Introduction
Paraquat dichloride (1,1′-dimethyl-4,4′-bipyridinium
dichloride; methyl viologen; PQ) is an effective and widely used
herbicide that can cause pulmonary fibrosis (1,2).
Previous studies have demonstrated that the lungs are one of the
primary target organs for PQ-induced toxicity in humans and animals
(1,3). The acute toxic effects of PQ,
including pulmonary edema and hypoxia, lead to irreversible
pulmonary fibrosis. Notably, 1,000s of mortalities due to
intentional or accidental ingestion of PQ have been reported
(4,5). Although certain drugs, including
glucocorticoids, antioxidants and cytotoxic drugs, can mitigate
toxicity (6–9), the treatment of acute PQ poisoning is
often poor and mortality rates remain high (10,11).
Since an effective PQ antidote has yet to be identified, studies on
the molecular mechanisms of PQ-induced pulmonary fibrosis are
critical for improving treatment and reducing mortality.
Connective tissue growth factor (CTGF) is a
cysteine-rich extracellular matrix-associated heparin-binding
protein that belongs to the CCN family (12,13).
CTGF is widely expressed in numerous tissues at low levels,
however, is markedly upregulated in fibrotic and cancerous tissue
(14). CTGF regulates various
biological processes associated with fibro-genesis, including
cellular adhesion, proliferation, migration, differentiation,
extracellular matrix (ECM) production (15–17)
and angiogenesis (18). In
particular, CTGF has been found to promote deposition of several
ECM proteins, including collagen, fibronectin and tenascin C
(19,20). Aberrant ECM production by lung
fibroblasts has been associated with fibrosis in several models of
pulmonary injury (21,22). ECM production could further be
stimulated as a consequence of lung fibroblast proliferation,
migration and differentiation into myofibroblasts (23). Accordingly, CTGF has been
demonstrated to affect various cell types involved in the
fibrogenic process, including type II alveolar epithelial cells,
endothelial cells, mesenchymal stem cells and lung fibroblasts
(16).
Although CTGF has been demonstrated to be important
in pulmonary fibrosis induced by bleomycin and hyperoxia (24,25),
an association between this growth factor and PQ-induced lung
injury has not been established. In particular, the effect of CTGF
on ECM production, cellular proliferation, migration and
myofibroblast differentiation in association with PQ poisoning
remains to be elucidated. The aim of the present study was to
investigate the effects of PQ on CTGF expression and subsequent
pulmonary fibrosis.
Materials and methods
Ethics statement
All animals were handled in accordance with
guidelines approved by the Experimentation Ethics Review Committee
of Nanjing University (Nanjing, China). The rats were fed with
commercial rat chow, provided with water ad libitum and kept
on a 12:12 h light-dark cycle.
Cell culture
MRC-5 lung fibroblasts (human lung fibroblasts;
American Type Culture Collection, Manassas, VA, USA; cat. no. CCL
171) were cultured in high Dulbecco’s modified Eagle’s medium
(DMEM; HyClone Laboratories, Inc., Logan, UT, USA) with 10% fetal
bovine serum (FBS; Invitrogen Life Technologies, Carlsbad, CA, USA)
supplemented with 1% L-glutamine and 1% penicillin/streptomycin
solution. Cells were incubated at 37°C in 5% CO2 and
routinely passaged upon reaching 80% confluency, using 0.25%
trypsin and a 1:3 cell dilution for each passage.
Cell viability
The viability of lung fibroblasts was evaluated
using a Cell Counting kit-8 (CCK-8; Dojindo Laboratories, Kumamoto,
Japan) assay. Cells were plated in 6-well plates at a density of
2×106 cells/ml for 12 h and treated with various
concentrations of CTGF (50–200 ng/ml; PeproTech, Inc., Rocky Hill,
NJ, USA) for 24, 48 or 72 h. The cells were then transferred into a
96-well culture plate (n=8) at a density of 2×104
cells/100 μl/well for 12 h. The culture medium was removed
and 100 μl of serum-free medium containing 10 μl of
CCK-8 solution was added to each well. After a 4 h incubation
period, the absorbance was measured at an optical density (OD) of
450 nm using a multi-detection microplate reader (VersaMax;
Molecular Devices, Sunnyvale, CA, USA).
Cell migration
A double-chamber system was used to perform a
transwell migration assay to determine the migration ability of
lung fibroblasts. Cells were seeded in a 6-well plate with
serum-free medium at a density of 4×104 cells/per upper
chamber. Culture medium (1 ml) with 10% FBS containing various
concentrations of CTGF (50–200 ng/ml) was simultaneously added to
the lower chamber. The upper and lower chambers were separated by a
permeable polycarbonate membrane with a pore size of 8 μm.
The cells were permitted to migrate to the lower chamber for 24 h.
Following this incubation period, cells that had entered the lower
surface of the filter membrane were fixed with a cold solution of
4% paraformaldehyde for 30 min. Cells were then washed three times
with phosphate-buffered saline (PBS) and stained with 0.1% crystal
violet for 30 min. Cells that remained in the upper surface were
gently scraped off with a cotton swab. A total of 10 different
fields of view from each membrane were randomly selected and
captured using a photomicroscope (BX51; Olympus, Tokyo, Japan).
Cell migration was quantified by counting the number of migrated
cells. Experiments were performed three times in duplicate.
Animal groups and experimental
protocol
A total of 48 adult male Sprague-Dawley (SD) rats
weighing 200–250 g were purchased from the Animal Center of Nanjing
University (Nanjing, China). The SD rats were randomly divided into
two groups (n=24 for each group). The experimental group received a
single intraperitoneal injection of PQ (30 mg/kg), while the
control group received an equivalent volume of sterile saline. The
rats were then sacrificed by cervical dislocation after 7, 14 and
28 days.
