Introduction
Laryngeal cancer is one of the leading malignant
tumors of the head and neck and is reported to have a higher
incidence in highly developed countries (1). Male patients are at particular risk,
with an annual rate of incidence of ~3.5–5.5/100,000 cases. By
contrast, female patients are at a markedly lower risk with an
annual rate of 0.6/100,000 cases. The mortality rate among men was
~2.4/100,000 cases and in women, the rate was ~0.3/10 million cases
(1). There are various options for
treatment of laryngeal cancer including the conventional approaches
of surgery, chemotherapy and radiotherapy. However, the curative
effect and rate remain poor and require significant
improvement.
Although a number of chemotherapeutic drugs are
available for the treatment of cancer, a highly effective and less
toxic approach for treating laryngeal cancer is lacking. One
potential resource for a new generation of therapeutics for the
prevention and treatment of laryngeal cancer may be natural
substances. For example, epidemiological studies suggest that the
intake of broccoli, cauliflower and other cruciferous vegetables
can significantly reduce the incidence of cancers of the bladder,
pancreas, colon, lung and stomach (2–5).
Indole-3-carbinol (I3C) has recently been identified as an
important tumoricidal component found in cruciferous vegetables and
particularly in members of the genus Brassica(6,7). A
number of studies have shown that I3C induces cell cycle arrest in
the G1 phase in cancer cells (8,9),
promotes apoptosis (8–10) and prevents tumor invasion and
metastasis (11). It has been found
that I3C promotes cell cycle arrest by downregulating
cyclin-proteins such as cyclin D1 and cyclin E. Additionally, it is
thought that IC3 induces apoptosis by mechanisms that depend on
downregulation of the anti-apoptotic genes Bcl-2, Bcl-xL and
survivin and by enhancing expression of Bax and by functionally
activating caspase-3 and caspase-9 (9–12).
The phosphatidylinositol-3 kinase/serine-threonine
kinase (PI3K/Akt) signaling pathway is involved in the activation
of anti-apoptotic mechanisms, glucose metabolism and protein
synthesis, all of which influence cell growth and proliferation
(13,14). Abnormal activation of the PI3K/Akt
pathway is found in many malignancies. Moreover, it is becoming
apparent that tyrosine kinase-mediated activation of PI3K may be
important, for example, in the context of phosphorylated tyrosine
kinase interacting with the p85 subunit or in the context of
mutated Ras-binding to PI3K, which leads to the activation of
PI3K.
In addition, somatic mutations may also play an
important role. For example, mutation in the PTEN tumor-suppressor
gene may disrupt the ability of PTEN to switch off the PI3K
pathway. Recently a PIK3CA mutation was found to occur in ~30% of
epithelial tumors. Abnormal activation of PI3K and somatic
mutations can collectively drive uncontrolled growth and cell
proliferation during the development of tumors, including ovarian,
breast, pancreatic, lung and colon cancer or indeed other
malignancies (13,15,16).
To the best of our knowledge there are few reports
describing the application of I3C in the treatment of laryngeal
cancer. Similarly, there currently exists no report concerning the
mechanism of action of I3C and its putative relationship with
PI3K/Akt. The present study aimed to determine the influence of I3C
on the proliferation and apoptosis of human laryngeal cancer using
the Hep-2 cell line and to determine the relevance of the PI3K/Akt
signaling pathway. The main objective was to elucidate the
potential tumoricidal mechanism of I3C and to provide a basis for
tumor-targeting treatment and drug screening.
Materials and methods
Cell culture and preparation of I3C
The human laryngeal carcinoma Hep-2 cell line was
obtained from the central laboratory at Wuhan University. Hep-2
cells were cultured in RPMI-1640 culture medium (Invitrogen Life
Technologies Carlsbad, CA, USA) and supplemented with 10%
heat-inactivated fetal bovine serum (FBS; HyClone, Logan, UT, USA),
100 U/ml penicillin and 20 μg/ml streptomycin in a fully humidified
incubator at 37°C and an atmosphere of 5% CO2 in air.
I3C (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in dimethyl
sulphoxide (DMSO) to a stock concentration of 1 M, and was
subsequently diluted to concentrations of 50, 100 and 150 μM for
use in the experiments. The final concentration of DMSO in the I3C
preparations was <0.5%. In order to demonstrate that the final
concentration of DMSO did not affect the laryngeal cancer cells,
Hep-2 cells cultured in complete medium alone, without exposure to
I3C (as the untreated control), and thus DMSO (<0.5%), were used
as the negative control group (0 μM).
Assay of Hep-2 cell proliferation using
CCK-8 reagent
Hep-2 cells were seeded into 96-well plates at a
density of 1×103/ml in complete culture medium and
stimulated with I3C at 0, 50, 100 or 150 μM in replicates of 6
wells per concentration of I3C. An additional 6 wells contained
culture medium alone as a blank control. After 0, 24, 48 and 72 h,
cells were treated with 10 μl of CCK-8 reagent (Dongji, Japan) and
incubated at 37°C for 1 h. An automatic microtitre plate reader was
set to zero according to the blank control wells. The absorbance
(A) of the wells was read at a wavelength of 450 nm. The percentage
of cell proliferation inhibition was calculated according to the
following formula: Cell proliferation inhibition (%) = (1 − average
absorbance (A) of the experimental group/average absorbance (A) of
the control group) × 100%.
Morphological assessment of Hep-2
cells
Hep-2 cells were seeded into 6-well plates in
complete culture medium containing 0, 50, 100 and 150 μM of I3C.
Separate wells were seeded with culture medium alone, as an
untreated control group. The experimental group, negative control
group (0 μM of I3C) and blank control group were seeded in
triplicate. After 48 h of culture, the cells were stained with 2
μg/ml Hoechst 33258 (Beyotime, Shanghai, China) and observed under
a fluorescence microscope equipped with a blue filter for the
assessment of morphological changes. The nuclei of normal cells
were uniformly blue, and by contrast, apoptotic cells showed
densely stained pyknosis and nuclear fragmentation.
Assay of cell apoptosis by dual staining with
Annexin V and propidium iodide (PI).
Hep-2 cells were seeded into 6-well plates and in
complete culture medium containing 0, 50, 100 and 150 μM of I3C,
and separate wells were seeded with culture medium alone as an
untreated control group. Each group was seeded in triplicate. After
48 h of culture, the cells were harvested by centrifugation,
resuspended in binding buffer, and successively incubated with 5 μl
Annexin V-FITC and 5 μl of PI (Multi Sciences, China) at room
temperature for 15 min. Apoptosis was determined by flow cytometric
analysis using a FACSCanto™II spectrophotometer (BD Biosciences,
San Jose, CA, USA). For the flow cytometric dot plot, Annexin V
staining was set as the horizontal axis and PI staining was set as
the vertical axis. Mechanically damaged cells were located in the
upper left quadrant, apoptotic or necrotic cells in the upper right
quadrant, dual negative and normal cells in the lower left quadrant
and early apoptotic cells in the lower right quadrant of the flow
cytometric dot plot.
