Introduction
Osteoporosis is described as a ‘silent disease’,
characterized by gradual bone loss that occurs in the absence of
other symptoms over a period of years (1,2). Due
to its increasing prevalence, osteoporosis severely affects human
health and quality of life (1).
Osteoporosis is a metabolic disease that is common in
postmenopausal women and the elderly (1). Phenotypically, it is characterized by
bone loss, bone microstructural damage, bone fragility and
increased susceptibility to fractures (1). The approved medications for the
prevention and treatment of osteoporosis in clinical practice are
frequently ineffective and cause adverse reactions (1–5). New
therapeutic strategies are therefore urgently required.
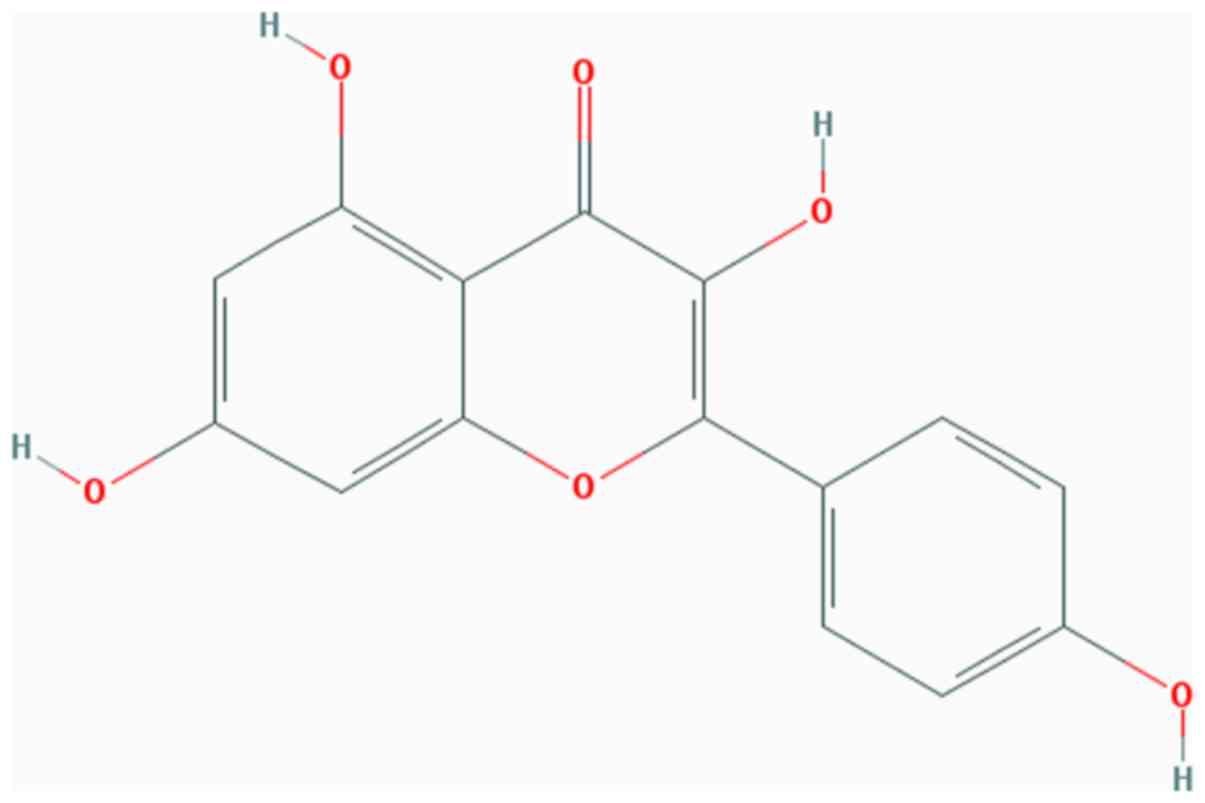
Kaempferol (Kae; Fig.
1) is a type of natural flavonoid, extracted from the rhizome
of Kaempferia galangal L. (6). As a natural flavonol, Kae is present
in a variety of Chinese medicinal herbs, plants, fruits and beans
(6), and is prized for its
medicinal properties, which include anti-inflammatory (7) and antitumoral (8) effects, as well as being beneficial
for the treatment of diabetes (9),
cardiovascular disease (10) and
osteoporosis (11). Kae has been
previously confirmed to be beneficial to bone microarchitecture by
increasing bone density and reversing osteoporosis in
ovariectomized (OVX) rats. However, the precise mechanism(s)
governing these effects have not been defined.
Mammalian target of rapamycin (mTOR) is a member of
the phosphatidylinositol 3-kinase-related kinase family of protein
kinases (12). mTOR functions
through two structurally and functionally distinct multi-protein
complexes, mTORC1 and mTORC2, which are involved in cell growth,
proliferation, survival, protein synthesis, autophagy and
transcription (13). mTOR is also
an important regulator of bone metabolism known to promote
osteoblastic differentiation and increase bone matrix synthesis
(14,15). In addition, mTORC1 and mTORC2 have
been implicated in the regulation of bone homeostasis (16–18).
Therefore, mTOR was hypothesized to be a novel
target for the development of new and effective osteoporosis
therapies. The aim of the present study was to investigate whether
Kae was able to enhance the osteogenic differentiation and function
of bone marrow mesenchymal stem cells (BMSCs) via mTOR
activation.
Materials and methods
Reagents
Kae (purity>98%) was purchased from the National
Institute for the Control of Pharmaceutical and Biological Products
(Beijing, China). Kae was dissolved in DMSO and diluted to 0.01% in
PBS. Rapamycin (Rapa), a specific inhibitor of mTOR, was purchased
from Selleck Chemicals. The Alizarin Red S (ARS) staining buffer
and alkaline phosphatase (ALP) detection kits were purchased from
Nanjing Jiancheng Bioengineering Institute. Anti-runt-related
transcription factor 2 (Runx2; cat. no. ab23981) and anti-Osterix
(cat. no. ab22552) was purchased from Abcam. Anti-eukaryotic
translation initiation factor 4E-binding protein 1 (4E/BP1; cat.
no. 94525), anti-phosphorylated (p)-4E/BP 1 (cat. no. 2855) and
anti-ribosomal protein S6 kinase B1 (S6K1; cat. no. 9202),
anti-p-S6K1 (cat. no. 9204) was obtained from Cell Signaling
Technologies, Inc. Horseradish peroxidase-labeled
anti-immunoglobulin G secondary antibody (goat anti-mouse lgG; cat.
no. SA00001-1; and goat anti-rabbit lgG; cat. no. SA00001-2) was
obtained from ProteinTech Group, Inc. Anti-β-actin antibody (cat.
no. KL002) was provided by Nanjing Jiancheng Bioengineering
Institute.
Animals
A total of 30 adult (age, 6–8 weeks) female
Sprague-Dawley (SD) rats weighing 180–220 g were obtained from the
Nanchang University Laboratory Animal Center (Nanchang, China) and
maintained under a 12-h dark/light cycle at 22–25°C and 40–70%
humidity. Animals were allowed access to food and water ad
libitum. Experiments were performed according to the Guide for
the Care and Use of Laboratory (National Institutes of Health), and
were approved by the Ethics Committee of Nanchang University (no.
2017-0122).
Experimental groups and
treatments
A total of 30 rats were randomly divided into five
groups (n=6 in each group) as follows: i) Sham group, in which the
abdominal cavities of the rats were opened and fat tissue around
the ovaries removed; ii) OVX group, in which the rats were
ovariectomized; iii) Kae treatment group (OVX + Kae), in which the
OVX rats were continuously given Kae (100 mg/kg/day) via gavage for
8 weeks; iv) Kae and Rapa administration group (OVX + Kae + Rapa),
in which the rats were treated as in the Kae treatment group and in
addition received intraperitoneal injections of Rapa (0.2
mg/kg/day) for 8 weeks; and v) Rapa group (OVX + Rapa), in which
the OVX rats received intraperitoneal injections of Rapa (0.2
mg/kg/day) for 8 weeks. The dose and timing of Kae and Rapa
administration were ascertained in preliminary experiments.
Briefly, the OVX rats were given Kae at 25, 50 and 100 mg/kg/day.
Bone mineral density (BMD) was detected after 8 weeks to select the
working concentration. Kae at 100 mg/kg/day was the most effective
concentration in improving BMD in OVX rats. Rapa was administered
at 0.2 mg/kg/day according to the manufacturer's protocol (Selleck
Chemicals). Subsequent experiments were performed following the 8
weeks of treatment.
Assessment of BMD and bone
microarchitecture
Following treatment, the rats were sacrificed and
their right femurs and tibias were dissected. The 2D total bone
mineral content was used to calculate the BMD as previously
described (19). The right femurs
of the rats were analyzed using dual-energy X-ray absorptiometry
with a Lunar Prodigy Advance system (version 13.6; GE
Healthcare).
