Introduction
According to GLOBOCAN, cancer is the first cause of
mortality worldwide, accounting for 14.1 million new cancer cases
reported in 2012, and 8.2 million of deaths induced by cancer. One
of the leading causes of the high cancer mortality is the
limitation of actual treatments based on drugs and radiation. These
limitations include lack of specificity, reduced drug
bioavailability, drug rapid blood clearance, poor drug solubility,
patient resistance and disease relapse. The most efficient
treatment for cancer is the use of chemotherapeutics (1). These substances, like cisplatin or
taxol, have been preferred from other therapies because they have a
superior efficiency of killing cancer cells preferentially by
inhibiting replication or inducing apoptosis (1,2).
Chemotherapeutics with anthracyclines and cyclophosphamide causes
serious side effects in patients killing healthy cells (3) and tissues like bone marrow,
epithelial cells, and hair follicles (4). Hence, there seems to be an urgent
need for developing more efficient treatments that may offer fewer
side-effects in comparison to the actual therapies (5). New technologies for cancer treatment
have been developed in the recent years based on the research and
application of nanotechnology and molecular biology (1,2).
Nanotechnology can be defined as the design,
fabrication, and use of nanoparticles, which are structures
generally from 10 to 100 nm. At least one of its dimensions must be
in the nanometer scale in order to be considered a nanomaterial.
Thus, nanomaterials may be formed from a few hundred to millions of
atoms. Because of their small size, the properties of nanoparticles
vary from their bulk form. This has been linked to various effects
such as the high percentage of atoms on the surface, high surface
free energy of nanomaterials, spatial confinement and fewer
imperfections in their structure. Different chemical, physical and
biological characteristics, unique in nanoparticles, can be
controlled during their synthesis to be used in biomedical
applications. These physicochemical properties such as size, shape,
surface charge, hydrophilic interactions, magnetism, and electrical
characteristics as well as the lack of ability to produce an
undesirable immunological reaction, make nanoparticles a promising
tool in medicine (6). A special
condition that makes nanotechnology so attractive for medicine is
the capacity to target the therapy directly to the cancerous tissue
without affecting healthy tissues, avoiding side effects. This
characteristic can be possible because some cancer cells, such as
those from breast, ovary, prostate, pancreas, lung and liver
express exclusively, or over-express on the surface some protein
receptors (like growth factor receptors or hormone receptors) which
can be identified by individual elements added to the nanoparticles
(4). In cancer therapy, the
research of nanoparticles for killing cancer cells includes a
variety of applications such as drug delivery (1), photodynamic therapy (5), protein delivery (7) and hyperthermia (8). A recent application of nanoparticles
in cancer treatment is their improvement in gene therapy as a
non-viral vector, loading nucleic acids in their surface and taking
it directly inside the target cell nucleus (9).
The development of molecular biology allows the
application of nucleic acid manipulation techniques in the
treatment of several genetic diseases like cancer. Gene therapy
consists in the transfer of genetic material into a target cell
nucleus for therapeutic issues with relatively minimal side
effects. This genetic material could be DNA or RNA, the complete
gene sequence, gene segments or an oligonucleotide. Based on the
different cancer gene therapy objectives, there are two main
classifications: Corrective cancer gene therapy or death-induced
gene therapy. Corrective cancer gene therapy deactivates oncogenes
or activates non-functioning tumor suppressor genes, restoring the
normal cellular functions. Death-induced gene therapy does not aim
for turning cancer cells into healthy cells, instead it aims for
the complete death of the cancerous tissues by the activation of
different cellular pathways (9).
Death-induced gene therapy
The objective of this kind of treatment is to
produce the death of cancerous cells by the expression of genes
whose protein product activates different death pathways. These
genes usually are administrated in pDNA (9). Plasmids are double-stranded circular
DNA molecules, and they can be replicated and transcribed once
inside the target nucleus cell (10). The commercial plasmid used in this
application needs to have in their sequence an eukaryotic
transcription promoter, such as CMV, SV40, AFPS or AFPL, to be able
to work in mammal cells. With the use of modifier enzymes of DNA,
such as restriction enzymes and ligases, the sequence of the
death-inducing gene can be added to the commercial plasmid. For
monitoring the expression of the recombinant plasmid, a reporter
gene sequence can be inserted into the plasmid. The protein
products of reporter genes, like green fluorescent protein (GFP) or
luciferase, can be easily quantified by a colorimetric method
(11–13). There are several advantages of
using pDNA vs. the protein itself; the purification of pDNA is less
expensive and time-consuming even at large scale, plasmid does not
trigger an immune response, and tumor cells do not develop
resistance to the pDNA. There are three principal ways to induce
cell death in gene therapy: a) apoptosis-induced, b) toxin-induced
and c) gene-directed enzyme prodrug therapy (GDEPT) (4).
a) Apoptosis-induced cancer
therapy
A strategy in death-induced gene therapy is the
expression of apoptotic genes. Genes expressed by cancer cells
without affecting healthy tissues are preferred (4). Examples of these genes are H19
(encodes a long non-coding tumor suppressor RNA), CEA (encodes a
cell surface glycoprotein which regulates differentiation,
apoptosis, and cell polarity), and UPAR (encodes the receptor for
urokinase plasminogen activator, activating the degradation of the
extracellular matrix) (14).
Another important commonly studied gene is TRAIL, which encodes a
cytokine that belongs to the tumor necrosis factor (TNF) ligand
family. This protein induces apoptosis preferentially in cancer
cells (14–15). Other genes members of the
TNF-receptor family expressed in normal apoptosis process are FAS
(encodes induced cellular death), FASL (encodes a membrane protein
for the induction of apoptosis), and BCL2 (encodes a membrane
receptor for tumor necrosis factor α) (14,16).
b) Toxin-induced gene therapy
Toxins are chemical substances that interact with
the cells at molecular levels, with biological macromolecules such
as enzymes and cellular receptors, causing toxic effects. These
toxins are found in some microbes, plants, and animals as part of
their defensive or predation strategies. Different toxins have been
proposed for biomedical applications in Toxin-induced gene therapy
(17). Examples of bacterial
toxins studied for cancer treatment are diphtheria toxin (DT) from
Corynebacterium diphtheria, whose toxin inhibits the protein
synthesis by inactivating eukaryotic elongation factor-2 (EF-2);
exotoxin A (ETA) from Pseudomonas aeruginosa which inhibits
the protein synthesis by the inhibition of EF-2 as well;
streptolysin O (SLO) from Streptococcus which binds to
membrane cholesterol and oligomerizes making a large pore; and
membrane protein product of gef gene from Escherichia
coli which induces arrest of cellular respiration. An example
of an animal toxin that can be used in gene therapy against cancer
is the melittin (Mel), an apoptosis inducer from bee poison
(18). Some plant toxin genes can
be utilized for cancer treatment too. The most common are the
ribosome-inactivating proteins (RIPs) which have a rRNA
N-glycosidase activity. They cleave the bond between adenine and
ribose (depurination) in the 28S rRNA, blocking the recruitment of
translation elongation factors and the protein synthesis (19). Some examples of RIPs are ricin from
the seeds of Ricinus communis, saporin from Saponaria
officinalis, and lunasin from Glycine max seeds
(20).