Western blot assay of PI3K, Akt, p-c-Raf
and GSK3-β expression
After 48 h of treatment with different
concentrations of I3C, the cells were lysed in RIPA buffer to
extract total cellular protein. Protein concentration was
determined according to the BCA quantitative method, and 30 μg of
each protein sample was resolved by SDS-PAGE and the protein bands
were transferred to a nitrocellulose membrane. Following protein
transfer, the membrane was blocked for 1 h in the presence of 5%
skimmed milk proteins, following by incubation at 4°C overnight
with the primary antibodies (Cell Signaling Technology, Inc.,
Beverly, MA, USA) targeted against PI3K p110α, PI3K p110β, PI3K
class III, p-PDK1, p-Akt, Akt, p-c-Raf, GSK3-β and GAPDH (as an
internal reference housekeeping protein). On the following day, the
blots were incubated with a secondary antibody at room temperature
for 1 h, and specific protein bands were visualized by an enhanced
chemiluminescence (ECL) assay kit (Pierce Biotechnology, Inc.,
Rockford, IL, USA).
Animal selection and breeding
Specific pathogen-free (SPF) female BALB/c nude mice
(aged 4 weeks and weighing ~18–20 g) were obtained from Beijing HFK
Bioscience Co., Ltd. (China). All animal husbandry practices and
experimentation were carried out under SPF conditions. Prior to
initiating the experiment, animals were provided with conventional
feed for 1 week (provided by Beijing HFK Bioscience Co., Ltd).
Following this adaptive feeding, I3C was incorporated into the
conventional feed at a ratio of 0.5% (w/w). All elements of this
study were approved by the local Ethics and Animal Care and Use
Committee of Wuhan University, China.
Establishing tumor-bearing animal models
in BALB/c nude mice
Twenty-four female BALB/c mice were randomly
assigned to a pretreatment group, a treatment group and a control
group (n=8 mice/group). The Hep-2 cells were cultured to a
confluence of 80–90%, harvested by treatment with trypsin, and
following centrifugation were resuspended in plasma. Hep-2 cells
were adoptively transferred into mice at a density of
1×107 cells/ml, by injecting a 200-μl cell suspension in
plasma subcutaneously into the right side of the back of nude mice.
The pretreatment group was received the conventional feed
supplemented with I3C at a ratio of 0.5% (w/w) for 2 weeks prior to
adoptive Hep-2 cell transfer and then received to a conventional
feed following injection of the tumor cells. The treatment group
received a normal conventional diet until one week after adoptive
transfer with the Hep-2 cells. At this time point, the tumors had
grown to a size of 5×5 mm, and mice then received a feed
supplemented with a 0.5% (w/w) ratio of I3C. Mice were continued on
the I3C supplemented feed, until the experiment had reached the
endpoint at week 8. Mice in the control group were fed only
conventional feed. At the end of each week following the adoptive
transfer of the Hep-2 cells into the mice, the long diameter (a)
and the short diameter (b) were measured in order to calculate the
volume using the formula: V = 1/2ab2 for statistical
analysis. Eight weeks thereafter the mice were sacrificed, and
specimens were collected from the tumor, heart, liver and
kidney.
Protein extraction and expression
assay
Tumor specimens from the sacrificed mice were cut
into regularly sized pieces and minced using a tissue homogenizer.
Immediately following this preparation, the cells were lysed in
RIPA buffer, and the extracted proteins were quantified by the BCA
method. Western blot assay was applied to detect the specific
protein expression of PI3K, Akt, p-c-Raf and GSK3-β.
Morphological observation of the heart,
liver and kidney tissue sections
Biopsy specimens of the heart, liver and kidney
obtained from the nude mice were fixed in 10% formalin, dehydrated,
embedded in wax, and sectioned into 5- to 8-μm specimens and pasted
onto slides. Next, the specimens were de-waxed and stained with
hematoxylin and eosin (H&E) for final histological evaluation
under a standard light microscope.
Statistical analysis
All statistical analyses were assessed by the SPSS
statistical software package, version 16.0 for Microsoft Windows
(SPSS Inc., Chicago, IL, USA). Results are expressed as means ±
standard deviation (SD). Differences between multiple groups were
compared by ANOVA. P<0.05 was considered to indicate a
statistically significant result.
Results
Marked inhibition of laryngeal cancer
cell growth following I3C treatment
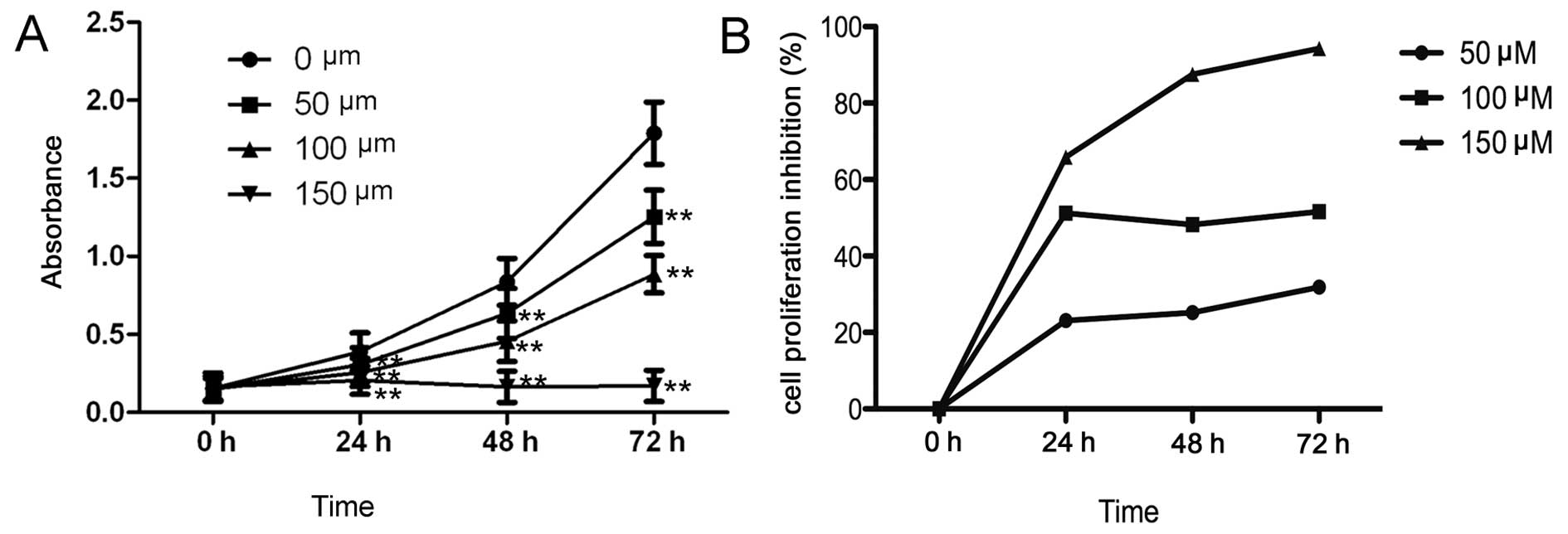
Following the dose-dependent (50, 100 and 150 μM)
and time-dependent (24, 48 and 72 h) treatment of Hep-2 cells with
I3C, significant differences in cell growth were noted between the
treated and negative control groups (P<0.01) (Fig. 1A). In the group treated with 50 μM
I3C at the indicated time points of 24, 48 and 72 h, the percentage
of Hep-2 cell proliferation inhibition was 23.12, 25.21 and 31.89%,
respectively. Additionally, in the group treated with 100 μm I3C
the percentage of cell proliferation inhibition at 24, 48 and 72 h
was 51.17, 48.22 and 51.62% respectively. In the group treated with
150 μM I3C, the percentage of cell proliferation inhibition at 24,
48 and 72 h was 65.97, 87.45 and 94.26% respectively (Fig. 1B). Inhibition of the proliferation
of Hep-2 laryngeal carcinoma cells by I3C was highly
dose-dependent.