Based on the median values of total BMD, selected
trabecular microarchitecture of the femoral metaphysis was
evaluated using micro-computed tomography (micro-CT; Scanco Medical
AG) with Scanco image processing language software (version 5.08b;
Scanco Medical AG). Scans were performed from the proximal growth
plate in the distal direction (18 µm/slice) as the distal femur has
a high concentration of trabecular bone compared with the proximal
and middle regions of the femur. A volume of interest (VOI) was
selected, defined as the cross-sectional area spanning 100 slices
from the proximal growth plate. The 2D scans were used to produce
3D reconstructions of the bone microarchitecture, which were used
to measure bone morphometric parameters of the selected VOI,
including the bone volume fraction [bone volume/tissue volume
(BV/TV)], trabecular number (Tb.N), trabecular separation (Tb.Sp),
trabecular thickness (Tb.Th) and structure model index (SMI). The
operator conducting the CT analysis was blinded to the treatments
associated with the specimens. All examinations were conducted
according to the principles and procedures described in the most
recent National Research Council publication of the Guide for the
Care and Use of Laboratory Animals and the ARRIVE guidelines
(20).
Isolation of rat BMSCs
BMSCs were flushed from the femurs and tibias of
normal SD and OVX rats with PBS in a biosafety cabinet, using a 5
ml syringe fitted with a needle (21G). Mononucleated cells were
isolated by density gradient centrifugation at 400 × g for 20 min
at room temperature in rat lymphocyte separation medium (TBD
Science) at a concentration of 1.091 g/ml. Isolated cells were
cultured in low-glucose DMEM (HyClone; GE Healthcare Life Sciences)
containing 15% fetal bovine serum (HyClone; GE Healthcare Life
Sciences). All cells were maintained in a 37°C incubator within an
atmosphere containing 5% CO2. BMSCs were identified by
CD44 and CD34 labeling (21). The
culture medium was regularly replaced and cells were passaged after
reaching 70–80% confluence. Third-generation BMSCs were harvested
for subsequent experiments.
Cytotoxicity assays
The cytotoxicity of BMSCs was determined using an
MTT assay. Briefly, rat BMSCs were plated into 96-well plates at a
density of 1×105 cells/well. After 24 h of culture in a
5% CO2 incubator at 37°C, adherent cells were treated
with a range of Kae concentrations (0.1, 1, 10 and 100 µM) for 24
h. The MTT reagent (10 µl; 10 mg/ml) was subsequently added to each
well, and cells were incubated for a further 4–6 h in a 5%
CO2 incubator at 37°C. The medium was removed and 100 µl
DMSO was added to each well. Plates were shaken for 10 min, and the
absorbance was measured at 492 nm using a Spectra Max Paradigm
microplatereader (Molecular Devices, LLC). Cytotoxicity was
calculated as follows: Cytotoxicity (%)=(1-absorbance of
sample/absorbance of control) ×100.
Experimental groups and
treatments
Third-passage BMSCs were randomly divided into 5
groups: i) Control group, in which BMSCs derived from normal rats
were treated with osteogenic induction medium (Cyagen Biosciences,
Inc.) to induce osteoblast differentiation; ii) OVX group, in which
BMSCs derived from OVX rats were treated in the same way as the
control group; iii) OVX + Kae group, in which BMSCs derived from
OVX rats were incubated with 0.1, 1, 10 or 100 µM Kae and treated
in the same way as the control group; iv) OVX + Kae + Rapa group,
in which BMSCs derived from OVX rats were incubated with Kae and 10
µM Rapa and then treated in the same way as the control group; v)
OVX + Rapa group, where BMSCs derived from OVX rats were incubated
with 10 µM Rapa and treated in the same way as the control group.
The dose of Rapa was determined during preliminary experiments.
Briefly, MTT was detected to evaluate the optimal concentration of
Rapa. Rapa (at 0.1, 1, 10 or 100 µM) was incubated with BMSCs. Rapa
was found to exhibit a concentration-dependent cytotoxic effect.
Therefore, 10 µM was selected. BMSCs were harvested at day 15 in
all groups.
ARS staining
ARS staining was used to assess the osteogenic
differentiation of BMSCs. Briefly, rat BMSCs were plated into
24-well plates at a density of 2.5×105 cells/well. After
induction of osteogenesis, cells were fixed with 95% alcohol for 15
min at room temperature and the BMSCs were stained with ARS for 5
min at room temperature. Mature osteoblasts that differentiated
from rBMSCs displayed intense brown-red staining after 5 min of ARS
staining (magnification, ×40). Cells were imaged at ×400
magnification by light microscopy, after the addition of 10% (w/v)
cetylpyridinium chloride to precipitate calcium ions. The
absorbance of each well was measured at 562 nm using a Spectra Max
Paradigm microplatereader (Molecular Devices, LLC).
Alkaline phosphatase activity
assay
The effects of Kae on osteoblasts were assessed
using an ALP assay kit. The absorbance/optical density (OD) of each
sample was measured at 492 nm using a microplate reader. ALP
activity in the osteogenic induction medium (Cyagen Biosciences,
Inc.) was determined as follows: ALP activity (Jinshi unit/100
ml)=(T-B)/(S-B)×0.02 mg/ml ×100 ml × a, where T=OD of the test
sample; B=OD of the blank; S=OD of the standard; and a=dilution
factor of the sample. According to the conditions above, a Jinshi
unit is defined as the ALP activity that releases of 1 mg of phenol
every 15 min after mixing with 100 ml liquid matrix at 37°C.
Western blot analysis
Cells were lysed in RIPA lysis buffer containing 50
mM Tris-HCl (pH 7.4), 150 mM NaCl, 1% sodium deoxycholate, 1%
NP-40, 1 mM PMSF and 1 mM EDTA. Then, extracts were centrifuged at
24,750 × g at 4°C for 15 min to remove insoluble material. Total
protein concentrations were determined using a bicinchoninic acid
assay (Beyotime Institute of Biotechnology), according to the
manufacturer's protocol. An equivalent quantity of total protein
(40 µg) per well was diluted in sample buffer containing 100 mM
dithiothreitol and heated to 98°C for 5 min. Lysates were separated
using 10–15% SDS-PAGE (Bio-Rad Laboratories, Inc.) and subsequently
transferred to PVDF membranes. The membranes were blocked in 5%
non-fat dry milk for 2 h at room temperature and incubated
overnight at 4°C using primary antibodies (1:500) against β-actin,
Runx2, 4E/BP 1, p-4E/BP1, S6K1 and p-S6K1. Membranes were washed in
TBS with Tween-20 (TBS-T), and incubated with horseradish
peroxidase-conjugated secondary antibody (1:2,000) at room
temperature for 1 h. Membranes were washed three times for 20 min
in TBS-T, and protein bands were visualized by enhanced
chemiluminescence (Proteintech Group, Inc.). Band intensities were
measured and quantitated using Quantity One software (version
4.6.6; Bio-Rad Laboratories, Inc.) and β-actin was used for
normalization.
Statistical analysis
SPSS (version 20.0; IBM Corp.) was used for
statistical analysis. Data are presented as the mean ± SEM from six
independent experiments. The variance homogeneity test and one-way
ANOVA were performed between groups. Newman-Keuls test was used
following ANOVA. P<0.05 was considered to indicate a
statistically significant difference.
Results
Kae ameliorates OVX-induced
osteoporosis in rats
The 3D trabecular bone microarchitecture, used for
the assessment of distal femoral metaphysis, was calculated from
micro-CT images (Fig. 2). The mean
BV/TV, Tb.N, Tb.Th and BMD values in the OVX group were
significantly lower than those of the sham group, while Tb.Sp and
SMI were higher (P<0.05). Compared with the OVX group, BV/TV,
Tb.N, Tb.Th and BMD values were significantly greater, while Tb.Sp
and SMI were lower in the OVX + Kae group (P<0.05). Thus,
treating OVX rats with Kae significantly improved bone
microarchitecture and mass. The opposite trend was observed in the
OVX + Kae + Rapa and OVX + Rapa groups: OVX rats treated with Kae +
Rapa or Rapa only exhibited lower BV/TV, Tb.N and Tb.Th values and
higher Tb.Sp and SMI values compared to the OVX + Kae group
(P<0.05 vs. OVX + Kae).