c) GDEPT
It consists of the administration of an
enzyme-coding gene in combination with a prodrug. The enzyme is a
non-toxic protein, but their binding with the prodrug turns the
complex into a toxic compound (21). An example of the most common GDEPT
system is the TK/GCV suicide system. This system is constituted of
two elements: the transfection with the gene that codifies the
enzyme herpes simplex virus thymidine kinase (HSV-Tk) and the
prodrug ganciclovir (GCV). The enzyme HSV-Tk is a non-toxic
product, but once the GCV interacts with the HSV-Tk, it turns into
ganciclovir triphosphate (GCV-3P). GCV-3P is a toxic compound which
inhibits DNA polymerase causing death (22). Other systems are: cytosine
deaminase/5-fluorocytosine system, which inhibits thymidylate
synthase; nitroreductase/CB1954 system, which triggers an extensive
DNA damage; and carboxypeptidase G2/nitrogen mustard system, which
links with DNA (23).
Vectors for DNA transfection
The transfection with the selected gene can be
possible transferring naked pDNA into target cells, but there are
some disadvantages of this method. First, the pDNA exhibits a short
half-life time in blood circulation because of the degradation
caused by circulating and intracellular nucleases. The rapid
clearance makes it unavailable for intravenous application in the
clinic (24). Another pDNA
property is the high negative charge, same as the cell membrane,
decreasing the transfection effectiveness by simple passive
diffusion because of the repellency of the electric charges. These
disadvantages can be improved by the use of a vector. A vector is a
structure that works as a vehicle, carrying DNA through blood
circulation, distributing it until it is endocytosed by a cell.
There are two main types of vectors: viral vectors and non-viral
vectors as shown in Fig. 1
(25). Principles of action of
non-viral vectors can be classified into physical and chemical
mechanisms (26).
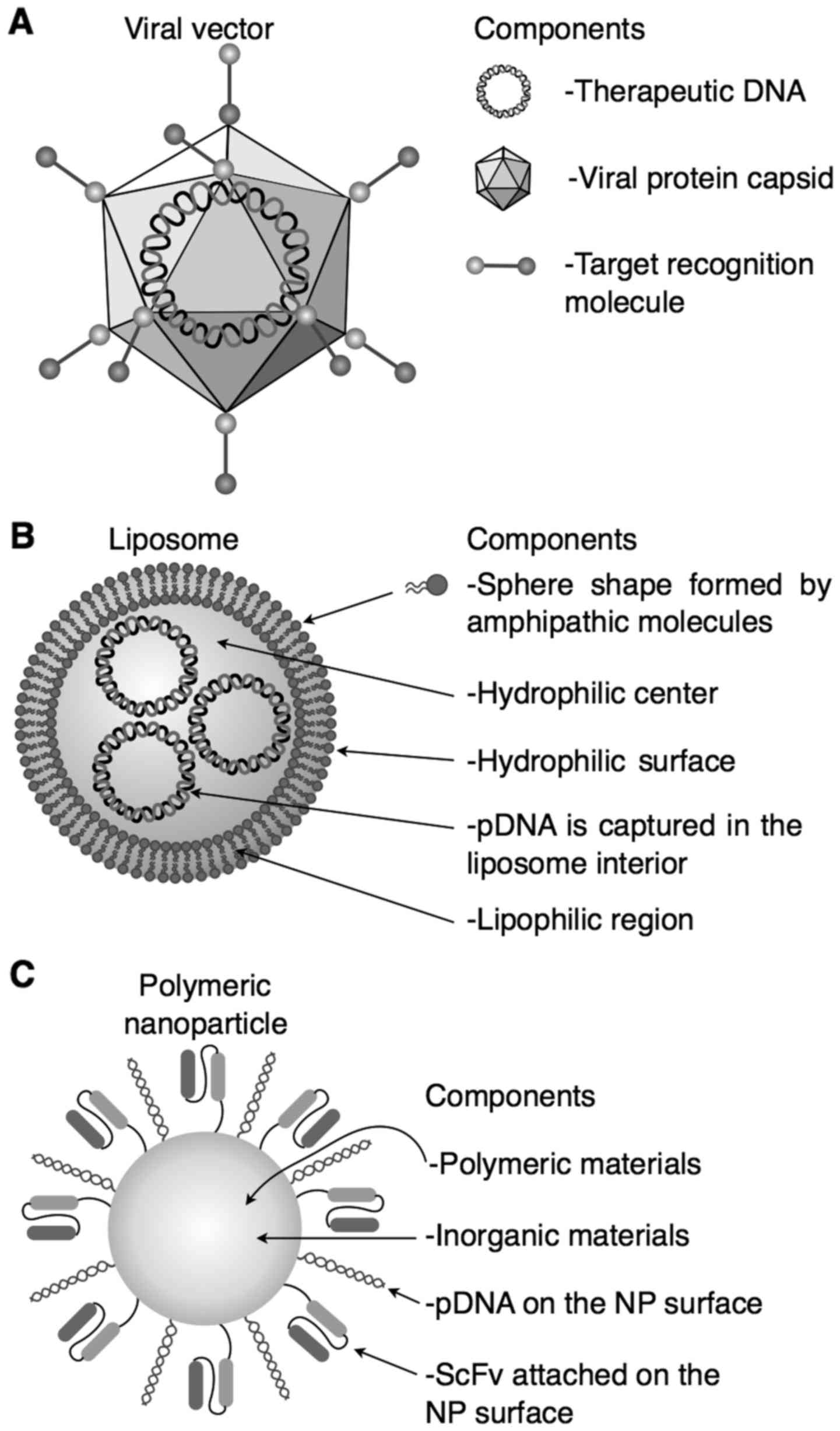 | Figure 1.Viral (A) and non-viral (B and C)
vectors commonly used for therapy and their principal components
are described. (A) Viral vector. It has excellent transfection
efficiency, but triggers an antiviral immune response. (B)
Liposome. It has an excellent in vitro transfection
efficiency, but in vivo is an inefficient system because the
high surface charges, and (C) polymeric nanoparticle. It is stable,
sterilizable, with scale-up reproducibility, the biodegradation is
controllable, it has unique physical properties, low
immunigenicity, and it is non-toxic; but it has low
biocompatibility in some inorganic components, the cost of
production is expensive, and obtaining of recognition molecules is
difficult. ScFv, single-stranded variable fragment; NP,
nanoparticle. |
The structure of a virus consists of a genome
surrounded by a protein coat. For viral replication, the virus
needs to insert its genetic material into a host cell to use the
host replication and transcription machine. A virus can be
genetically modified in order to make it harmless, changing their
infectious genome for the therapeutic genetic material. Because of
their transfection efficiency viral vectors are the most commonly
studied vectors for gene therapy. Viruses that can be modified for
their use in gene therapy are adenovirus, herpes simplex virus,
vaccinia virus, retrovirus, lentivirus, adeno-associated virus, and
reovirus. The transfection efficiency of viruses is superior from
other methods, but there are several disadvantages like immune
recognition for most of the viruses, mutagenic interaction of
retrovirus and lentivirus, and inflammatory toxicity of adenovirus
(22,27).
