Introduction
Autoimmunity is the failure of an organism to
recognize its own constituents as self, which causes an immune
response against its own cells and tissues. Any disease resulting
from this aberrant immune response is termed autoimmune disease
(AID) (1). AID is frequently
observed clinically and therapy for AID often includes treatment
with hormones and immunosuppressants (1). These agents are effective in the
short-term, however, long-term use increases the incidence of
tumors and uncontrolled infectious diseases due to various side
effects (1,2). Therefore, AID is recognized worldwide
as a ‘5D’ disease, which comprises discomfort, disability, drug
toxicity, dollar loss and death (3).
Psoriasis is a common AID and the primary
pathological features include the abnormal proliferation of
epidermal cells and infiltration of inflammatory cells. Although
the exact pathogenesis of psoriasis has not been fully elucidated,
the importance of T cells has been demonstrated in previous studies
(4,5). Cytokines within psoriatic lesions,
including tumor necrosis factor (TNF)-α, interleukin (IL)-1, IL-6
and IL-8 (6–8), are subject to complex interactions
and regulation, suggesting their importance in the development of
this inflammatory disease. Therefore, understanding the
interactions involved in the cytokine network has been a major area
of focus in the investigation of the pathogenesis of psoriasis
(9). In the present study, to
further understand the association between the cytokine network and
psoriasis, human keratinocytes (HaCaT cells) were cultured in
vitro (10,11) to investigate the effects of the
N-terminal 24 amino acids (N24) and the expression of the p55
regulatory subunit of phosphatidylinositol 3-kinase (p55PIK) on the
regulation of endotoxin (LPS)-induced cytokine secretion.
Materials and methods
Cells and plasmids
The HaCaT cell line was purchased from the American
Type Culture Collection (Manassas, VA, USA) and was subcultured in
the laboratory and stored at −80°C. The cells were cultured in
Dulbecco’s modified Eagle’s medium (DMEM; Gibco Life Technologies,
Carlsbad, CA, USA) containing 15% newborn calf serum (Evergreen,
Hangzhou, China) and 100 μg/ml penicillin and streptomycin
(Evergreen) and incubated at 37°C with 5% CO2. The N24
and p55PIK adenoviruses (Evergreen) were purified and stored in the
laboratory as previously described (12).
Reagents
Trypsin was purchased from Gibco Life Technologies,
LPS (<0.008% protein from E. coli K235) was purchased
from Sigma-Aldrich (St. Louis, MO, USA) and the TNF-α and IL
enzyme-linked immunosorbent assay (ELISA) kits were purchased from
Westang Biotechnology (Shanghai, China).
Cryopreservation of the HaCaT cells and
in vitro culture
The HaCaT cells stored in cryovials (Biyuntian
Health Care Products Co., Hangzhou, China) were removed from the
−80°C freezer and rapidly thawed by submersing the cryovial in a
37°C water bath. The cryovial tubes were opened in a laminar flow
hood and the cell suspension was transferred into centrifuge tubes
and centrifuged (Beckman Coulter, Brea, CA, USA) at a low speed
(100 × g for 5 min). The supernatant was discarded and the cell
pellet was gently resuspended in DMEM containing 15% newborn calf
serum and 100 μg/ml penicillin and streptomycin and transferred
into a culture flask (Biyutian Health Care Products Co.). The cells
were cultured in a 37°C thermostatic incubator with 5%
CO2 and the medium was replaced every 2–3 days until a
confluent monolayer had formed. The cells were washed three times
with D-Hanks solution (Biyutian Health Care Products Co.) prior to
the addition of 1 ml of 0.25% trypsin and incubated for 10 min at
37°C in a 5% CO2 atmosphere. DMEM (10 ml) was added to
terminate the digestion upon detachment of the adherent cells from
the bottom of the flask. The cells were gently dissociated by
pipetting up and down into a single cell suspension and
1×104 cells were transferred into a new flask. The
medium was replaced every 2–3 days and the cells were passaged
every ~6 days. Viable cells with a healthy morphology were used for
experiments.
ELISA detection of inflammatory cytokine
secretion by HaCaT cells
The following groups were evaluated: Blank control,
LPS-stimulation, adenovirus (AD) expressing-green fluorescent
protein (GFP), AD-N24-GFP, AD-p55PIK-GFP, AD-GFP+LPS,
AD-N24-GFP+LPS and AD-p55PIK-GFP+LPS. Viable cells were seeded into
six-well plates, incubated at 37°C and transduced with AD-GFP,
AD-N24-GFP or AD-p55PIK-GFP viruses until the cells reached 70%
confluence. The transduction efficiency multiplicity of infection
value was set to 100 and the cells were further cultured for 24 h
at 37°C. The cells were stimulated using LPS (10 μl) at a final
concentration of 100 ng/ml for 4 h, and the culture supernatants
were collected and stored at −80°C until use. The assays were
performed within two weeks.
