Introduction
Autologous adipocyte transplantation is increasingly
applied in plastic surgery because of its numerous advantages
(1). Autologous adipocytes were
initially used as filling material to permanently maintain
morphological features after successful transplantation; the
morphology of such tissues is natural and resembles that of normal
tissue. Liposuction is a simple surgical method that can easily
yield large amounts of adipose tissue from numerous body regions to
satisfy various tissue requirements (2). Furthermore, because the tissue is
derived from the patient, immune rejection, allergic reactions and
tissue toxicity exhibited towards some artificial materials do not
occur (3). In addition, in the
long term, adipose tissue is a relatively safe tissue-filling
substance (4). However, after
autologous fat transplantation, autologous tissue can become
necrotic and is reabsorbed over time. The survival rate of
autologous adipocytes is only 20–70% of the intraoperative filling
volume (4,5). The reduction in tissue volume often
leads to depressions, asymmetric morphology and even failure of the
surgical filling. To achieve the desired filling effects, the
patient may need to undergo multiple operations.
Mechanical damage to the adipose tissue that has
been extracted can occur during liposuction, which results in
necrosis and loss of the transplanted tissue. The lack of vascular
reconstruction in the transplanted adipose tissue also affects the
survival rate of adipocytes (6,7).
Billings et al (8) first
reported that ‘fibroblast-like’ mesenchymal cells in the adipose
tissue may facilitate adipose tissue survival. Adipose-derived stem
cells (ADSCs) are mesodermal cells derived from adipose tissues.
Their morphology is fusiform, and they can differentiate into
adipose, bone, cartilage, muscle, epithelial, neuronal and other
tissue types. Previous studies investigating the use of stem cell
transplantation to treat tissue injury have reported that stem
cells promote tissue repair (2,3,9).
Furthermore, an increasing number of studies have reported that the
paracrine signaling from stem cells is an important mechanism that
promotes various physiological activities in cells (10,11).
Various cellular factors secreted by ADSCs are involved in forming
and regenerating blood vessels, and significantly improve the
survival rate of adipocytes (12,13).
Studies also reported that increasing the number of ADSCs in
adipose tissue can significantly promote the survival rate of
adipose tissue (14). Abundant
ADSCs can be obtained during liposuction; however, mechanical
damage during adipose tissue extraction, ischemia and hypoxia in
the tissue, cell rupture and death, and the release of numerous
reactive oxygen species can lead to oxidative stress and finally
apoptosis (15). Understanding the
mechanism underlying the induction of apoptosis of ADSCs may lead
to the development of methods for decreasing their apoptotic rate
and increasing their numbers in tissues, which would help to
improve the survival rate of transplanted adipose tissue.
Maternally expressed gene 3 (MEG3) is a long
non-coding RNA (lncRNA) (1.6 kb) expressed in normal human tissues
but frequently downregulated in tumor cells (16). MEG3 overexpression has been
reported to promote apoptosis in various tissues and cells;
however, the mechanism of action of MEG3 in ADSCs is unclear
(17–20). Some studies have reported that MEG3
may influence apoptosis through activation of p53, which in turn
regulates downstream genes (21,22).
Tumor suppressor p53 regulates cell cycle initiation and
contributes to apoptosis, genomic stability and angiogenesis. Upon
irreversible DNA damage, p53 triggers apoptosis. The present study
evaluated the roles of MEG3 and p53 in apoptosis of ADSCs by
analyzing the expression of p53 and its related downstream
molecules.
Materials and methods
Cell culture
Human ADSCs were purchased from the Cell Bank of the
Shanghai Institutes for Biological Sciences. Cells were cultured in
DMEM (Gibco; Thermo Fisher Scientific, Inc.) containing 10% fetal
bovine serum (Gibco; Thermo Fisher Scientific, Inc.) and 100 U/ml
penicillin/streptomycin (Gibco; Thermo Fisher Scientific, Inc.).
Cells were incubated at 37°C in a humidified chamber with 5%
CO2. Cells were harvested using 0.25% trypsin-0.02% EDTA
solution (Gibco; Thermo Fisher Scientific, Inc.) and sub-cultured
to 80% confluence. A cell oxidative stress model was established by
incubating cells with 130.6 µM H2O2 at 37°C
for 24 h. Cells cultured in normal medium were used as a control
group.
Transfection
The MEG3 sequence was synthesized (GenBank
NR_002766) and subcloned into the pCDNA3.1 vector (Shanghai
GeneChem Co., Ltd.). MEG3 was ectopically expressed by transfection
of pCDNA-MEG3. The empty pCDNA vector was used as a control. The
small interfering (si)RNA targeting MEG3 (forward,
5′-GGUUGUUGUGAGAAUUAAAUG-3′ and reverse,
5′-UUUAAUUCUCACAACAACCCU-3′) and its negative control (NC; forward,
5′-GGUAAUGUUAAUGAGUUGAUG-3′ and reverse,
5′-UCCCAUUACAAUUACUCAACU-3′) were purchased from Shanghai GeneChem
Co., Ltd. ADSCs were seeded (0.5×105 cells/well) into
24-well plates and cultured at 37°C for 24 h. At 90% confluence,
cells were transfected with pCDNA3.1 vectors (2 mg/l) or siRNAs (40
nmol/l) using Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.), according to the manufacturer's protocol.
