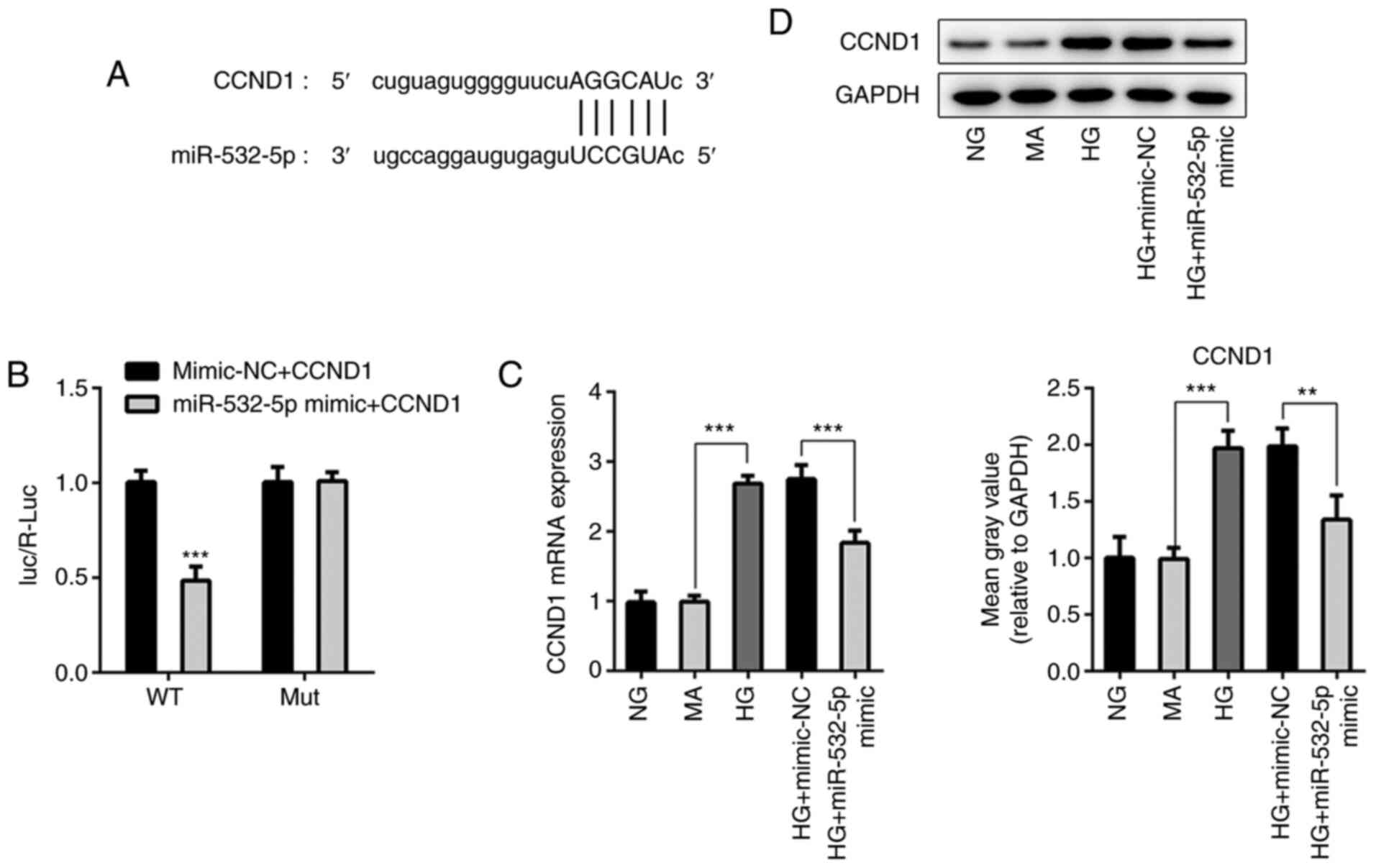
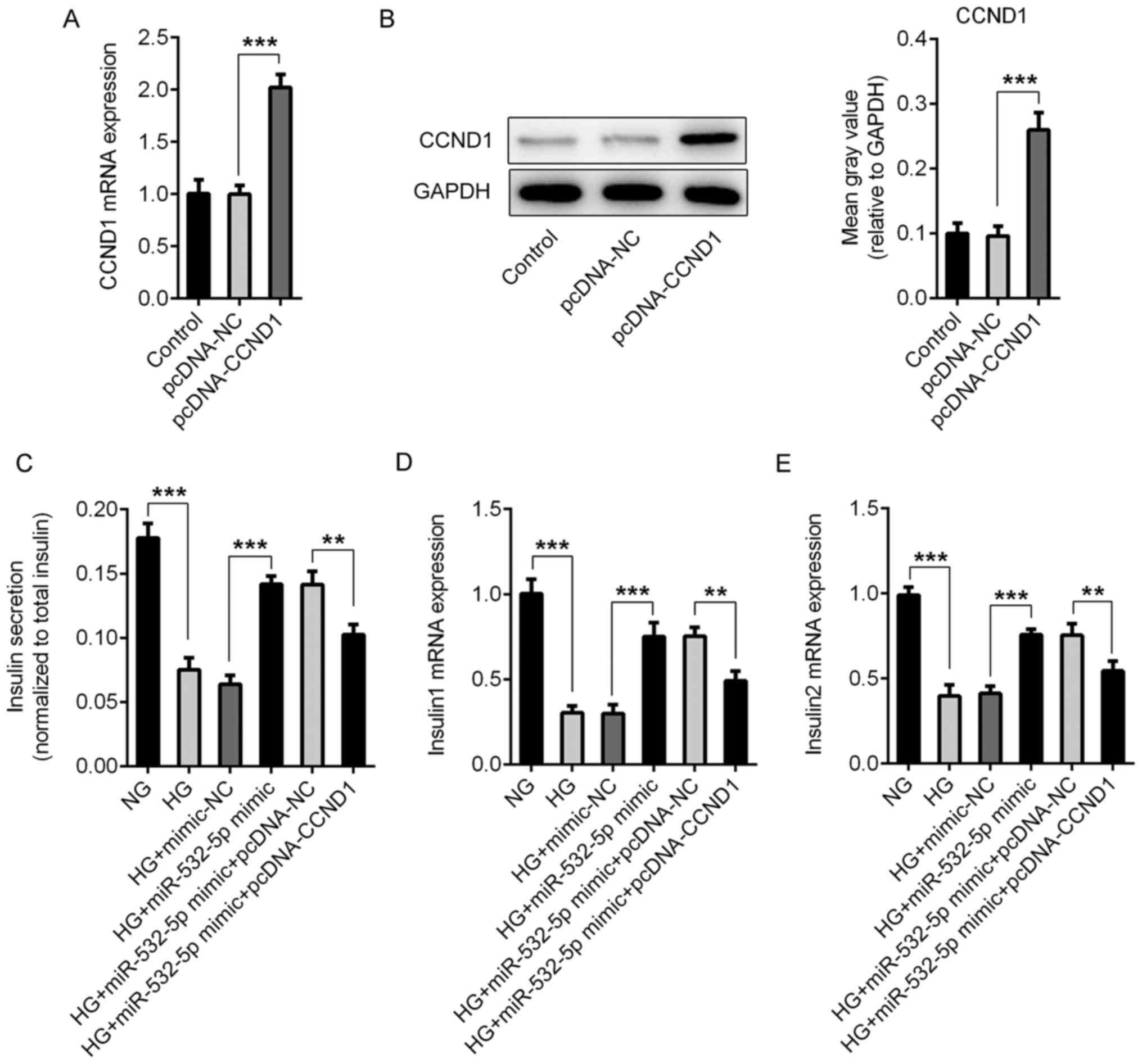
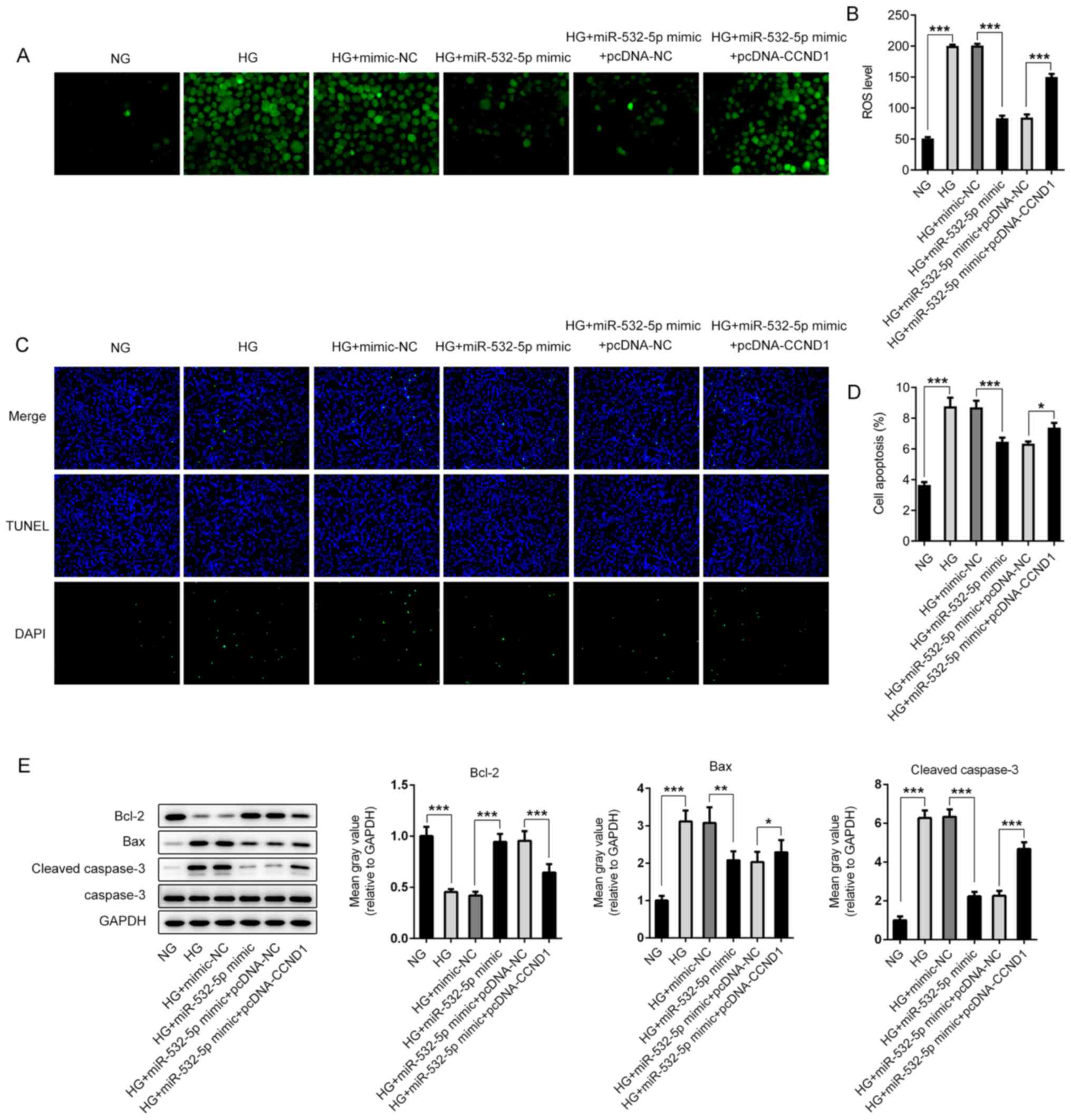
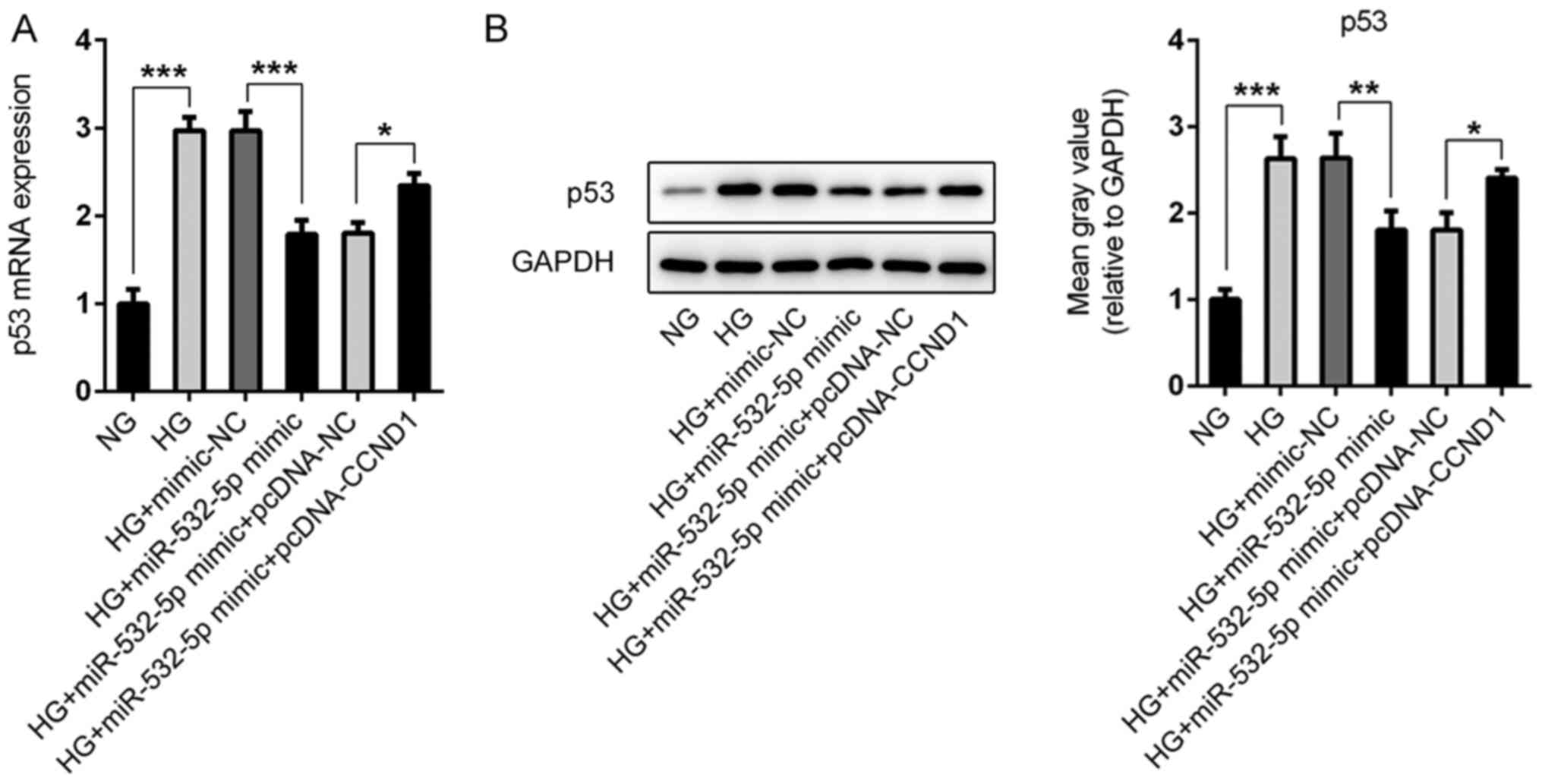
|
1
|
Yang X, Zhang Y, Xu W, Deng R, Liu Y, Li
F, Wang Y, Ji X, Bai M, Zhou F, et al: Potential role of Hsp90 in
rat islet function under the condition of high glucose. Acta
Diabetol. 53:621–628. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhao YC, Zhu J, Song GY and Li XS:
Relationship between thioredoxin-interacting protein (TXNIP) and
islet β-cell dysfunction in patients with impaired glucose
tolerance and hypertriglyceridemia. Int J Clin Exp Med.