Reverse transcription quantitative
polymerase chain reaction (RT-qPCR) to assess the effects of
AD-GFP, AD-N24-GFP and AD-p55PIK-GFP on the mRNA expression levels
of TNF-α, IL-6 and IL-8 mRNA in LPS-stimulated HaCaT cells
The primers for RT-qPCR were synthesized by
Invitrogen Biotechnology Co., Ltd. (Shanghai, China), based on the
receptor mRNA template from the GenBank nucleotide sequence
database (TNF-α, NM000594.2, 8 July 2012; IL-6, NM000600.3, 11 May
2014 and IL-8 NM000584.2, 6 February 2011). The primers were
purified using a PAGE method (Invitrogen Biotechnology Co., Ltd)
and the following primer sequences were used: TNF-α, forward
5′-CGAGTGACAAGCCTGTAGCC-3′ and reverse
5′-TGAAGAGGACCTGGGAGTAGAT-3′; IL-6, forward
5′-AGCCACTCACCTCTTCAGAACG-3′ and reverse
5′-TGCCTCTTTGCTGCTTTCACA-3′; IL-8, forward
5′-CTCCAAACCTTTCCACCCC-3′ and reverse 5′-TCCACAACCCTCTGCACCC-3′ and
GAPDH, forward 5′-GATTTGGTCGTATTGGGCG-3′ and reverse
5′-CCTGGAAGATGGTGATGGG-3′. To ensure the consistency of the RNA
samples, GAPDH was used as an internal control. The total RNA from
the HaCaT cells was extracted using TRIzol Total RNA Isolation
reagent (Tiangen Biotech Co., Beijing, China) according to the
manufacturer’s instructions. RT-qPCR was performed based on the
features of the primers and the conditions for the PCR reactions
were determined from pilot experiments using a SYBR Green mix
(Tiangen Biotech Co., Beijing, China), according to the
manufacturer’s instructions. The PCR results were analyzed for the
target gene expression by the 2−ΔΔCT method. In this
formula, the cycle threshold (CT) is defined as the number of
cycles required for the fluorescent signal to cross the threshold
and exceed the background level. ΔΔCT = (experimental group CT
target gene - CT housekeeping gene) - (control group Ct target gene
- Ct housekeeping gene). Therefore, 2−ΔΔCT represents
the fold-change of the target gene in the experimental group
relative to the control group. This method directly calculates the
levels of the target gene expression relative to those of the
housekeeping gene.
Immunohistochemistry assay for nuclear
factor (NF)-κB activity
Viable cells were cultured on cover glasses and
subjected to immunohistochemical staining with a Diaminobenzidine
Histochemistry kit (Biyuntian Health Care Products Co.) to detect
NF-κB p65-positive cells. NF-κB p65-positive cells were detected
based on brown staining in the cell nuclei. To calculate the
numbers of positive cells, five regions from each cover glass
(Tiangen Biotech Co., Beijing, China) were randomly selected under
a magnification of ×200 (Leica DMIL 090-135.002; Leica Microsystems
GmbH, Wetzlar, Germany). The positive cell ratio was calculated by
comparing the number of positively stained cells with the total
number of cells. Positive cell ratio (%) = number of positive
cells/number of total cells × 100%.
Western blot analysis to detect the
protein expression levels of the Toll-like receptor
(TLR)2/TLR4/myeloid differentiation factor 88 (MyD88)/PI3K/Akt and
mitogen-activated protein kinase (MAPK) signaling pathways and of
the IκB-α NF-κB inhibitor
The expression levels of these proteins were
monitored in the HaCaT cells expressing either AD-N24-GFP or
AD-p55PIK-GFP at different time-points following LPS stimulation to
confirm the timing of the peak expression. The proteins were
extracted, according to the instructions provided by Santa Cruz
Biotechnology, Inc. (Dallas, TX, USA) and bovine serum albumin
Bio-Rad Laboratories, Inc., Hercules, CA, USA) was used as the
standard. The cells from all the groups were lysed in cold NP-40
protein lysis buffer [50 mM Tris (pH 7.4), 150 mM NaCI, 0.5% NP-40,
20% glycerol with 1 mM dithiothreitol, 1 mM phenylmethylsulfonyl
fluoride, leupeptin and pepstatin (10 g/mI each) and 100 pg/mI
aprotinin] for 20 min. Lysates were clarified by centrifugation at
10,000 × g and 4°C for 20 min. The standard curve for protein
concentration was calculated using a Bio-Rad Protein Assay kit
(Bio-Rad Laboratories, Inc.) according to the manufacturer’s
instructions. The absorbance values were detected at 595 nm on a
spectrophotometer (NanoDrop1000; Thermo Fisher Scientific,
Wilmington, DE, USA) and the target protein concentration was
calculated based on the standard curve. The total protein (20 μg)
was loaded onto a 12% polyacrylamide gel for electrophoresis and
then transferred onto a polyvinylidene difluoride membrane at 100 V
for 1 h. Primary antibodies (Santa Cruz Biotechnology, Inc.) were
diluted 1:1,000 in PBS and added to the membrane following blocking
with 5% skim milk. GAPDH was used as a loading control. The
antibodies used were as follows: rabbit polyclonal immunoglobulin G
(IgG) anti-NFκB p65 (C-20; sc-372); rabbit polyclonal IgG anti-TLR2
(H-175; sc-10739); rabbit polyclonal IgG anti-TLR4 (H-80;
sc-10741); rabbit polyclonal IgG anti-MyD88 (HFL-296; sc-11356);
rabbit polyclonal IgG anti-Akt1/2/3 (H-136; sc-8312); rabbit
polyclonal IgG anti-p-Akt1/2/3 (Ser473; sc-7985)-R; rabbit
polyclonal IgG anti-ERK 1/2 (H-72; sc-292838); goat polyclonal IgG
p-ERK 1/2 (Thr202/Tyr204; sc-16982); rabbit IgG JNK (FL; sc-571);
rabbit polyclonal IgG p-JNK (Thr183; sc-135642); rabbit polyclonal
IgG GAPDH (FL-335; sc-25778); rabbit polyclonal IgG p38 (H-147;
sc-7149) and rabbit polyclonal IgG p-p38 (Tyr182; sc-101759).
Horseradish peroxidase-conjugated secondary antibodies (diluted
1:5,000; donkey anti-goat IgG-HRP, sc-2020, and goat anti-rabbit
IgG-HRP, sc-2004; Santa Cruz Biotechnology, Inc.) and an enhanced
chemiluminescence detection kit (SuperSignal West Femto Maximum
Sensitivity Substrate Trial kit; Pierce Biotechnology, Inc., Thermo
Fisher Scientific, Rockford, IL, USA) were used to detect the
hybridization signal.
Statistical analysis
The statistical significance was assessed using SPSS
11.5 software (SPSS, Inc., Chicago, IL, USA) and the date represent
the mean ± standard deviation. Differences between the groups were
analyzed using unpaired two-tailed Student’s t-tests. P<0.05 was
considered to indicate a statistically significant difference.
