Introduction
Hypoxia inducible factor-1 (HIF1) has been
identified for ~20 years (1) and
is necessary to reprogram cellular metabolism, which is essential
during physiological and pathological processes (2). HIF-1 regulates the transcription of
hundreds of genes, which are involved in energy metabolism
(3). The role of HIF1 in glucose
metabolism has been well addressed. Previous investigations have
suggested that HIF1 is also important in lipid metabolism,
including regulating lipid uptake and trafficking, fatty acid
metabolism, sterol metabolism, triacylglycerol synthesis and
phospholipid metabolism, lipid droplet formation and lipid
signaling (4–6).
Adipose differentiation-related protein (ADRP) is a
structural protein in lipid droplets, which regulates lipid
accumulation. Overexpression of the ADRP gene stimulates lipid
accumulation in mouse fibroblasts (7) and in human macrophages (8), whereas knockdown of the ADRP gene
reduces the level of triglycerides and esterified cholesterol in
THP-1 macrophages (8). In
addition, in a mouse model, inactivation of ADRP gene results in a
significant reduction in lipid droplet accumulation following
overnight incubation with oxidized low density lipoprotein (oxLDL)
(9). These results suggest that
ADRP facilitates lipid accumulation.
It has been reported that the expression of ADRP
increases under hypoxic conditions (10–12).
However, whether ADRP is a direct target gene of HIF1 is unknown.
In a previous study, lipid accumulation was observed in human
breast cancer and it was identified that enhancement of LDL and
VLDL uptake induced by HIF1 under hypoxia contributes to lipid
accumulation in the breast cancer cell line MCF7 (13). However, whether ADRP is also
involved in lipid accumulation and the uptake of LDL and VLDL under
hypoxia in breast cancer, remains to be elucidated. In the present
study, a dual luciferase assay and chromatin immunoprecipitation
(ChIP) experiment was performed to determine the presence of a
functional hypoxia response element (HRE) in the ADRP gene promoter
and determine whether ADRP is a direct target gene of HIF1. The
results of our previous study showed that hypoxia promoted LDL and
very-(V)LDL uptake, in an HIF1-dependent manner, thus the present
study investigated the role of ADRP in LDL and VLDL uptake and
lipid accumulation under hypoxia.
Materials and methods
Cell culture, hypoxic exposure, DFO, LDL
and VLDL treatment
MCF7, a breast cancer cell line used for
investigating the role of hypoxia in lipid accumulation in a
previous study (13), was
purchased from the Cell Center of the Institute of Basic Medical
Science, Chinese Academy of Medical Sciences (Beijing, China). MCF7
cells (1×106) were grown in Dulbecco's modified Eagle's
medium (DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) supplemented with 10% fetal bovine serum (FBS), 100 U/ml
penicillin and 100 µg/ml streptomycin (all from Gibco;
Thermo Fisher Scientific, Inc.). Cells incubated in normoxic cells
were maintained at 37°C in a 5% CO2 and 95% air
incubator. The cells exposed to hypoxia were placed in a three gas
incubator (YCP-50S; Huaxi Electronic Technologies, Changsha, China)
with 5% CO2, 94% N2 and 1% O2 at
37°C. The cells reached 70% confluence prior to treatment with DFO,
LDL or VLDL. For DFO treatment, 100 µM DFO (Sigma-Aldrich,
St. Louis, MO, USA) was added to the culture medium for 24 h. For
LDL treatment, the culture medium was replaced with fresh medium
(10% FBS) with, or without, LDL at a final concentration of 200
µg/ml. For VLDL treatment, the culture medium was replaced
with fresh medium (10% FBS), with or without, VLDL at a final
concentration of 100 µg/ml. The cells were placed in either
normoxic or hypoxic incubators, cultured for 24 h, and then fixed
with formaldehyde (Sigma-Aldrich, St. Louis, MO, USA) and
recollected for analysis. Human LDL and VLDL were purchased from
Peking Union Bio Co., Ltd. (Beijing, China).
Plasmid construction
The promoter region of the ADFP gene was
amplified from human genomic DNA using polymerase chain reaction
(PCR) and cloned into a luciferase reporter vector (pGL3-Basic;
Promega Corporation, Madison, WI, USA). Briefly, human genomic DNA
was extracted using a Quick Genomic DNA Extraction kit (Guangzhou
Dongsheng Biotech Co., Ltd., Guangzhou, China) according to
manufacturer's instructions. A total of 50 ng genomic DNA was then
used as a template to amplify the promoter region of the ADFP gene
in 20 µl reaction system containing 1 µl of 10
µM primers (Sangon Biotech Co., Ltd. Shanghai, China), 1
µl of 25 µM dNTP mixture (Beijing TransGen Biotech
Co., Ltd., Beijing, China) and 1 µl DNA polymerase (Beijing
TransGen Biotech Co., Ltd.). The following primer sequences were
used: ADFP, forward 5′-agacgcgtCATGCCTGGCTATTTAGTG-3′ and reverse
5′-ccctcgagCTCATGCCGGTAATCCCAGCA-3′. The PCR reaction was performed
in a PCR Thermocycler (Thermo Fisher Scientific, Inc.) with the
following reaction conditions: 95°C for 5 min, followed by 40
cycles of 95°C for 30 sec, 60°C for 30 sec and 72°C for 1 min. As a
positive control, the nucleotide sequence containing the identified
HRE of the erythropoietin (EPO) gene (14) was also cloned into the
pGL3-promoter.
Western blotting
Total protein was extracted from the tissue samples
or cells using lysis buffer (Beyotime Institute of Biotechnology,
Shanghai, China), and subsequently quantified using the Bradford
method (Bio-Rad Laboratories, Inc., Hercules, CA, USA). A total of
20 µg protein was separated by 10% SDS-PAGE (Bio-Rad
Laboratories, Inc., Hercules, CA, USA) and transferred onto a
polyvinylidene fluoride membrane (Bio-Rad Laboratories, Inc.).