At 24 h post-transfection, subsequent experiments were
performed.
Cell viability assay
Cell viability was assessed using an MTT assay.
Cells (0.1×105 cell/well) were seeded in 96-well plates
for 24 h. Cells were pretreated with different concentrations of
H2O2 (0–300 µM) at 37°C for 24 h.
Subsequently, 10 µl MTT (5 mg/ml) solution was added to each well
and incubated for 4 h at 37°C. To dissolve the formazan crystals,
150 µl DMSO was added to each well. The absorbance of each well was
measured at a wavelength of 490 nm using a microplate reader
(Thermo Fisher Scientific, Inc.). Data are expressed as percentages
of the untreated control.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR) analysis
Total RNA was isolated using TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), according to
the manufacturer's protocol. Toral RNA was reverse transcribed into
cDNA using Super M-MLV Reverse Transcriptase and 2×Power Taq PCR
MasterMix (Bioteke Corporation), according to the manufacturer's
protocol. Briefly, for RT, 1 µl oligo (dT)15 and 2 µl
dNTPs (2.5 mM each) were added, and then ddH2O was added
to a total volume of 14.5 µl. The following thermocycling
conditions were used for reverse transcription: 10 min at 25°C, 50
min at 42°C and 5 min at 95°C. The both forward and reverse primers
for MEG3, TP53 and ACTB were provided by Shanghai
GenePharma Co., Ltd. (Table
I).
 | Table I.Primer sequences for reverse
transcription-quantitative PCR. |
Table I.
Primer sequences for reverse
transcription-quantitative PCR.
| Gene | Primer sequences
(5′→3′) |
|---|
| MEG3 | F:
GCTGGGTCGGCTGAAGAAC |
|
| R:
CGTGGCTGTGGAGGGATTT |
| TP53 | F:
ACCACCATCCACTACAACTACAT |
| | R:
CAGGACAGGCACAAACACG |
| ACTB | F:
CTTAGTTGCGTTACACCCTTTCTTG |
|
| R:
CTGTCACCTTCACCGTTCCAGTTT |
qPCR was performed using the following thermocycling
conditions: Initial denaturation at 95°C for 3 min; 40 cycles of
amplification at 95°C for 12 sec and 60°C for 40 sec; and final
extension (72°C for 5 min). qPCR was performed using the SYBR Green
qPCR Detection kit (Tiangen Biotech Co., Ltd.) and the Roche
LightCycler 480 Detection system (Roche Diagnostics). Relative mRNA
expression levels of MEG3 and TP53 were normalized to
the internal reference gene ACTB using the 2−ΔΔCq
method (23). RT-qPCR was
performed in triplicate.
Flow cytometry
Cells were harvested and washed twice in ice-cold
PBS for 48 h. Apoptosis was detected by the tagging of membrane
phosphatidylserine with the fluorescent dye Annexin V-APC/7AAD
Apoptosis Detection kit (Nanjing KeyGEN Biotech, Co. Ltd.) in
accordance with the manufacturer's protocols. In each sample,
~5×105 cells were analyzed and were immediately
subjected to bivariate flow cytometric analysis using a FACScan (BD
Biosciences) equipped with CellQuest (version 7.5.3; BD
Biosciences).
Morphological analysis of apoptotic
cells
Cells were cultured in 24-well plates; after
treatment with H2O2 for 24 h, the cells were
stained with 125 µl Hoechst 33258 (Sigma-Aldrich; Merck KGaA)
staining solution at 37°C for 5 min, washed twice with PBS, and
observed using a fluorescence microscope fitted with a camera
(Olympus Corporation; magnification, ×400).
Western blot analysis
Cells from each group were lysed using RIPA lysis
buffer (Beijing Solarbio Science & Technology Co., Ltd.). The
protein fraction was harvested by centrifugation at 14,000 × g at
5°C for 20 min and quantified using the BCA protein assay kit
(Beijing Solarbio Science & Technology Co., Ltd.). Equal
amounts of protein (20 µg) were separated via 10% SDS-PAGE and
electro-transferred onto a polyvinylidene difluoride membrane (EMD
Millipore). The membranes were blocked with 5% nonfat milk in
Tris-buffered saline-0.1% Tween-20 (TBST) at 20°C for 2 h.