Results
Effects of AD-N24-GFP and AD-p55PIK-GFP
on LPS-induced inflammatory cytokine release by HaCaT cells
ELISA was used to measure changes in the release of
the inflammatory cytokines, IL-6 and IL-8, upon LPS-stimulation
following AD-N24-GFP or AD-p55PIK-GFP transfection in the HaCaT
cells. IL-6 and IL-8 were detected in quiescent cells (6.44±0.95
and 50.97±13.14 pg/ml, respectively), however, the levels of IL-6
and IL-8 in the culture supernatant increased to different extents
following LPS-stimulation for 4 h (88.32±7.43 and 314.57±30.82
pg/ml, respectively). A more marked increase was observed in the
expression of IL-8 compared with IL-6, although statistically
significant differences were observed for each protein (P<0.05).
Upon transfection with AD-N24-GFP, the LPS-induced release of IL-6
and IL-8 by the HaCaT cells decreased to 51.25±4.79 and
174.61±26.02 pg/ml, respectively. These differences were
statistically significant (P<0.05). Upon transfection with
AD-p55PIK-GFP, the expression levels of IL-6 and IL-8 increased to
120.99±13.74 and 450.28±32.2 pg/ml, respectively. These differences
were also statistically significant (P<0.05). The data are shown
in Fig. 1.
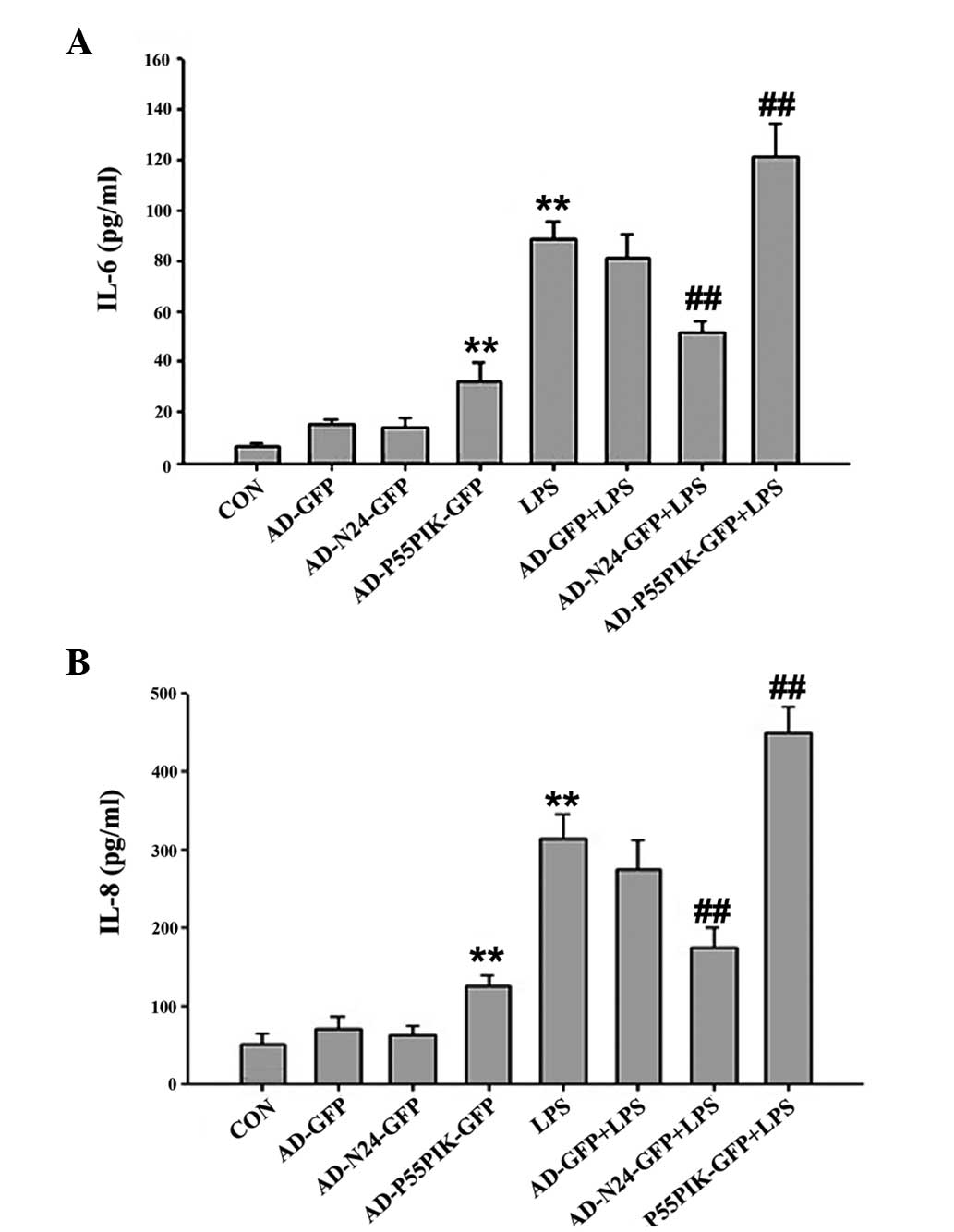 | Figure 1Enzyme-linked immunosorbent assay
results indicating the effect of AD-N24-GFP and AD-p55PIK-GFP on
the LPS-stimulated release of the (A) IL-6 and (B) IL-8
inflammatory factors (**P<0.01, compared with the
control group; ##P<0.01, compared with the LPS
group). LPS, endotoxin; IL, interleukin, GFP, green fluorescent
protein, CON, control, N24, N-terminal 24 amino acids; AD,
adenovirus. Values are expressed as the mean ± standard error of
the mean of three independent experiments. |
RT-qPCR to determine the LPS-induced mRNA
expression of TNF-α, IL-6 and IL-8 by HaCaT cells prior to and
following AD-N24-GFP or AD-p55PIK-GFP transfection
Compared with the unstimulated group,
LPS-stimulation led to an almost five-fold increase in the mRNA
expression levels of TNF-α, IL-6 and IL-8 following transfection
with AD-N24-GFP. This was significantly lower compared with that of
the untransfected group in response to LPS-stimulation. The mRNA
expression levels of the inflammatory cytokines TNF-α, IL-6 and
IL-8 induced by LPS-stimulation were significantly increased upon
transfection with AD-p55PIK-GFP, indicating that an increase in the
intracellular expression of p55PIK promoted inflammatory cytokine
release. The results are shown in Fig.