Histological analysis
Following sacrificing the animals, the lungs were
removed and the left lung was fixed with 4% paraformaldehyde for 16
h. The tissue samples were then processed using graded alcohol,
xylene and paraffin and blocked in paraffin. The paraffin-embedded
sections (5 μm thick) were stained using a hematoxylin and
eosin (H&E) kit (Biyuntian, Inc., Nantong, China) and a
Masson’s trichrome staining kit (Nanjing Jiancheng Bioengineering
Institute, Nanjing, China), according to the manufacturer’s
instructions. The slides with H&E staining were examined using
light microscopy (Eclipse TE2000-S; Nikon Corporation, Tokyo,
Japan) and images were captured to determine the integrity of the
tissue. The severity of pulmonary fibrosis in the lung sections
stained for collagen with Masson’s trichrome stain was determined
by a histopathologist who was blinded to the protocol design.
Immunohistochemical staining
Formalin-fixed, paraffin-embedded skin sections were
stained with rabbit polyclonal anti-CTGF antibodies (cat. no.
ab6992; Abcam, Inc., Cambridge, MA, USA). Following transferal
through a graded series of alcohol and xylene, lung tissue samples
were embedded in paraffin and sectioned (5 μm). The sample
sections were mounted onto poly-L-lysine-coated slides and
processed for immunohistochemical analysis, according to a similar
procedure as previously reported (26). Briefly, sections were incubated
overnight at room temperature with 3% bovine serum albumin (BSA)
for 30 min at 37°C for blockade of non-specific binding sites and
then incubated overnight at 4°C with the primary antibody targeting
CTGF (1:200 dilution). Specificity of the antibody was examined
using normal rabbit serum instead of the primary antibody. The
slides were then incubated with a secondary biotinylated goat
anti-rabbit antibody (1:200 dilution; cat. no. BA1003; Wuhan Boster
Biological Technology, Ltd., Wuhan, China) for 30 min at 37°C.
Following rinsing, the slides were incubated with horseradish
peroxidase-conjugated streptavidin and then washed with deionized
water. The samples were then exposed to 3,3′-diaminobenzidine
substrate solution for 10 min, coun-terstained with hematoxylin and
mounted with coverslips. Images were captured on a Nikon Eclipse
TE2000-S microscope (Nikon Corporation). A brown reaction product
was considered a positive result.
Immunofluorescence
To evaluate the effect of CTGF on myofibroblast
differentiation of human lung fibroblasts, MRC-5 lung fibroblasts
were treated with 100 ng/ml CTGF (PeproTech, Inc.) for 3 days.
Immunofluorescence staining was performed as previously described
(21). Briefly, MRC-5 cells were
fixed with 4% paraformaldehyde and permeabilized with 0.1% Triton
X-100 for 10 min. The cells were then incubated with PBS containing
2% BSA for 1 h at 37°C to block unspecific binding sites. The fixed
cells were then incubated with the following primary antibodies:
Rabbit polyclonal anti-α-smooth muscle actin (α-SMA; 1:200
dilution; cat. no. ab5694) and rabbit polyclonal anti-collagen I
(1:200 dilution; cat. no. ab34710; Abcam, Inc., Cambridge, MA, USA)
at 4°C for 16 h. Following three washes with PBS, cells were
incubated with a secondary antibody (1:400 dilution; goat
anti-rabbit Alexa Fluor 594 or 488; Invitrogen Life Science,
Gaithersburg, MD, USA) in 2% BSA for 1 h at 37°C in the dark.
Nuclear staining was performed using 5 mg/ml
4′,6-diamidino-2-phenylindole (Biyuntian, Inc.). Cells were
visualized using a confocal fluorescence microscope (Fluoview
FV10i; Olympus).
Western blot analysis
In order to investigate the effects of PQ on protein
levels of CTGF, MRC-5 cells were treated with various
concentrations of PQ (50–500 μM; Sigma-Aldrich, St. Louis,
MO, USA) for 72 h. Protein samples were obtained from cultured
cells and animals (right lung, n=8) treated with PQ. Western blot
analysis of protein lysates was performed as previously described
(21). Briefly, cells and lung
tissues were lysed in ice-cold RIPA extraction buffer (150 mM NaCl,
10 mM Tris-HCl, pH 7.4, 1% Triton X-100, 1% sodium deoxycholate and
0.1% SDS) containing a protease inhibitor cocktail (Roche
Diagnostics, Indianapolis, IN, USA) for 30 min. The whole lysates
were then centrifuged at 12,000 × g for 30 min and the protein
concentration in the supernatant was determined using a
bicinchoninic acid assay (Wuhan Boster Biological Technology,
Ltd.). The protein samples were boiled for 10 min and 20 μl
aliquots were then subjected to 12% SDS-polyacrylamide gel
electrophoresis. The protein bands were electrophoretically
transferred onto a polyvinylidene fluoride membrane, which was
incubated in blocking buffer (1X PBS, 0.1% Tween-20, 1% BSA and 5%
non-fat milk) for 1 h at 37°C. The membrane was then exposed to
primary antibodies (at a 1:3,000 dilution) against rabbit α-SMA,
rabbit collagen I and mouse β-actin (Abcam, Inc.) in blocking
buffer overnight at 4°C. Following three washes in PBS containing
0.05% Tween 20, the membrane was incubated with the secondary
antibody (horseradish peroxidase-conjugated goat anti-rabbit/mouse
IgG; Wuhan Boster Biological Technology, Ltd.) at 37°C for 1 h.
Visualization of immunoreactive protein bands was performed with an
enhanced chemiluminescence detection kit (Amersham Biosciences,
Piscataway, NJ, USA) using an Odyssey Scanning System (LI-COR,
Inc., Lincoln, NE, USA).
Hydroxyproline (HP) content
The quantity of collagen in the lung tissue was
determined by analysis of HP content according to the
manufacturer’s instructions of the detection kit (Nanjing Jiancheng
Bioengineering Institute). The absorbance was measured at 550 nm
and the HP content was determined using a standard curve (0–100
mg/ml).
Statistical analysis
Data are expressed as the mean ± standard deviation.
