Introduction
Colorectal cancer (CRC) is ranked as one of the most
common types of cancer. In fact, according to data from the USA,
for the male population it is ranked third of the most diagnosed,
after lung and prostate cancer, and for the female population, it
is ranked second, following breast cancer (1). Moreover, it is considered as one of
the leading causes of cancer-related death worldwide (2,3) In the
USA alone, CRC is responsible for the second greatest number of
cancer-related deaths. As a matter of fact, the American Cancer
Society estimated that for 2013 alone, the number of first
diagnosed CRC cases and that of deaths due to CRC was as high as
142,820 and about 50,830, respectively (4). However, regardless of the fact that
both public and medical awareness concerning CRC has risen during
the last decade, approximately 50% of the patients referred with
CRC, at the time of diagnosis present distant metastases. It is
clear that CRC is a rather heterogeneous disease by means of its
various clinical manifestations, biological behavior and in-tumor
variety of mutations (5,6) making it a true challenge, not only to
detect in an early stage, but also to treat or even manage in the
long term. Nowadays, it is evident that CRC is a
multifactorial/polygenic disease arising both due to epigenetic as
well as genetic manifestations occurring in a number of genes with
an unparalleled role for the maintenance of normal cellular
homeostasis. Such genes may be tumor suppressor genes, oncogenes,
mismatch repair genes and cell cycle regulating genes in colon
mucosal cells (7). Intense research
has highlighted the importance of three major molecular pathways as
the main cause of these genetic alterations that in turn result in
carcinogenesis: Chromosomal instability (CIN), microsatellite
instability (MSI), and the CpG island methylation phenotype (CIMP)
(5,8). All these pathways attribute to the
transformation of an adenoma to carcinoma, a multistep carcinogenic
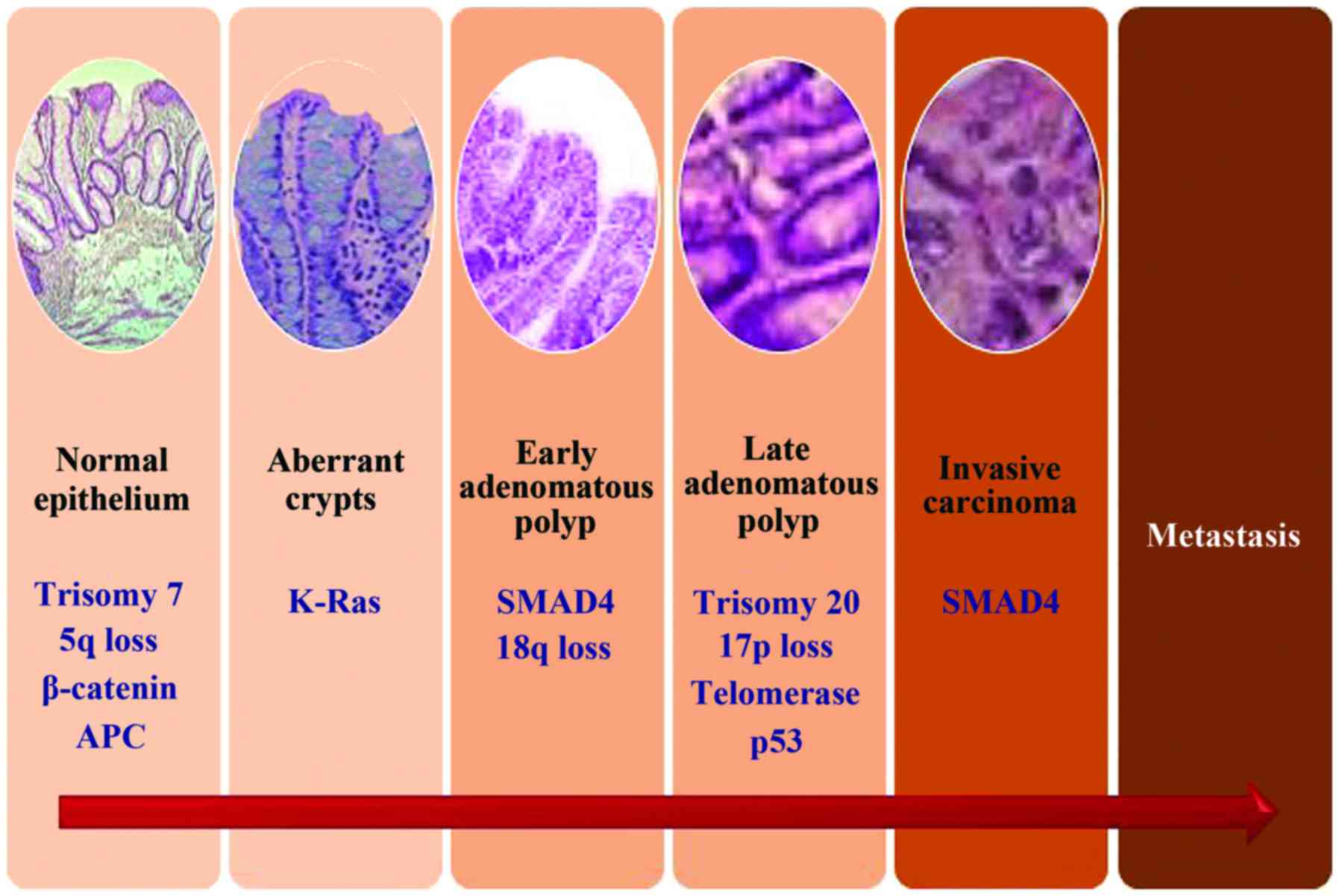
process known as the adenoma-carcinoma sequence (9), which is thought to be a common site
for all CRCs. An illustrated form of this process is presented in
Fig. 1. However, the period between
those two hallmarks provides a diagnostic window for the early
detection of CRC. As a result, the current research trend aims to
describe new markers for diagnosis (these markers are meant to be
used for risk stratification and early detection of colorectal
polyps), prognosis (these markers will give an indication of the
likely progression of the disease) and prediction of the biological
behavior of a certain therapeutic regimen, making the clinician
able to transform the knowledge of the tumor biology into a
personalized decision-making process for each patient (8,10,11).
Therefore, the information coming from CRC biology combined with
the assessment of serum and tissue markers with prognostic and
predictive value, currently constitutes, the pillars in the
treatment of early-stage cases as well in the clinical management
of advanced disease offering new methods to estimate the
therapeutic efficacy and the overall outcome. An overview of the
current and potential biomarkers used in CRC is shown in Fig. 2. In addition to CRC biomarkers,
novel molecular imaging techniques using hybrid positron emission
tomography (PET)/computed tomography (CT) systems enable accurate
initial staging, efficient assessment of treatment response, and
follow-up, facilitating individualized treatment strategies in CRC
patients (12). Furthermore, novel
positron emitting radiopharmaceuticals together with hybrid
PET/magnetic resonance (MR) systems, which provide enhanced soft
tissue resolution and incremental diagnostic information from
functional MR methods such as diffusion-weighted imaging, hold
promise for increased accuracy in CRC staging, restaging, early
detection of recurrence, and accurate treatment planning for
radiotherapy (13).
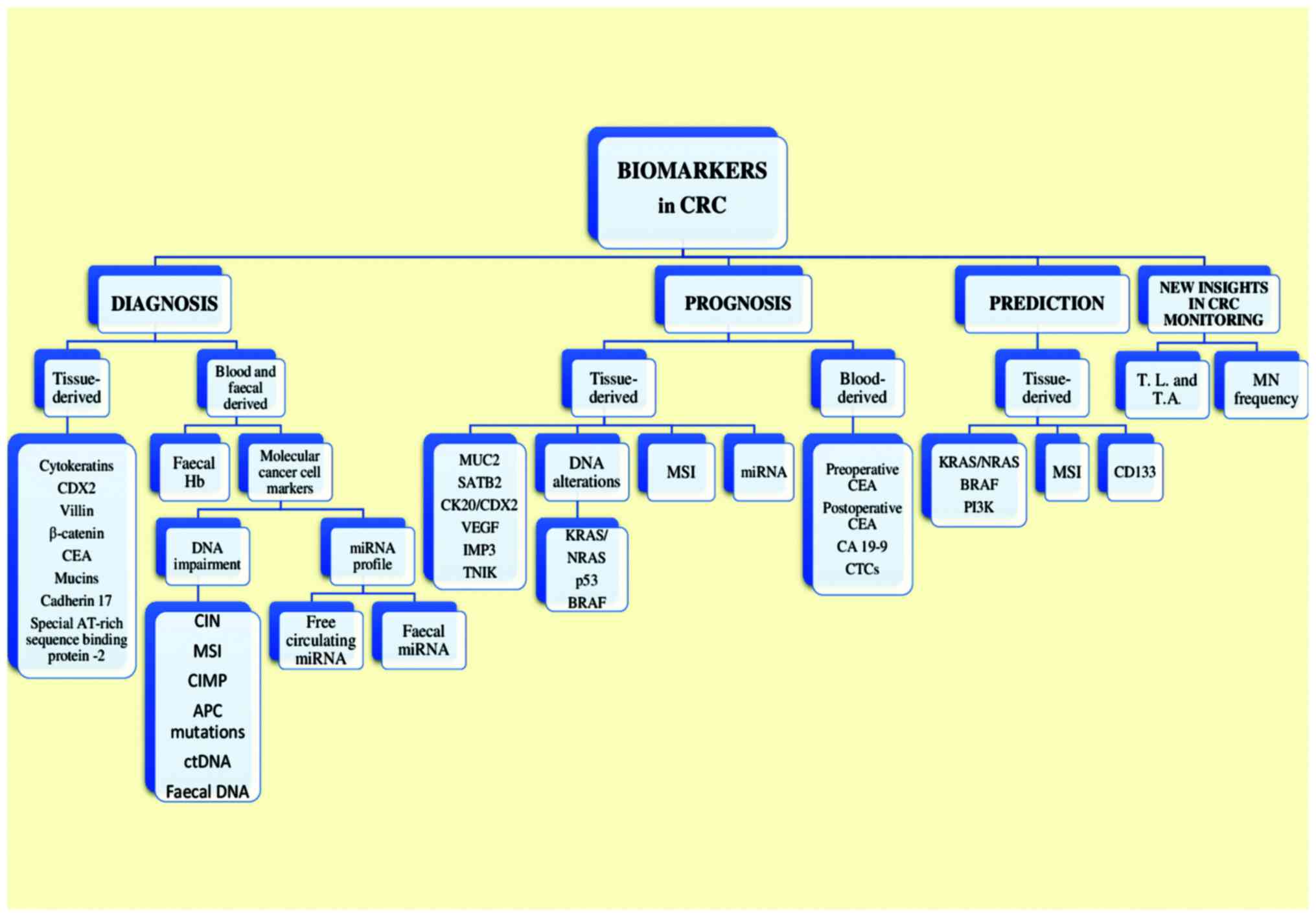 | Figure 2.A graphic overview of the current and
potential biomarkers used in CRC. CRC, colorectal cancer; CDX2,
caudal type homeobox 2; CEA, carcinoembryonic antigen; CIN,
chromosomal instability; MSI, microsatellite instability; CIMP, CpG
island methylation phenotype; APC, adenomatous polyposis coli;
ctDNA, circulating tumor DNA; miRNA, microRNA; SATB2, special
AT-rich sequence binding protein 2; CK, cytokeratin; VEGF, vascular
endothelial growth factor; IMP3, insulin-like growth factor-II
mRNA-binding protein 3; TNIK, Traf2- and Nck-interacting kinase;
BRAF, B-rapidly accelerated fibro-sarcoma (proto-oncogene); MSI,
microsatellite instability; CA 19-9, cancer antigen 19-9; CTCs,
circulating tumor cells; PI3K, phosphoinositide 3-kinase; MN,
micronuclei. |
Diagnostic markers
General
Concerning the detection of CRC in the general
population, the screening methods most commonly offered until now
have included faecal occult blood testing (FOBT), flexible
sigmoidoscopy and colonoscopy while CT colonography is a more
recent addition to the CRC screening modalities (14). Even though screening has clearly
proved to decrease the risk of CRC-associated mortality, screening
effectiveness is restricted by limitations of test performance,
inadequate access to CRC screening tests and loose screening
compliance. Consequently, a great number of patients at the time of
diagnosis present with locally advanced or metastatic disease, a
phenomenon that is observed even among prosperous nations,
including the United States (15).
In accordance to that, CRC researchers focus their research on
innovative ideas to identify molecular markers for the development
of highly accurate, non-invasive screening tests for CRC in the
hope of increasing the compliance of the population and to decrease
potential unwanted side-effects which accompany the more invasive
techniques. Several molecular classes have been tested for their
potential use in CRC screening: DNA (16–18),
proteins (19), messenger RNA
(mRNA) (20) and microRNA (miRNA)
(21–24), and have all proven to be quite
promising in early phase biomarker studies (25). However, until now, only two tests
(faecal haemoglobin and DNA-based markers) meet the pre-clinical
and clinical criteria required for their efficient transduction
from the laboratory to the clinical setting. In fact, recently, a
multi-target stool DNA (MT-sDNA) test has proven better
sensitivity, although with lower specificity, to faecal haemoglobin
by immunochemical testing for the detection of curable-stage CRC
and advanced adenomas while exhibiting overall cancer detection
similar to colonoscopy (26). As a
result, stool DNA testing was approved in the United States by FDA
for general population screening of average risk, asymptomatic
individuals in 2014.
Invasive techniques and diagnostic tissue
markers
The most commonly invasive technique used,
colonoscopy, provides the clinician a great advantage, to evaluate
in real time the presence/absence of a polyp and to resect it at
the same time, if possible, thus representing the standard tool of
practice for CRC evaluation (10).
Resecting a polyp gives the clinician an opportunity to use
immunohistochemical (IHC) staining, typically used in the diagnosis
of gastrointestinal neoplasms, in order to facilitate accurate
tumor classification. By doing that, two main goals are set: The
first one is to exclude morphologic mimickers or to identify the
closest morphologically tissue or organ of origin in cases of
metastatic carcinoma of unknown origin and thus confirm the
diagnosis. The second one is to help estimate the most likely
prognosis and even predict response to a given chemotherapy or
novel molecular-targeted therapy (15,26).
Diagnostic tissue biomarkers therefore provide additional and
fundamental information that complement clinical colonoscopy
findings.
Cytokeratins (CKs)
CKs, proteins expressed by epithelial cells, are
members of the intermediate filaments family along with vimentin,
desmin, neuro-filament, and glial-filamen. Numerous studies have
attempted to identify a possible expression pattern of CKs and
connect it with either the organ of origin (in order to determine
whether it is a primary CRC) or with tumor progression. However, as
more and more studies are conducted it is becoming clear that such
a connection is not likely to be identified in the near future. To
begin with, CK7 and CK20 are helpful when the clinician needs to
differentiate metastases from CRC, which are usually
CK7−/CK20+, from other tumors (27). CK20 almost selectively stains the
normal gland cells of the colonic mucosa and Merkel cells while its
expression is rarely may be seen in the urothelium or other mucosas
(28,29). By contrast, CK7 is usually expressed
in urinary bladder and female genital tract epithelia, mesothelium,
normal lung, and, rarely, it may be observed in gastric and
intestinal normal glands. However, the majority of researchers
agree that it is not found in normal colonic mucosa (28,30).
Based on these findings, the immunophenotype CK7/CK20 is used as a
routine in order to differentiate CK20-expressing metastasis of
colorectal adenocarcinomas from lung, ovarian or bladder
carcinomas, which are usually stained with CK7 (31). However, it is reported that
non-neoplastic colonic mucosa proximal to the rectum exhibits a
CK7−/CK20+ phenotype, as is the case for 90%
of CRCs (32). When CK17 is
included in the diagnostic panel, the efficacy of the test is
improved as less than 10% of CRCs express CK17 in contrast to other
carcinomas that are more often positive for CK17 (including
stomach, endometrium and urine bladder). In addition, pancreatic
ductal carcinomas are consistently positive and a number of
carcinomas from other sites, may exhibit CK17 expression (33). Furthermore, when CRC progression is
studied, CK20 and CK7 can be useful. Results indicate that advanced
CRCs were more often CK20+/CK7+ compared to
early-stage cancers, which were predominantly
CK20+/CK7−. Thus, CK7 expression may be a
differentiating marker for the progression of CRC (34).
Caudal type homeobox 2 (CDX2)
CDX2 is a transcription factor encoded by
CDX2 gene, a member of the caudal subgroup of homeobox
genes. Its main role is to ensure maintenance of a cellular
intestinal phenotype during the in utero and ex utero
life (35). CDX2 presents strong
expression patterns in epithelia of the normal small intestine,
appendix, colon, and rectum as well as in the pancreatic
centroacinar and interacinar ductal cells (36). It is revealed that loss of CDX2 may
give birth to human CRC. CRCs, beside those exhibiting MSI, are
consistently CDX2-positive (37).