2.
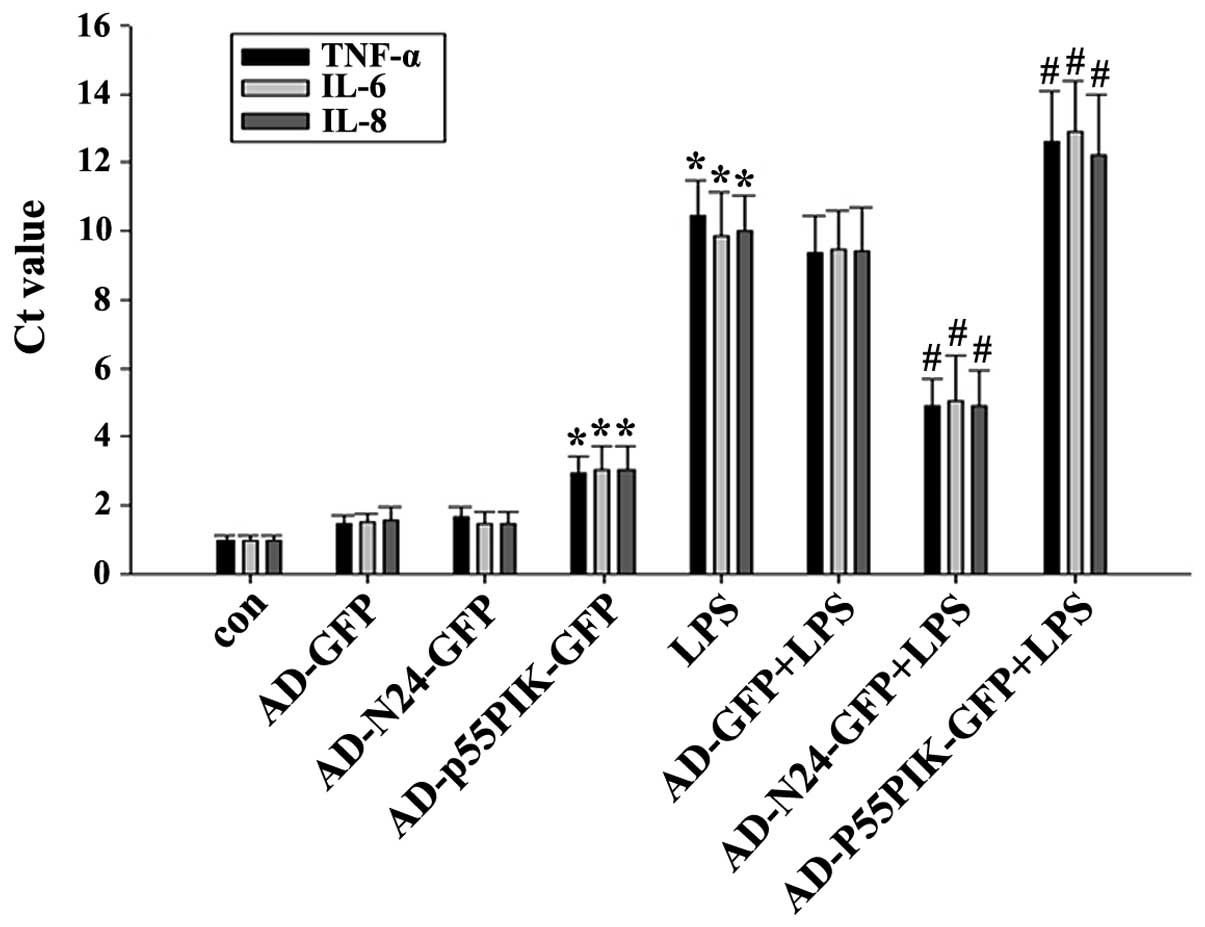 | Figure 2Reverse transcription quantitative
polymerase chain reaction detection of the mRNA expression levels
of TNF-α, IL-6 and IL-8 in the HaCaT cells (*P<0.05,
compared with the control group; #P<0.05, compared
with the LPS group). Ct, cycle threshold; con, control; GFP, green
fluorescent protein; LPS, endotoxin; IL, interleukin, N24,
N-terminal 24 amino acids; P55PIK, p55 regulatory subunit of
phosphatidylinositol 3-kinase; TNF, tumor necrosis factor; HaCaT,
human keratinocytes; AD, adenovirus. Values are expressed as the
mean ± standard error of the mean of three independent
experiments. |
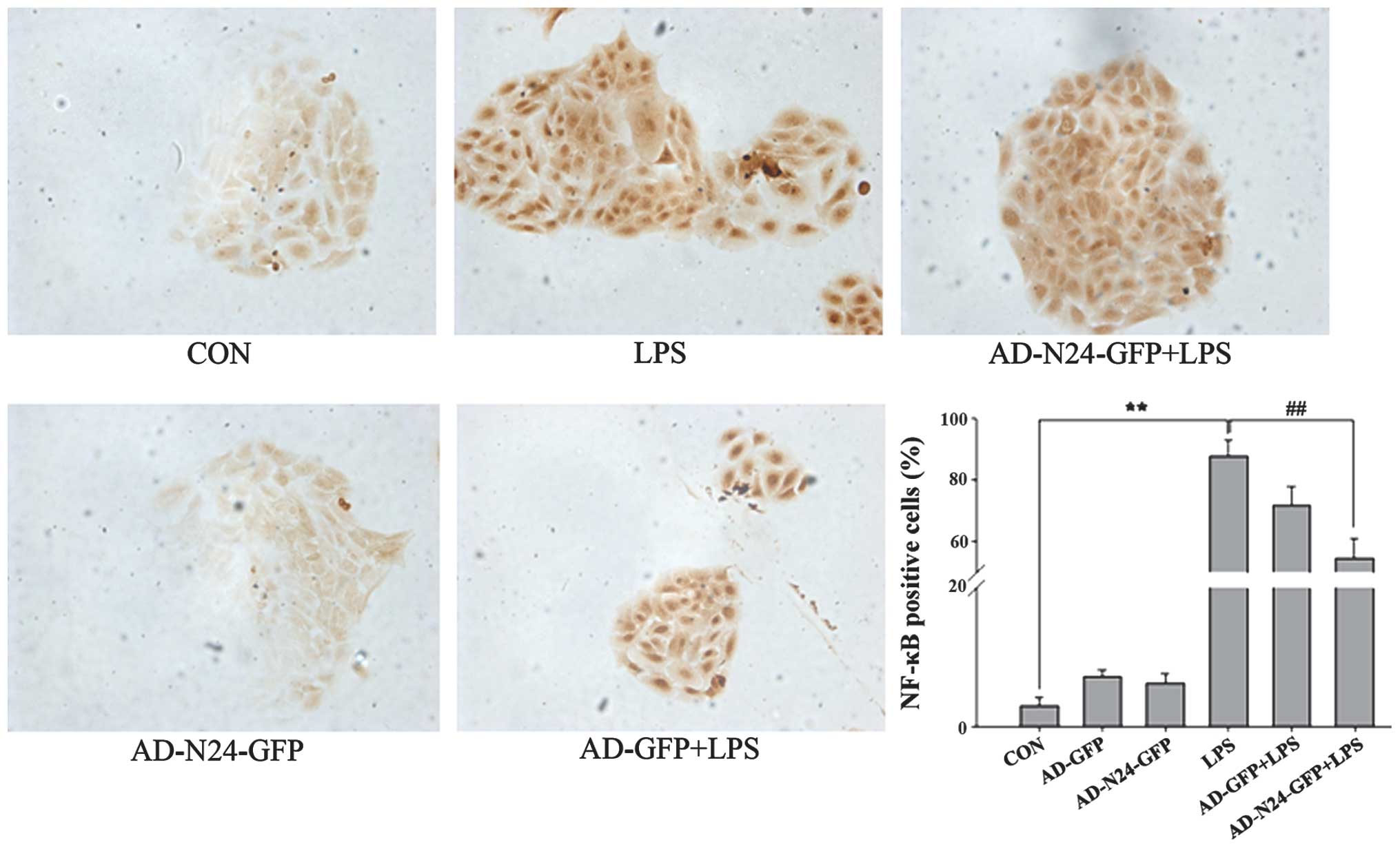
Immunocytochemistry to detect the
expression of NF-κB
HaCaT cells were cultured on cover glass, fixed and
immunocytochemistry was performed. The numbers of LPS-induced NF-κB
p65-positive HaCaT cells prior to and following transfection with
AD-N24-GFP were counted under a microscope. The quiescent HaCaT
cells contained few NF-κB p65-positive cells (3.04±1.13%) compared