Induction of laryngeal cancer Hep-2 cell
apoptosis following I3C treatment
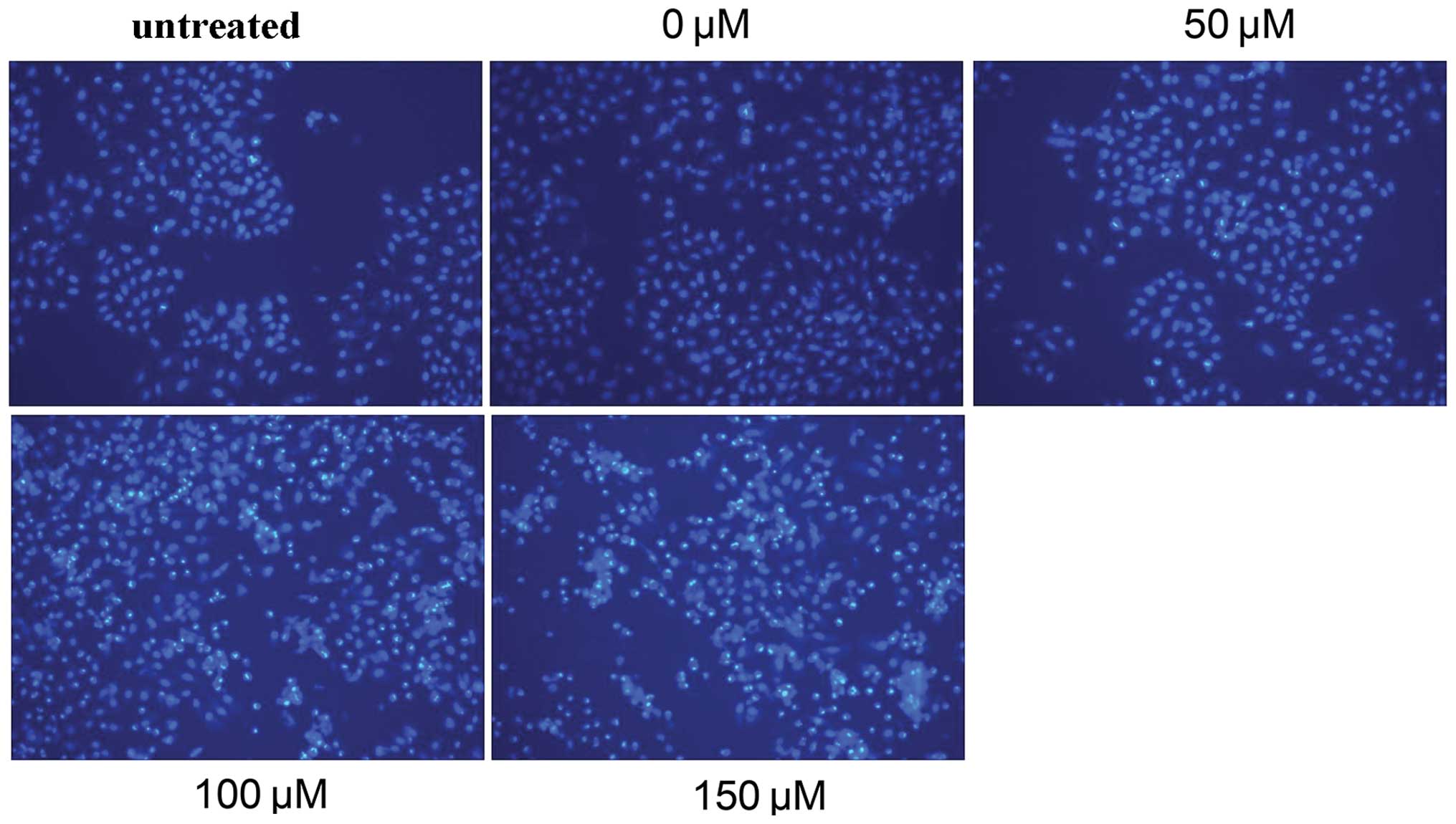
After Hep-2 cells were treated with I3C for 48 h,
the cells were stained with Hoechst 33258 and observed by
fluorescence microscopy. In the negative (0 μM I3C) and untreated
control groups, Hep-2 cell nuclei displayed a normal and complete
blue appearance. By contrast, in Hep-2 cells treated with 50, 100
or 150 μM I3C, the cells displayed enhanced fragmentation or
pyknosis of the nuclei, which were typical changes associated with
cellular apoptosis. Nuclear pyknosis and fragmentation in Hep-2
cells were significantly increased by treatment with I3C in a
dose-dependent manner. Morever, at an I3C concentration of 150 μM,
the ratio of pyknosis was ~62±6% (Fig.
2).