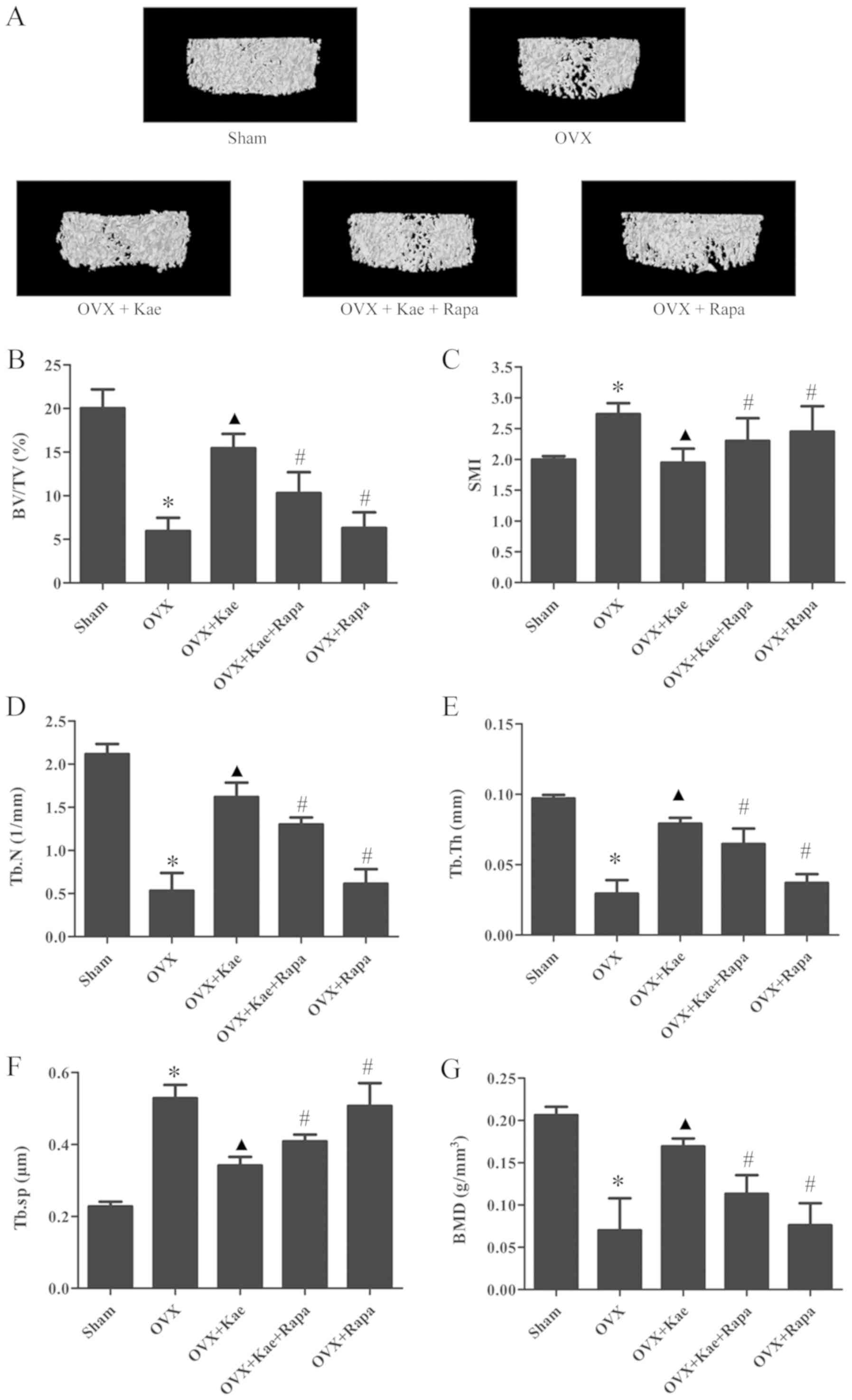 | Figure 2.Effects of Kae on bone
microarchitecture in rats. (A) Representative trabecular bone
microarchitecture of the femoral metaphysis for each group,
obtained from micro-CT images. Quantification of various parameters
of bone microarchitecture for each group: (B) BV/TV, (C) SMI, (D)
Tb.N, (E) Tb.Th, and (F) Tb.Sp. (G) BMD parameters measured using
micro-CT in each group. The data are expressed as the mean ± SEM
(n=6). *P<0.05 vs. control group; ▲P<0.05 vs. OVX
group; #P<0.05 vs. OVX + Kae group. Kae, kaempferol;
micro-CT, micro-computed tomography; BV/TV, bone volume/tissue
volume; Tb.N, trabecular number; Tb.Sp, trabecular separation,
Tb.Th, trabecular thickness; SMI, structure model index; BMD, bone
mineral density; Rapa, rapamycin; OVX, ovariectomized. |
Assessment of the optimal Kae dose
that promotes osteoblast differentiation
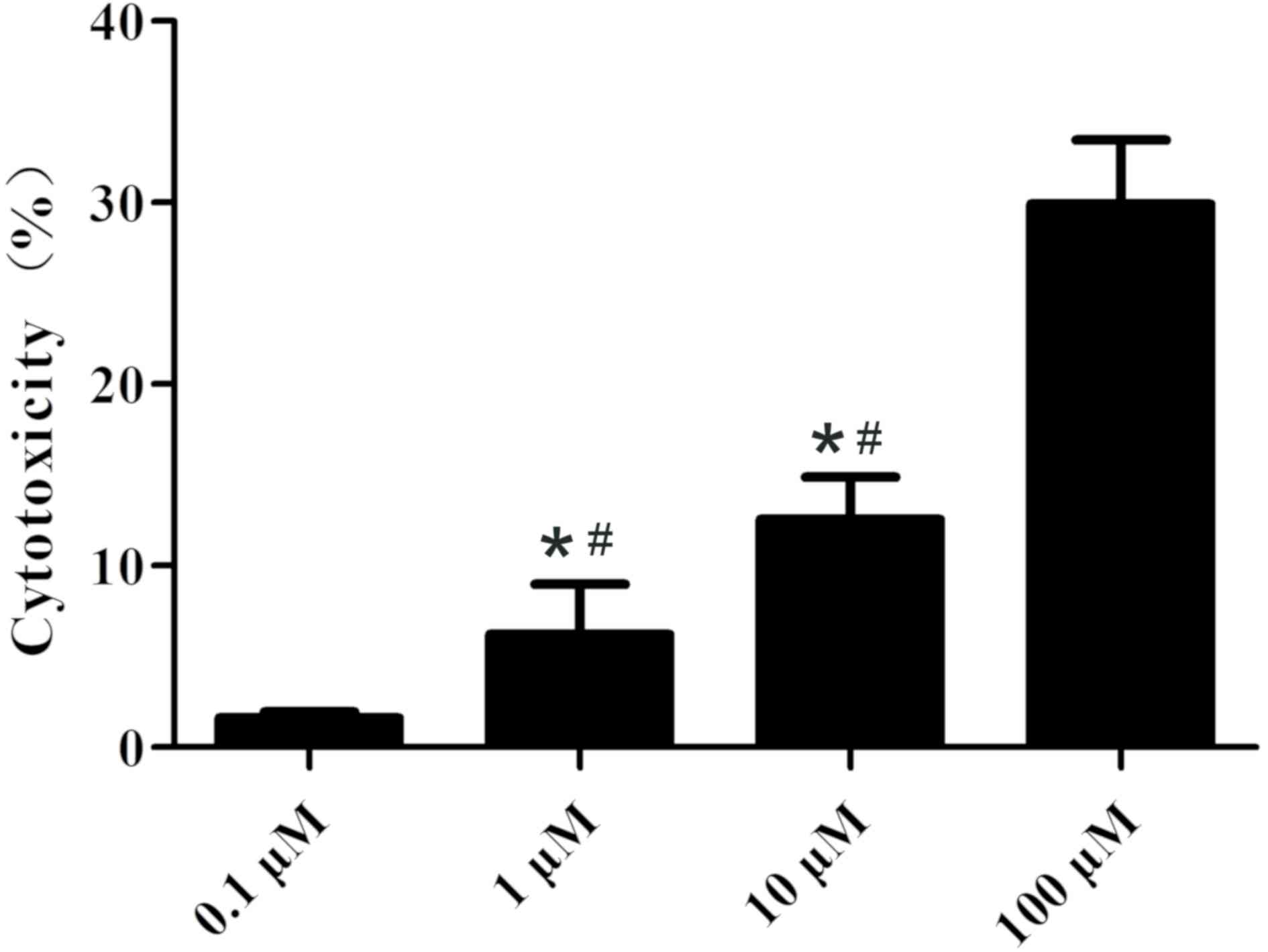
The cytotoxicity of various concentrations of Kae
was tested on BMSCs. As shown in Fig.
3, Kae displayed a concentration-dependent cytotoxic effect,
demonstrating high cytotoxicity at 100 µM. Additionally, it was
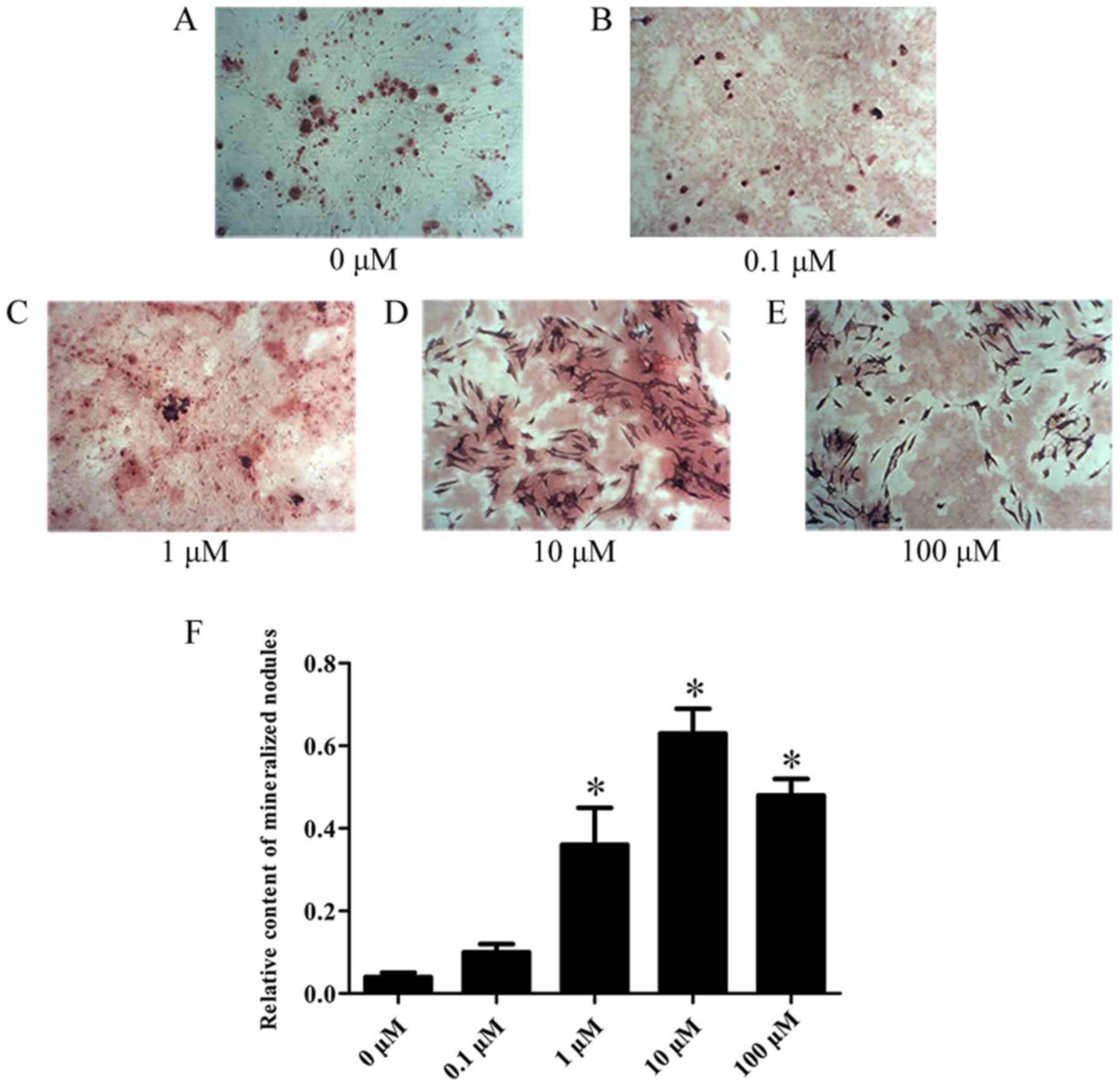
observed that Kae induced osteoblast differentiation (Fig. 4). On day 15 following osteogenic
induction, ARS staining revealed increased calcium deposition with
increasing Kae concentrations. However, the number of osteoblasts
unexpectedly decreased when cells were treated with 100 µM Kae.