Differences among groups were evaluated by one-way analysis of
variance using Statistical Package for the Social Sciences (SPSS)
version 18.0 software (SPSS, Inc., Chicago IL, USA). P<0.05 was
considered to indicate a statistically significant difference.
Results
PQ-induced lung injury and pulmonary
fibrosis
After PQ administration for 7, 14 and 28 days, rats
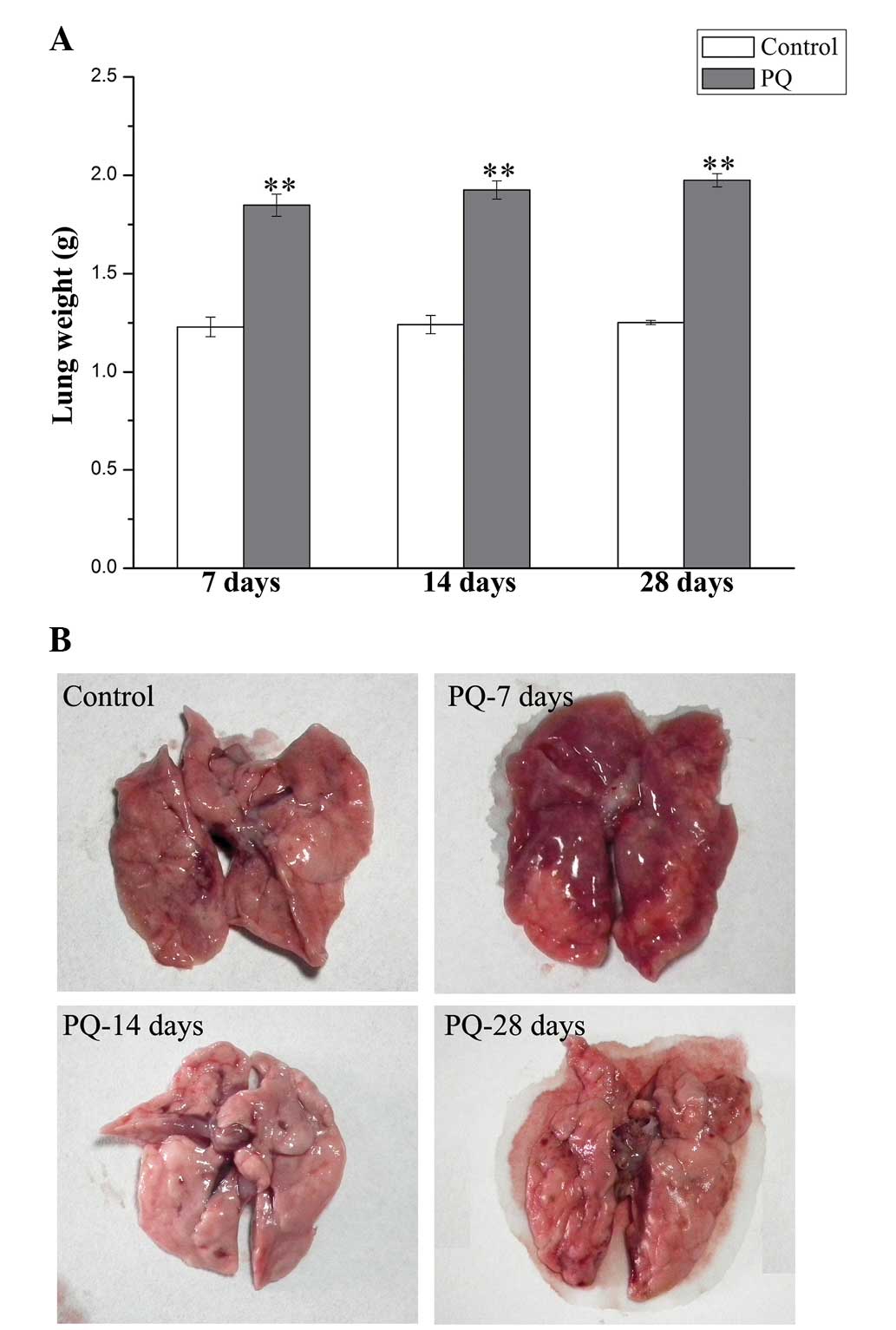
were sacrificed and the lungs were collected. As shown in Fig. 1A, the weight of the lungs increased
in response to PQ administration, indicating the presence of
pulmonary inflammation and fibrosis. In addition, the macroscopic
appearance of the lungs was fibrotic (Fig. 1B). These results suggest that PQ is
able to induce lung injury, inflammation and fibrosis in this rat
model.