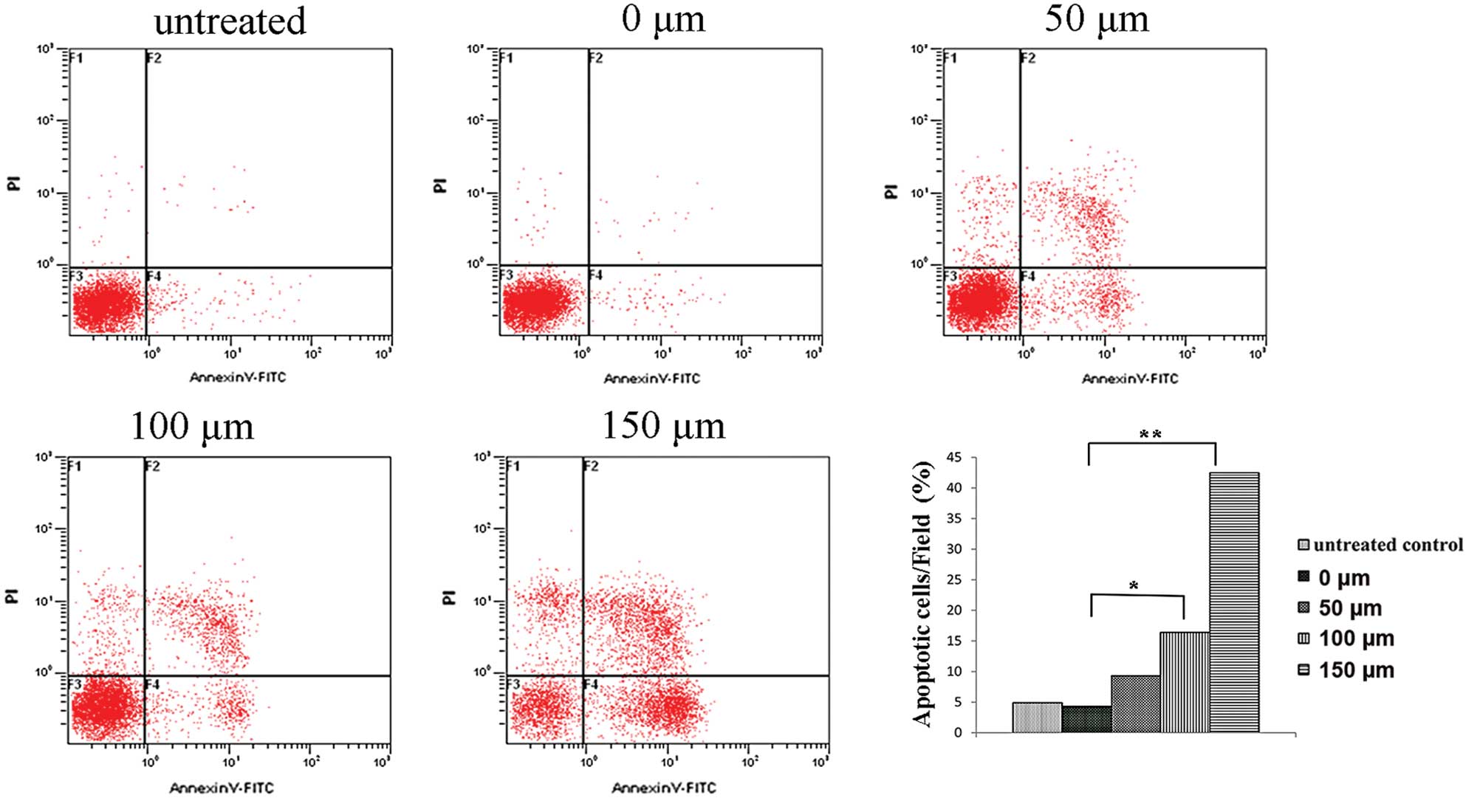
The analysis of Hep-2 cell apoptosis by Annexin V-PI
double staining showed that after 48 h of treatment with 0, 50, 100
or 150 μM I3C, the apoptosis rate was 4.9±0.85, 9.3±1.56, 16.4±2.28
and 42.5±5.32%, respectively. For the untreated control the
apoptosis rate was 4.3±0.76%. Additionally, when comparing the 50,
0 μM and the untreated control groups, there was no statistically
significant difference in the rate of apoptosis. Significant
increases in the apoptosis rates were noted in the 100 and 150 μM
groups as compared with the 0 μM and the untreated control groups
(P<0.05, P<0.01). This showed that a concentration of 100 μM
I3C induced apoptosis of Hep-2 cells, and a concentration of 150 μM
significantly increased the proportion of apoptotic cells and
displayed an apoptosis rate >40% (Fig. 3). This suggested that the proportion
of cells undergoing apoptosis following treatment with I3C was
dose-dependent (Fig. 3).
Significantly reduced protein expression
of PI3K/Akt in Hep-2 cells following treatment with I3C
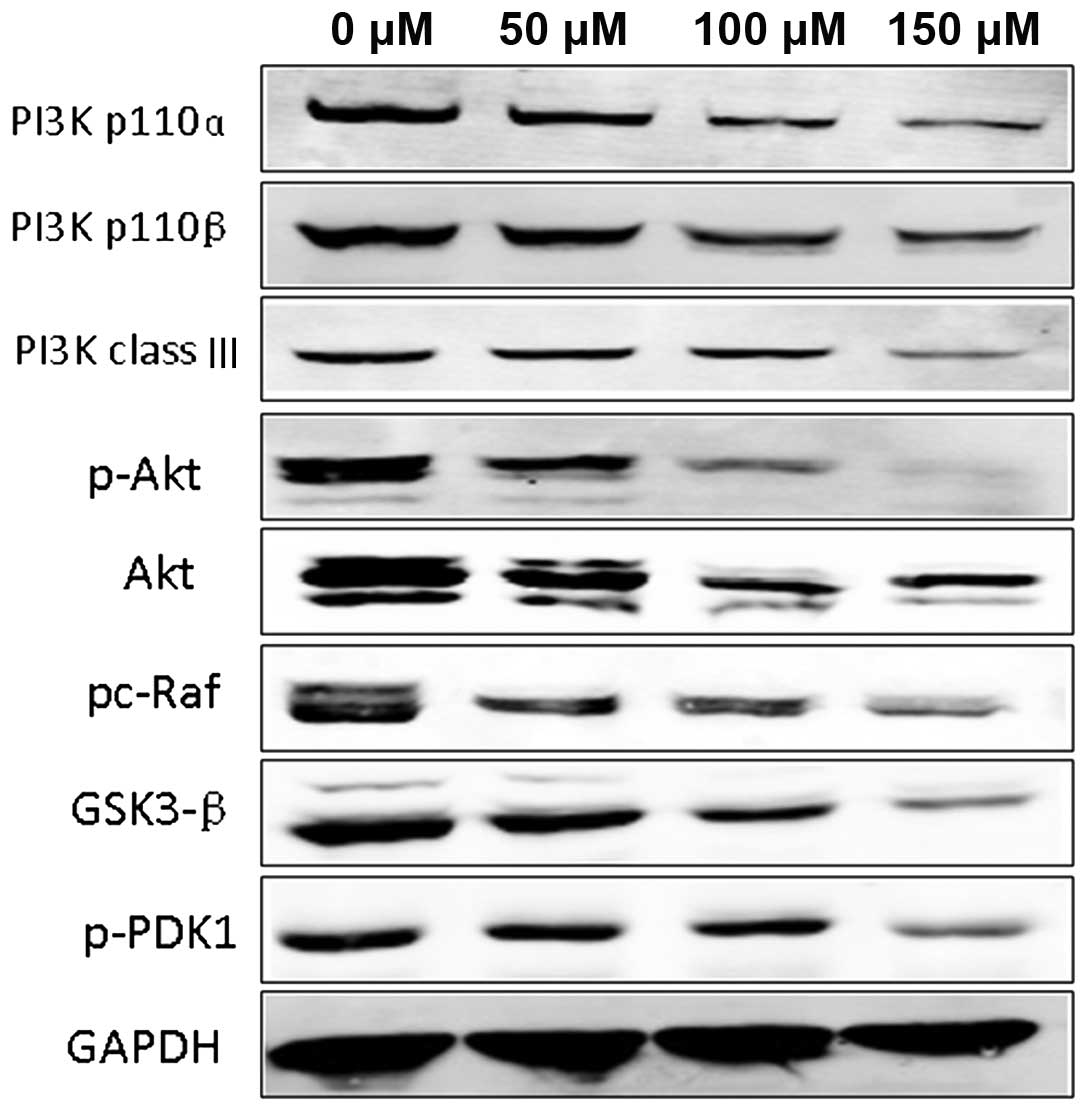
We examined the expression of key signaling proteins
of the PI3K/Akt pathway and downstream signaling proteins
associated with cancer cell proliferation and apoptosis. We found
that in the untreated control and negative control groups, the
protein expression of PI3K p110α, PI3K p110β, PI3K class III,
p-PDK1, Akt, p-Akt, p-c-Raf and GSK3-β were all highly increased.
