impairs
notch signaling and promotes apoptosis in chronically ischemic
myocardium. J Thorac Cardiovasc Surg. 148:1048–1055. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Guo D, Ye J, Dai J, Li L, Chen F, Ma D and
Ji C: Notch-1 regulates Akt signaling pathway and the expression of
cell cycle regulatory proteins cyclin D1, CDK2 and p21 in T-ALL
cell lines. Leuk Res. 33:678–685. 2009. View Article : Google Scholar
|
|
227
|
Sweetwyne MT, Gruenwald A, Niranjan T,
Nishinakamura R, Strobl LJ and Susztak K: Notch1 and Notch2 in
podocytes play differential roles during diabetic nephropathy
development. Diabetes. 64:4099–4111. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Bheeshmachar G, Purushotaman D, Sade H,
Gunasekharan V, Rangarajan A and Sarin A: Evidence for a role for
notch signaling in the cytokine-dependent survival of activated T
cells. J Immunol. 177:5041–5050. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Sholler GS, Currier EA, Dutta A, Slavik
MA, Illenye SA, Mendonca MC, Dragon J, Roberts SS and Bond JP:
PCI-24781 (abexinostat), a novel histone deacetylase inhibitor,
induces reactive oxygen species-dependent apoptosis and is
synergistic with bortezomib in neuroblastoma. J Cancer Ther Res.
2:212013. View Article : Google Scholar
|
|
230
|
Yu HC, Bai L, Yue SQ, Wang DS, Wang L, Han
H and Dou KF: Notch signal protects non-parenchymal cells from
ischemia/reperfusion injury in vitro by repressing ROS. Ann
Hepatol. 12:815–821. 2013.PubMed/NCBI
|
|
231
|
Naik S, MacFarlane M and Sarin A: Notch4
signaling confers susceptibility to TRAIL-induced apoptosis in
breast cancer cells. J Cell Biochem. 116:1371–1380. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Wang C, Qi R, Li N, Wang Z, An H, Zhang Q,
Yu Y and Cao X: Notch1 signaling sensitizes tumor necrosis
factor-related apoptosis-inducing ligand-induced apoptosis in human
hepatocellular carcinoma cells by inhibiting Akt/Hdm2-mediated p53
degradation and up-regulating p53-dependent DR5 expression. J Biol
Chem. 284:16183–16190. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Chung AS, Lee J and Ferrara N: Targeting
the tumour vascu-lature: Insights from physiological angiogenesis.
Nat Rev Cancer. 10:505–514. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Carmeliet P, Moons L, Dewerchin M, Mackman
N, Luther T, Breier G, Ploplis V, Müller M, Nagy A, Plow E, et al:
Insights in vessel development and vascular disorders using
targeted inactivation and transfer of vascular endothelial growth
factor, the tissue factor receptor and the plasminogen system. Ann
N Y Acad Sci. 811:191–206. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Lymboussaki A, Olofsson B, Eriksson U and
Alitalo K: Vascular endothelial growth factor (VEGF) and VEGF-C
show overlapping binding sites in embryonic endothelia and distinct
sites in differentiated adult endothelia. Circ Res. 85:992–999.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
McColl BK, Stacker SA and Achen MG:
Molecular regulation of the VEGF family-inducers of angiogenesis
and lymphangiogenesis. APMIS. 112:463–480. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Przybylski M: A review of the current
research on the role of bFGF and VEGF in angiogenesis. J Wound
Care. 18:516–519. 2009. View Article : Google Scholar
|
|
238
|
Li JL and Harris AL: Crosstalk of VEGF and
Notch pathways in tumour angiogenesis: Therapeutic implications.
