Introduction
In American males, prostate cancer is the most
common noncutaneous malignancy and is the second leading cause of
cancer-associated mortality (1).
In 2013, the American Cancer Society predicted the diagnosis of
~238,590 novel cases of prostate cancer in the USA with 29,720
fatalities (2,3). The majority of prostate
cancer-associated mortality is attributed to metastatic disease
rather than primary, organ-confined prostate cancer (4,5).
Previous studies have demonstrated that, due to dietary factors,
the incidence of metastatic prostate cancer is lower in Asian males
compared with Western males (6)
According to these results, it may be possible to identify agents
from the differences of Asian and Western food, for use as
chemopreventive agents.
The flavonoid, 4′,5,7-trihydroxyflavone (apigenin),
is one of the most common flavonoids and is widely distributed in
several types of fruit and vegetable, including parsley, onions,
orange, tea, chamomile, wheat sprouts and seasonings (7). It has been increasingly recognized as
a chemopreventive agent against cancer, exhibiting antimutagenic,
anti-inflammatory, antiviral and purgative effects (8). The chemopreventive activity of
apigenin in multiple organ sites is likely to be associated with
its ability to modulate pathways involved in cell cycle control
(9), apoptosis (9,10)
and signal transduction (11). One
important reason why apigenin has become noteworthy as a beneficial
and chemopreventive agent is its low intrinsic toxicity (12).
Epithelial mesenchymal transition (EMT) is important
in the invasion and metastasis of human cancer (13) EMT is an important process, in which
epithelial cells lose their polarity and convert into more motile
and invasive mesenchymal phenotypes. Previous studies have
demonstrated the inhibitory effect of apigenin on other human
cancer cells (14–17). The present study aimed to
demonstrate the effect of apigenin on EMT in prostate cancer.
The present study aimed to determine whether
apigenin decreased the migration and invasion ability of prostate
cancer cells and its underlying mechanism.
Materials and methods
Reagents and cell culture
Apigenin (≥99% pure) and MTT were purchased from
Sigma (St. Louis, MO, USA). Primary antibodies against E-cadherin,
Snail, vimentin and GAPDH and secondary antibodies were purchased
from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). The
antibodies were all rabbit-type and monoclonal. The bicinchoninic
acid protein assay kit was obtained from Pierce Biotechnology
(Rockford, IL, USA).
The DU145 human prostate cancer cell line was
obtained from the Shanghai Institute of Cell Biology, Chinese
Academy of Sciences (Shanghai, China). The cells were cultured in
RPMI-1640 medium (HyClone, Logan, UT, USA) supplemented with 10%
heat-inactivated fetal bovine serum (FBS; JRH Biosciences, Lenexa,
KS, USA), 100 U/ml penicillin and 100 mg/l streptomycin (Sigma, St.
Louis, MO, USA). The cultures were then maintained in a humidified
atmosphere of 5% CO2 at 37°C.
Cell viability assay
The effect of apigenin on the viability of DU145
cells was evaluated using an MTT assay. The DU145 cells
(~10×104) were seeded onto 96-well plates. Following
incubation overnight, the cells were treated with apigenin at
different concentrations (0–80 μM) in dimethyl sulfoxide (DMSO) for
24 and 48 h in a humidified atmosphere of 5% CO2 at
37°C. Following incubation for the indicated duration, MTT (20 μl
of 5 mg/ml) was added to each well and incubated at 37°C for 4 h.
The MTT solution in the medium was then removed. To achieve
solubilization of the formazan crystal formed in the viable cells,
150 μl DMSO was added to each well prior to measuring the
absorbance at 490 nm using an MRX II absorbance reader (Dynex
Technologies, Chantilly, VA, USA). The results were expressed as a
percentage of growth.
Wound healing assays
Cells were grown to ~100% confluence in 6-well
plates following treatment with apigenin. The cell monolayers were
wounded through scraping with a micropipette tip and the wound
closure was observed after 24 h. Images were captured under a
phase-contrast microscope DP80 (Olympus, Tokyo, Japan) immediately
or 24 h after wounding. The experiments were repeated three
times.
In vitro invasion and motility
assays
The invasion and motility assays were performed, as
previously described (18) with
minor modifications. The cells were plated onto a 6-well plate
(8×104 cells/well). Following incubation overnight, the
cells were treated with apigenin at different concentrations (0–20
μM) in DMSO for 24 h and harvested. The treated and control cells
were suspended in medium at a concentration of 4×105
cells/ml and 0.2 ml of the medium with suspended cells was added to
the top chamber of uncoated and Matrigel-coated PET (BD Bioscience,
Franklin Lakes, NJ, USA) membranes for motility and invasion
assays, respectively (24-well insert; 8 μm pore size; Millipore,
Bedford, MA, USA). The medium (0.6 ml), supplemented with 20% FBS,
was then added to each well of the plate to act as a
chemoattractant in the lower chamber. The cells were incubated for
24 h and any cells that did not migrate through the pores to the
lower surface of the membrane were removed using a cotton swab by
scraping the upper membrane surface. The cells that migrated to the
lower surface of the membrane were fixed in 100% methanol for 5 min
and stained with 0.1% crystal violet for 2 min (Sigma). These
experiments were performed a minimum of three times.
