Regulation of hematopoiesis by hedgehog signaling (Review)
- Authors:
- Published online on: March 28, 2023 https://doi.org/10.3892/mmr.2023.12987
- Article Number: 100
Abstract
Introduction
Three types of hedgehog (Hh) ligands, Sonic hedgehog (Shh), Indian hedgehog (Ihh) and Desert hedgehog (Dhh) have been found in mammals exhibiting different expression patterns and biological functions (1). Shh is extensively expressed in a number of tissues in both the embryo and adult. However, Ihh is specifically expressed in hematopoietic cells, bones, cartilage and the eyes, whereas Dhh is restricted to the gonads, external genitalia, eyes and peripheral nerves (2–5). Hh ligands that bind to a transmembrane protein, patched (Ptch), transduce an intracellular signal that is widely involved in the differentiation, proliferation and survival of numerous cells at a series of developmental stages from embryogenesis to adulthood (1). It has been reported that Hh signaling spatially and temporally paves the way for the development and differentiation of hematopoietic stem and progenitor cells (HSPCs) and blood cells in embryonic and adult hematopoietic system developing tissues. Defining the role of Hh signaling in hematopoiesis, and the potential impact in embryonic hematopoiesis and the development of multi-lineage blood cells in adults may provide a novel theoretical basis for improving regeneration following injury or hematopoietic stem cell (HSC) transplantation.
Hh signaling pathway in mammals
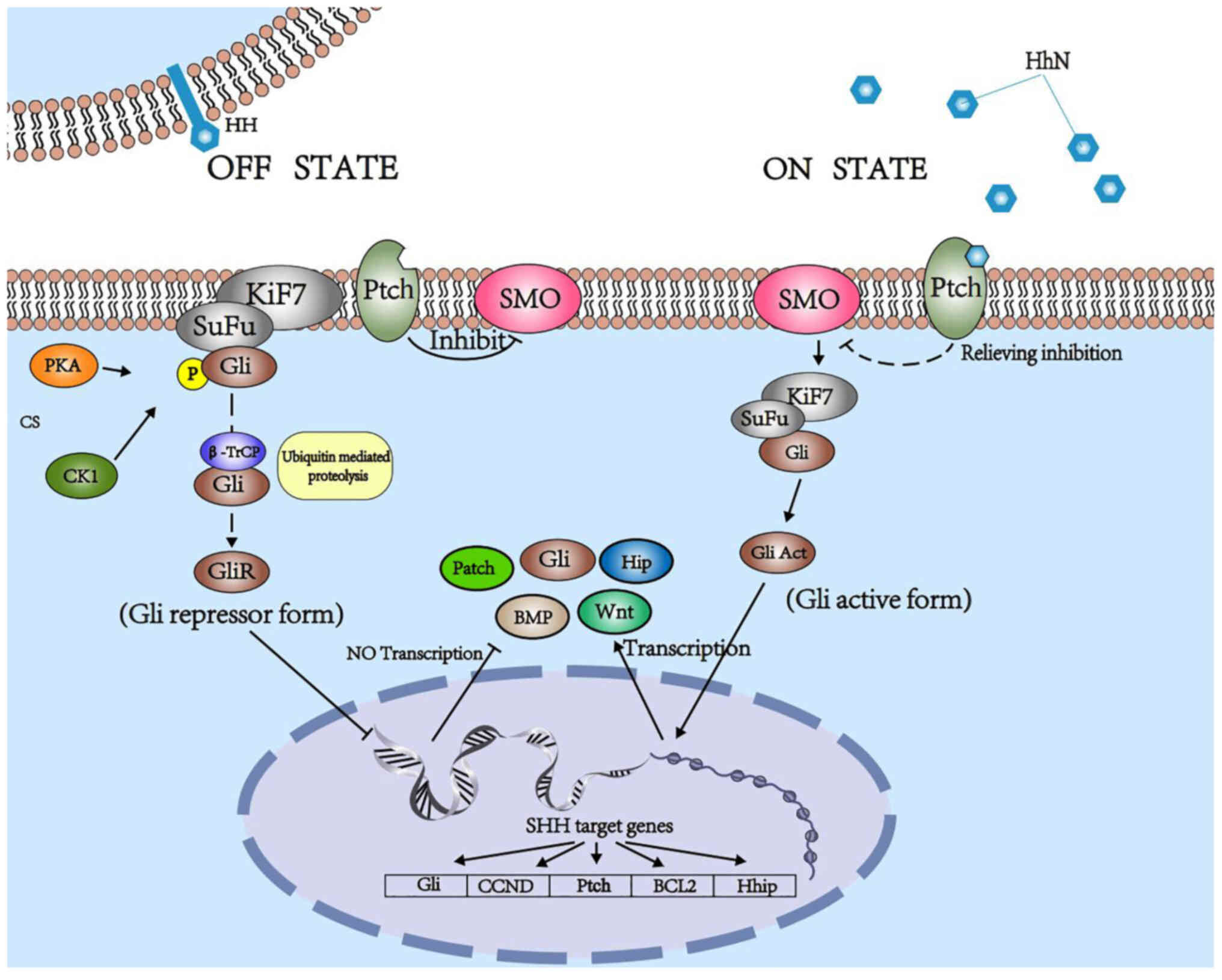
In mammals, canonical Hh signaling initiates from the binding of Hh ligands to a 12-transmembrane domain receptor protein, Ptch. This binding leads to alleviation of the Ptch-induced suppressive effects on smoothened (Smo). Smo is subsequently uninhibited, and activates zinc finger transcription factor glioma-associated oncogene (Gli) proteins, which are dissociated from a suppressive complex-containing scaffold protein, suppressor of fused (SuFu), and translocate into the nucleus to promote or depress target gene transcription (5,6), including that of Gli1, CCND, Ptch, BCL2 and Hhip. Three Gli proteins (Gli1, Gli2 and Gli3) are expressed in vertebrates with overlapping and partially redundant domains. Only Gli1 usually acts as a transcriptional activator, while Gli2 and Gli3 act as either activators or repressors, which is determined by post-transcriptional and post-translational modifications (7–9). Lack of SuFu leads to a decrease in the stability of Gli2/3 protein (10). Kinesin-like protein KIF7 (Kif7) negatively regulates Smo; however, it enhances Hh signaling in certain cases (Fig. 1) (11).