Front Biosci (Landmark Ed). 14:3094–3110. 2009. View Article : Google Scholar
|
|
239
|
Dimova I, Popivanov G and Djonov V:
Angiogenesis in cancer-general pathways and their therapeutic
implications. J BUON. 19:15–21. 2014.PubMed/NCBI
|
|
240
|
Phng LK and Gerhardt H: Angiogenesis: A
team effort coordinated by notch. Dev Cell. 16:196–208. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
241
|
Benedito R, Roca C, Sörensen I, Adams S,
Gossler A, Fruttiger M and Adams RH: The notch ligands Dll4 and
Jagged1 have opposing effects on angiogenesis. Cell. 137:1124–1135.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
242
|
Boas SE and Merks RM: Tip cell overtaking
occurs as a side effect of sprouting in computational models of
angiogenesis. BMC Syst Biol. 9:862015. View Article : Google Scholar : PubMed/NCBI
|
|
243
|
Garcia-Pascual CM, Zimmermann RC, Ferrero
H, Shawber CJ, Kitajewski J, Simón C, Pellicer A and Gomez R:
Delta-like ligand 4 regulates vascular endothelial growth factor
receptor 2-driven luteal angiogenesis through induction of a
tip/stalk phenotype in proliferating endothelial cells. Fertil
Steril. 100:1768–1776.e1. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
244
|
Brzozowa M, Wojnicz R, Kowalczyk-Ziomek G
and Helewski K: The Notch ligand Delta-like 4 (DLL4) as a target in
angiogenesis-based cancer therapy? Contemp Oncol (Pozn).
17:234–237. 2013.
|
|
245
|
Fukuhara S, Sako K, Noda K, Zhang J,
Minami M and Mochizuki N: Angiopoietin-1/Tie2 receptor signaling in
vascular quiescence and angiogenesis. Histol Histopathol.
25:387–396. 2010.PubMed/NCBI
|
|
246
|
Cao Y, Cao R and Hedlund EM: R Regulation
of tumor angiogenesis and metastasis by FGF and PDGF signaling
pathways. J Mol Med (Berl). 86:785–789. 2008. View Article : Google Scholar
|
|
247
|
van Meeteren LA, Goumans MJ and ten Dijke
P: TGF-β receptor signaling pathways in angiogenesis; emerging
targets for anti-angiogenesis therapy. Curr Pharm Biotechnol.
12:2108–2120. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
248
|
Orlova VV, Liu Z, Goumans MJ and ten Dijke
P: Controlling angiogenesis by two unique TGF-β type I receptor
signaling pathways. Histol Histopathol. 26:1219–1230.
2011.PubMed/NCBI
|
|
249
|
Taniyama Y, Morishita R, Aoki M, Nakagami
H, Yamamoto K, Yamazaki K, Matsumoto K, Nakamura T, Kaneda Y and
Ogihara T: Therapeutic angiogenesis induced by human hepatocyte
growth factor gene in rat and rabbit hindlimb ischemia models:
Preclinical study for treatment of peripheral arterial disease.
Gene Ther. 8:181–189. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
250
|
Wu L, Fu Z, Zhou S, Gong J, Liu CA, Qiao Z
and Li S: HIF-12α and HIF-22α: Siblings in promoting angiogenesis
of residual hepatocellular carcinoma after high-intensity focused
ultrasound ablation. PLoS One. 9:e889132014. View Article : Google Scholar
|
|
251
|
Hayashi H and Kume T: Foxc transcription
factors directly regulate Dll4 and Hey2 expression by interacting
with the VEGF-Notch signaling pathways in endothelial cells. PLoS
One. 3:e24012008. View Article : Google Scholar : PubMed/NCBI
|
|
252
|
Mitsuhashi N, Shimizu H, Ohtsuka M,
Wakabayashi Y, Ito H, Kimura F, Yoshidome H, Kato A, Nukui Y and
Miyazaki M: Angiopoietins and Tie-2 expression in angiogenesis and
proliferation of human hepatocellular carcinoma. Hepatology.