An alternative from the use of potentially dangerous
viral vectors are those based on physical or chemical mechanisms.
Vectors based on physical methods such as hydrodynamic methods,
gene gun, and electroporation have good results ex vivo, but
in vivo protocols need to be developed to increase their
transfection efficiency, avoiding risk for the patient (22). Nanoparticles are vectors based on
chemical principles and they have several properties that make them
a promising vector for pDNA transfection. An essential
characteristic that makes nanoparticles available for gene
transfection is a cationic surface charge. pDNA have a high
negative charge, so nanoparticles must have positive surface
charges to generate electrostatic interactions with pDNA. pDNA and
cationic nanoparticles interact by simple contact, and the pDNA is
loaded on the surface of the nanoparticles creating a complex. This
complex is easily taken inside the cell in comparison to naked pDNA
because the cellular membrane is negatively charged and there is no
electrostatic repulsion. Another characteristic of pDNA bound to
nanoparticles is the protection against degradation by blood
nucleases, improving the bioavailability of the therapeutic agent
(28,29).
Nanoparticles for pDNA transfection
Even when there is a background about the use of
nanoparticles in the clinic, there are no clinical protocols
approved for death-induced gene therapy. Since 1995, the Food and
Drug Administration (FDA) has approved the use of some
nanoparticles by oral, local, topical and systemic administration
for cancer treatment. These are structures that carry a
chemotherapeutic agent all over the patient's body (30). Despite the lack of clinical studies
of nanoparticles carrying nucleic acids as therapeutic agents,
there are several pieces of evidence of the preclinical efficiency
of these nanosystems in gene therapy (31–34).
Companies have a commercial interest in the production of
nanoparticles, because of their easy large-scale production. The
most efficient techniques for scale-up production of pharmaceutical
grade nanoparticles are emulsification solvent diffusion and
nanoprecipitation. These techniques allow the formation of
particles with a uniform and controlled size (35).
Nanoparticles used as nucleic acid carriers include
liposomes, inorganic nanoparticles, and polymeric nanoparticles.
Liposomes are self-assembled vesicles formed by lipid bilayer
membranes of amphiphilic phospholipids. Liposomes have an excellent
in vitro transfection capacity; however, they have some
deficiencies in vivo, due to their high positive charge,
which induces hemotoxicity, caused by the aggregates produced due
to the interaction between liposomes and blood proteins. Also,
these aggregates promote a low transfection rate. Other problems
with liposomes are low stability, industrial production, and
difficulties in sterilization. In contrast, polymeric and inorganic
nanoparticles can be easily modified to reduce the excesses of
positive charges; they are more stable, more reproducible and
easily sterilizable (36,37). For this reason, polymeric and
inorganic nanoparticles are considered by several authors as
promising vectors for gene transfection (38,39).
Currently, biodegradable cationic polymer
nanoparticles are the most studied. These systems are
submicron-sized colloidal particles. The properties of the
nanoparticles depend on their composition, solubility,
crystallinity, molecular weight, backbone stability, hydrophobicity
and polydispersity of the selected polymer. It is critical that the
polymers selected for the preparation of nanoparticles are
biocompatible and biodegradable. The advantage of using
biodegradable polymers is its controlled degradation. This
degradation releases the plasmid into the cellular cytoplasm once
the nanosystem is inside the cell. Polymeric nanoparticles also
protect nucleic acids from nuclease degradation. The cationic
property allows the binding of the nucleic acid into the
nanoparticle surface. Natural and synthetic polymers can be used
for gene transfection (40,41).
Some examples of natural polymers used in gene therapy are
polysaccharides such as chitosan (41,42)
and alginate (43), or proteins
like albumin (44). Also,
synthetic polymers can be employed, such as polycaprolactone (PLC)
(45), polylactic acid (PLA)
(46), polyethyleneimine (PEI)
(47), poly (lactic-co-glycolic
acid) (PLGA) (46,48) or polyethylene glycol (PEG)
(49).
Inorganic nanoparticles can be easily manipulated in
size, shape, composition and chemical properties. They are easy to
prepare in large-scale, easily functionalized and have a high
degree of transfection. They have various physical properties such
as electrical, magnetic and optical properties, which can be
modified during synthesis. These properties can be manipulated to
obtain optimal conditions to improve transfection efficiency
(35). Examples of this type of
nanoparticles are carbon nanotubes (50), calcium phosphate nanoparticles
(51), gold (13), silica (52), and magnetic nanoparticles (53). For the use of magnetic
nanoparticles such as magnetite, once the nanosystem is inside the
patient's circulatory system, an external electromagnetic field can
be applied. In gene therapy, the process of directing nucleic acids
with the support of external magnetic fields is known as
magnetofection. The efficiency of transfection increases at the
moment of applying an external electromagnetic field, because it
accelerates the absorption on the cellular surface (54).
Properties of nanoparticles
Because the nanosystem is a foreign body to the
organism, it must be designed with some unique properties to avoid
immediate recognition of the immune system, and be quickly
eliminated from circulation. The first molecular event required to
discard the nanosystem from circulation is opsonization, which is
the binding of serum proteins to the surface of the nanosystem.
This binding is produced by the physicochemical proprieties of the
nanoparticles, such as the electrostatic charge on the nanoparticle
surface. Once the nanoparticle is coated by these proteins,
including complement proteins and immunoglobulins, the immune
system can recognize the complex and eliminate it from circulation.
That is why specific properties are required to avoid the
elimination of the nanosystem from the body and to ensure its
internationalization into the cell. We describe these proprieties
briefly (55).
i) Shape. Spherical shapes are
preferred from sharp corners
Some nanoparticles like nanocubes tend to damage
smaller blood vessels and capillaries (55).
ii) Size. The usual scale of spherical
structures is from 30 to 150 nm
Nanoscale size is required for the system to cross
biological barriers, like epithelia. Nanoparticles smaller than 150
nm are capable of performing this action. These dimensions are also
optimal to be endocyted by the target cells. However, if the size
is lower than 30 nm, the nanoparticles are immediately discarded by
the kidneys and liver from the circulatory system. At dimensions
bigger than 30 nm the nanoparticles cannot pass the glomerular
filtration, so they remain in the blood. At the range of 30 to 150
nm, the blood circulation time is relative to the size; smaller
size has a longer circulation time, while the larger particles are
more easily recognized by the macrophages of the immune system
(56,57).
iii) Surface electrostatic charge
Nanoparticle surface charge has to be positive to
interact with negative charges over the surface of cell membranes.