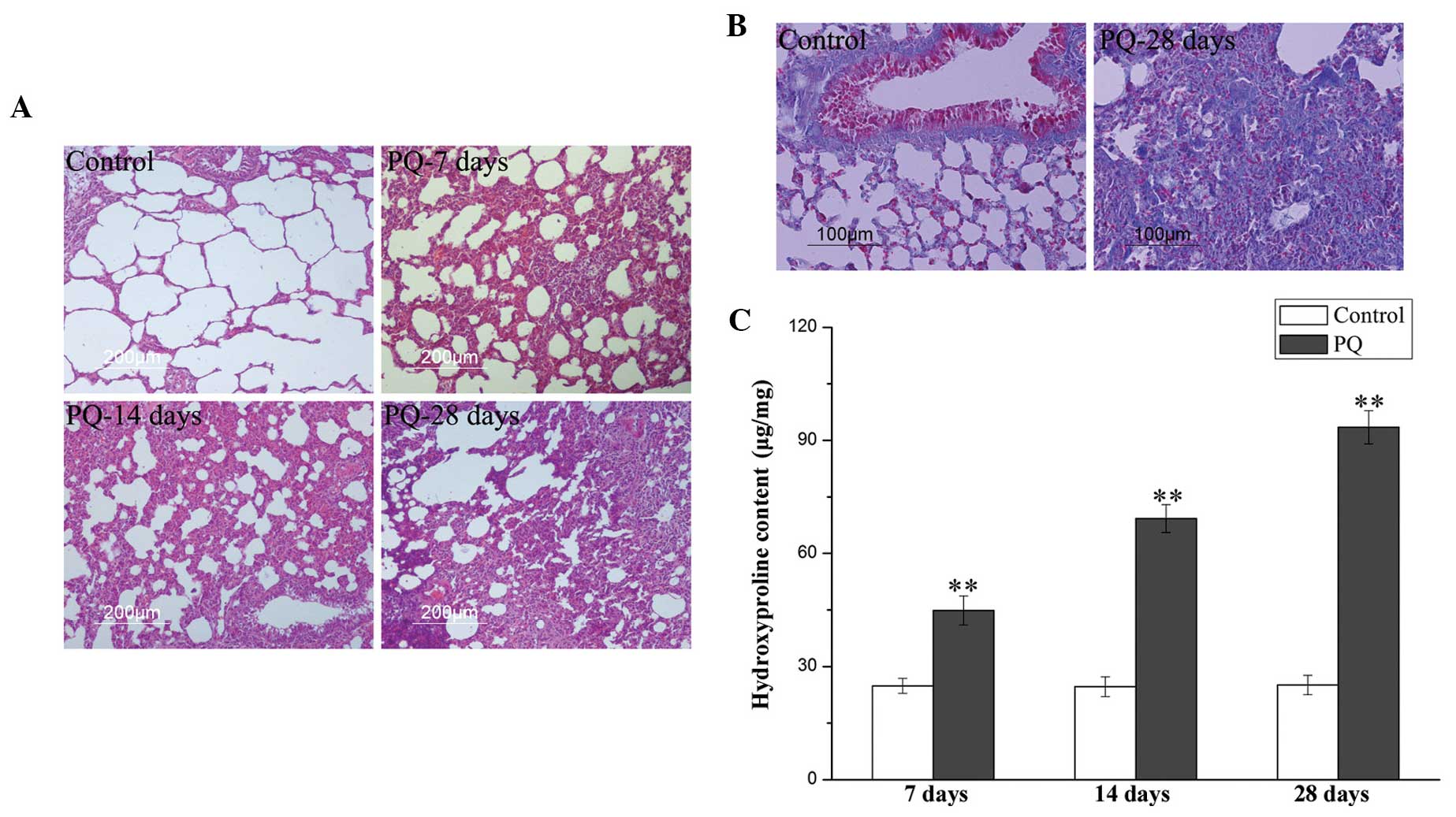
Histopathological analyses of rat lungs
treated with PQ
To investigate the effects of PQ on lung tissue,
serial lung sections from PQ-treated rats (7, 14 and 28 days after
PQ administration) were stained with H&E or Masson’s trichrome
and examined using a light microscope. Representative histological
sections from lungs of rats in each experimental group are shown in
Fig. 2A. Lung tissues in the
control group appeared histologically normal, showing no signs of
inflammation or epithelial damage. By contrast, histopathological
examination of the lung tissue following PQ administration revealed
alterations in the lung structure. After 7 days, the lungs
exhibited marked alterations in tissue structure, exhibiting signs
of acute injury with interstitial edema and widespread inflammatory
cell infiltration in the alveolar space and septum. In addition, an
infiltration of mononuclear inflammatory cells, fibroblast
proliferation, extensive cellular thickening and fibrosis was
evident after 14 days. Similarly, at day 28, H&E staining
indicated thickened alveolar walls and pulmonary interstitial
fibrosis. Masson’s trichrome staining revealed that collagen
deposition (blue staining) in the PQ group was increased compared
with the control group (Fig. 2B).
In conclusion, histological examination demonstrated that PQ
administration induced lung injury and pulmonary fibrosis.
HP content in the lungs following PQ
administration
In order to evaluate collagen deposition in the lung
tissues, the HP content was determined 7, 14 and 28 days after PQ
administration. As shown in Fig.
2C, PQ caused a statistically significant (P<0.05) increase
in the amount of HP at all time points. On day 28, the HP content
in the PQ group was more than three times higher than in the