Hh signaling promotes embryonic hematopoiesis
Hh signaling is predominantly involved in the later stages of embryonic hematopoiesis, which includes two spatially and temporally distinguishable hematogenic waves, namely pro-definitive and definitive hematopoiesis, to individually give rise to bipotent hepatic progenitor cells (HPCs) or HSCs (12). Mutants in the Hh pathway (the shh mutant and the slow muscle-omitted, smu, mutant) or treatment with Hh signaling inhibitor, cyclopamine, in zebrafish embryos severely reduced the number of runt-related transcription factor (runx1)+ definitive blood cells, while the number of βE1+ primitive erythrocytes were unaffected. This requirement for Hh signaling coincides with the time of three consecutive steps in dorsal aorta formation and intersomitic vessel sprouting, indicating that Hh signaling is required for HSC formation (13). Using mouse embryonic explants, Dyer et al (14) demonstrated that Ihh secreted by visceral endoderm and mature yolk sacs alone are sufficient in inducing endothelial and hematopoietic differentiation, following increased Ptch, Smo and Gli1, as well as Bmp4 within anterior epiblasts (15). HSC numbers in the aorta-gonad-mesonephros region (AGM) increased, leading to increased colony-forming units spleen day 11 (CFU-S11) in the AGM with ventral tissues explant culture and in vivo transplantation compared with AGM explants, suggesting an important role of ventral tissues in the AGM HSC activity. Considering the increased level of Gli1 expression in AGM (c-Kit−CD34−) mesenchymal cells, positively regulating HSC activity by both Ihh and Shh proteins, the Hh signaling pathway is identified as a HSC inducing signal for definitive hematopoietic development in embryonic day 10 AGM (16).
Through a combined analysis of differentiating mouse embryonic stem cells (ESCs), mouse embryo cultures and zebrafish embryos, Kim et al (17) constructed a model of later embryonic hematopoietic development from mesoderm stage to hematopoietic cell differentiation. This hierarchy of differentiation consists of Flk1+ mesoderm patterning to the endothelium with arterial identity (VE-cadherin+CD41−CD45−) through the activation of Notch signaling, and promoting blood formation (CD41/45+) from hemogenic endothelial cells upon upregulation of the protein, stem cell leukemia.
Expression of transcriptional factors that promote hemogenic endothelium differentiation, such as BRACHYURY, GATA2 and RUNX1, was increased in human ESCs following treatment with a Smo agonist, purmorphamine, for 6 days (18,19). However, treatment with a Smo antagonist, SANT-1, reduced the expression of RUNX1 and BRACHURY in hESCs, and increased the expression of markers of endocardiogenic endothelium differentiation, suggesting a crucial role of Shh for differentiation of hESCs toward a hemogenic lineage (19,20).
Hh signaling regulates HSPC proliferation and differentiation
The role of Hh signaling in HSPCs remains controversial. Bhardwaj et al (21) showed that Hh signaling components, including Shh, Ptch and Smo, were expressed in primitive and mature CD19+, CD33+ and CD3+ cells, as well as stromal cells isolated from adult bone marrow (BM) and endothelial cells from human umbilical veins, while Gli1, Gli2 and Gli3 were absent in both myeloid and lymphoid lineages. In NOD-SCID mice transplanted with Shh-treated human CD34+CD38−Lin− cells, the proliferation of CD34+CD38−Lin− cells was enhanced and multi-lineage blood cells, including myeloid CD15+CD33+, lymphoid CD19+CD20+, CD34+ and rare CD34+CD38− cells increased. In addition, the total number of HPCs increased when Hh signaling was blocked by an anti-Hh antibody, suggesting a positive regulatory role in HSPCs. This effect was also observed in vitro. The promoter action of Shh on HSPCs and hematopoietic reconstitution is dependent on downstream Bmp4 signaling, as noggin, a specific inhibitor of Bmp4, is capable of inhibiting Shh-induced proliferation in a similar manner to anti-Hh antibody. Together with mesenchymal stem cells, Shh protein promotes the proliferation of HSCs, which is associated with increased expression of angiogenic factor receptor Tie-2 in HSCs cells, and angiogenic factors VEGF and Ang1 in MSCs (22). Expansion of human HSCs in vitro and in vivo stimulated by Ihh in the stromal supportive culture system was observed in an independent study (23).