In fact, a quite interesting research recently investigated the
effect of restoration of CDX2 expression on colon cancer cell
viability, colony formation, cell cycle distribution, apoptosis,
invasion ability and xenograft tumor growth in nude mice (38). According to the researchers, CDX2
upregulation significantly reduced the life span and inhibited
colony formation, and the invasion and migration ability of LoVo
cells. Moreover, it was able to induce cell cycle arrest and
apoptosis in vitro, especially under hypoxic culture
conditions (38). According to data
from histological studies, expression patterns of CDX2 are found in
a variety of neoplastic tissues such as adenocarcinomas that
exhibit intestinal-type differentiation, including adenocarcinomas
of the gastroesophageal junction, bladder, urachus, small bowel,
pancreas, appendix, and ovary (37).
Villin
Villin is a cytoskeletal actin-binding protein that
is associated with the microfilament family. It is normally found
in cells that exhibit highly specialized, brush border-type
microvilli, similar to enterocytes (39). Villin is associated with elimination
of polarity that the epithelium exhibits, thus altering tissue
architecture (40). Thus, in CRCs
containing a micropapillary pattern, villin IHC has been effective
for tracking the polarity in this type of CRC (41). The specificity of villin as a marker
of intestinal origin is limited, similar to CDX2, as positivity may
also be marked in adenocarcinomas with intestinal differentiation
arising from a wide variety of organs including stomach, lung, and
ovary as well as in malignancies of other sites such as the
endocervix and liver (the latter more rarely, though) (42).
β-catenin
β-catenin is a multifunctional protein that is
involved both in cell adhesion and intracellular signaling, with
the latter being accomplished through β-catenin's actions through
the Wnt signaling pathway (43).
Activation of the Wnt signaling pathway (Fig. 3) increases the cytoplasmic pool of
free β-catenin and, to a smaller extent, the nuclear pool where it
induces proliferation. The Wnt signaling pathway in its normal form
is highly activated in the majority of CRCs as a result of mutant
adenomatous polyposis coli (APC) or β-catenin. Moreover,
upregulated Wnt signaling has a key role in the pathogenesis of CRC
(44). In the absence of functional
APC, as it often happens in CRC, nuclear β-catenin can be
identified immunohistochemically (44,45).
Although nuclear expression of β-catenin is not unique to CRC, it
can be proven useful as part of a diagnostic panel.
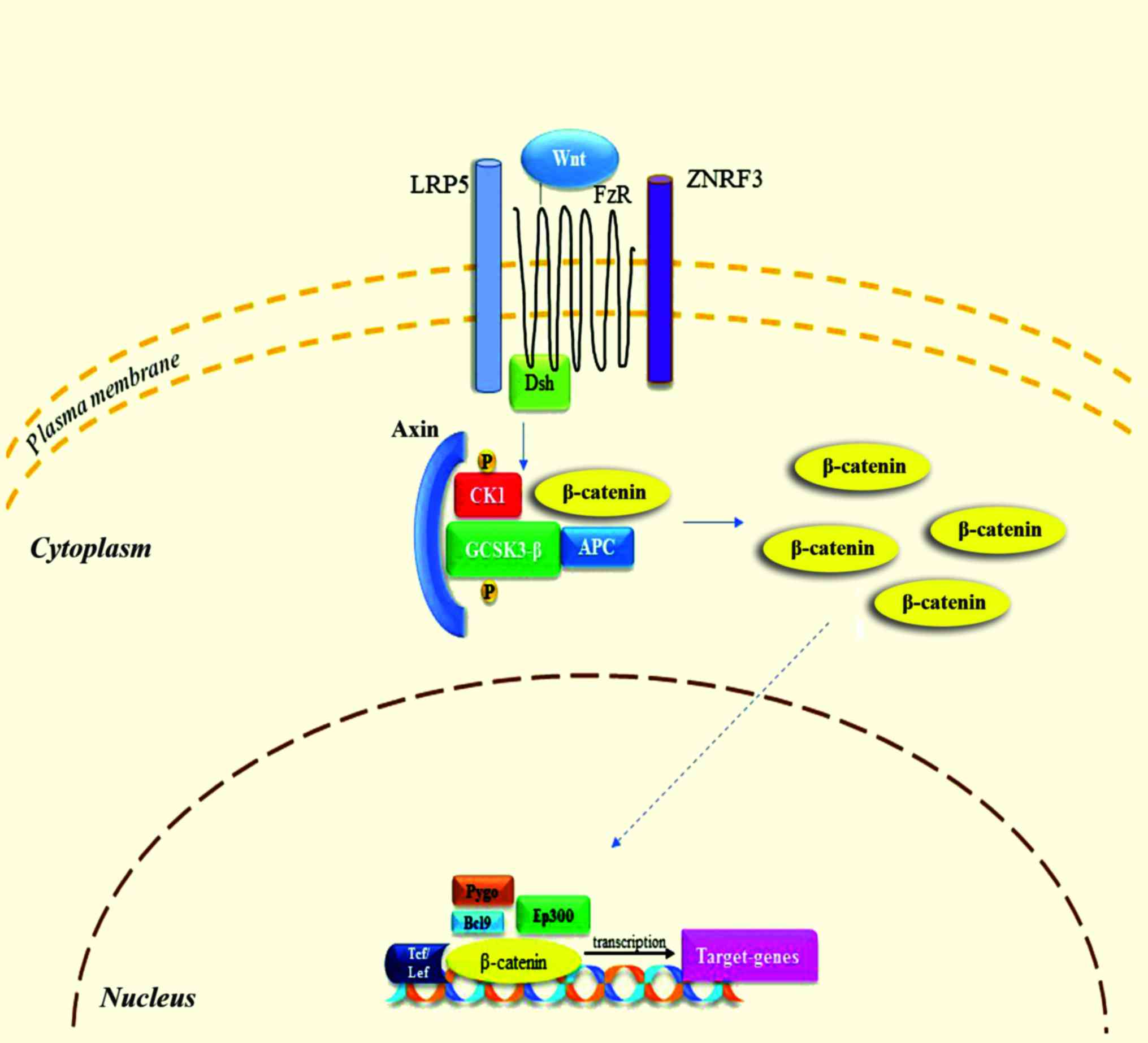 | Figure 3.Wnt/β-catenin signaling pathway. When
Wnt is present, it binds to FzR and LRP5 receptor. Dsh enables the
phosphorylation of GSK-3β and CK1, resulting in the binding of
axin. Following to accumulation of β-catenin and translocation to
the nucleus, it binds to various factors in order to modulate the
transcription of target-genes. These processes lead to
antiapoptotic results and promote cellular proliferation. FzR,
frizzled receptor; LRP5, low-density lipoprotein receptor-related
protein 5; GCSK3-β, glycogen-synthase kinase 3β; CK1, casein kinase
1; ZNRF3, zinc and ring finger 3; APC, adenomatosis polyposis coli;
Pygo, pygopus; Bcl9, B-cell CLL/lymphoma 9; Ep300, E1A binding
protein p300; Tcf, T-cell factor; Lef, lymphoid enhancer-binding
factor 1. |
Carcinoembryonic antigen (CEA)
CEA are membrane-associated glycoproteins playing a
number of roles in cell adhesion or signal transduction (46). Monoclonal CEA (mCEA) may be
expressed in a wide variety of adenocarcinomas, including those
arising from the colon (47).
Although this lack of specificity limits its value as a diagnostic
marker of CRC, it remains a useful component of a broad diagnostic
panel. In a meta-analysis study by Tan et al (48) based on 20 different studies, serum
CEA has been demonstrated to comprise an exam of elevated
specificity, although sensitivity was inadequate when tracing CRC
recurrent conditions. The cut-off range varied among these studies
from 3 to 15 ng/ml, suggesting that a measure of 2.2 ng/ml would be
the optimal regarding sensitivity and specificity (48). Evidently, at this point circulating
CEA may constitute a primary means of recording progress in
patients' surgical follow-up, in accordance with the complementary
tools including clinical picture, radiological response and
histological results.
Mucins
Mucins are high molecular-weighted, heavily
glycosylated proteins (49). Within
the polypeptide of these glycoprotein molecules tandemly repeated
sequences of amino acids rich in threonine and serine moieties lead
to various combinations of these repeats making each mucin type
unique. In colon, a mixture of neutral, sialomucin, and sulphomucin
are normally met, with MUC2 being the most prominent, primarily in
goblet cells. MUC4 is also abundant in the colon and its expression
is found both in goblet and columnar cells, whereas MUC3 appears to
be expressed primarily within enterocytes (50). MUC1, MUC5AC, and MUC6 under normal
conditions are not expressed in the colonic mucosa (50). MUC2 is frequently expressed in
mucinous CRC as well as mucinous carcinomas of the ovary, breast,
and pancreas (50). Gastric mucins
may also be expressed in CRC (51).
MUC5AC expression is associated with mucinous differentiation and
MSI, while most mucinous carcinomas exhibit a
MUC2+/MUC5AC+ phenotype (52). Additionally, MUC21 has recently
proven to be expressed in CRCs (53). Notably, although a correlation
between mucin expression and clinical characteristics was observed
by Wang et al (54) an
increased expression of MUC5 was found to be associated with poor
cellular differentiation, lymph node metastasis, advanced tumor
stage and a poor overall prognosis in CRC. By contrast, a decreased
expression of MUC2 was reported in cases with lymph node
metastasis, poor cellular differentiation and an advanced tumor
stage in CRC. These results suggest that MUC2 and MUC5 levels could
be associated with tumor progression and even be used in order to
facilitate the early diagnosis and clinical characterization of CRC
(54).
Cadherin 17 (CDH17)
CDH17 was first described in the liver and intestine
of rats (55). Later on, the
research in humans revealed that its expression is limited to the
intestine (both small and large) and in a part of the pancreatic
duct. As for its function, it serves as an intestinal peptide
transporter (56). However, its
clinical applicability in GI tumors diagnosis was only recently
recognized (57). Recent data
indicate that positive CDH17 immuno-reactivity is frequently seen
in colorectal adenocarcinomas up to 96% and to a significant
portion of gastric (58),
pancreatic, and biliary adenocarcinomas (25–50%) (59). By contrast, it is rarely found in
adenocarcinomas arising from extra-GI tract (1–10%). Notably, even
though CDH17 is transcriptionally regulated by CDX2, it is more
sensitive and specific than CDX2 for the identification of primary
and metastatic colorectal adenocarcinoma (57–59).
CDH17 also seems to be useful in diagnosing CRC variant with poorly
differentiated or even undifferentiated morphology, such as
medullary carcinoma, which characteristically lacks the expression
of conventional intestinal differential markers, such as CK20 and
CDX2 (57). In a study where the
effect of the downregulation of CDH17 upon human colon cancer cells
was examined, the researchers found that the downregulation of
extrinsic CDH17 gene can conspicuously promote
apoptosis-inducing effects of noscapine on specific human colon
cancer cell lines, which may indicate a novel strategy to improve
the chemotherapeutic effects on colon cancer (60).
Special AT-rich sequence binding
protein 2 (SATB2)
SATB2 is a member of the nuclear matrix-associated
transcription factors family whose role is to serve as epigenetic
regulators of gene expression with high tissue selectivity
(61). Research has shown that
SATB2 has a variety of biologic functions. However, the exact role
of SATB2 in the GI tract is still unknown. Recently, Magnusson
et al (61) suggested that
SATB2 immunoreactivity was mainly encountered in the glandular
lining cells of the lower GI tract, including appendix, colon, and
rectum. In fact, SATB2 is a highly sensitive and specific marker
for adenocarcinomas of the colon and rectum, with a diagnostic
sensitivity of 97% (121 of 125 cases) in CRCs and of 81% in CRC
metastases (61). Moreover, Bian
et al (59) found that the
combination of CDH17 and SATB2 serves as potential optimal markers
for the differential diagnosis of pulmonary enteric adenocarcinoma,
a rare type of non-small cell lung cancer exhibiting similar
histological and immunohistochemical morphology to colorectal
adenocarcinoma, and metastatic colorectal adenocarcinoma (59).
Non-invasive techniques and diagnostic
blood-derived and faecal markers
Faecal heamoglobin detection
tests
Until recently, non-invasive techniques used for
diagnostic purposes were represented mainly by the guaiac faecal
occult blood test (gFOBT) and faecal immunochemical test (FIT). The
gFOBT detects blood loss from peptic ulcer and gastrointestinal
cancer. However, its validity in terms of CRC diagnosis, is rather
restricted as gFOBT is, not only unable to distinguish bleeding
between the upper and the lower GI tract, but also lacks the
ability to specifically distinguish human from non-human haeme
(8,10). Moreover, gFOBTs are not sensitive to
small bleedings, while specificity can be affected by diet or drugs
and they have a fixed hemoglobin concentration cut-off determining
positivity. As a result, gFOBT exhibits low sensitivity in terms of
the detection of cancerous and preneoplastic lesions (30–40%)
(62), whereas the sensitivity to
detect an advanced adenoma (polyp, >9 mm; villous features or
high grade dysplasia) is only 18% (63). On the other hand, FIT is able to
detect human globin by means of an antibody-based assay providing a
qualitative or quantitative result (depending on the kit used)
expressed as faecal haemoglobin concentration per gram of faeces.
FITs are analytically more specific, capable of quantitation and
hence provide flexibility to adjust cut-off concentration for
positivity and the balance between sensitivity and specificity.
FITs are clinically more sensitive for cancers and advanced
adenomas, and because they are easier to use, acceptance rates are
higher (64). However, FIT can only
detect bleeding from colonic pre-neoplastic lesions. Thus, when
used for CRC screening, the sensitivity FITs demonstrates in
detecting adenomas >1 cm in diameter is only 20–30%, while for
advanced neoplasia or large adenomas sensitivity ranges between 20
and 67%, which is comparable or superior to the sensitivity of
gFBOT (64,65). Another very important aspect that
limits occult blood testing is the fact that it detects
significantly more in the left- than right-sited lesions in the
colon (66), which is a significant
issue given the increased incidence of right-sided CRCs that has
developed over the last two decades (67). In other words, it is evident that
those two non-invasive diagnostic techniques are limited by their
intrinsic lack of sensitivity and specificity (with the latter
being the case for gFBOT) making them unable to stand on their own
as diagnostic tools. Table I
presents a summarization of bibliographical references regarding
FOBT sensitivity and specificity.
 | Table I.A summarization of bibliographical
references to FOBT sensitivity and specificity. |
Table I.
A summarization of bibliographical
references to FOBT sensitivity and specificity.
| Author/(Refs.) | Faecal occult blood
testing |
|---|
| Kronborg et
al (185), Scholefield et
al (186) | Reduction in CRC
mortality with gFOBT biannually [relative risk reductions of 13%
(UK trial) and 16% (Danish trial)] |
|
| No significant
reduction in overall mortality |
|
| gFOBT: Low
sensitivity for CRC detection (UK trial, 45%; Danish trial,
54%) |
|
| True-positive rate:
50% (UK and Danish trials) |
|
| False-positive
rate: 5–10% (UK and Danish trials) |
|
| True-negative rate:
90–95% (UK and Danish trials) |
|
| False-negative rate
50% (UK and Danish trials) |
| Medical Advisory
Secretariat (187), Dancourt et
al (188), Faivre et al
(189) | iFOBT sensitivity
superior to those of gFOBT for CRC detection: Two studies showed
sensitivity for gFOBT, 13 and 25%, respectively; pooled iFOBT
sensitivity, 81% iFOBT and gFOBT: Lower sensitivities for adenoma
detection than for CRC detection: Rehydrated gFOBT, 22%; pooled
iFOBT, 28% |
| Lin et al
(190) | FIT sensitivity,
73.8% (95% CI, 62.3 to 83.3) for quantitative (n=9,989) test
categories; 78.6% (95% CI, 61.0 to 90.5) for qualitative (n=18,296)
test categories |
| Koo et al
(191), Moss et al
(192) | Positive predictive
value of FIT > positive predictive value gFOBT for advanced
adenoma (1.73 vs. 0.35%) and all neoplasias (3.74 vs. 0.76%) FIT
detects twice more CRCs and advanced adenomas |
| Gonzalez-Pons and
Cruz-Correa (8) | gFOBT: ↓ ability to
define bleeding between upper/lower GI tract |
| Kuipers et
al (10) | gFOBT: ↓ ability to
distinguish human haeme |
| Valori et al
(62) | gFOBT: Νot
sensitive in small bleedings |
|
| gFOBT ↓ sensitivity
in detecting cancerous/preneoplastic lesions |
|
| gFOBT: Specificity
affected by diet/drugs |
| Lieberman et
al (63) | gFOBT: 18%
sensitivity in detecting advanced adenomas |
| Whitlock et
al (65), Young et al
(64) | FITs sensitivity
for advanced adenomas: ~20–67% (↑ than FOBT) |
| Dancourt et
al (188) | FIT detects more
CRC and advanced neoplasia than gFBOT (similar positive predictive
value) |
| Imperiale et
al (25) |
|
| Rozen et al
(193) | Comparative
performance of gFOBT and FIT depends on the number of samples and
threshold chosen for the quantitative FIT |
| Hoffman et
al (194) | Screening adherence
with FIT was higher than with gFOBT (61.4 vs. 50.5%) |
| Brenner and Tao
(195) | Sensitivity of FITs
for detecting CRC/any advanced neoplasm/any neoplasm: 2–3 times
higher than gFBOT |
|
| Increased levels of
FITs specificity vs. gFOBT |
| Fitzpatrick-Lewis
et al (196) | iFOBT vs. gFOBT on
mortality outcomes: iFOBT has higher sensitivity and comparable
specificity (insufficient evidence from RCTs) |
| Murphy et al
(197) | Total financial
burden: Lower for FIT at any threshold (expressed in µg Hb/g
faeces) than for gFOBT, and this difference increases as the FIT
threshold is decreased (Cohort-based Markov state transition
model) |
| Lee et al
(198) | FIT sensitivity,
79%; FIT specificity, 94% |
| Morikawa et
al (66) | gFOBT detect
notably more lesions in the left (compared to the right colon) |
Blood-derived and faecal molecular
cancer cell markers
Our improved knowledge of the molecular pathogenesis
that conditions the polyp→adenocarcinoma progression sequence has
made clear that the molecular changes found in polyps and
adenocarcinomas have the potential to serve as neoplasm-specific
molecular markers for these lesions. The concept of using these
molecular markers for CRC screening is indeed the next step for a
well-established non-invasive detection method for CRC. In contrast
to heme detecting tests, tests focusing on molecular markers
derived from neoplastic cells of the colon can prove to be more
accurate. CRCs are known to bear distinguishable genetic and
epigenetic changes as they develop and progress, which forms the
rational of stool-based DNA and RNA testing.