Treatment of Hep-2 cells with I3C markedly decreased the protein
expression of PI3K p110α, PI3K p110β, PI3K class III, p-PDK1, Akt,
p-Akt, p-c-Raf and GSK3-β when compared with these levels in the
untreated groups, and the decreased expression levels of the
signaling proteins was I3C dose-dependent (Fig. 4).
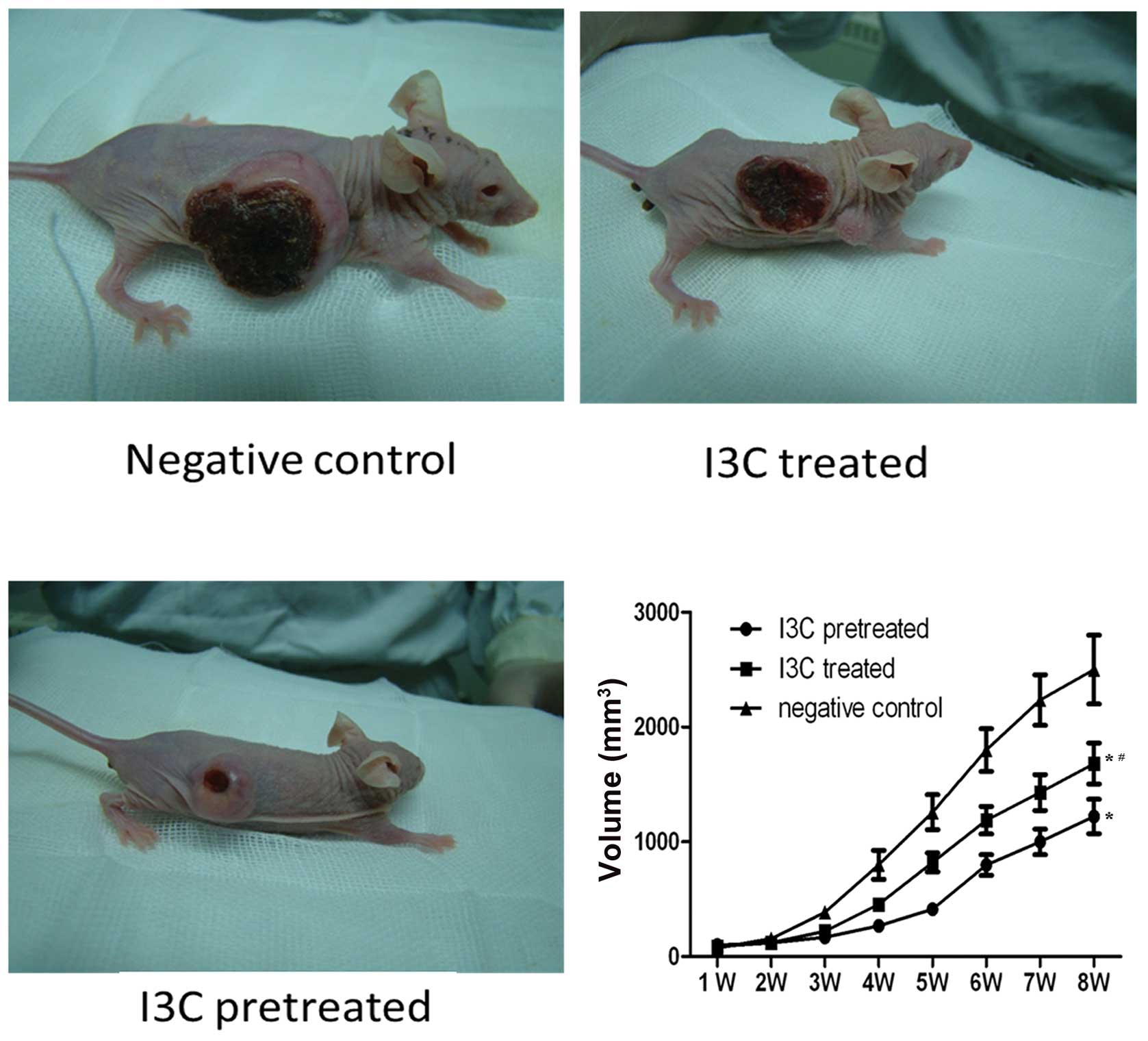
Tumoricidal effects of I3C in a BALB/c
nude mouse model
We further investigated the tumoricidal effect of
I3C in animal model experiments. The control mouse group received
only conventional feed. These mice subsequently developed tumors of
a large size; at week 8; the average volume of the tumors was
2456.5±266.3 mm3. By contrast, the mean volumes of the
tumors in the I3C treatment group and the pretreatment group were
1732.7±153.5 mm3 and 1263.6±134.4 mm3,
respectively (P<0.05). This suggests that I3C significantly
inhibited tumor growth (P<0.05), and that administration of I3C
prior to tumor transplantation was more effective than
administration after transplantation of the tumor was established
(P<0.05) (Fig. 5).