Using a Ptch-1+/− mouse model, which exhibited increased Hh signaling activity, Trowbridge et al (24) found that Hh signaling induced cycling and expansion of primitive BM HPCs during homeostasis and stress at the expense of HSC exhaustion. Compared with wild-type (WT) mice, Ptch-1+/− mice had significantly higher numbers of Lin−Sca-1+c-Kit+ (LSK) cells and CFUs, and exhibited accelerated recovery of peripheral blood leukocytes after 5-fluorouracil (5-FU) treatment. Ptch-1+/−Lin− BM cells had a greater short-term regeneration capability but reduced long-term reconstitution efficiency. Downregulation of cell cycle-related genes (Tfdp2, Skp1a, CyclinA2, Rad51, CyclinH, CDC16, CDC2a, Mre11a and RPA3) in the Ptch-1+/− group was associated with the absence of long-term hematopoiesis. The regenerative capacity of Ptch-1+/− HSCs could be restored by cyclopamine through the upregulation of cycling Lin−Sca-1+ cells. Homozygous deletion of Gli1 (Gli1null) increased the number of long-term HSCs, which survived for a prolonged period following engraftment. However, in vitro colony-forming assays showed that the number of granulocyte CFUs derived from Gli1null BM was almost 2-fold lower compared with those derived from Gli1WT mice. Furthermore, the number of neutrophils and platelets in Gli1null mice recovered at a reduced rate following 5-FU treatment, indicating that loss of Gli1 affected myeloid progenitor function and impaired their subsequent ability to recover (25).
An independent study demonstrated that Smo deletion or overexpression exerted no significant effect on HSC self-renewal, differentiation and reconstitution ability. In Smo-deficient LSKs, <10% (70/739) of HSC gene signatures changed (upregulated or downregulated) and the expression of genes closely associated with long-term HSC activity was not altered (26). The number of T and B cells, erythroid and myeloid cells, and megakaryocytes and LSK cells in SmoNull mice were not changed, similar to the results obtained using SmoWT mice. Furthermore, deletion of Smo did not affect BM reconstitution after transplantation, and no significant difference was observed in hematopoietic colony formation potential, long-term survival ability of competitive transplantation and recovery ability of BM under 5-FU-induced stress (27).
Hh signaling participates in lineage differentiation of blood cells
T-cell differentiation
Hh signaling components are expressed in the human and mouse thymus, and participate in T-cell development at a series of stages (28–32). Hh signaling may affect the differentiation, maturation and distribution of γδ or αβ T cells in the thymus. After treatment with recombinant Shh in a fetal thymus organ culture system, CD3+γδ T-cell receptor (TCR+) and mature CD44+ cells were significantly increased. By contrast, treatment with a Hh-neutralizing antibody reduced the number of γδ T cells and promoted αβ T-cell maturation (33,34).
Targeted deletion has demonstrated that Shh, Ihh and Ptch expressed in the mouse thymus are necessary for αβ T cell differentiation from double-negative (DN; CD4−CD8−) cells to double-positive (DP; CD4+CD8+) cells (31). Results of a previous study demonstrated that the proportion of CD44+CD25− (DN1) was increased, and the proportion of CD44+CD25+ (DN2) and DP cells was decreased. In addition, single-positive (SP; CD4+ or CD8+) T-cell production were observed in Shh−/− and Ihh−/− embryonic thymi as well as Ptc−/− thymi, suggesting that the Hh signaling pathway positively regulates the transition from DN1 to DN2 and the transition from CD44−CD25− (DN4) to DP without affecting mature SP cells (31,32,35). Results of a previous study using mutants of Smo, Shh and Ihh demonstrated that Gli2 and Gli3 are required for transitions from DN1 to DN2 mediated by Smo-dependent Shh signaling (36–38). In addition, the Hh pathway also showed negative regulation of pre-TCR induced differentiation from CD44−CD25+ (DN3) to DP, in a Shh- and Ihh-mediated and Gli2-dependent manner upon analysis of Gli2−/−, Shh−/−, Ihh−/−, Gli2ΔN2-transgenic and Gli2ΔC2-transgenic thymi (30,36,38). Gli1 was shown to promote the differentiation of DN thymocytes prior to pre-TCR signal transduction and exerted an inhibitory function following pre-TCR signaling. Introduction of a class I-restricted transgenic TCR into the adult Gli1-deficient and embryonic Gli2-deficient thymus identified that both Gli1 and Gli2 influence its selection to the CD8 lineage (39). A significant reduction in the proportion of SP CD4+ (SP4) cells and the ratio of CD4/CD8 SP thymocytes was detected in the Gli2 transgenic mouse thymus (40). Furthermore, rShh significantly increased the proportion of CD3high DP cells and decreased the proportion of CD3high SP4 and the ratio of CD4/CD8 SP cells in thymus explants (41). Analysis of Gli3−/− fetal mice thymocytes showed that the expression of Shh protein was significantly increased and that the development of DP to SP was impaired. Neutralization of Hh protein in the Gli3−/− thymus enhanced SP4 differentiation, suggesting that Gli3 expressed in mouse thymic epithelial cells stimulates the differentiation of thymocytes from DP to SP, through inhibition of Shh (42). In particular, the Hh pathway activation regulator, Kif7, is required for healthy T-cell development, by which differentiation of DP and mature CD8+ T cells is enhanced and T-cell activation in vitro is promoted (31).