DNA impairment
Approximately 90% of CRCs develop sporadically, and
only a few cases (<10%) are hereditary, with familial
adenomatous polyposis, hereditary non-polyposis colorectal cancer
(HNPCC), MUTYH-associated polyposis, Peutz-Jeghers syndrome (PJS)
and serrated polyposis syndrome being the main representatives of
hereditary causes of CRC. Currently, three major paths for CRC
development have been described, with CIN being the most common
accounting for 70–80% of CRCs, the MSI pathway positioned in the
second place accounting for 5–20% of tumors and in the third place
the CIMP, which represents approximately 15% of CRCs (68). These pathogenetic pathways can be
examined either in serum samples (CIN, MSI, CIMP, APC) using
immunohistochemical techniques or in faeces (CIMP, APC).
a) CIN
CIN is the hallmark characteristic of most CRC cases
(80–85%), and its main characteristic is the extensive abnormality
in chromosome number (aneuploidy) and loss of heterozygosity. CIN
can be observed in several forms, including chromosomal numerical
abnormalities, small sequence modifications such as base deletions
or insertions, chromosomal rearrangements and gene amplification
(5).
b) MSI
Microsatellites (MSs) are short tandem-repeated base
pairs of 1–6 scattered all over the genome. For the normal human
genome the number of MSs is approximately half a million. Genome
studies revealed that MSs are prone to duplication errors. However
these errors are usually corrected by the MMR system (5,69,70).
Consequently it is logical to assume that a defective MMR system
would result in the accumulation of DNA mistakes and thus MSI.
Indeed, MSI arises by the inhibition of MMR system either via
defective methylation of MLH1 in CpG island or point
mutation of any MMR genes (hMLH1, hMSH2, hMSH6, PMS1 and
PMS2) especially hypermethylation of hMLH1 promoter
(5). It is estimated that about
15–20% of CRC patients present MSI with a small fraction of which
2–4% are related to HNPCC (71). In
order to estimate MS status, Bethesda panel was agreed in which
five MS loci were included (BAT25, BAT26, D5S346, D2S123,
and D17S250) (72). However,
some researchers suggested an expanded Bethesda panel include 10
loci. Based on this panel, MSI can be divided into three groups:
MSI-high (MSI-H), defined as having ≥30% unstable loci using
mononucleotide or dinucleotide markers (8,73);
MSI-low (MSI-L), with 10–30% unstable loci; and microsatellite
stable (MSS), with <10% unstable loci (73). MSI status varies according to a
given CRC stage: Stage II CRC exhibits high prevalence of MSI (20%)
while in stage IV CRC MSI is approximately 4% (74,75).
Moreover, differences based on the MSI status are found when
prognosis is examined. For example, cases with MSI-H CRC share a
better prognosis than that with MSS CRC (5,76).
c) CIMP
It is well accepted that DNA methylation is a key
process for the normal growth of eukaryotes. If occurs in cytosine
number 5 within CpG island which is present in 50% of tumor
suppressor gene promoters (77).
CpG island accounts for >70% of CG sequences that extend to 0.4
kB on the genome (78). Even though
hypermethylation of CpG island cytosine represents a hallmark for
cancer progression, both hypomethylation and hypermethylation may
lead to the transformation of normal mucosa to adenoma and
subsequently to the development of CRC (79). Disturbance of epigenetic programming
(epigenetic modification including DNA methylation, histone
modification and post-transcriptional gene regulation) is closely
related to the development of CRC (80). It is reported that a wide spectrum
of aberrant methylated genes in CRC, regulates crucial functions in
the normal cell regarding proliferation and maintenance of genome
stability (77). In the clinical
setting, abnormal DNA methylation patterns can be detected in
patient's blood or faeces samples from which CRC cells are
examined. A number of abnormal methylated genes with variable level
of specificity and sensitivity can serve as diagnostic biomarkers
in CRC patients. These genes include WiF-1, AIX4, PGR, FBNI,
P53, TIMP3, SEPT9, MGMT, Vimentin and GATA4 (77,80). A
number of studies have indicated that instead of looking for a
single gene methylation pattern, using a panel of several
methylated genes is better in terms of specificity and sensitivity.
Lind et al (81) tested a
panel of epigenetic biomarkers for use as CRC and adenoma
screening. In this study, approximately 523 human samples were
examined using a gene panel consisting of: CNRIP1, FBN1, INA,
MAL, SNCA and SPG20. According to their study, high
level of sensitivity and specificity was achieved. Hence, those
authors suggested that a combination of the six genes may serve as
a non-invasive biomarker with high specificity and sensitivity for
early diagnosis of CRC (81).
d) APC mutations
APC gene regulates the Wnt signaling pathway
via encoding a multifunctional protein. Specifically, APC
regulates Wnt pathway through the destruction of the transcription
factor β-catenin, which enhances the activity of Wnt pathway.
Hence, APC conversely organizes Wnt signaling (82). In addition, APC gene is
involved in cell cycle regulation, cytoskeleton stabilization,
intracellular adhesion, as well as apoptosis. In an attempt to
determine the exact role of APC gene in CRC, Dow et
al (83) investigated whether
APC mutation is essential for CRC protection. For their
study a CRC mouse model with inhibited APC was used.
According to their findings, inhibiting APC gives rise to
adenomas in colon and small intestine (83). In general, about 90% of CRC patients
demonstrate APC gene mutation, which highlights its
applicability as a molecular biomarker for CRC diagnosis (8). Liang et al (84) conducted a meta-analysis study to
correlate APC polymorphism (D1822V, E1317Q, I1307K)
and CRC risk. They concluded that E1317Q significantly
increased adenoma risk. However, I1307K is associated with high
risk of CRC (84). CRC-related
tumor suppressor genes are thought to be altered in the early phase
of cancer development, and APC mutation is the first step in
the translation of normal mucosa to neoplastic tissue, leading to
the activation of the WNT pathway. Subsequent mutations that occur
in genes, such as KRAS, TP53, SMAD4 and type II TGF-β
receptor (TGFBR2), lead to the progression from polyp to
cancer similar to the process that takes place in other
gastrointestinal carcinomas (85).
e) Circulating tumor DNA (ctDNA)
Circulating free DNA (cfDNA) is thought to be a
natural phenomenon, existing as a result of cellular apoptosis and
necrosis of both normal and cancer cells, with secretion from
cancer cells being an additional source that remains to be
elucidated (86). ctDNA, a subclass
of a patient's cfDNA, is the material used for testing in order to
identify potential DNA impairments that have been associated with
CRC. Owing to the circulating DNAases, the half life ctDNA is
restricted to a few hours allowing clinicians to have a more
accurate look at a patient's cfDNA profile. Findings have shown
that the cfDNA quantity is quite higher in patients with cancer
than that in healthy individuals. Moreover, cfDNA quantity seems to
be positively correlated with the cancer stage (87). Of note, benign tumors or
non-neoplastic lesions in general do not lead to ctDNA avoiding,
therefore, a potential pitfall of false positives (88). To date, the main DNA defect that is
studied is the hypermethylated SEPT9 gene in the plasma.
However, following the initial attempts to set a diagnostic tool
based solely on SEPT9, researchers realized that combining
multiple gene loci in a single panel in order to achieve better
sensitivity and specificity is optimal. For example, Tänzer et
al (89) showed that the
combination of SEPT9 with ALX4 achieved a sensitivity
of 71% and a specificity of 95% for advanced adenomas, thus
supporting SEPT9/ALX4 as a biomarker for precancerous
lesions (89). In parallel to that,
apart from SEPT9, a number of genes have been investigated
including HJC1, CYCD2, PAX5, RB1, SRBC, NPY, PENK, WIF1, ALX4,
HLFT, HPP1, MLH1, APC, CDKN2A/P16h, TMEFF2, NGFR, FRP2, NEUROG1
and RUNX3 (90).
f) Faecal DNA
As with circulating DNA, stool DNA is examined for
the presence of abnormalities in specific genes. Stool-based assays
have proven to be the most efficient assay type for a number of
reasons. Direct histologic observations show that CRCs and polyps
exfoliate neoplastic cells and their debris into the mucocellular
layer of the colonic lumen at a steady continuous rate (91). However, at first sight, one would
wonder how exfoliated cells arising from a right-sided polyp or CRC
could survive in the intraluminal environment in order to be
detected. Indeed, most of these cells go through lysis, preserving
however to a certain extent their DNA content thus allowing a DNA
analysis to be conducted. To date, along with mutant KRAS a variety
of hypermethylated genes including APC, ATM, BMP3, CDKN2A,
SFRP2, GATA4, GSTP1, HLTF, MLH1, MGMT, NDRG4, RASSF2A, SFRP2,
TFPI2, VIM and WIF1 have been analyzed in faecal DNA for
the early detection of CRC, with SFRP2 and VIM
proving to be the most promising ones (92,93).
When these tests for both mutant and methylated DNA markers and
FIT, collectively known as MT-sDNA, are combined, it is proven that
they demonstrate the best clinical performance of CRC molecular
marker screening assays to date.
Blood and faecal miRNA profile
miRNAs are single-stranded small non-coding RNAs
that are 18–25 nucleotides in length. Their role remained unknown
until 1993 when it was recognized that they act as negative
post-transcriptional regulators in Caenorhabditis elegans
(94,95). Until 1993, miRNA (as any other
member of the non-coding RNA family) was considered to be a useless
RNA product of ‘junk DNA’. However, it was shown that this is not
the case as it was proven that it is able to regulate gene
expression at a post-transcriptional level, either by blocking mRNA
translation or by inducing their degradation. By binding miRNA to
its target mRNA, miRNA can trigger the degradation of its target or
otherwise inhibit its translation into protein, with the degree of
sequence complementarity between the miRNA and mRNA determining
which mechanism is employed (96–98).
Over the last few years, research in human cancer focusing on the
potential of miRNA to serve as a biomarker has increased
dramatically mainly due to their unique properties. First of all,
miRNAs are very stable under a variety of conditions both in the
experimental and laboratory setting. Moreover, RNases cannot
degrade them owing to their small size and the hairpin-loop
structure (99). In addition to
that, cell-free miRNAs occur in large numbers as they are packed in
high density lipoprotein particles, apoptotic bodies,
microvesicles, exosomes or by their binding to argonaute-2,
properties that synergistically result in an increased stability
(100,101). Thus, it is easy to isolate them
from different forms of clinical specimens (99). Lastly, miRNAs are actively secreted
by cancer cells into the circulatory system and digestive tract
(102). Hence, in the clinical
setting, circulating cell-free miRNAs and faecal miRNA are the main
forms of RNA used as diagnostic biomarkers.
a) Blood-derived (circulating)
cell-free miRNA
Various studies have shown that a series of miRNAs
are pathologically excreted in CRC plasma or serum samples.
However, due to the fact that standardized techniques for miRNA
extraction, normalization and quantification are yet to be found,
one needs to bear in mind that most of the available results in
literature are not easily reproducible thus making sometimes
controversial. Circulating cell-free miRNA was first evaluated in a
more comprehensive and systematic way in patients with CRC by Ng
et al (103) in 2009 who
found an altered miRNA expression profile in tissue and plasma
samples from CRC patients and healthy subjects. However, a very
important observation was that two miRNAs, miR-92a and miR-17-3p,
were able to indicate patients with CRC, differentiating from
healthy subjects, based on their high expression profile
(sensitivity: 64 89%, specificity: 70, 70% for each miRNA,
respectively) (103). Since then,
numerous miRNAs have been identified as indicators of CRC. However,
even though a few single miRNAs have proven to be enough to make
the distinction with increased sensitivity and specificity, the
addition of even more dysregulated miRNAs into a single panel
usually achieved better diagnostic results. Indicatively, some of
the dysregulated miRNAs and their testing samples are described:
miR-17-3p plasma, miR-18 plasma, miR-21 plasma, miR-21 serum,
miR-21 serum (exosome), miR-29a plasma, miR-92a plasma, miR-92a
serum, miR-155 serum, miR-200c plasma, miR-221 plasma, miR-21,
miR-31, miR-92a, miR-181b, miR-203, let-7g (panel), miR-7, miR-93
and miR-409-3p (panel) (104).
However, only three upregulated miRNAs, miR-19a-3p, miR-21 and
miR-92, were identified as promising diagnostic biomarkers by more
than one study (105,106).
b) Faecal miRNA
Identification of CRC-specific miRNAs is also
feasible in stools. Link et al (107), using RT-PCR and microarray
analysis proved that faeces from patients with CRC and colorectal
adenoma contained higher levels of miR-21 and miR-106a as opposed
to that from healthy controls (107). A subsequent study that assessed
faecal miR-21 and miR-92a levels revealed that faecal miR-92a
expression was able to differentiate patients with CRC or adenoma
from healthy subjects or even those with lower-risk polyps
(22). More interesting data came
from the study of Zhu et al (108) as they found that stool miR-29 was
significantly present in patients with rectal cancer than in those
with cancer in the rest of the large intestine. Thus, the use of a
panel of miRNA expression patterns can create a form of a cancer
fingerprint (108). A sum of the
useful stool-derived dysregulated miRNAs for diagnosis is miR-17-92
cluster, miR-20a, miR-21 up miR-135 miR-144 miR-29a, miR-223,
miR-221, miR-92a and miR-224 (109).
Prognostic markers
General
Once CRC is diagnosed, the clinician has to take
the next step in the clinical management, that is to evaluate the
prognosis for this patient or in other words to estimate the likely
progression of the cancer and the aggressiveness that it may
exhibit (recurrence likelihood, progression and/or chance for
metastasis despite adjuvant therapy). The current practice for
prognosis assessment is based on radiological (CT, MRI) and
pathological (TNM, lymphovascular, perineural and venous invasion)
criteria. In fact, TNM staging remains the strongest prognostic
tool (110). However, not only do
none of the prognostic tools mentioned above provide clear evidence
on which of these CRC cases is more prone to relapse, give
metastases or are proven to be resistant to chemotherapy, but they
also are not suitable for the personalized treatment of each
patient. Thus, much effort has been made for the evaluation of the
potential of several molecules and gene alterations to serve as
prognostic biomarkers, taking us a step closer to a true
personalized treatment.
Tissue-derived prognostic biomarkers
Molecular prognostic biomarkers
Some of the tissue-derived molecular biomarkers
meant for diagnostic purposes, have demonstrated a promising
potential for also serving as prognostic markers. Such markers are
mucins (MUC2), SATB2 protein, CK20/CDX2, VEGF, insulin-like growth
factor-II mRNA-binding protein 3 (IMP3) and Traf2- and
Nck-interacting kinase (TNIK) expression. Even though some MUC2
expression profile studies have yielded mixed results, it is
evident that MUC2 loss of expression may be an indicator of poor
prognosis in both mis-match repair protein (MMR)-proficient and
MLH1-negative CRC (111).