control group. The results of HP quantification were consistent
with the histopathological results.
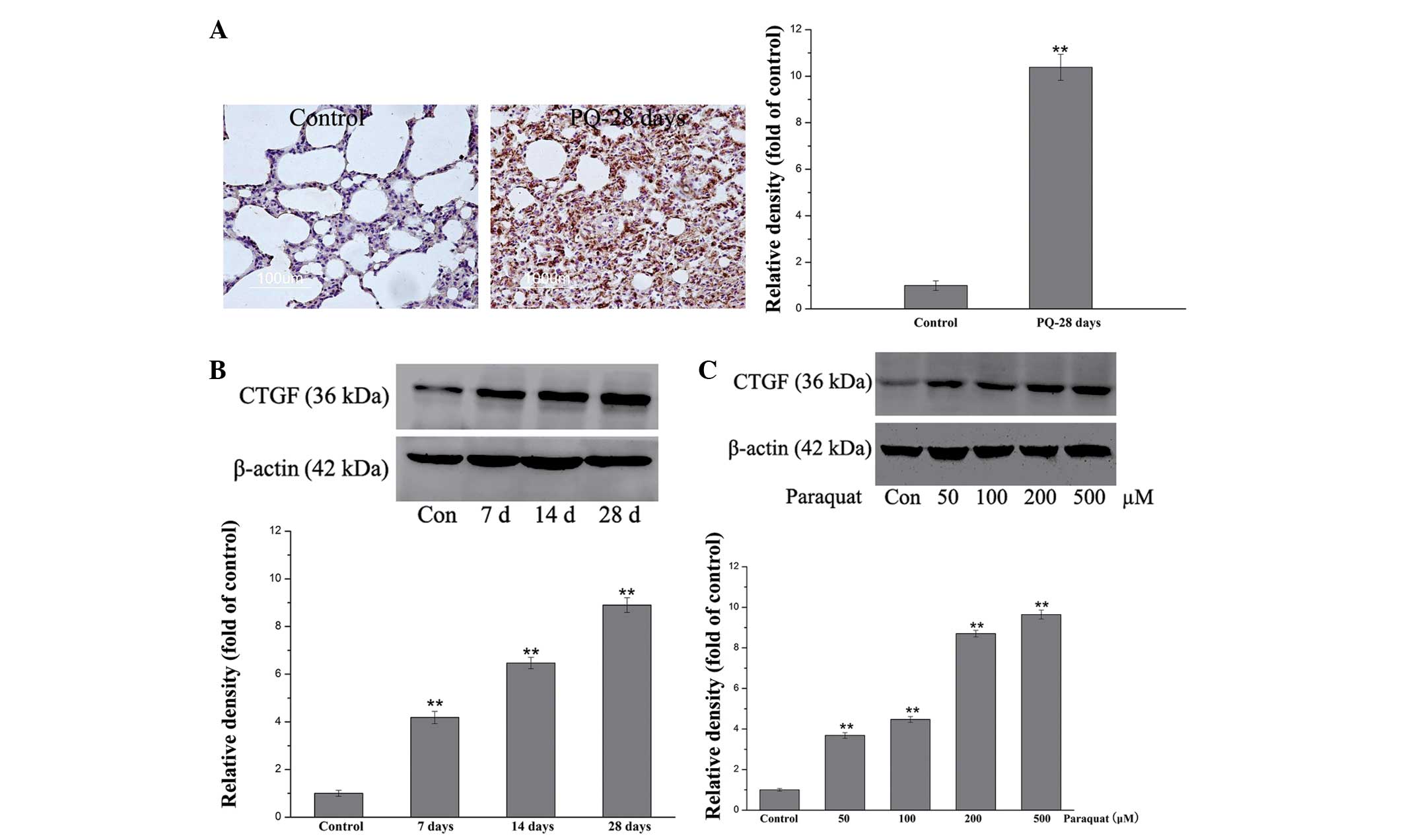
PQ induces expression of CTGF in lung
tissues and lung fibroblasts
Immunohistochemical staining was used to detect the
expression of CTGF in lung tissue. As shown in Fig. 3A, the CTGF expression was
significantly increased 28 days after PQ administration
(P<0.01). Western blot analysis was also performed to confirm
that the CTGF expression was enhanced in response to PQ. As shown
in Fig. 3B, PQ-induced a
time-dependent increase in CTGF protein levels. Furthermore,
western blot analysis was performed using MRC-5 cell lysates to
evaluate whether the same trend could be observed in vitro.
Following exposure to various concentrations of PQ (50–500
μM), the results indicated that the cells exhibited
significantly higher levels of CTGF protein (P<0.01; Fig. 3C). The results suggest that
PQ-induced CTGF expression may contribute to pulmonary
fibrogenesis.
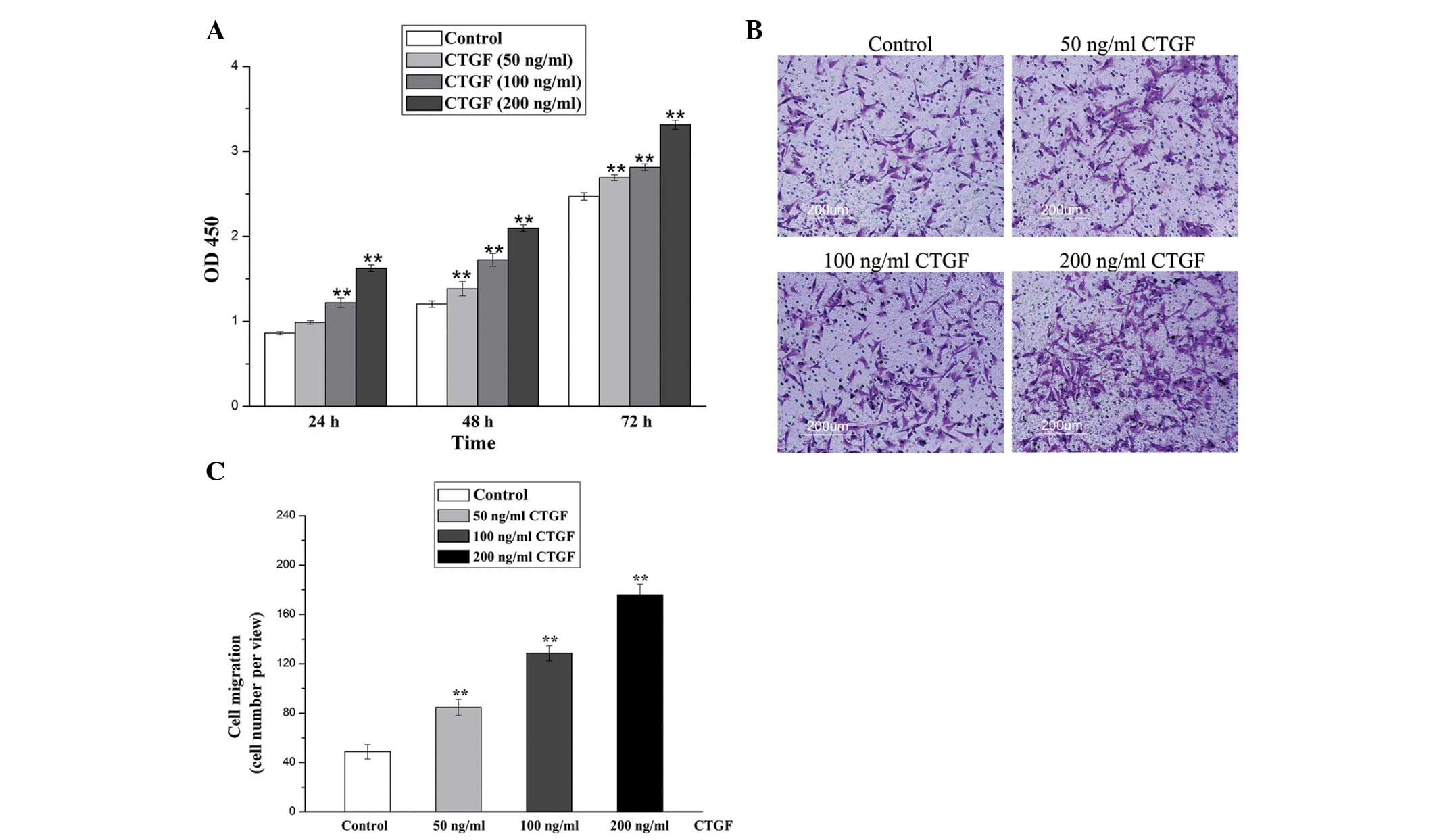
CTGF promotes the proliferation and
migration of lung fibroblasts
The effect of CTGF on the proliferation and
migration of lung fibroblasts was evaluated, as these biological
processes are important in pulmonary fibrogenesis. As shown in
Fig. 4A, the cell viability was
significantly increased in response to CTGF (P<0.01). The data
indicate that CTGF promotes cell proliferation in a dose- and
time-dependent manner. As shown in Figs. 4B and C, the results from the
transwell migration assay reveal that CTGF improves lung fibroblast
migration in a statistically significant manner (P<0.01). In
particular, 200 ng/ml of CTGF caused a 3.6-fold increase in cell
migratory capacity. These results highlight the possibility that
CTGF is able to mediate PQ-induced pulmonary fibrogenesis.