Cell cycle assay
The cells were plated onto six-well culture dishes
at concentrations 80,000 cells/well that were determined to yield
60–70% confluence within 24 h. The cells were then treated with
apigenin at a range of concentrations (0–80 μM) in DMSO. After 24
h, the cells were washed twice with phosphate-buffered saline (PBS)
and centrifuged at 800 × g for 5 min. The pellet was fixed with 70%
ethanol for 1 h at 4°C. Following washing with PBS, the cells were
resuspended with propidium iodide solution (0.05 mg/ml) containing
RNase and incubated in the dark at room temperature for 30 min. The
DNA content was then analyzed using an FC500 flow cytometer (BD
Bioscience).
Western blot analysis
The cells were harvested 24 h after treatment with
apigenin. These cells were washed and lysed with lysis buffer (10
mmol/l Tris-hydrochloride, 0.25 mol/l sucrose, 5 mmol/l EDTA, 50
mmol/l NaCl, 30 mmol/l sodium pyrophosphate, 50 mmol/l NaF, 1
mmol/l Na3VO4, 1 mmol/l PMSF and 2% cocktail;
pH 7.5). The protein concentration in the resulting lysate was
determined using a bicinchoninic acid protein assay (Merck KGaA,
Darmstadt, Germany). Appropriate quantities of protein (20–30 μg)
were separated by electrophoresis on 10–12% Tris-glycine
polyacrylamide gels and transferred onto nitrocellulose membranes.
The membranes were inhibited and incubated overnight with the