On the other hand, studies on the SATB2 expression profile showed
that an upregulated SATB2 was connected with good prognosis in CRC
and could even increase sensitivity to chemotherapy and radiation,
whereas a downregulated SATB2 in cases of colorectal
adenocarcinomas was associated with poor prognosis, as tumor
invasion, infiltrated lymph nodes and distant metastases were
observed (112,113). Studies on CDX2 revealed that loss
of CDX2 expression is associated with proximal origin, infiltrative
characteristics and advanced TNM stage. Moreover, it was found to
be an independent poor prognostic factor of progression-free
survival (PFS) and overall survival (OS) (114). Vascular endothelial growth factor
or VEGF is one of the main angiogenic factors in CRC as it is
expressed in approximately 50% of the cases Thus, VEGF-1 expression
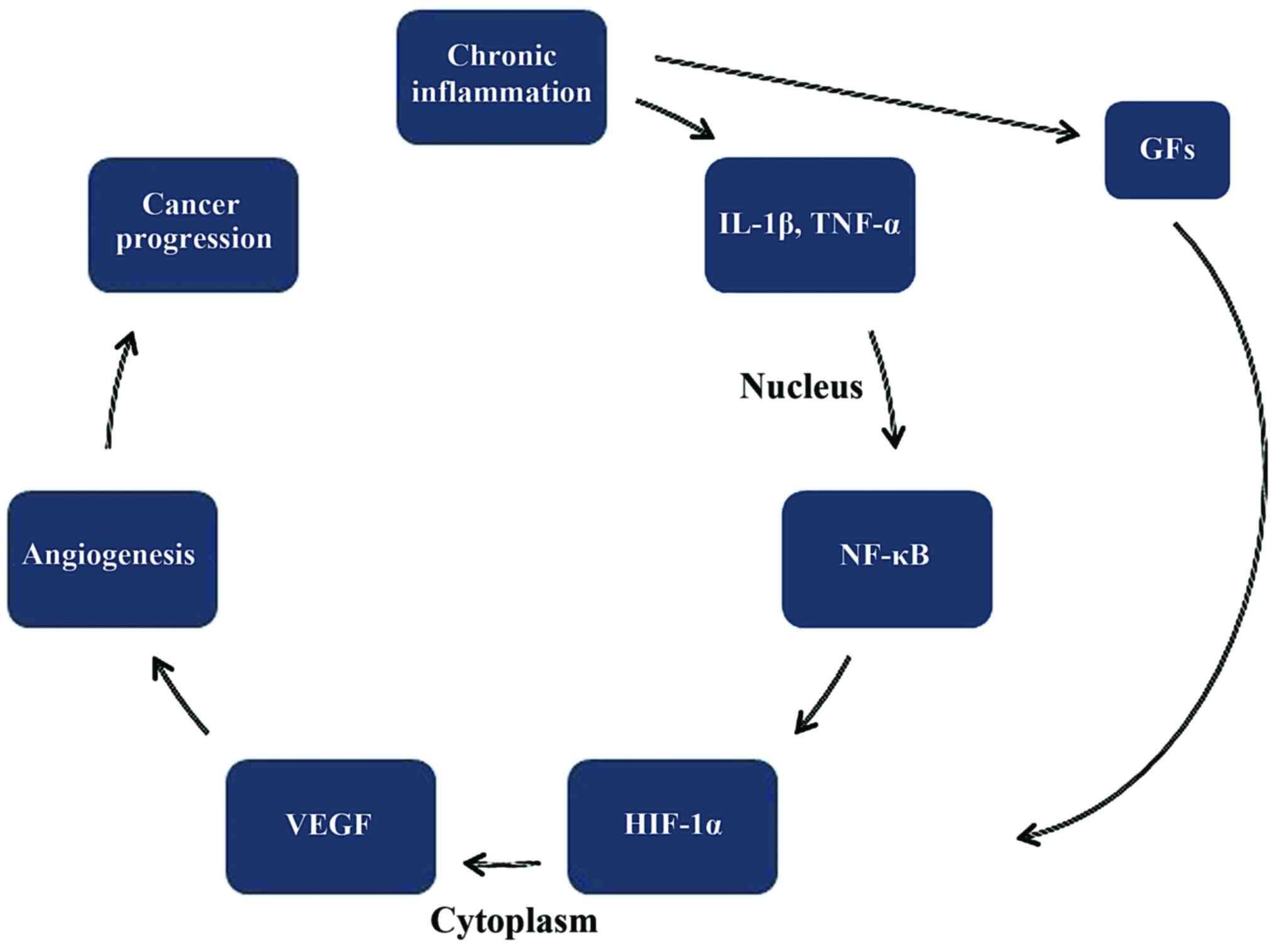
is correlated with a worse prognosis (115). Fig.
4 shows the connection between chronic inflammation and the
development of CRC. Moreover, studies on IMP3 found an increased
expression in colon cancer compared to normal colonic mucosa. Of
note is the fact that its expression was found to be higher in
cases with lymph node infiltration with cancer cells (93%) than in
primary colon cancer (65%) or normal mucosa (3.9%) (116). Lastly, research on TNIK reported
that high levels of TNIK protein in primary tumors could indicate
distant metastasis after surgery of stage II and III CRC patients
as well as invasive characteristics of CRC (117).
DNA alterations with prognostic
value
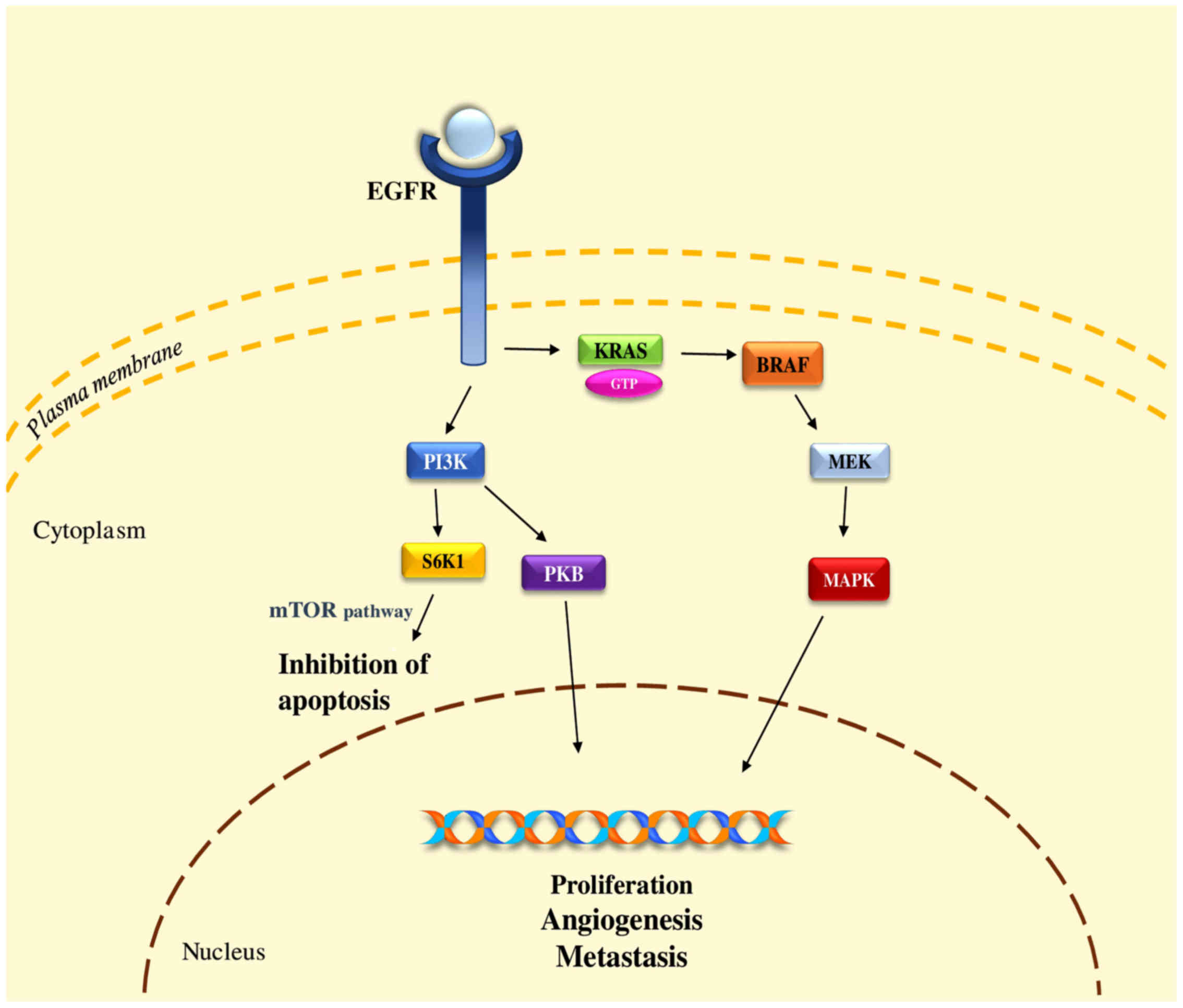
KRAS and NRAS
The family of RAS proteins (H-, K-, and N-RAS) is
located in the intracellular side of the cell membrane involved in
G-protein mediated signal transduction. Activation of the epidermal
growth factor receptor (EGFR) from its ligand (e.g., EGF, TGF-α,
amphiregulin) results in a change from GDP- to GTP-form of the
KRAS, leading to increased concentrations of B-rapidly accelerated
fibrosarcoma (proto-oncogene) (BRAF) to the plasma membrane
(Fig. 5). BRAF activates the
mitogen-activated protein kinases (MAPK) signaling pathway that
results in the expression of proteins involved in several pathways
with a crucial role including cell proliferation, differentiation,
survival, angiogenesis, and cell motility. Mutant KRAS proteins
present as locked in the active form as a result of an impaired
GTPase activity, leading to an increased proliferating rate that is
unregulated and thus resulting in an overall malignant
transformation of the cells. Therefore, it is logical to assume
that mutation of these oncogenes poses a great threat for
carcinogenesis (118). Indeed, it
is well documented that alterations of KRAS gene can act as
the first step towards carcinogenesis in approximately 50% of the
CRC cases (119). However, it is
not that clear whether it could serve as a prognostic marker in the
clinical setting. Recently, a meta-analysis of seven studies failed
to associate KRAS mutation status with prognosis (however, a
common limitation of such studies is the small pool of patients)
(120). On the other hand, two
large multicenter studies, demonstrated that only one mutation of
KRAS, of codon 12, could be linked with a more aggressive
progression of cancer cells. However, according to their data,
KRAS mutations failed to be associated with tumor location
or stage and recurrence of disease (121,122). On the other hand, the possible
prognostic value of NRAS mutations is less examined, even
though they appear to act in a similar way with KRAS as for
the degree of negative prognostic significance (123).
p53
p53 is a transcription factor that participates in
a variety of cellular reactions to several stress situations such
as mutagenic DNA damage, oncogene activation, hypoxia and telomere
shortening. In contrast to other mutations, p53 mutation seems to
occur late in the development of CRC since few or even no cases of
mutations are described in precancerous lesions from sporadic
adenomas and polyps. Interestingly, data from one study showed that
mutations of p53 in CRC exhibits a dependence on the primary
tumor site thus suggesting a prognostic value of this marker. In
more detail, patients with a primary tumor site in the proximal
colon (caecum, ascending colon) and mutant p53 gene
exhibited better survival when treated with a combination of
chemotherapy and surgical removal compared to those treated by
surgery alone (124).
BRAF
BRAF is serine-threonine protein kinase that is
recognized downstream in the KRAS signaling cascade. BRAF
mutations are linked with a poor outcome thus proving their
clinical applicability as a prognostic marker in the adjuvant
setting. Another indication of a poor outcome is found in stage II
and III CRC cases where BRAF mutations were associated with
worse OS (125). A rather
interesting finding is that BRAF V600E mutation is able to
predict a poor prognosis in right-sided MSS CRC (126,127). Further examination of the
BRAF prognostic value was carried with a study investigating
the correlation of BRAF mutation with MSI. The existence of
BRAF wild-type and MSI-H exhibited favorable outcomes.
Furthermore, BRAF-wild/MSS and BRAF-mutated/MSI-H
exhibited intermediate outcomes (128).
MSI
MSI status is a well-studied diagnostic marker for
CRC as mentioned above. However, its clinical relevance does not
stop in the diagnostic setting but continues in the prognostic as
well. MSI-H is associated with better survival rates than both
MSI-L and MSS, not only in HNPCC, but also in sporadic cases
(129,130).
miRNA with prognostic value
During the last few years, a wide variety of miRNAs
has been evaluated in order to identify their prognostic value in
CRC. Indeed, numerous miRNAs, among which are miR-101, the let7
family, miR-133b, miR-126, miR-337, miR-944, miR-646, miR-497 and
miR-142-3p have been identified to behave as tumor suppressors
(tumor-suppressive miRNAs) (131–137) while others, including miR-7,
miR-20a, miR-21, miR-29a, miR-92a, miR-130b, miR-155 and miR-552,
were characterized as key players in creating a favorable
microenvironment for cancer cells. These miRNAs are known as
oncomirs (oncogenic miRNAs) (138–141). miR-20a-5p was found to promote
tumor invasion and metastasis via downregulation of SMAD4 (142). Moreover, elevated miR-21 levels
correlate with CRC cell proliferation, invasion, lymph node
metastases, advanced clinical stage, poor overall and disease-free
survival in the different Duke stages (143,144). Increased expression of miR-29a is
found to strongly correlate with metastases and especially liver
metastases as, if present, it can serve as an early detector of
liver metastases (145,146). High preoperative miR-155 closely
correlates with advanced stage and metastasis while persistent
postoperative expression correlates with recurrence and metastasis.
Thus, miR-155 could be considered a prognostic marker for overall
and disease-free survival (147).
In a recent study, Zhao et al (148) presented that miR-411 functions as
a tumor-suppressive miRNA directly targeting PIK3R3 and indirectly
regulating the AKT/mTOR signaling pathway. In other words, an
increase of miR-411 would result in the downregulation of PIK3R3
directly and AKT/mTOR indirectly. Additionally, they observed that
a decreased expression of miR-411 was correlated with worse
findings such as lymph node metastasis, distant metastasis and
worse TNM stage (148).
Blood-derived prognostic biomarkers
Preoperative CEA levels
CEA may be tested in the preoperative setting in
patients with CRC in order to assist staging and surgical treatment
planning. Previous findings showed that increased preoperative CEA
(>5 ng/ml) correlates with poorer prognosis. In fact, a study
with 2,230 patients proved that pre-operative CEA levels was an
important independent prognostic factor when outcome prediction was
encountered (149). Similarly,
another study with 1,146 patients with CRC found that, following
use of a multivariate analysis, preoperative CEA levels proved a
highly significant prognostic factor even when stage and grade were
introduced in the model (150). In
addition, elevated preoperative CEA in stage III and IV CRC are
considered to be a potent independent risk factor as far as local
relapse, short disease-free survival and OS are concerned (151).
Postoperative CEA levels
After surgical removal of the tumor, CEA levels
should be checked as it is shown that persistent by elevated CEA
levels suggest further evaluation for metastatic disease. In
addition, elevated CEA is very efficient for revealing recurrence
in asymptomatic patients and is the most sensitive detector for
liver metastases (152).
Cancer antigen 19-9 (CA 19-9)
levels
CA 19-9, also termed sialyl Lewis a, is a
documented marker with prognostic value for CRC. It is shown that
cases with increased CA 19-9 present more frequently metastases
thus making it a marker of poor prognosis (152). Similarly, a recent study with
stage IV CRC proved that the preoperative serum CA 19-9 level can
be a promising marker of tumor recurrence and prognosis in cases
submitted to curative resection. In detail, high levels of CA 19-9
were connected with worse 3-year relapse-free survival and 3-year
overall than that in the normal CA 19-9 group (153).
Circulating tumor cells (CTCs)
Detection of CTC has demonstrated prognostic
significance in predicting patient outcome in cases of metastatic
CRC. Recently, two groups investigated the prognostic significance
of CTCs in CRC (154,155). Both of them found that patients
with high CTC count were more likely to experience worse PFS and OS
in contrast to those patients with low CTC count.
Predictive markers
General
The final step in the clinical management of a
patient with cancer, is the determination of the most suitable
therapeutic regimen. It is well known that patients with cancer,
exhibit different responses to a certain therapy and some patients
can benefit the most from the use of a biologic factor. Thus, it is
of paramount importance for us to be able to know beforehand which
patient will receive each therapy or in other words to predict the
most likely response to a given therapy. For this reason,
predictive markers are used.