B lymphopoiesis
Shh has an effect on the proliferation of B220+CD43+ pro-B cells in a dose-dependent manner (43). Smo and Gli3 are also important regulators in the development of B lymphocytes. Depletion of Smo from osteoblastic cells, significantly reduced BM pro-B and pre-B cells, and recovery of BM B progenitors was delayed in the chemical ablation of the BM (44). Gli3 deficiency increased Shh transcription and the expression of Hh-target genes in the fetal liver, leading to the inhibition of early B-cell maturation. The proportion of CD19+, B220+, CD19+B220+, CD19+B220− and CD19+BP1+ in Shh−/− and Shh+/− E14.5 fetal hepatocytes was significantly higher than that in Shh+/+ (WT) mice. The proportion of CD19+ cells decreased significantly following treatment with rShh, suggesting that Gli3 promotes B-cell development through repression of Shh (45).
Myelopoiesis and erythropoiesis
Three Hh ligands have been reported to modulate myelopoiesis and erythropoiesis through different actions under normal or stress conditions at multiple stages of differentiation, despite sharing a common signaling pathway.
Dhh is expressed in stromal cells in the BM and by non-hematopoietic cells of the spleen stroma (46,47). The population of Sca-1−c-kit+ progenitors was increased in Dhh−/− BM compared with that in the WT. Following expression of CD34 and FcγRII/III, the earlier common myeloid progenitor (CMP) was demonstrated to increase in Dhh−/− mouse BM. Furthermore, there was a statistically significant increase in megakaryocyte-erythrocyte progenitor cell proportion and a concomitant reduction in the granulocyte-monocyte progenitor proportion. These data therefore suggest that Dhh acts on the CMP population and negatively regulates early erythroid differentiation, but is required for normal granulocyte production (48). Shh was reported to play a critical role in the granulopoietic response to bacterial infection. Expression of Shh mRNA and protein in BM cells, consistent with increased Gli1 expression in HSPCs and BrdU+ cells in the LKS cell subpopulation, was markedly increased in mice with bacteremia induced by injection of E. coli. Gli1 deletion did not affect the baseline activity of BrdU incorporation into LKS cells; however, attenuated bacteremia induced activation of BrdU incorporation into LKS cells, following an inhibition of the increase in granulocytes in the bloodstream after systemic E. coli infection. These findings demonstrated that Shh/Gli1 pathway positively regulated granulopoietic response to serious bacterial infection through the activation of HSPCs (49).
Numerous components of the Hh signaling pathway are present in the fetal liver, with high expression of Ihh and Gli1 in the stroma, and high expression of Ptc1 in HSPCs. Cridland et al (50) showed that nearly one-half of the Ihh knockout (Ihh−/−) embryos did not survive between E13.5 and E14.5. Circulating red blood cells in Ihh−/− embryos were significantly reduced, consistent with a significant reduction in α-globin gene expression in E13.5 fetal livers, which were small and pale upon observation. Embryonic red cells in Ihh−/− embryos were less terminally differentiated with an abnormal structure. The expression of Gli1, but not Ptch-1, was significantly downregulated, suggesting that Ihh acts upon Gli1 transcription factors during definitive erythropoiesis. However, an independent study showed that Dhh was a negative regulator of normal and stress-induced erythropoiesis. In Dhh-deficient mice, erythrocyte progenitor cell counts in the BM were increased. Erythroblasts were reliant on Dhh both in vitro and ex vivo, and were negatively regulated by Dhh. Erythrocyte differentiation was accelerated in both the spleen and BM in Dhh-deficient mice under irradiation (48).