37:1105–1113. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
253
|
Wulff C, Wilson H, Largue P, Duncan WC,
Armstrong DG and Fraser HM: Angiogenesis in the human corpus
luteum: Localization and changes in angiopoietins, tie-2 and
vascular endothelial growth factor messenger ribonucleic acid. J
Clin Endocrinol Metab. 85:4302–4309. 2000.PubMed/NCBI
|
|
254
|
Weinmaster G: Notch signaling: Direct or
what? Curr Opin Genet Dev. 8:436–442. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
255
|
Reizis B and Leder P: Direct induction of
T lymphocyte-specific gene expression by the mammalian Notch
signaling pathway. Genes Dev. 16:295–300. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
256
|
Nakano N, Nishiyama C, Yagita H, Hara M,
Motomura Y, Kubo M, Okumura K and Ogawa H: Notch signaling enhances
FcεRI-mediated cytokine production by mast cells through direct and
indirect mechanisms. J Immunol. 194:4535–4544. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
257
|
Wüstehube J, Bartol A, Liebler SS, Brütsch
R, Zhu Y, Felbor U, Sure U, Augustin HG and Fischer A: Cerebral
cavernous malformation protein CCM1 inhibits sprouting angiogenesis
by activating DELTA-NOTCH signaling. Proc Natl Acad Sci USA.
107:12640–12645. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
258
|
You C, Sandalcioglu IE, Dammann P, Felbor
U, Sure U and Zhu Y: Loss of CCM3 impairs DLL4-Notch signalling:
Implication in endothelial angiogenesis and in inherited cerebral
cavernous malformations. J Cell Mol Med. 17:407–418. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
259
|
Patel NS, Li JL, Generali D, Poulsom R,
Cranston DW and Harris AL: Up-regulation of delta-like 4 ligand in
human tumor vasculature and the role of basal expression in
endothelial cell function. Cancer Res. 65:8690–8697. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
260
|
Rath S, Liebl J, Furst R, Vollmar AM and
Zahler S: Regulation of endothelial signaling and migration by
v-ATPase. Angiogenesis. 17:587–601. 2014. View Article : Google Scholar
|
|
261
|
Hernandez SL, Banerjee D, Garcia A,
Kangsamaksin T, Cheng WY, Anastassiou D, Funahashi Y,
Kadenhe-Chiweshe A, Shawber CJ, Kitajewski JK, et al: Notch and
VEGF pathways play distinct but complementary roles in tumor
angiogenesis. Vasc Cell. 5:172013. View Article : Google Scholar : PubMed/NCBI
|
|
262
|
Jin Y, Kaluza D and Jakobsson L: VEGF,
Notch and TGFβ/BM Ps in regulation of sprouting angiogenesis and
vascular patterning. Biochem Soc Trans. 42:1576–1583. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
263
|
Chintala H, Krupska I, Yan L, Lau L, Grant
M and Chaqour B: The matricellular protein CCN1 controls retinal
angiogenesis by targeting VEGF, Src homology 2 domain phosphatase-1
and Notch signaling. Development. 142:2364–2374. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
264
|
Kiec-Wilk B, Grzybowska-Galuszka J, Polus
A, Pryjma J, Knapp A and Kristiansen K: The MAPK-dependent
regulation of the Jagged/Notch gene expression by VEGF, bFGF or
PPAR gamma mediated angiogenesis in HUVEC. J Physiol Pharmacol.
61:217–225. 2010.PubMed/NCBI
|
|
265
|
Cao L, Arany PR, Wang YS and Mooney DJ:
Promoting angio-genesis via manipulation of VEGF responsiveness
with notch signaling. Biomaterials. 30:4085–4093. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
266
|
Thurston G and Kitajewski J: VEGF and
Delta-Notch: Interacting signalling pathways in tumour
angiogenesis. Br J Cancer. 99:1204–1209. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
267
|
Zechariah A, ElAli A, Doeppner TR, Jin F,
Hasan MR, Helfrich I, Mies G and Hermann DM: Vascular endothelial
growth factor promotes pericyte coverage of brain capillaries,
improves cerebral blood flow during subsequent focal cerebral
ischemia, and preserves the metabolic penumbra. Stroke.