The Z potential of the nanoparticles (a measure used to
characterize surface charges) must be around +25 mV to have stable
nanoparticles. Nanoparticles in this range of Z potential do not
form aggregates due to the electrostatic repulsion generated
between nanoparticles. Nanoparticles with a very low Z potential
tends to form aggregates because of the lack of electrostatic
repulsion interactions. Nanoparticles with a very high Z potential
can cause problems of hemocompatibility due to the interaction
between nanoparticles and cellular components of the blood, causing
hemolysis and platelet aggregation (55,57).
iv) Water interactions
If the nanoparticles present hydrophilicity, the
chemical interactions between nanoparticles and water delay the
process of opsonization, generating a longer lifetime of the
nanoparticles in blood. The immune system rapidly discards
nanoparticles with hydrophobic properties due to its faster
opsonization (55).
v) Biodegradability
Biodegradable materials are required because the
presence of the nanoparticle in the body must not be permanent.
Biodegradable materials are eliminated once the therapeutic action
is completed, through normal excretion pathways, or they can be
integrated into the normal metabolic pathways of the cell (55,56).
Biodegradability is related to the microstructure and composition
and its interaction with the external physiological environment.
Examples of these materials include polymers, ceramics and
composites (56).
vi) Pegylation
Nanoparticles must have a positive charge to
immobilize the nucleic acids over their surface, but a high
positive charge also generates hemocompatibility problems. A
strategy widely used to eliminate the excess of positive charges is
the functionalization with PEG. PEG is a polymer with no
electrostatic charge. PEG regulates the excess of positive charges
in a nanosystem. Many authors who develop nanoparticles with
different medical applications prefer the use of PEG to improve
their physicochemical proprieties (58–61).
Targeting the nanoparticles
One of the key points to improve death-induced gene
therapy in the clinic is to avoid the expression of suicide genes
in healthy tissues. The greatest challenge of intelligent
treatments is to selectively release the therapeutic agent in
cancerous tissues; several methods are currently under
investigation. In diseases such as cancer, tumor cells usually
express certain exclusive receptor proteins, or they can
overexpress those commonly found in healthy tissues (4). Examples of these proteins are human
epidermal growth factor receptor-2 in HER2+ breast cancers
(62); mesothelin, urokinase
plasminogen activator receptor, and some growth factor receptors in
pancreatic cancer (63); the α
isoform of folate receptor in ovarian cancer (64); scavenger receptor type B-1 in
chronic lymphocytic leukemia (65); folate receptor in acute myeloid
leukemia (66); prostate-specific
membrane antigen (67) and matrix
metalloproteinase-2 in prostate cancer (68,69).
These membrane proteins have become targets for specific
recognition, easily identified by recognition molecules such as
antibodies, aptamers, proteins and peptides (70). Passive non-selective endocytosis of
nanoparticles is possible because of hydrophobic and electrostatic
interactions. To improve active endocytosis, nanoparticle surface
must be modified specifically with recognition molecules (55), immobilized on the surface of the
nanoparticles by methods like glutaraldehyde (71) or carbodiimide (72).
The most used recognition molecules are monoclonal
antibodies (mAbs). Antibodies are proteins (~150 kDa) produced by
the immune system that have extremely high specificity and affinity
for their targeted analytes. The use of antibodies has an advantage
over the use of peptides and aptamers because of the low
immunogenicity. There is preclinical evidence of their efficiency
for recognition of targeted cells in cancer therapies. However, its
size is too big; thus, when nanoparticles are functionalized with
antibodies, the nanosystem increases its size considerably. A large
size hinders the intracellular penetration of the nanosystems into
solid tumors. Another disadvantage is the increase of
immunogenicity of the system. Finally, there is an extensive need
of optimization for scale-up manufacturing (73–75).
An alternative to this is the use of single-stranded variable
fragments (scFv). The antibody contains two Fab fragments which
also have a similar specificity compared to complete protein. scFv
are 27 kDa molecular weight fragments from the fusion of two chains
from the variable regions of the heavy chain (VH) and the light
chain (VL) of the antibody, joined by a peptide of 10 to 25
aminoacids. There are in vivo studies where nanoparticles
functionalized with scFv against HER2 in breast cancer can decrease
tumor size. A disadvantage of the use of MAbs and scFv is the high
cost they generate, so large scale production strategies are still
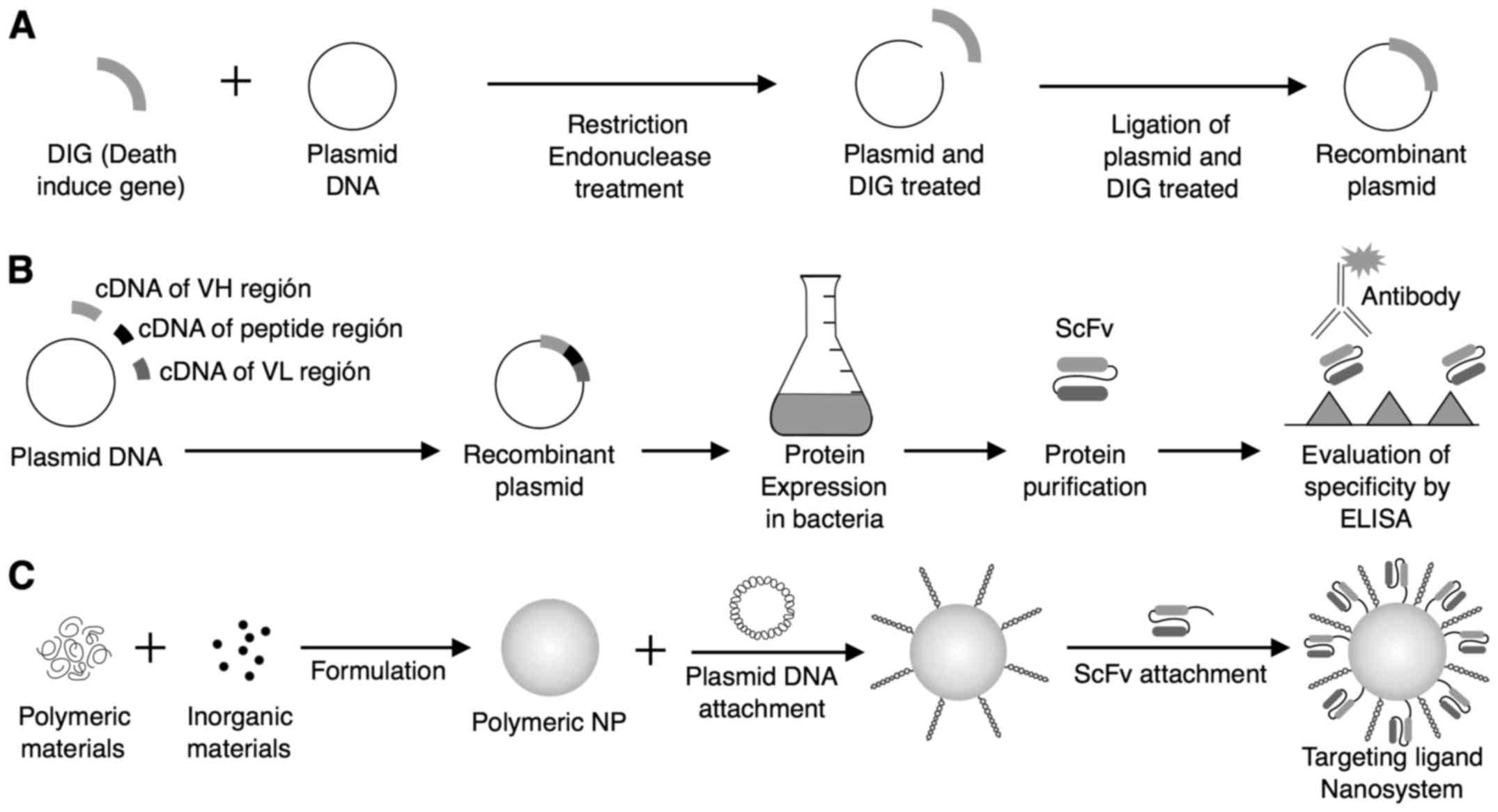
a challenge (75–78). Fig.