Hh signaling is involved in hematopoietic injury
Expression of Shh, Ptch-1, Smo and Gli1 was significantly reduced in mice with pesticide-induced aplasia (51). Expression of Shh, Ihh and Dhh of Hh ligands was markly reduced in BM stromal cells. Supplementation of the recombinant mouse Shh (rmShh) in vitro promoted CFUs of granulocytes, erythrocytes, monocytes and megakaryocytes, and CFUs of granulocyte-macrophage progenitors, and augmented fibroblastic colony formation, suggesting that rmShh can minimize the suppression of different pesticide mixtures on hematopoiesis and rescue stromal and hematopoietic precursors from pesticide-induced cytotoxicity in vitro (52). Downregulation of Hh signaling may be involved in hematopoietic injury and failure. Results of our previous study demonstrated that expression of Shh in the BM of patients with aplastic anemia was significantly lower than that in patients with iron deficiency anemia. The expression level of Shh was positively associated with white blood cell count, lymphocyte count and CD4+/CD8+ ratio (unpublished data).
Conclusion
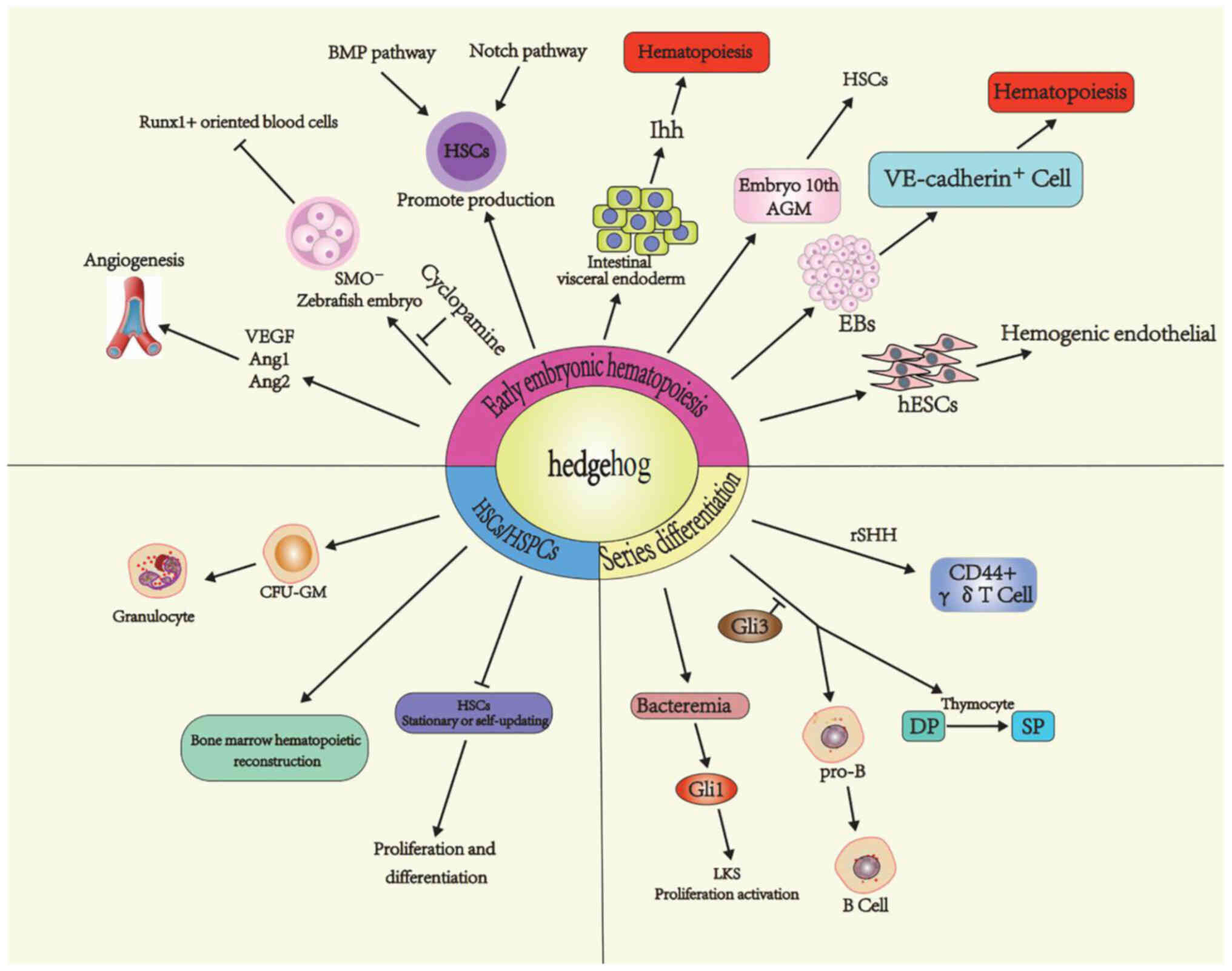
While progress has been made in elucidating the role of Hh signaling on hematopoiesis (Fig. 2), there are still a number of questions that need to be addressed. The disagreement between results may be attributed to a set of methodological differences, such as different hematopoietic microenvironments (AGM, fetal liver or BM), developmental stages (primitive, early or late definitive hematopoiesis), species (zebrafish or mouse) and physiological state (stress damage or normal hematopoiesis). Whether the Hh signaling pathway exhibits potential as a target for the diagnosis and treatment of blood disorders requires further investigation.