From the range of concentrations tested, the optimal concentration
of Kae that promoted osteoblast differentiation was 10 µM. This
concentration was therefore used in subsequent experiments.
Kae promotes osteogenic
differentiation
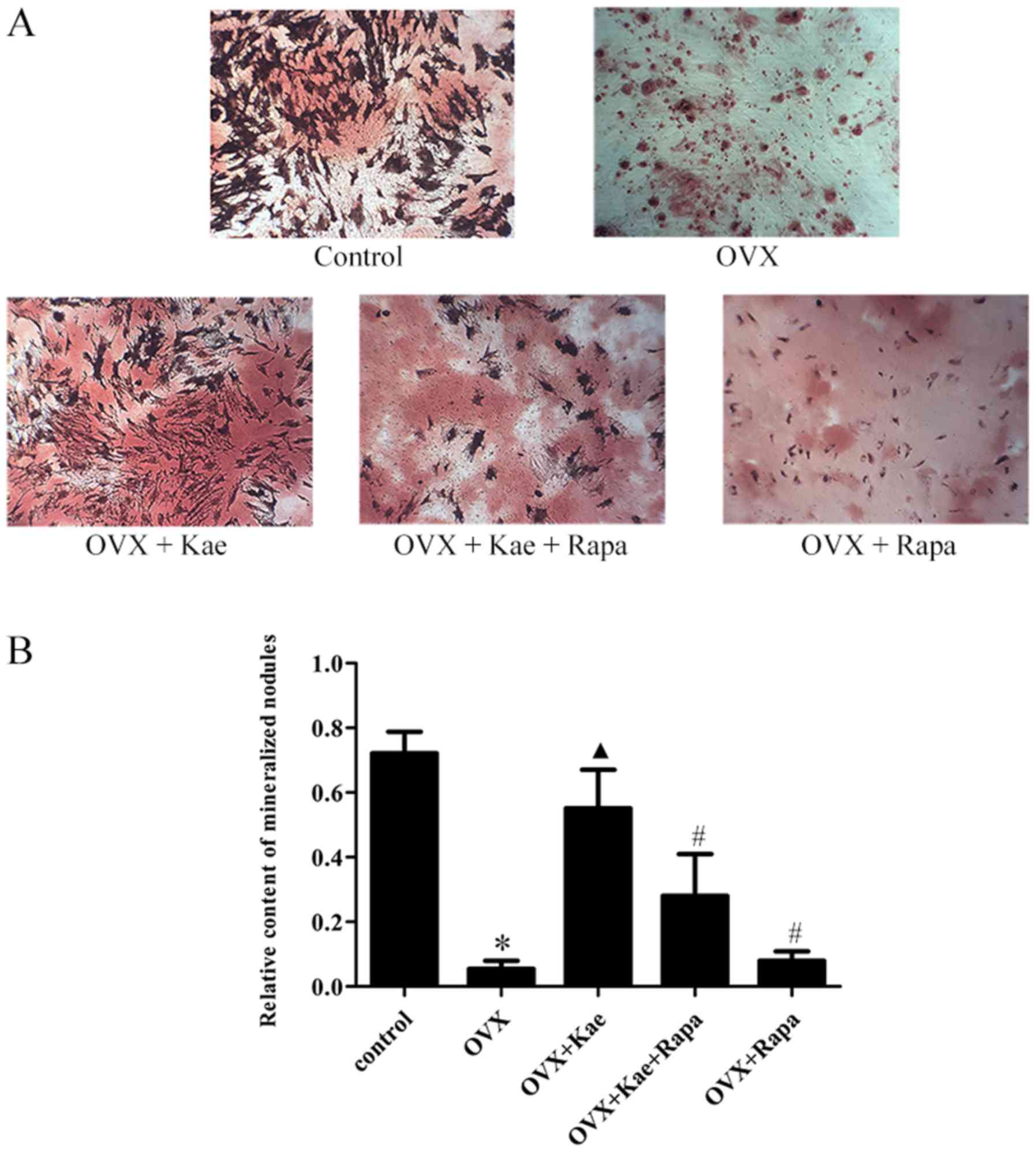
ARS staining was used to assess the induction of
osteogenesis in BMSCs. As illustrated in Fig. 5, following the induction of
osteogenic differentiation, the rate of calcific nodule formation
decreased in the OVX group compared to the control group
(P<0.05). Moreover, treatment with Kae significantly increased
the number of calcified nodules in the OVX + Kae group (P<0.05
vs. OVX group). However, the protective effect of Kae on
osteoblasts was antagonized by the co-administration of Rapa
(P<0.05 vs. OVX + Kae).
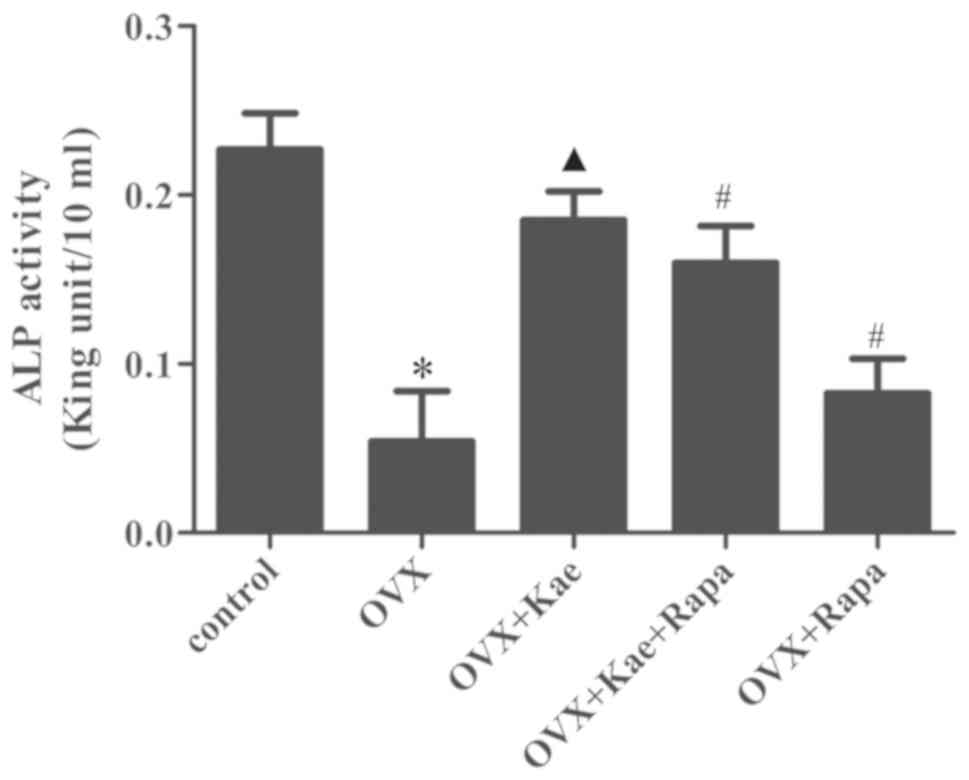
Kae increases ALP expression in
BMSCs
ALP activity was measured during osteogenesis. As
shown in Fig. 6, the expression of
ALP in the OVX group was significantly reduced compared with the
control group (P<0.05). In addition, ALP activity was
significantly higher in the OVX + Kae group compared to the OVX
group (P<0.05 vs. OVX group). Pretreatment with Kae + Rapa or
with Rapa alone led to reduced levels of ALP activity compared to
Kae treatment alone (P<0.05).