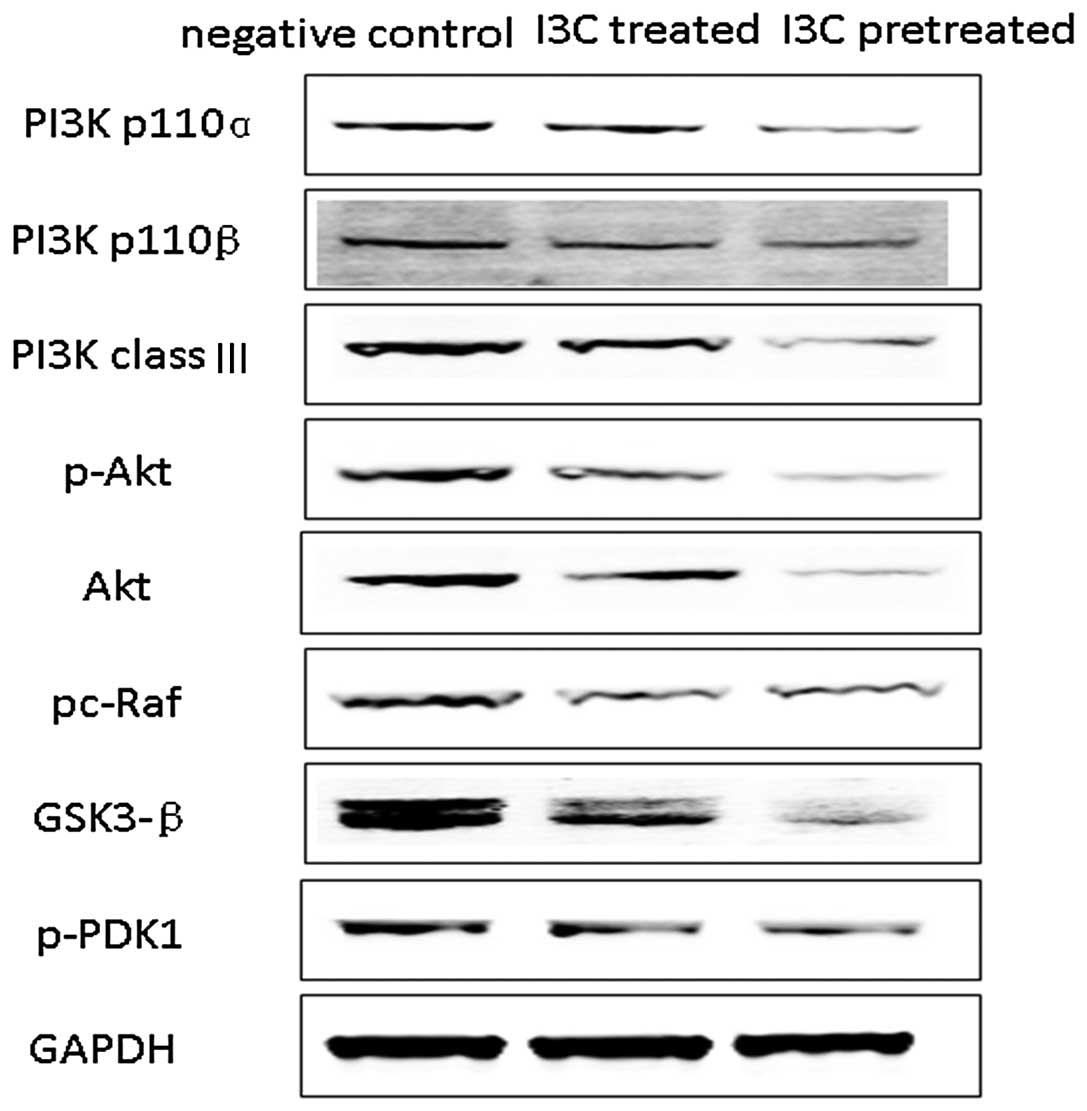
I3C treatment significantly reduces
expression of PI3K/AKT and downstream signaling proteins in the
transplanted tumors
We analyzed the expression of proteins related to
the PI3K/AKT pathway and downstream signaling pathways in the
transplanted tumors and found that increasing concentrations of I3C
were associated with reduced expression of PI3K p110α, PI3K p110β,
PI3K class III, p-PDK1, Akt, p-Akt, p-c-Raf and GSK3-β signaling
proteins. The lowest signaling protein expression was detected in
the pretreatment group, followed by the treatment group. As
expected, the highest expression of the signaling proteins was
found in the control group (Fig.
6).
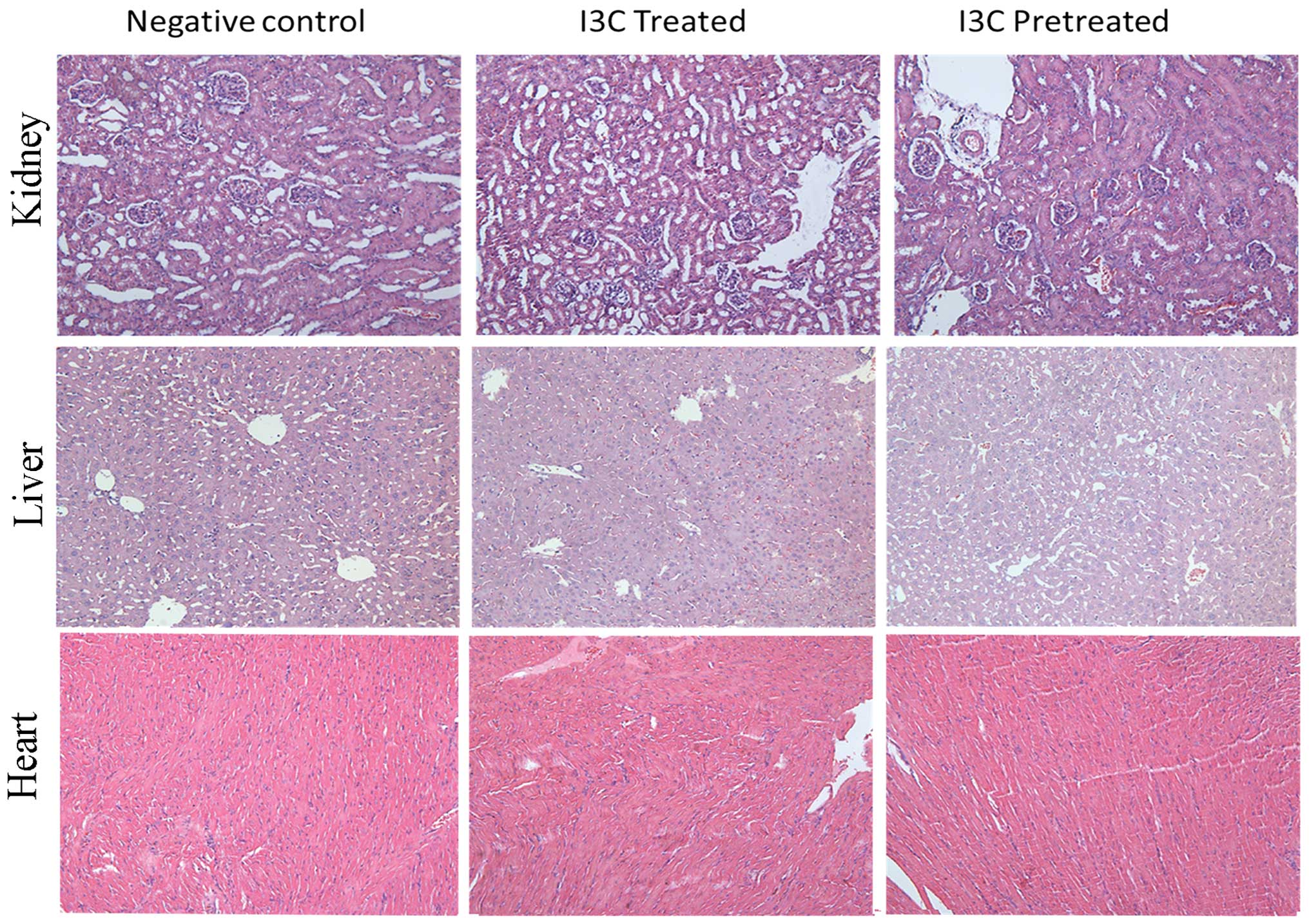
No harmful side effects are noted in the
heart, liver and kidney of I3C-treated nude mice
Specimens of the heart, liver and kidney obtained
from the nude mice were sectioned and stained with H&E.