Following blocking with 1% bovine serum albumin (Sigma-Aldrich),
the membrane was incubated with primary antibodies overnight at
4°C, then washed with Tris-buffered saline with 0.1% Tween 20
buffer (TBST; Beyotime Institute of Biotechnology) at room
temperature, prior to incubation with secondary antibody for 1 h at
room temperature. Following washing with TBST buffer, the signals
were detected using an Electrochemiluminescence kit (EMD Millipore,
Billerica, MA, USA) and analyzed with ImageJ version 1.41o
(National Institutes of Health, Bethesda, MD, USA). The antibodies
used in the present study included: Mouse anti-human HIF-1α
monoclonal antibody (dilution 1:500; cat. no. sc-13515; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), rabbit anti-human β-actin
monoclonal antibody (dilution 1:1000; cat. no. sc-1616-R; Santa
Cruz Biotechnology, Inc.), rabbit anti-human ADRP polyclonal
antibody (dilution 1:500; cat. no. ab78920; Abcam, Cambridge, MA,
USA), and horseradish peroxidase-conjugated goat anti-mouse
(dilution 1:10,000; cat. no. sc-2005; Santa Cruz Biotechnology,
Inc.) and goat anti-rabbit (dilution 1:10,000; cat. no. sc-2004;
Santa Cruz Biotechnology, Inc.) secondary antibody.
Transient transfection and luciferase
assay
The MCF7 cells were plated at a density of
1×105 cells into 24-well plates to reach ~50–70%
confluence the following day. The cells were co-transfected with
the pGL3-basic-based construct and pRL-TK plasmid DNAs using
Lipofectamine Plus (Invitrogen; Thermo Fisher Scientific, Inc.).
The transfection medium was replaced with complete medium after 6
h. The cells were incubated in normoxia or hypoxia for an
additional 24 h. Following incubation, the cells were lysed and
reporter gene expression was assessed using a dual-luciferase
reporter assay system (Promega Corporation). The cells were lysed
and reporter gene expression was assessed using a Dual
Luciferase® Reporter Assay system (Promega Corporation)
according to manufacturer's instructions. Briefly, the cells were
lysed with passive lysis buffer (Promega Corporation) at room
temperature, the firefly and Renilla luciferase activities
of the lysates were determined, and the firefly luciferase activity
levels were normalized to that of Renilla luciferase.
RNA isolation and PCR analysis
The cells were dissolved in TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc.), and total RNA was
extracted, according to the manufacturer's protocol. Total RNA (1
µg) was converted into 1 µg cDNA using an M-MLV
reverse-transcription system (Invitrogen; Thermo Fisher Scientific,
Inc.) in the presence of oligo (dT)18 (Beijing TransGen
Biotech Co., Ltd.). Reverse transcription-quantitative (RT-q)PCR
was performed using an ABI-7500 Fast Sequence Detection system
(Applied Biosystems; Thermo Fisher Scientific, Inc.) with
SYBR® Green PCR mix (Beijing TransGen Biotech Co.,
Ltd.). The reaction system contained 10 µl 2X
SYBR® Green PCR Master mix (Beijing TransGen Biotech
Co., Ltd.), 4 pmol of each primer (Sangon Biotech Co., Ltd.) and
0.2 µl RT reaction product. The samples were set in
triplicate. The thermocycling parameters were as follows: 95°C for
10 min, followed by 40 cycles of 95°C for 15 sec and 60°C for 30
sec, and a detection step at 72°C for 30 sec. The specific gene
primers were as follows: HIF-1α forward, 5′-AGG TGG ATA TGT CTG
GGTTG-3′, HIF-1α reverse, 5′-AAG GAC ACA TTC TGT TTG TTG-3′; ADRP
forward, 5′-GGC TAG ACA GGA TTG AGG AGAG-3′, and ADRP reverse,
5′-TCA CTG CCC CTT TGG TCTTG-3′. The relative abundance of the
HIF-1α and ADRP transcript was quantified using the comparative Cq
method (15), with β-actin as an
internal control.
ChIP assay
The MCF7 cells were plated (2×106) into a
15 cm plate and grown to ~70% confluence. Then the cells were
exposed to hypoxia for 24 h, and a ChIP assay was performed, as
previously described (16).
Briefly, the cells were fixed with 1% (v/v) formaldehyde
(Sigma-Aldrich) for 10 min at room temperature. Crosslinking was
blocked by the addition of 0.125 M glycine (Sigma-Aldrich) for 5
min at room temperature. The cells were then washed with ice-cold
phosphate-buffered saline (PBS) and resuspended in cell lysis
buffer provided in the EZ-Magna ChIP kit (cat. no. 17-409; EMD
Millipore). The lysates were centrifuged at 10,000 x g at 4°C and
the pellets were resuspended in nuclear lysis buffer provided in
the EZ-Magna ChIP kit (EMD Millipore). The nuclear lysates were
sonicated for chromatin fraction. The chromatin fraction was
immunoprecipitated overnight at 4°C with 5 µg rabbit
anti-human HIF-1α polyclonal antibody (cat. no. ab2185; Abcam) and
5 µg rabbit anti-human IgG polyclonal antibody (cat. no.
ab2410; Abcam) respectively. The precipitated DNA was amplified by
PCR with the following primers: P1, 5′-ACTCGGGCTTGGGACAGGGC-3′; P2,
5′- GCGAAAGGCGAAGAGCAGGCG-3′; P3, 5′-T GACAT CA AG CAGT C CAC C C
-3′; a n d P4, 5′-CTCAGGAGGGAGAAGTAAAGTTG-3′. PCR reactions were
performed in a 20 µl system containing 1 µl of 10
µM primers (Sangon Biotech Co., Ltd.), 1 µl of 25
µM dNTP mixture (Beijing TransGen Biotech Co., Ltd.) and 1
µl DNA polymerase (Beijing TransGen Biotech Co., Ltd.) for
30 cycles consisting of denaturing for 10 sec at 94°C, annealing
for 30 sec at 58°C and extension for 1 min at 72°C. As a positive
control, the protein phosphatase 1, regulatory subunit 3C (PPP1R3C)
promoter region, which contained a known HIF-1 binding site, was
amplified using primers as previously described (17).