8:4363–4368. 2015.PubMed/NCBI
|
|
3
|
Nelson P, Smith N, Ciupe S, Zou W, Omenn
GS and Pietropaolo M: Modeling dynamic changes in type 1 diabetes
progression: Quantifying beta-cell variation after the appearance
of islet-specific autoimmune responses. Math Biosci Eng. 6:753–778.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dahan T, Ziv O, Horwitz E, Zemmour H, Lavi
J, Swisa A, Leibowitz G, Ashcroft FM, In't Veld P, Glaser B and Dor
Y: Pancreatic beta-cells express the fetal islet hormone gastrin in
rodent and human diabetes. Diabetes. 66:426–436. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Saliminejad K, Khorram Khorshid HR,
Soleymani Fard S and Ghaffari SH: An overview of microRNAs:
Biology, functions, therapeutics, and analysis methods. J Cell
Physiol. 234:5451–5465. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jones A, Danielson KM, Benton MC, Ziegler
O, Shah R, Stubbs RS, Das S and Macartney-Coxson D: MiRNA
signatures of insulin resistance in obesity. Obesity (Silver
Spring). 25:1734–1744. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ortega FJ, Mercader JM, Moreno-Navarrete
JM, Rovira O, Guerra E, Esteve E, Xifra G, Martínez C, Ricart W,
Rieusset J, et al: Profiling of circulating microRNAs reveals
common microRNAs linked to type 2 diabetes that change with insulin
sensitization. Diabetes Care. 37:1375–1383. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ma J, Zhang J, Wang Y, Long K, Wang X, Jin
L, Tang Q, Zhu L, Tang G, Li X and Li M: MiR-532-5p alleviates
hypoxia-induced cardiomyocyte apoptosis by targeting PDCD4. Gene.
675:36–43. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Taneera J, Fadista J, Ahlqvist E, Zhang M,
Wierup N, Renström E and Groop L: Expression profiling of cell
cycle genes in human pancreatic islets with and without type 2
diabetes. Mol Cell Endocrinol. 375:35–42. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Shen J and Zhu B: Integrated analysis of
the gene expression profile and DNA methylation profile of obese
patients with type 2 diabetes. Mol Med Rep. 17:7636–7644.
2018.PubMed/NCBI
|
|
11
|
Bustany S, Bourgeais J, Tchakarska G, Body
S, Hérault O, Gouilleux F and Sola B: Cyclin D1 unbalances the
redox status controlling cell adhesion, migration, and drug
resistance in myeloma cells. Oncotarget. 7:45214–45224. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhong Q, Hu Z, Li Q, Yi T, Li J and Yang
H: Cyclin D1 silencing impairs DNA double strand break repair,
sensitizes BRCA1 wildtype ovarian cancer cells to olaparib. Gynecol
Oncol. 152:157–165. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Pistritto G, Trisciuoglio D, Ceci C,
Garufi A and D'Orazi G: Apoptosis as anticancer mechanism: Function
and dysfunction of its modulators and targeted therapeutic
strategies. Aging (Albany NY). 8:603–619. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu RX, Ma Y, Hu XL, Liao YP, Hu XN, He BC
and Sun WJ: Pioglitazone/metformin adduct regulates insulin
secretion and inhibits high glucose-induced apoptosis via
p21-p53-MDM2 signaling in INS-1 cells. J Cell Biochem.