Subsequently, the membranes were incubated overnight at 4°Cin with
the following primary antibodies in TBST: anti-Bax (1:2,000; cat.
no. ab182733; Abcam), anti-Bcl-2 (1:2,000; cat.no. ab182858;
Abcam), anti-caspase3 (1:5,000; cat.no. ab32351; Abcam),
anti-caspase9 (1:2,000; cat.no. ab202068; Abcam), anti-p53
(1:1,000; cat. no. 2527; Cell Signaling Technology, Inc.) and
anti-β-actin (1:1,000; cat. no. sc-130656; Santa Cruz
Biotechnology, Inc.). After three washes with TBST, the membrane
was incubated with an anti-rabbit horseradish peroxidase-conjugated
secondary antibody (1:2,000; cat. no. 7074; Cell Signaling
Technology, Inc.) at 20°C for 2 h. Protein bands were visualized
using a SuperSignal™ West Pico PLUS Chemiluminescent substrate
(Thermo Fisher Scientific, Inc.). Protein expression levels were
quantified using Image-Pro Plus software (version 6.0; Media
Cybernetics, Inc.).
Statistical analysis
All experiments were performed in triplicate.
Continuous variables are presented as the mean ± SEM values. SPSS
version 13.0 software (SPSS, Inc.) was used for statistical
analysis. Differences between groups were analyzed using an
unpaired Student's t-test. All other analyses were performed using
one-way ANOVA and post-hoc analyses were performed using Tukey's
multiple comparisons test. P<0.05 was considered to indicate a
statistically significance difference.
Results
MEG3 regulates apoptosis of ADSCs
To investigate the regulatory effect of MEG3 on
apoptosis of ADSCs, MEG3 was transfected into ADSCs to obtain a
MEG3 overexpression (MEG3-up) group and a MEG3-silenced (MEG3-down)
group. Compared with the vector group, the expression of MEG3 was
significantly higher in the MEG3-up group as determined by RT-qPCR
(P<0.01). Compared with the NC group, the expression of MEG3 was
significantly lower in the MEG3-down group (P<0.01) (Fig. 1A).
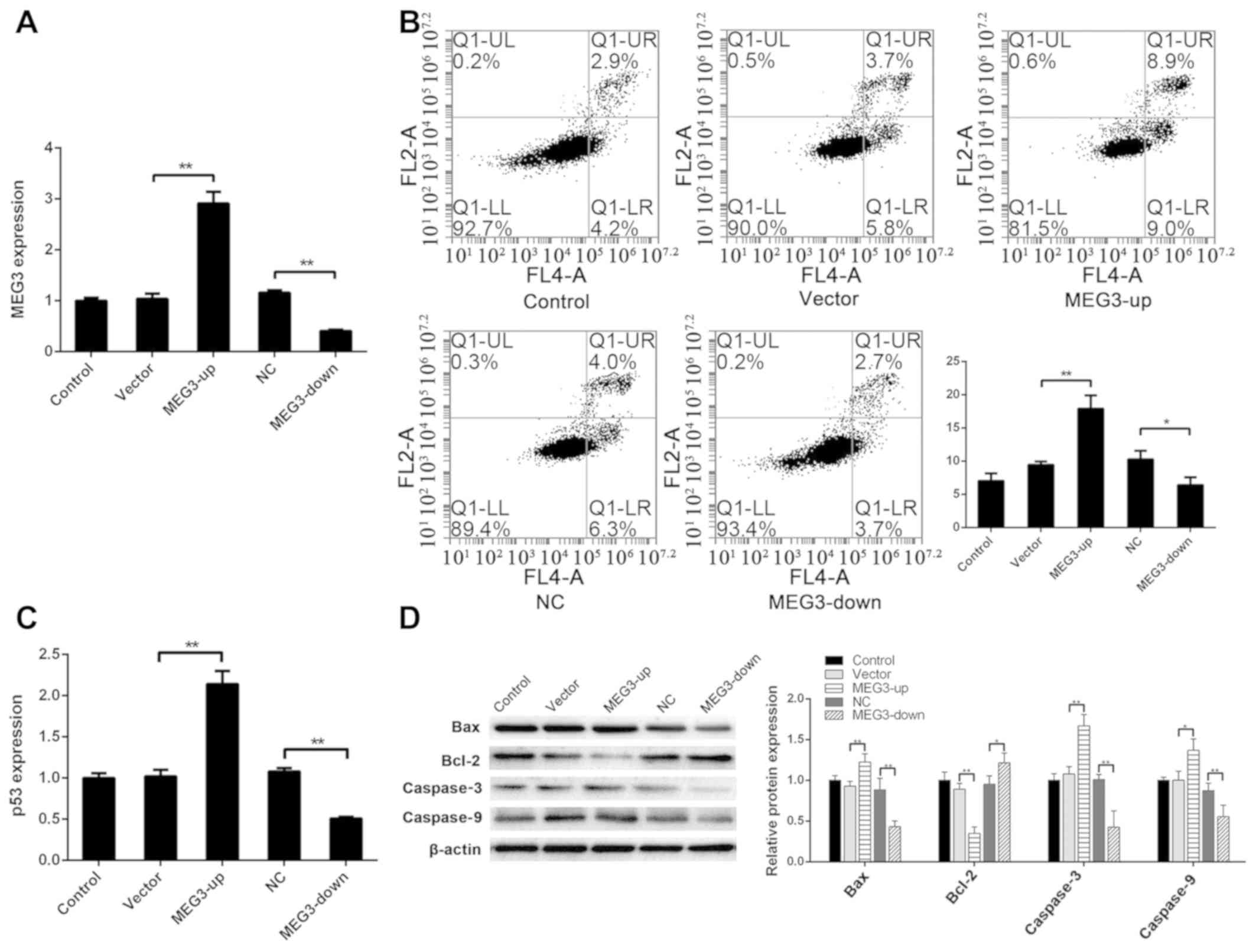 | Figure 1.MEG3 regulates apoptosis of ADSCs.