appropriate primary antibody at dilutions according to the
manufacturer’s instructions. The membranes were subsequently washed
and incubated with the corresponding horseradish
peroxidase-conjugated secondary antibody at 1:1,000 dilution in
Tris-buffered saline-Tween 20 (10 mM Tris-Cl; pH 7.4, 150 mM NaCl
and 0.1% Tween-20). The bound secondary antibody was then detected
using an enhanced chemiluminescence system (Pierce Biotechnology
Inc., Rockford, IL, USA).
Statistical analysis
Statistical significance was compared between the
various treatment groups and the controls using analysis of
variance. P<0.05 was considered to indicate a statistically
significant difference. All statistical analyses were performed
using Prism software (GraphPad Prism 6; GraphPad, La Jolla, CA,
USA).
Results
Apigenin inhibits cell growth in DU145
cells
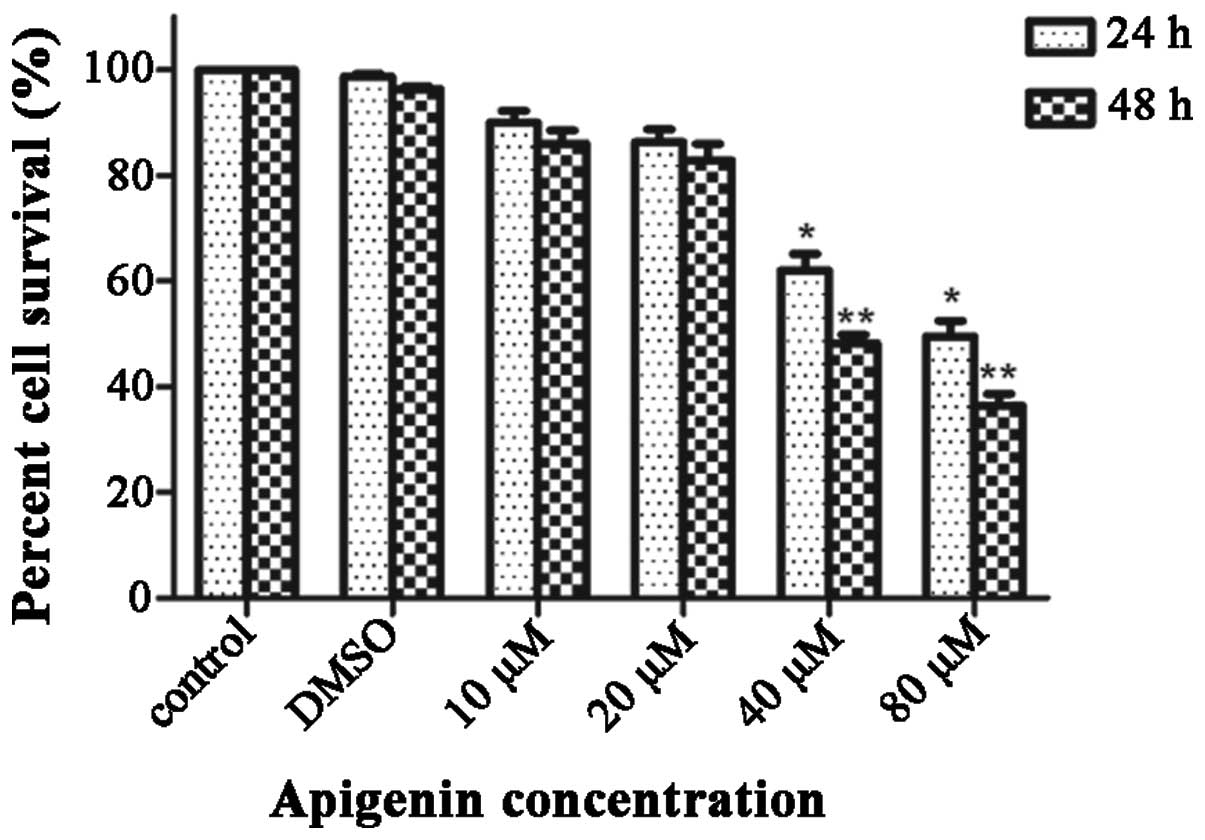
The MTT assay revealed that treatment with apigenin
with the vehicle DMSO (1 μl/ml) at varying concentrations (10–80
μM) for different time periods (24–48 h), resulted in dose and
time-dependent inhibition of DU145 cell growth compared with the
untreated control. As is shown in Fig.
1, no significant difference was identified between the
untreated control and the vehicle control indicating that DMSO did
not affect the proliferation of DU145 cells. Treatment with 10 μM
apigenin caused minimal change in the viability of the cells,