44:1690–1697. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
268
|
Yang JP, Liu HJ and Liu XF: VEGF promotes
angiogenesis and functional recovery in stroke rats. J Invest Surg.
23:149–155. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
269
|
Dzietko M, Derugin N, Wendland MF, Vexler
ZS and Ferriero DM: Delayed VEGF treatment enhances angiogenesis
and recovery after neonatal focal rodent stroke. Transl Stroke Res.
4:189–200. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
270
|
Lee HJ, Kim KS, Park IH and Kim SU: Human
neural stem cells over-expressing VEGF provide neuroprotection,
angiogenesis and functional recovery in mouse stroke model. PLoS
One. 2:e1562007. View Article : Google Scholar : PubMed/NCBI
|
|
271
|
Esposito E, Hayakawa K, Maki T, Arai K and
Lo EH: Effects of postconditioning on neurogenesis and angiogenesis
during the recovery phase after focal cerebral ischemia. Stroke.
46:2691–2694. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
272
|
Oh TW, Park KH, Jung HW and Park YK:
Neuroprotective effect of the hairy root extract of Angelica gigas
NAKAI on transient focal cerebral ischemia in rats through the
regulation of angiogenesis. BMC Complement Altern Med. 15:1012015.
View Article : Google Scholar : PubMed/NCBI
|
|
273
|
Duan S, Shao G, Yu L and Ren C:
Angiogenesis contributes to the neuroprotection induced by
hyperbaric oxygen preconditioning against focal cerebral ischemia
in rats. Int J Neurosci. 125:625–634. 2015. View Article : Google Scholar
|
|
274
|
Hayward NM, Yanev P, Haapasalo A,
Miettinen R, Hiltunen M, Grohn O and Jolkkonen J: Chronic
hyperperfusion and angiogenesis follow subacute hypoperfusion in
the thalamus of rats with focal cerebral ischemia. J Cereb Blood
Flow Metab. 31:1119–1132. 2011. View Article : Google Scholar :
|
|
275
|
Guo F, Lv S, Lou Y, Tu W, Liao W, Wang Y
and Deng Z: Bone marrow stromal cells enhance the angiogenesis in
ischaemic cortex after stroke: Involvement of notch signalling.
Cell Biol Int. 36:997–1004. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
276
|
Dao M, Tate CC, McGrogan M and Case CC:
Comparing the angiogenic potency of naïve marrow stromal cells and
Notch-transfected marrow stromal cells. J Transl Med. 11:812013.
View Article : Google Scholar
|
|
277
|
Lähteenvuo JE, Lähteenvuo MT, Kivelä A,
Rosenlew C, Falkevall A, Klar J, Heikura T, Rissanen TT, Vähakängas
E, Korpisalo P, et al: Vascular endothelial growth factor-B induces
myocardium-specific angiogenesis and arteriogenesis via vascular
endothelial growth factor receptor-1- and neuropilin
receptor-1-dependent mechanisms. Circulation. 119:845–856. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
278
|
Semenza GL: Vasculogenesis, angiogenesis,
and arteriogenesis: Mechanisms of blood vessel formation and
remodeling. J Cell Biochem. 102:840–847. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
279
|
Carmeliet P: Mechanisms of angiogenesis
and arteriogenesis. Nat Med. 6:389–395. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
280
|
Buschmann I and Schaper W: Arteriogenesis
Versus Angiogenesis: Two Mechanisms of Vessel Growth. News Physiol
Sci. 14:121–125. 1999.
|
|
281
|
Rangarajan A, Talora C, Okuyama R, Nicolas
M, Mammucari C, Oh H, Aster JC, Krishna S, Metzger D, Chambon P, et
al: Notch signaling is a direct determinant of keratinocyte growth
arrest and entry into differentiation. EMBO J. 20:3427–3436. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
282
|
Gunaratne A, Chan E, El-Chabib TH, Carter
D and Di Guglielmo GM: aPKC alters the TGFβ response in NSCLC cells
through both Smad-dependent and Smad-independent pathways. J Cell
Sci. 128:487–498. 2015. View Article : Google Scholar
|
|
283
|
Wang Y, Pan L, Moens CB and Appel B:
Notch3 establishes brain vascular integrity by regulating pericyte
number. Development. 141:307–317. 2014. View Article : Google Scholar :
|
|
284
|
Blasi F, Wei Y, Balkaya M, Tikka S,
Mandeville JB, Waeber C, Ayata C and Moskowitz MA: Recognition
memory impairments after subcortical white matter stroke in mice.