Tissue-derived predictive markers
DNA alterations
KRAS and NRAS
Mutations of KRAS gene have proven their
clinical use as a predictive biomarker in response to the EGFR
inhibitors clinically used, as various mutations of KRAS
present resistance to therapy with EGFR receptor monoclonal
antibody blockers such as cetuximab (156). In detail, anti-EGFR therapy on
cases of metastatic CRC who display KRAS mutations either of
the codon 12 or 13, present no benefit (157). However, a mutant KRAS does
not always mean that it is a sign of bad response. For example, De
Roock et al (158) have
found that patients with metastatic CRC with P.G13D KRAS
mutation treated with cetuximab exhibited better results than other
KRAS mutations (158).
Under the light of these findings, evaluation of an extended panel
of RAS mutations including mutations in KRAS exon 2,
3 and 4 and NRAS exons 2, 3, and 4 can better discriminate
which patients are not good candidates for treatment with anti-EGFR
therapy (159).
BRAF
BRAF mutations and especially BRAF
V600E is among the most common mutations found in CRC cases as
it is present in approximately 8–10% of all cases (160). BRAF mutation is often used
as a discrimination factor between familial and sporadic CRC as the
existence of BRAF V600E mutation in MSI CRCs can virtually
exclude Lynch syndrome (161).
However, numerous studies have shown that tumors that exhibit
BRAF mutation are resistant to anti-EGFR therapy (162).
Phosphoinositide 3-kinase (PI3K)
PI3K is a downstream mediator of the EGFR signaling
cascade along with AKT and PTEN. An interesting finding regarding
PI3K mutations lies on the use of cetuximab in patients with
CRCs who display mutations of PI3K and especially at the
PIK3CA exon 20 (which is the kinase domain) in contrast to patients
with PIK3CA-wild-type CRCs. Patients with mutant PI3K
exhibit much worse results (163).
However, the mutation of a different exon, PIK3CA exon 9
(which is the helical domain), cannot serve as a predictive marker
for anti-EGFR therapy, a fact that reflects the high complexity of
the effects of specific mutations on different functions of the
mutant kinases (163).
MSI status
Recent studies have investigated the application of
level of MSI status as a potential predictive marker of adjuvant
therapy. While there is enough evidence supporting that MSI-H may
predict response to 5-fluorouracil (5-FU)-based adjuvant therapy in
stage III colon cancer, numerous recent studies demonstrated that
there is no significant difference between patients with MSI-H and
MSS tumors when 5-FU-based adjuvant therapy is used (164–166). This finding is very important for
the group with stage II disease, in which adjuvant chemotherapy
(5-FU alone) is reported to improve survival by approximately 3%,
allowing some investigators to suggest that stage II colon tumors
should be analyzed for MSI status as well in order to assist guide
decisions on the use of adjuvant therapy (165).
CD133
CD133 is a surface protein that has been associated
with tumor angiogenesis and recurrence and especially with VEGF
cascade (167). Previous findings
have shown that the expression of CD133 was associated with worse
survival rates in patients treated with surgery as a monotherapy
and in patients treated with 5-FU-based chemotherapy. By contrast,
CD133 expression in patients with stage III tumors, was able to
determine that the absence of CD133 could benefit more from 5-FU
treatment as expressed by their survival rates but not those with
present CD133 expression. Positive expression of CD133 was also
associated with worse clinical response to chemotherapy in stage IV
patients (168).
New insights in CRC monitoring
General
Going through the list of the markers used for
diagnostic, prognostic and predictive purposes, it is evident that
even though a plethora of markers are available, the clinician
still cannot achieve the best possible monitoring of his patient's
disease, even when a wide panel of markers is used. For this
reason, great effort is made by researchers worldwide in order to
identify new markers that will be able to lead to a better, if not
the best, disease monitoring. A number of these markers seem to be
quite promising. We will discuss three of these markers [telomere
length (TL), telomerase activity and micronuclei (MN)
frequency].
TL and telomerase activity
Telomeres are DNA-protein complexes that are
enrolled to guard the ends of eukaryotic chromosomes and composed
of a repetitive nucleotide sequence (5′-TTAGGG-3′) which is added
on by the telomerase. Telomerase is an enzyme complex that consists
of two subunits, the reverse transcriptase protein human telomerase
reverse transcriptase (hTERT), which is the catalytic subunit, and
the telomerase RNA component (TERC), an RNA template-hTR (human
telomere RNA), which serves as a template for directing the
appropriate telomeric sequences onto the 3′ end of a telomeric
primer (169). When telomeres
become too short, cells may be unable proliferate, a situation that
has been linked to the development of a variety of age-related
diseases, such as cancer, cardiovascular diseases, diabetes and
psychiatric disorders (170–172). A number of studies have examined
the role of telomeres and telomerase in CRC in generating CIN. In
CRC telomere, shortening is an initial event that directly reflects
pathologic cell proliferation because of telomere's shortening in
CRCs and in well-differentiated tumors (173,174). Telomeres and telomerase have been
proposed as potential prognostic and diagnostic biomarkers in CRC.
Telomeres and telomerase activity have universal changes along the
CRC process (175).
Fernández-Marcelo et al (176) examined the ratio of TL in cancer
to non-cancer tissue, telomerase activity and TERT levels and their
role as prognostic markers. In tumors, TL was shorter than that in
non-tumor tissues and more than 80% of CRCs displayed telomerase
activity and thus the use of telomere status as a prognostic factor
has been suggested (176). Gertler
et al (177) analyzed TL
and hTERT expression in matched cancer and adjacent non-cancer
mucosa samples and found that telomeres in CRC tissues were
significantly shorter compared with adjacent normal mucosa samples.
Another study comparing peripheral blood cell TL in CRC patients
and healthy subjects, TL has an anomalous behavior. We can conclude
that telomeres and telomerase emerge as useful diagnostic and
prognostic markers in the clinical management of CRC (5).
MN frequency
MN are extra-nuclear bodies recognized in dividing
cells, that contain chromosome fragments and/or whole chromosomes
that failed to incorporate into the nucleus after cell division. It
is well established that MN formation can be induced by defects in
the DNA repairing system, leading to the accumulation of DNA
damages and chromosomal aberrations (178). As MN formation is a result of CIN
and indicative of a malfunctioning cell, it is reasonable to assume
that MN could, under certain circumstances, be linked with cancer
development (179). Indeed, high
MN frequency is recognized in a number of cancer types.
Specifically, in lung cancer MN frequency has become a useful
marker for the identification of small and non-small cell lung
cancer using peripheral blood samples (180). As far as CRC is concerned, data
from various studies using peripheral blood samples indicate that
MN frequency is a promising biomarker for the early detection of
CRC while it could also be used as a prognostic biomarker (181–184). However, more studies are needed in
order to describe with certainty the true potential of this
biomarker.
Acknowledgements
Not applicable.
Funding
Not applicable.
Availability of data and materials
All data generated or analyzed during this study
are included in this published article.
Authors' contributions
JT conceived and designed the study. TKN and LV
researched the literature, performed analysis of data and drafted
the manuscript. PF and GP drafted the manuscript. JT, TMS, DAS and
AMT critically revised the article for important intellectual
content.
Ethics approval and consent to
participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Demetrios A. Spandidos is the Editor-in-Chief for
the journal, but had no personal involvement in the reviewing
process, or any influence in terms of adjudicating on the final
decision, for this article.
Glossary
Abbreviations
Abbreviations:
|
CRC
|
colorectal cancer
|
|
CIN
|
chromosomal instability
|
|
MSI
|
microsatellite instability
|
|
CIMP
|
CpG island methylation phenotype
|
|
CEA
|
carcinoembryonic antigen
|
|
ctDNA
|
circulating tumor DNA
|
|
CA 19-9
|
cancer antigen 19-9
|
|
CTCs
|
circulating tumor cells
|
|
PI3K
|
phosphoinositide 3-kinase
|
|
CKs
|
cytokeratins
|
|
APC
|
adenomatous polyposis coli
|
|
CDH17
|
cadherin 17
|
|
PFS
|
progression-free survival
|
|
OS
|
overall survival
|
|
hTERT
|
human telomerase reverse
transcriptase
|
|
MN
|
micronuclei
|
|
TERC
|
telomerase RNA component
|
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Haggar FA and Boushey RP: Colorectal
cancer epidemiology: Incidence, mortality, survival, and risk
factors. Clin Colon Rectal Surg. 22:191–197. 2009. View Article : Google Scholar
|
|
3
|
Dušek L, Mužík J, Malúšková D and
Šnajdrová L: Epidemiology of colorectal cancer: International
comparisonInstitute of Biostatistics and Analyses. Masaryk
University; Brno, Czech Republic:
|
|
4
|
Bardhan K and Liu K: Epigenetics and
colorectal cancer pathogenesis. Cancers (Basel). 5:676–713. 2013.
View Article : Google Scholar
|
|
5
|
Tsiaoussis J, Vassilopoulou L,
Nikolouzakis T, Rakitskii VN, Vakonaki E, Fragkiadaki P,
Stivaktakis P and Tsatsakis AM: Biomolecular profile of colorectal
cancer - the role of telomerase as a potent biomarker. Farmacia.
65:643–659. 2017.
|
|
6
|
Souglakos J, Philips J, Wang R, Marwah S,
Silver M, Tzardi M, Silver J, Ogino S, Hooshmand S, Kwak E, et al:
Prognostic and predictive value of common mutations for treatment
response and survival in patients with metastatic colorectal
cancer. Br J Cancer. 101:465–472. 2009. View Article : Google Scholar
|
|
7
|
Migliore L, Migheli F, Spisni R and
Coppedè F: Genetics, cytogenetics, and epigenetics of colorectal
cancer. J Biomed Biotechnol. 2011:7923622011. View Article : Google Scholar
|
|
8
|
Gonzalez-Pons M and Cruz-Correa M:
Colorectal cancer biomarkers: Where are we now? BioMed Res Int.
2015:1490142015. View Article : Google Scholar
|
|
9
|
Cunningham D, Atkin W, Lenz HJ, Lynch HT,
Minsky B, Nordlinger B and Starling N: Colorectal cancer. Lancet.
375:1030–1047. 2010. View Article : Google Scholar
|
|
10
|
Kuipers EJ, Rösch T and Bretthauer M:
Colorectal cancer screening - optimizing current strategies and new
directions. Nat Rev Clin Oncol. 10:130–142. 2013. View Article : Google Scholar
|
|
11
|
Labianca R and Merelli B: Screening and
diagnosis for colorectal cancer: Present and future. Tumori.
96:889–901. 2010. View Article : Google Scholar
|
|
12
|
Mahmud A, Poon R and Jonker D: PET imaging
in anal canal cancer: A systematic review and meta-analysis. Br J
Radiol. 90:201703702017. View Article : Google Scholar
|
|
13
|
Paspulati RM and Gupta A: PET/MR imaging
in cancers of the gastrointestinal tract. PET Clin. 11:403–423.
2016. View Article : Google Scholar
|
|
14
|
Bond JH: Fecal occult blood test screening
for colorectal cancer. Gastrointest Endosc Clin N Am. 12:11–21.
2002. View Article : Google Scholar
|
|
15
|
Siegel R, Desantis C and Jemal A:
Colorectal cancer statistics, 2014. CA Cancer J Clin. 64:104–117.
2014. View Article : Google Scholar
|
|
16
|
Ahlquist DA: Molecular detection of
colorectal neoplasia. Gastroenterology. 138:2127–2139. 2010.
View Article : Google Scholar
|
|
17
|
Shah R, Jones E, Vidart V, Kuppen PJ,
Conti JA and Francis NK: Biomarkers for early detection of
colorectal cancer and polyps: Systematic review. Cancer Epidemiol
Biomarkers Prev. 23:1712–1728. 2014. View Article : Google Scholar
|
|
18
|
Alix-Panabières C and Pantel K: Clinical
applications of circulating tumor cells and circulating tumor DNA
as liquid biopsy. Cancer Discov. 6:479–491. 2016. View Article : Google Scholar
|
|
19
|
Shastri YM, Loitsch S, Hoepffner N, Povse
N, Hanisch E, Rösch W, Mössner J and Stein JM: Comparison of an
established simple office-based immunological FOBT with fecal tumor
pyruvate kinase type M2 (M2-PK) for colorectal cancer screening:
Prospective multicenter study. Am J Gastroenterol. 103:1496–1504.
2008. View Article : Google Scholar
|
|
20
|
Takai T, Kanaoka S, Yoshida K, Hamaya Y,
Ikuma M, Miura N, Sugimura H, Kajimura M and Hishida A: Fecal
cyclooxygenase 2 plus matrix metalloproteinase 7 mRNA assays as a
marker for colorectal cancer screening. Cancer Epidemiol Biomarkers
Prev. 18:1888–1893. 2009. View Article : Google Scholar
|
|
21
|
Huang Z, Huang D, Ni S, Peng Z, Sheng W
and Du X: Plasma microRNAs are promising novel biomarkers for early
detection of colorectal cancer. Int J Cancer. 127:118–126. 2010.
View Article : Google Scholar
|
|
22
|
Wu CW, Ng SS, Dong YJ, Ng SC, Leung WW,
Lee CW, Wong YN, Chan FK, Yu J and Sung JJ: Detection of miR-92a
and miR-21 in stool samples as potential screening biomarkers for
colorectal cancer and polyps. Gut. 61:739–745. 2012. View Article : Google Scholar
|
|
23
|
Pan C, Yan X, Li H, Huang L, Yin M, Yang
Y, Gao R, Hong L, Ma Y, Shi C, et al: Systematic literature review
and clinical validation of circulating microRNAs as diagnostic
biomarkers for colorectal cancer. Oncotarget. 8:68317–68328. 2017.
View Article : Google Scholar
|
|
24
|
Kanaan Z, Roberts H, Eichenberger MR,
Billeter A, Ocheretner G, Pan J, Rai SN, Jorden J, Williford A and
Galandiuk S: A plasma microRNA panel for detection of colorectal
adenomas: A step toward more precise screening for colorectal
cancer. Ann Surg. 258:400–408. 2013. View Article : Google Scholar
|
|
25
|
Imperiale TF, Ransohoff DF, Itzkowitz SH,
Levin TR, Lavin P, Lidgard GP, Ahlquist DA and Berger BM:
Multitarget stool DNA testing for colorectal-cancer screening. N
Engl J Med. 370:1287–1297. 2014. View Article : Google Scholar
|
|
26
|
Imperiale TF, Ransohoff DF, Itzkowitz SH,
Turnbull BA and Ross ME: Colorectal Cancer Study Group: Fecal DNA
versus fecal occult blood for colorectal-cancer screening in an
average-risk population. N Engl J Med. 351:2704–2714. 2004.
View Article : Google Scholar
|
|
27
|
Bayrak R, Yenidünya S and Haltas H:
Cytokeratin 7 and cytokeratin 20 expression in colorectal
adenocarcinomas. Pathol Res Pract. 207:156–160. 2011. View Article : Google Scholar
|
|
28
|
Righi A, Betts CM, Marchetti C, Marucci G,
Montebugnoli L, Prati C, Eusebi LH, Muzzi L, Ragazzini T and
Foschini MP: Merkel cells in the oral mucosa. Int J Surg Pathol.
14:206–211. 2006. View Article : Google Scholar
|
|
29
|
Stenling R, Lindberg J, Rutegård J and
Palmqvist R: Altered expression of CK7 and CK20 in preneoplastic
and neoplastic lesions in ulcerative colitis. APMIS. 115:1219–1226.
2007. View Article : Google Scholar
|
|
30
|
Radović S, Selak I, Babić M,
Vukobrat-Bijedić Z and Knezević Z: Anti-cytokeratin 7: A positive
marker for epithelial dysplasia in flat bowel mucosa. Bosn J Basic
Med Sci. 4:24–30. 2004.
|
|
31
|
Gurzu S and Jung I: Aberrant pattern of
the cytokeratin 7/cytokeratin 20 immunophenotype in colorectal
adenocarcinomas with BRAF mutations. Pathol Res Pract. 208:163–166.
2012. View Article : Google Scholar
|
|
32
|
Chu P, Wu E and Weiss LM: Cytokeratin 7
and cytokeratin 20 expression in epithelial neoplasms: A survey of
435 cases. Mod Pathol. 13:962–972. 2000. View Article : Google Scholar
|
|
33
|
Miettinen M, Nobel MP, Tuma BT and
Kovatich AJ: Keratin 17: Immunohistochemical mapping of its
distribution in human epithelial tumors and its potential
applications. Appl Immunohistochem. 5:152–159. 1997. View Article : Google Scholar
|
|
34
|
Hernandez BY, Frierson HF Jr, Moskaluk CA,
Li YJ, Clegg L, Cote TR, McCusker ME, Hankey BF, Edwards BK and
Goodman MT: CK20 and CK7 protein expression in colorectal cancer:
Demonstration of the utility of a population-based tissue
microarray. Hum Pathol. 36:275–281. 2005. View Article : Google Scholar
|
|
35
|
Silberg DG, Swain GP, Suh ER and Traber
PG: Cdx1 and cdx2 expression during intestinal development.