2 shows the preparation and construction of a nanosystem for
death-induced gene therapy using scFv along with recombinant
plasmid (71).
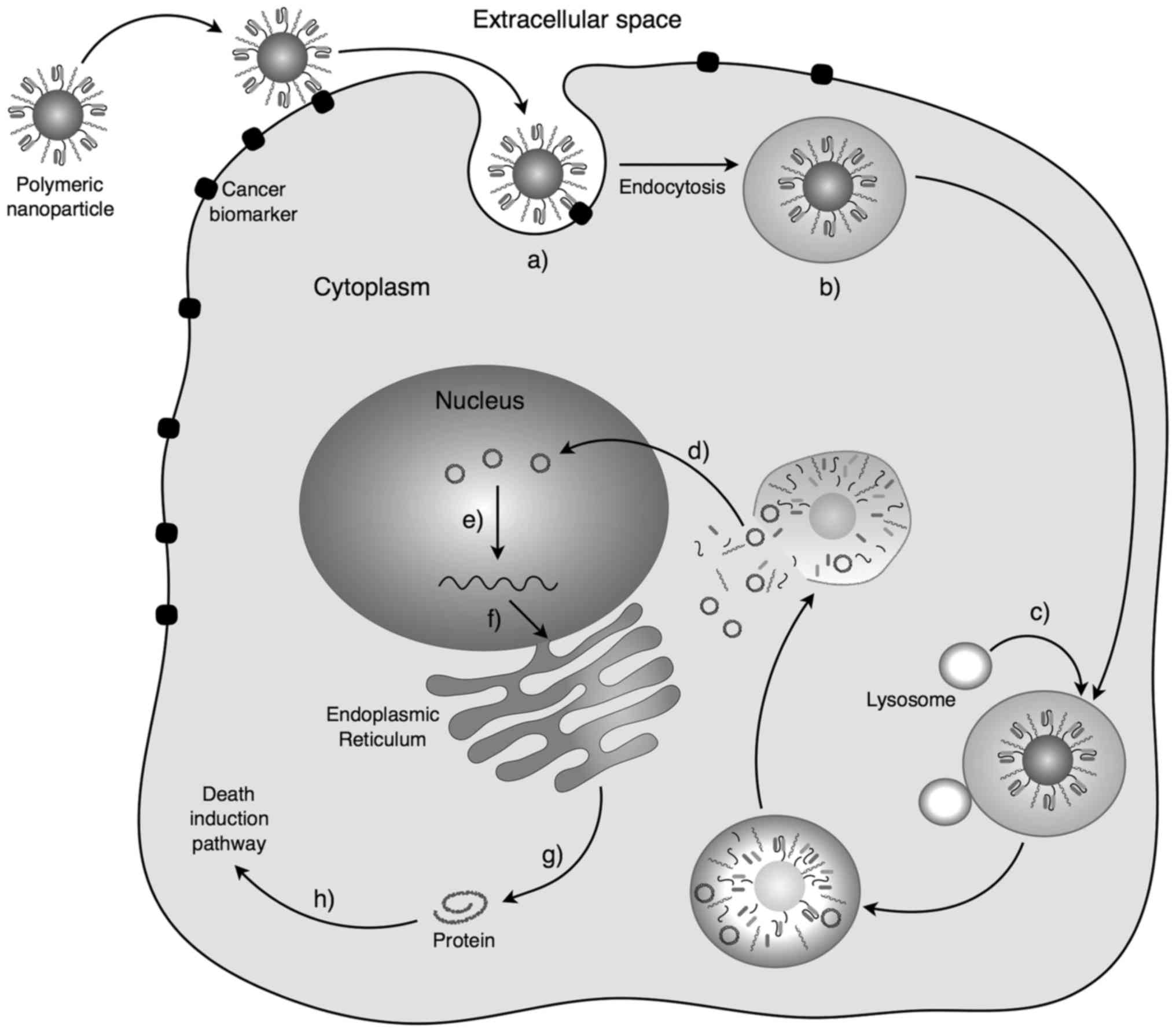
Active endocytosis in death-induced gene therapy
begins in the first instance by administration of the nanosystem,
usually intravenously. The nanosystem should recirculate through
the circulatory blood system, avoiding natural elimination
mechanisms of the host, such as opsonization and subsequent
phagocytosis by macrophages. The nanosystem has the physicochemical
properties to interact and cross the epithelium (55–56).
When the cancer tissue is located, recognition molecules generate a
specific binding to the protein receptors attached on the surface
of cancer cells. This binding is called ligand. The ligand
activates different molecular pathways conducing to endocytosis.
Ligand-mediated endocytosis increases the affinity and specificity
of the nanosystem endocytosis (Fig.
3) (4). The nanosystem is
internalized in the cell into a vesicle generated by the cell
membrane. The vesicle enters into the endosomal cellular system
where it is degraded. The nanosystem is released into the cytoplasm
where it begins to degrade and it releases all its components into
the medium. Nanosystem residues are removed from the cell by
standard excretion mechanisms, or they are incorporated into
cellular metabolic pathways depending on their nature (4,55).
The recombinant plasmid, which is the active substance, enters into
the nucleus through the nuclear pores. Once inside, it is
recognized by the transcription system, generating the mRNA of the
death-induced gene. This mRNA goes from the nucleus to the
cytoplasm, and it is translated into protein. These proteins
trigger different cellular pathways ending in cellular death
(20).