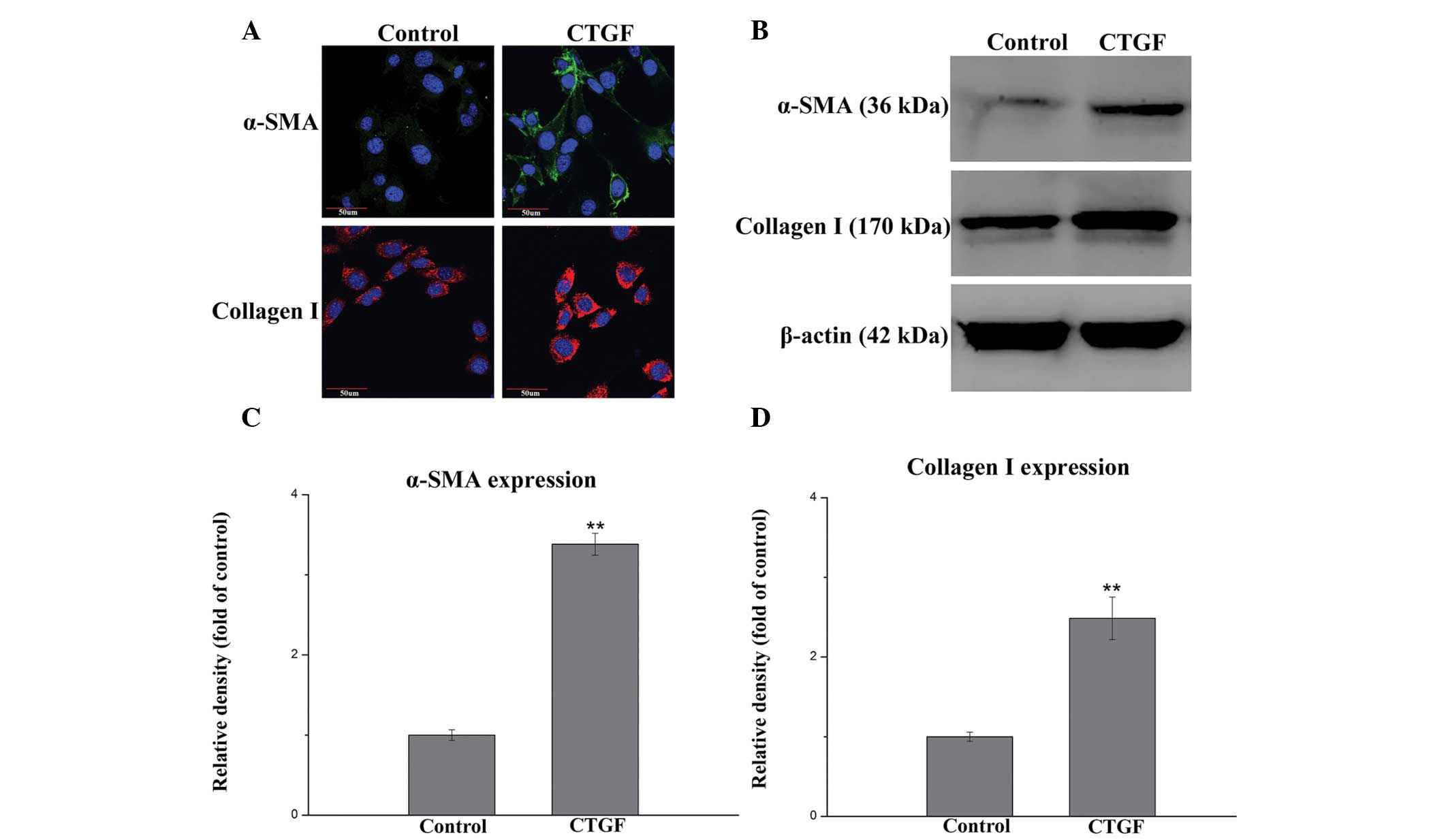
CTGF induces myofibroblast
differentiation of lung fibroblasts
To further investigate the role of CTGF in pulmonary
fibrogenesis, the differentiation of lung fibroblasts into
myofibroblasts was examined. MRC-5 cells were incubated with 100
ng/ml of CTGF for 3 days. Immunofluorescence and western blot
analysis were used to measure expression levels of α-SMA and
collagen I, which are myofibrolast differentiation markers. As
shown in Fig. 5A,
immunofluorescence staining indicated that α-SMA (stained green)
and collagen I (stained red) protein expression was increased in
the CTGF-induced group, as compared with the control group. In
addition, as shown in Figs. 5B–D,
western blotting demonstrated that CTGF induced a pronounced
increase in the protein expression levels of α-SMA and collagen I,
indicating the differentiation of lung fibroblasts into
myofibroblasts. This differentiation process has previously been
associated with lung fibrosis (27).