Gastroenterology. 119:961–971. 2000. View Article : Google Scholar
|
|
36
|
Moskaluk CA, Zhang H, Powell SM, Cerilli
LA, Hampton GM and Frierson HF Jr: Cdx2 protein expression in
normal and malignant human tissues: An immunohistochemical survey
using tissue microarrays. Mod Pathol. 16:913–919. 2003. View Article : Google Scholar
|
|
37
|
Werling RW, Yaziji H, Bacchi CE and Gown
AM: CDX2, a highly sensitive and specific marker of adenocarcinomas
of intestinal origin: An immunohistochemical survey of 476 primary
and metastatic carcinomas. Am J Surg Pathol. 27:303–310. 2003.
View Article : Google Scholar
|
|
38
|
Zheng J, He S, Qi J, Wang X, Yu J, Wu Y,
Gao Q, Wang K and Sun X: Targeted CDX2 expression inhibits
aggressive phenotypes of colon cancer cells in vitro and
in vivo. Int J Oncol. 51:478–488. 2017. View Article : Google Scholar
|
|
39
|
Bretscher A and Weber K: Villin: The major
microfilament-associated protein of the intestinal microvillus.
Proc Natl Acad Sci USA. 76:2321–2325. 1979. View Article : Google Scholar
|
|
40
|
Patnaik S, George SP, Pham E, Roy S, Singh
K, Mariadason JM and Khurana S: By moonlighting in the nucleus,
villin regulates epithelial plasticity. Mol Biol Cell. 27:535–548.
2016. View Article : Google Scholar
|
|
41
|
Kuroda N and Yorita K: Colon cancer with
micropapillary carcinoma component: A clinopathologic study of 9
cases. Pol J Pathol. 68:102–108. 2017. View Article : Google Scholar
|
|
42
|
Bacchi CE and Gown AM: Distribution and
pattern of expression of villin, a gastrointestinal-associated
cytoskeletal protein, in human carcinomas: A study employing
paraffin-embedded tissue. Lab Invest. 64:418–424. 1991.
|
|
43
|
Willert K and Nusse R: Beta-catenin: A key
mediator of Wnt signaling. Curr Opin Genet Dev. 8:95–102. 1998.
View Article : Google Scholar
|
|
44
|
Wang JL, Qi Z, Li YH, Zhao HM, Chen YG and
Fu W: TGFβ induced factor homeobox 1 promotes colorectal cancer
development through activating Wnt/β-catenin signaling. Oncotarget.
8:70214–70225. 2017.
|
|
45
|
Clevers H: Wnt/β-catenin signaling in
development and disease. Cell. 127:469–480. 2006. View Article : Google Scholar
|
|
46
|
Sheahan K, O'Brien MJ, Burke B, Dervan PA,
O'Keane JC, Gottlieb LS and Zamcheck N: Differential reactivities
of carcinoembryonic antigen (CEA) and CEA-related monoclonal and
polyclonal antibodies in common epithelial malignancies. Am J Clin
Pathol. 94:157–164. 1990. View Article : Google Scholar
|
|
47
|
Zhou M, Chinnaiyan AM, Kleer CG, Lucas PC
and Rubin MA: Alpha-methylacyl-CoA racemase: A novel tumor marker
over-expressed in several human cancers and their precursor
lesions. Am J Surg Pathol. 26:926–931. 2002. View Article : Google Scholar
|
|
48
|
Tan E, Gouvas N, Nicholls RJ, Ziprin P,
Xynos E and Tekkis PP: Diagnostic precision of carcinoembryonic
antigen in the detection of recurrence of colorectal cancer. Surg
Oncol. 18:15–24. 2009. View Article : Google Scholar
|
|
49
|
Andrianifahanana M, Moniaux N and Batra
SK: Regulation of mucin expression: Mechanistic aspects and
implications for cancer and inflammatory diseases. Biochim Biophys
Acta. 1765:189–222. 2006.
|
|
50
|
Hanski C, Hofmeier M, Schmitt-Gräff A,
Riede E, Hanski ML, Borchard F, Sieber E, Niedobitek F, Foss HD,
Stein H and Riecken EO: Overexpression or ectopic expression of
MUC2 is the common property of mucinous carcinomas of the colon,
pancreas, breast, and ovary. J Pathol. 182:385–391. 1997.
View Article : Google Scholar
|
|
51
|
Zlatian OM, Comănescu MV, Roşu AF, Roşu L,
Cruce M, Găman AE, Călina CD and Sfredel V: Histochemical and
immunohistochemical evidence of tumor heterogeneity in colorectal
cancer. Rom J Morphol Embryol. 56:175–181. 2015.
|
|
52
|
Park SY, Lee HS, Choe G, Chung JH and Kim
WH: Clinicopathological characteristics, microsatellite
instability, and expression of mucin core proteins and p53 in
colorectal mucinous adenocarcinomas in relation to location.
Virchows Arch. 449:40–47. 2006. View Article : Google Scholar
|
|
53
|
King RJ, Yu F and Singh PK: Genomic
alterations in mucins across cancers. Oncotarget. 8:67152–67168.
2017. View Article : Google Scholar
|
|
54
|
Wang H, Jin S, Lu H, Mi S, Shao W, Zuo X,
Yin H, Zeng S, Shimamoto F and Qi G: Expression of survivin, MUC2
and MUC5 in colorectal cancer and their association with
clinicopathological characteristics. Oncol Lett. 14:1011–1016.
2017. View Article : Google Scholar
|
|
55
|
Dantzig AH, Hoskins JA, Tabas LB, Bright
S, Shepard RL, Jenkins IL, Duckworth DC, Sportsman JR, Mackensen D,
Rosteck PR Jr, et al: Association of intestinal peptide transport
with a protein related to the cadherin superfamily. Science.
264:430–433. 1994. View Article : Google Scholar
|
|
56
|
Su MC, Yuan RH, Lin CY and Jeng YM:
Cadherin-17 is a useful diagnostic marker for adenocarcinomas of
the digestive system. Mod Pathol. 21:1379–1386. 2008. View Article : Google Scholar
|
|
57
|
Panarelli NC, Yantiss RK, Yeh MM, Liu Y
and Chen YT: Tissue-specific cadherin CDH17 is a useful marker of
gastrointestinal adenocarcinomas with higher sensitivity than CDX2.
Am J Clin Pathol. 138:211–222. 2012. View Article : Google Scholar
|
|
58
|
Stănculescu D, Mărgăritescu C, Stepan A
and Mitruţ AO: E-cadherin in gastric carcinomas related to
histological prognostic parameters. Rom J Morphol Embryol. 52
Suppl:1107–1112. 2011.
|
|
59
|
Bian T, Zhao J, Feng J, Zhang Q, Qian L,
Liu J, Jiang D, Liu Y and Zhang J: Combination of cadherin-17 and
SATB homeobox 2 serves as potential optimal makers for the
differential diagnosis of pulmonary enteric adenocarcinoma and
metastatic colorectal adenocarcinoma. Oncotarget. 8:63442–63452.
2017. View Article : Google Scholar
|
|
60
|
Tian X, Liu M, Zhu Q, Tan J, Liu W, Wang
Y, Chen W, Zou Y, Cai Y, Han Z and Huang X: Down-regulation of
liver-intestine cadherin enhances noscapine-induced apoptosis in
human colon cancer cells. Expert Rev Anticancer Ther. 17:857–863.
2017. View Article : Google Scholar
|
|
61
|
Magnusson K, de Wit M, Brennan DJ, Johnson
LB, McGee SF, Lundberg E, Naicker K, Klinger R, Kampf C, Asplund A,
et al: SATB2 in combination with cytokeratin 20 identifies over 95%
of all colorectal carcinomas. Am J Surg Pathol. 35:937–948. 2011.
View Article : Google Scholar
|
|
62
|
Valori R, Rey JF, Atkin WS, Bretthauer M,
Senore C, Hoff G, Kuipers EJ, Altenhofen L, Lambert R and Minoli G:
International Agency for Research on Cancer: European guidelines
for quality assurance in colorectal cancer screening and diagnosis
First Edition - quality assurance in endoscopy in colorectal cancer
screening and diagnosis. Endoscopy. 44 Suppl 3:SE88–SE105.
2012.
|
|
63
|
Lieberman DA and Weiss DG: Veterans
Affairs Cooperative Study Group 380: One-time screening for
colorectal cancer with combined fecal occult-blood testing and
examination of the distal colon. N Engl J Med. 345:555–560. 2001.
View Article : Google Scholar
|
|
64
|
Young GP, Symonds EL, Allison JE, Cole SR,
Fraser CG, Halloran SP, Kuipers EJ and Seaman HE: Advances in fecal
occult blood tests: The FIT revolution. Dig Dis Sci. 60:609–622.
2015. View Article : Google Scholar
|
|
65
|
Whitlock EP, Lin J, Liles E, Beil T, Fu R,
O'Connor E, Thompson RN and Cardenas T: Screening for Colorectal
Cancer: An Updated Systematic ReviewAgency for Healthcare Research
and Quality. Rockville, MD: 2008
|
|
66
|
Morikawa T, Kato J, Yamaji Y, Wada R,
Mitsushima T and Shiratori Y: A comparison of the immunochemical
fecal occult blood test and total colonoscopy in the asymptomatic
population. Gastroenterology. 129:422–428. 2005. View Article : Google Scholar
|
|
67
|
Gupta AK, Melton LJ III, Petersen GM,
Timmons LJ, Vege SS, Harmsen WS, Diehl NN, Zinsmeister AR and
Ahlquist DA: Changing trends in the incidence, stage, survival, and
screen-detection of colorectal cancer: A population-based study.
Clin Gastroenterol Hepatol. 3:150–158. 2005. View Article : Google Scholar
|
|
68
|
Puccini A, Berger MD, Naseem M, Tokunaga
R, Battaglin F, Cao S, Hanna DL, McSkane M, Soni S, Zhang W and
Lenz HJ: Colorectal cancer: Epigenetic alterations and their
clinical implications. Biochim Biophys Acta. 1868:439–448.
2017.
|
|
69
|
Losso GM, Moraes RS, Gentili AC and
Messias-Reason IT: Microsatellite instability - MSI markers (BAT26,
BAT25, D2S123, D5S346, D17S250) in rectal cancer. Arq Bras Cir Dig.
25:240–244. 2012. View Article : Google Scholar
|
|
70
|
de la Chapelle A and Hampel H: Clinical
relevance of microsatellite instability in colorectal cancer. J
Clin Oncol. 28:3380–3387. 2010. View Article : Google Scholar
|
|
71
|
Merok MA, Ahlquist T, Røyrvik EC,
Tufteland KF, Hektoen M, Sjo OH, Mala T, Svindland A, Lothe RA and
Nesbakken A: Microsatellite instability has a positive prognostic
impact on stage II colorectal cancer after complete resection:
Results from a large, consecutive Norwegian series. Ann Oncol.
24:1274–1282. 2013. View Article : Google Scholar
|
|
72
|
Umar A, Boland CR, Terdiman JP, Syngal S,
de la Chapelle A, Rüschoff J, Fishel R, Lindor NM, Burgart LJ,
Hamelin R, et al: Revised Bethesda Guidelines for hereditary
nonpolyposis colorectal cancer (Lynch syndrome) and microsatellite
instability. J Natl Cancer Inst. 96:261–268. 2004. View Article : Google Scholar
|
|
73
|
Laghi L, Bianchi P and Malesci A:
Differences and evolution of the methods for the assessment of
microsatellite instability. Oncogene. 27:6313–6321. 2008.
View Article : Google Scholar
|
|
74
|
Roth AD, Tejpar S, Yan P, Fiocca R,
Dietrich D, Delorenzi M, Labianca R, Cunningham D, Van Cutsem E and
Bosman F: Stage-specific prognostic value of molecular markers in
colon cancer: Results of the translational study on the PETACC
3-EORTC 40993-SAKK 60–00 trial. J Clin Oncol. 27 15
Suppl:40022009.
|
|
75
|
Koopman M, Kortman GA, Mekenkamp L,
Ligtenberg MJ, Hoogerbrugge N, Antonini NF, Punt CJ and van Krieken
JH: Deficient mismatch repair system in patients with sporadic
advanced colorectal cancer. Br J Cancer. 100:266–273. 2009.
View Article : Google Scholar
|
|
76
|
Ulamec M and Krušlin B: Colorectal cancer,
novel biomarkers and immunohistochemistry - an overview. Rad Med
Sci. 520:41–49. 2014.
|
|
77
|
Hashimoto Y, Zumwalt TJ and Goel A: DNA
methylation patterns as noninvasive biomarkers and targets of
epigenetic therapies in colorectal cancer. Epigenomics. 8:685–703.
2016. View Article : Google Scholar
|
|
78
|
Jair KW, Bachman KE, Suzuki H, Ting AH,
Rhee I, Yen RW, Baylin SB and Schuebel KE: De novo CpG island
methylation in human cancer cells. Cancer Res. 66:682–692. 2006.
View Article : Google Scholar
|
|
79
|
Beggs AD, Jones A, El-Bahrawy M, Abulafi
M, Hodgson SV and Tomlinson IP: Whole-genome methylation analysis
of benign and malignant colorectal tumours. J Pathol. 229:697–704.
2013. View Article : Google Scholar
|
|
80
|
Toiyama Y, Okugawa Y and Goel A: DNA
methylation and microRNA biomarkers for noninvasive detection of
gastric and colorectal cancer. Biochem Biophys Res Commun.
455:43–57. 2014. View Article : Google Scholar
|
|
81
|
Lind GE, Danielsen SA, Ahlquist T, Merok
MA, Andresen K, Skotheim RI, Hektoen M, Rognum TO, Meling GI, Hoff
G, et al: Identification of an epigenetic biomarker panel with high
sensitivity and specificity for colorectal cancer and adenomas. Mol
Cancer. 10:852011. View Article : Google Scholar
|
|
82
|
Cheung AF, Carter AM, Kostova KK, Woodruff
JF, Crowley D, Bronson RT, Haigis KM and Jacks T: Complete deletion
of Apc results in severe polyposis in mice. Oncogene. 29:1857–1864.
2010. View Article : Google Scholar
|
|
83
|
Dow LE, O'Rourke KP, Simon J,
Tschaharganeh DF, van Es JH, Clevers H and Lowe SW: Apc restoration
promotes cellular differentiation and reestablishes crypt
homeostasis in colorectal cancer. Cell. 161:1539–1552. 2015.
View Article : Google Scholar
|
|
84
|
Liang J, Lin C, Hu F, Wang F, Zhu L, Yao
X, Wang Y and Zhao Y: APC polymorphisms and the risk of colorectal
neoplasia: A HuGE review and meta-analysis. Am J Epidemiol.
177:1169–1179. 2013. View Article : Google Scholar
|
|
85
|
Docea AO, Mitruţ P, Grigore D, Pirici D,
Călina DC and Gofiţă E: Immunohistochemical expression of TGF beta
(TGF-β), TGF beta receptor 1 (TGFBR1), and Ki67 in intestinal
variant of gastric adenocarcinomas. Rom J Morphol Embryol. 53
Suppl:683–692. 2012.
|
|
86
|
Jahr S, Hentze H, Englisch S, Hardt D,
Fackelmayer FO, Hesch RD and Knippers R: DNA fragments in the blood
plasma of cancer patients: Quantitations and evidence for their
origin from apoptotic and necrotic cells. Cancer Res. 61:1659–1665.
2001.
|
|
87
|
Frattini M, Gallino G, Signoroni S,
Balestra D, Lusa L, Battaglia L, Sozzi G, Bertario L, Leo E,
Pilotti S and Pierotti MA: Quantitative and qualitative
characterization of plasma DNA identifies primary and recurrent
colorectal cancer. Cancer Lett. 263:170–181. 2008. View Article : Google Scholar
|
|
88
|
Diehl F, Li M, Dressman D, He Y, Shen D,
Szabo S, Diaz LA Jr, Goodman SN, David KA, Juhl H, et al: Detection
and quantification of mutations in the plasma of patients with
colorectal tumors. Proc Natl Acad Sci USA. 102:16368–16373. 2005.
View Article : Google Scholar
|
|
89
|
Tänzer M, Balluff B, Distler J, Hale K,
Leodolter A, Röcken C, Molnar B, Schmid R, Lofton-Day C, Schuster T
and Ebert MP: Performance of epigenetic markers SEPT9 and ALX4 in
plasma for detection of colorectal precancerous lesions. PLoS One.