Cell transfection with small interfering
(si)RNA
At 1 day prior to transfection, the cells were
plated (5×105) into 6-well plates. The cells were grown
to 50% confluence and then transfected with 25 nM (final
concentration) of siGENOME non-targeting siRNA2, human HIF1A,
siGENOME SMART pool, or human ADRP siGENOME SMART pool (Thermo
Fisher Scientific, Inc.) using Dharma FECT1 transfection reagent,
according to the manufacturer's protocol (Thermo Fisher Scientific,
Inc.). Following 24 h incubation in normoxic conditions, the
transfection medium was replaced with complete medium, and the
cells were incubated in hypoxic conditions for another 24 h. The
total RNA and cell lysates were collected for qPCR and western
blotting, respectively.
Flow cytometric analysis
The cells were plated (5×105) into 6-well
plate. Following incubation and treatment, the cells were digested
with 0.25% trypsin-EDTA (Gibco; Thermo Fisher Scientific, Inc.),
collected and washed with PBS three times at room temperature.
Subsequently, the cells were fixed with 3.7% formaldehyde
(Sigma-Aldrich) for 30 min at room temperature. The cells were then
rinsed immediately with PBS twice at room temperature, following
which the cells were stained with 1 ml Nile Red working solution
for 20 min at 37°C. The cells were then rinsed with PBS twice at
room temperature, resuspended in PBS and analyzed immediately using
an Accuri C6 flow cytometer system (Accuri cytometers, Ann Arbor,
MI, USA). The Nile Red stock solution was purchased from Genmed
Scientifics (Arlington, MA, USA), and was prepared by diluting 0.5
µl of the stock solution in 1 ml PBS, and mixing well.
Intracellular lipid qualification
The 1×105 MCF7 cells were plated into
24-well plate following incubation and treatment, and intracellular
lipid quantification was performed using a Steatosis Colorimetric
Assay kit (cat. no. 10012643-1; Cayman Chemical Co., Ann Arbor, MI,
USA) according to the manufacture's protocol. Briefly, the cells
were fixed with 1% (v/v) formaldehyde (Sigma-Aldrich) for 15 min at
room temperature, washed twice with PBS (Thermo Fisher Scientific,
Inc.) at room temperature for 5 min each, following which the well
was dried completely and Oil Red O (Cayman Chemical Co.) working
solution added to all cells, and incubated for 20 min at room
temperature. The cells were washed with distilled water five times
at room temperature for 5 min, and the cells were dried completely.
Finally, dye extraction solution (Cayman Chemical Co.) was added to
each well, gently mixed for 30 min at room temperature, and the
absorbance was measured at 490 nm with a microplate reader (BioTek
Instruments Inc., Winooski, VT, USA).
Statistical analysis
The data were presented as the mean ± standard
deviation, and subjected to one-way analysis of variance. Student's
t test was used to compare the relative expression levels of target
genes and the relative quantity of cellular lipid. The SPSS version
10.0 software package for Windows (SPSS, Inc., Chicago, IL, USA)
was used for statistical analysis. P<0.05 was considered to
indicate a statistically significant result.
Results
Hypoxia induces ADRP in an HIF1-dependent
manner
To examine whether the gene expression of
ADRP was oxygen regulated, the MCF7 cells were incubated
under normoxic (21% O2) or hypoxic (1% O2)
conditions, or in medium containing 100 µM deferoxamine
mesylate salt (DFO) for 24 h. Subsequent qPCR assays (Fig. 1A) revealed an increase in the mRNA
levels of ADRP in the cells under hypoxic conditions or following
DFO treatment. The protein levels of ADRP in the MCF7 cells were
determined using immunoblotting (Fig.
1B). Increased protein levels of ADRP were observed with
increasing protein levels of HIF1A in the cells under hypoxic
conditions or following DFO treatment. These results indicated
ADRP as a hypoxia-inducible gene. In agreement, a
significant decrease in the mRNA and protein levels of ADRP
(Fig. 1C and D) were detected in
the MCF7 cells transfected with siRNA targeting HIF1A, which
suggested that inhibition of the HIF1A gene reduced the
hypoxic induction of ADRP. Taken together, these results
demonstrated that hypoxia induced the expression of ADRP in
a HIF1-dependent manner.