Conclusion
The use of nanotechnology in medicine promises to be
a valuable tool in the clinic in the future, both in diagnosis and
treatment. It promises to reduce the aggressiveness, the number of
cases, and the mortality of diseases like cancer. There are some
nanoparticles already approved in the clinic by international
organizations, but all of them work as a vehicle of a drug. There
are other promising applications of nanoparticles such as
hyperthermia or photodynamic therapy. In this review, we showed a
less known promising application of nanoparticles in medicine: as a
vector for death induction by gene therapy. Because of their
chemical, physical and biological properties, nanoparticles are
superior as a nucleic acid carrier than viruses or other physical
transfection methods. Death-induced gene therapy bet on the total
elimination of cancerous cells, so the challenge of this therapy is
to produce the death only in cancerous tissues and not in the
healthy ones. Despite this, preclinical in vitro and in
vivo evidence shows that there is possible to transfect cancer
cells exclusively with the death-induced gene. This kind of genes
produces different types of proteins which interact with cancerous
cells, triggering the process of apoptosis. Toxic proteins cannot
be administrated directly to the patient because the body readily
degrades proteins. Nanoparticles systems with pDNA are more stable
than proteins, are easily constructed and easily modified to obtain
the necessary properties for their use in a patient.
There are still many challenges in the building of
the transfection vector with the perfect properties of size,
surface charge, shape, biodegradation rate, biocompatibility,
stability, scale-up construction, easiness of sterilization,
avoidance of opsonization, biodistribution, and specificity. Thanks
to their versatility, nanoparticles have demonstrated to be a
suitable candidate as a pDNA carrier. Perfecting the design of
different nanoparticles, recognition-molecules and death-inductor
genes could lead death-induced gene therapy systems into the
clinical application.
References
|
1
|
Qin SY, Zhang AQ, Cheng SX, Rong L and
Zhang XZ: Drug self-delivery systems for cancer therapy.
Biomaterials. 112:234–247. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sun NF, Liu ZA, Huang WB, Tian AL and Hu
SY: The research of nanoparticles as gene vector for tumor gene
therapy. Crit Rev Oncol Hematol. 89:352–357. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
van Ramshorst MS, van Werkhoven E, Honkoop
AH, Dezentjé VO, Oving IM, Mandjes IA, Kemper I, Smorenburg CH,
Stouthard JM, Linn SC, et al: Toxicity of dual HER2-blockade with
pertuzumab added to anthracycline versus non-anthracycline
containing chemotherapy as neoadjuvant treatment in HER2-positive
breast cancer: The TRAIN-2 study. Breast. 29:153–159. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Vago R, Collico V, Zuppone S, Prosperi D
and Colombo M: Nanoparticle-mediated delivery of suicide genes in
cancer therapy. Pharmacol Res. 111:619–641. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Banerjee SM, MacRobert AJ, Mosse CA,
Periera B, Bown SG and Keshtgar MR: Photodynamic therapy: Inception
to application in breast cancer. Breast. 31:105–113. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Sanchez-Dominguez CN, Gallardo-Blanco HL,
Rodriguez-Rodriguez AA, Vela-Gonzalez AV and Sanchez-Dominguez M:
Nanoparticles vs cancer: A multifuncional tool. Curr Top Med Chem.
14:664–675. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yu M, Wu J, Shi J and Farokhzad OC:
Nanotechnology for protein delivery: Overview and perspectives. J
Control Release. 240:24–37. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Beik J, Abed Z, Ghoreishi FS,
Hosseini-Nami S, Mehrzadi S, Shakeri-Zadeh A and Kamrava SK:
Nanotechnology in hyperthermia cancer therapy: From fundamental
principles to advanced applications. J Control Release.
235:205–221. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Amer MH: Gene therapy for cancer: Present
status and future perspective. Mol Cell Ther. 2:272014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tolmasky ME: Plasmids. Reference Module in
Life Sciences: Elsevier. 2017. View Article : Google Scholar
|
|
11
|
Fang CY, Tsai YD, Lin MC, Wang M, Chen PL,
Chao CN, Huang YL, Chang D and Shen CH: Inhibition of human bladder
cancer growth by a suicide gene delivered by JC polyomavirus
virus-like particles in a mouse model. J Urol. 193:2100–2106. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kim HA, Nam K, Lee M and Kim SW:
Hypoxia/hepatoma dual specific suicide gene expression plasmid
delivery using bio-reducible polymer for hepatocellular carcinoma
therapy. J Control Release. 171:1–10. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lazarus GG and Singh M: In vitro cytotoxic
activity and transfection efficiency of polyethyleneimine
functionalized gold nanoparticles. Colloids Surf B Biointerfaces.
145:906–911. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pruitt KD, Brown GR, Hiatt SM,
Thibaud-Nissen F, Astashyn A, Ermolaeva O, Farrell CM, Hart J,
Landrum MJ, McGarvey KM, et al: RefSeq: An update on mammalian
reference sequences. Nucleic Acids Res. 42:(Database Issue).
D756–D763. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Luo C, Miao L, Zhao Y, Musetti S, Wang Y,
Shi K and Huang L: A novel cationic lipid with intrinsic antitumor
activity to facilitate gene therapy of TRAIL DNA. Biomaterials.
102:239–248. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Inoue N, Watanabe M, Ishido N, Kodu A,
Maruoka H, Katsumata Y, Hidaka Y and Iwatani Y: Involvement of
genes encoding apoptosis regulatory factors (FAS, FASL, TRAIL,
BCL2, TNFR1 and TNFR2) in the pathogenesis of autoimmune thyroid
diseases. Hum Immunol. 77:944–951. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhan C, Li C, Wei X and Lu W and Lu W:
Toxins and derivatives in molecular pharmaceutics: Drug delivery
and targeted therapy. Adv Drug Deliv Rev. 90:101–118. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Glinka EM: Eukaryotic expression vectors
bearing genes encoding cytotoxic proteins for cancer gene therapy.
Plasmid. 68:69–85. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Walsh MJ, Dodd JE and Hautbergue GM:
Ribosome-inactivating proteins: Potent poisons and molecular tools.
Virulence. 15:774–784. 2013. View Article : Google Scholar
|
|
20
|
Glinka EM: Eukaryotic expression vectors
containing genes encoding plant proteins for killing of cancer
cells. Cancer Epidemiol. 37:1014–1019. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Malekshah OM, Chen X, Nomani A, Sarkar S
and Hatefi A: Enzyme/prodrug systems for cancer gene therapy. Curr
Pharmacol Rep. 2:299–308. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Duarte S, Carle G, Faneca H, de Lima MC
and Pierrefite-Carle V: Suicide gene therapy in cancer: Where do we
stand now? Cancer Lett. 324:160–170. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Karjoo Z, Chen X and Hatefi A: Progress
and problems with the use of suicide genes for targeted cancer
therapy. Adv Drug Deliv Rev. 99:113–128. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lila Abu AS, Uehara Y, Ishida T and Kiwada
H: Application of polyglycerol coating to plasmid DNA lipoplex for
the evasion of the accelerated blood clearance phenomenon in
nucleic acid delivery. J Pharm Sci. 103:557–566. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Badrinath N, Heo J and Yoo SY: Viruses as
nanomedicine for cancer. Int J Nanomedicine. 11:4835–4847. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dizaj SM, Jafari S and Khosroushahi AY: A
sight on the current nanoparticle-based gene delivery vectors.