Stroke. 45:1468–1473. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
285
|
Yao H, Duan M, Hu G and Buch S:
Platelet-derived growth factor B chain is a novel target gene of
cocaine-mediated Notch1 signaling: Implications for HIV-associated
neurological disorders. J Neurosci. 31:12449–12454. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
286
|
Manda VK, Mittapalli RK, Geldenhuys WJ and
Lockman PR: Chronic exposure to nicotine and saquinavir decreases
endothelial Notch-4 expression and disrupts blood-brain barrier
integrity. J Neurochem. 115:515–525. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
287
|
Nakatsu MN, Sainson RC, Aoto JN, Taylor
KL, Aitkenhead M, Pérez-del-Pulgar S, Carpenter PM and Hughes CC:
Angiogenic sprouting and capillary lumen formation modeled by human
umbilical vein endothelial cells (HUVEC) in fibrin gels: The role
of fibroblasts and Angiopoietin-1. Microvasc Res. 66:102–112. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
288
|
Li Z, Wang J, Zhao C, Ren K, Xia Z, Yu H
and Jiang K: Acute Blockage of Notch signaling by DAPT induces
neuroprotection and Neurogenesis in the Neonatal rat brain after
stroke. Transl Stroke Res. 7:132–140. 2016. View Article : Google Scholar
|
|
289
|
Marumo T, Takagi Y, Muraki K, Hashimoto N,
Miyamoto S and Tanigaki K: Notch signaling regulates
nucleocytoplasmic Olig2 translocation in reactive astrocytes
differentiation after ischemic stroke. Neurosci Res. 75:204–209.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
290
|
Shimada IS, Borders A, Aronshtam A and
Spees JL: Proliferating reactive astrocytes are regulated by
Notch-1 in the peri-infarct area after stroke. Stroke.
42:3231–3237. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
291
|
Uyttendaele H, Closson V, Wu G, Roux F,
Weinmaster G and Kitajewski J: Notch4 and Jagged-1 induce
microvessel differentiation of rat brain endothelial cells.
Microvasc Res. 60:91–103. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
292
|
Xiao MJ, Han Z, Shao B and Jin K: Notch
signaling and neuro-genesis in normal and stroke brain. Int J
Physiol Pathophysiol Pharmacol. 1:192–202. 2009.
|
|
293
|
Carlén M, Meletis K, Göritz C, Darsalia V,
Evergren E, Tanigaki K, Amendola M, Barnabe-Heider F, Yeung MS,
Naldini L, et al: Forebrain ependymal cells are Notch-dependent and
generate neuroblasts and astrocytes after stroke. Nat Neurosci.
12:259–267. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
294
|
Yang T, Liu LY, Ma YY and Zhang W: Notch
signaling-mediated neural lineage selection facilitates
intrastriatal transplantation therapy for ischemic stroke by
promoting endogenous regeneration in the hippocampus. Cell
Transplant. 23:221–238. 2014. View Article : Google Scholar
|
|
295
|
Yasuhara T, Matsukawa N, Hara K, Maki M,
Ali MM, Yu SJ, Bae E, Yu G, Xu L, McGrogan M, et al: Notch-induced
rat and human bone marrow stromal cell grafts reduce ischemic cell
loss and ameliorate behavioral deficits in chronic stroke animals.
Stem Cells Dev. 18:1501–1514. 2009. View Article : Google Scholar : PubMed/NCBI
|