Discussion
PQ has previously been found to cause acute lung
injury and pulmonary fibrosis with interstitial collagen
deposition, which leads to reduced functional capacity (1). PQ poisoning is a severe health
problem, as numerous human mortalities have occurred as a
consequence of PQ ingestion (4,5). The
lung is the major target organ for this toxic agent, as alveoli
type II epithelial cells absorb PQ through an active polyamine
uptake process (28–30). PQ can accumulate in lung tissue and
reach peak plasma concentrations within 2 h after ingestion
(31). Notably, the concentration
of PQ in the lung parenchyma can be 10–20 times higher than that in
the plasma (32). The signaling
pathways that lead to PQ-induced pulmonary fibrosis remain to be
elucidated. Previous studies have focused on clarifying the
molecular mechanisms of PQ poisoning to determine useful molecular
targets for developing therapeutic strategies. The present study
examined the role of CTGF in PQ-induced collagen production and
myofibroblast differentiation of human lung fibroblasts.
CTGF is a downstream cooperative mediator of the
transforming growth factor-β signaling pathway and is widely
expressed in numerous tissues at low physiological levels. However,
this growth factor is markedly upregulated at the pathological
sites of numerous animal models of human disease, including
pulmonary fibrosis, liver fibrosis, skin fibrosis, cancer and
various types of malignancy (14,33,34).
In particular, increased levels of CTGF have previously been
reported in patients with severe pulmonary fibrosis and animal
models of pulmonary fibrosis (25). In the present study, PQ exposure
caused alterations in lung architecture, which was evident from
interstitial edema, extensive cellular thickening of interalveolar
septa, increased interstitial cells with a fibroblastic appearance
and excessive collagen deposition. Concurrently, it was found that
PQ exposure induces CTGF expression in vitro and in
vivo. A previous in vitro study also indicated that CTGF
can exert an effect on a number of cell types, thereby promoting
biological processes associated with fibrogenesis, including cell
proliferation, migration and ECM production (16). The present study demonstrated that
CTGF can induce the proliferation and migration of MRC-5 lung
fibroblasts. Therefore, it is likely that CTGF is a mediator of
PQ-induced pulmonary fibrogenesis.
Accumulating evidence suggests that the
overexpression of collagen and α-SMA in lung fibrotic lesions is
associated with fibrotic lung disease (35). Collagen is the major ECM component
of the lungs and increases at the early stages of acute lung
injury, affecting respiratory mechanics. Myofibroblasts that
originate from the differentiation of lung fibroblasts exhibit
morphological and biochemical characteristics of lung fibroblasts
and smooth muscle cells, which have been considered the main source
of ECM within the impaired lungs of patients with idiopathic
pulmonary fibrosis (36,37). The results from the present study
are supported by a previous study demonstrating that CTGF knockdown
animals have a reduced pathogenic fibrotic response (29). The mechanism responsible for
protection against fibrosis in CTGF knockdown cells was found to be
reduced collagen synthesis (38).
In the present study, the results indicate that PQ exposure causes
increased levels of collagen and CTGF, in a time-dependent manner.
Immunofluorescent staining and western blot analysis reveal that
lung fibroblasts treated with CTGF have increased expression of
collagen and α-SMA. This observation suggests that CTGF can induce
myofibroblast differentiation.
In conclusion, our observations imply that
PQ-induced overexpression of CTGF may be responsible for pulmonary
fibrosis, through promoting the proliferation, migration and
myofibroblast differentiation of lung fibroblasts. The present
study supports the theory that pharmacological inhibition of CTGF
is a feasible strategy to reduce the magnitude of pulmonary
fibrosis induced by PQ.
Acknowledgments
This study was supported by grants from the National
Natural Science Foundation of China (grant no. 81401583), the Major
Projects Foundation of Nanjing Military Region (grant no. 12Z32),
the Medical Science Foundation for Young Cultivation Project of PLA
(grant no. 13QNP038) and the Natural Science Foundation of Jinling
Hospital (grant nos. 2013023 and 2014004).
References
|
1
|
Dinis-Oliveira RJ, Duarte JA,
Sánchez-Navarro A, et al: Paraquat poisonings: mechanisms of lung
toxicity, clinical features, and treatment. Crit Rev Toxicol.
38:13–71. 2008. View Article : Google Scholar
|
|
2
|
Neves FF, Sousa RB, Pazin-Filho A, et al:
Severe paraquat poisoning: clinical and radiological findings in a
survivor. J Bras Pneumol. 36:513–516. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen CM and Lua AC: Lung toxicity of
paraquat in the rat. J Toxicol Environ Health A. 60:477–487. 2000.
View Article : Google Scholar
|
|
4
|
Baltazar T, Dinis-Oliveira RJ, Duarte JA,
et al: Paraquat research: do recent advances in limiting its
toxicity make its use safer? Br J Pharmacol. 168:44–45. 2013.
View Article : Google Scholar :
|
|
5
|
Novaes RD, Gonçalves RV, Cupertino MC, et
al: Bark extract of Bathysa cuspidata attenuates extra-pulmonary
acute lung injury induced by paraquat and reduces mortality in
rats. Int J Exp Pathol. 93:225–233. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li LR, Sydenham E, Chaudhary B and You C:
Glucocorticoid with cyclophosphamide for paraquat-induced lung
fibrosis. Cochrane Database Syst Rev. 16:CD0080842010.
|
|
7
|
Lin JL, Lin-Tan DT, Chen KH, et al:
Improved survival in severe paraquat poisoning with repeated pulse
therapy of cyclophosphamide and steroids. Intensive Care Med.