(A) After transfecting MEG3 into ADSCs, MEG3 expression in the
MEG3-up group was significantly higher than that in the vector
group, and MEG3 expression in the MEG3-down group was significantly
lower than that in the NC group. (B) Apoptotic rate (UR+LR) was
7.04±1.10% in the control group, 9.44±0.52% in the vector group,
17.91±2.02% in the MEG3-up group, 10.28±1.31% in the NC group and
6.38±1.21% in the MEG3-down group. The apoptotic rate in the
MEG3-up group was significantly higher than that in the vector
group, and that in the MEG3-down group was lower than that in the
NC group. (C) Compared with the vector group, the expression of p53
was significantly higher in the MEG3-up group. Compared with the NC
group, the expression of p53 was significantly lower in the
MEG3-down group. (D) Compared with the control group, Bax, caspase3
and caspase9 were upregulated in the MEG3-up group. Compared with
the NC group, Bax, caspase3 and caspase9 were downregulated in the
MEG3-down group. Compared with the vector group, Bcl-2 was
downregulated in the MEG3-up group. Compared with the NC group,
Bcl-2 was upregulated in the MEG3-down group. *P<0.05,
**P<0.01. ADSC, adipose-derived stem cells; MEG3, maternally
expressed gene 3; NC, negative control. |
Apoptosis in each group was detected via flow
cytometry to analyze the effect of MEG3 expression on apoptosis.
The apoptotic rate was 7.04±1.10% in the control group, 9.44±0.52%
in the vector group, 17.91±2.02% in the MEG3-up group, 10.28±1.31%
in the NC group and 6.38±1.21% in the MEG3-down group. The
apoptotic rate in the MEG3-up group was significantly higher than
that in the vector group (P<0.01) and that of the MEG3-down
group was lower than that of the NC group (P<0.05) (Fig. 1B). These findings indicated that
the apoptotic rate of ADSCs was increased and decreased when MEG3
expression levels were increased and decreased, respectively,
suggesting that MEG3 expression affects apoptosis of ADSCs.
The mechanism underlying the effect of MEG3 on
apoptosis in cells may be associated with p53 regulation by MEG3.
To evaluate this hypothesis and explore the mechanism underlying
apoptotic regulation in ADSCs by MEG3 in more detail, RT-qPCR
analysis was performed for TP53 in each group of cells.
Compared with the vector group, the expression of TP53 was
significantly higher in the MEG3-up group (P<0.01). Compared
with the NC group, the expression of TP53 was significantly
lower in the MEG3-down group (P<0.01) (Fig. 1C). These findings indicated that
p53 may be regulated by MEG3, thus influencing apoptosis of
ADSCs.
To further assess apoptosis of ADSCs, key proteins
in the apoptosis-related cell signaling pathway regulated by p53
were detected via western blotting. Compared with the vector group,
Bax was upregulated in the MEG3-up group (P<0.01), whereas it
was downregulated in the MEG3-down group compared with the NC group
(P<0.01). Bcl-2 was downregulated in the MEG3-up group compared
with the vector group (P<0.01), and was upregulated in the
MEG3-down group compared with the NC group (P<0.05). Caspase3
was upregulated in the MEG3-up group compared with the vector group
(P<0.01), and downregulated in the MEG3-down group compared with
the NC group (P<0.01). Caspase9 was upregulated in the MEG3-up
group compared with the vector group (P<0.05), and downregulated
in the MEG3-down group compared with the NC group (P<0.01)
(Fig. 1D). These results agree
with the change in p53 expression and suggested that MEG3 regulated
apoptosis of ADSCs by regulating p53, in turn regulating the
downstream apoptotic Bcl-2/Bax pathway.
H2O2-induced
apoptosis of ADSCs
To further assess apoptosis of ADSCs under oxidative
stress, a cell oxidative stress model was established with
H2O2 to induce apoptosis of ADSCs. To
determine the appropriate H2O2 concentration,
ADSCs were first treated with a gradient of
H2O2 and a survival curve of ADSCs was
plotted using the MTT assay. The mortality of ADSCs was 50% at a
concentration of 130.6 µM H2O2 (Fig. 2A). Hence, the half maximal
inhibitory concentration (IC50) was used in the
following experiment.