therefore the migration and invasion assays were performed using
concentrations of 0–20 μM apigenin. With increasing concentration
and time, a marked reduction in cell viability was observed,
particularly at concentrations of 40 and 80 μM. The inhibitory
concentration 50% values for apigenin treatment were estimated to
be 80.8 and 49.3 μM for 24 and 48 h, respectively. These data
indicated that apigenin exerted a significant inhibitory effect on
the DU145 cells.
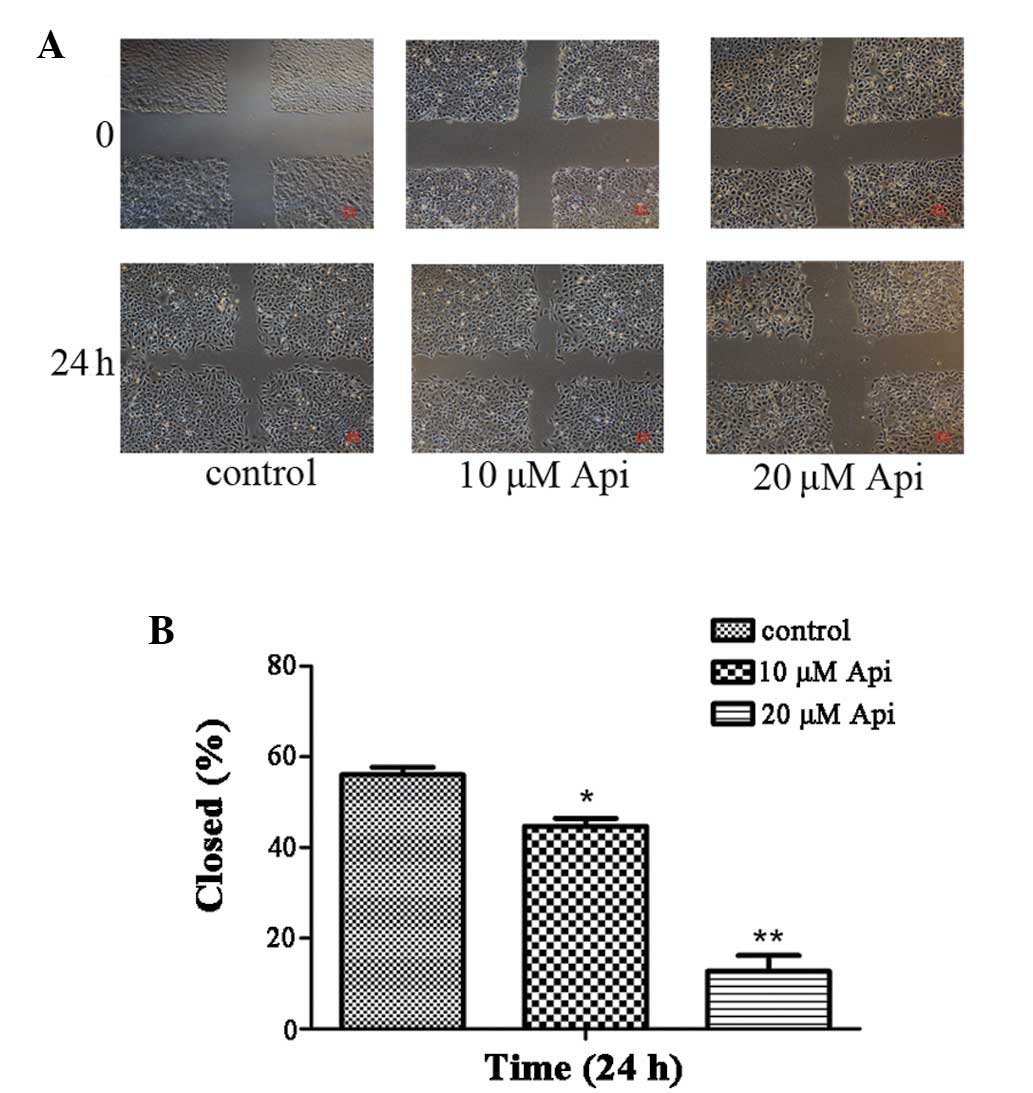
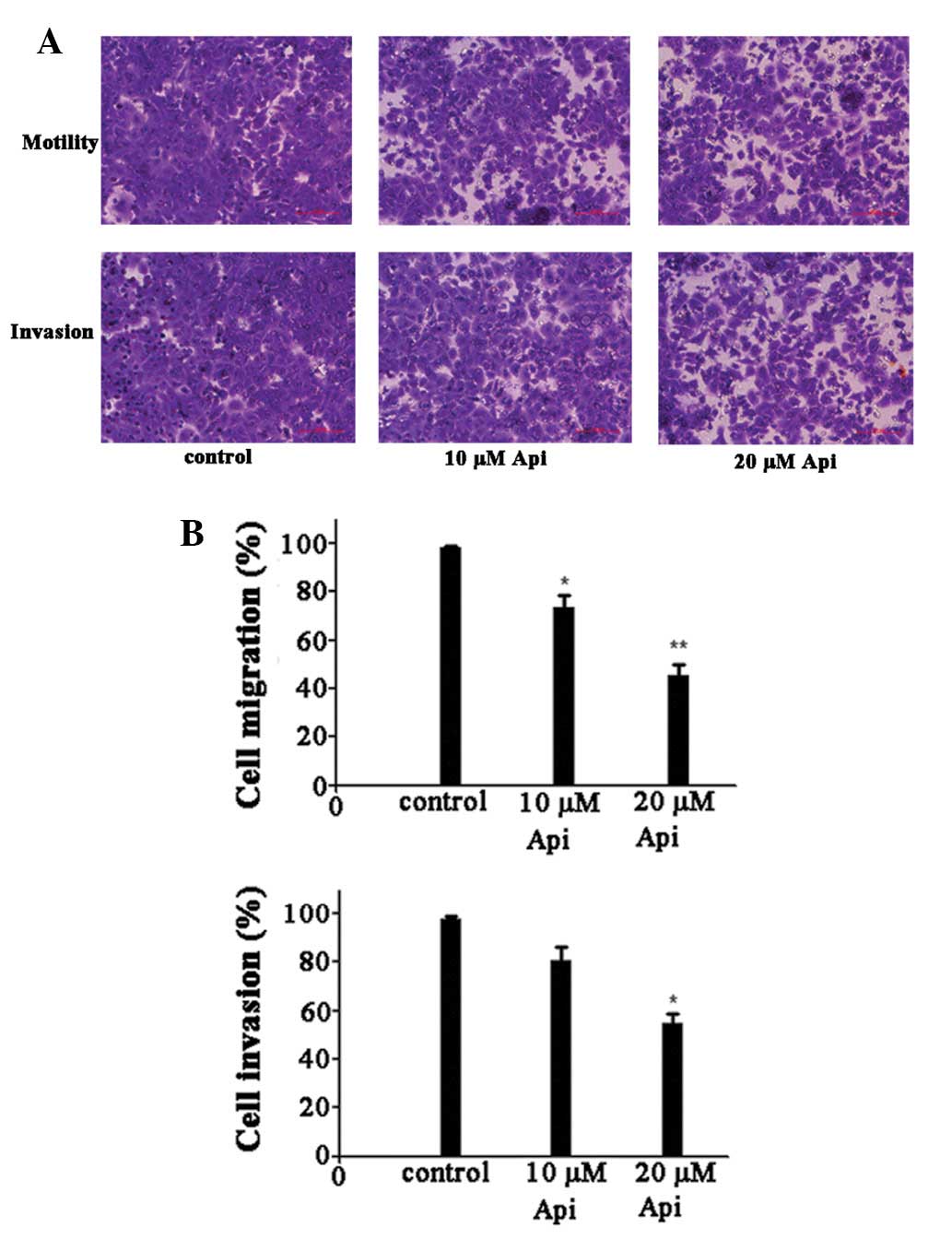
Apigenin inhibits DU145 cell migration
and invasion
As the low concentration of apigenin did not induce
significant DU145 cell death, the DU145 cells were treated with
0–20 μM apigenin to detect whether the migration and invasion
potential of the DU145 cells was decreased at the low
concentration. Wound healing assays and transwell assays were
performed to observe the function. As shown in Fig. 2, the wound healing assays revealed
that apigenin-treated cells exhibited retarded wound closure and
fewer stained cells in a dose-dependent manner. As shown in
Fig. 3, a significant decrease in
motility and invasion was observed compared with the untreated
control. These results demonstrated that treatment with apigenin
caused significant suppression of the migratory and invasive
capability of the DU145 cells.