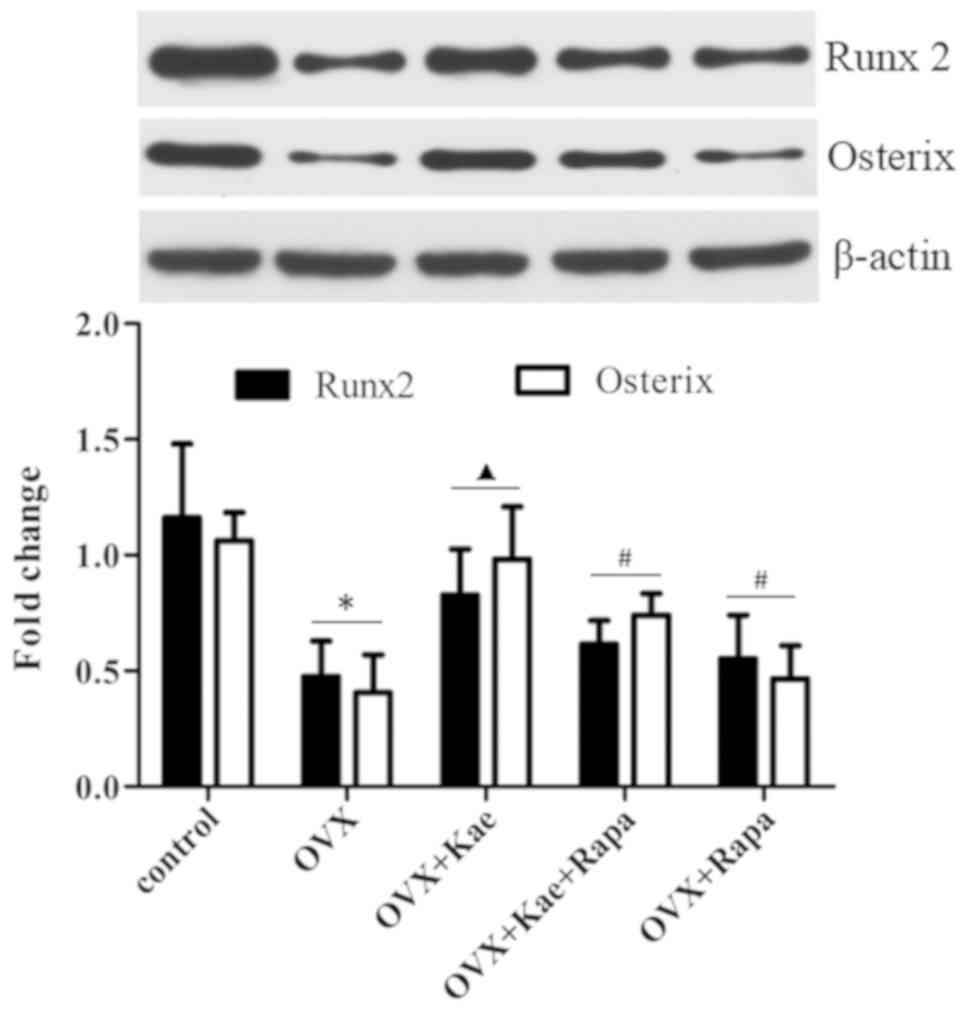
Kae upregulates Runx2 and Osterix
expression
Western blot analysis was used to assess the
expression of the osteogenesis-related transcription factors Runx2
and Osterix during osteogenesis. Fig.
7 demonstrates that a significant downregulation of Runx2 and
Osterix expression occurred in the OVX group compared with the
control group (P<0.05). Kae significantly increased the
expression of Runx2 and Osterix compared with the OVX group
(P<0.05). However, their expression was significantly lower in
the OVX + Kae + Rapa and OVX + Rapa groups compared with the OVX +
Kae group (P<0.05).
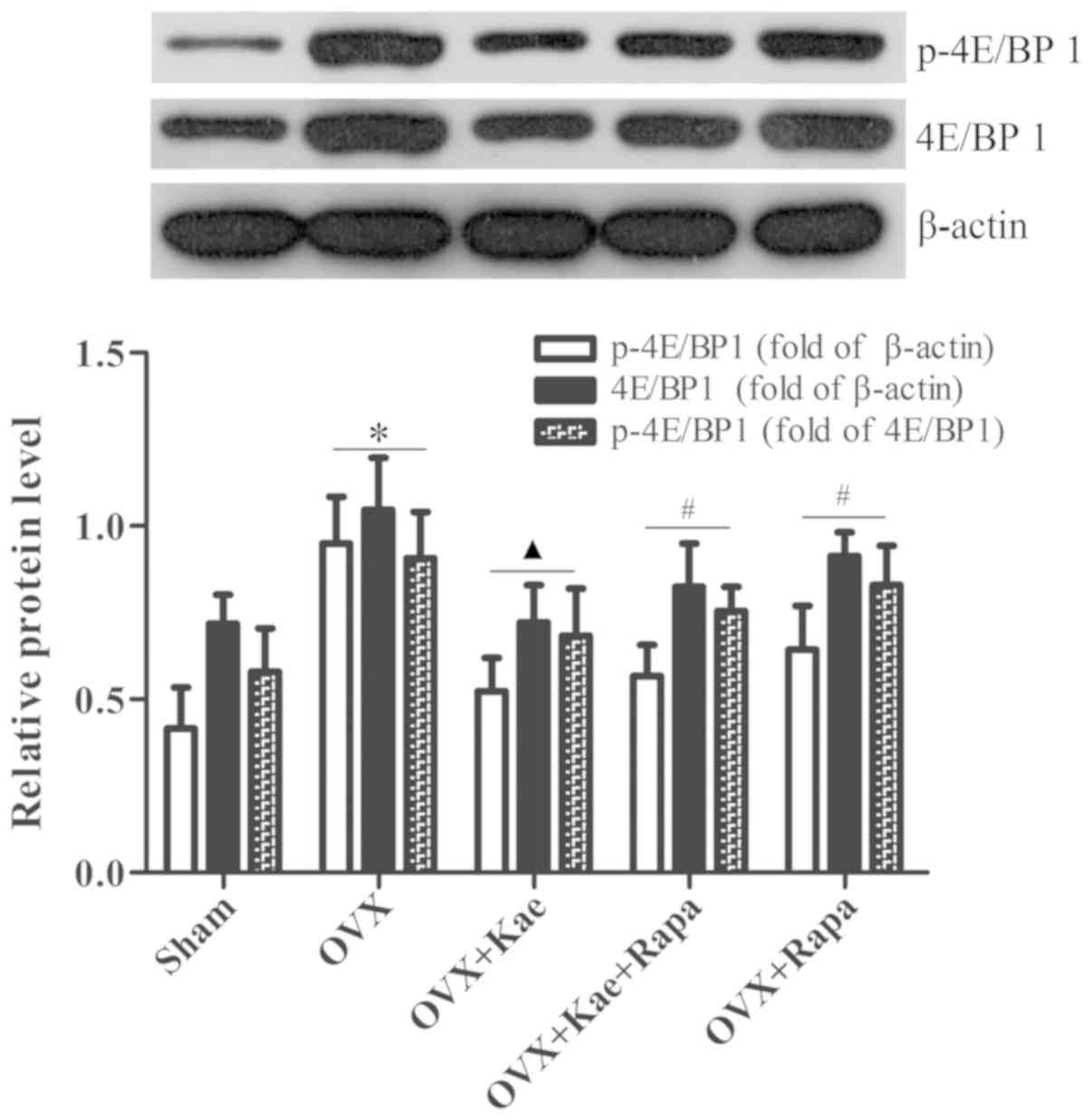
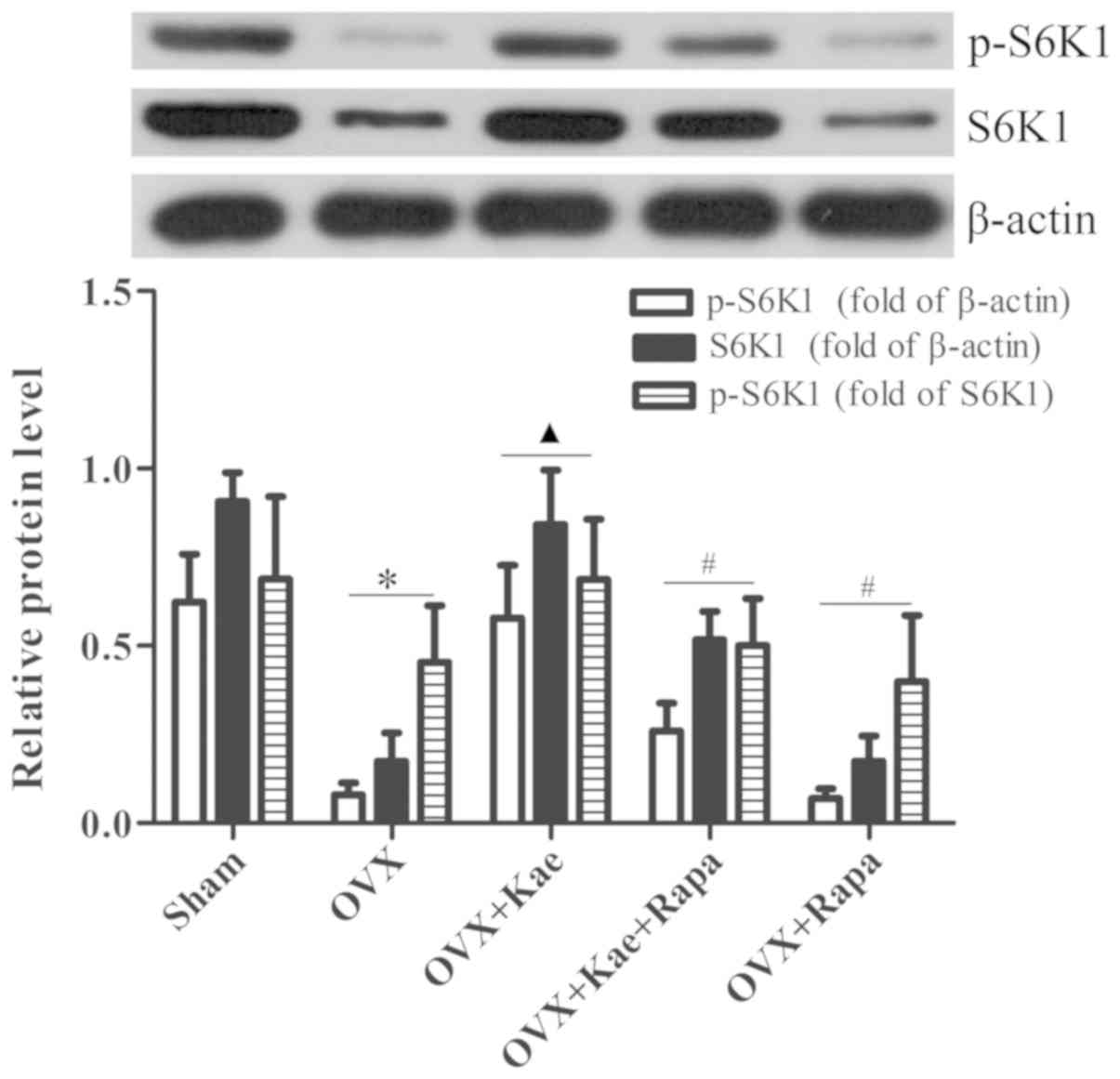
Kae alters the phosphorylation of
4E/BP1 and S6K1
Western blot analysis was used to determine the
levels of phosphorylated and total 4E/BP1 and S6K1, important
downstream regulators of the mTOR pathway. As shown in Fig. 8, the levels of p-4E/BP1 in the OVX
+ Kae group were significantly lower than in the OVX group
(P<0.05). However, an notably increased level of 4E/BP1 p-4EB/P1
was observed in the OVX + Kae + Rapa and OVX + Rapa groups. In
addition, Kae increased S6K1 and p-S6K1 levels (Fig. 9; P<0.05 vs. OVX group).