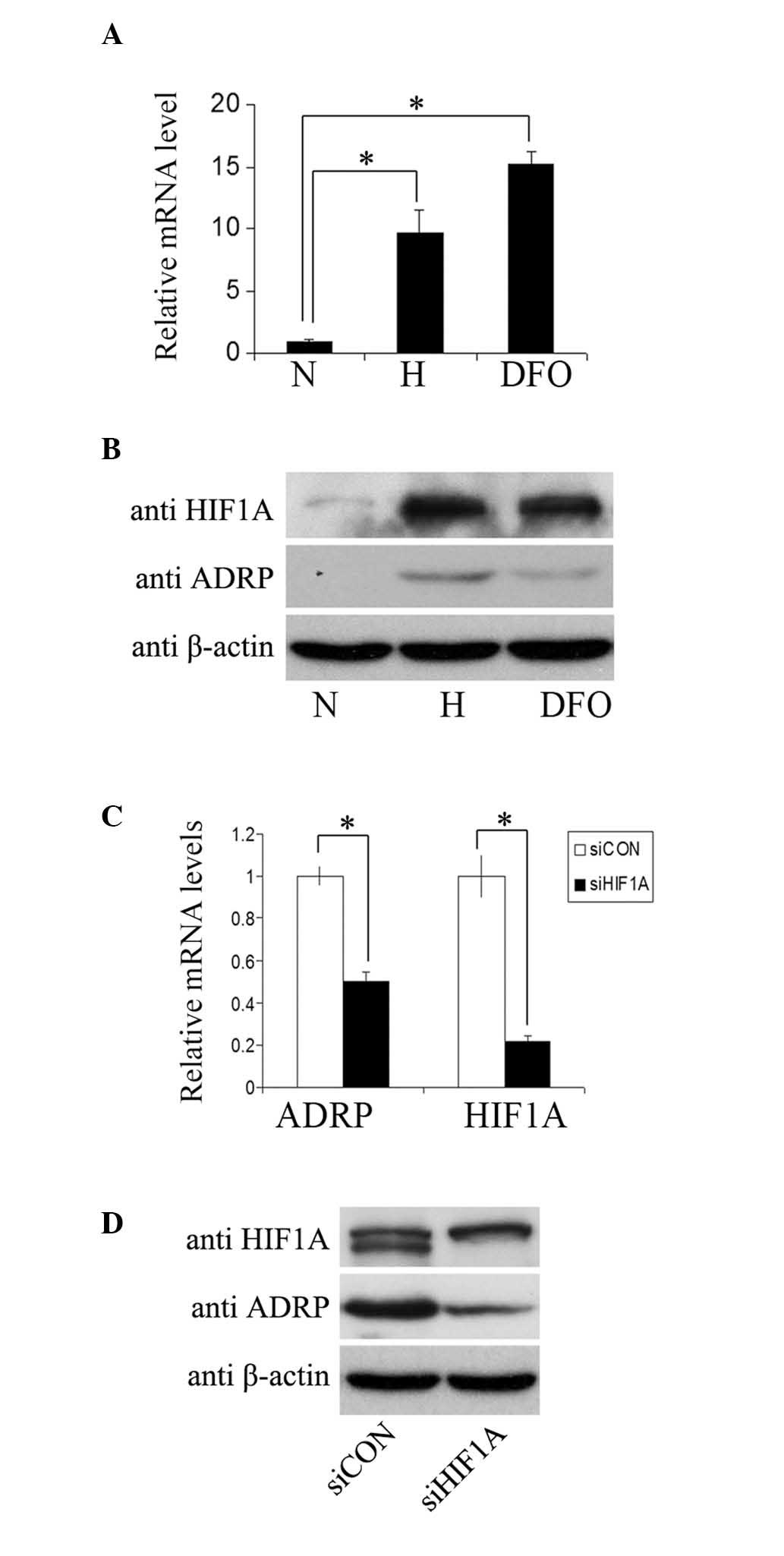 | Figure 1ADRP is induced by hypoxia in
an HIF-dependent manner. (A) mRNA levels of ADRP were analyzed
using qPCR in MCF7 cells cultured in normoxia or hypoxia, or in
medium containing 100 µM DFO for 24 h. The mRNA levels of
ADRP were normalized to those of β-actin. The relative mRNA
expression of ADRP is presented as the values in hypoxia, relative
to normoxia. Data are expressed as the mean ± standard
deviation.(B) Protein levels of HIF1α, ADRP and β-actin in the MCF7
cells were determined using western blotting under the different
treatment conditions. Anti-β-actin antibody was used as a control
for equal protein loading. (C) mRNA levels of ADRP and HIF1α were
determined using qPCR in MCF7 cells transfected with siHIF1A and
siCON. The relative mRNA expression levels are presented as the
fold values of mRNA levels in cells transfected with siHIF1A/cells
transfected with siCON. Data are expressed as the mean ± standard
deviation (D) Protein levels of HIF1α, ADRP and β-actin in the
transfected MCF7 cells were determined using western blot assays,
as above. *P<0.05. ADRP, Adipose
differentiation-related protein; HIF1, hypoxia-inducible factor-1;
N, normoxia; H, hypoxia; DFO, deferoxamine mesylate salt; si, small
interfering RNA; CON, control. |
Identification and validation of HRE in
the human ADRP gene
To determine whether ADRP was a direct target of
HIF1 under hypoxia, the present study examined the promoter region
of the human ADRP gene for a consensus HRE sequence, as
previously described (3). Several
putative HREs were identified (Fig.
2A), however, only the HRE at ~-33 in position is conserved in
human, mouse and rat (Fig. 2B). To
determine whether this was a functional HRE, the promoter region of
the ADRP gene was amplifed and inserted it into the
luciferase reporter plasmid, pGL3-promoter. The plasmid of the
pGL3-promoter was used as a negative control. A construct of the
pGL3-promoter with the insertion of the identified HRE of the
EPO gene was used as a positive control. As shown in
Fig. 2C, the region between −754
and +635 markedly increased luciferase activity in the MCF7 cells
under hypoxia. Analysis of the deletion constructs suggested that
the conserved HRE was functional (Fig.
2C). Mutation of the conserved HRE significantly impaired the
induction of luciferase activity by hypoxia, however, mutation of
the putative HRE in exon 1 did not impair the luciferase activity
by hypoxia (Fig. 2D and E), which
also suggested that the conserved HRE may be a functional HRE.