with the LPS stimulated group, in which LPS almost fully activated
NF-κB p65 (87.57± 5.38%). Following transfection with AD-N24-GFP,
the number of NF-κB p65-positive HaCaT cells decreased to
54.43±6.54%, which was statistically significant compared with the
LPS group (P<0.01). The results are shown in Fig. 3.
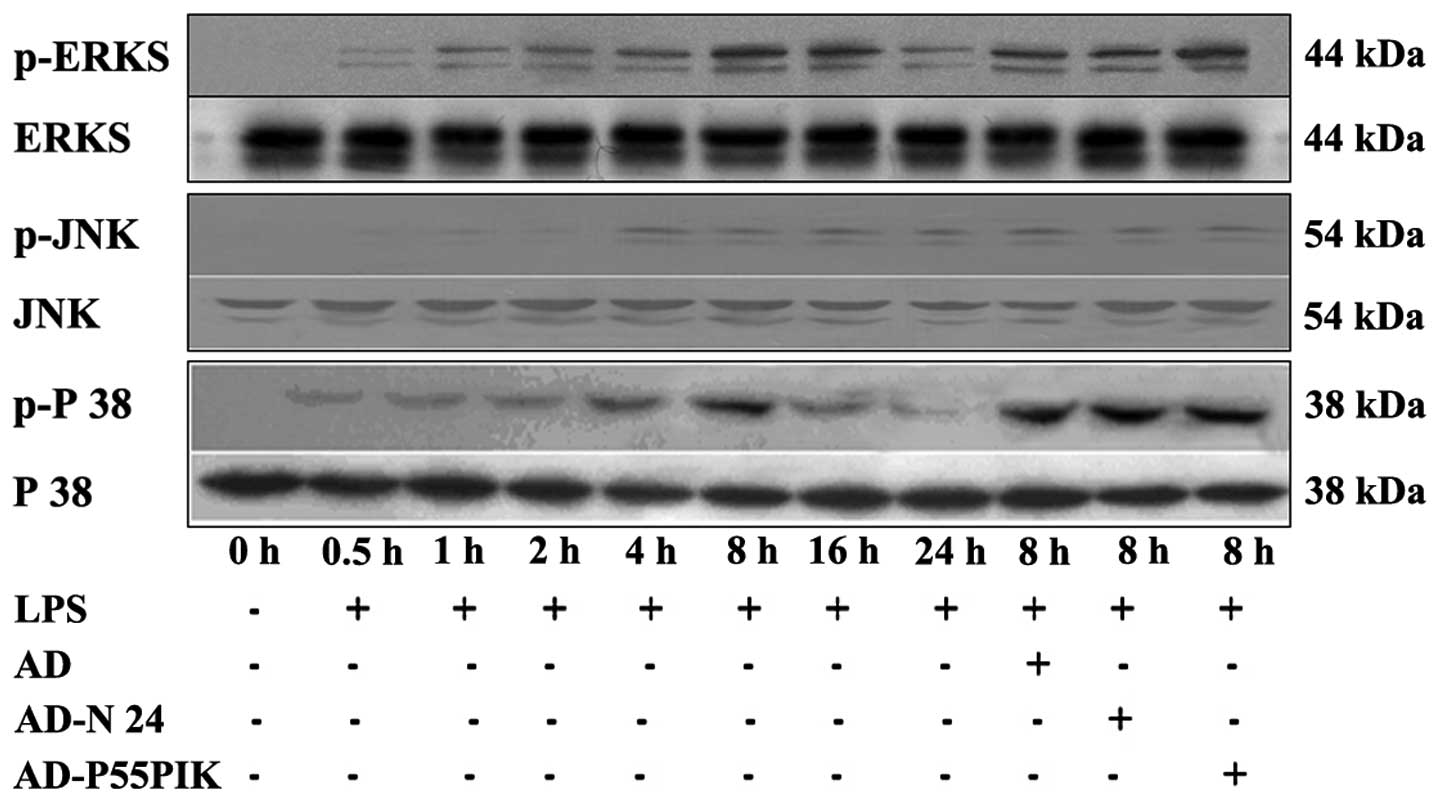
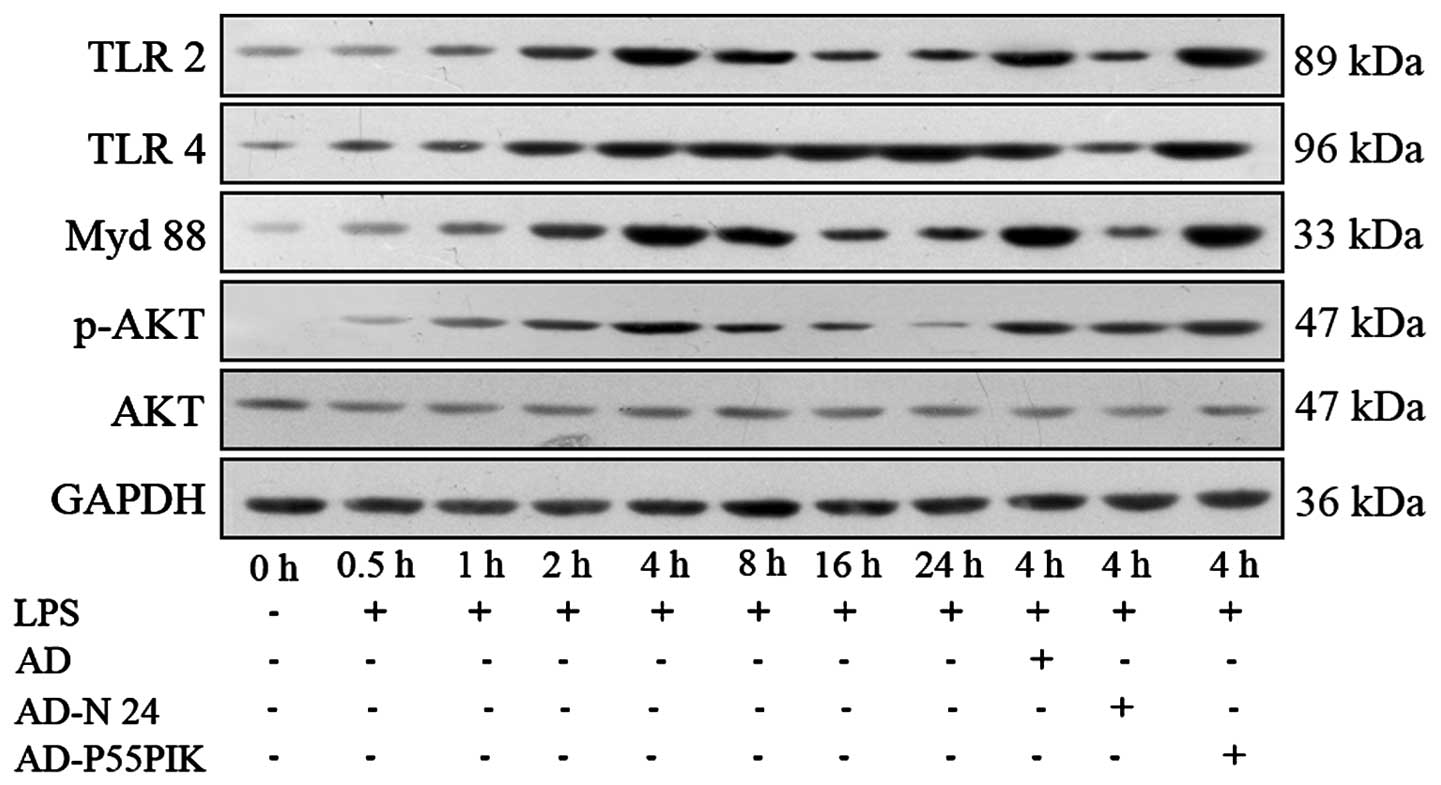
Protein expression analyses of the TLR
signaling pathways in the LPS-simulated HaCaT cells expressing
AD-N24-GFP or AD-p55PIK-GFP
The expression levels of the
TLR2/TLR4/MyD88/PI3K/Akt signaling pathway proteins were
investigated. As shown in Fig. 5,
TLR2 and TLR4 were expressed in the HaCaT cells. LPS-stimulation
increased the expression levels of TLR2 and TLR4 in the HaCaT
cells, with the peak increase observed 4 h and 8 h
post-stimulation. These levels remained higher than normal for at
least 24 h. The expression of MyD88 gradually increased following
LPS-stimulation, peaked at 4 h and then gradually decreased,
however remained higher compared with the normal level at 24 h. The
phosphorylation of Akt gradually increased (P<0.05) 30 min after
LPS-stimulation, peaked at 4 h (P<0.05) and then decreased
rapidly and returned to normal levels by 24 h. Transfection with
AD-GFP had no affect on the expression of the proteins, whereas
transfection with AD-N24-GFP significantly downregulated the
protein expression levels of TLR2 and TLR4 following LPS
stimulation compared with the LPS-stimulated group. The protein
expression of MyD88 also decreased and no change in the expression
of phosphorylated Akt was observed, suggesting that the AD-N24-GFP
protein downregulated the expression levels of TLR2/TLR4/MyD88 in
the HaCaT cells with no significant effect on the PI3K/Akt
signaling pathway. Transfection with AD-p55PIK-GFP increased the
protein expression levels of TLR2, TLR4 (to an extent), MyD88 and
phosphorylated Akt following LPS stimulation (Fig. 4). These results suggested that the
AD-p55PIK-GFP affected the protein expression levels of
TLR2/TLR4/MyD88 by upregulating phosphorylated Akt in the HaCaT
cells.