Pathological evaluation showed that treatment with I3C exerted no
adverse effects on the heart, liver or kidney in the three groups
of nude mice. No tissue damage such as structural degeneration or
tissue necrosis was noted in any of the tissue specimens. These
studies indicated that the supplementation of the feed with 0.5%
(w/w) I3C provoked no toxic side effects in any of the organs
examined in the experimental mice (Fig.
7).
Discussion
Indole-3-carbinol is extracted from cruciferous
vegetables by hydrolysis of glucosinolates (6). Many previous in vitro studies
have shown that I3C causes significant growth inhibition of a
variety of tumor cell lines by inducing cell cycle arrest and
apoptosis (8,10,12,17,18).
However, the role of I3C in laryngeal cancer has remained largely
unexplored. Our findings found that I3C significantly inhibited
proliferation of Hep-2 cells and that I3C at a dose of 150 μM was
an optimal concentration with the tumor inhibition rate of 65.97,
87.45 and 94.26% noted at 24, 48 and 72 h, respectively. The
inhibitory mechanism involved alteration of the proliferation of
cancer cells and induction of apoptosis. We observed that a dose of
100 μM of I3C induced an apoptotic effect and that the apoptotic
rate increased in an I3C dose-dependent manner. Rahman et
al(10) reported that treatment
of breast cancer cells with 30–100 μM of I3C for 24–72 h induced
apoptosis.
The PI3K/Akt pathway is associated with receptor
tyrosine kinase signaling pathways. Mutational activation of
pathway components can inappropriately activate PI3K and downstream
target proteins Akt and phosphorylated Akt (p-Akt), and can
subsequently result in phosphorylation and activation of mTORGSK3,
mdm2, Bad, and members of the caspase family, that collectively
play an important role in promoting tumor cell growth,
proliferation, suppression of apoptosis, promotion of cellular
invasion, tumor metastasis and angiogenesis (19). Abnormalities in the PI3K/Akt
signaling pathway, in particular, and abnormal phosphorylation of
Akt have been found to be closely associated with the development
of tongue carcinoma and head and neck squamous cell carcinomas
(15,20).
To further clarify the potential relationship
between I3C-induced apoptosis and the PI3K/Akt signaling pathway in
laryngeal cancer, we analyzed the differential expression of
PI3K/Akt pathway-specific signaling proteins and their cognate
downstream proteins both before and after treatment with I3C.
Markedly decreased expression of PI3K p110α, PI3K p110β, PI3K class
III, p-PDK1, Akt, p-Akt, p-c-Raf and GSK3-β in Hep-2 cells was
noted following treatment with I3C as compared with levels in the
untreated cells. This observation suggests that inhibition of
laryngeal cancer cell proliferation and induction of apoptosis is
closely associated with the PI3K/Akt signaling pathway and the
findings are concordant with those found in breast or prostate
cancer (8,21).
Previous animal studies (16,17)
demonstrated that I3C inhibits the occurrence and development of
solid tumors. Previous studies of cervical cancer revealed that the
antitumor mechanism of I3C was partly dependent on the increase in
the permeability of the tumor cell membrane, activation of
caspase-3 and thus induction of cancer cell apoptosis (22). When mice with prostate cancer
(TRAMP) were fed diets containing 1% (w/w) I3C over a period of 10
weeks, tumor cell apoptosis was induced (23) and Nrf2 was activated, which is a
pathway known to regulate antioxidant signaling pathways. Newfield
et al(24) reported that the
immune response of mice fed with I3C could be triggered to produce
the anti-proliferative agent 2-hydroxy estrone. Our animal
experiments confirmed that I3C effectively inhibited the growth of
laryngeal cancer and that I3C intervention significantly decreased
laryngeal tumor growth in nude mice when compared with the control
mice. It is worth noting here that the graft tumor growth rate was
slower in the pretreatment group than that noted in the treated
mouse group, suggesting that I3C effectively prevents the
occurrence and development of solid tumors. This observation also
suggests that I3C can be considered as an alternative therapeutic
approach for the prevention and treatment of laryngeal cancer.
Consistent with the results obtained from the in
vitro experiments in the pretreated and treated nude mice, we
found that the expression levels of PI3K p110α, PI3K p110β, PI3K
class III, p-PDK1, Akt, p-Akt, p-c-Raf and GSK3-β were
significantly downregulated when compared with the levels in the
control group not treated with I3C. This observation indicates that
abnormal activation of the PI3K/Akt pathway may play a key role in
the process of laryngeal squamous cell carcinoma. Our animal
experiments also confirmed that I3C downregulated protein
expression in the PI3K/Akt signaling pathway in the transplanted
tumors.
Using I3C to treat MCF10CA1a breast cancer cells and
homologous CF10A mammary epithelial cells, Rahman et
al(10) found that I3C only
induced MCF10CA1a breast cancer cell apoptosis (10), suggested that I3C was without risk
to nontumor cells. Consistently, our experiment also confirmed that
I3C caused growth inhibition only in the transplanted tumors in
nude mice, and did not provoke any degenerative harm to the heart,
liver, kidney and other organs with low cytotoxicity to normal
cells.