37:1006–1013. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Moon JM and Chun BJ: The efficacy of high
doses of vitamin C in patients with paraquat poisoning. Hum Exp
Toxicol. 30:844–850. 2011. View Article : Google Scholar
|
|
9
|
Suntres ZE: Role of antioxidants in
paraquat toxicity. Toxicology. 180:65–77. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Koo JR, Kim JC, Yoon JW, et al: Failure of
continuous venovenous hemofiltration to prevent death in paraquat
poisoning. Am J Kidney Dis. 39:55–59. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Koo JR, Yoon JW, Han SJ, et al: Rapid
analysis of plasma paraquat using sodium dithionite as a predictor
of outcome in acute paraquat poisoning. Am J Med Sci. 338:373–377.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Harlow CR and Hillier SG: Connective
tissue growth factor in the ovarian paracrine system. Mol Cell
Endocrinol. 187:23–27. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Takigawa M, Nakanishi T, Kubota S and
Nishida T: Role of CTGF/HCS24/ecogenin in skeletal growth control.
J Cell Physiol. 194:256–266. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang Q, Usinger W, Nichols B, et al:
Cooperative interaction of CTGF and TGF-β in animal models of
fibrotic disease. Fibrogenesis Tissue Repair. 4:42011. View Article : Google Scholar
|
|
15
|
Moussad EE and Brigstock DR: Connective
tissue growth factor: what’s in a name? Mol Genet Metab.
71:276–292. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ponticos M, Holmes AM, Shi-wen X, et al:
Pivotal role of connective tissue growth factor in lung fibrosis:
MAPK-dependent transcriptional activation of type I collagen.
Arthritis Rheum. 60:2142–2155. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bogatkevich GS, Ludwicka-Bradley A,
Singleton CB, et al: Proteomic analysis of CTGF-activated lung
fibroblasts: identification of IQGAP1 as a key player in lung
fibroblast migration. Am J Physiol Lung Cell Mol Physiol.
295:L603–L611. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shimo T, Kubota S, Kondo S, et al:
Connective tissue growth factor as a major angiogenic agent that is
induced by hypoxia in a human breast cancer cell line. Cancer Lett.
174:57–64. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chujo S, Shirasaki F, Kawara S, et al:
Connective tissue growth factor causes persistent proalpha2(I)
collagen gene expression induced by transforming growth factor-beta
in a mouse fibrosis model. J Cell Physiol. 203:447–456. 2005.
View Article : Google Scholar
|
|
20
|
Heng EC, Huang Y, Black SA Jr and Trackman
PC: CCN2, connective tissue growth factor, stimulates collagen
deposition by gingival fibroblasts via module 3 and alpha6- and
beta1 integrins. J Cell Biochem. 98:409–420. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun Z, Gong X, Zhu H, et al: Inhibition of
Wnt/β-catenin signaling promotes engraftment of mesenchymal stem
cells to repair lung injury. J Cell Physiol. 229:213–224. 2014.
View Article : Google Scholar
|
|
22
|
McAnulty RJ: Fibroblasts and
myofibroblasts: their source, function and role in disease. Int J
Biochem Cell Biol. 39:666–671. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Scotton CJ and Chambers RC: Molecular
targets in pulmonary fibrosis: the myofibroblast in focus. Chest.
132:1311–1321. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bonniaud P, Margetts PJ, Kolb M, et al:
Adenoviral gene transfer of connective tissue growth factor in the
lung induces transient fibrosis. Am J Respir Crit Care Med.
168:770–778. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chen CM, Wang LF, Chou HC, Lang YD and Lai
YP: Up-regulation of connective tissue growth factor in
hyperoxia-induced lung fibrosis. Pediatr Res. 62:128–133. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sun Z, Wang C, Shi C, et al: Activated Wnt
signaling induces myofibroblast differentiation of mesenchymal stem
cells, contributing to pulmonary fibrosis. Int J Mol Med.
33:1097–1109. 2014.PubMed/NCBI
|
|
27
|
Phan SH: The myofibroblast in pulmonary
fibrosis. Chest. 122(6 Suppl): 286S–289S. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ahmed AA: Protective effect of montelukast
on paraquat-induced lung toxicity in rats. Biosci Trends. 3:63–72.
2009.
|
|
29
|
Tomita M, Okuyama T, Katsuyama H, et al:
Mouse model of paraquat-poisoned lungs and its gene expression
profile. Toxicology. 231:200–209. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hoet PH and Nemery B: Polyamines in the
lung: polyamine uptake and polyamine-linked pathological or
toxicological conditions. Am J Physiol Lung Cell Mol Physiol.
278:L417–L433. 2000.PubMed/NCBI
|
|
31
|
Smith LL: Mechanism of paraquat toxicity
in lung and its relevance to treatment. Hum Toxicol. 6:31–36. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Smith LL, Lewis CP, Wyatt I and Cohen GM:
The importance of epithelial uptake systems in lung toxicity.
Environ Health Perspect. 85:25–30. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Lacerda AC, Rodrigues-Machado Mda G,
Mendes PL, et al: Paraquat (PQ)-induced pulmonary fibrosis
increases exercise metabolic cost, reducing aerobic performance in
rats. J Toxicol Sci. 34:671–679. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tung JN, Lang YD, Wang LF and Chen CM:
Paraquat increases connective tissue growth factor and collagen
expression via angiotensin signaling pathway in human lung
fibroblasts. Toxicol In Vitro. 24:803–808. 2010. View Article : Google Scholar
|
|
35
|
Bhogal RK, Stoica CM, McGaha TL and Bona
CA: Molecular aspects of regulation of collagen gene expression in
fibrosis. J Clin Immunol. 25:592–603. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Phan SH: Biology of fibroblasts and
myofibroblasts. Proc Am Thorac Soc. 5:334–337. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kendall RT and Feghali-Bostwick CA:
Fibroblasts in fibrosis: novel roles and mediators. Front
Pharmacol. 5:1232014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu X, Wu W and Chen H: Effects of CTGF
gene silencing on the proliferation and myofibroblast
differentiation of human lung fibroblasts. Sheng Wu Yi Xue Gong
Cheng Xue Za Zhi. 25:407–412. 2008.In Chinese. PubMed/NCBI
|