Expression of MAPK signaling pathway
proteins
The MAPK signaling pathway is important in the
LPS-mediated cellular response. In mammalian cells, at least four
MAPK subfamilies have been identified, namely the
extracellular-signal-regulated kinases (ERK)1/2, c-Jun N-terminal
kinases (JNK), p38 and NIMA-associated protein kinase 5. As shown
in Fig. 5, the present study
demonstrated that the phosphorylation of ERK increased 30 min
post-LPS stimulation (P<0.05), peaked at 8 h (P<0.05) and
then gradually declined, however, it remained significantly higher
compared with the baseline until 24 h (P<0.05). The
phosphorylation patterns of JNK and p38 were similar, however, the
phosphorylation of p38 decreased rapidly subsequent to peaking at 8
h (P<0.05) and almost returned to basal levels at 24 h. The
expression of phosphorylated JNK decreased subsequent to peaking at
4 h (P<0.05) and remained marginally higher compared with basal
levels at 24 h (Fig. 5).
Transfection with either AD-GFP, AD-N24-GFP or AD-p55PIK-GFP caused
no affect on the expression levels of phosphorylated ERK1/2, JNK or
p38, suggesting that AD-N24-GFP and AD-p55PIK-GFP had no
significant affect on the MAPK signaling pathway.
Discussion
The skin is the body’s first line of defense against
outside pathogens and is important in innate immunity. The
differentiation features of HaCaT cells and normal human
keratinocytes are similar (13)
and have been widely used to investigate the activity and
functionality of anti-inflammatory drugs (14). TLRs are primarily expressed in
immune cells, including dendritic cells, lymphocytes and
monocytes/macrophages. In addition, TLRs are expressed in
epithelial cells and other non-immune cells (15). TLR signaling consists of at least
two distinct pathways: The MyD88-dependent and the
MyD88-independent pathways. MyD88 is essential in intracellular
signal transduction through TLRs. The C-terminus of MyD88 contains
a Toll/interleukin-1 receptor (TIR) domains, which binds to the TIR
domain of TLR, IL-1R and IL-18R. The N-terminus contains a death
domain (DD), which is responsible for recruiting signaling
molecules with DDs, including TNF receptor-associated factor 6 and
TAK1 binding protein 1 and 2. This recruitment induces the
activation of NF-κB-inducing kinase and the phosphorylation of the
NF-kB inhibitor, IκB kinase (IKK1-IKK2-IKKK2). The activation of
IKK promotes the ubiquitination and degradation of IκB-α, resulting
in the activation of NF-κB, ERK, p38, JNK and other MAPKs, which
then activate activator protein 1. Activation of this pathway
induces the expression levels of IL-1, IL-6, IL-8, IL-12, TNF-α and
other inflammatory cytokines (16).
Phosphoinositide 3-kinases (PI3Ks) are pivotal in
inflammatory processes. In vivo and in vitro studies
have demonstrated that leukocytes and macrophages lacking PI3Kγ
exhibit defective chemotaxis towards various stimuli (17,18).
The inhibitory capacity of the selective inhibitor, AS041164, on
peritoneal leukocyte chemotaxis towards RANTES is three times
higher compared with that of LY294002 (19). LY294002 is a general PI3K
inhibitor, whereas AS-041164 is a potent inhibitor of PI3K with
selectivity for the class IB isoform PI3Kγ. When administered
orally to mice, AS-041164 demonstrates a pharmacokinetic profile
that is similar to the general PI3K inhibitor LY294002 but is three
times more potent at blocking neutrophil recruitment induced by
RANTES. In addition, endothelial PI3K facilitates the adhesion of
leukocytes to the inflammatory vessel wall (20). The expression of PI3Kδ in
endothelial cells promotes the adhesion of reactive leukocytes and
the transmembrane migration of endothelial cells induced by TNF-α
and leukotriene B4 (21). These
observations demonstrate that PI3Kδ and PI3Kγ are required for the
effective recruitment of white blood cells by endothelial cells in
response to cytokine stimuli.
The present study established an in vitro
model to examine the effects of N24 and p55PIK on the expression
levels of TLR signaling pathway proteins in LPS-stimulated HaCaT
cells. The NF-κB p65 protein translocation into the nucleus was
altered upon LPS stimulation and N24 significantly suppressed this
translocation. Furthermore, the ubiquitination and subsequent
degradation of NF-κB p65 was observed in LPS-stimulated HaCaT
cells. N24, but not p55PIK, significantly inhibited this
ubiquitination and degradation, suggesting that N24 can effectively
inhibit NF-κB p65 translocation into the nucleus and prevent the
release of pro-inflammatory cytokines. The expression levels of
TLR2, TLR4 and MyD88 were demonstrated to increase with prolonged
LPS stimulation in the HaCaT cells. The expression of N24 caused a
reduction in the expression levels of TLR2, TLR4 and Myd88 in
response to LPS stimulation. By contrast, p55PIK promoted the
expression levels of TLR2, TLR4 and MyD88, suggesting that
activation of PI3K increased the immune response to LPS, while N24
functioned as a PI3K inhibitor. The effects of N24 and p55PIK on
the MAPK pathway, another TLR-mediated inflammatory signaling
pathway, were also investigated. N24 and p55PIK caused no
significant change to the phosphorylation of ERK1/2, JNK or p38,
suggesting that N24 and p55PIK affect TLRs/MyD88-mediated signaling
in an MAPK-independent manner.