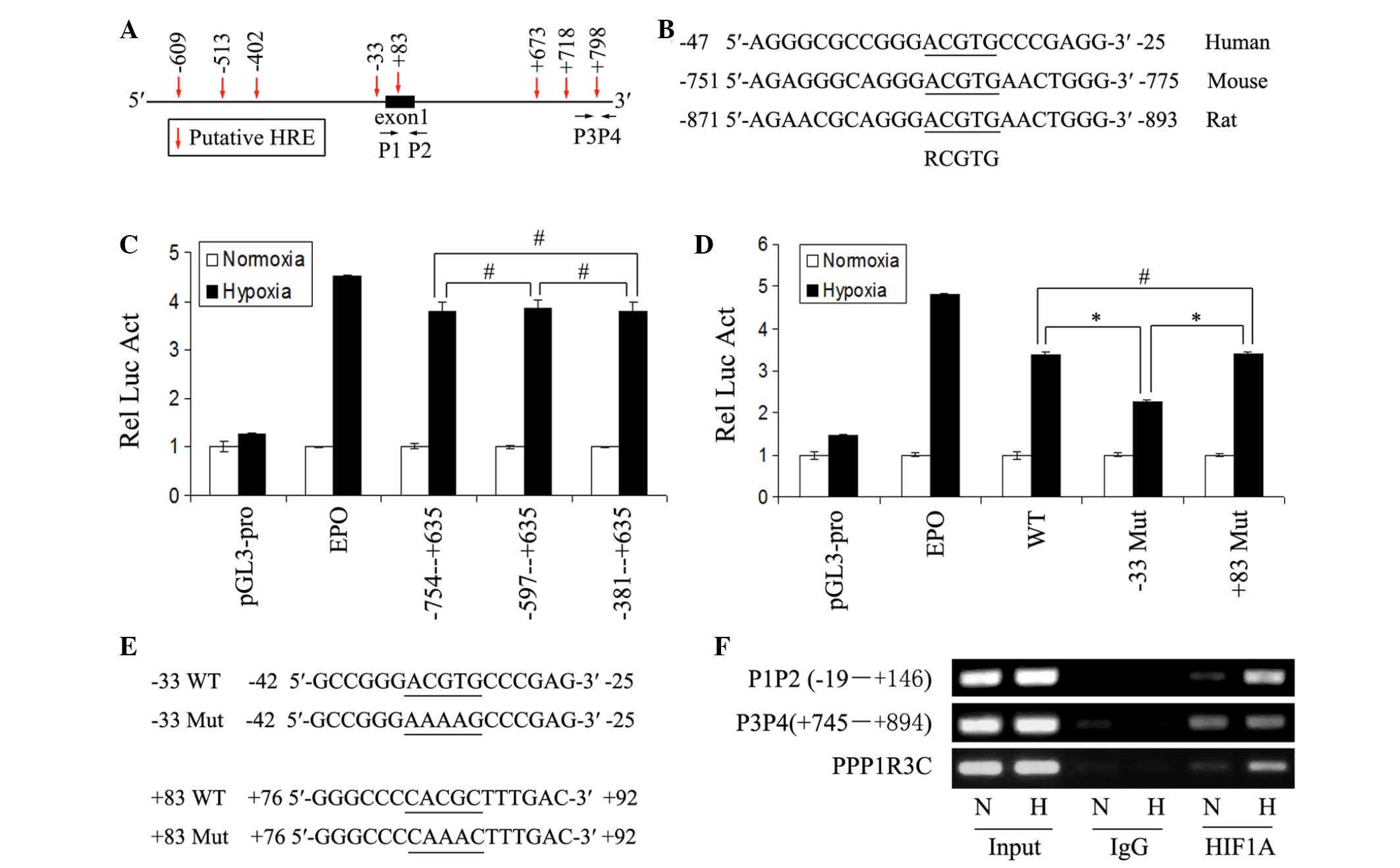 | Figure 2Identification and validation of HREs
in the ADRP gene promoter. (A) Sketch map of the ADRP
promoter region. The putative HREs are indicated by arrows. The
nucleotide sequences are numbered in relation to the transcription
initiation site, which is designated '+1'. P1, P2, P3 and P4
indicate the primers used for polmterase chain reaction
amplification of the immunoprecipitated chromatin fragments in
Fig. 2E. (B) Comparison of the
conserved HRE and flanking nucleotides identified in the human, rat
and mouse ADRP gene promoter regions. (C and D) Luciferase reporter
assays were performed in MCF7 cells transfected with the constructs
containing the indicated sequences from the human ADRP gene
promoter region. Each transfection experiment was performed in
triplicate. The relative mean luciferase activity in the cells
under hypoxia is shown as the fold over the mean activity in the
cells under normoxia. Error bars represent the mean ± standard
deviation. pGL3-pro and EPO represent negative and positive
control, respectively. (E) Mutant sequence of the putative HREs in
the construct of Fig. 2D are
indicated. (F) ChIP-PCR assays were performed using the indicated
primers and antibodies, to demonstrate binding of HIF1α to the -33
HRE of ADRP in the MCF7 cells under normoxic and hypoxic
conditions. PPP1R3C was used as a positive control. ADRP, adipose
differentiation-related protein; HIF1, hypoxia-inducible factor-1;
N, normoxiaa; H, hypoxia; HRE, hypoxia response element; EPO,
erythropoietin; WT, wild-type; Mut, mutant; PPP1R3C, protein
phosphatase 1, regulatory subunit 3C; Rel Luc Act, relative
luciferase activity. *P<0.05 and
#P>0.05. |
To determine whether HIF1 binds to the conserved HRE
within living cells, the present study performed ChIP assays using
antibodies against HIF1A (IgG as a negative control) in the
normoxic- and hypoxic-cultured MCF7 cells. The sequence containing
the potential functional HRE in the ADRP gene promoter was
detected, and hypoxia significantly increased HIF1A binding to the
HRE (Fig. 2F). The sequence
containing the binding site of HIF1 in the known HIF1 target gene
PPP1R3C, served as a positive control (17). The sequence between +745 and +894
in the ADRP promoter was detected, with primers P3 and P4 used as a
negative control (Fig. 2F). Taken
together, these results demonstrated that ADRP was a direct target
of HIF1 under hypoxia.