Conversely, the levels of S6K1 and p-S6K1 were significantly lower
in the OVX + Kae + Rapa and OVX + Rapa groups compared with the OVX
+ Kae group (P<0.05). Taken together, these results indicated
that Kae may promote osteogenesis through mTOR signaling as 4E/BP1
was activated by mTOR, whereas S6K1 exhibited the opposite
trend.
Discussion
Estrogen has a significant role in bone metabolism
(22), inhibiting bone resorption
and stimulating bone formation (23). Flavonoids are estrogen-like
chemicals used as estrogen substitutes, which may exert protective
effects against postmenopausal bone loss (11,24).
In addition, Kae is a phytoestrogen that possesses osteogenic
properties (6). Guo et al
(25) demonstrated that Kae
stimulates osteogenic differentiation in cultured osteoblasts
through estrogen receptor signaling. In addition, evidence suggests
that Kae has a positive effect on bone metabolism, since it was
observed to promote osteogenesis and to inhibit osteoclast
activity, adipocyte formation and autophagy (26–30).
From the in vivo assessment of bone microarchitecture, the
present study verified that the bone mass parameters (BMD, BV/TV,
Tb.N and Tb.Th) were significantly elevated, and the parameters of
osteoporosis (SMI and Tb.Sp) reduced in OVX rats treated with Kae
compared with OVX group rats. These changes in bone morphology
induced by Kae in OVX rats are similar to those found in previously
published research (26,27). However, the concentration of Kae
used in the in vivo experiment of the present study was
higher than that of previous studies (26,27).
The similarity of the results may be associated with differences in
delivery time or the vehicle solution used in the present study.
Further research is required to effectively assess the effect of
different doses of Kae on bone metabolism. Additionally, when BMSCs
were isolated from OVX rats and osteogenesis was induced in
vitro, Kae was found to promote osteogenic differentiation and
increase ALP activity, consistent with Kim et al (30), although their data suggest that Kae
stimulates osteogenic differentiation through increased expression
of autophagy-related factors. In addition, Trivedi et al
(26) also reported that treatment
with Kae led to a reduction in adipogenic differentiation of BMCs
in a Kae + OVX group, verified by a significant reduction in
adipocyte number compared with an OVX group using Oil Red O
staining. Both osteoblasts and adipocytes share a common precursor
(MSCs), and the commitment to either fate is maintained in a
dynamic balance under physiological conditions, serving a critical
role in the microenvironment of the bone marrow (31–33).
However, the imbalance between adipogenesis and osteogenesis has
been linked to a number of pathophysiological processes, such as
osteoporosis, osteopenia, obesity and aging (31–33).
The observations of the present study lend support to these
previously published data. Kae principally exerts its osteogenic
function via promotion of bone formation, but whether it functions
via inhibition of adipogenesis requires further investigation.
Together, the in vitro and in vivo experiments
suggest that Kae prevented OVX-induced bone loss by promotion of
osteoblast function. To determine the molecular mechanism of
Kae-dependent osteogenic differentiation, an assessment of the
activation of different signaling pathways was performed.
Runx-2, also termed core binding factor α-1,
activates and initiates the differentiation of BMSCs into
osteoblasts and regulates osteoblast maturation (34). Runx-2 is a key transcription factor
during osteoblast differentiation and serves a crucial role in bone
formation and reconstruction (35). Osterix, a zinc finger-containing
transcription factor required for osteoblast differentiation, was
first discovered by Nakashima et al in 2002 (36). A study revealed that the silencing
of mouse Osterix induces chondrogenesis and chondrocyte
differentiation; in addition, bone formation is impaired but Runx2
expression is unaffected (37).
Osterix was not found to be expressed in Runx2-deficient mice,
suggesting that it acts downstream of Runx2 during osteoblast
differentiation (37). Thus, both
Runx2 and Osterix are master transcription factors that promote
osteoblast differentiation and bone formation. In the present
study, western blot analysis was used to detect the expression
levels of Runx2 and Osterix in BMSCs after the induction of
osteogenesis. Kae was found to significantly upregulate Runx2 and
Osterix expression. This suggested that the effect of Kae on
osteogenic differentiation is mediated via Runx2 and Osterix.
mTOR is a member of the phosphatidylinositol
3-kinase-related kinase family of protein kinases (12). As a core component of intracellular
signaling, mTOR regulates cell growth, proliferation and survival,
protein synthesis, autophagy and transcription through two
functionally and structurally distinct multi-component complexes,
mTORC1 and mTORC2 (13). mTORC1
consists of mTOR, regulatory-associated protein of mTOR (Raptor)
and G protein β-unit-like protein (38). mTORC1 promotes the phosphorylation
of S6K1, but inhibits the phosphorylation of 4E/BP1 (39,40).
Activation of mTORC1 improves growth factor metabolism, cell growth
and proliferation (39,40). mTORC2 consists of mTOR, mTOR
associated protein, LST8 homolog, Raptor independent companion of
mTOR complex 2 and mSIN1, phosphorylating and activating Akt
(phosphorylation site, S473) and glycogen synthase kinase
phosphorylation (41,42).
Studies have demonstrated that mTOR signaling has
numerous key regulatory functions in various diseases, including
cancer (43–46), infectious diseases (47,48),
atherosclerosis, and degenerative and autoimmune diseases (49–51),
and has been suggested to be a novel therapeutic target for the
treatment of these diseases (43–51).
Other studies have shown that mTOR signaling is also involved in
the regulation of multiple aspects of skeletal development
(52–54). However, controversy remains as to
whether mTOR promotes (17,18,55,56)
or inhibits (56–59) osteogenic differentiation. A recent
study indicated that knockdown of DEP domain containing MTOR
interacting protein (Deptor) promotes BMSC osteogenesis both in
vitro and in vivo, with higher expression of Deptor
contributing to the progression of osteoporosis (56). It is well known that overexpression
of Deptor downregulates the activity of mTORC1 and mTORC2,
indicating that mTOR may play a crucial role during osteogenesis.
Similarly, a previous study demonstrated that treatment with Rapa
attenuated the expression of ALP, downregulated the expression and
phosphorylation of S6K1 and inhibited osteoblastic differentiation
in vascular smooth muscle cells (VSMCs), suggesting that mTOR
participates in the osteoblastic differentiation of VSMCs (18). In the present study, Kae was found
to increase the expression of important downstream regulatory
factors in the mTOR pathway, which also indicates that mTOR
signaling may be involved in osteogenesis. However, a number of
researchers have published contrary results. For instance, Dai
et al (59,60) indicated that deletion of Raptor (an
indispensable component of mTORC1) in osteoclasts leads to
increased bone volume and decreased bone resorption. In other
words, mTORC1 is critical for osteoclast formation. Raptor
deficiency in osteoclasts impairs bone resorption without affecting
bone formation, ultimately leading to increased bone mass in mice.