In conclusion, the present study demonstrated the
effect of N24 and p55PIK on TLR signaling and the suitability of
PI3K as a potential novel target for immunosuppressant therapy and
N24 as a novel candidate immunosuppressant. The underlying
mechanism of N24 was also examined. These findings provide a
theoretical basis for the identifcation of immunosuppressive drugs
and therapeutic strategies against AID.
Acknowledgements
This study was supported by a grant from the
National Science Foundation of China (no. 81072431).
References
|
1
|
Cho JH and Gregersen PK: Genomics and the
multifactorial nature of human autoimmune disease. N Engl J Med.
365:1612–1623. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bogdanos DP, Smyk DS, Rigopoulou EI, et
al: Twin studies in autoimmune disease: genetics, gender and
environment. J Autoimmun. 38:J156–J169. 2012. View Article : Google Scholar
|
|
3
|
Bruce B1 and Fries JF: The Stanford Health
Assessment Questionnaire: a review of its history, issues,
progress, and documentation. J Rheumatol. 30:167–178.
2003.PubMed/NCBI
|
|
4
|
Bos JD and De Rie MA: The pathogenesis of
psoriasis: Immunological facts and speculations. Immunol Today.
20:40–46. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lowes MA, Kikuchi T, Fuentes-Duculan J, et
al: Psoriasis vulgaris lesions contain discrete populations of Th1
and Th17 T cells. J Invest Dermatol. 128:1207–1211. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cooper KD, Hammerberg C, Baadsgaard O,
Elder JT, Chan LS, Sauder DN, Voorhees JJ and Fisher G: IL-1
activity is reduced in psoriatic skin. Decreased IL-1 alpha and
increased nonfunctional IL-1 beta. J Immunol. 144:4593–4603.
1990.PubMed/NCBI
|
|
7
|
Gomi T, Shiohara T, Munakata T, Imanishi K
and Nagashima M: Interleukin 1 alpha, tumor necrosis factor alpha
and interferon gamma in psoriasis. Arch Dermatol. 127:827–830.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Neuner P, Urbanski A, Trautinger F, Moller
A, Kirnbauer R, Kapp A, Schopf E, Schwarz T and Luger TA: Increased
IL-6 production by monocytes and keratinocytes in patients with
psoriasis. J Invest Dermatol. 97:27–33. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Luger TA and Schwarz T: Evidence for an
epidermal cytokine network. J Invest Dermatol. 95:100S–104S. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gilhar A, Pillar T and Etzioni A: Possible
role of cytokines in cellular proliferation of the skin
transplanted onto nude mice. Arch Dermatol. 131:38–42. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Stein M, Bernd A, Ramirez-Bosca A,
Kippenberger S and Holzmann H: Measurement of anti-inflammatory
effects of glucocorticoids on human keratinocytes in vitro.
Comparison of normal human keratinocytes with the keratinocyte cell
line HaCaT. Arzneimittelforschung. 47:1266–1270. 1997.
|
|
12
|
Hu J, Xia X, Cheng A, Wang G, Luo X, Reed
MF, Fojo T, Oetting A, Gong J and Yen PM: A peptide inhibitor
derived from p55PIK phosphatidylinositol 3-kinase regulatory
subunit: a novel cancer therapy. Mol Cancer Ther. 7:3719–3728.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mutis T, De Bueger M, Bakker A and
Ottenhoff TH: HLA class II+ human keratinocytes present
Mycobacterium leprae antigens to CD4+ Th1-like cells. Scand J
Immunol. 37:43–51. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Barker JN, Mitra RS, Griffiths CE, Dixit
VM and Nickoloff BJ: Keratinocytes as initiators of inflammation.
Lancet. 337:211–214. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Akira S: Toll-like receptors and innate
immunity. Adv Immunol. 78:1–56. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Su SB, Silver PB, Grajewski RS, Agarwal
RK, Tang J, Chan CC and Caspi RR: Essential role of the MyD88
pathway, but nonessential roles of TLRs 2, 4, and 9, in the
adjuvant effect promoting Th1-mediated autoimmunity. J Immunol.
175:6303–6310. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Okkenhaug K, Bilancio A, Farjot G, Priddle
H, Sancho S, Peskett E, Pearce W, Meek SE, Salpekar A, Waterfield
MD, et al: Impaired B and T cell antigen receptor signaling in
p110delta PI 3-kinase mutant mice. Science. 297:1031–1034.
2002.PubMed/NCBI
|
|
18
|
Iwasaki A and Medzhitov R: Toll-like
receptor control of the adaptive immune responses. Nat Immunol.
5:987–995. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li Z, Jiang H, Xie W, Zhang Z, Smrcka AV
and Wu D: Roles of PLC-beta2 and -beta3 and PI3Kgamma in
chemoattractant-mediated signal transduction. Science.
287:1046–1049. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Puri KD, Doggett TA, Huang CY, Douangpanya
J, Hayflick JS, Turner M, Penninger J and Diacovo TG: The role of
endothelial PI3Kgamma activity in neutrophil trafficking. Blood.
106:150–157. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Puri KD, Doggett TA, Douangpanya J, Hou Y,
Tino WT, Wilson T, Graf T, Clayton E, Turner M, Hayflick JS, et al:
Mechanisms and implications of phosphoinositide 3-kinase delta in
promoting neutrophil trafficking into inflamed tissue. Blood.
103:3448–3456. 2004. View Article : Google Scholar : PubMed/NCBI
|