ADRP is not important in hypoxia mediated
LDL and VLDL uptake or lipid accumulation
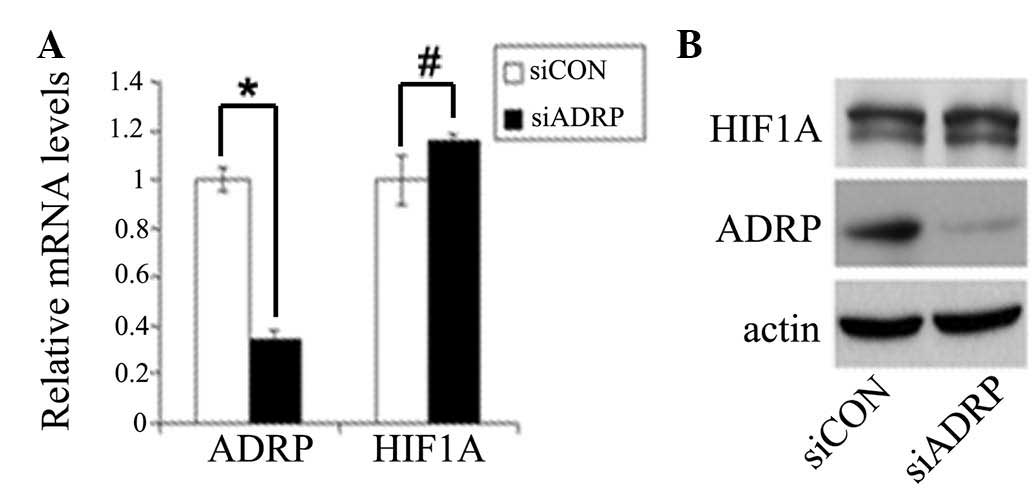
Our previous study demonstrated that hypoxia
significantly increases LDL and VLDL uptake, and enhances lipid
accumulation in MCF7 cells (13),
and another investigation demonstrated that ADRP also promotes
lipid accumulation following overnight incubation with oxLDL
(9). Therefore, the present study
aimed to determine whether ADRP induced by HIF1 also contributes to
lipid accumulation under hypoxia. To investigate this, the MCF7
cells were transfected with ADRP siRNA and treated with LDL or
VLDL, followed by exposure to normoxia or hypoxia for 24 h.
Reductions in the mRNA and protein levels of ADRP were observed in
the cells transfected with the specific siRNA targeting ADRP
(Fig. 3A and B). Flow cytometry
and a steatosis colorimetric assay kit were used to analyze
intracellular lipid content. Consistent with our previous findings,
the results showed that hypoxia induced lipid accumulation and
LDL/VLDL uptake, in an HIF1-dependent manner (Fig. 4A and B). However, compared with the
cells transfected with non-targeting siRNA (control), knockdown of
ADRP did not reduce hypoxia-induced LDL and VLDL uptake or lipid
accumulation (Fig. 4C–E). These
results suggested that HIF1 promoted LDL and VLDL uptake, and lipid
accumulation, but not through the induction of ADRP under
hypoxia.
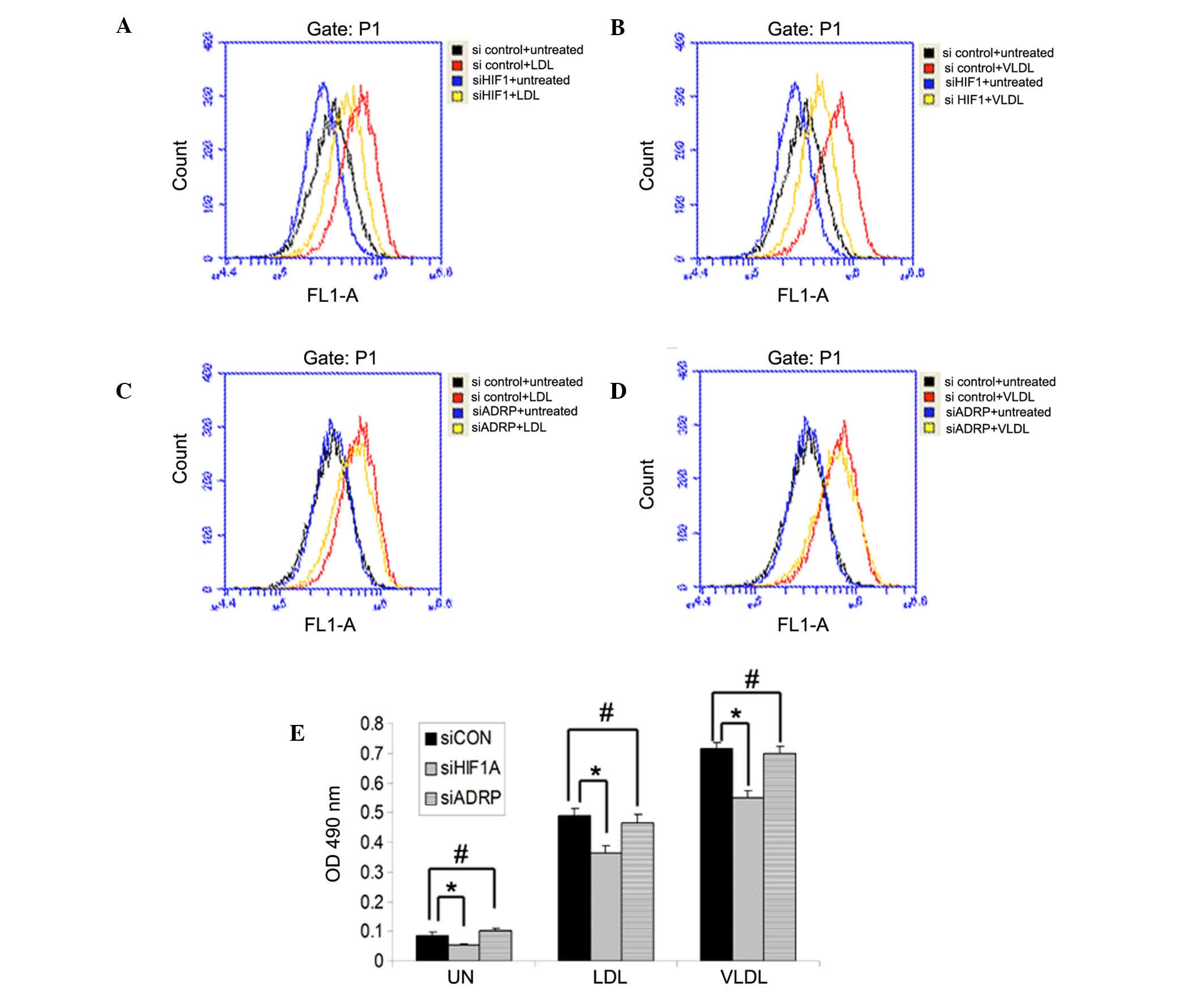 | Figure 4ADRP does not affect LDL and VLDL
uptake or lipid accumulation under hypoxia. MCF7 cells were
transfected with siHIF1α or siCON for 24 h, following which the
cells were treated with (A) LDL or (B) VLDL and incubated under
hypoxia for an additional 24 h. MCF7 cells were transfected with
siADRP or siCON for 24 h, following which the cells were treated
with (C) LDL or (D) VLDL and incubated under hypoxia for an
additional 24 h. Cells (5×104) were stained with Nile
Red, and flow cytometry was used to measure intracellular lipid
levels by detecting the value of OD575. (E) MCF7 cells were
transfected with siRNAs and treated with LDL or VLDL, followed by
incubation under hypoxia. Intracellular lipid content was measured
using a steatosis colorimetric assay kit. Data are presented as the
mean ± standard deviation. ADRP, adipose differentiation-related
protein; HIF1, hypoxia-inducible factor-1; LDL, low density
lipoprotein; VLDL, very-LDL; si, small interfering RNA; CON,
control; UN, untreated; OD, optical density. *P<0.05
and #P>0.05. |
Discussion
ADRP is a structural protein of lipid droplets and
it has been reported that ADRP is expressed at high levels and is
associated with lipid accumulation in solid tumors, particularly in
clear cell lesions (18,19). In step-wise carcinogenesis, the
expression of ADRP is correlated with the proliferation rate and is
upregulated during early tumorigenesis (18). The expression of ADRP is also
associated with atherosclerosis (9). It has been reported that ADRP is
induced under hypoxic conditions in several studies (10–12),
however, whether ADRP is a direct HIF1 target gene remains to be
elucidated. Thus, the present study examined whether ADRP is a
direct target gene of HIF1. The present study demonstrated that
ADRP was a HIF1 direct target gene, and identified a functional HRE
in the ADRP gene promoter; results which are consistent with
previous reports (10–12).
It has been reported that hypoxia significantly
increases LDL and VLDL uptake, and enhances lipid accumulation in
arterial SMCs (20–22), cardiomyocytes (23,24)
and cancer cell lines (13). As
ADRP promotes lipid accumulation and is upregulated under hypoxic
conditions, the present study investigated whether ADRP also
contributes to lipid accumulation under hypoxic conditions. The
presents study examined the impact of ADRP on LDL and VLDL uptake,
and lipid accumulation under hypoxia. However, ADRP did not affect
HIF1-mediated LDL and VLDL uptake or lipid accumulation under