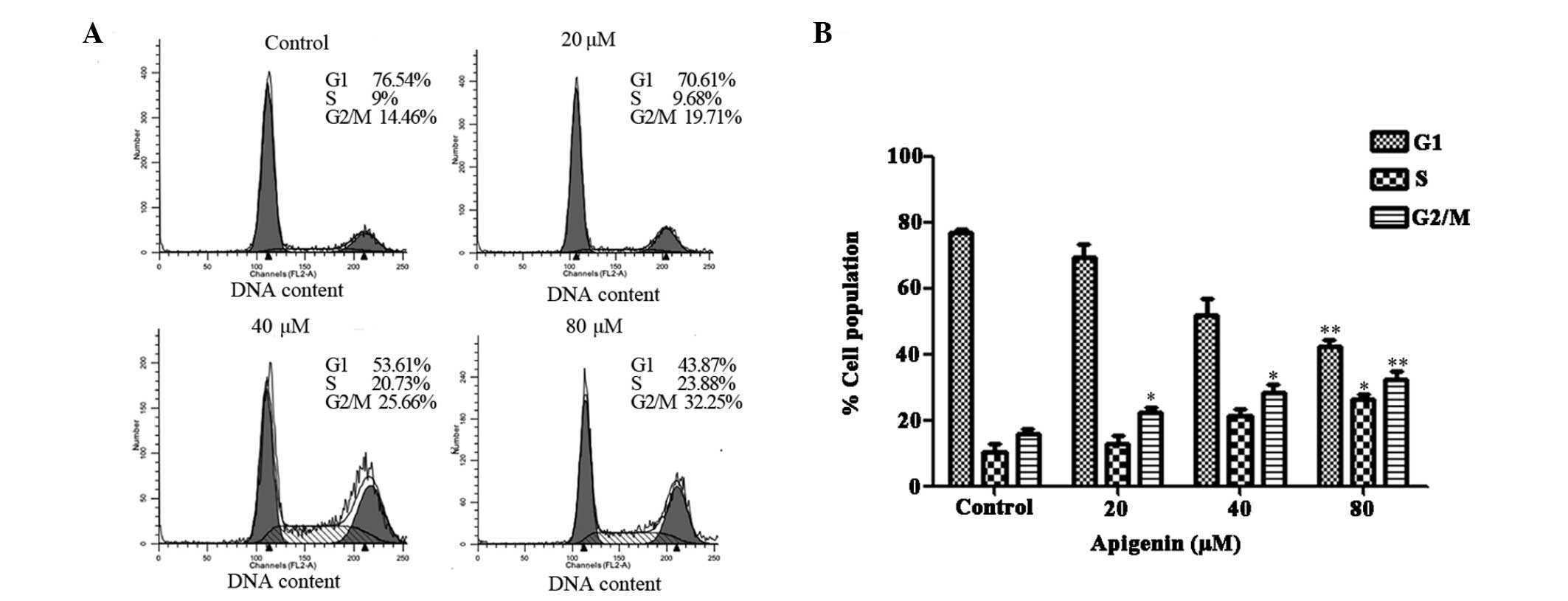
Apigenin induces G2/M phase cell cycle
arrest
The effect of apigenin on cell cycle perturbations
was also assessed. Compared with the untreated controls, treatment
with apigenin led to a marked arrest of the DU145 cells in the G2/M
phase of the cell cycle. Cell cycle analysis demonstrated that the
population of control cells in the G2/M phase was 14.46% and the
percentage of cells in the G2/M phase increased significantly
following treatment with apigenin at different concentrations for
24 h (Fig. 4A). This increase in
the population of cells in the G2/M phase was accompanied by a
concomitant decrease in the number of cells in the G1 phase of the
cell cycle (Fig. 4B).