Acknowledgements
Not applicable.
Funding
Research grants were obtained from the following non-commercial foundations: The Innovation Training Program for College Students (grant no. 202010573023) and the China Postdoctoral Science Foundation (grant no. 2017M612848).
Availability of data and materials
Not applicable.
Authors' contributions
ZC was responsible for study conception and design. JC and YS drafted and revised the manuscript. All authors have read and approved the final manuscript. Supervision of the study was provided by ZC. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing interests.
References
|
Petrova R and Joyner AL: Roles for hedgehog signaling in adult organ homeostasis and repair. Development. 141:3445–3457. 2014. View Article : Google Scholar : PubMed/NCBI | |
|
Pachernegg S, Georges E and Ayers K: The desert hedgehog signalling pathway in human gonadal development and differences of sex development. Sex Dev. 16:98–111. 2022. View Article : Google Scholar : PubMed/NCBI | |
|
Ohba S: Hedgehog signaling in skeletal development: Roles of Indian hedgehog and the mode of its action. Int J Mol Sci. 21:66652020. View Article : Google Scholar : PubMed/NCBI | |
|
Pathi S, Pagan-Westphal S, Baker DP, Garber EA, Rayhorn P, Bumcrot D, Tabin CJ, Blake Pepinsky R and Williams KP: Comparative biological responses to human sonic, Indian, and desert hedgehog. Mech Dev. 106:107–117. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Sasai N, Toriyama M and Kondo T: Hedgehog signal and genetic disorders. Front Genet. 10:11032019. View Article : Google Scholar : PubMed/NCBI | |
|
Carballo GB, Honorato JR, de Lopes GPF and Spohr TCLSE: A highlight on sonic hedgehog pathway. Cell Commun Signal. 16:112018. View Article : Google Scholar : PubMed/NCBI | |
|
Kim J, Kato M and Beachy PA: Gli2 trafficking links hedgehog-dependent activation of smoothened in the primary cilium to transcriptional activation in the nucleus. Proc Natl Acad Sci USA. 106:21666–21671. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Varjosalo M and Taipale J: Hedgehog: Functions and mechanisms. Genes Dev. 22:2454–2472. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Matissek SJ and Elsawa SF: GLI3: A mediator of genetic diseases, development and cancer. Cell Commun Signal. 18:542020. View Article : Google Scholar : PubMed/NCBI | |
|
Chen MH, Wilson CW, Li YJ, Law KK, Lu CS, Gacayan R, Zhang X, Hui CC and Chuang PT: Cilium-independent regulation of Gli protein function by Sufu in hedgehog signaling is evolutionarily conserved. Genes Dev. 23:1910–1928. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Hsu SH, Zhang X, Yu C, Li ZJ, Wunder JS, Hui CC and Alman BA: Kif7 promotes hedgehog signaling in growth plate chondrocytes by restricting the inhibitory function of Sufu. Development. 138:3791–3801. 2011. View Article : Google Scholar : PubMed/NCBI | |
|
Tesanovic S, Krenn PW and Aberger F: Hedgehog/GLI signaling in hematopoietic development and acute myeloid leukemia-from bench to bedside. Front Cell Dev Biol. 10:9447602022. View Article : Google Scholar : PubMed/NCBI | |
|
Gering M and Patient R: Hedgehog signaling is required for adult blood stem cell formation in zebrafish embryos. Dev Cell. 8:389–400. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
Dyer MA, Farrington SM, Mohn D, Munday JR and Baron MH: Indian hedgehog activates hematopoiesis and vasculogenesis and can respecify prospective neurectodermal cell fate in the mouse embryo. Development. 128:1717–1730. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Farrington SM, Belaoussoff M and Baron MH: Winged-helix, hedgehog and Bmp genes are differentially expressed in distinct cell layers of the murine yolk sac. Mech Dev. 62:197–211. 1997. View Article : Google Scholar : PubMed/NCBI | |
|
Peeters M, Ottersbach K, Bollerot K, Orelio C, de Bruijn M, Wijgerde M and Dzierzak E: Ventral embryonic tissues and hedgehog proteins induce early AGM hematopoietic stem cell development. Development. 136:2613–2621. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Kim PG, Albacker CE, Lu YF, Jang IH, Lim Y, Heffner GC, Arora N, Bowman TV, Lin MI, Lensch MW, et al: Signaling axis involving hedgehog, notch, and Scl promotes the embryonic endothelial-to-hematopoietic transition. Proc Natl Acad Sci USA. 110:E141–E150. 2013.PubMed/NCBI | |
|