Nanoscale Res Lett. 9:2522014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Crespo-Barreda A, Encabo-Berzosa MM,
González-Pastor R, Ortíz-Teba P, Iglesias M, Serrano JL and
Duque-Martin P: Chapter 11-viral and nonviral vectors for in vivo
and ex vivo gene therapies A2-laurence, JeffreyTrans Regenerative
Med Clinic. Boston: Academic Press; pp. 155–177. 2016, View Article : Google Scholar
|
|
28
|
Zou W, Liu C, Chen Z and Zhang N:
Preparation and characterization of cationic PLA-PEG nanoparticles
for delivery of plasmid DNA. Nanoscale Res Lett. 4:982–992. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Raju D, Vishwakarma RK, Khan BM, Mehta UJ
and Ahmad A: Biological synthesis of cationic gold nanoparticles
and binding of plasmid DNA. Mater Lett. 129:159–161. 2014.
View Article : Google Scholar
|
|
30
|
Anselmo AC and Mitragotri S: Nanoparticles
in the clinic. Bioeng Trans Med. 1:10–29. 2016.
|
|
31
|
Gebremedhin S, Singh A, Koons S, Bernt W,
Konopka K and Duzgunes N: Gene delivery to carcinoma cells via
novel non-viral vectors: Nanoparticle tracking analysis and suicide
gene therapy. Eur J Pharm Sci. 60:72–79. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gao S, Tian H, Xing Z, Zhang D, Guo Y, Guo
Z, Zhu X and Chen X: A non-viral suicide gene delivery system
traversing the blood brain barrier for non-invasive glioma
targeting treatment. J Control Release. 243:357–369. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Eslaminejad T, Nematollahi-Mahani SN and
Ansari M: Synthesis, characterization and cytotoxicity of the
plasmid EGFP-p53 loaded on pullulan-spermine magnetic
nanoparticles. J Magn Magn Mater. 402:34–43. 2016. View Article : Google Scholar
|
|
34
|
McBride JW, Massey AS, McCaffrey J,
McCrudden CM, Coulter JA, Dunne NJ, Robson T and McCarthy HO:
Development of TMTP-1 targeted designer biopolymers for gene
delivery to prostate cancer. Int J Pharm. 500:144–153. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang K, Kievit FM and Zhang M:
Nanoparticles for cancer gene therapy: Recent advances, challenges,
and strategies. Pharmacol Res. 114:56–66. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Islam MA, Park TE, Singh B, Maharjan S,
Firdous J, Cho MH, Kang SK, Yun CH, Choi YJ and Cho CS: Major
degradable polycations as carriers for DNA and siRNA. J Control
Release. 193:74–89. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pérez-Herrero E and Fernández-Medarde A:
Advanced targeted therapies in cancer: Drug nanocarriers, the
future of chemotherapy. Eur J Pharm Biopharm. 93:52–79. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bishop CJ, Majewski RL, Guiriba TR, Wilson
DR, Bhise NS, Quiñones-Hinojosa A and Green JJ: Quantification of
cellular and nuclear uptake rates of polymeric gene delivery
nanoparticles and DNA plasmids via flow cytometry. Acta Biomater.
37:120–130. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Guo J, O'Driscoll CM, Holmes JD and Rahme
K: Bioconjugated gold nanoparticles enhance cellular uptake: A
proof of concept study for siRNA delivery in prostate cancer cells.
Int J Pharm. 509:16–27. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Masood F: Polymeric nanoparticles for
targeted drug delivery system for cancer therapy. Mater Sci Eng C
Mater Biol Appl. 60:569–578. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Carrillo C, Suñé JM, Pérez-Lozano P,
García-Montoya E, Sarrate R, Fàbregas A, Miñarro M and Ticó JR:
Chitosan nanoparticles as non-viral gene delivery systems:
Determination of loading efficiency. Biomed Pharmacother.
68:775–783. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bor G, Mytych J, Zebrowski J, Wnuk M and
Şanli-Mohamed G: Cytotoxic and cytostatic side effects of chitosan
nanoparticles as a non-viral gene carrier. Int J Pharm.
513:431–437. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhou J, Deng W, Wang Y, Cao X, Chen J,
Wang Q, Xu W, Du P, Yu Q, Chen J, et al: Cationic carbon quantum
dots derived from alginate for gene delivery: One-step synthesis
and cellular uptake. Acta Biomater. 42:209–219. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Tirkey B, Bhushan B, Kumar Uday S and
Gopinath P: Prodrug encapsulated albumin nanoparticles as an
alternative approach to manifest anti-proliferative effects of
suicide gene therapy. Mater Sci Eng C Mater Biol Appl. 73:507–515.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Shi S, Shi K, Tan L, Qu Y, Shen G, Chu B,
Zhang S, Su X, Li X, Wei Y and Qian Z: The use of cationic
MPEG-PCL-g-PEI micelles for co-delivery of Msurvivin T34A gene and
doxorubicin. Biomaterials. 35:4536–4547. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gaspar VM, Baril P, Costa EC, de
Melo-Diogo D, Foucher F, Queiroz JA, Sousa F, Pichon C and Correia
IJ: Bioreducible poly(2-ethyl-2-oxazoline)-PLA-PEI-SS triblock
copolymer micelles for co-delivery of DNA minicircles and
Doxorubicin. J Control Release. 213:175–191. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Peng SF, Hsu HK, Lin CC, Cheng YM and Hsu
KH: Novel PEI/Poly-γ-Gutamic Acid Nanoparticles for high efficient
siRNA and Plasmid DNA Co-Delivery. Molecules. 22:pii: E86. 2017.
View Article : Google Scholar
|
|
48
|
Cocco E, Deng Y, Shapiro EM, Bortolomai I,
Lopez S, Lin K, Bellone S, Cui J, Menderes G, Black JD, et al:
Dual-targeting nanoparticles for in vivo delivery of suicide genes
to chemotherapy-resistant ovarian cancer cells. Mol Cancer Ther.
16:323–333. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Frede A, Neuhaus B, Klopfleisch R, Walker
C, Buer J, Müller W, Epple M and Westendorf AM: Colonic gene
silencing using siRNA-loaded calcium phosphate/PLGA nanoparticles
ameliorates intestinal inflammation in vivo. J Control Release.