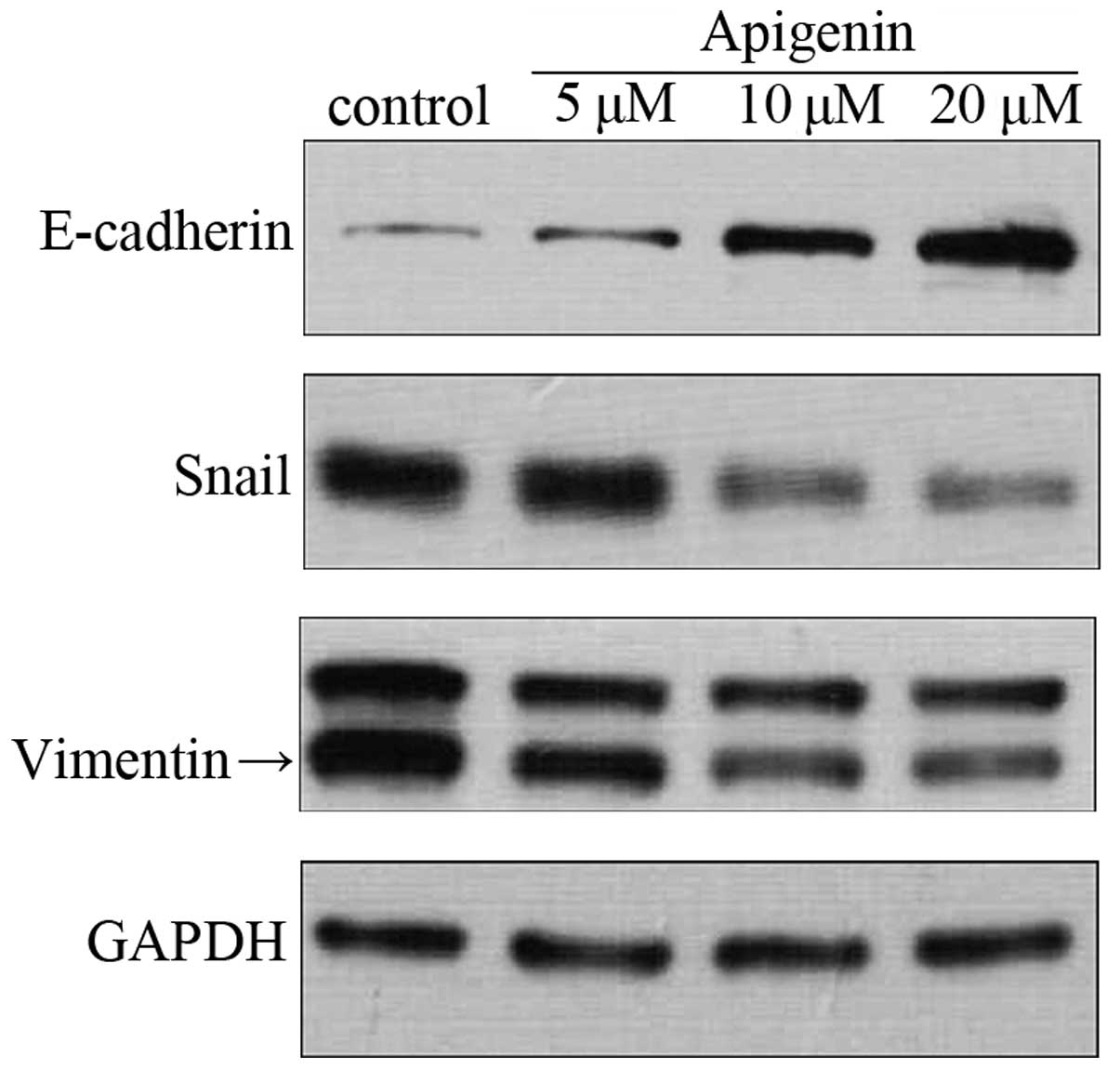
Apigenin reverses EMT in DU145 cells
EMT is important in metastasis. The loss of
E-cadherin is key in EMT (19),
therefore the dose-dependent effects of apigenin on the
constitutive protein levels of E-cadherin, Snail and vimentin in
DU145 cells was examined. The western blot analysis revealed a
significant increase in the expression of E-cadherin, while in
sharp contrast, the protein expression of Snail and vimentin was
significantly decreased by apigenin treatment in a dose-dependent
manner (Fig. 5). These results
demonstrated that apigenin reversed the process of EMT.