First, the cells were stained with Hoechst 33258 to
detect apoptosis of cells under H2O2-induced
oxidative stress. Compared with the control group, the number of
apoptotic cells in the IC50 group was markedly
increased, as revealed by inverted fluorescence microscopy
(Fig. 2B). Furthermore, upon flow
cytometric detection, the apoptotic rate of ADSCs in the
IC50 group was 43.63±5.2%, which was significantly
higher than that in the control group (5.64±1.72%; P<0.01),
suggesting that ADSCs undergo apoptosis during oxidative stress
induced by H2O2 (Fig. 2C).
After confirming the effect of
H2O2 on apoptosis of ADSCs, MEG3 expression
was analyzed in ADSCs by RT-qPCR analysis. The results showed that
MEG3 was significantly upregulated in the IC50 group
compared with the control group under oxidative stress induced by
H2O2 (P<0.01; Fig. 2D). These results suggested that
MEG3 may be involved in apoptosis of ADSCs induced by
H2O2 and plays an important regulatory
role.
The key proteins associated with apoptosis were
detected via western blotting. Under the influence of
H2O2, p53, Bax, caspase3 and caspase9 were
upregulated, whereas Bcl-2 was downregulated in the IC50
group compared with the control group (P<0.01; Fig. 2E), which was associated with MEG3
upregulation upon H2O2 treatment.
Role of MEG3 in
H2O2-induced apoptosis
To confirm that MEG3 serves a key role in
H2O2-induced apoptosis of ADSCs, the MEG3-up
group, vector group, MEG3-down group and its NC group were assessed
following treatment with H2O2. As determined
by flow cytometry, the apoptotic rates of the vector group, MEG3-up
group, NC group and MEG3-down group were 40.93±1.82, 71.53±2.31,
41.13±4.35 and 24.09±2.54%, respectively. MEG3 silencing
effectively decreased H2O2-induced apoptosis
in response to the IC50 concentration of
H2O2, and the apoptotic rate in the MEG3-down
group was significantly lower than that in the NC group
(P<0.01). However, upon MEG3 upregulation in the MEG3-up group,
apoptotic induction by H2O2 was significantly
greater than that in the vector group (P<0.01) (Fig. 3A). These data suggested that MEG3
may be the key factor regulating ADSCs apoptosis induced by
H2O2.
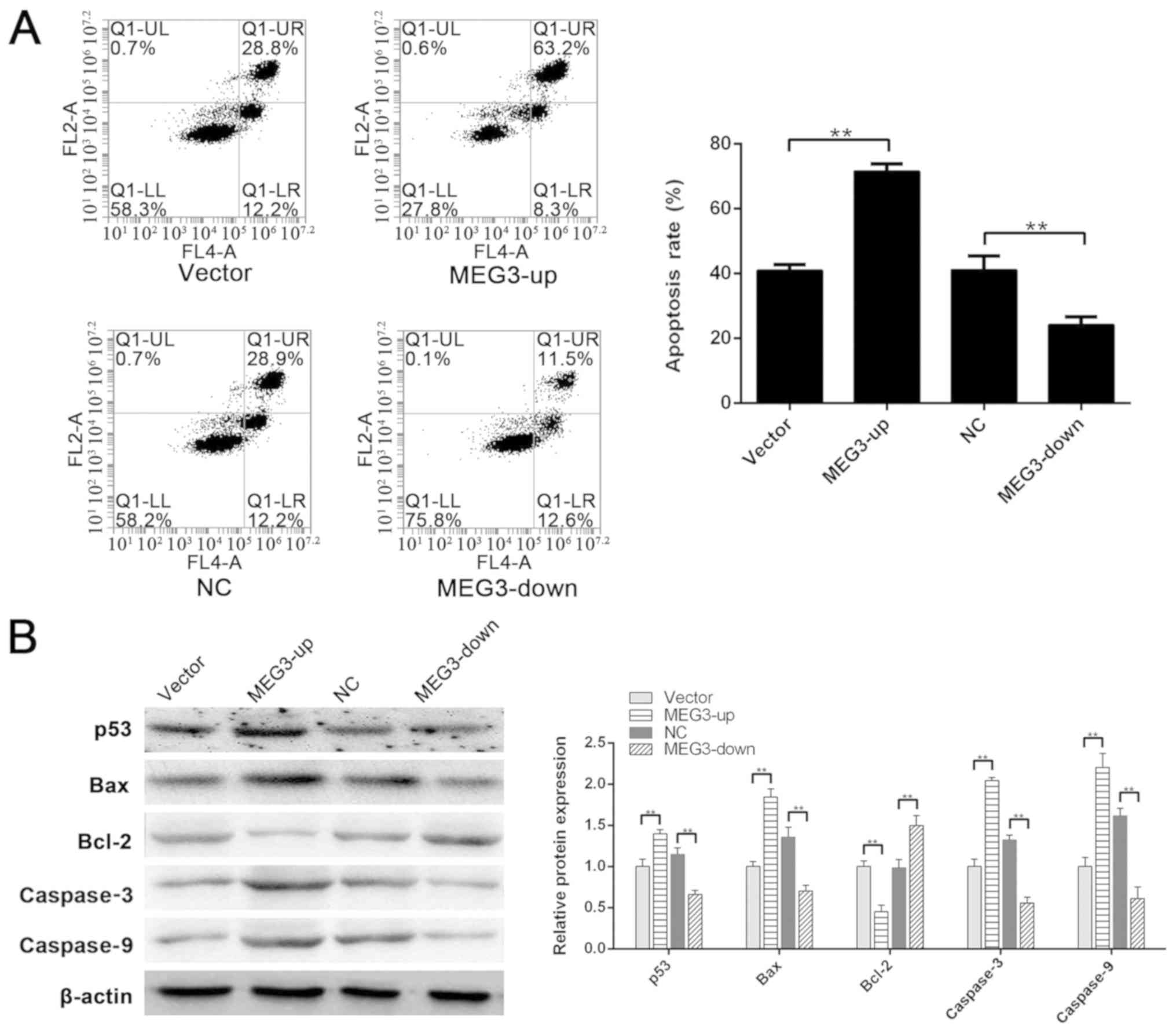 | Figure 3.Role of MEG3 in
H2O2-induced apoptosis. (A) Apoptotic rates
of the vector group, MEG3-up group, NC group and MEG3-down group
were 40.93±1.82, 71.53±2.31, 41.13±4.35 and 24.09±2.54%,
respectively. Apoptotic rate in the MEG3-up group was significantly
greater than that in the vector group. Apoptotic rate in the
MEG3-down group was significantly lower than that in the NC group.