hypoxic conditions in the MCF7 cells. These data are not consistent
with previous investigations, and the different experimental
conditions may offer an explanation for the different results. The
previous study was performed under normoxic conditions and used
ox-LDL to incubate cells (9). In
the present study, the cells were treated with LDL and VLDL under
hypoxic conditions. In addition, lipid uptake and storage are two
separate process. Several proteins are involved in lipid storage.
Hypoxia-inducible protein 2 (HIG2), a novel protein identified as
being associated with lipid droplets, is upregulated by hypoxia and
is a direct and specific target of HIF1. Normoxic overexpression of
HIG2 is sufficient to increase lipid droplets in HeLa cells
(25). When knocking down ADRP,
other HIF1 target genes involving lipid storage, including HIG2,
may compensate the function of ADRP. The combined knockdown of
these genes may affect HIF1-mediated LDL and VLDL uptake, and lipid
accumulation. Therfore, further investigations are required to
identify the genes, which are involved in lipid storage and are
regulated by HIF1 under hypoxic conditions.
Acknowledgments
This study was supported by the National Natural
Science Foundation of China (grant. nos. 31301076, 81201688 and
81401961 to Dr Guomin Shen, Dr Ning Ning and Dr Xiaobo Li,
respectively), the Science and Technology Plan Foundation of Inner
Mongolia Autonomous Region (grant. no. 20130404 to Dr Xingsheng
Zhao), the Hospital Foundation of Inner Mongolia Autonomous Region
People's Hospital (grant. no. 201301 to Dr Xi Liu) and the
Wu-Lian-De Youth Science Foundation of Harbin Medical University
(grant. no. WLD-QN1411 to Dr Xiaobo Li) and the Postdoctoral
Scientific Research Development Fund of Heilongjiang Province
(grant no. LBH-Q14104 to Dr Xiaobo Li).
References
|
1
|
Wang GL, Jiang BH, Rue EA and Semenza GL:
Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS
heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci
USA. 92:5510–5514. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Semenza GL: Hypoxia-inducible factors in
physiology and medicine. Cell. 148:399–408. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Semenza GL: Defining the role of
hypoxia-inducible factor 1 in cancer biology and therapeutics.
Oncogene. 29:625–634. 2010. View Article : Google Scholar :
|
|
4
|
Scarfo LM, Weller PF and Farber HW:
Induction of endothelial cell cytoplasmic lipid bodies during
hypoxia. Am J Physiol Heart Circ Physiol. 280:H294–H301. 2001.
|
|
5
|
Belanger AJ, Luo Z, Vincent KA, Akita GY,
Cheng SH, Gregory RJ and Jiang C: Hypoxia-inducible factor 1
mediates hypoxia-induced cardiomyocyte lipid accumulation by
reducing the DNA binding activity of peroxisome
proliferator-activated receptor alpha/retinoid X receptor. Biochem
Biophys Res Commun. 364:567–572. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chabowski A, Gorski J, Calles-Escandon J,
Tandon NN and Bonen A: Hypoxia-induced fatty acid transporter
translocation increases fatty acid transport and contributes to
lipid accumulation in the heart. FEBS Lett. 580:3617–3623. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Imamura M, Inoguchi T, Ikuyama S,
Taniguchi S, Kobayashi K, Nakashima N and Nawata H: ADRP stimulates
lipid accumulation and lipid droplet formation in murine
fibroblasts. Am J Physiol Endocrinol Metab. 283:E775–E783. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Larigauderie G, Furman C, Jaye M, Lasselin
C, Copin C, Fruchart JC, Castro G and Rouis M: Adipophilin enhances
lipid accumulation and prevents lipid efflux from THP-1
macrophages: Potential role in atherogenesis. Arterioscler Thromb
Vasc Biol. 24:504–510. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Paul A, Chang BH, Li L, Yechoor VK and
Chan L: Deficiency of adipose differentiation-related protein
impairs foam cell formation and protects against atherosclerosis.