Discussion
The present study investigated whether apigenin
affected EMT in DU145 prostate cancer cells. Emerging evidence has
suggested that EMT is a morphological event that is crucial for
tumor progression in terms of its physiological and metastatic
development (20). EMT is defined
by the loss of cell-cell adhesion, modification of cell morphology
and the increase of cellular migration activity (20). Downregulation of the epithelial
marker E-cadherin, the main transmembrane adhesion molecule
responsible for cell-to-cell adhesions and tissue organization in
epithelial tissue, and upregulation of the mesenchymal marker
vimentin is one of the basic mechanisms involved in EMT (21). This suggests that the loss of
E-cadherin expression induces the breakdown of adherens junctions
and, in addition to other signaling events, promotes changes in the
expression of the robust gene (22) and suppresses the dissociation of
epithelial cells from their location (23). In the present study, when the DU145
cells were treated with different concentrations of apigenin, the
expression of E-cadherin was rescued and that of vimentin was
markedly inhibited, indicating that the progression of EMT was
reversed and metastatic capability was suppressed.
There is now clear evidence that a number of factors
are involved in the induction of EMT and that their signaling
pathways communicate extensively with each other and with
oncogenes. The activation of Ras, a receptor tyrosine kinase, and
its signaling system not only provides a continuous growth stimulus
for cancer, but also potentially induces EMT (24). Additionally, all pathways
downstream of Ras (phosphoinositide 3-kinase; PI3K, Raf and
Ral-GEF) have been demonstrated to induce EMT (25). Transforming growth factor β, a
cytokine produced by mesenchymal stromal and inflammatory cells
(26), may be the most extensively
investigated of the stromal factors facilitating the induction of
malignant cells to undergo EMT. The transcriptional repressors,
Snail, Slug, Zeb and Twist also control EMT. Snail and Zeb are
zinc-finger transcription factors that bind to the E-boxes of the
E-cadherin-encoding gene (CDH1) promoter, thereby repressing the
expression of CDH1 (27). In the
present study, the downregulation of Snail expression suggested
that apigenin inhibited EMT by targeting the Snail regulator. Our
previous study demonstrated that apigenin treatment in T24 bladder
cancer cells inhibited the PI3K/Akt pathway (28), which suggested that apigenin
reversed EMT, possibly through the PI3K/protein kinase B pathway.
In future studies, the complex mechanism between apigenin and EMT
requires further examination.
Previous studies have also revealed that apigenin is
a potent inhibitor of cell-cycle progression in several cell lines
(29,30). The present study also measured the
effect of apigenin on the cell cycle of DU145 cells and found that
apigenin led to G2/M phase arrest. Although G1 arrest by apigenin
in human diploid fibroblasts has been observed previously (30), the present study observed G2/M
arrest in apigenin-treated DU145 cells and the difference between
these results may be attributed to the types of cell assessed.
In conclusion, the present study demonstrated that
apigenin induced cell death and inhibited the migration and
invasion ability of DU145 prostate cancer cells in a dose and
time-dependent manner. Apigenin led to a reversal of EMT and also
caused G2/M phase arrest. All these results indicated that apigenin
can be used as a chemopreventive agent in prostate cancer. To the
best of our knowledge, this is the first study demonstrating the
effect of apigenin on EMT in prostate cancer in vitro.
However, further investigation of the mechanism underlying
apigenin-treated cell inhibition is required.
Acknowledgements
This study was supported by the National Key
Clinical Specialty Construction Project of China, Key Medical
Disciplines of Zhejiang Province, Combination of Traditional
Chinese and Western Medicine Key Disciplines of Zhejiang Province
(no. 2012-XK-A23), the National Natural Science Foundation of China
(no. 81101717), the Natural Science Foundation of Zhejiang Province
(nos. LY13H160009 and Y2110120) and the Medical Scientific Research
Foundation of Zhejiang Province, China (no. 2013KYB100).
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siegel R, DeSantis C, Virgo K, et al:
Cancer treatment and survivorship statistics, 2012. CA Cancer J
Clin. 62:220–241. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
American Cancer Society. Prostate cancer
facts and statistics. http://www.cancer.org/cancer/prostatecancer/index.
November 13–2013
|
|
4
|
Cook LS, Goldoft M, Schwartz SM and Weiss
NS: Incidence of adenocarcinoma of the prostate in Asian immigrants
to the United States and their descendants. J Urol. 161:152–155.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Arya M, Bott SR, Shergill IS, Ahmed HU,
Williamson M and Patel HR: The metastatic cascade in prostate
cancer. Surg Oncol. 15:117–128. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Adlercreutz H: Western diet and Western
diseases: some hormonal and biochemical mechanisms and
associations. Scand J Clini Lab Invest Suppl. 50:3–23. 1990.
View Article : Google Scholar
|
|
7
|
Birt DF, Mitchell D, Gold B, Pour P and
Pinch HC: Inhibition of ultraviolet light induced skin
carcinogenesis in SKH-1 mice by apigenin, a plant flavonoid.