(B) Compared with the vector group, the expression of p53, Bax,
caspase3 and caspase9 was significantly higher in the MEG3-up
group. Compared with the NC group, the expression of p53, Bax,
caspase3 and caspase9 was significantly lower in the MEG3-down
group. Compared with the vector group, Bcl-2 was downregulated in
the MEG3-up group. Compared with the NC group, Bcl-2 was
upregulated in the MEG3-down group. **P<0.01. MEG3, maternally
expressed gene 3; NC, negative control. |
To further investigate the mechanism underlying
apoptotic regulation by MEG3, western blotting was performed to
analyze the expression of related proteins. p53 was significantly
downregulated in the MEG3-down group compared with the NC group
(P<0.01), and MEG3 upregulation in the MEG3-up group
significantly enhanced p53 compared with in the vector group
(P<0.01). The expression of Bax, caspase3 and caspase9
downstream of p53 displayed a similar tendency (P<0.01).
However, Bcl-2 was significantly upregulated in the MEG3-down group
compared with the NC group (P<0.01; Fig. 3B). Furthermore, Bcl-2 expression
was significantly downregulated in the MEG3-up group compared with
the vector group (P<0.01; Fig.
3B). These results indicated that p53 may be regulated by MEG3,
and the downstream Bcl-2/Bax signaling pathway could regulate
apoptosis of these cells.
Discussion
In 1893, Neuber first proposed autologous
transplantation of adipose tissue for filling (24); since then, autologous
transplantation of adipose tissue has improved. This method is
increasingly used in plastic surgery, and is considered safe and
easy. Subcutaneous adipose tissue in the waist, abdomen and leg are
used as donor tissue for autologous transplantation, and these
tissues are obtained through vacuum suction and mechanical
curettage. Saline washing, static precipitation or low-speed
centrifugation are used to improve the purity and cytoactivity of
adipose tissue to improve its survival rate. In 2002, Coleman
(25) proposed a method for
further treating adipose tissue by centrifugation, decanting and
wicking to eliminate unviable components and improve the survival
rate after transplantation. This method is currently widely used in
the clinical setting. In a study using an animal model aimed at
improving the survival rate of adipose tissue transplantation,
ADSCs were infused into adipose tissue and the cell-assisted
lipotransfer method was proposed, which has been applied clinically
in recent years (26,27). The important role of ADSCs in the
survival rate of adipose transplantation has been increasingly
recognized (28).
lncRNAs were originally considered a type of
nonfunctional genetic noise; however, the functions of lncRNA in
regulating gene expression, chromatin remodeling, transcription and
post-transcriptional processing have been increasingly reported
(29). Upregulation of MEG3 has
been reported to promote apoptosis in bladder cancer, gastric
cancer, lung cancer, osteosarcoma and other cells (23,30–32).
In ADSCs, MEG3 overexpression significantly increased the apoptotic
rate of ADSCs, whereas MEG3 silencing significantly decreased the
apoptotic rate of ADSCs, thus confirming that MEG3 significantly
contributes to apoptosis of ADSCs.
MEG3 regulates cellular physiology by regulating p53
and downstream signaling (23,32).
RT-qPCR analysis of the regulatory association in ADSCs revealed
that p53 expression levels were positively associated with MEG3
expression levels. Furthermore, western blotting indicated that the
Bcl-2/Bax pathway downstream of p53 may regulate apoptosis. When
MEG3 was upregulated, p53 and Bax were upregulated, whereas Bcl-2
protein expression was suppressed, thus promoting apoptosis.