5:e90612010. View Article : Google Scholar
|
|
90
|
Danese E and Montagnana M: Epigenetics of
colorectal cancer: Emerging circulating diagnostic and prognostic
biomarkers. Ann Transl Med. 5:2792017. View Article : Google Scholar
|
|
91
|
Ahlquist DA, Harrington JJ, Burgart LJ and
Roche PC: Morphometric analysis of the ‘mucocellular layer’
overlying colorectal cancer and normal mucosa: Relevance to
exfoliation and stool screening. Hum Pathol. 31:51–57. 2000.
View Article : Google Scholar
|
|
92
|
Chen WD, Han ZJ, Skoletsky J, Olson J, Sah
J, Myeroff L, Platzer P, Lu S, Dawson D, Willis J, et al: Detection
in fecal DNA of colon cancer-specific methylation of the
nonexpressed vimentin gene. J Natl Cancer Inst. 97:1124–1132. 2005.
View Article : Google Scholar
|
|
93
|
Itzkowitz S, Brand R, Jandorf L, Durkee K,
Millholland J, Rabeneck L, Schroy PC III, Sontag S, Johnson D,
Markowitz S, et al: A simplified, noninvasive stool DNA test for
colorectal cancer detection. Am J Gastroenterol. 103:2862–2870.
2008. View Article : Google Scholar
|
|
94
|
Lee RC, Feinbaum RL and Ambros V: The
C. elegans heterochronic gene lin-4 encodes small RNAs with
antisense complementarity to lin-14. Cell. 75:843–854. 1993.
View Article : Google Scholar
|
|
95
|
Wightman B, Ha I and Ruvkun G:
Posttranscriptional regulation of the heterochronic gene lin-14 by
lin-4 mediates temporal pattern formation in C. elegans.
Cell. 75:855–862. 1993. View Article : Google Scholar
|
|
96
|
He L and Hannon GJ: MicroRNAs: Small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531. 2004.
View Article : Google Scholar
|
|
97
|
Mendell JT: MicroRNAs: Critical regulators
of development, cellular physiology and malignancy. Cell Cycle.
4:1179–1184. 2005. View Article : Google Scholar
|
|
98
|
Vasudevan S, Tong Y and Steitz JA:
Switching from repression to activation: MicroRNAs can up-regulate
translation. Science. 318:1931–1934. 2007. View Article : Google Scholar
|
|
99
|
Creemers EE, Tijsen AJ and Pinto YM:
Circulating microRNAs: Novel biomarkers and extracellular
communicators in cardiovascular disease? Circ Res. 110:483–495.
2012. View Article : Google Scholar
|
|
100
|
Arroyo JD, Chevillet JR, Kroh EM, Ruf IK,
Pritchard CC, Gibson DF, Mitchell PS, Bennett CF,
Pogosova-Agadjanyan EL, Stirewalt DL, et al: Argonaute2 complexes
carry a population of circulating microRNAs independent of vesicles
in human plasma. Proc Natl Acad Sci USA. 108:5003–5008. 2011.
View Article : Google Scholar
|
|
101
|
Vickers KC, Palmisano BT, Shoucri BM,
Shamburek RD and Remaley AT: MicroRNAs are transported in plasma
and delivered to recipient cells by high-density lipoproteins. Nat
Cell Biol. 13:423–433. 2011. View Article : Google Scholar
|
|
102
|
Toiyama Y, Takahashi M, Hur K, Nagasaka T,
Tanaka K, Inoue Y, Kusunoki M, Boland CR and Goel A: Serum miR-21
as a diagnostic and prognostic biomarker in colorectal cancer. J
Natl Cancer Inst. 105:849–859. 2013. View Article : Google Scholar
|
|
103
|
Ng EK, Chong WW, Jin H, Lam EK, Shin VY,
Yu J, Poon TC, Ng SS and Sung JJ: Differential expression of
microRNAs in plasma of patients with colorectal cancer: A potential
marker for colorectal cancer screening. Gut. 58:1375–1381. 2009.
View Article : Google Scholar
|
|
104
|
Carter JV, Galbraith NJ, Yang D, Burton
JF, Walker SP and Galandiuk S: Blood-based microRNAs as biomarkers
for the diagnosis of colorectal cancer: A systematic review and
meta-analysis. Br J Cancer. 116:762–774. 2017. View Article : Google Scholar
|
|
105
|
Okugawa, Grady WM and Goel A: Epigenetic
alterations in colorectal cancer: Emerging biomarkers.
Gastroenterology. 149:1204–1225.e12. 2015. View Article : Google Scholar
|
|
106
|
Chen M, Lin M and Wang X: Over expression
of miR-19a inhibits colorectal cancer angiogenesis by suppressing
KRAS expression. Oncol Rep. 39:619–626. 2018.
|
|
107
|
Link A, Balaguer F, Shen Y, Nagasaka T,
Lozano JJ, Boland CR and Goel A: Fecal microRNAs as novel
biomarkers for colon cancer screening. Cancer Epidemiol Biomarkers
Prev. 19:1766–1774. 2010. View Article : Google Scholar
|
|
108
|
Zhu Y, Xu A, Li J, Fu J, Wang G, Yang Y,
Cui L and Sun J: Fecal miR-29a and miR-224 as the noninvasive
biomarkers for colorectal cancer. Cancer Biomark. 16:259–264. 2016.
View Article : Google Scholar
|
|
109
|
Masuda T, Hayashi N, Kuroda Y, Ito S,
Eguchi H and Mimori K: MicroRNAs as biomarkers in colorectal
cancer. Cancers (Basel). 9:1242017. View Article : Google Scholar
|
|
110
|
Edge SB and Compton CC: The American Joint
Committee on Cancer: The 7th edition of the AJCC Cancer Staging
Manual and the future of TNM. Ann Surg Oncol. 17:1471–1474. 2010.
View Article : Google Scholar
|
|
111
|
Perez RO, Bresciani BH, Bresciani C,
Proscurshim I, Kiss D, Gama-Rodrigues J, Pereira DD, Rawet V,
Cecconnello I and Habr-Gama A: Mucinous colorectal adenocarcinoma:
Influence of mucin expression (Muc1, 2 and 5) on
clinico-pathological features and prognosis. Int J Colorectal Dis.
23:757–765. 2008. View Article : Google Scholar
|
|
112
|
Wang S, Zhou J, Wang XY, Hao JM, Chen JZ,
Zhang XM, Jin H, Liu L, Zhang YF, Liu J, et al: Down-regulated
expression of SATB2 is associated with metastasis and poor
prognosis in colorectal cancer. J Pathol. 219:114–122. 2009.
View Article : Google Scholar
|
|
113
|
Eberhard J, Gaber A, Wangefjord S, Nodin
B, Uhlén M, Lindquist Ericson K and Jirström K: A cohort study of
the prognostic and treatment predictive value of SATB2 expression
in colorectal cancer. Br J Cancer. 106:931–938. 2012. View Article : Google Scholar
|
|
114
|
Bae JM, Lee TH, Cho NY, Kim TY and Kang
GH: Loss of CDX2 expression is associated with poor prognosis in
colorectal cancer patients. World J Gastroenterol. 21:1457–1467.
2015. View Article : Google Scholar
|
|
115
|
Falchook GS and Kurzrock R: VEGF and
dual-EGFR inhibition in colorectal cancer. Cell Cycle.
14:1129–1130. 2015. View Article : Google Scholar
|
|
116
|
Li D, Yan D, Tang H, Zhou C, Fan J, Li S,
Wang X, Xia J, Huang F, Qiu G and Peng Z: IMP3 is a novel
prognostic marker that correlates with colon cancer progression and
pathogenesis. Ann Surg Oncol. 16:3499–3506. 2009. View Article : Google Scholar
|
|
117
|
Takahashi H, Ishikawa T, Ishiguro M,
Okazaki S, Mogushi K, Kobayashi H, Iida S, Mizushima H, Tanaka H,
Uetake H and Sugihara K: Prognostic significance of Traf2- and
Nck-interacting kinase (TNIK) in colorectal cancer. BMC Cancer.
15:7942015. View Article : Google Scholar
|
|
118
|
Spandidos DA, Glarakis IS, Kotsinas A,
Ergazaki M and Kiaris H: Ras oncogene activation in benign and
malignant colorectal tumours. Tumori. 81 Suppl:7–11. 1995.
|
|
119
|
Kiaris H and Spandidos D: Mutations of ras
genes in human tumors (Review). Int J Oncol. 7:413–421. 1995.
|
|
120
|
Rui YY, Zhang D, Zhou ZG, Wang C, Yang L,
Yu YY and Chen HN: Can K-ras gene mutation be utilized as
prognostic biomarker for colorectal cancer patients receiving
chemotherapy? A meta-analysis and systematic review. PLoS One.
8:e779012013. View Article : Google Scholar
|
|
121
|
Andreyev HJN, Norman AR, Cunningham D,
Oates JR and Clarke PA: Kirsten ras mutations in patients with
colorectal cancer: The multicenter ‘RASCAL’ study. J Natl Cancer
Inst. 90:675–684. 1998. View Article : Google Scholar
|
|
122
|
Andreyev HJN, Norman AR, Cunningham D,
Oates J, Dix BR, Iacopetta BJ, Young J, Walsh T, Ward R, Hawkins N,
et al: Kirsten ras mutations in patients with colorectal cancer:
The ‘RASCAL II’ study. Br J Cancer. 85:692–696. 2001. View Article : Google Scholar
|
|
123
|
Wang Y, Velho S, Vakiani E, Peng S, Bass
AJ, Chu GC, Gierut J, Bugni JM, Der CJ, Philips M, et al: Mutant
N-RAS protects colorectal cancer cells from stress-induced
apoptosis and contributes to cancer development and progression.
Cancer Discov. 3:294–307. 2013. View Article : Google Scholar
|
|
124
|
Russo A, Bazan V, Iacopetta B, Kerr D,
Soussi T and Gebbia N: TP53-CRC Collaborative Study Group: The TP53
colorectal cancer international collaborative study on the
prognostic and predictive significance of p53 mutation: Influence
of tumor site, type of mutation, and adjuvant treatment. J Clin
Oncol. 23:7518–7528. 2005. View Article : Google Scholar
|
|
125
|
French AJ, Sargent DJ, Burgart LJ, Foster
NR, Kabat BF, Goldberg R, Shepherd L, Windschitl HE and Thibodeau
SN: Prognostic significance of defective mismatch repair and BRAF
V600E in patients with colon cancer. Clin Cancer Res. 14:3408–3415.
2008. View Article : Google Scholar
|
|
126
|
Toon CW, Chou A, DeSilva K, Chan J,
Patterson J, Clarkson A, Sioson L, Jankova L and Gill AJ: BRAFV600E
immunohistochemistry in conjunction with mismatch repair status
predicts survival in patients with colorectal cancer. Mod Pathol.
27:644–650. 2014. View Article : Google Scholar
|
|
127
|
Zlobec I, Bihl MP, Schwarb H, Terracciano
L and Lugli A: Clinicopathological and protein characterization of
BRAF- and K-RAS-mutated colorectal cancer and implications for
prognosis. Int J Cancer. 127:367–380. 2010.
|
|
128
|
Ogino S, Shima K, Meyerhardt JA, McCleary
NJ, Ng K, Hollis D, Saltz LB, Mayer RJ, Schaefer P, Whittom R, et
al: Predictive and prognostic roles of BRAF mutation in stage III
colon cancer: Results from intergroup trial CALGB 89803. Clin
Cancer Res. 18:890–900. 2012. View Article : Google Scholar
|
|
129
|
Gryfe R, Kim H, Hsieh ET, Aronson MD,
Holowaty EJ, Bull SB, Redston M and Gallinger S: Tumor
microsatellite instability and clinical outcome in young patients
with colorectal cancer. N Engl J Med. 342:69–77. 2000. View Article : Google Scholar
|
|
130
|
Ribic CM, Sargent DJ, Moore MJ, Thibodeau
SN, French AJ, Goldberg RM, Hamilton SR, Laurent-Puig P, Gryfe R,
Shepherd LE, et al: Tumor microsatellite-instability status as a
predictor of benefit from fluorouracil-based adjuvant chemotherapy
for colon cancer. N Engl J Med. 349:247–257. 2003. View Article : Google Scholar
|
|
131
|
Schee K, Boye K, Abrahamsen TW, Fodstad Ø
and Flatmark K: Clinical relevance of microRNA miR-21, miR-31,
miR-92a, miR-101, miR-106a and miR-145 in colorectal cancer. BMC
Cancer. 12:5052012. View Article : Google Scholar
|
|
132
|
Xuan Y, Yang H, Zhao L, Lau WB, Lau B, Ren
N, Hu Y, Yi T, Zhao X, Zhou S and Wei Y: MicroRNAs in colorectal
cancer: Small molecules with big functions. Cancer Lett.
360:89–105. 2015. View Article : Google Scholar
|
|
133
|
Shen WW, Zeng Z, Zhu WX and Fu GH:
MiR-142-3p functions as a tumor suppressor by targeting CD133,
ABCG2, and Lgr5 in colon cancer cells. J Mol Med (Berl).
91:989–1000. 2013. View Article : Google Scholar
|
|
134
|
Liu X, Wang Y and Zhao J: MicroRNA-337
inhibits colorectal cancer progression by directly targeting KRAS
and suppressing the AKT and ERK pathways. Oncol Rep. 38:3187–3196.
2017. View Article : Google Scholar
|
|
135
|
Wen L, Li Y, Jiang Z, Zhang Y, Yang B and
Han F: miR-944 inhibits cell migration and invasion by targeting
MACC1 in colorectal cancer. Oncol Rep. 37:3415–3422. 2017.
View Article : Google Scholar
|
|
136
|
Dai H, Hou K, Cai Z, Zhou Q and Zhu S:
Low-level miR-646 in colorectal cancer inhibits cell proliferation
and migration by targeting NOB1 expression. Oncol Lett.
14:6708–6714. 2017.
|
|
137
|
Xu Y, Chen J, Gao C, Zhu D, Xu X, Wu C and
Jiang J: MicroRNA-497 inhibits tumor growth through targeting
insulin receptor substrate 1 in colorectal cancer. Oncol Lett.
14:6379–6386. 2017.
|
|
138
|
Yang IP, Tsai HL, Miao ZF, Huang CW, Kuo
CH, Wu JY, Wang WM, Juo SH and Wang JY: Development of a
deregulating microRNA panel for the detection of early relapse in
postoperative colorectal cancer patients. J Transl Med. 14:1082016.
View Article : Google Scholar
|
|
139
|
Bovell LC, Shanmugam C, Putcha BD,
Katkoori VR, Zhang B, Bae S, Singh KP, Grizzle WE and Manne U: The
prognostic value of microRNAs varies with patient race/ethnicity
and stage of colorectal cancer. Clin Cancer Res. 19:3955–3965.
2013. View Article : Google Scholar
|
|
140
|
Peng Q, Zhang X, Min M, Zou L, Shen P and
Zhu Y: The clinical role of microRNA-21 as a promising biomarker in
the diagnosis and prognosis of colorectal cancer: A systematic
review and meta-analysis. Oncotarget. 8:44893–44909. 2017.
|
|
141
|
Cao J, Yan XR, Liu T, Han XB, Yu JJ, Liu
SH and Wang LB: MicroRNA-552 promotes tumor cell proliferation and
migration by directly targeting DACH1 via the Wnt/β-catenin
signaling pathway in colorectal cancer. Oncol Lett. 14:3795–3802.
2017. View Article : Google Scholar
|
|
142
|
Cheng D, Zhao S, Tang H, Zhang D, Sun H,
Yu F, Jiang W, Yue B, Wang J, Zhang M, et al: MicroRNA-20a-5p
promotes colorectal cancer invasion and metastasis by
downregulating Smad4. Oncotarget. 7:45199–45213. 2016. View Article : Google Scholar
|
|
143
|
Fukushima Y, Iinuma H, Tsukamoto M,
Matsuda K and Hashiguchi Y: Clinical significance of microRNA-21 as
a biomarker in each Dukes' stage of colorectal cancer. Oncol Rep.
33:573–582. 2015. View Article : Google Scholar
|
|
144
|
Shibuya H, Iinuma H, Shimada R, Horiuchi A
and Watanabe T: Clinicopathological and prognostic value of
microRNA-21 and microRNA-155 in colorectal cancer. Oncology.
79:313–320. 2010. View Article : Google Scholar
|
|
145
|
Wang LG and Gu J: Serum microRNA-29a is a
promising novel marker for early detection of colorectal liver
metastasis. Cancer Epidemiol. 36:e61–e67. 2012. View Article : Google Scholar
|
|
146
|
He PY, Yip WK, Chai BL, Chai BY, Jabar MF,
Dusa N, Mohtarrudin N and Seow HF: Inhibition of cell migration and
invasion by miR 29a 3p in a colorectal cancer cell line through
suppression of CDC42BPA mRNA expression. Oncol Rep. 38:3554–3566.