119:5449–5459. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu X, Chen H, Hou Y, Ma X, Ye M, Huang R,
Hu B, Cao H, Xu L, Liu M, et al: Adaptive EGF expression sensitizes
pancreatic cancer cells to ionizing radiation through activation of
the cyclin D1/P53/PARP pathway. Int J Oncol. 54:1466–1480.
2019.PubMed/NCBI
|
|
16
|
Ruan D, Liu Y, Wang X, Yang D and Sun Y:
MiR-149-5p protects against high glucose-induced pancreatic beta
cell apoptosis via targeting the BH3-only protein BIM. Exp Mol
Pathol. 110:1042792019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Abouzaripour M, Pasbakhsh P, Atlasi N,
Shahverdi AH, Mahmoudi R and Kashani IR: In vitro differentiation
of insulin secreting cells from mouse bone marrow derived
stage-specific embryonic antigen 1 positive stem cells. Cell J.
17:701–710. 2016.PubMed/NCBI
|
|
19
|
Ye D, Zhang T, Lou G, Xu W, Dong F, Chen G
and Liu Y: Plasma miR-17, miR-20a, miR-20b and miR-122 as potential
biomarkers for diagnosis of NAFLD in type 2 diabetes mellitus
patients. Life Sci. 208:201–207. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lakhter AJ, Pratt RE, Moore RE, Doucette
KK, Maier BF, DiMeglio LA and Sims EK: Beta cell extracellular
vesicle miR-21-5p cargo is increased in response to inflammatory
cytokines and serves as a biomarker of type 1 diabetes.
Diabetologia. 61:1124–1134. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tang Q, Len Q, Liu Z and Wang W:
Overexpression of miR-22 attenuates oxidative stress injury in
diabetic cardiomyopathy via Sirt 1. Cardiovasc Ther. 36:2018.
View Article : Google Scholar
|
|
22
|
Wang J, Zhang J, Chen X, Yang Y, Wang F,
Li W, Awuti M, Sun Y, Lian C, Li Z, et al: MiR-365 promotes
diabetic retinopathy through inhibiting Timp3 and increasing
oxidative stress. Exp Eye Res. 168:89–99. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Cheng Y, Kuang W, Hao Y, Zhang D, Lei M,
Du L, Jiao H, Zhang X and Wang F: Downregulation of miR-27a* and
miR-532-5p and upregulation of miR-146a and miR-155 in LPS-induced
RAW264.7 macrophage cells. Inflammation. 35:1308–1313. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yan X, Zeng D, Zhu H, Zhang Y, Shi Y, Wu
Y, Tang H and Li D: MiRNA-532-5p regulates CUMS-induced
depression-like behaviors and modulates LPS-induced proinflammatory
cytokine signaling by targeting STAT3. Neuropsychiatr Dis Treat.
16:2753–2764. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cai X, Wang S, Hong L, Yu S, Li B, Zeng H,
Yang X, Zhang P and Shao L: Long noncoding RNA taurine-upregulated
gene 1 knockdown protects cardiomyocytes against
hypoxia/reoxygenation-induced injury through regulating
miR-532-5p/Sox8 axis. J Cardiovasc Pharmacol. 76:556–563. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gurke J, Schindler M, Pendzialek SM,
Thieme R, Grybel KJ, Heller R, Spengler K, Fleming TP, Fischer B
and Navarrete Santos A: Maternal diabetes promotes mTORC1
downstream signalling in rabbit preimplantation embryos.
Reproduction. 151:465–476. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li N, Zeng J, Sun F, Tong X, Meng G, Wu C,
Ding X, Liu L, Han M, Lu C and Dai F: p27 inhibits CDK6/CCND1
complex formation resulting in cell cycle arrest and inhibition of
cell proliferation. Cell Cycle. 17:2335–2348. 2018. View Article : Google Scholar : PubMed/NCBI
|