During adipose tissue transplantation, currently
used methods for obtaining adipose tissue often result in the
incorporation of numerous damaged and dead cells in the tissue; in
addition, after transplantation, the microvascular system in the
tissue has not formed yet and the cells are in a state of ischemia
and anoxia (18,33). These conditions subject
transplanted ADSCs to oxidative stress, thus increasing reactive
oxygen species production in cells and inducing apoptosis. A
reduction in the survival rate of ADSCs directly affects the
survival rate of transplanted tissue (33). In the present study, oxidative
stress was induced in ADSCs with H2O2, thus
increasing the apoptotic rate of ADSCs. Furthermore, RT-qPCR
analysis revealed that MEG3 was upregulated, suggesting that MEG3
may contribute to apoptosis induced by oxidative stress in ADSCs.
After overexpression and silencing of MEG3 in ADSCs, the apoptotic
rate of ADSCs induced by H2O2 was assessed.
MEG3 silencing decreased apoptotic induction by
H2O2, whereas MEG3 overexpression aggravated
apoptosis. Detection of pathway proteins via western blotting
revealed that p53 was regulated by MEG3 and in turn regulated
apoptosis of ADSCs through the Bcl-2/Bax pathway.
In conclusion, the present results revealed that
MEG3 significantly contributed to spontaneous apoptosis of ADSCs
and apoptosis induced by oxidative stress. This effect was
regulated by MEG3 and it was suggested that p53 may modulate the
downstream Bcl-2/Bax pathway. The present results highlight the
role of MEG3 in apoptosis of ADSCs and provide a foundation for
further improving the survival rate of ADSCs. Future work to
further verify the mechanism of apoptosis underlying ADSCs in
vivo and in vitro should be conducted to lay a
foundation for future clinical research. These results may be
useful for improving the survival rate of adipose tissue grafts and
surgical effects, as well as the success rate of plastic surgery
for filling with adipose tissue.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Youth
Backbone Support Project of China Medical University (grant no.
QGZD2018025).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
YS designed the study, performed the research,
analyzed data and wrote the paper. The author read and approved the
final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The author declares that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ADSC
|
adipose-derived stem cell
|
|
TBST
|
Tris-buffered saline-Tween
|
References
|
1
|
Rohrich RJ: The American Society of
Plastic Surgeons' procedural statistics: What they really mean.
Plast Reconstr Surg. 112:1389–1392. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Claro F Jr, Figueiredo JC, Zampar AG and
Pinto-Neto AM: Applicability and safety of autologous fat for
reconstruction of the breast. Br J Surg. 99:768–780. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang CF, Zhou Z, Yan YJ, Zhao DM, Chen F
and Qiao Q: Clinical analyses of clustered microcalcifications
after autologous fat injection for breast augmentation. Plast
Reconstr Surg. 127:1669–1673. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Coleman SR: Structural fat grafting: More
than a permanent filler. Plast Reconstr Surg. 118 (3
Suppl):S108–S120. 2006. View Article : Google Scholar
|
|
5
|
Lu F, Li J, Gao J, Ogawa R, Ou C, Yang B
and Fu B: Improvement of the survival of human autologous fat
transplantation by using VEGF-transfected adipose-derived stem
cells. Plast Reconstr Surg. 124:1437–1446. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kirkham JC, Lee JH, Medina MA III,
McCormack MC, Randolph MA and Austen WG Jr: The impact of
liposuction cannula size on adipocyte viability. Ann Plast Surg.
69:479–481. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nguyen A, Pasyk KA, Bouvier TN, Hassett CA
and Argenta LC: Comparative study of survival of autologous adipose
tissue taken and transplanted by different techniques. Plast
Reconstr Surg. 85:378–389. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Billings E Jr and May JW Jr: Historical
review and present status of free fat graft autotransplantation in
plastic and reconstructive surgery. Plast Reconstr Surg.
83:368–381. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pal R, Venkataramana NK, Bansal A,
Balaraju S, Jan M, Chandra R, Dixit A, Rauthan A, Murgod U and
Totey S: Ex vivo-expanded autologous bone marrow-derived
mesenchymal stromal cells in human spinal cord injury/paraplegia: A
pilot clinical study. Cytotherapy. 11:897–911. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zuk PA: The adipose-derived stem cell:
Looking back and looking ahead. Mol Biol Cell. 21:1783–1787. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ionescu L, Byrne RN, van Haaften T,
Vadivel A, Alphonse RS, Rey-Parra GJ, Weissmann G, Hall A, Eaton F
and Thébaud B: Stem cell conditioned medium improves acute lung
injury in mice: In vivo evidence for stem cell paracrine action. Am
J Physiol Lung Cell Mol Physiol. 303:L967–L977. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fouraschen SM, Pan Q, de Ruiter PE, Farid
WR, Kazemier G, Kwekkeboom J, Ijzermans JN, Metselaar HJ, Tilanus
HW, de Jonge J and van der Laan LJ: Secreted factors of human
liver-derived mesenchymal stem cells promote liver regeneration
early after partial hepatectomy. Stem Cells Dev. 21:2410–2419.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell
JW, Katz AJ, Benhaim P, Lorenz HP and Hedrick MH: Multilineage
cells from human adipose tissue: Implications for cell-based
therapies. Tissue Eng. 7:211–228. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kern S, Eichler H, Stoeve J, Klüter H and
Bieback K: Comparative analysis of mesenchymal stem cells from bone
marrow, umbilical cord blood, or adipose tissue. Stem Cells.