Circ Res. 102:1492–1501. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Saarikoski ST, Rivera SP and Hankinson O:
Mitogen-inducible gene 6 (MIG-6), adipophilin and tuftelin are
inducible by hypoxia. FEBS Lett. 530:186–190. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xia X, Lemieux ME, Li W, Carroll JS, Brown
M, Liu XS and Kung AL: Integrative analysis of HIF binding and
transactivation reveals its role in maintaining histone methylation
homeostasis. Proc Natl Acad Sci USA. 106:4260–4265. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bostrom P, Magnusson B, Svensson PA,
Wiklund O, Borén J, Carlsson LM, Ståhlman M, Olofsson SO and Hultén
LM: Hypoxia converts human macrophages into triglyceride-loaded
foam cells. Arterioscler Thromb Vasc Biol. 26:1871–1876. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shen GM, Zhao YZ, Chen MT, Zhang FL, Liu
XL, Wang Y, Liu CZ, Yu J and Zhang JW: Hypoxia-inducible factor-1
(HIF-1) promotes LDL and VLDL uptake through inducing VLDLR under
hypoxia. Biochem J. 441:675–683. 2012. View Article : Google Scholar
|
|
14
|
Wang GL and Semenza GL: Characterization
of hypoxia-inducible factor 1 and regulation of DNA binding
activity by hypoxia. J Biol Chem. 268:21513–21518. 1993.PubMed/NCBI
|
|
15
|
Li X, Zhang J, Gao L, McClellan S, Finan
MA, Butler TW, Owen LB, Piazza GA and Xi Y: MiR-181 mediates cell
differentiation by interrupting the Lin28 and let-7 feedback
circuit. Cell Death Differ. 19:378–386. 2012. View Article : Google Scholar :
|
|
16
|
Pescador N, Villar D, Cifuentes D,
Garcia-Rocha M, Ortiz-Barahona A, Vazquez S, Ordoñez A, Cuevas Y,
Saez-Morales D, Garcia-Bermejo ML, et al: Hypoxia promotes glycogen
accumulation through hypoxia inducible factor (HIF)-mediated
induction of glycogen synthase 1. PLoS One. 5:e96442010. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shen GM, Zhang FL, Liu XL and Zhang JW:
Hypoxia-inducible factor 1-mediated regulation of PPP1R3C promotes
glycogen accumulation in human MCF-7 cells under hypoxia. FEBS
Lett. 584:4366–4372. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Straub BK, Herpel E, Singer S, Zimbelmann
R, Breuhahn K, Macher-Goeppinger S, Warth A, Lehmann-Koch J,
Longerich T, Heid H and Schirmacher P: Lipid droplet-associated
PAT-proteins show frequent and differential expression in
neoplastic steatogenesis. Mod Pathol. 23:480–492. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Ostler DA, Prieto VG, Reed JA, Deavers MT,
Lazar AJ and Ivan D: Adipophilin expression in sebaceous tumors and
other cutaneous lesions with clear cell histology: An
immunohistochemical study of 117 cases. Mod Pathol. 23:567–573.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wada Y, Sugiyama A, Yamamoto T, Naito M,
Noguchi N, Yokoyama S, Tsujita M, Kawabe Y, Kobayashi M, Izumi A,
et al: Lipid accumulation in smooth muscle cells under LDL loading
is independent of LDL receptor pathway and enhanced by hypoxic
conditions. Arterioscler Thromb Vasc Biol. 22:1712–1719. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Castellano J, Aledo R, Sendra J, Costales
P, Juan-Babot O, Badimon L and Llorente-Cortés V: Hypoxia
stimulates low-density lipoprotein receptor-related protein-1
expression through hypoxia-inducible factor-1α in human vascular
smooth muscle cells. Arterioscler Thromb Vasc Biol. 31:1411–1420.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cal R, Castellano J, Revuelta-López E,
Aledo R, Barriga M, Farré J, Vilahur G, Nasarre L, Hove-Madsen L,
Badimon L and Llorente-Cortés V: Low-density lipoprotein
receptor-related protein 1 mediates hypoxia-induced very low
density lipoprotein-cholesteryl ester uptake and accumulation in
cardiomyocytes. Cardiovasc Res. 94:469–479. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Perman JC, Boström P, Lindbom M, Lidberg
U, StÅhlman M, Hägg D, Lindskog H, Scharin Täng M, Omerovic E,
Mattsson Hultén L, et al: The VLDL receptor promotes lipotoxicity
and increases mortality in mice following an acute myocardial
infarction. J Clin Invest. 121:2625–2640. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Castellano J, Farré J, Fernandes J,
Bayes-Genis A, Cinca J, Badimon L, Hove-Madsen L and
Llorente-Cortés V: Hypoxia exacerbates Ca(2+)-handling disturbances
induced by very low density lipoproteins (VLDL) in neonatal rat
cardiomyocytes. J Mol Cell Cardiol. 50:894–902. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gimm T, Wiese M, Teschemacher B, Deggerich
A, Schödel J, Knaup KX, Hackenbeck T, Hellerbrand C, Amann K,
Wiesener MS, et al: Hypoxia-inducible protein 2 is a novel lipid
droplet protein and a specific target gene of hypoxia-inducible
factor-1. FASEB J. 24:4443–4458. 2010. View Article : Google Scholar : PubMed/NCBI
|