On the other hand, Xu et al (58) reported that the activation of
mTORC1 in B lymphocytes promotes osteoclast formation and causes
osteoporosis in mice by promoting TNF superfamily member 11
expression with negative regulation of Akt and β-catenin. A
separate study by Wang et al (61) also indicated that suppression of
the mTOR signaling pathway by rapamycin was able to promote BMSC
differentiation into osteoblasts in the context of degenerative
scoliosis. These conflicting results may be attributed to different
methodologies (different cell lines, drug concentrations or
experimental design), and underline the requirement for further
research. Thus, for further validation, BMSCs treated with Rapa, a
specific inhibitor of mTOR, exhibited a marked decline in
osteogenic activity and decreased Runx2 and Osterix expression.
This treatment also reversed the positive effects of Kae exposure.
Similarly, a number of studies have revealed that Kae can exert
cytoprotective effects via regulation of mTOR signaling in
different cell lines (62–64). Taken together, these data support
the hypothesis that the mTOR-Runx2/Osterix signaling axis is
associated with the osteogenic effect of Kae in OVX rats and
respective BMSCs.
In summary, the present study demonstrated that Kae
has a positive effect on bone formation and osteogenesis in OVX
rats, which is possibly exerted via mTOR-Runx2/Osterix signaling.
Since Kae naturally occurs in a variety of foods, it is likely that
it may exert beneficial effects as a food supplement by improving
postmenopausal osteoporosis. Further large scale randomized in
vivo trials are now required to confirm the potential of Kae in
the treating osteoporosis.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Traditional
Chinese Medicine Project of the Health and Family Planning
Commission of Jiangxi Province, China (grant no. 2017A274) and the
Science and Technology Program of Health and Family Planning
Commission of Jiangxi Province, China (grant no. 20185232).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JL conceived and designed the present study and
revised the manuscript for important intellectual content. JZ was
involved in all experiments and was a major contributor in writing
the manuscript. JW modified the study design and designed the
structure of the article. YL performed the experiments, analyzed
the data and wrote the manuscript. JM and XL completed the data
analysis. BX and ZY performed the cell culture, ARS staining, ALP
activity assay, western blotting. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Experiments were performed according to the Guide
for the Care and Use of Laboratory (National Institutes of Health),
and were approved by the Ethics Committee of Nanchang University
(no. 2017-0122).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
Kae
|
kaempferol
|
|
BMSCs
|
bone mesenchymal stem cells
|
|
mTOR
|
mammalian target of rapamycin
|
|
OVX
|
ovariectomized
|
|
Rapa
|
rapamycin
|
References
|
1
|
Cosman F, de Beur SJ, LeBoff MS, Lewiecki
EM, Tanner B, Randall S and Lindsay R; National Osteoporosis
Foundation, : Clinician's guide to prevention and treatment of
osteoporosis. Osteoporos Int. 25:2359–2381. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Das S and Crockett JC: Osteoporosis-a
current view of pharmacological prevention and treatment. Drug Des
Devel Ther. 7:435–448. 2013.PubMed/NCBI
|
|
3
|
Rizzoli R, Reginster JY, Boonen S, Bréart
G, Diez-Perez A, Felsenberg D, Kaufman JM, Kanis JA and Cooper C:
Adverse reactions and drug-drug interactions in the management of
women with postmenopausal osteoporosis. Calcif Tissue Int.
89:91–104. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Adler RA, El-Hajj Fuleihan G, Bauer DC,
Camacho PM, Clarke BL, Clines GA, Compston JE, Drake MT, Edwards
BJ, Favus MJ, et al: Managing osteoporosis in patients on long-term
bisphosphonate treatment: Report of a task force of the American
society for bone and mineral research. J Bone Miner Res. 31:16–35.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Drieling RL, LaCroix AZ, Beresford SAA,
Boudreau DM, Kooperberg C, Chlebowski RT, Ko MG and Heckbert SR:
Long-Term oral bisphosphonate therapy and fractures in older women:
The Women's health initiative. J Am Geriatr Soc. 65:1924–1931.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Huang L, Yagura T and Chen S: Sedative
activity of hexane extract of Keampferia galangal L. and its
active compounds. J Ethnopharmacol. 120:123–125. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Devi KP, Malar DS, Nabavi SF, Sureda A,
Xiao J, Nabavi SM and Daglia M: Kaempferol and inflammation: From
chemistry to medicine. Pharmacol Res. 99:1–10. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Qin Y, Cui W, Yang X and Tong B:
Kaempferol inhibits the growth and metastasis of cholangiocarcinoma
in vitro and in vivo. Acta Biochim Biophys Sin (Shanghai).
48:238–245. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Suchal K, Malik S, Khan SI, Malhotra RK,
Goyal S, Bhatia J, Ojha S and Arya D: Molecular pathways involved
in the amelioration of myocardial injury in diabetic rats by
kaempferol. Int J Mol Sci. 18(pii): E10012017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
An M and Kim M: Protective effects of
kaempferol against cardiac sinus node dysfunction via CaMKII
deoxidization. Anat Cell Biol. 48:235–243. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mühlbauer RC and Li F: Effect of
vegetables on bone metabolism. Nature. 401:343–344. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mitra A, Luna JI, Marusina AI, Merleev A,
Kundu-Raychaudhuri S, Fiorentino D, Raychaudhuri SP and Maverakis
E: Dual mTOR inhibition is required to prevent TGF-β-mediated
fibrosis: Implications for scleroderma. J Invest Dermatol.
135:2873–2876. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Conciatori F, Ciuffreda L, Bazzichetto C,
Falcone I, Pilotto S, Bria E, Cognetti F and Milella M: mTOR
Cross-talk in cancer and potential for combination therapy. Cancers
(Basel). 10(pii): E232018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Karner CM, Lee SY and Long F: Bmp induces
osteoblast differentiation through both Smad4 and mTORC1 signaling.
Mol Cell Biol. 37(pii): e00253–16. 2017.PubMed/NCBI
|
|
15
|
Fitter S, Matthews MP, Martin SK, Xie J,
Ooi SS, Walkley CR, Codrington JD, Ruegg MA, Hall MN, Proud CG, et
al: mTORC1 Plays an important role in skeletal development by
controlling preosteoblast differentiation. Mol Cell Biol. 37(pii):
e00668–16. 2017.PubMed/NCBI
|
|
16
|
Liu DM, Zhao L, Liu TT, Jiao PL, Zhao DD,
Shih MS, Tao B, Sun LH, Zhao HY and Liu JM: Rictor/mTORC2 loss in
osteoblasts impairs bone mass and strength. Bone. 90:50–58. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen J, Tu X, Esen E, Joeng KS, Lin C,
Arbeit JM, Rüegg MA, Hall MN, Ma L and Long F: WNT7B promotes bone
formation in part through mTORC1. PLoS Genet. 10:e10041452014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhan JK, Wang YJ, Wang Y, Wang S, Tan P,
Huang W and Liu YS: The mammalian target of rapamycin signalling
pathway is involved in osteoblastic differentiation of vascular
smooth muscle cells. Can J Cardiol. 30:568–575. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Min J, Yuan Z, Zhang Q, Lin S, Wang K and
Luo J: Analysis of anti-osteoporosis function of chlorogenic acid
by gene microarray profiling in ovariectomy rat model. Biosci Rep.
38(pii): BSR201807752018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kilkenny C, Browne WJ, Cuthill IC, Emerson
M and Altman DG: Improving bioscience research reporting: The
ARRIVE guidelines for reporting animal research. PLoS Biol.
8:e10004122010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jiang Y, Jahagirdar BN, Reinhardt RL,
Schwartz RE, Keene CD, Ortiz-Gonzalez XR, Reyes M, Lenvik T, Lund
T, Blackstad M, et al: Pluripotency of mesenchymal stem cells
derived from adult marrow. Nature. 418:41–49. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Riggs BL, Khosla S and Melton LJ III: Sex
steroids and the construction and conservation of the adult
skeleton. Endocr Rev. 23:279–302. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chien KR and Karsenty G: Longevity and
lineages: Toward the integrative biology of degenerative diseases
in heart, muscle, and bone. Cell. 120:533–544. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hegarty VM, May HM and Khaw KT: Tea
drinking and bone mineral density in older women. Am J Clin Nutr.
71:1003–1007. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Guo AJ, Choi RC, Zheng KY, Chen VP, Dong
TT, Wang ZT, Vollmer G, Lau DT and Tsim KW: Kaempferol as a
flavonoid induces osteoblastic differentiation via estrogen
receptor signaling. Chin Med. 7:102012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Trivedi R, Kumar S, Kumar A, Siddiqui JA,
Swarnkar G, Gupta V, Kendurker A, Dwivedi AK, Romero JR and
Chattopadhyay N: Kaempferol has osteogenic effect in ovariectomized
adult Sprague-Dawley rats. Mol Cell Endocrinol. 289:85–93. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nowak B, Matuszewska A, Nikodem A,
Filipiak J, Landwójtowicz M, Sadanowicz E, Jędrzejuk D, Rzeszutko
M, Zduniak K, Piasecki T, et al: Oral administration of kaempferol
inhibits bone loss in rat model of ovariectomy-induced osteopenia.