Anticancer Res. 17:85–91. 1997.PubMed/NCBI
|
|
8
|
Yang CS, Landau JM, Huang MT and Newmark
HL: Inhibition of carcinogenesis by dietary polyphenolic compounds.
Annu Rev Nutr. 21:381–406. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Patel D, Shukla S and Gupta S: Apigenin
and cancer chemoprevention: Progress, potential and promise
(review). Int J Oncology. 30:233–245. 2007.
|
|
10
|
Shukla S and Gupta S: Molecular targets
for apigenin-induced cell cycle arrest and apoptosis in prostate
cancer cell xenograft. Mol Cancer Ther. 5:843–852. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Van Dross RT, Hong X and Pelling JC:
Inhibition of TPA-induced cyclooxygenase-2 (COX-2) expression by
apigenin through downregulation of Akt signal transduction in human
keratinocytes. Mol Carcinog. 44:83–91. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Gupta S, Afaq F and Mukhtar H: Selective
growth-inhibitory, cell-cycle deregulatory and apoptotic response
of apigenin in normal versus human prostate carcinoma cells.
Biochem Biophys Res Commun. 287:914–920. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yilmaz M and Christofori G: EMT, the
cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev.
28:15–33. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Caltagirone S, Rossi C, Poggi A, et al:
Flavonoids apigenin and quercetin inhibit melanoma growth and
metastatic potential. Int J Cancer. 87:595–600. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang IK, Lin-Shiau SY and Lin JK:
Induction of apoptosis by apigenin and related flavonoids through
cytochrome c release and activation of caspase-9 and caspase-3 in
leukaemia HL-60 cells. Eur J Cancer. 35:1517–1525. 1999. View Article : Google Scholar
|
|
16
|
Wang W, Heideman L, Chung CS, Pelling JC,
Koehler KJ and Birt DF: Cell-cycle arrest at G2/M and growth
inhibition by apigenin in human colon carcinoma cell lines. Mol
Carcinog. 28:102–110. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yin F, Giuliano AE, Law RE and Van Herle
AJ: Apigenin inhibits growth and induces G2/M arrest by modulating
cyclin-CDK regulators and ERK MAP kinase activation in breast
carcinoma cells. Anticancer Res. 21:413–420. 2001.PubMed/NCBI
|
|
18
|
Albini A, Iwamoto Y, Kleinman HK, et al: A
rapid in vitro assay for quantitating the invasive potential of
tumor cells. Cancer Res. 47:3239–3245. 1987.PubMed/NCBI
|
|
19
|
Jaggi M, Johansson SL, Baker JJ, Smith LM,
Galich A and Balaji KC: Aberrant expression of E-cadherin and
beta-catenin in human prostate cancer. Urol Oncol. 23:402–406.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Thiery JP: Epithelial-mesenchymal
transitions in tumour progression. Nat Rev Cancer. 2:442–454. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Huber MA, Kraut N and Beug H: Molecular
requirements for epithelial-mesenchymal transition during tumor
progression. Curr Opin Cell Biol. 17:548–558. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sarkar FH, Li Y, Wang Z and Kong D:
Pancreatic cancer stem cells and EMT in drug resistance and
metastasis. Minerva Chir. 64:489–500. 2009.PubMed/NCBI
|
|
23
|
Vleminckx K, Vakaet L Jr, Mareel M, Fiers
W and van Roy F: Genetic manipulation of E-cadherin expression by
epithelial tumor cells reveals an invasion suppressor role. Cell.
66:107–119. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bates RC and Mercurio AM: The
epithelial-mesenchymal transition (EMT) and colorectal cancer
progression. Cancer Biol Ther. 4:365–370. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thiery JP: Epithelial-mesenchymal
transitions in development and pathologies. Curr Opin Cell Biol.
15:740–746. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Branton MH and Kopp JB: TGF-beta and
fibrosis. Microbes Infect. 1:1349–1365. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chaffer CL, Brennan JP, Slavin JL, Blick
T, Thompson EW and Williams ED: Mesenchymal-to-epithelial
transition facilitates bladder cancer metastasis: role of
fibroblast growth factor receptor-2. Cancer Res. 66:11271–11278.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhu Y, Mao Y, Chen H, et al: Apigenin
promotes apoptosis, inhibits invasion and induces cell cycle arrest
of T24 human bladder cancer cells. Cancer Cell Int. 13:542013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lepley DM, Li B, Birt DF and Pelling JC:
The chemopreventive flavonoid apigenin induces G2/M arrest in
keratinocytes. Carcinogenesis. 17:2367–2375. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lepley DM and Pelling JC: Induction of
p21/WAF1 and G1 cell-cycle arrest by the chemopreventive agent
apigenin. Mol Carcinog. 19:74–82. 1997. View Article : Google Scholar : PubMed/NCBI
|