In summary, our in vivo and in vitro
experiments demonstrated that I3C significantly inhibits laryngeal
cancer cell proliferation and induces apoptosis. We suggest that
the observed effects of I3C are likely associated with the
regulation of the PI3K/Akt signaling pathway and dowstream protein
expression. Due to the effective, non-toxic, natural antitumor
properties, I3C is considered to be a likely preventive and
curative candidate for laryngeal cancer. Additional studies are
required to determine the underlying mechanisms whereby I3C
suppresses laryngeal cancer at the molecular level, and to provide
a theoretical basis for the tumoricidal and clinical utility of
I3C.
Acknowledgements
This study was supported by grants from the National
Natural Science Foundation of China (no. 30901662), and the Science
and Technology Program of Wuhan City (nos. 200951199455 and
200950431168).
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Michaud DS, Spiegelman D, Clinton SK, Rimm
EB, Willett WC and Giovannucci EL: Fruit and vegetable intake and
incidence of bladder cancer in a male prospective cohort. J Natl
Cancer Inst. 91:605–613. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Annema N, Heyworth JS, McNaughton SA,
Iacopetta B and Fritschi L: Fruit and vegetable consumption and the
risk of proximal colon, distal colon, and rectal cancers in a
case-control study in Western Australia. J Am Diet Assoc.
111:1479–1490. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Larsson SC, Hakansson N, Näslund I,
Bergkvist L and Wolk A: Fruit and vegetable consumption in relation
to pancreatic cancer risk: a prospective study. Cancer Epidemiol
Biomarkers Prev. 15:301–305. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
van Poppel G, Verhoeven DT, Verhagen H and
Goldbohm RA: Brassica vegetables and cancer prevention.
Epidemiology and mechanisms. Adv Exp Med Biol. 472:159–168.
1999.
|
|
6
|
Keck AS and Finley JW: Cruciferous
vegetables: cancer protective mechanisms of glucosinolate
hydrolysis products and selenium. Integr Cancer Ther. 3:5–12. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shertzer H and Senft A: The micronutrient
indole-3-carbinol: implications for disease and chemoprevention.
Drug Metabol Drug Interact. 14:159–188. 2011.PubMed/NCBI
|
|
8
|
Chinni SR, Li Y, Upadhyay S, Koppolu PK
and Sarkar FH: Indole-3-carbinol (I3C) induced cell growth
inhibition, G1 cell cycle arrest and apoptosis in prostate cancer
cells. Oncogene. 20:2927–2936. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Brew CT, Aronchik I, Hsu JC, et al:
Indole-3-carbinol activates the ATM signaling pathway independent
of DNA damage to stabilize p53 and induce G1 arrest of human
mammary epithelial cells. Int J Cancer. 118:857–868. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rahman KM, Aranha O and Sarkar FH:
Indole-3-carbinol (I3C) induces apoptosis in tumorigenic but not in
nontumorigenic breast epithelial cells. Nutr Cancer. 45:101–112.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Meng Q, Qi M, Chen DZ, et al: Suppression
of breast cancer invasion and migration by indole-3-carbinol:
associated with up-regulation of BRCA1 and E-cadherin/catenin
complexes. J Mol Med. 78:155–165. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Aggarwal BB and Ichikawa H: Molecular
targets and anticancer potential of indole-3-carbinol and its
derivatives. Cell Cycle. 4:1201–1215. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Chang F, Lee JT, Navolanic PM, et al:
Involvement of PI3K/Akt pathway in cell cycle progression,
apoptosis, and neoplastic transformation: a target for cancer
chemotherapy. Leukemia. 17:590–603. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fresno Vara JA, Casado E, de Castro J,
Cejas P, Belda-Iniesta C and González-Barón M: PI3K/Akt signalling
pathway and cancer. Cancer Treat Rev. 30:193–204. 2004.PubMed/NCBI
|
|
15
|
Watanabe S, Sato K, Okazaki Y, Tonogi M,
Tanaka Y and Yamane GY: Activation of PI3K-AKT pathway in oral
epithelial dysplasia and early cancer of tongue. Bull Tokyo Dent
Coll. 50:125–133. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Oganesian A, Hendricks JD and Williams DE:
Long term dietary indole-3-carbinol inhibits
diethylnitrosamine-initiated hepatocarcinogenesis in the infant
mouse model. Cancer Lett. 118:87–94. 1997. View Article : Google Scholar
|
|
17
|
Plate AY and Gallaher DD: Effects of
indole-3-carbinol and phenethyl isothiocyanate on colon
carcinogenesis induced by azoxymethane in rats. Carcinogenesis.
27:287–292. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pappa G, Lichtenberg M, Iori R, Barillari
J, Bartsch H and Gerhäuser C: Comparison of growth inhibition
profiles and mechanisms of apoptosis induction in human colon
cancer cell lines by isothiocyanates and indoles from
Brassicaceae. Mutat Res. 599:76–87. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Brognard J, Clark AS, Ni Y and Dennis PA:
Akt/protein kinase B is constitutively active in non-small cell
lung cancer cells and promotes cellular survival and resistance to
chemotherapy and radiation. Cancer Res. 61:3986–3997.
2001.PubMed/NCBI
|
|
20
|
Cicenas J: The potential role of Akt
phosphorylation in human cancers. Int J Biol Markers. 23:1–9.
2008.PubMed/NCBI
|
|
21
|
Rahman KM, Li Y and Sarkar FH:
Inactivation of akt and NF-κB play important roles during
indole-3-carbinol-induced apoptosis in breast cancer cells. Nutr
Cancer. 48:84–94. 2004.
|
|
22
|
Chen DZ, Qi M, Auborn KJ and Carter TH:
Indole-3-carbinol and diindolylmethane induce apoptosis of human
cervical cancer cells and in murine HPV16-transgenic preneoplastic
cervical epithelium. J Nutr. 131:3294–3302. 2001.
|
|
23
|
Wu TY, Saw CL, Khor TO, Pung D,
Boyanapalli SS and Kong AN: In vivo pharmacodynamics of
indole-3-carbinol in the inhibition of prostate cancer in
transgenic adenocarcinoma of mouse prostate (TRAMP) mice:
involvement of Nrf2 and cell cycle/apoptosis signaling pathways.
Mol Carcinog. 51:761–770. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Newfield L, Goldsmith A, Bradlow HL and
Auborn K: Estrogen metabolism and human papillomavirus-induced
tumors of the larynx: chemo-prophylaxis with indole-3-carbinol.
Anticancer Res. 13:337–341. 1993.PubMed/NCBI
|