222:86–96. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ohta T, Hashida Y, Higuchi Y, Yamashita F
and Hashida M: In vitro cellular gene delivery employing a novel
composite material of single-walled carbon nanotubes associated
with designed peptides with pegylation. J Pharm Sci. 106:792–802.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Shekhar S, Roy A, Hong D and Kumta PN:
Nanostructured silicate substituted calcium phosphate (NanoSiCaPs)
nanoparticles-efficient calcium phosphate based non-viral gene
delivery systems. Mater Sci Eng C Mater Biol Appl. 69:486–495.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Li Y, Hei M, Xu Y, Qian X and Zhu W:
Ammonium salt modified mesoporous silica nanoparticles for dual
intracellular-responsive gene delivery. Int J Pharm. 511:689–702.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
El-Sherbiny IM, Elbaz NM, Sedki M,
Elgammal A and Yacoub MH: Magnetic nanoparticles-based drug and
gene delivery systems for the treatment of pulmonary diseases.
Nanomedicine (Lond). 12:387–402. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Sun J, Shi Z, Jia S and Zhang P: The force
analysis for superparamagnetic nanoparticles-based gene delivery in
an oscillating magnetic field. J Magn Magn Mater. 427:85–89. 2017.
View Article : Google Scholar
|
|
55
|
Sun T, Zhang YS, Pang B, Hyun DC, Yang M
and Xia Y: Engineered nanoparticles for drug delivery in cancer
therapy. Angew Chem Int Ed Engl. 53:12320–12344. 2014.PubMed/NCBI
|
|
56
|
Xu X, Ho W, Zhang X, Bertrand N and
Farokhzad O: Cancer nanomedicine: From targeted delivery to
combination therapy. Trends Mol Med. 21:223–232. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Yang Y and Yu C: Advances in silica based
nanoparticles for targeted cancer therapy. Nanomedicine.
12:317–332. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Suk JS, Xu Q, Kim N, Hanes J and Ensign
LM: PEGylation as a strategy for improving nanoparticle-based drug
and gene delivery. Adv Drug Deliv Rev. 99:28–51. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Casettari L, Vllasaliu D, Castagnino E,
Stolnik S, Howdle S and Illum L: PEGylated chitosan derivatives:
Synthesis, characterizations and pharmaceutical applications. Prog
Polym Sci. 37:659–685. 2012. View Article : Google Scholar
|
|
60
|
Palacio J, Agudelo NA and Lopez BL:
PEGylation of PLA nanoparticles to improve mucus-penetration and
colloidal stability for oral delivery systems. Curr Opin Chem Eng.
11:14–19. 2016. View Article : Google Scholar
|
|
61
|
Kim J, Kang Y, Tzeng SY and Green JJ:
Synthesis and application of poly (ethylene glycol)-co-poly
(β-amino ester) copolymers for small cell lung cancer gene therapy.
Acta Biomaterialia. 41:293–301. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Ahmed S, Sami A and Xiang J: HER2-directed
therapy: Current treatment options for HER2-positive breast cancer.
Breast Cancer. 22:101–116. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zhu L, Staley C, Kooby D, El-Rays B, Mao H
and Yang L: Current status of biomarker and targeted nanoparticle
development: The precision oncology approach for pancreatic cancer
therapy. Cancer Lett. 388:139–148. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Grunewald T and Ledermann JA: Targeted
Therapies for Ovarian Cancer. Best Pract Res Clin Obstet Gynaecol.
14:139–152. 2017. View Article : Google Scholar
|
|
65
|
McMahon KM, Scielzo C, Angeloni NL,
Deiss-Yehiely E, Scarfo L, Ranghetti P, Ma S, Kaplan J, Barbaglio
F, Gordon LI, et al: Synthetic high-density lipoproteins as
targeted monotherapy for chronic lymphocytic leukemia. Oncotarget.
8:11219–11227. 2017.PubMed/NCBI
|
|
66
|
Liu J, Zhao D, He W, Zhang H, Li Z and
Luan Y: Nanoassemblies from amphiphilic cytarabine prodrug for
leukemia targeted therapy. J Colloid Interface Sci. 487:239–249.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Pillai MR, Nanabala R, Joy A, Sasikumar A
and Knapp FF: Radiolabeled enzyme inhibitors and binding agents
targeting PSMA: Effective theranostic tools for imaging and therapy
of prostate cancer. Nucl Med Biol. 43:692–720. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Xie T, Dong B, Yan Y, Hu G and Xu Y:
Association between MMP-2 expression and prostate cancer: A
meta-analysis. Biomed Rep. 4:241–245. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Tarokh Z, Naderi-Manesh H and Nazari M:
Towards prostate cancer gene therapy: Development of a
chlorotoxin-targeted nanovector for toxic (melittin) gene delivery.
Eur J Pharm Sci. 99:209–218. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Sun L, Wu Q, Peng F, Liu L and Gong C:
Strategies of polymeric nanoparticles for enhanced internalization
in cancer therapy. Colloids Surf B Biointerfaces. 135:56–72. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Christian CG, Carlos MP, Alejandro MM,
Imelda OA, Oscar ZT, Adriana ME and Perla GC: Development of
antibody-coated magnetite nanoparticles for biomarker
immobilization. Journal of Nanomaterials. 2014:72014.
|
|
72
|
Thorek DL, Elias DR and Tsourkas A:
Comparative analysis of nanoparticle-antibody conjugations:
Carbodiimide versus click chemistry. Mol Imaging. 8:221–229.
2009.PubMed/NCBI
|
|
73
|
Crivianu-Gaita V and Thompson M: Aptamers,
antibody scFv, and antibody Fab' fragments: An overview and
comparison of three of the most versatile biosensor biorecognition
elements. Biosens Bioelectron. 85:32–45. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Kim J, Wilson DR, Zamboni CG and Green JJ:
Targeted polymeric nanoparticles for cancer gene therapy. J Drug
Target. 23:627–641. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Cheraghi R, Nazari M, Alipour M, Majidi A
and Hosseinkhani S: Development of a targeted anti-HER2 scFv
chimeric peptide for gene delivery into HER2-positive breast cancer
cells. Int J Pharm. 515:632–643. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cai Z, Chattopadhyay N, Yang K, Kwon YL,
Yook S, Pignol JP and Reilly RM: 111In-labeled trastuzumab-modified
gold nanoparticles are cytotoxic in vitro to HER2-positive breast
cancer cells and arrest tumor growth in vivo in athymic mice after
intratumoral injection. Nucl Med Biol. 43:818–826. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Yin XB, Wu LQ, Fu HQ, Huang MW, Wang K,
Zhou F, Yu X and Wang KY: Inhibitory effect of humanized
anti-VEGFR-2 ScFv-As2O3-stealth nanoparticles conjugate on growth
of human hepatocellular carcinoma: In vitro and in vivo studies.
Asian Pac J Trop Med. 7:337–343. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Xiangbao Y, Linquan W, Mingwen H, Fan Z,
Kai W, Xin Y, Kaiyang W and Huaqun F: Humanized anti-VEGFR-2
ScFv-As2O3-stealth nanoparticles, an antibody conjugate with potent
and selective anti-hepatocellular carcinoma activity. Biomed
Pharmacother. 68:597–602. 2014. View Article : Google Scholar : PubMed/NCBI
|