Sinha S and Chen JK: Purmorphamine activates the hedgehog pathway by targeting smoothened. Nat Chem Biol. 2:29–30. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Pethe P, Noel VS and Kale V: Deterministic role of sonic hedgehog signalling pathway in specification of hemogenic versus endocardiogenic endothelium from differentiated human embryonic stem cells. Cells Dev. 166:2036852021. View Article : Google Scholar : PubMed/NCBI | |
|
Rohatgi R, Milenkovic L, Corcoran RB and Scott MP: Hedgehog signal transduction by smoothened: Pharmacologic evidence for a 2-step activation process. Proc Natl Acad Sci USA. 106:3196–3201. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Bhardwaj G, Murdoch B, Wu D, Baker DP, Williams KP, Chadwick K, Ling LE, Karanu FN and Bhatia M: Sonic hedgehog induces the proliferation of primitive human hematopoietic cells via BMP regulation. Nat Immunol. 2:172–180. 2001. View Article : Google Scholar : PubMed/NCBI | |
|
Guo J, Wang SY, Zhu XF, Li ST, Lin FL, Li XM and Huang CL: Effect of Shh and BM-MSC synergism on the proliferation of hematopoietic stem cells. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 26:1523–1530. 2018.(In Chinese). PubMed/NCBI | |
|
Kobune M, Ito Y, Kawano Y, Sasaki K, Uchida H, Nakamura K, Dehari H, Chiba H, Takimoto R, Matsunaga T, et al: Indian hedgehog gene transfer augments hematopoietic support of human stromal cells including NOD/SCID-beta2m-/- repopulating cells. Blood. 104:1002–1009. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Trowbridge JJ, Scott MP and Bhatia M: Hedgehog modulates cell cycle regulators in stem cells to control hematopoietic regeneration. Proc Natl Acad Sci USA. 103:14134–14139. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Merchant A, Joseph G, Wang Q, Brennan S and Matsui W: Gli1 regulates the proliferation and differentiation of HSCs and myeloid progenitors. Blood. 115:2391–2396. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Gao J, Graves S, Koch U, Liu S, Jankovic V, Buonamici S, El Andaloussi A, Nimer SD, Kee BL, Taichman R, et al: Hedgehog signaling is dispensable for adult hematopoietic stem cell function. Cell Stem Cell. 4:548–558. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Hofmann I, Stover EH, Cullen DE, Mao J, Morgan KJ, Lee BH, Kharas MG, Miller PG, Cornejo MG, Okabe R, et al: Hedgehog signaling is dispensable for adult murine hematopoietic stem cell function and hematopoiesis. Cell Stem Cell. 4:559–567. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Sacedón R, Varas A, Hernández-López C, Gutiérrez-deFrías C, Crompton T, Zapata AG and Vicente A: Expression of hedgehog proteins in the human thymus. J Histochem Cytochem. 51:1557–1566. 2003. View Article : Google Scholar : PubMed/NCBI | |
|
Gutiérrez-Frías C, Sacedón R, Hernández-López C, Cejalvo T, Crompton T, Zapata AG, Varas A and Vicente A: Sonic hedgehog regulates early human thymocyte differentiation by counteracting the IL-7-induced development of CD34+ precursor cells. J Immunol. 173:5046–5053. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Outram SV, Varas A, Pepicelli CV and Crompton T: Hedgehog signaling regulates differentiation from double-negative to double-positive thymocyte. Immunity. 13:187–197. 2000. View Article : Google Scholar : PubMed/NCBI | |
|
Outram SV, Hager-Theodorides AL, Shah DK, Rowbotham NJ, Drakopoulou E, Ross SE, Lanske B, Dessens JT and Crompton T: Indian hedgehog (Ihh) both promotes and restricts thymocyte differentiation. Blood. 113:2217–2228. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Shah DK, Hager-Theodorides AL, Outram SV, Ross SE, Varas A and Crompton T: Reduced thymocyte development in sonic hedgehog knockout embryos. J Immunol. 172:2296–2306. 2004. View Article : Google Scholar : PubMed/NCBI | |
|
Mengrelis K, Lau CI, Rowell J, Solanki A, Norris S, Ross S, Ono M, Outram S and Crompton T: Sonic hedgehog is a determinant of γδ T-cell differentiation in the thymus. Front Immunol. 10:16292019. View Article : Google Scholar : PubMed/NCBI | |
|
Furmanski AL, Saldana JI, Rowbotham NJ, Ross SE and Crompton T: Role of hedgehog signalling at the transition from double-positive to single-positive thymocyte. Eur J Immunol. 42:489–499. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Uhmann A, Dittmann K, Nitzki F, Dressel R, Koleva M, Frommhold A, Zibat A, Binder C, Adham I, Nitsche M, et al: The hedgehog receptor patched controls lymphoid lineage commitment. Blood. 110:1814–1823. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Hager-Theodorides AL, Dessens JT, Outram SV and Crompton T: The transcription factor Gli3 regulates differentiation of fetal CD4-CD8-double-negative thymocytes. Blood. 106:1296–1304. 2005. View Article : Google Scholar : PubMed/NCBI | |