24:1294–1301. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Su M, Guan H, Zhang F, Gao Y, Teng X and
Yang W: HDAC6 regulates the chaperone-mediated autophagy to prevent
oxidative damage in injured neurons after experimental spinal cord
injury. Oxid Med Cell Longev. 2016:72637362016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen X, Yan L, Guo Z, Chen Z, Chen Y, Li
M, Huang C, Zhang X and Chen L: Adipose-derived mesenchymal stem
cells promote the survival of fat grafts via crosstalk between the
Nrf2 and TLR4 pathways. Cell Death Dis. 7:e23692016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang X, Rice K, Wang Y, Chen W, Zhong Y,
Nakayama Y, Zhou Y and Klibanski A: Maternally expressed gene 3
(MEG3) noncoding ribonucleic acid: Isoform structure, expression,
and functions. Endocrinology. 151:939–947. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Luo G, Wang M, Wu X, Tao D, Xiao X, Wang
L, Min F, Zeng F and Jiang G: Long non-coding RNA MEG3 inhibits
cell proliferation and induces apoptosis in prostate cancer. Cell
Physiol Biochem. 37:2209–2220. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Peng W, Si S, Zhang Q, Li C, Zhao F, Wang
F, Yu J and Ma R: Long non-coding RNA MEG3 functions as a competing
endogenous RNA to regulate gastric cancer progression. J Exp Clin
Cancer Res. 34:792015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang X, Zhou Y, Mehta KR, Danila DC,
Scolavino S, Johnson SR and Klibanski A: A pituitary-derived MEG3
isoform functions as a growth suppressor in tumor cells. J Clin
Endocrinol Metab. 88:5119–5126. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lu KH, Li W, Liu XH, Sun M, Zhang ML, Wu
WQ, Xie WP and Hou YY: Long non-coding RNA MEG3 inhibits NSCLC
cells proliferation and induces apoptosis by affecting p53
expression. BMC Cancer. 13:4612013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou Y, Zhong Y, Wang Y, Zhang X, Batista
DL, Gejman R, Ansell PJ, Zhao J, Weng C and Klibanski A: Activation
of p53 by MEG3 non-coding RNA. J Biol Chem. 282:24731–24742. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Neuber F: Fettransplantation. Chir Kongr
Verhandl Dsch Gesellch Chir. 22:661893.
|
|
25
|
Coleman SR: Hand rejuvenation with
structural fat grafting. Plast Reconstr Surg. 110:1731–1747. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Matsumoto D, Sato K, Gonda K, Takaki Y,
Shigeura T, Sato T, Aiba-Kojima E, Iizuka F, Inoue K, Suga H and
Yoshimura K: Cell-assisted lipotransfer: Supportive use of human
adipose-derived cells for soft tissue augmentation with
lipoinjection. Tissue Eng. 12:3375–3382. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Moseley TA, Zhu M and Hedrick MH:
Adipose-derived stem and progenitor cells as fillers in plastic and
reconstructive surgery. Plast Reconstr Surg. 118 (3
Suppl):S121–S128. 2006. View Article : Google Scholar
|
|
28
|
Toyserkani NM, Quaade ML and Sørensen JA:
Cell-Assisted Lipotransfer: A systematic review of its efficacy.
Aesthetic Plast Surg. 40:309–318. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ponting CP, Oliver PL and Reik W:
Evolution and functions of long noncoding RNAs. Cell. 136:629–641.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ying L, Huang Y, Chen H, Wang Y, Xia L,
Chen Y, Liu Y and Qiu F: Downregulated MEG3 activates autophagy and
increases cell proliferation in bladder cancer. Mol Biosyst.
9:407–411. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sun M, Xia R, Jin F, Xu T, Liu Z, De W and
Liu X: Downregulated long noncoding RNA MEG3 is associated with
poor prognosis and promotes cell proliferation in gastric cancer.
Tumour Biol. 35:1065–1073. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shi Y, Lv C, Shi L and Tu G: MEG3 inhibits
proliferation and invasion and promotes apoptosis of human
osteosarcoma cells. Oncol Lett. 15:1917–1923. 2018.PubMed/NCBI
|
|
33
|
Carrière A, Ebrahimian TG, Dehez S, Augé
N, Joffre C, André M, Arnal S, Duriez M, Barreau C, Arnaud E, et
al: Preconditioning by mitochondrial reactive oxygen species
improves the proangiogenic potential of adipose-derived cells-based
therapy. Arterioscler Thromb Vasc Biol. 29:1093–1099. 2009.
View Article : Google Scholar : PubMed/NCBI
|

