2017.
|
|
147
|
Lv ZC, Fan YS, Chen HB and Zhao DW:
Investigation of microRNA-155 as a serum diagnostic and prognostic
biomarker for colorectal cancer. Tumour Biol. 36:1619–1625. 2015.
View Article : Google Scholar
|
|
148
|
Zhao J, Xu J and Zhang R: MicroRNA-411
inhibits malignant biological behaviours of colorectal cancer cells
by directly targeting PIK3R3. Oncol Rep. 39:633–642. 2018.
|
|
149
|
Park YJ, Park KJ, Park JG, Lee KU, Choe KJ
and Kim JP: Prognostic factors in 2230 Korean colorectal cancer
patients: Analysis of consecutively operated cases. World J Surg.
23:721–726. 1999. View Article : Google Scholar
|
|
150
|
Park YJ, Youk EG, Choi HS, Han SU, Park
KJ, Lee KU, Choe KJ and Park JG: Experience of 1446 rectal cancer
patients in Korea and analysis of prognostic factors. Int J
Colorectal Dis. 14:101–106. 1999. View Article : Google Scholar
|
|
151
|
Wu ZY, Wan J, Zhao G, Peng L, Du JL, Yao
Y, Liu QF and Lin HH: Risk factors for local recurrence of middle
and lower rectal carcinoma after curative resection. World J
Gastroenterol. 14:4805–4809. 2008. View Article : Google Scholar
|
|
152
|
Locker GY, Hamilton S, Harris J, Jessup
JM, Kemeny N, Macdonald JS, Somerfield MR, Hayes DF and Bast RC Jr:
ASCO: ASCO 2006 update of recommendations for the use of tumor
markers in gastrointestinal cancer. J Clin Oncol. 24:5313–5327.
2006. View Article : Google Scholar
|
|
153
|
Ozawa T, Ishihara S, Kawai K, Nozawa H,
Yamaguchi H, Kitayama J and Watanabe T: Prognostic significance of
preoperative serum carbohydrate antigen 19-9 in patients with stage
IV colorectal cancer. Clin Colorectal Cancer. 15:e157–e163. 2016.
View Article : Google Scholar
|
|
154
|
Tol J, Koopman M, Miller MC, Tibbe A, Cats
A, Creemers GJ, Vos AH, Nagtegaal ID, Terstappen LW and Punt CJ:
Circulating tumour cells early predict progression-free and overall
survival in advanced colorectal cancer patients treated with
chemotherapy and targeted agents. Ann Oncol. 21:1006–1012. 2010.
View Article : Google Scholar
|
|
155
|
Sastre J, Maestro ML, Gómez-España A,
Rivera F, Valladares M, Massuti B, Benavides M, Gallén M, Marcuello
E, Abad A, et al: Circulating tumor cell count is a prognostic
factor in metastatic colorectal cancer patients receiving
first-line chemotherapy plus bevacizumab: A Spanish Cooperative
Group for the Treatment of Digestive Tumors study. Oncologist.
17:947–955. 2012. View Article : Google Scholar
|
|
156
|
Dinu D, Dobre M, Panaitescu E, Bîrlă R,
Iosif C, Hoara P, Caragui A, Boeriu M, Constantinoiu S and
Ardeleanu C: Prognostic significance of KRAS gene mutations in
colorectal cancer - preliminary study. J Med Life. 7:581–587.
2014.
|
|
157
|
Peeters M, Douillard JY, Van Cutsem E,
Siena S, Zhang K, Williams R and Wiezorek J: Mutant KRAS codon 12
and 13 alleles in patients with metastatic colorectal cancer:
Assessment as prognostic and predictive biomarkers of response to
panitumumab. J Clin Oncol. 31:759–765. 2013. View Article : Google Scholar
|
|
158
|
De Roock W, Jonker DJ, Di Nicolantonio F,
Sartore-Bianchi A, Tu D, Siena S, Lamba S, Arena S, Frattini M,
Piessevaux H, et al: Association of KRAS p.G13D mutation with
outcome in patients with chemotherapy-refractory metastatic
colorectal cancer treated with cetuximab. JAMA. 304:1812–1820.
2010. View Article : Google Scholar
|
|
159
|
Hecht JR, Douillard JY, Schwartzberg L,
Grothey A, Kopetz S, Rong A, Oliner KS and Sidhu R: Extended RAS
analysis for anti-epidermal growth factor therapy in patients with
metastatic colorectal cancer. Cancer Treat Rev. 41:653–659. 2015.
View Article : Google Scholar
|
|
160
|
Rajagopalan H, Bardelli A, Lengauer C,
Kinzler KW, Vogelstein B and Velculescu VE: Tumorigenesis: RAF/RAS
oncogenes and mismatch-repair status. Nature. 418:9342002.
View Article : Google Scholar
|
|
161
|
Toon CW, Walsh MD, Chou A, Capper D,
Clarkson A, Sioson L, Clarke S, Mead S, Walters RJ, Clendenning M,
et al: BRAFV600E immunohistochemistry facilitates universal
screening of colorectal cancers for Lynch syndrome. Am J Surg
Pathol. 37:1592–1602. 2013. View Article : Google Scholar
|
|
162
|
Kuan SF, Navina S, Cressman KL and Pai RK:
Immunohistochemical detection of BRAF V600E mutant protein using
the VE1 antibody in colorectal carcinoma is highly concordant with
molecular testing but requires rigorous antibody optimization. Hum
Pathol. 45:464–472. 2014. View Article : Google Scholar
|
|
163
|
De Roock W, Claes B, Bernasconi D, De
Schutter J, Biesmans B, Fountzilas G, Kalogeras KT, Kotoula V,
Papamichael D, Laurent-Puig P, et al: Effects of KRAS, BRAF, NRAS,
and PIK3CA mutations on the efficacy of cetuximab plus chemotherapy
in chemotherapy-refractory metastatic colorectal cancer: A
retrospective consortium analysis. Lancet Oncol. 11:753–762. 2010.
View Article : Google Scholar
|
|
164
|
Popat S, Hubner R and Houlston RS:
Systematic review of microsatellite instability and colorectal
cancer prognosis. J Clin Oncol. 23:609–618. 2005. View Article : Google Scholar
|
|
165
|
Sargent DJ, Marsoni S, Thibodeau SN,
Labianca R, Hamilton SR, Torri V, Monges G, Ribic C, Grothey A and
Gallinger S: Confirmation of deficient mismatch repair (dMMR) as a
predictive marker for lack of benefit from 5-FU based chemotherapy
in stage II and III colon cancer (CC): A pooled molecular
reanalysis of randomized chemotherapy trials. J Clin Oncol. 26 15
suppl:40082008. View Article : Google Scholar
|
|
166
|
Jover R, Zapater P, Castells A, Llor X,
Andreu M, Cubiella J, Balaguer F, Sempere L, Xicola RM, Bujanda L,
et al: Gastrointestinal Oncology Group of the Spanish
Gastroenterological Association: The efficacy of adjuvant
chemotherapy with 5-fluorouracil in colorectal cancer depends on
the mismatch repair status. Eur J Cancer. 45:365–373. 2009.
View Article : Google Scholar
|
|
167
|
Pohl A, El-Khoueiry A, Yang D, Zhang W,
Lurje G, Ning Y, Winder T, Hu-Lieskoven S, Iqbal S, Danenberg KD,
et al: Pharmacogenetic profiling of CD133 is associated with
response rate (RR) and progression-free survival (PFS) in patients
with metastatic colorectal cancer (mCRC), treated with
bevacizumab-based chemotherapy. Pharmacogenomics J. 13:173–180.
2013. View Article : Google Scholar
|
|
168
|
Ong CW, Kim LG, Kong HH, Low LY, Iacopetta
B, Soong R and Salto-Tellez M: CD133 expression predicts for
non-response to chemotherapy in colorectal cancer. Mod Pathol.
23:450–457. 2010. View Article : Google Scholar
|
|
169
|
Nakamura TM, Morin GB, Chapman KB,
Weinrich SL, Andrews WH, Lingner J, Harley CB and Cech TR:
Telomerase catalytic subunit homologs from fission yeast and human.
Science. 277:955–959. 1997. View Article : Google Scholar
|
|
170
|
Wu X, Amos CI, Zhu Y, Zhao H, Grossman BH,
Shay JW, Luo S, Hong WK and Spitz MR: Telomere dysfunction: a
potential cancer predisposition factor. J Natl Cancer Inst.
95:1211–1218. 2003. View Article : Google Scholar
|
|
171
|
Willeit P, Willeit J, Mayr A, Weger S,
Oberhollenzer F, Brandstätter A, Kronenberg F and Kiechl S:
Telomere length and risk of incident cancer and cancer mortality.
JAMA. 304:69–75. 2010. View Article : Google Scholar
|
|
172
|
Calado RT and Young NS: Telomere diseases.
N Engl J Med. 361:2353–2365. 2009. View Article : Google Scholar
|
|
173
|
O'Sullivan J, Risques RA, Mandelson MT,
Chen L, Brentnall TA, Bronner MP, Macmillan MP, Feng Z, Siebert JR,
Potter JD and Rabinovitch PS: Telomere length in the colon declines
with age: A relation to colorectal cancer? Cancer Epidemiol
Biomarkers Prev. 15:573–577. 2006. View Article : Google Scholar
|
|
174
|
Raynaud CM, Jang SJ, Nuciforo P,
Lantuejoul S, Brambilla E, Mounier N, Olaussen KA, André F, Morat
L, Sabatier L and Soria JC: Telomere shortening is correlated with
the DNA damage response and telomeric protein down-regulation in
colorectal preneoplastic lesions. Ann Oncol. 19:1875–1881. 2008.
View Article : Google Scholar
|
|
175
|
Aghagolzadeh P and Radpour R: New trends
in molecular and cellular biomarker discovery for colorectal
cancer. World J Gastroenterol. 22:5678–5693. 2016. View Article : Google Scholar
|
|
176
|
Fernández-Marcelo T, Sánchez-Pernaute A,
Pascua I, De Juan C, Head J, Torres-García A-J and Iniesta P:
Clinical relevance of telomere status and telomerase activity in
colorectal cancer. PLoS One. 11:e01496262016. View Article : Google Scholar
|
|
177
|
Gertler R, Rosenberg R, Stricker D,
Friederichs J, Hoos A, Werner M, Ulm K, Holzmann B, Nekarda H and
Siewert JR: Telomere length and human telomerase reverse
transcriptase expression as markers for progression and prognosis
of colorectal carcinoma. J Clin Oncol. 22:1807–1814. 2004.
View Article : Google Scholar
|
|
178
|
Norppa H and Falck GC: What do human
micronuclei contain? Mutagenesis. 18:221–233. 2003. View Article : Google Scholar
|
|
179
|
Farhadi S and Mohamadi M and Mohamadi M:
Repair index in examination of nuclear changes in the buccal mucosa
of smokers: a useful method for screening of oral cancer. Asian Pac
J Cancer Prev. 18:3087–3090. 2017.
|
|
180
|
El-Zein RA, Abdel-Rahman S, Santee KJ, Yu
R and Shete S: Identification of small and non-small cell lung
cancer markers in peripheral blood using cytokinesis-blocked
micronucleus and spectral karyotyping assays. Cytogenet Genome Res.
152:122–131. 2017. View Article : Google Scholar
|
|
181
|
Maffei F, Moraga Zolezzi JM, Angelini S,
Zenesini C, Musti M, Festi D, Cantelli-Forti G and Hrelia P:
Micronucleus frequency in human peripheral blood lymphocytes as a
biomarker for the early detection of colorectal cancer risk.
Mutagenesis. 29:221–225. 2014. View Article : Google Scholar
|
|
182
|
Karaman A, Binici DN, Kabalar ME and
Calikuşu Z: Micronucleus analysis in patients with colorectal
adenocarcinoma and colorectal polyps. World J Gastroenterol.
14:6835–6839. 2008. View Article : Google Scholar
|
|
183
|
Ravegnini G, Moraga Zolezzi JM, Maffei F,
Musti M, Zenesini C, Simeon V, Sammarini G, Festi D, Hrelia P and
Angelini S: Simultaneous analysis of SEPT9 promoter methylation
status, micronuclei frequency, and folate-related gene
polymorphisms: The potential for a novel blood-based colorectal
cancer biomarker. Int J Mol Sci. 16:28486–284897. 2015. View Article : Google Scholar
|
|
184
|
Ionescu EM, Nicolaie T, Ionescu MA,
Becheanu G, Andrei F, Diculescu M and Ciocirlan M: Predictive
cytogenetic biomarkers for colorectal neoplasia in medium risk
patients. J Med Life. 8:398–403. 2015.
|
|
185
|
Kronborg O, Jørgensen OD, Fenger C and
Rasmussen M: Randomized study of biennial screening with a faecal
occult blood test: Results after nine screening rounds. Scand J
Gastroenterol. 39:846–851. 2004. View Article : Google Scholar
|
|
186
|
Scholefield JH, Moss S, Sufi F, Mangham CM
and Hardcastle JD: Effect of faecal occult blood screening on
mortality from colorectal cancer: Results from a randomised
controlled trial. Gut. 50:840–844. 2002. View Article : Google Scholar
|
|
187
|
Health Quality Ontario, . Fecal occult
blood test for colorectal cancer screening: An evidence-based
analysis. Ont Health Technol Assess Ser. 9:1–40. 2009.
|
|
188
|
Dancourt V, Lejeune C, Lepage C, Gailliard
MC, Meny B and Faivre J: Immunochemical faecal occult blood tests
are superior to guaiac-based tests for the detection of colorectal
neoplasms. Eur J Cancer. 44:2254–2258. 2008. View Article : Google Scholar
|
|
189
|
Faivre J, Dancourt V, Denis B, Dorval E,
Piette C, Perrin P, Bidan JM, Jard C, Jung S, Levillain R, et al:
Comparison between a guaiac and three immunochemical faecal occult
blood tests in screening for colorectal cancer. Eur J Cancer.
48:2969–2976. 2012. View Article : Google Scholar
|
|
190
|
Lin JS, Piper MA, Perdue LA, Rutter CM,
Webber EM, O'Connor E, Smith N and Whitlock EP: Screening for
colorectal cancer: Updated evidence report and systematic review
for the US Preventive Services Task Force. JAMA. 315:2576–2594.
2016. View Article : Google Scholar
|
|
191
|
Koo S, Neilson LJ, Von Wagner C and Rees
CJ: The NHS bowel cancer screening program: Current perspectives on
strategies for improvement. Risk Manag Healthc Policy. 10:177–187.
2017. View Article : Google Scholar
|
|
192
|
Moss S, Mathews C, Day TJ, Smith S, Seaman
HE, Snowball J and Halloran SP: Increased uptake and improved
outcomes of bowel cancer screening with a faecal immunochemical
test: Results from a pilot study within the national screening
programme in England. Gut. 66:1631–1644. 2017. View Article : Google Scholar
|
|
193
|
Rozen P, Levi Z, Hazazi R, Waked A, Vilkin
A, Maoz E, Birkenfeld S and Niv Y: Quantitative colonoscopic
evaluation of relative efficiencies of an immunochemical faecal
occult blood test and a sensitive guaiac test for detecting
significant colorectal neoplasms. Aliment Pharmacol Ther.
29:450–457. 2009. View Article : Google Scholar
|
|
194
|
Hoffman RM, Steel S, Yee EFT, Massie L,
Schrader RM and Murata GH: Colorectal cancer screening adherence is
higher with fecal immunochemical tests than guaiac-based fecal
occult blood tests: A randomized, controlled trial. Prev Med.
50:297–299. 2010. View Article : Google Scholar
|
|
195
|
Brenner H and Tao S: Superior diagnostic
performance of faecal immunochemical tests for haemoglobin in a
head-to-head comparison with guaiac based faecal occult blood test
among 2235 participants of screening colonoscopy. Eur J Cancer.
49:3049–3054. 2013. View Article : Google Scholar
|
|
196
|
Fitzpatrick-Lewis D, Ali MU, Warren R,
Kenny M, Sherifali D and Raina P: Screening for colorectal cancer:
A systematic review and meta-analysis. Clin Colorectal Cancer.
15:298–313. 2016. View Article : Google Scholar
|
|
197
|
Murphy J, Halloran S and Gray A:
Cost-effectiveness of the faecal immunochemical test at a range of
positivity thresholds compared with the guaiac faecal occult blood
test in the NHS Bowel Cancer Screening Programme in England. BMJ
Open. 7:e0171862017. View Article : Google Scholar
|
|
198
|
Lee JK, Liles EG, Bent S, Levin TR and
Corley DA: Accuracy of fecal immunochemical tests for colorectal
cancer: Systematic review and meta-analysis. Ann Intern Med.
160:1712014. View Article : Google Scholar
|