|
El Andaloussi A, Graves S, Meng F, Mandal M, Mashayekhi M and Aifantis I: Hedgehog signaling controls thymocyte progenitor homeostasis and differentiation in the thymus. Nat Immunol. 7:418–426. 2006. View Article : Google Scholar : PubMed/NCBI | |
|
Rowbotham NJ, Hager-Theodorides AL, Furmanski AL, Ross SE, Outram SV, Dessens JT and Crompton T: Sonic hedgehog negatively regulates pre-TCR-induced differentiation by a Gli2-dependent mechanism. Blood. 113:5144–5156. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Drakopoulou E, Outram SV, Rowbotham NJ, Ross SE, Furmanski AL, Saldana JI, Hager-Theodorides AL and Crompton T: Non-redundant role for the transcription factor Gli1 at multiple stages of thymocyte development. Cell Cycle. 9:4144–4152. 2010. View Article : Google Scholar : PubMed/NCBI | |
|
Rowbotham NJ, Hager-Theodorides AL, Cebecauer M, Shah DK, Drakopoulou E, Dyson J, Outram SV and Crompton T: Activation of the hedgehog signaling pathway in T-lineage cells inhibits TCR repertoire selection in the thymus and peripheral T-cell activation. Blood. 109:3757–3766. 2007. View Article : Google Scholar : PubMed/NCBI | |
|
Saldaña JI, Solanki A, Lau CI, Sahni H, Ross S, Furmanski AL, Ono M, Holländer G and Crompton T: Sonic hedgehog regulates thymic epithelial cell differentiation. J Autoimmun. 68:86–97. 2016. View Article : Google Scholar : PubMed/NCBI | |
|
Solanki A, Yanez DC, Ross S, Lau CI, Papaioannou E, Li J, Saldaña JI and Crompton T: Gli3 in fetal thymic epithelial cells promotes thymocyte positive selection and differentiation by repression of Shh. Development. 145:dev1469102018. View Article : Google Scholar : PubMed/NCBI | |
|
Cooper CL, Hardy RR, Reth M and Desiderio S: Non-cell-autonomous hedgehog signaling promotes murine B lymphopoiesis from hematopoietic progenitors. Blood. 119:5438–5448. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Lu W, Dordai D, Huso D and Desiderio S: Smoothened signaling in the mouse osteoblastoid lineage is required for efficient B lymphopoiesis. Blood. 131:323–327. 2018. View Article : Google Scholar : PubMed/NCBI | |
|
Solanki A, Lau CI, Saldaña JI, Ross S and Crompton T: The transcription factor Gli3 promotes B cell development in fetal liver through repression of Shh. J Exp Med. 214:2041–2058. 2017. View Article : Google Scholar : PubMed/NCBI | |
|
Perry JM, Harandi OF, Porayette P, Hegde S, Kannan AK and Paulson RF: Maintenance of the BMP4-dependent stress erythropoiesis pathway in the murine spleen requires hedgehog signaling. Blood. 113:911–918. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Hegde GV, Peterson KJ, Emanuel K, Mittal AK, Joshi AD, Dickinson JD, Kollessery GJ, Bociek RG, Bierman P, Vose JM, et al: Hedgehog-induced survival of B-cell chronic lymphocytic leukemia cells in a stromal cell microenvironment: A potential new therapeutic target. Mol Cancer Res. 6:1928–1936. 2008. View Article : Google Scholar : PubMed/NCBI | |
|
Lau CI, Outram SV, Saldaña JI, Furmanski AL, Dessens JT and Crompton T: Regulation of murine normal and stress-induced erythropoiesis by desert hedgehog. Blood. 119:4741–4751. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Shi X, Wei S, Simms KJ, Cumpston DN, Ewing TJ and Zhang P: Sonic hedgehog signaling regulates hematopoietic stem/progenitor cell activation during the granulopoietic response to systemic bacterial infection. Front Immunol. 9:3492018. View Article : Google Scholar : PubMed/NCBI | |
|
Cridland SO, Keys JR, Papathanasiou P and Perkins AC: Indian hedgehog supports definitive erythropoiesis. Blood Cells Mol Dis. 43:149–155. 2009. View Article : Google Scholar : PubMed/NCBI | |
|
Chaklader M, Das P, Pereira JA, Chaudhuri S and Law S: Altered canonical hedgehog-gli signalling axis in pesticide-induced bone marrow aplasia mouse model. Arh Hig Rada Toksikol. 63:271–282. 2012. View Article : Google Scholar : PubMed/NCBI | |
|
Chaklader M and Law S: Alteration of hedgehog signaling by chronic exposure to different pesticide formulations and unveiling the regenerative potential of recombinant sonic hedgehog in mouse model of bone marrow aplasia. Mol Cell Biochem y. 401:115–131. 2015. View Article : Google Scholar : PubMed/NCBI |