Pharmacol Rep. 69:1113–1119. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim CJ, Shin SH, Kim BJ, Kim CH, Kim JH,
Kang HM, Park BS and Kim IR: The effects of Kaempferol-inhibited
autophagy on osteoclast formation. Int J Mol Sci. 19(pii):
E1252018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Adhikary S, Choudhary D, Ahmad N, Karvande
A, Kumar A, Banala VT, Mishra PR and Trivedi R: Dietary flavonoid
kaempferol inhibits glucocorticoid-induced bone loss by promoting
osteoblast survival. Nutrition. 53:64–76. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kim IR, Kim SE, Baek HS, Kim BJ, Kim CH,
Chung IK, Park BS and Shin SH: The role of kaempferol-induced
autophagy on differentiation and mineralization of osteoblastic
MC3T3-E1 cells. BMC Complement Altern Med. 16:3332016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Chen Q, Shou P, Zheng C, Jiang M, Cao G,
Yang Q, Cao J, Xie N, Velletri T, Zhang X, et al: Fate decision of
mesenchymal stem cells: Adipocytes or osteoblasts? Cell Death
Differ. 23:1128–1139. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Eriksen EF: Cellular mechanisms of bone
remodeling. Rev Endocr Metab Disord. 11:219–227. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zuk PA, Zhu M, Ashjian P, De Ugarte DA,
Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P and Hedrick
MH: Human adipose tissue is a source of multipotent stem cells. Mol
Biol Cell. 13:4279–4295. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lucero CM, Vega OA, Osorio MM, Tapia JC,
Antonelli M, Stein GS, van Wijnen AJ and Galindo MA: The
cancer-related transcription factor Runx2 modulates cell
proliferation in human osteosarcoma cell lines. J Cell Physiol.
228:714–723. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wysokinski D, Pawlowska E and Blasiak J:
RUNX2: A master bone growth regulator that may be involved in the
DNA damage response. DNA Cell Biol. 34:305–315. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Nakashima K, Zhou X, Kunkel G, Zhang Z,
Deng JM, Behringer RR and de Crombrugghe B: The novel zinc
finger-containing transcription factor osterix is required for
osteoblast differentiation and bone formation. Cell. 108:17–29.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Nishimura R, Wakabayashi M, Hata K,
Matsubara T, Honma S, Wakisaka S, Kiyonari H, Shioi G, Yamaguchi A,
Tsumaki N, et al: Osterix regulates calcification and degradation
of chondrogenic matrices through matrix metalloproteinase 13
(MMP13) expression in association with transcription factor Runx2
during endochondral ossification. J Biol Chem. 287:33179–33190.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zoncu R, Efeyan A and Sabatini DM: mTOR:
From growth signal integration to cancer, diabetes and ageing. Nat
Rev Mol Cell Biol. 12:21–35. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Howell JJ and Manning BD: mTOR couples
cellular nutrient sensing to organismal metabolic homeostasis.
Trends Endocrinol Metab. 22:94–102. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Dai N, Rapley J, Angel M, Yanik MF, Blower
MD and Avruch J: mTOR phosphorylates IMP2 to promote IGF2 mRNA
translation by internal ribosomal entry. Genes Dev. 25:1159–1172.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Cybulski N and Hall MN: TOR complex 2: A
signaling pathway of its own. Trends Biochem Sci. 34:620–627. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Alessi DR, Pearce LR and García-Martínez
JM: New insights into mTOR signaling: MTORC2 and beyond. Sci
Signal. 2:pe272009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Steelman LS, Martelli AM, Cocco L, Libra
M, Nicoletti F, Abrams SL and McCubrey JA: The therapeutic
potential of mTOR inhibitors in breast cancer. Br J Clin Pharmacol.
82:1189–1212. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu ZG, Tang J, Chen Z and Zhang H, Wang
H, Yang J and Zhang H: The novel mTORC1/2 dual inhibitor INK128
enhances radiosensitivity of breast cancer cell line MCF-7. Int J
Oncol. 49:1039–1045. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ling S, Song L, Fan N, Feng T, Liu L, Yang
X, Wang M, Li Y, Tian Y, Zhao F, et al: Combination of metformin
and sorafenib suppresses proliferation and induces autophagy of
hepatocellular carcinoma via targeting the mTOR pathway. Int J
Oncol. 50:297–309. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Guo H, Xu Y, Wang F, Shen Z, Tuo X, Qian
H, Wang H and Wang K: Clinical associations between ASCT2 and
p-mTOR in the pathogenesis and prognosis of epithelial ovarian
cancer. Oncol Rep. 40:3725–3733. 2018.PubMed/NCBI
|
|
47
|
Nicoletti F, Fagone P, Meroni P, McCubrey
J and Bendtzen K: mTOR as a multifunctional therapeutic target in
HIV infection. Drug Discov Today. 16:715–721. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Donia M, McCubrey JA, Bendtzen K and
Nicoletti F: Potential use of rapamycin in HIV infection. Br J Clin
Pharmacol. 70:784–793. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mammana S, Bramanti P, Mazzon E, Cavalli
E, Basile MS, Fagone P, Petralia MC, McCubrey JA, Nicoletti F and
Mangano K: Preclinical evaluation of the PI3K/Akt/mTOR pathway in
animal models of multiple sclerosis. Oncotarget. 9:8263–8277. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Donia M, Mangano K, Amoroso A, Mazzarino
MC, Imbesi R, Castrogiovanni P, Coco M, Meroni P and Nicoletti F:
Treatment with rapamycin ameliorates clinical and histological
signs of protracted relapsing experimental allergic
encephalomyelitis in Dark Agouti rats and induces expansion of
peripheral CD4+CD25+Foxp3+ regulatory T cells. J Autoimmun.
33:135–140. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Oaks Z, Winans T, Huang N, Banki K and
Perl A: Activation of the mechanistic target of Rapamycin in SLE:
Explosion of evidence in the last five years. Curr Rheumatol Rep.
18:732016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Chen J and Long F: mTOR signaling in
skeletal development and disease. Bone Res. 6:12018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Wu H, Wu Z, Li P, Cong Q, Chen R, Xu W,
Biswas S, Liu H, Xia X, Li S, et al: Bone size and quality
regulation: Concerted actions of mTOR in mesenchymal stromal cells
and osteoclasts. Stem Cell Reports. 8:1600–1616. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Chen C, Akiyama K, Wang D, Xu X, Li B,
Moshaverinia A, Brombacher F, Sun L and Shi S: mTOR inhibition
rescues osteopenia in mice with systemic sclerosis. J Exp Med.
212:73–91. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Li SF, Tang JJ, Chen J, Zhang P, Wang T,
Chen TY, Yan B, Huang B, Wang L, Huang MJ, et al: Regulation of
bone formation by baicalein via the mTORC1 pathway. Drug Des Devel
Ther. 9:5169–5183. 2015.PubMed/NCBI
|
|
56
|
Chen S, Jia L, Zhang S, Zheng Y and Zhou
Y: DEPTOR regulates osteogenic differentiation via inhibiting
MEG3-mediated activation of BMP4 signaling and is involved in
osteoporosis. Stem Cell Res Ther. 9:1852018. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Martin SK, Fitter S, Bong LF, Drew JJ,
Gronthos S, Shepherd PR and Zannettino AC: NVP-BEZ235, a dual pan
class I PI3 kinase and mTOR inhibitor, promotes osteogenic
differentiation in human mesenchymal stromal cells. J Bone Miner
Res. 25:2126–2137. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Xu S, Zhang Y, Liu B, Li K, Huang B, Yan
B, Zhang Z, Liang K, Jia C, Lin J, et al: Activation of mTORC1 in B
lymphocytes promotes osteoclast formation via regulation of
β-catenin and RANKL/OPG. J Bone Miner Res. 31:1320–1333. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Dai Q, Han Y, Xie F, Ma X, Xu Z, Liu X,
Zou W and Wang J: A RANKL-based osteoclast culture assay of mouse
bone marrow to investigate the role of mTORC1 in osteoclast
formation. J Vis Exp. Mar 15–2018.doi: 10.3791/56468. View Article : Google Scholar
|
|
60
|
Dai Q, Xie F, Han Y, Ma X, Zhou S, Jiang
L, Zou W and Wang J: Inactivation of Regulatory-associated protein
of mTOR (Raptor)/mammalian target of rapamycin complex 1 (mTORC1)
signaling in osteoclasts increases bone mass by inhibiting
osteoclast differentiation in mice. J Biol Chem. 292:196–204. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wang Y, Yi XD and Li CD: Suppression of
mTOR signaling pathway promotes bone marrow mesenchymal stem cells
differentiation into osteoblast in degenerative scoliosis: In vivo
and in vitro. Mol Biol Rep. 44:129–137. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Che J, Liang B, Zhang Y, Wang Y, Tang J
and Shi G: Kaempferol alleviates ox-LDL-induced apoptosis by
up-regulation of autophagy via inhibiting PI3K/Akt/mTOR pathway in
human endothelial cells. Cardiovasc Pathol. 31:57–62. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Varshney R, Gupta S and Roy P:
Cytoprotective effect of kaempferol against palmitic acid-induced
pancreatic β-cell death through modulation of autophagy via
AMPK/mTOR signaling pathway. Mol Cell Endocrinol. 448:1–20. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Kim GD: Kaempferol inhibits angiogenesis
by suppressing HIF-1α and VEGFR2 Activation via ERK/p38 MAPK and
PI3K/Akt/mTOR signaling pathways in endothelial cells. Prev Nutr
Food Sci. 22:320–326. 2017. View Article : Google Scholar : PubMed/NCBI
|